Cure Modelling and Monitoring for Isothermal Processing of Fast-Curing Epoxy Resin
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterisation
2.2.1. Mathematical Model of Glass Transition Temperature
2.2.2. Modelling of Reaction Kinetics
3. Results and Discussion
3.1. Evaluation of DSC Measurements
3.2. Modelling of Glass Transition Temperature
3.3. Modelling of the Degree of Cure
3.4. Parameter Sensitivity Analysis
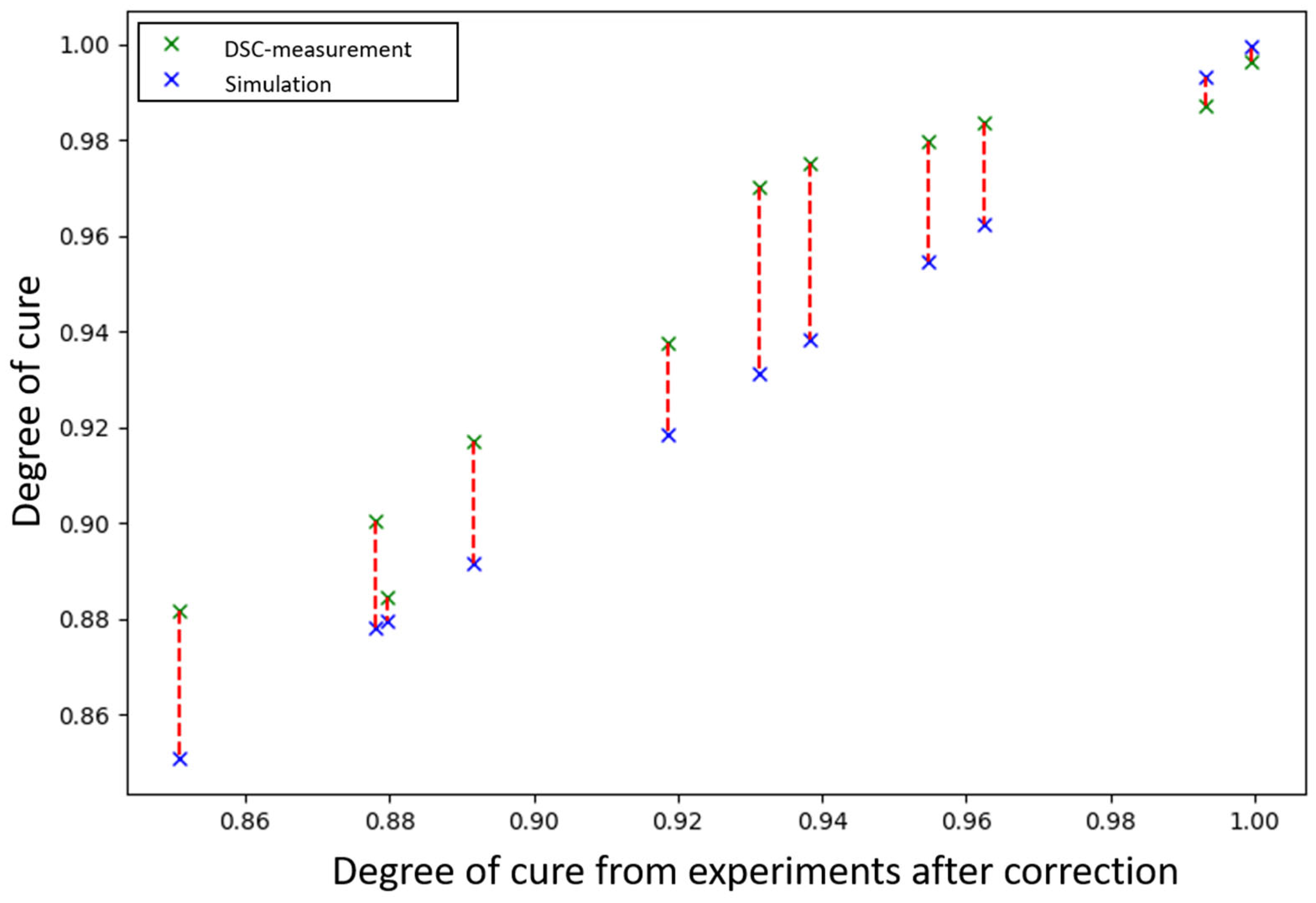
3.5. Experimental Investigation of the Degree of Cure
- The residual heat in the sample part and the heat released by the exothermic curing reaction cause the reaction to continue after the measurement has ended.
