Enhancement of the Solubility and Dissolution Profile of Rivaroxaban by the Antisolvent Precipitation Technique: A Promising Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Dry RIV Nanosuspensions by the Antisolvent Precipitation Method
2.2.2. Preparation of RIV Physical Mixtures
2.2.3. Physicochemical Characterization of the Dry RIV Nanosuspensions
2.2.4. Formulation of Tablets Containing Dry RIV Nanosuspensions
2.2.5. Tablets Manufacturing Process
2.3. Tablets Characterization
2.3.1. Organoleptic Properties
2.3.2. Dimensions (Diameter and Thickness)
2.3.3. Mass Uniformity
2.3.4. Hardness
2.3.5. Friability
2.3.6. In Vitro Disintegration Time
2.4. In Vitro Dissolution Study
2.5. Data Analysis
2.5.1. Dissolution Efficiency
2.5.2. Similarity Factor (f2)
2.5.3. Dissolution Kinetic Modeling
2.6. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Characterization of the Dry RIV Nanosuspensions
3.1.1. FTIR Characterization of Materials
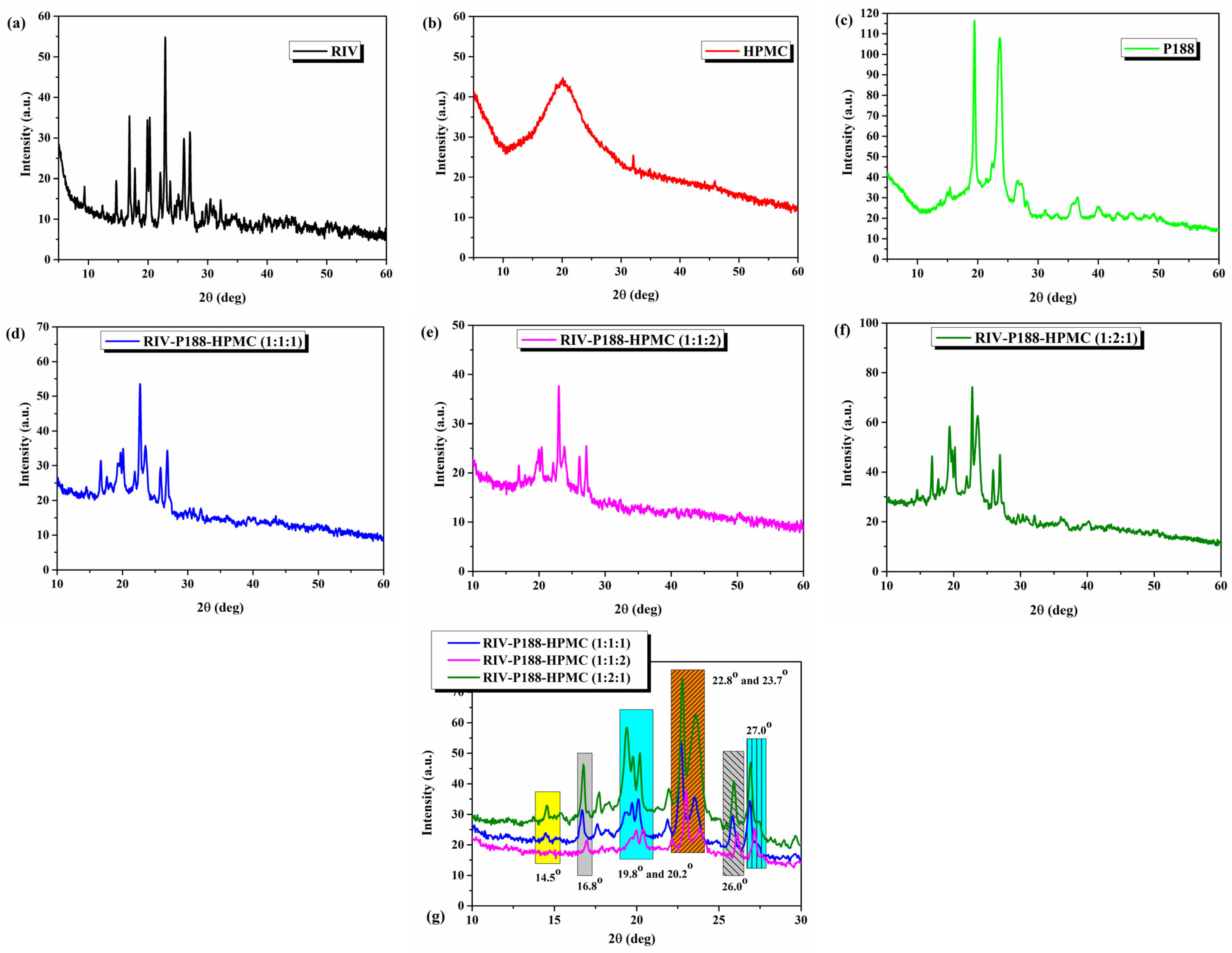
3.1.2. XRD Analysis
3.1.3. SEM Analysis
3.1.4. DLS Analysis
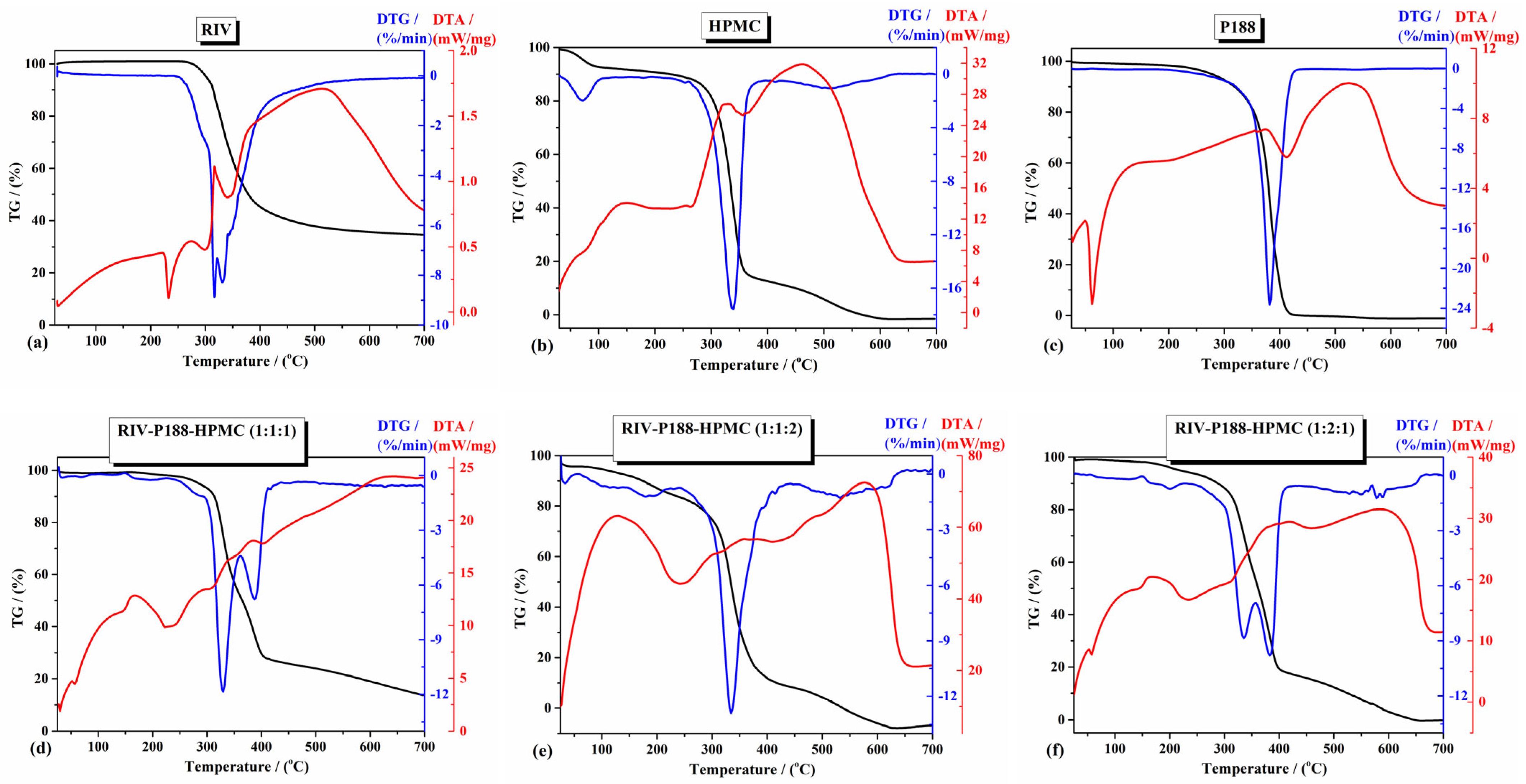
3.1.5. Thermal Analysis
3.2. Tablets Characterization
3.3. In Vitro Dissolution Profiles
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jacob, S.; Kather, F.S.; Boddu, S.H.S.; Attimarad, M.; Nair, A.B. Nanosuspension Innovations: Expanding Horizons in Drug Delivery Techniques. Pharmaceutics 2025, 17, 136. [Google Scholar] [CrossRef]
- Subbaiah, M.A.M.; Rautio, J.; Meanwell, N.A. Prodrugs as empowering tools in drug discovery and development: Recent strategic applications of drug delivery solutions to mitigate challenges associated with lead compounds and drug candidates. Chem. Soc. Rev. 2024, 53, 2099–2210. [Google Scholar] [CrossRef]
- Ma, Y.; Cong, Z.; Gao, P.; Wang, Y. Nanosuspensions technology as a master key for nature products drug delivery and In vivo fate. Eur. J. Pharm. Sci. 2023, 185, 106425. [Google Scholar] [CrossRef]
- Noyes, A.A.; Whitney, W.R. The rate of solution of solid substances in their own solutions. J. Am. Chem. Soc. 1897, 19, 930–934. [Google Scholar] [CrossRef]
- Manca, M.L.; Lai, F.; Pireddu, R.; Valenti, D.; Schlich, M.; Pini, E.; Ailuno, G.; Fadda, A.M.; Sinico, C. Impact of nanosizing on dermal delivery and antioxidant activity of quercetin nanocrystals. J. Drug Deliv. Sci. Tecnol. 2020, 55, 101482. [Google Scholar] [CrossRef]
- Müller, R.H.; Becker, R.; Kruss, B.; Peters, K. Pharmaceutical Nanosuspensions for Medicament Administration as System of Increased Saturation Solubility and Rate of Solution. U.S. Patent 08/836,305, 12 January 1999. [Google Scholar]
- Yadollahi, R.; Vasilev, K.; Simovic, S. Nanosuspension technologies for delivery of poorly soluble drugs. J. Nanomat. 2015, 2015, 216375. [Google Scholar] [CrossRef]
- Zabihi, F.; Yang, M.; Leng, Y.; Zhao, Y. PLGA–HPMC nanoparticles prepared by a modified supercritical anti-solvent technique for the controlled release of insulin. J. Super. Fluids 2015, 9, 15–22. [Google Scholar] [CrossRef]
- Ginoya, S.; Atara, S. Nanosuspension: A novel approach towards the drug delivery system. Pharma Sci. Monit. 2013, 4, 100–122. [Google Scholar]
