Solar-Driven Photocatalytic Degradation of Dye Pollutant Using MnO2-Modified Biochar via Fenton-like Reactions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Biochar Preparation
2.3. Biochar Catalyst Characterization Techniques
2.4. Fenton Reaction Sample Preparation
2.5. Reaction Set-Up and Analytical Calculations
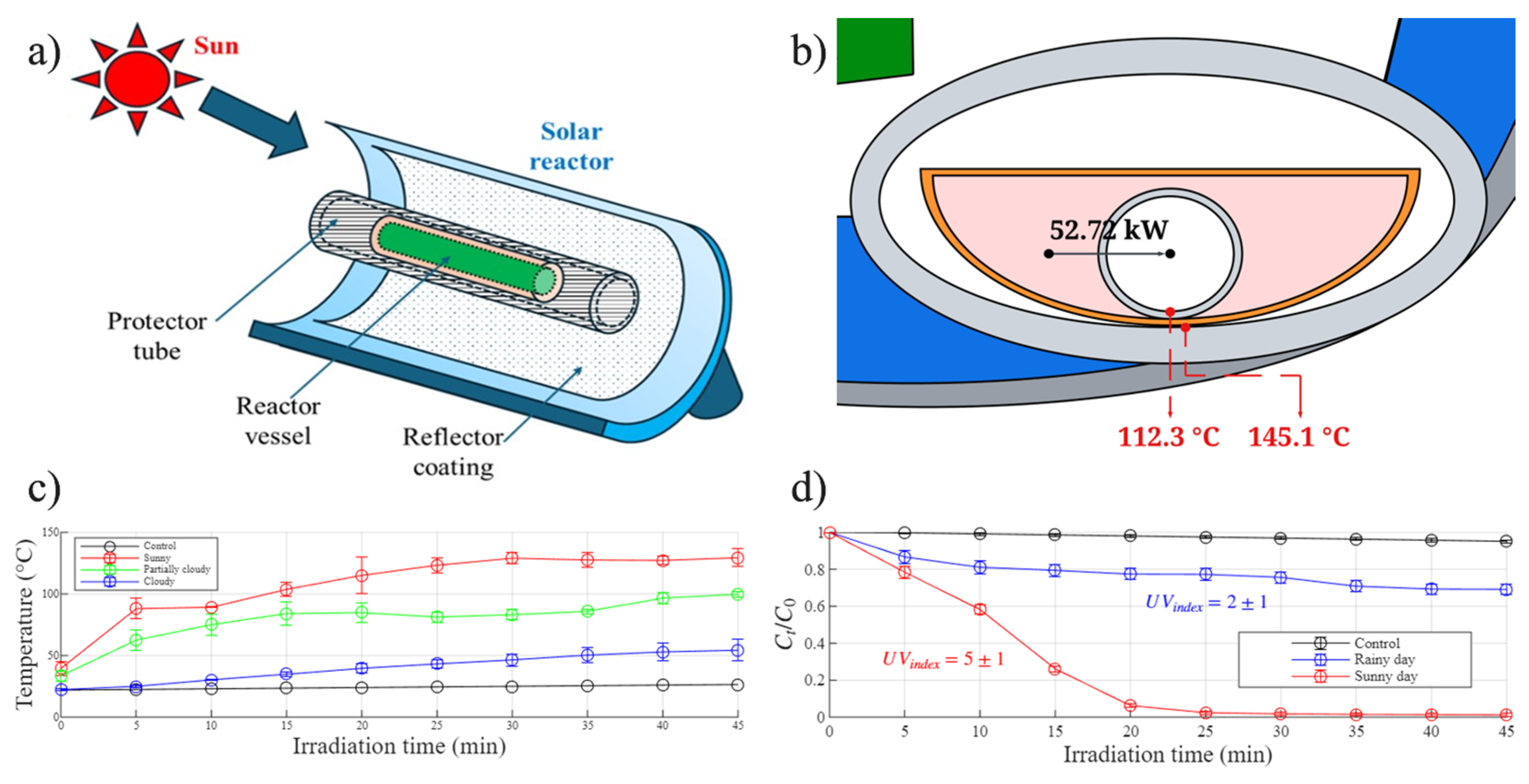
2.5.1. Solar Fenton Reactor
2.5.2. Dye Degradation Efficiency Evaluation and Reaction Kinetics Analysis
2.5.3. UV Reactor
2.5.4. Irradiation Efficiency Calculation Based on UV Index
2.5.5. Energy Balance Calculation
2.5.6. Catalyst Reusability Test
3. Results and Discussion
3.1. Comparison Between Solar and UV Fenton Systems
3.2. Catalyst Characterizations
3.2.1. X-Ray Diffraction (XRD) Analysis
3.2.2. Fourier Transform Infrared (FTIR) Analysis
3.2.3. Surface Morphology and Elemental Distribution Analysis
3.3. Effect of the Initial Dye Concentration and Catalyst Type
3.4. Reaction Kinetics of Different Catalysts
3.4.1. Reaction Kinetics Studied Using MW-Mn Catalysts
3.4.2. Reaction Kinetics Studied Using MW-HDPE-Mn Catalysts
3.4.3. Reaction Kinetics Studied Using MW-APS-Mn Catalysts
3.5. Prediction of Reactor Temperature and Dye Degradation Efficiency
3.6. Effects of Sunlight Spectrum Filtration
3.7. Reusability in MW-APS Biochar Catalyst
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| RhB | Rhodamine B |
| UV | Ultraviolet |
| AOPs | Advanced Oxidation Processes |
| ROS | Reactive oxygen species |
| MW | Maple wood |
| APS | Aerobic paper sludge |
| HDPE | high-density polyethylene |
| XRD | X-ray diffraction |
| FTIR | Fourier transform infrared |
| SEM | Scanning electron microscopy |
| EDS | Energy-dispersive spectroscopy |
| IMDA | Integral Method of Data Analysis |
References
- Al-Tohamy, R.; Ali, S.S.; Li, F.; Okasha, K.M.; Mahmoud, Y.A.G.; Elsamahy, T.; Jiao, H.; Fu, Y.; Sun, J. A critical review on the treatment of dye-containing wastewater: Ecotoxicological and health concerns of textile dyes and possible remediation approaches for environmental safety. Ecotoxicol. Environ. Saf. 2022, 231, 113160. [Google Scholar] [CrossRef]
- Amalina, F.; Abd Razak, A.S.; Krishnan, S.; Zularisam, A.W.; Nasrullah, M. A review of eco-sustainable techniques for the removal of Rhodamine B dye utilizing biomass residue adsorbents. Phys. Chem. Earth 2022, 128, 103267. [Google Scholar] [CrossRef]
- Yalasangi, V.; Mayilswamy, N.; Kandasubramanian, B. Biochar-derived adsorbents for removal of Rhodamine B from wastewater. Bioresour. Technol. Rep. 2024, 28, 101987. [Google Scholar] [CrossRef]