- When the reaction has reached its maximum, a large amount of heat is released. Due to the thermal inertia of the mould and the sensor, this is not recorded immediately. In this case, the resin temperature is higher than the measured value.
3.6. Uncertainty Analysis of Temperature Measurement
- Intrinsic sensor accuracy;
- Uncertainty of the data acquisition system;
- Measurement repeatability;
- Installation-related effects caused by sensor placement and thermal coupling.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Jin, F.-L.; Li, X.; Park, S.-J. Synthesis and application of epoxy resins: A review. J. Ind. Eng. Chem. 2015, 29, 1–11. [Google Scholar] [CrossRef]
- Müller-Pabel, M.; Rodríguez Agudo, J.A.; Gude, M. Measuring and understanding cure-dependent viscoelastic properties of epoxy resin: A review. Polym. Test. 2022, 114, 107701. [Google Scholar] [CrossRef]
- Hiken, A. The Evolution of the Composite Fuselage: A Manufacturing Perspective. In Aerospace Engineering; Dekoulis, G., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Sourour, S.; Kamal, M.R. Differential scanning calorimetry of epoxy cure: Isothermal cure kinetics. Thermochim. Acta 1976, 14, 41–59. [Google Scholar] [CrossRef]
- Grindling, J. Simulation zur Verarbeitung von reaktiven Non-Post-Cure-Epoxidharz-Systemen im Druckgelieren und konventionellen Vergießen. In Schriftenreihe der Verfahrenstechnik Universität Paderborn; Shaker: Aachen, Germany, 2006; Volume 31. [Google Scholar]
- DiBenedetto, A.T. Prediction of the glass transition temperature of polymers: A model based on the principle of corresponding states. J. Polym. Sci. Part B Polym. Phys. 1987, 25, 1949–1969. [Google Scholar] [CrossRef]
- Kyriazis, A.; Pommer, C.; Lohuis, D.; Rager, K.; Dietzel, A.; Sinapius, M. Comparison of Different Cure Monitoring Techniques. Sensors 2022, 22, 7301. [Google Scholar] [CrossRef] [PubMed]
- TD_SR_8500_SZ_8525_20151228. 24 January 2014. Available online: https://sicomin.com/datasheets/sr8500_sz8525.pdf (accessed on 16 March 2026).
- Vyazovkin, S.; Burnham, A.K.; Criado, J.M.; Pérez-Maqueda, L.A.; Popescu, C.; Sbirrazzuoli, N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim. Acta 2011, 520, 1–19. [Google Scholar] [CrossRef]
- Fava, R.A. Differential scanning calorimetry of epoxy resins. Polymer 1968, 9, 137–151. [Google Scholar] [CrossRef]
- Hutchinson, J.M. Determination of the glass transition temperature: Methods correlation and structural heterogeneity. J. Therm. Anal. Calorim. 2009, 98, 579–589. [Google Scholar] [CrossRef]
- Plazek, D.J.; Frund, Z.N. Epoxy resins (DGEBA): The curing and physical aging process. J. Polym. Sci. Part B Polym. Phys. 1990, 28, 431–448. [Google Scholar] [CrossRef]
- Bernath, A.; Kärger, L.; Henning, F. Accurate Cure Modeling for Isothermal Processing of Fast Curing Epoxy Resins. Polymers 2016, 8, 390. [Google Scholar] [CrossRef] [PubMed]