- Sadeghi, F.; Ashofteh, M.; Homayouni, A.; Abbaspour, M.; Nokhodchi, A.; Garekani, H.A. Antisolvent precipitation technique: A very promising approach to crystallize curcumin in presence of polyvinyl pyrrolidon for solubility and dissolution enhancement. Colloids Surf. B Biointerfaces 2016, 147, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Homayouni, A.; Amini, M.; Sohrabi, M.; Varshosaz, J.; Nokhodchi, A. Curcumin nanoparticles containing poloxamer or soluplus tailored by high pressure homogenization using antisolvent crystallization. Int. J. Pharm. 2019, 562, 124–134. [Google Scholar] [CrossRef]
- Wu, C.Y.; Wang, W. Application of Antisolvent Precipitation Method for Formulating Excipient-Free Nanoparticles of Psychotropic Drugs. Pharmaceutics 2022, 14, 819. [Google Scholar] [CrossRef]
- Li, Z.; Gong, Z.; Zhang, B.; Nawaz, A. Investigation of the Influence of Anti-Solvent Precipitation Parameters on the Physical Stability of Amorphous Solids. Molecules 2024, 29, 1275. [Google Scholar] [CrossRef]
- Metre, S.; Mukesh, S.; Samal, S.K.; Chand, M.; Sangamwar, A.T. Enhanced Biopharmaceutical Performance of Rivaroxaban through Polymeric Amorphous Solid Dispersion. Mol. Pharm. 2018, 15, 652–668. [Google Scholar] [CrossRef] [PubMed]
- Alamri, A.H.; Ishrat, G.; Zahid, F.; Ali, Z.; Alqahtani, A.; Lahiq, A.A.; Alsharif, S.T.; Sahab, M.A.; Din, F.U. Development and evaluation of the rivaroxaban loaded nanostructured lipid carriers for improved oral bioavailability and safety. Sci. Rep. 2025, 15, 23350. [Google Scholar] [CrossRef]
- Solomon, C.; Anuța, V.; Sarbu, I.; Ozon, E.A.; Musuc, A.M.; Bratan, V.; Rusu, A.; Surdu, V.-A.; Croitoru, C.; Chandak, A.; et al. Enhancing the Drug Release and Physicochemical Properties of Rivaroxaban via Cyclodextrin Complexation: A Comprehensive Analytical Approach. Pharmaceuticals 2025, 18, 761. [Google Scholar] [CrossRef] [PubMed]
- Ozon, E.A.; Mati, E.; Karampelas, O.; Anuta, V.; Sarbu, I.; Musuc, A.M.; Mitran, R.-A.; Culita, D.C.; Atkinson, I.; Anastasescu, M.; et al. The development of an innovative method to improve the dissolution performance of rivaroxaban. Heliyon 2024, 10, E33162. [Google Scholar] [CrossRef]
- Jin, C.; Wu, F.; Hong, Y.; Shen, L.; Lin, X.; Zhao, L.; Feng, Y. Updates on applications of low-viscosity grade Hydroxypropyl methylcellulose in coprocessing for improvement of physical properties of pharmaceutical powders. Carbohydr. Polym. 2023, 311, 120731. [Google Scholar] [CrossRef]
- Deshmukh, K.; Basheer Ahamed, M.; Deshmukh, R.R.; Khadheer Pasha, S.K.; Bhagat, P.R.; Chidambaram, K. Biopolymer Composites with High Dielectric Performance: Interface Engineering. In Biopolymer Composites in Electronics; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 27–128. [Google Scholar] [CrossRef]
- Sheskey, P.J.; Cook, W.G.; Cable, C.G. (Eds.) Handbook of Pharmaceutical Excipients, 8th ed.; Pharmaceutical Press: London, UK, 2017. [Google Scholar]
