Effect of Inorganic Salts on Synthesis of Poly(glycidyl methacrylate) Microspheres, Their Functionalization with Poly(ethyleneimine) and Evaluation of Its Use for Removal of Acid Red 27, Acetaminophen and Nitrites
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Synthesis of MgONPs and CaCO3NPs
2.3. Synthesis of the Crosslinked PGMA
2.3.1. Synthesis of PGMA Particles
2.3.2. PEI Chain Grafts on PGMA Spheres
2.3.3. Analysis of the Effect of InAds on Aqueous PVA Solutions
2.3.4. PGMA–(InAds)–PEI Resin
2.4. Characterization Analyses
2.4.1. Infrared and UV–Vis Spectroscopic Characterization
2.4.2. Thermal Characterization
2.4.3. Morphological Characterization
2.4.4. Determination of Water Absorption Capacity (WAC)
2.4.5. Determination of Epoxy Group Content
2.4.6. Adsorption Experiments of , AR-27, and
2.4.7. Desorption of AR-27 and
3. Results and Discussion
3.1. Synthesis of MgONPs and CaCO3NPs
3.2. Effect of Polymerization Type and MBA Content on PGMA Synthesis
3.3. PGMA Resins Obtained by Suspension Polymerization with Addition of NaCl, MgONPs and CaCO3NPs
3.3.1. Effect of the Aqueous-Phase Saline Composition on Suspension Polymerization
3.3.2. Relationship Between Aqueous-Phase Composition and PGMA Morphology
3.3.3. Morphological Characterization by SEM
3.3.4. Spectroscopic Characterization of PGMA
3.3.5. Thermal Characterization of PGMA by TGA and DSC
3.3.6. WAC and EV of PGMA
3.4. Anchoring of PEI Groups onto PGMA
3.4.1. Spectroscopic Characterization
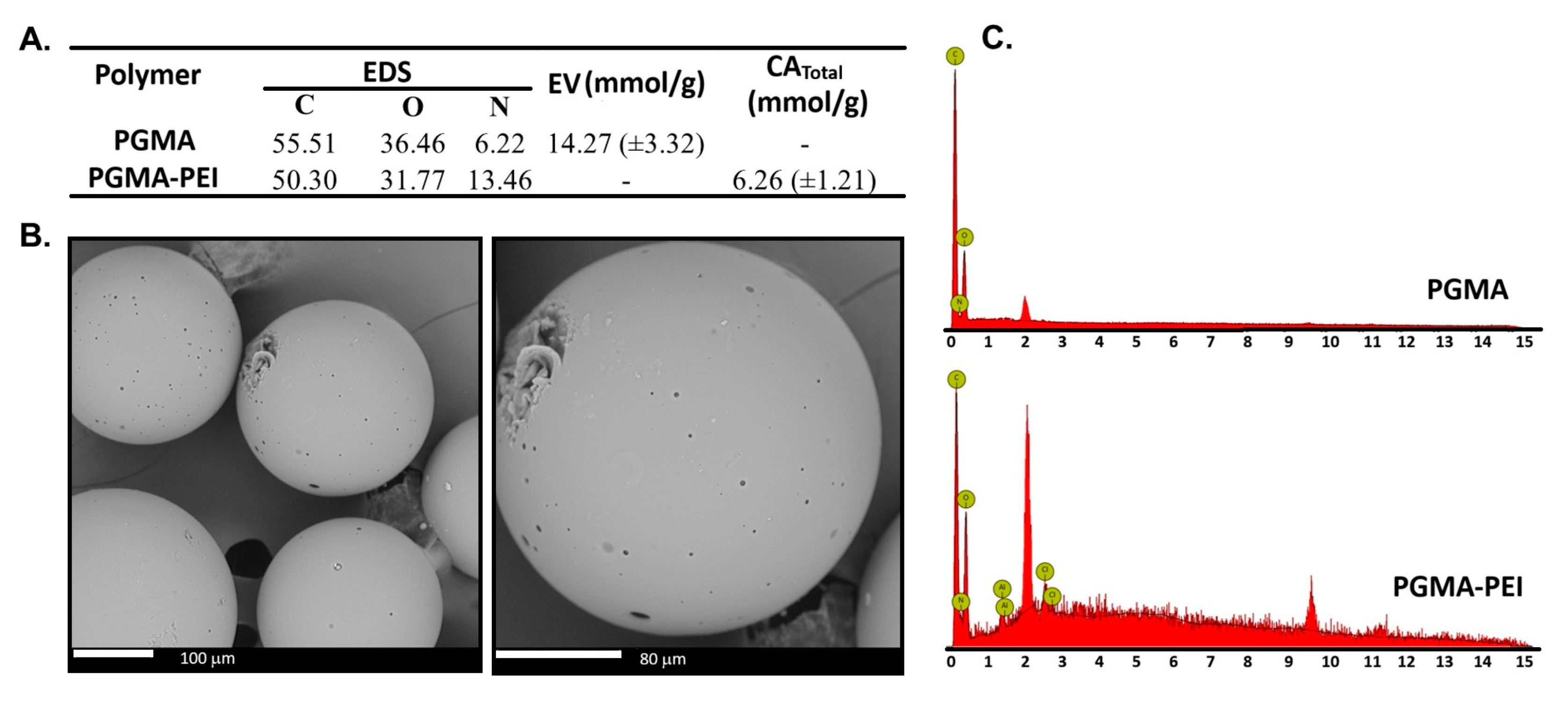
3.4.2. WAC, EV and SEM–EDS
3.4.3. Thermal Analysis: TGA
3.5. Adsorption Behavior of AR-27 Dye and Act on PGMA–PEI
Retention Experiments: AR-27
3.6. Adsorption of on PGMA–PEI
3.7. Desorption Experiments
3.7.1. Desorption of AR-27
3.7.2. Desorption of
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACT | Acetaminophen |
| AD-1 or -2 | Adsorption–desorption cycles |
| Amax | Maximum absorbance |
| AR-27 | Acid red 27 |
| ATR | Attenuated total reflectance |
| BzO2 | Benzoyl peroxide |
| Total functional group loading | |
| CaCO3NPs | CaCO3 nanoparticles |
| Initial concentration | |
| DSC | Differential scanning calorimetry |
| DLS | Dynamic light scattering |
| EDS | Energy-dispersive X-ray spectroscopy |
| EV | Epoxy value |
| FTIR | Fourier transform infrared spectroscopy |
| GMA | Glycidyl methacrylate |
| InAds | Inorganic additives |
| IS | Ionic strength |
| MBAAm | N,N′-methylenebisacrylamide |
| Wetted membrane–funnel assembly (Equation (1)) | |
| Mass of the wetted membrane–funnel–wet resin assembly (Equation (1)) | |