- Bushra, R.; Mohamad, S.; Alias, Y.; Jin, Y.; Ahmad, M. Current approaches and methodologies to explore the perceptive adsorption mechanism of dyes on low-cost agricultural waste: A review. Microporous Mesoporous Mater. 2021, 319, 111040. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, M.; Liu, H.; Gu, J.; Liu, Y. Environmental sustainability: A pressing challenge to biological sewage treatment processes. Curr. Opin. Environ. Sci. Health 2019, 12, 1–5. [Google Scholar] [CrossRef]
- Kumari, P.; Kumar, A. Advanced oxidation process: A remediation technique for organic and non-biodegradable pollutant. Results Surf. Interfaces 2023, 11, 100122. [Google Scholar] [CrossRef]
- dos Santos, C.R.; Rodrigues, A.C.; Vilasbôas, F.d.S.; Mistura, C.M.; Schneider, I.A.H. On the Fenton’s process for the treatment of effluents from the dyeing of agates containing Rhodamine B and ethanol. Discov. Chem. Eng. 2025, 5, 14. [Google Scholar] [CrossRef]
- Zhang, Y.; Shaad, K.; Vollmer, D.; Ma, C. Treatment of textile wastewater using advanced oxidation processes—A critical review. Water 2021, 13, 3515. [Google Scholar] [CrossRef]
- Xu, S.-L.; Wang, W.; Song, Y.; Tang, R.; Hu, Z.-H.; Zhou, X.; Yu, H.-Q. Expanding the pH range of Fenton-like reactions for pollutant degradation: The impact of acidic microenvironments. Water Res. 2025, 270, 122851. [Google Scholar] [CrossRef]
- Cuong, D.V.; Wu, P.-C.; Chen, L.-I.; Hou, C.-H. Active MnO2/biochar composite for efficient As(III) removal: Insight into the mechanisms of redox transformation and adsorption. Water Res. 2021, 188, 116495. [Google Scholar] [CrossRef]
- Angaru, G.K.R.; Pal, C.A.; Lingamdinne, L.P.; Husain, Z.M.; Kulkarni, R.; Choi, Y.-L.; Koduru, J.R.; Chang, Y.-Y. High-performance MnO2 embedded fly ash zeolite applied for effective mineralization of bisphenol-A and sorption of Congo red: Mechanism, real water application, and toxicity assessment. Chem. Eng. Sci. 2024, 286, 119700. [Google Scholar] [CrossRef]
- Laishram, D.; Kim, S.B.; Lee, S.Y.; Park, S.J. Advancements in biochar as a sustainable adsorbent for water pollution mitigation. Adv. Sci. 2025, 12, 2410383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, X.; Bian, W.; Peng, J.; Wan, H.; Zhao, J. The key role of persistent free radicals on the surface of hydrochar and pyrocarbon in the removal of heavy metal–organic combined pollutants. Bioresour. Technol. 2020, 318, 124046. [Google Scholar] [CrossRef] [PubMed]
- Martin-Montero, A.; Zapanti, A.M.; Pliego, G.; Casas, J.A.; Garcia-Costa, A.L. Solar photo-Fenton: An effective method for MCPA degradation. Processes 2025, 13, 2257. [Google Scholar] [CrossRef]
- Brillas, E. Solar photoelectro-Fenton: A very effective and cost-efficient electrochemical advanced oxidation process for the removal of organic pollutants from synthetic and real wastewaters. Chemosphere 2023, 327, 138532. [Google Scholar] [CrossRef]