- Zupanc, A.; Petkovšek, M.; Zdovc, B.; Žagar, E.; Zupanc, M. Degradation of hydroxypropyl methylcellulose (HPMC) by acoustic and hydrodynamic cavitation. Ultrason. Sonochemistry 2024, 109, 107020. [Google Scholar] [CrossRef]
- Viridén, A.; Wittgren, B.; Larsson, A. Investigation of critical polymer properties for polymer release and swelling of HPMC matrix tablets. Eur. J. Pharm. Sci. 2009, 36, 297–309. [Google Scholar] [CrossRef]
- Mobilawon, F.E.; Iroegbu, A.O.C.; Zinyemba, O.; Meijboom, R. Hydroxypropyl methylcellulose (HPMC) in sustainable pharmaceutical synthesis—Mechanistic insights, green metrics and outlook. Sus. Chem. Pharm. 2025, 48, 102233. [Google Scholar] [CrossRef]
- Veena Chaudhary, N. A state of the art an exploration of HPMC and its opportunities in the construction industry. Constr. Build. Mater. 2025, 495, 143579. [Google Scholar] [CrossRef]
- Chen, W.N.; Shaikh, M.F.; Bhuvanendran, S.; Date, A.; Ansari, M.T.; Radhakrishnan, A.K.; Othman, I. Poloxamer 188 (P188), A Potential Polymeric Protective Agent for Central Nervous System Disorders: A Systematic Review. Curr. Neuropharmacol. 2022, 20, 799–808. [Google Scholar] [CrossRef]
- Sherif, A.Y.; Elzayat, E.M. Development of Bioresponsive Poloxamer-Based Self-Nanoemulsifying System for Enhanced Febuxostat Bioavailability: Solidification Strategy Using I-Optimal Approach. Pharmaceutics 2025, 17, 975. [Google Scholar] [CrossRef]
- Han, J.; Zhou, X.; Fu, J.; Gao, G.; Zuo, C.; Guo, Y.; Han, M.; Wang, X. Annonaceous acetogenins nanosuspensions stabilized by poloxamer 188: Preparation, properties and in vivo evaluation. J. Drug Deliv. Sci. Technol. 2021, 66, 102676. [Google Scholar] [CrossRef]
- Nakka, V.N.J.; GY, S.K. Development and Evaluation of Rivaroxaban Nanosuspension by Solvent Evaporation Method. Int. J. Pharm. Sci. Nanotechnol. 2025, 18. [Google Scholar] [CrossRef]
- Demir, H.; Gulsun, T.; Sener, E.; Sahin, S.; Oner, L. Preparation and Characterization of Rivaroxaban Nanocrystals Prepared by Combination of Wet Ball Milling and High Pressure Homogenization Methods. Lat. Am. J. Pharm. 2018, 37, 2482–2490. [Google Scholar]
- Abd-Ali, K.J.; Toma, N.M. Preparation and in vitro evaluation of rivaroxaban nanocrystal. J. Res. Pharm. 2026, 30, 296–306. [Google Scholar] [CrossRef]
- Solomon, C.; Sarbu, I.; Anuța, V.; Ozon, E.A.; Musuc, A.M.; Rusu, A.; Surdu, V.-A.; Chandak, A.; Gavriloaia, R.M.; Fița, A.C.; et al. Pharmaceutical Co-Crystal Formulation of Rivaroxaban with Niacinamide: Preparation, Characterization, and In Vitro Release Evaluation. Materials 2026, 19, 1336. [Google Scholar] [CrossRef] [PubMed]
- Xia, D.; Quan, P.; Piao, H.; Piao, H.; Sun, S.; Yin, Y.; Cui, F. Preparation of stable nitrendipine nanosuspensions using the precipitation-ultrasonication method for enhancement of dissolution and oral bioavailability. Eur. J. Pharm. Sci. 2010, 40, 325–334. [Google Scholar] [CrossRef]
- Kakran, M.; Sahoo, N.G.; Li, L.; Judeh, M.A. Fabrication of quercetin nanoparticles by anti-solvent precipitation method for enhanced dissolution. Powder Technol. 2012, 223, 59–64. [Google Scholar] [CrossRef]
