Stabilizing the Sealing Performance of EPDM by the Incorporation of a ZIF-8 Network
Abstract
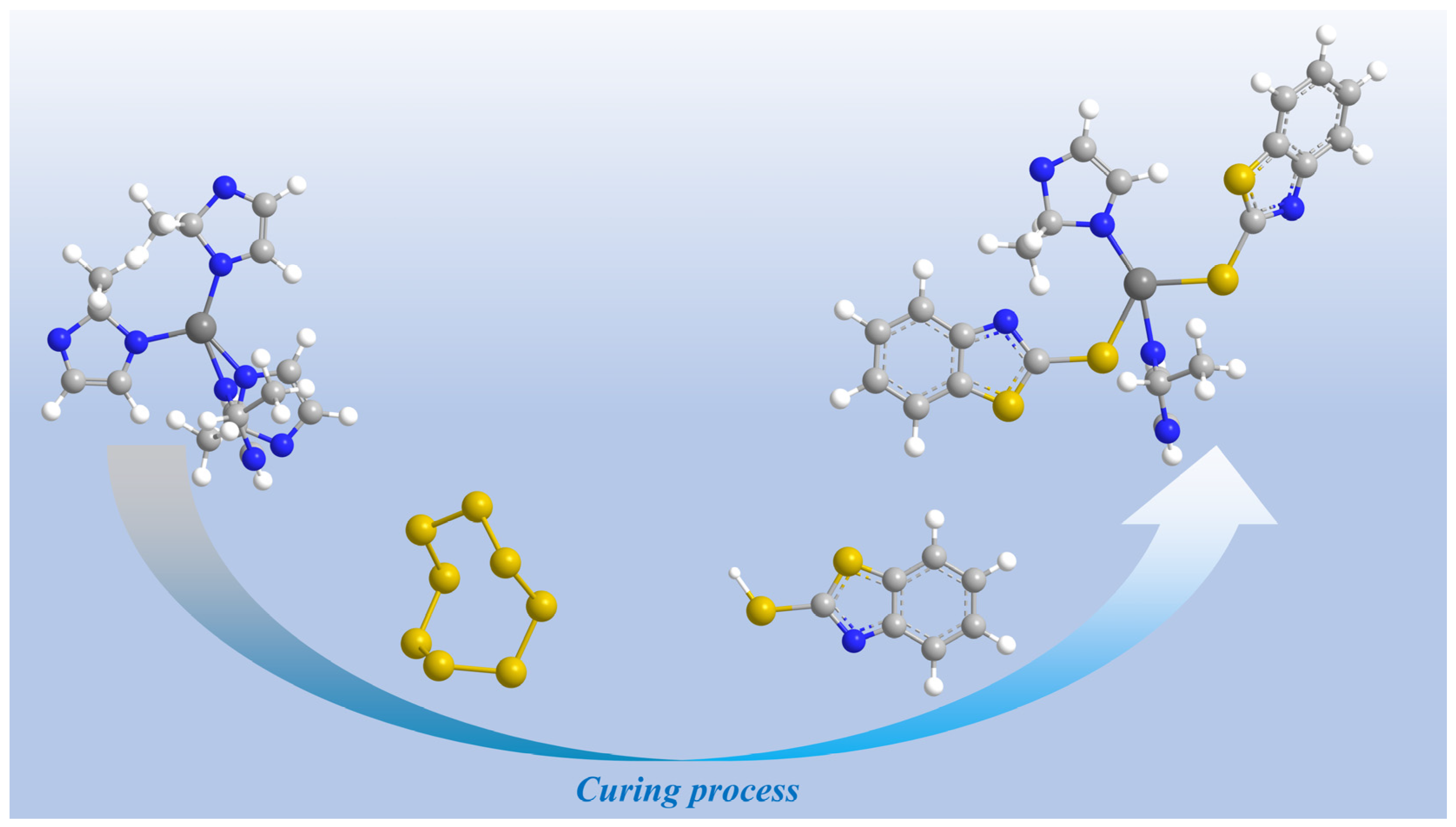
1. Introduction
2. Experimental Procedure
2.1. Materials
2.2. Accelerated Aging Experiments
2.3. Transmission Electron Microscopy (TEM)
2.4. Crosslink Density
2.5. X-Ray Photoelectron Spectroscopy (XPS)
2.6. ATR-FTIR Analysis
2.7. Thermogravimetric Analysis (TGA)
3. Results and Discussion
3.1. Modification of EPDM
3.1.1. ZIF-8 Preparation
3.1.2. ZIF-8 Characterization
3.1.3. EPDM Mixing and ZIF-8 Incorporation
3.2. Mechanical Performance and Aging
3.3. Analysis of Performance Degradation After Aging
3.4. Characterization Before Aging
3.5. Characterization After Aging
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ziegler, A.D.; Fox, J.M.; Xu, J. The Rubber Juggernaut. Science 2009, 324, 1024–1025. [Google Scholar] [CrossRef]
- Wang, M.; Li, Y.; Qian, C.; Wang, S.; Liu, D. Relationship between the aging thermal oxygen and mechanical properties of nitrile butadiene rubber reinforced by RD-loaded carboxylated carbon nanotubes. Appl. Surf. Sci. 2024, 655, 159569. [Google Scholar] [CrossRef]
- Choi, E.Y.; Kim, C.K.; Park, C.B. Fabrication of MA-EPDM grafted MWCNTs by reactive extrusion for enhanced interfacial adhesion and mechanical properties of PP/MA-EPDM composite. Compos. Part B 2022, 242, 110043. [Google Scholar] [CrossRef]
- Qiao, X.; Miao, Y.; Zhang, X.; Shen, H.; Hua, Y.; Sun, L.; Huang, Y.; Xu, J.; Wu, D. An analytical model for stress distribution of NBR O-ring during dynamic hydrogen permeation. Int. J. Hydrogen Energy 2025, 109, 164–173. [Google Scholar] [CrossRef]
- Colom, X.; Marín, M.; Saeb, M.R.; Formela, K.; Cañavate, J. Recycling devulcanized EPDM to improve engineering properties of SBR rubber compounds. Resour. Conserv. Recycl. Adv. 2024, 23, 200227. [Google Scholar] [CrossRef]
- Krejsa, M.R.; Koenig, J.L. A Review of Sulfur Crosslinking Fundamentals for Accelerated and Unaccelerated Vulcanization. Rubber Chem. Technol. 1993, 66, 376–410. [Google Scholar] [CrossRef]
- Ducháček, V.; Kuta, A.; Pr̆ibyl, P. Efficiency of metal activators of accelerated sulfur vulcanization. J. Appl. Polym. Sci. 1993, 47, 743–746. [Google Scholar] [CrossRef]
- Suzuki, T.; Osaka, N.; Endo, H.; Shibayama, M.; Ikeda, Y.; Asai, H.; Higashitani, N.; Kokubo, Y.; Kohjiya, S. Nonuniformity in Cross-Linked Natural Rubber as Revealed by Contrast-Variation Small-Angle Neutron Scattering. Macromolecules 2010, 43, 1556–1563. [Google Scholar] [CrossRef]
- Ikeda, Y.; Higashitani, N.; Hijikata, K.; Kokubo, Y.; Morita, Y.; Shibayama, M.; Osaka, N.; Suzuki, T.; Endo, H.; Kohjiya, S. Vulcanization: New Focus on a Traditional Technology by Small-Angle Neutron Scattering. Macromolecules 2009, 42, 2741–2748. [Google Scholar] [CrossRef]