- Tran, H.D.; Nguyen, D.Q.; Do, P.T.; Tran, U.N. Kinetics of photocatalytic degradation of organic compounds: A mini-review and new approach. RSC Adv. 2023, 13, 16915–16925. [Google Scholar] [CrossRef]
- Amakiri, K.T.; Angelis-Dimakis, A.; Ramirez Canon, A. Recent advances, influencing factors, and future research prospects using photocatalytic process for produced water treatment. Water Sci. Technol. 2022, 85, 769–788. [Google Scholar] [CrossRef]
- Fu, Q.; Xu, X.; Miao, R.; Wang, M.; Zhou, H.; He, L.; Guan, Q. Mn-embedded porous rubber seed shell biochar for enhanced removal of copper ions and catalytic efficacy of the used adsorbent for hydrogenation of furfural. Chem. Eng. J. 2022, 441, 136065. [Google Scholar] [CrossRef]
- Du, J.; Gao, L.; Yang, Y.; Chen, G.; Guo, S.; Omran, M.; Chen, J.; Ruan, R. Study on thermochemical characteristics properties and pyrolysis kinetics of the mixtures of waste corn stalk and pyrolusite. Bioresour. Technol. 2021, 324, 124660. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Q.; Zhang, J.; Liu, H.; Wang, H.; Zhang, J. Enhanced CO2 absorption in amine-based carbon capture aided by coconut shell-derived nitrogen-doped biochar. Sep. Purif. Technol. 2025, 353, 128451. [Google Scholar] [CrossRef]
- Tomin, O.; Yazdani, M.R. Production and characterization of porous magnetic biochar: Before and after phosphate adsorption insights. J. Porous Mater. 2022, 29, 849–859. [Google Scholar] [CrossRef]
- Zhang, X.; Tong, G.; Zhou, Y.; Li, G.; Zhang, H. Enhancing paper sludge dewatering by waste polyester fiber and FeCl3 for preparation of Fe-rich biochar. BioResources 2021, 16, 2326. [Google Scholar] [CrossRef]
- Niu, J.; Fan, S.; Wu, Z. Preparation of biochar from papermaking sludge and its adsorption characteristics for tetracycline. Toxics 2025, 13, 1050. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Khan, M.U.; Lin, X.; Cai, H.; Lei, H. Temperature varied biochar as a reinforcing filler for high-density polyethylene composites. Compos. Part B Eng. 2019, 175, 107151. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, D.; Lu, W.; Khan, M.U.; Xu, H.; Yi, W.; Lei, H.; Huo, E.; Qian, M.; Zhao, Y.; et al. Production of high-density polyethylene biocomposites from rice husk biochar: Effects of varying pyrolysis temperature. Sci. Total Environ. 2020, 738, 139910. [Google Scholar] [CrossRef]
- Dutta, B.; Raghavan, V.G.S.; Orsat, V.; Ngadi, M. Surface characterisation and classification of microwave pyrolysed maple wood biochar. Biosyst. Eng. 2015, 131, 49–64. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Liu, Y.; Chen, Y.; Chen, L.; Yan, H.; Chen, Y.; Xu, F.; Li, M.; Li, L. Modification of sludge biochar by MnO2 to degrade methylene blue: Synergistic catalysis and degradation mechanisms. J. Water Process Eng. 2022, 48, 102864. [Google Scholar] [CrossRef]