- Hao, L.; Wang, X.; Zhang, D.; Xu, Q.; Song, S.; Wang, F.; Li, C.; Guo, H.; Liu, Y.; Zheng, D.; et al. Studies on the preparation, characterization and pharmacokinetics of Amoitone B nanocrystals. Int. J. Pharm. 2012, 433, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Hollis, C.P.; Zhang, Q.; Li, T. Preparation and antitumor study of camptothecin nanocrystals. Int. J. Pharm. 2011, 415, 293–300. [Google Scholar] [CrossRef]
- Mititelu, M.; Moroșan, E.; Nicoară, A.C.; Secăreanu, A.A.; Musuc, A.M.; Atkinson, I.; Pandele Cusu, J.; Nițulescu, G.M.; Ozon, E.A.; Sarbu, I.; et al. Development of Immediate Release Tablets Containing Calcium Lactate Synthetized from Black Sea Mussel Shells. Mar. Drugs 2022, 20, 45. [Google Scholar] [CrossRef]
- Mitu, M.A.; Cretu, E.A.; Novac, M.; Karampelas, O.; Nicoara, A.; Nitulescu, G.; Lupuleasa, D. The Flowing Characteristics of Some Composed Powders Containing Inclusion Complexes in Beta-Cyclodextrin. In Proceedings of the Romanian National Congress of Pharmacy, 17th Edition: 21st Century Pharmacy—Between Intelligent Specialization and Social Responsibility, Bucharest, Romania, 26–29 September 2018; Draganescu, D., Arsene, A., Eds.; Filodiritto: Bologna, Italy, 2018; pp. 129–133. [Google Scholar]
- Musuc, A.M.; Anuta, V.; Atkinson, I.; Sarbu, I.; Popa, V.T.; Munteanu, C.; Mircioiu, C.; Ozon, E.A.; Nitulescu, G.M.; Mitu, M.A. Formulation of Chewable Tablets Containing Carbamazepine-β-cyclodextrin Inclusion Complex and F-Melt Disintegration Excipient. The Mathematical Modeling of the Release Kinetics of Carbamazepine. Pharmaceutics 2021, 13, 915. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, C.K.; Rao, S.R.M.; Sudhakar, M. HPMC a Biomedical Polymer in Pharmaceutical Dosage Forms. J. Chem. Pharm. Sci. 2015, 8, 875–881. [Google Scholar]
- Fristiohady, A.; Suryani, S.; Fitrawan, L.O.M.; Andriani, R.; Aspadiah, V.; Saripuddin, S.; Purnama, L.O.M.J.; Hamsidi, R.; Indalifiany, A. Characterization, Formulation, and Evaluation of Tablet Containing Johar Leaves (Cassia siamea Lamk.) Extract with Hydroxypropyl Methylcellulose (HPMC) as Tablet Binder. Lett. Appl. NanoBioSci. 2020, 10, 2070–2077. [Google Scholar]
- Vlad, R.-A.; Pintea, A.; Pintea, C.; Rédai, E.-M.; Antonoaea, P.; Bîrsan, M.; Ciurba, A. Hydroxypropyl Methylcellulose—A Key Excipient in Pharmaceutical Drug Delivery Systems. Pharmaceutics 2025, 17, 784. [Google Scholar] [CrossRef]
- Council of Europe. European Pharmacopoeia, 10th ed.; EDQM, Council of Europe: Strasbourg, France, 2019. [Google Scholar]
- USP. Rivaroxaban Tablets Monograph. In USP-NF; United States Pharmacopeia: Rockville, MD, USA, 2023. [Google Scholar]
- Solís-Cruz, B.; Hernández-Patlán, D.; Hipólito, E.M.; Téllez-Isaías, G.; Pineda, A.A.; López-Arellano, R. Discriminative Dissolution Method Using the Open-Loop Configuration of the USP IV Apparatus to Compare Dissolution Profiles of Metoprolol Tartrate Immediate-Release Tablets: Use of Kinetic Parameters. Pharmaceutics 2023, 15, 2191. [Google Scholar] [CrossRef]
- FDA. Guidance for Industry: Dissolution Testing of Immediate Release Solid Oral Dosage Forms; FDA: Silver Spring, MD, USA, 1997.