| 60 °C | 392.73 | 95.04 | 0.8051 |
| 80 °C | 450.24 | 40.27 | 0.9179 |
| 100 °C | 473.47 | 18.08 | 0.9632 |
| 120 °C | 397.22 | 9.36 | 0.9760 |
| 1 K/min | 445.07 |
| 2.5 K/min | 462.29 |
| 5 K/min | 455.78 |
| 10 K/min | 461.73 |
| 15 K/min | 448.09 |
| Experiment | ||
|---|---|---|
| 60 °C isothermal | 0.8051 | 66.31 °C |
| 80 °C isothermal | 0.9179 | 101.7 °C |
| 100 °C isothermal | 0.9632 | 109.3 °C |
| 120 °C isothermal | 0.9760 | 116.87 °C |
| 2.5 K/min dynamic | 0 | −37.2 °C |
| Parameter | Unit | |
|---|---|---|
| 0.7182 | - | |
| 2.375 | - | |
| 397.867 | K | |
| 0.4477 | - |
| Parameter | Unit | |
|---|---|---|
| - | ||
| - | ||
| - | ||
| - | ||
| - | ||
| - |
| ID | Duration in s | Average Temperature During Experiment in °C | Degree of Cure Based on Model | Comment |
|---|---|---|---|---|
| #01 | 1106.13 | 97.95 | 0.9447 | |
| #02 | 978.32 | 98.15 | 0.9346 | |
| #03 | 686.66 | 98.05 | 0.9109 | |
| #04 | 624.51 | 97.55 | 0.8919 | |
| #05 | 1331.46 | 78.55 | 0.8614 | |
| #06 | 943.13 | 79.35 | 0.7793 | |
| #07 | 1109.28 | 79.85 | 0.8249 | |
| #08 | - | - | - | Failed due to premature opening of mould |
| #09 | - | - | - | Failed due to premature opening of mould |
| #10 | 1016.40 | 118.15 | 0.9859 | |
| #11 | 2427.76 | 69.65 | 0.8617 | |
| #12 | 2320.53 | 79.65 | 0.9023 | |
| #13 | 1983.72 | 126.15 | 0.9992 |
| ID | Degree of Cure Based on Model After Correction | Degree of Cure Based on DSC Measurement | Deviation |
|---|---|---|---|
| #01 | 0.9624 | 0.9836 | 0.0212 |
| #02 | 0.9548 | 0.9796 | 0.0248 |
| #03 | 0.9384 | 0.9750 | 0.0366 |
| #04 | 0.9314 | 0.9702 | 0.0388 |
| #05 | 0.8916 | 0.9172 | 0.0256 |
| #06 | 0.8509 | 0.8817 | 0.0308 |
| #07 | 0.8780 | 0.9006 | 0.0226 |
| #10 | 0.9932 | 0.9872 | 0.006 |
| #11 | 0.8797 | 0.8844 | 0.0047 |
| #12 | 0.9187 | 0.9377 | 0.019 |
| #13 | 0.9996 | 0.9966 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Schaible, P.; Schwaiberger, D.; Schabel, S.; Fleischer, J. Cure Modelling and Monitoring for Isothermal Processing of Fast-Curing Epoxy Resin. Polymers 2026, 18, 952. https://doi.org/10.3390/polym18080952
Schaible P, Schwaiberger D, Schabel S, Fleischer J. Cure Modelling and Monitoring for Isothermal Processing of Fast-Curing Epoxy Resin. Polymers. 2026; 18(8):952. https://doi.org/10.3390/polym18080952
Chicago/Turabian StyleSchaible, Patrick, David Schwaiberger, Sebastian Schabel, and Jürgen Fleischer. 2026. "Cure Modelling and Monitoring for Isothermal Processing of Fast-Curing Epoxy Resin" Polymers 18, no. 8: 952. https://doi.org/10.3390/polym18080952
APA StyleSchaible, P., Schwaiberger, D., Schabel, S., & Fleischer, J. (2026). Cure Modelling and Monitoring for Isothermal Processing of Fast-Curing Epoxy Resin. Polymers, 18(8), 952. https://doi.org/10.3390/polym18080952