| MgONPs | MgO nanoparticles |
| Average molecular weight in number | |
| Mass of the dry resin (Equation (1)) | |
| MRC | Maximum retention capacity |
| Mw | Average molecular weight in mass |
| PDI | Polydispersity index |
| PEI | Polyethyleneimine |
| PGMA | Poly(glycidyl methacrylate) |
| PVA | Poly(vinyl alcohol) |
| R | Removal percentage |
| Rh | Hydrodynamic radius |
| SEM | Scanning electron microscopy |
| TGA | Thermogravimetric analysis |
| Tpol | Polymerization temperature |
| UV-vis | Ultraviolet-visible spectroscopy |
| Volume of NaOH consumed in the sample (Equation (2)) | |
| Volume of NaOH consumed in the blank (Equation (2)) | |
| Mass of the sample (Equation (2)) | |
| WAC | Water absorption capacity |
References
- Benaglia, M.; Alberti, A.; Giorgini, L.; Magnoni, F.; Tozzi, S. Poly(glycidyl methacrylate): A highly versatile polymeric building block for post-polymerization modifications. Polym. Chem. 2013, 4, 124–132. [Google Scholar] [CrossRef]
- Arslan, O.N.; Güntürkün, D.; Göksu, Y.A.; Altinbay, A.; Özgür Özer, H.; Nofar, M. Poly(glycidyl methacrylate) modified cellulose nanocrystals and their PBAT-based nanocomposites. Int. J. Biol. Macromol. 2023, 253, 126851. [Google Scholar] [CrossRef]
- Rehan, K.M.; Basha, K.A.; Safiullah, S.M. A Review on Engineering of Poly(Glycidyl Methacrylate) Microspheres. Polym. Sci. Ser. A 2024, 65, 593–615. [Google Scholar] [CrossRef]
- Shang, J.; Lin, S.; Theato, P. Fabrication of color changeable CO2 sensitive nanofibers. Polym. Chem. 2017, 8, 7446–7451. [Google Scholar] [CrossRef]
- Faria, M.; Vilela, C.; Mohammadkazemi, F.; Silvestre, A.J.D.; Freire, C.S.R.; Cordeiro, N. Poly(glycidyl methacrylate)/bacterial cellulose nano-composites: Preparation, characterization and post-modification. Int. J. Biol. Macromol. 2019, 127, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Vivaldo-Lima, E.; Wood, P.E.; Hamielec, A.E.; Penlidis, A. An Updated Review on Suspension Polymerization. Ind. Eng. Chem. Res. 1997, 36, 939–965. [Google Scholar] [CrossRef]
- Safiullah, S.M.; Wasi, K.A.; Basha, K.A. Preparation of poly(Glycidyl methacrylate)-copper nanocomposite by in-situ suspension polymerization—A novel synthetic method. Mater. Lett. 2014, 133, 60–63. [Google Scholar] [CrossRef]
- Avşar, A.; Gökbulut, Y.; Ay, B.; Serin, S. A novel catalyst system for the synthesis of N,N′-methylenebisacrylamide from acrylamide. Des. Monomers Polym. 2017, 20, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Qu, D.; Wang, S.; Qi, S.; Zuo, H. Structure, Property Optimization, and Adsorption Properties of N,N′-methylenebisacrylamide Cross-Linked Polyacrylic Acid Hydrogels under Different Curing Conditions. Polymers 2024, 16, 1990. [Google Scholar] [CrossRef] [PubMed]