- Stevens, R.; Gray, V.; Dorantes, A.; Gold, L.; Pham, L. Scientific and Regulatory Standards for Assessing Product Performance Using the Similarity Factor, f2. AAPS J. 2015, 17, 301–306. [Google Scholar] [CrossRef]
- Muselík, J.; Komersová, A.; Kubová, K.; Matzick, K.; Skalická, B. A Critical Overview of FDA and EMA Statistical Methods to Compare In Vitro Drug Dissolution Profiles of Pharmaceutical Products. Pharmaceutics 2021, 13, 1703. [Google Scholar] [CrossRef]
- Mircioiu, C.; Voicu, V.; Anuta, V.; Tudose, A.; Celia, C.; Paolino, D.; Fresta, M.; Sandulovici, R.; Mircioiu, I. Mathematical Modeling of Release Kinetics from Supramolecular Drug Delivery Systems. Pharmaceutics 2019, 11, 140. [Google Scholar] [CrossRef]
- Preda, I.A.; Mircioiu, I.; Mircioiu, C.; Corlan, G.; Pahomi, G.; Prasacu, I.; Anuta, V. Research concerning the development of a biorelevant dissolution test for formulations containing norfloxacin. I. Modelling of in vitro release kinetics. Farmacia 2012, 60, 675–687. [Google Scholar]
- Ritger, P.; Peppas, N. A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J. Control. Release 1987, 5, 37–42. [Google Scholar] [CrossRef]
- Wu, I.; Bala, S.; Škalko-Basnet, N.; Di Cagno, M. Interpreting non-linear drug diffusion data: Utilizing Korsmeyer-Peppas model to study drug release from liposomes. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2019, 138, 105026. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wu, S.-P.; Liu, X.-J.; Zhang, L.-J.; Lu, J. Crystal characterization and transformation of the forms I and II of anticoagulant drug rivaroxaban. Cryst. Res. Technol. 2017, 52, 1600379. [Google Scholar] [CrossRef]
- Meng, Y.; Tan, F.; Yao, J.; Cui, Y.; Feng, Y.; Li, Z.; Wang, Y.; Yang, Y.; Gong, W.; Yang, M.; et al. Preparation, characterization, and pharmacokinetics of rivaroxaban cocrystals with enhanced in vitro and in vivo properties in beagle dogs. Int. J. Pharm. X 2022, 4, 100119. [Google Scholar] [CrossRef]
- Al Kiey, S.A.; Basser, R.A.; Kamel, S. Simultaneous detection of ascorbic acid, dopamine, and uric acid using an rGO/PE/PVP@ HPMC/GCE electrode in biological matrices. Cellulose 2026, 33, 881–900. [Google Scholar] [CrossRef]
- Okolišan, D.; Potinteu, A.-I.; Vlase, G.; Budiul, M.M.; Matica, M.A.; Pahomi, A.; Mogoş, A.; Vlase, T. Novel Ternary Biopolymer Films Incorporating Amygdalin: FTIR, TG, and In Vitro Evaluation on Model Bacteria. Materials 2026, 19, 897. [Google Scholar] [CrossRef] [PubMed]
- Mohyeldin, S.M.; Mehanna, M.M.; Elgindy, N.A. The relevancy of controlled nanocrystallization on rifampicin characteristics and cytotoxicity. Int. J. Nanomed. 2016, 11, 2209–2222. [Google Scholar] [CrossRef][Green Version]
- Shoukat, H.M.J.; Sheikh, F.A.; Tayyab, M.; Bukhari, S.N.A.; Haseeb, M.T.; Nazir, I.; Alafar, R.A.; Abdelgawad, M.A.; Al-Sanea, M.M.; Zafar, A.; et al. Statistically optimized orodispersible film loaded with Febuxostat nanosuspension to enhance oral bioavailability. J. Drug Deliv. Sci. Technol. 2026, 117, 108021. [Google Scholar] [CrossRef]
- Shetty, G.R.; Rao, B.L.; Asha, S.; Wang, Y.; Sangappa, Y. Preparation and characterization of silk fibroin/hydroxy-propyl methyl cellulose (HPMC) blend films. Fibers Polym. 2015, 16, 1734–1741. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, X.; Zu, Y.; Zhang, Y. Preparation and characterization of paclitaxel nanosuspension using novel emulsification method by combining high speed homogenizer and high pressure homogenization. Int. J. Pharm. 2015, 490, 324–333. [Google Scholar] [CrossRef]
- Daebis, N.A.O.; El-Massik, M.; Abdelkader, H. Formulation and characterization of Itraconazole Oral Nanosuspension: Methyl cellulose as promising stabilizer. Ely. J. Pharm. Res. 2015, 1, 102. [Google Scholar]
- Mou, D.; Chen, H.; Wan, J.; Xu, H.; Yang, X. Potent dried drug nanosuspensions for oral bioavailability enhancement of poorly soluble drugs with pH-dependent solubility. Int. J. Pharm. 2011, 413, 237–244. [Google Scholar] [CrossRef]
- Ghosh, I.; Schenck, D.; Bose, S.; Ruegger, C. Optimization of formulation and process parameters for the production of nanosuspension by wet media milling technique: Effect of vitamin E TPGS and nanocrystal particle size on oral absorption. Eur. J. Pharm. Sci. 2012, 47, 718–728. [Google Scholar] [CrossRef]
- Newa, M.; Bhandari, K.H.; Li, D.X.; Kwon, T.-H.; Kim, J.A.; Yoo, B.K.; Woo, J.S.; Lyoo, W.S.; Yong, C.S.; Choi, H.G. Preparation, characterization and in vivo evaluation of ibuprofen binary solid dispersions with poloxamer 188. Int. J. Pharm. 2007, 343, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Kesisoglou, F.; Panmai, S.; Wu, Y. Nanosizing—Oral formulation development and biopharmaceutical evaluation. Adv. Drug Deliv. Rev. 2007, 59, 631–644. [Google Scholar] [CrossRef] [PubMed]