- Ikeda, Y.; Yasuda, Y.; Ohashi, T.; Yokohama, H.; Minoda, S.; Kobayashi, H.; Honma, T. Dinuclear Bridging Bidentate Zinc/Stearate Complex in Sulfur Cross-Linking of Rubber. Macromolecules 2015, 48, 462–475. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, L.; Wu, W.; Cheng, Z.; Sun, Y.; Jiang, H.; Li, C. Zinc oxide with dominant (1 0 0) facets boosts vulcanization activity. Eur. Polym. J. 2019, 113, 148–154. [Google Scholar] [CrossRef]
- Lee, Y.H.; Cho, M.; Nam, J.-D.; Lee, Y. Effect of ZnO particle sizes on thermal aging behavior of natural rubber vulcanizates. Polym. Degrad. Stab. 2018, 148, 50–55. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, Y.; Lu, G.; Wang, Y.; Li, S.; Cui, C.; Wu, J.; Xu, Z.; Tian, D.; Huang, W. Mesoporous metal-organic frameworks with size-, shape-, and space-distribution-controlled pore structure. Adv. Mater. 2015, 27, 2923–2929. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-Z.; Wang, C.; Wu, Z.-Y.; Xiong, Y.; Xu, Q.; Yu, S.-H.; Jiang, H.-L. From Bimetallic Metal-Organic Framework to Porous Carbon: High Surface Area and Multicomponent Active Dopants for Excellent Electrocatalysis. Adv. Mater. 2015, 27, 5010–5016. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Hu, P. Progress on fundamentals of adsorption transport of metal-organic frameworks materials and sustainable applications for water harvesting and carbon capture. J. Clean. Prod. 2023, 393, 136253. [Google Scholar] [CrossRef]
- Xiong, Q.; Chen, Y.; Yang, D.; Wang, K.; Wang, Y.; Yang, J.; Li, L.; Li, J. Constructing strategies for hierarchically porous MOFs with different pore sizes and applications in adsorption and catalysis. Mater. Chem. Front. 2022, 6, 2944–2967. [Google Scholar] [CrossRef]
- Klajn, K.; Gozdek, T.; Bieliński, D.M. Metal Organic Frameworks: Current State and Analysis of Their Use as Modifiers of the Vulcanization Process and Properties of Rubber. Materials 2023, 16, 7631. [Google Scholar] [CrossRef]
- Tian, X.; Hu, J.; Sun, X.; Duan, Y.; Jiang, M. Novel application of Zn-containing Zeolitic Imidazolate Frameworks in promoting the vulcanization and mechanical properties of natural rubber composites. J. Appl. Polym. Sci. 2023, 140, e54294. [Google Scholar] [CrossRef]
- Yang, M.; Yasin, S.; Chen, Q.; Peng, W.; Lv, Z.; Qi, Z.; Shi, J. Suppressing high pressure hydrogen blistering in NBR via Zn-based MOF vulcanization: Microstructural and mechanical insights. Int. J. Hydrogen Energy 2025, 181, 151810. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, X.; Gao, J. Aging of ethylene–propylene–diene monomer (EPDM) in artificial weathering environment. Polym. Degrad. Stab. 2007, 92, 1841–1846. [Google Scholar] [CrossRef]