- Pistel, K.-F.; Kissel, T. Effects of salt addition on the microencapsulation of proteins using W/O/W double emulsion technique. J. Microencapsul. 2000, 17, 467–483. [Google Scholar] [CrossRef]
- Shafiei, M.; Kazemzadeh, Y.; Martyushev, D.A.; Dai, Z.; Riazi, M. Effect of chemicals on the phase and viscosity behavior of water in oil emulsions. Sci. Rep. 2023, 13, 4100. [Google Scholar] [CrossRef]
- Zhao, Y.; Liang, T.; Miao, P.; Chen, T.; Han, X.; Hu, G.; Gao, J. Green Preparation of Aminated Magnetic PMMA Microspheres via EB Irradiation and Its Highly Efficient Uptake of Ce(III). Materials 2022, 15, 6553. [Google Scholar] [CrossRef]
- Barroca, L.R.; Velho, P.; Macedo, E.A. Removal of Acetaminophen (Paracetamol) from Water Using Aqueous Two-Phase Systems (ATPSs) Composed of Choline-Amino Acid Ionic Liquids. J. Chem. Eng. Data 2024, 69, 215–226. [Google Scholar] [CrossRef]
- Mpayipheli, N.; Mpupa, A.; Madala, N.E.; Nomngongo, P.N. Quantification of Acetaminophen, Ibuprofen, and β-Blockers in Wastewater and River Water Bodies During the COVID-19 Pandemic. Environments 2025, 12, 278. [Google Scholar] [CrossRef]
- Ramírez-Rodríguez, A.E.; Morales-Barrera, L.; Cristiani-Urbina, E. Continuous biosorption of acid red 27 azo dye by Eichhornia crassipes leaves in a packed-bed column. Sci. Rep. 2021, 11, 18413. [Google Scholar] [CrossRef]
- Lerma, T.A.; Chamorro, A.F.; Palencia, M.; Combatt, E.; Valle, H. Organoclay Microparticle-Enhanced Microfiltration for the Removal of Acid Red 27 in Aqueous Systems. Water 2025, 17, 2817. [Google Scholar] [CrossRef]
- Wu, H.; Zhou, J.; Zhang, S.; Niu, P.; Li, H.; Liu, Z.; Zhang, N.; Li, C.; Wang, L.; Wang, Y. A Comparative Study of Removal of Acid Red 27 by Adsorption on Four Different Chitosan Morphologies. Polymers 2024, 16, 1019. [Google Scholar] [CrossRef] [PubMed]
- Rivas, B.L.; Aguirre, M.d.C. Nitrite removal from water using water-soluble polymers in conjunction with liquid-phase polymer-based retention technique. React. Funct. Polym. 2007, 67, 1487–1494. [Google Scholar] [CrossRef]
- Hurtado, C.F.; Cancino-Madariaga, B.; Torrejón, C.; Villegas, P.P. Separation of nitrite and nitrate from water in aquaculture by nanofiltration membrane. Desalination Water Treat. 2016, 57, 26050–26062. [Google Scholar] [CrossRef]