- Anwer, M.K.; Mohammad, M.; Iqbal, M.; Ansari, M.N.; Ezzeldin, E.; Fatima, F.; Alshahrani, S.M.; Aldawsari, M.F.; Alalaiwe, A.; Alzahrani, A.A.; et al. Sustained release and enhanced oral bioavailability of rivaroxaban by PLGA nanoparticles with no food effect. J. Thromb. Thrombolysis 2020, 49, 404–412. [Google Scholar] [CrossRef]
- Ozon, E.A.; Burloiu, A.M.; Musuc, A.M.; Manda, G.; Anuta, V.; Dinu-Pîrvu, C.E.; Lupuliasa, D.; Neagoe, I.V.; Anastasescu, M.; Socoteanu, R.P.; et al. Cellulose-Derived Gels for Topical Delivery: HPMC as a Functional Matrix for Porphyrinic Photosensitizers. Gels 2025, 11, 824. [Google Scholar] [CrossRef] [PubMed]
- Nousheen, L.; Rajasekaran, S.; Qureshi, M.S. Solubility enhancement of lornoxicam with poloxamer 188 by solvent evaporation method. Int. J. Health Sci. 2022, 6, 8186–8195. [Google Scholar] [CrossRef]
- Liu, J.; Qu, S.; Wang, T.; Yang, B.; Liu, X.; Wang, G.; Li, P.; Yu, Q.; Ling, F. Nanosuspensions as an approach for improved solubility and anti-Ichthyophthirius multifiliis activity of magnolol. Aquaculture 2024, 579, 740133. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, Y.; Zhang, L.; Wang, Q.; Zhang, D. Stability of nanosuspensions in drug delivery. J. Control. Release 2013, 172, 1126–1141. [Google Scholar] [CrossRef]
- Xia, D.; Ouyang, M.; Wu, J.X.; Jiang, Y.; Piao, H.; Sun, S.; Zheng, L.; Rantanen, J.; Cui, F.; Yang, M. Polymer-mediated anti-solvent crystallization of nitrendipine: Monodispersed spherical crystals and growth mechanism. Pharm. Res. 2012, 29, 158–169. [Google Scholar] [CrossRef]
- Jakubowska, E.; Ciepluch, N. Blend Segregation in Tablets Manufacturing and Its Effect on Drug Content Uniformity—A Review. Pharmaceutics 2021, 13, 1909. [Google Scholar] [CrossRef]
- Yohana Chaerunisaa, A.; Sriwidodo, S.; Abdassah, M. Microcrystalline Cellulose as Pharmaceutical Excipient. In Pharmaceutical Formulation Design—Recent Practices; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Saigal, N.; Baboota, S.; Ahuja, A.; Ali, J. Microcrystalline cellulose as a versatile excipient in drug research. J. Young Pharm. 2009, 1, 6–12. [Google Scholar]
- Lamešić, D.; Grilc, B.; Roškar, R.; Kolokytha, S.; Hofmann, J.; Malekos, A.; Kaufmann, R.; Planinšek, O. Spherical Agglomerates of Lactose Reduce Segregation in Powder Blends and Improve Uniformity of Tablet Content at High Drug Loads. AAPS Pharmscitech 2021, 23, 17. [Google Scholar] [CrossRef]
- Rojas, J.; Buckner, I.; Kumar, V. Co-proccessed excipients with enhanced direct compression functionality for improved tableting performance. Drug Dev. Ind. Pharm. 2012, 38, 1159–1170. [Google Scholar] [CrossRef]
- Alhusban, F.; Murgatroyd, E.F. Impact testing as a new approach to determine mechanical strength of pharmaceutical tablets. Int. J. Pharm. 2024, 653, 123891. [Google Scholar] [CrossRef] [PubMed]
- Adeleye, O.A. Relationship between compression pressure, mechanical strenghth and release properties of tablets. Polim. Med. 2019, 49, 27–33. [Google Scholar] [CrossRef]
- Sun, C.C.; Hao, H. Improving mechanical properties of caffeine and methyl gallate crystals by cocrystallization. Crysth Growth Des. 2008, 8, 1575–1579. [Google Scholar] [CrossRef]
- Osei-Yeboah, F.; Sun, C.C. Validation and applications of an expedited tablet friability method. Int. J. Pharm. 2015, 484, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Steffens, K.E.; Wagner, K.G. Immediate-Release Formulations Produced via Twin-Screw Melt Granulation: Systematic Evaluation of the Addition of Disintegrants. AAPS PharmSciTech 2021, 22, 183. [Google Scholar] [CrossRef]
- Kaul, G.; Huang, J.; Chatlapalli, R.; Ghosh, K.; Nagi, A. Quality-by-design case study: Investigation of the role of poloxamer in immediate-release tablets by experimental design and multivariate data analysis. AAPS PharmSciTech 2011, 12, 1064–1076. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lai, F.; Sinico, C.; Ennas, G.; Marongiu, F.; Marongiu, G.; Fadda, A.M. Diclofenac nanosuspensions: Influence of preparation procedure and crystal form on drug dissolution behaviour. Int. J. Pharm. 2009, 373, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Namdeo Tukaram, B.; Vidaya Rajagopalan, I.; Sushi Ikumar Shartchandra, P. The Effects of Lactose, Microcrystalline Cellulose and Dicalcium Phosphate on Swelling and Erosion of Compressed HPMC Matrix Tablets: Texture Analyzer. Iran. J. Pharm. Res. 2010, 9, 349–358. [Google Scholar]
- Kondo, A.; Koide, T.; Fukami, T. Evaluation of the Effect of Disintegrant Distribution on the Dissolution Behavior of Pharmaceutical Tablets Using Raman Chemical Imaging. Chem. Pharm. Bull. 2023, 71, 454–458. [Google Scholar] [CrossRef]