- Zhou, L.; Huang, Y.; Qiu, W.; Sun, Z.; Liu, Z.; Song, Z. Adsorption properties of nano-MnO2–biochar composites for copper in aqueous solution. Molecules 2017, 22, 173. [Google Scholar] [CrossRef]
- Chen, Y.-D.; Ho, S.-H.; Wang, D.; Wei, Z.-S.; Chang, J.-S.; Ren, N.-Q. Lead removal by a magnetic biochar derived from persulfate-ZVI treated sludge together with one-pot pyrolysis. Bioresour. Technol. 2018, 247, 463–470. [Google Scholar] [CrossRef]
- Biswas, B.; Pandey, N.; Bisht, Y.; Singh, R.; Kumar, J.; Bhaskar, T. Pyrolysis of agricultural biomass residues: Comparative study of corn cob, wheat straw, rice straw and rice husk. Bioresour. Technol. 2017, 237, 57–63. [Google Scholar] [CrossRef]
- Siddique, M.A.B.; Bithi, U.H.; Ahmed, A.N.; Gafur, M.; Reaz, A.H.; Roy, C.K.; Islam, M.M.; Firoz, S.H. Preparation of manganese oxide nanoparticles with enhanced capacitive properties utilizing gel formation method. ACS Omega 2022, 7, 48007–48017. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zou, H. Ultra-efficient catalytic degradation of malachite green dye wastewater by KMnO4-modified biochar (Mn/SRBC). RSC Adv. 2022, 12, 27002–27011. [Google Scholar] [CrossRef]
- Li, R.; Wang, Z.; Zhao, X.; Li, X.; Xie, X. Magnetic biochar-based manganese oxide composite for enhanced fluoroquinolone antibiotic removal from water. Environ. Sci. Pollut. Res. 2018, 25, 31136–31148. [Google Scholar] [CrossRef]
- Zhang, M.; Lin, K.; Li, X.; Wu, L.; Yu, J.; Cao, S.; Zhang, D.; Xu, L.; Parikh, S.J.; Ok, Y.S. Removal of phosphate from water by paper mill sludge biochar. Environ. Pollut. 2022, 293, 118521. [Google Scholar] [CrossRef]










Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sandoval, J.A.S.; Ragib, A.A.; Kozinski, J.; Rakshit, S.K.; Kang, K. Solar-Driven Photocatalytic Degradation of Dye Pollutant Using MnO2-Modified Biochar via Fenton-like Reactions. Polymers 2026, 18, 1119. https://doi.org/10.3390/polym18091119
Sandoval JAS, Ragib AA, Kozinski J, Rakshit SK, Kang K. Solar-Driven Photocatalytic Degradation of Dye Pollutant Using MnO2-Modified Biochar via Fenton-like Reactions. Polymers. 2026; 18(9):1119. https://doi.org/10.3390/polym18091119
Chicago/Turabian StyleSandoval, Jorge A. Soto, Abdullah Al Ragib, Janusz Kozinski, Sudip K. Rakshit, and Kang Kang. 2026. "Solar-Driven Photocatalytic Degradation of Dye Pollutant Using MnO2-Modified Biochar via Fenton-like Reactions" Polymers 18, no. 9: 1119. https://doi.org/10.3390/polym18091119
APA StyleSandoval, J. A. S., Ragib, A. A., Kozinski, J., Rakshit, S. K., & Kang, K. (2026). Solar-Driven Photocatalytic Degradation of Dye Pollutant Using MnO2-Modified Biochar via Fenton-like Reactions. Polymers, 18(9), 1119. https://doi.org/10.3390/polym18091119