- Suneetha, M.; Ravindhranath, K. Adsorption of Nitrite Ions from Wastewater Using Bio-sorbents Derived from Azadirachta indica Plant. Asian J. Water Environ. Pollut. 2017, 14, 71–79. [Google Scholar] [CrossRef]
- Wu, J.-L.; Liu, Z.-H.; Ma, Q.-G.; Dai, L.; Dang, Z. Occurrence, removal and risk evaluation of ibuprofen and acetaminophen in municipal wastewater treatment plants: A critical review. Sci. Total Environ. 2023, 891, 164600. [Google Scholar] [CrossRef]
- Matin, A.; Jillani, S.M.S.; Baig, U.; Ihsanullah, I.; Alhooshani, K. Removal of pharmaceutically active compounds from water sources using nanofiltration and reverse osmosis membranes: Comparison of removal efficiencies and in-depth analysis of rejection mechanisms. J. Environ. Manag. 2023, 338, 117682. [Google Scholar] [CrossRef] [PubMed]
- Lai, N.D.; Nguyen, D.T.C. Adsorptive removal of methyl red dye from the aqueous environment: A critical review. J. Ind. Eng. Chem. 2025; in press. [CrossRef]
- Domga, R.; Doumbi, R.T.; Kom, B.; Nen-Oure, B.S.; Motue Waffo, L.C.; Guillaume, E.N.; Bosco, T.J. Simultaneous removal of methyl orange and methyl red dyes by electrocoagulation process in aqueous medium using central composite design. Sustain. Chem. Environ. 2026, 13, 100298. [Google Scholar] [CrossRef]
- Rather, I.A.; Khan, S.A.; Ali, R.; Khan, T.A. One-Walled Phthalimide Extended Calix[4]Pyrrole-Based Supramolecular Adsorbent for Alleviating Nitrate From Simulated Water. ChemistrySelect 2025, 10, e202405302. [Google Scholar] [CrossRef]
- Yang, H.; Cheng, H. Controlling nitrite levels in drinking water by chlorination and chloramination. Sep. Purif. Technol. 2007, 56, 392–396. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, D.; Zhan, Y.; Chen, N.; Hu, W.; Wang, G.; Feng, C.; Tong, S. Electrostimulation enhances nitrate removal and mitigates nitrite accumulation from denitrification-induced alkalization. Environ. Res. 2026, 291, 123536. [Google Scholar] [CrossRef]
- Tan, X.; Nie, W.-B.; Lu, Y.; Wang, X.-W.; Dang, C.-C.; Wang, X.; Liu, L.-Y.; Ren, N.-Q.; Ni, B.-J.; Xie, G.-J. Anaerobic methane oxidation drives simultaneous nitrite and nitrous oxide removal. Bioresour. Technol. 2025, 423, 132247. [Google Scholar] [CrossRef]
- Yadav, P.; Saini, R.; Bhaduri, A. Facile synthesis of MgO nanoparticles for effective degradation of organic dyes. Environ. Sci. Pollut. Res. 2023, 30, 71439–71453. [Google Scholar] [CrossRef]
- Ruiz, Y. Síntesis de Nanopartículas de Carbonato de Calcio: Control de Tamaño y Fase Cristalina; Universidad de Valle: Cali, Colombia, 2015; Available online: https://hdl.handle.net/10893/15630 (accessed on 10 February 2026).