- Nickerson, B.; Kong, A.; Gerst, P.; Kao, S. Correlation of dissolution and disintegration results for an immediate-release tablet. J. Pharm. Biomed. Anal. 2018, 150, 333–340. [Google Scholar] [CrossRef]
- Markl, D.; Zeitler, J.A. A Review of Disintegration Mechanisms and Measurement Techniques. Pharm. Res. 2017, 34, 890–917. [Google Scholar] [CrossRef]
- Bou-Chacra, N.; Melo, K.J.C.; Morales, I.A.C.; Stippler, E.S.; Kesisoglou, F.; Yazdanian, M.; Löbenberg, R. Evolution of Choice of Solubility and Dissolution Media After Two Decades of Biopharmaceutical Classification System. Aaps J. 2017, 19, 989–1001. [Google Scholar] [CrossRef]
- Wu, C.N.; Liu, Y.; He, Z.G.; Sun, J. Insight into the Development of Dissolution Media for BCS Class II Drugs: A Review from Quality Control and Prediction of In Vivo Performance Perspectives. Curr. Drug Deliv. 2016, 13, 1004–1020. [Google Scholar] [CrossRef]
- Kushwah, V.; Arora, S.; Tamás Katona, M.; Modhave, D.; Fröhlich, E.; Paudel, A. On Absorption Modeling and Food Effect Prediction of Rivaroxaban, a BCS II Drug Orally Administered as an Immediate-Release Tablet. Pharmaceutics 2021, 13, 283. [Google Scholar] [CrossRef]
- Peltonen, L.; Strachan, C.J. Degrees of Order: A Comparison of Nanocrystal and Amorphous Solids for Poorly Soluble Drugs. Int. J. Pharm. 2020, 586, 119492. [Google Scholar] [CrossRef]
- Chogale, M.; Chodake, V.; Patravale, V. Performance parameters and Characterizations of Nanocrystals: A Brief review. Pharmaceutics 2016, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Bonhoeffer, B.; Kordikowski, A.; John, E.; Juhnke, M. Numerical modeling of the dissolution of drug nanocrystals and its application to industrial product development. ADMET DMPK 2022, 10, 253–287. [Google Scholar] [CrossRef]
- Ferrar, J.A.; Sellers, B.D.; Chan, C.; Leung, D.H. Towards an improved understanding of drug excipient interactions to enable rapid optimization of nanosuspension formulations. Int. J. Pharm. 2020, 578, 119094. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Nguyen-Dinh, T.D.; Le, N.T.T.; Huynh, T.K.C.; Nguyen, D.H. Study of the Influence of Poloxamer 184, 188, and 407 As Capping Agents on the Dual-Drug Delivery Ability of Hollow Mesoporous Silica Nanoparticles. ACS Appl. Eng. Mater. 2025, 3, 2697–2707. [Google Scholar] [CrossRef]
- Khan, W.H.; Asghar, S.; Khan, I.U.; Irfan, M.; Alshammari, A.; Riaz Rajoka, M.S.; Munir, R.; Shah, P.A.; Khalid, I.; Razzaq, F.A.; et al. Effect of hydrophilic polymers on the solubility and dissolution enhancement of rivaroxaban/beta-cyclodextrin inclusion complexes. Heliyon 2023, 9, e19658. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Lee, P. Evolution of supersaturation of amorphous pharmaceuticals: The effect of rate of supersaturation generation. Mol. Pharm. 2013, 10–11, 4330–4346. [Google Scholar] [CrossRef]
- Lynnerup, J.T.; Eriksen, J.; Bauer-Brandl, A.; Holsæter, A.M.; Brandl, M. Insight into the mechanism behind oral bioavailability-enhancement by nanosuspensions through combined dissolution/permeation studies. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2023, 184, 106417. [Google Scholar] [CrossRef]
- Budiman, A.; Citraloka, Z.G.; Muchtaridi, M.; Sriwidodo, S.; Aulifa, D.; Rusdin, A. Inhibition of Crystal Nucleation and Growth in Aqueous Drug Solutions: Impact of Different Polymers on the Supersaturation Profiles of Amorphous Drugs—The Case of Alpha-Mangostin. Pharmaceutics 2022, 14, 2386. [Google Scholar] [CrossRef]






| Ingredients | Quantity mg/Tablet | Role in Formulation | |||||
|---|---|---|---|---|---|---|---|
| F1 | F2 | F3 | F4 | F5 | F6 | ||
| RIV-P188-HPMC (1:1:1 dry nanosuspension) | 30 | - | - | - | - | - | Active ingredient |
| RIV-P188-HPMC (1:1:1 physical mixture) | - | 30 | - | - | - | - | Active ingredient |
| RIV-P188-HPMC (1:2:1 dry nanosuspension) | - | - | 40 | - | - | - | Active ingredient |
| RIV-P188-HPMC (1:2:1 physical mixture) | - | - | - | 40 | - | - | Active ingredient |
| RIV-P188-HPMC (1:1:2 dry nanosuspension) | - | - | - | - | 40 | - | Active ingredient |
| RIV-P188-HPMC (1:1:2 physical mixture) | - | - | - | - | - | 40 | Active ingredient |
| Avicel® PH 102—microcrystalline cellulose | 83 | 83 | 78 | 78 | 78 | 78 | Filler Binder |
| Flowlac® 100—spray-dried lactose | 83 | 83 | 78 | 78 | 78 | 78 | Filler Binder |
| EXPLOTAB®—Sodium starch glycolate | 2 | 2 | 2 | 2 | 2 | 2 | Superdisintegrant |
| LIGAMED® MF-2-V—Magnesium stearate | 2 | 2 | 2 | 2 | 2 | 2 | Glidant |
| TOTAL | 200 | 200 | 200 | 200 | 200 | 200 | |
| Functional Group | υ(N-H) | C-H Stretching Vibration | υ(C=O) | β(N-H) | C-O-C Stretching Vibration | C-H Deformation Vibration |