- Zheng, C.; Wang, Y.; Phua, S.Z.F.; Lim, W.Q.; Zhao, Y. ZnO–DOX@ZIF-8 core–shell nanoparticles for pH-responsive drug delivery. ACS Biomater. Sci. Eng. 2017, 3, 2223–2229. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-R.; Dong, X.; Pan, S.-Y.; Luo, L.; Lei, H.-T.; Xu, Z.-L. Design to enhance sensing performance of ZIF-8 crystals. Prog. Nat. Sci. Mater. Int. 2024, 34, 240–250. [Google Scholar] [CrossRef]
- Venna, S.R.; Jasinski, J.B.; Carreon, M.A. Structural evolution of zeolitic imidazolate framework-8. J. Am. Chem. Soc. 2010, 132, 18030–18033. [Google Scholar] [CrossRef] [PubMed]
- Mott, P.H.; Roland, C.M. Aging of Natural Rubber in Air and Seawater. Rubber Chem. Technol. 2001, 74, 79–88. [Google Scholar] [CrossRef]
- Delor-Jestin, F.; Barrois-Oudin, N.; Cardinet, C.; Lacoste, J.; Lemaire, J. Thermal ageing of acrylonitrile-butadiene copolymer. Polym. Degrad. Stab. 2000, 70, 1–4. [Google Scholar] [CrossRef]
- Andrews, R.D.; Tobolsky, A.V.; Hanson, E.E. The Theory of Permanent Set at Elevated Temperatures in Natural and Synthetic Rubber Vulcanizates. J. Appl. Phys. 1946, 17, 352–361. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, R.; Iervolino, R.; Barbera, S. Investigation of crosslinking in the thermooxidative aging of nitrile–butadiene rubber. J. Appl. Polym. Sci. 2015, 132, 41319. [Google Scholar] [CrossRef]
- Yang, Y.; Qiao, X.; Fan, X.; Liao, S.; Sun, Z. Use of ZIF-8 as vulcanization additive to optimize natural rubber composites to lower the amount of ZnO. J. Vinyl Addit. Tech. 2024, 30, 1039–1051. [Google Scholar] [CrossRef]
- Xie, W.-L.; Hou, Z.-B.; Liu, G.-X.; Liao, S.; Luo, M.-C. Nucleophiles realizing the reduction of ZnO content for vulcanization accompanied by the improvement in thermo-oxidative resistance for vulcanized isoprene rubbers. J. Appl. Polym. Sci. 2024, 141, e55380. [Google Scholar] [CrossRef]
- Sharma, S.; Sudarshan, K.; Yadav, A.; Jha, S.; Bhattacharyya, D.; Pujari, P. Investigation of compression-induced deformations in local structure and pore architecture of ZIF-8 using FTIR, X-ray absorption, and positron annihilation spectroscopy. J. Phys. Chem. C 2019, 123, 22273–22280. [Google Scholar] [CrossRef]
- Chang-In, T.; Anancharoenwong, E.; Suchat, S. Synergistic Effects of Nitrosamine-Safe Accelerators for Enhanced Natural Rubber Latex Balloon in Sulfur Conventional Vulcanizing System. Polymers 2026, 18, 438. [Google Scholar] [CrossRef] [PubMed]