- Donia, A.M.; Atia, A.; El-Boraey, H.A.; Mabrouk, D.H. Adsorption of Ag(I) on Glycidyl Methacry-late/N,N′-Methylene Bis-Acrylamide Chelating Resins with Embedded Iron Oxide. Sep. Purif. Technol. 2006, 48, 281–287. [Google Scholar] [CrossRef]
- Costa, L.; Monteiro, R.; Castro, H.; Ribeiro, T.; Oliveira, A.; Torquato, E.; Arcanjo, M.; Marques, M. Glycidyl Methacrylate-Ethylene Glycol Dimethacrylate Copolymers with Varied Pore Structures Prepared with Different Reaction Parameters. Mater. Res. 2020, 23, e20190550. [Google Scholar] [CrossRef]
- Zhang, K.; Feng, W.; Jin, C. Protocol efficiently measuring the swelling rate of hydrogels. MethodsX 2020, 7, 100779. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Wang, Y.; Zhao, T.; Ye, Z.; Huang, H. Ultrasonication-assisted rapid determination of epoxide values in polymer mixtures containing epoxy resin. Anal. Methods 2014, 6, 4257–4261. [Google Scholar] [CrossRef]
- Lovell, P.A.; Schork, F.J. Fundamentals of Emulsion Polymerization. Biomacromolecules 2020, 21, 4396–4441. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Imhof, A. Surface morphology control of cross-linked polymer particles via dispersion polymerization. Soft Matter 2015, 11, 3589–3598. [Google Scholar] [CrossRef]
- Filova, B.; Musilova, L.; Mracek, A.; Ramos, M.L.; Veríssimo, L.M.P.; Valente, A.J.M.; Ribeiro, A.C.F. Effect of sodium salts on diffusion of poly(vinyl alcohol) in aqueous solutions. J. Mol. Liq. 2020, 304, 112728. [Google Scholar] [CrossRef]
- Hu, J.; Fu, M.; Li, M.; Luo, Y.; Ni, S.; Hou, L. Main Controlling Factors Affecting the Viscosity of Polymer Solution due to the Influence of Polymerized Cations in High-Salt Oilfield Wastewater. Processes 2024, 12, 791. [Google Scholar] [CrossRef]
- Bao, Y.; Huang, X.; Xu, D.; Xu, J.; Jiang, L.; Lu, Z.-Y.; Cui, S. Bound water governs the single-chain property of Poly(vinyl alcohol) in aqueous environments. Polymer 2022, 253, 124996. [Google Scholar] [CrossRef]
- Li, L.; Xu, X.; Liu, L.; Song, P.; Cao, Q.; Xu, Z.; Fang, Z.; Wang, H. Water governs the mechanical properties of poly(vinyl alcohol). Polymer 2021, 213, 123330. [Google Scholar] [CrossRef]
- Briscoe, B.; Luckham, P.; Zhu, S. The effects of hydrogen bonding upon the viscosity of aqueous poly(vinyl alcohol) solutions. Polymer 2000, 41, 3851–3860. [Google Scholar] [CrossRef]
- Yesayeu, M.A.; Kisialiova, H.A.; Shiman, D.I.; Kostjuk, S.V. Synthesis of polystyrene particles with controlled size via emulsion and dispersion radical polymerization: A practical guide. Colloids Surf. A Physicochem. Eng. Asp. 2025, 709, 136073. [Google Scholar] [CrossRef]
- Kurapati, R.; Natarajan, U. Factors Responsible for the Aggregation of Poly(vinyl alcohol) in Aqueous Solution as Revealed by Molecular Dynamics Simulations. Ind. Eng. Chem. Res. 2020, 59, 16099–16111. [Google Scholar] [CrossRef]
- Kang, B.; Tang, H.; Zhao, Z.; Song, S. Hofmeister Series: Insights of Ion Specificity from Amphiphilic Assembly and Interface Property. ACS Omega 2020, 5, 6229–6239. [Google Scholar] [CrossRef]
- Moghaddam, S.Z.; Thormann, E. The Hofmeister series: Specific ion effects in aqueous polymer solutions. J. Colloid Interface Sci. 2019, 555, 615–635. [Google Scholar] [CrossRef]