|---|---|---|---|---|---|---|
| RIV (cm−1) | 3358.47 | - | 1669.11 | 1645.49 | - | - |
| P188 (cm−1) | - | 2884.55 | - | - | 1105.9 | 842.2 |
| RIV-P188-HPMC (1:1:1) (cm−1) | 3356.08 | 2891.00 | 1668.15 | 1645.97 | 1104.5 | 841.7 |
| RIV-P188-HPMC (1:2:1) (cm−1) | 3361.85 | 2880.21 | 1670.10 | 1647.90 | 1103.1 | 842.7 |
| RIV-P188-HPMC (1:1:2) (cm−1) | 3353.17 | 2886.96 | 1669.11 | 1648.38 | 1106.4 | 843.2 |
| Compound | Peak Temperature (°C) and Mass Loss (%) (1st Step) | Temperature Range, Peak Temperature (°C) and Mass Loss (%) (2nd Step) | Temperature Range, Peak Temperature (°C) and Mass Loss (%) (3rd Step) | Residue (%) at 700 °C |
|---|---|---|---|---|
| RIV | TDTA = 231.9 °C (melting process) | 270–700 °C TDTA = 316.6 °C 65.5% | - | 34.5% |
| HPMC | TDTA = 76.6 °C (dehydration process) Below 114 °C/7.43% | 190–365 °C TDTA = 324 °C 78% | 365–650 °C TDTA = 461 °C 14.57% | No residue |
| P188 | TDTA = 62 °C (melting process) | 240–700 °C TDTG = 381.8 °C 100% | - | No residue |
| RIV-P188-HPMC (1:1:1) | TDTA = 57 °C (melting process of P188) TDTA = 224.6 °C (melting process of RIV) | 250–450 °C TDTA = 337.1 °C TDTA = 384.6 °C 74.4% | 450–700 °C 11.7% | 13.9% |
| RIV-P188-HPMC (1:1:2) | Below 60 °C (Dehydration process) 4.4% | 100–450 °C TDTG = 179.4 °C TDTA = 344.4 °C 87.6% | 450–700 °C 8.0% | No residue |
| RIV-P188-HPMC (1:2:1) | TDTA = 57.7 °C (melting process of P188) TDTA = 232.7 °C (melting process of RIV, with decomposition) | 190–400 °C TDTG = 335.2 °C TDTA = 382.7 °C 80.7% | 400–700 °C 19.3% | No residue |
| Product | Medium: pH 4.5 Acetate Buffer with 0.2% SDS | Medium: pH 6.8 Phosphate Buffer | ||||||
|---|---|---|---|---|---|---|---|---|
| Q15 (%) | Q30 (%) | Q120 (%) | DE% | Q30 (%) | Q60 (%) | Q180 (%) | DE% | |
| F1 | 59.6 ± 10.4 | 80.0 ± 11.3 | 104.4 ± 6.3 | 83.9 ± 2.8 | 49.0 ± 5.9 | 60.8 ± 3.1 | 67.9 ± 5.9 | 59.0 ± 0.9 |
| F2 | 18.5 ± 2.6 | 34.2 ± 3.3 | 72.4 ± 3.5 | 51.9 ± 2.1 | 32.9 ± 2.3 | 45.7 ± 1.8 | 58.3 ± 3.4 | 45.5 ± 0.3 |
| F3 | 67.6 ± 1.3 | 85.7 ± 5.9 | 106.5 ± 5.3 | 90.1 ± 1.9 | 57.7 ± 3.7 | 63.8 ± 1.6 | 67.8 ± 1.9 | 61.7 ± 1.1 |
| F4 | 61.7 ± 1.6 | 63.5 ± 2.2 | 73.0 ± 1.0 | 67.1 ± 1.5 | 45.6 ± 1.5 | 53.9 ± 2.1 | 57.7 ± 2.1 | 52.4 ± 1.2 |
| RIV powder | 54.1 ± 4.0 | 65.8 ± 4.3 | 69.8 ± 5.3 | 63.1 ± 2.3 | 11.8 ± 2.0 | 22.6 ± 2.1 | 34.4 ± 2.4 | 21.9 ± 0.8 |
| Xarelto® 10 mg | 85.0 ± 1.6 | 89.9 ± 0.3 | 93.8 ± 2.8 | 86.8 ± 1.6 | 45.8 ± 2.0 | 50.9 ± 2.3 | 54.3 ± 1.7 | 48.2 ± 1.2 |
| Comparison | f2 (pH 4.5) | f2 (pH 6.8) |
|---|---|---|
| F1 vs. Xarelto® 10 mg | N/A | 53.1 |
| F3 vs. Xarelto® 10 mg | N/A | 46.6 |
| F2 vs. Xarelto® 10 mg | N/A | 55.1 |
| F4 vs. Xarelto® 10 mg | N/A | 56.3 |
| F1 vs. F3 | 51.8 | 65.5 |
| F1 vs. F2 | 20.7 | 43.5 |
| F3 vs. F4 | 39.7 | 52.0 |
| F1 vs. RIV | 47.4 | 22.5 |
| F3 vs. RIV | 41.9 | 20.3 |
| F2 vs. RIV | 30.8 | 32.7 |
| F4 vs. RIV | 55.2 | 23.9 |
| Product | kKP (%·min−n) | n | tlag (min) | R2 |
|---|---|---|---|---|
| F1 | 17.55 | 0.32 | 4.52 | 0.9980 |
| F2 | 11.82 | 0.32 | 4.35 | 0.9784 |
| F3 | 15.86 | 0.39 | 3.10 | 0.9996 |
| F4 | 12.10 | 0.15 | 3.18 | 0.9634 |
| RIV powder | 4.06 | 0.41 | 14.07 | 0.9992 |
| Xarelto® 10 mg | 10.95 | 0.17 | 4.94 | 0.9457 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Benga, C.M.; Ozon, E.A.; Musuc, A.M.; Anuța, V.; Sârbu, I.; Surdu, V.-A.; Teodorescu, F.; Rusu, A.; Popa, L.; Ghica, M.V.; et al. Enhancement of the Solubility and Dissolution Profile of Rivaroxaban by the Antisolvent Precipitation Technique: A Promising Approach. Polymers 2026, 18, 1134. https://doi.org/10.3390/polym18091134
Benga CM, Ozon EA, Musuc AM, Anuța V, Sârbu I, Surdu V-A, Teodorescu F, Rusu A, Popa L, Ghica MV, et al. Enhancement of the Solubility and Dissolution Profile of Rivaroxaban by the Antisolvent Precipitation Technique: A Promising Approach. Polymers. 2026; 18(9):1134. https://doi.org/10.3390/polym18091134
Chicago/Turabian StyleBenga, Claudia Maria, Emma Adriana Ozon, Adina Magdalena Musuc, Valentina Anuța, Iulian Sârbu, Vasile-Adrian Surdu, Florina Teodorescu, Adriana Rusu, Lăcrămioara Popa, Mihaela Violeta Ghica, and et al. 2026. "Enhancement of the Solubility and Dissolution Profile of Rivaroxaban by the Antisolvent Precipitation Technique: A Promising Approach" Polymers 18, no. 9: 1134. https://doi.org/10.3390/polym18091134
APA StyleBenga, C. M., Ozon, E. A., Musuc, A. M., Anuța, V., Sârbu, I., Surdu, V.-A., Teodorescu, F., Rusu, A., Popa, L., Ghica, M. V., Chandak, A., & Pîrvu, C. E. D. (2026). Enhancement of the Solubility and Dissolution Profile of Rivaroxaban by the Antisolvent Precipitation Technique: A Promising Approach. Polymers, 18(9), 1134. https://doi.org/10.3390/polym18091134