- Tabbal, M.; Merel, P.; Moisa, S.; Chaker, M.; Gat, E.; Ricard, A.; Moisan, M.; Gujrathi, S. XPS and FTIR analysis of nitrogen incorporation in CNx thin films. Surf. Coat. Technol. 1998, 98, 1092–1096. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, Z.; Li, Y.; Lv, M.; Song, S.; Wu, Z.; Yan, J.; Han, S. Structural and optical properties of zinc nitride films prepared by rf magnetron sputtering. Appl. Surf. Sci. 2009, 255, 3544–3547. [Google Scholar] [CrossRef]
- He, X.; Zhou, H.; Zhang, Z.; Ren, T.; Wang, S. Thermo-oxidative Degradation Behavior of Natural Rubber Induced by Sulfur Crosslinking Structures. Polym. Degrad. Stab. 2025, 242, 111706. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, G.; Yu, Y.; Feng, Y.; Zheng, Y.; He, F.; Han, Z. An efficient method to enhance the stability of sulphide semiconductor photocatalysts: A case study of N-doped ZnS. Phys. Chem. Chem. Phys. 2015, 17, 1870–1876. [Google Scholar] [CrossRef]
- Li, B.; Li, S.-X.; Shen, M.-X.; Xiao, Y.-L.; Zhang, J.; Xiong, G.-Y.; Zhang, Z.-N. Tribological behaviour of acrylonitrile-butadiene rubber under thermal oxidation ageing. Polym. Test. 2021, 93, 106954. [Google Scholar] [CrossRef]
- Fernández-Berridi, M.J.; González, N.; Mugica, A.; Bernicot, C. Pyrolysis-FTIR and TGA techniques as tools in the characterization of blends of natural rubber and SBR. Thermochim. Acta 2006, 444, 65–70. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, J. Thermal Conductivity of Zeolitic Imidazolate Framework-8: A Molecular Simulation Study. J. Phys. Chem. C 2013, 117, 18441–18447. [Google Scholar] [CrossRef]









| Composition (Phr) | EPDM | EPDM-ZIF-8 |
|---|---|---|
| EPDM | 100 | 100 |
| Silica | 40 | 40 |
| Sulfur | 2 | 2 |
| Steric acid | 2 | 2 |
| MBT | 1 | 1 |
| 6PPD | 1 | 1 |
| ZnO | 2 | 0 |
| ZIF-8 capped ZnO | 0 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chen, J.; Peng, Q.; Liu, H.; Xiao, X.; Fu, X.; Wen, H.; Huang, Z.; Wang, F.; Zeng, X. Stabilizing the Sealing Performance of EPDM by the Incorporation of a ZIF-8 Network. Polymers 2026, 18, 874. https://doi.org/10.3390/polym18070874
Chen J, Peng Q, Liu H, Xiao X, Fu X, Wen H, Huang Z, Wang F, Zeng X. Stabilizing the Sealing Performance of EPDM by the Incorporation of a ZIF-8 Network. Polymers. 2026; 18(7):874. https://doi.org/10.3390/polym18070874
Chicago/Turabian StyleChen, Jiahui, Qian Peng, Huadong Liu, Xingtao Xiao, Xiaotao Fu, Hanlin Wen, Zhicheng Huang, Fangqiang Wang, and Xiaoliang Zeng. 2026. "Stabilizing the Sealing Performance of EPDM by the Incorporation of a ZIF-8 Network" Polymers 18, no. 7: 874. https://doi.org/10.3390/polym18070874
APA StyleChen, J., Peng, Q., Liu, H., Xiao, X., Fu, X., Wen, H., Huang, Z., Wang, F., & Zeng, X. (2026). Stabilizing the Sealing Performance of EPDM by the Incorporation of a ZIF-8 Network. Polymers, 18(7), 874. https://doi.org/10.3390/polym18070874