- Labidi, N.S.; Djebaili, A. Studies of The Mechanism of Polyvinyl Alcohol Adsorption on The Calcite/Water Interface in The Presence of Sodium Oleate. J. Miner. Mater. Charact. Eng. 2008, 7, 147–161. [Google Scholar] [CrossRef]
- Venugopal, G.; George, R.; Raghavan, N.; Srinivas, T.; Dakshinamurthy, A.; Paul, A.J.; Marahatta, A.B. Structural and Mechanical Properties of MgO-Poly(Vinyl Alcohol) Nanocomposite Film. Adv. Sci. Eng. Med. 2015, 7, 457–464. [Google Scholar] [CrossRef]
- Suhasini, M.R.; Rajeshwari, K.M.; Bindya, S.; Hemavathi, A.B.; Vishwanath, P.M.; Syed, A.; Eswaramoorthy, R.; Amachawadi, R.G.; Shivamallu, C.; Chattu, V.K.; et al. Pectin/PVA and pectin-MgO/PVA films: Preparation, characterization and biodegradation studies. Heliyon 2023, 9, e15792. [Google Scholar] [CrossRef]
- Sun, C.; Tang, T.; Uludag, H. A molecular dynamics simulation study on the effect of lipid substitution on polyethylenimine mediated siRNA complexation. Biomaterials 2013, 34, 2822–2833. [Google Scholar] [CrossRef]
- Skwarczynska-Wojsa, A.; Puszkarewicz, A. Removal of Acetaminophen from Aqueous Solutions in an Adsorption Process. Materials 2024, 17, 431. [Google Scholar] [CrossRef]
- Ismail, A.; Chou, S.-F. Polyethylenimine Carriers for Drug and Gene Delivery. Polymers 2025, 17, 2150. [Google Scholar] [CrossRef] [PubMed]
- De Britto, D.; Celi Goy, R.; Campana Filho, S.P.; Assis, O.B.G. Quaternary Salts of Chitosan: History, Anti-microbial Features, and Prospects. Int. J. Carbohyd. Chem. 2011, 2011, 312539. [Google Scholar] [CrossRef]
- Honciuc, A.; Solonaru, A.M.; Honciuc, M. Pickering Emulsion Polymerization Technology—Toward Nanostructured Materials for Applications in Metal Ion Extractions from Wastewaters. ACS Appl. Polym. Mater. 2023, 5, 8012–8022. [Google Scholar] [CrossRef]
- Subramani, S.E.; Thinakaran, N. Isotherm, kinetic and thermodynamic studies on the adsorption behaviour of textile dyes onto chitosan. Process Saf. Environ. Prot. 2017, 106, 1–10. [Google Scholar] [CrossRef]
- Altaie, O.T.S.; Zeidan, H.; Karakaya, N.; Karakaya, M.; Marti, M.E. Removal of Congo red from aqueous solutions by adsorption onto illite clay. Desalination Water Treat. 2023, 310, 226–237. [Google Scholar] [CrossRef]
- Tamer, T.M.; Khalifa, R.E.; Abou-Krisha, M.M.; Omer, A.M.; Alhamzani, A.G.; Youssef, M.E.; Yousef, T.A.; Salem, M.E.; Mohy-Eldin, M.S. Dichromate Contaminated Water Treatment using Novel Crystal Violet Azo Dye- Sulphonated Poly(Glycidyl methacrylate) Nano-Composite Adsorbent. Water Air Soil Pollut. 2025, 236, 35. [Google Scholar] [CrossRef]
- El-Aassar, M.R.; Tamer, T.M.; El-Sayed, M.Y.; Omer, A.M.; Althobaiti, I.O.; Youssef, M.E.; Alolaimi, R.F.; El-Agammy, E.F.; Alruwaili, M.S.; Rabhy, O.O.; et al. Development of Azo Dye Immobilized Sulfonated Poly (Glycidyl Methacrylate) Polymer Composite as Novel Adsorbents for Water Treatment Applications: Methylene Blue Immobilization Isotherm, Kinetic, Thermodynamic, and Simulations Studies. Molecules 2022, 27, 8418. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zheng, W. Polyethylenimine-functionalized polyacrylonitrile anion exchange fiber as a novel adsorbent for rapid removal of nitrate from wastewater. Chemosphere 2020, 258, 12737. [Google Scholar] [CrossRef]
- Suzaimi, N.D.; Goh, P.S.; Malek, N.A.N.N.; Lim, J.W.; Ismail, A.F. Performance of branched polyethyleneimine grafted porous rice husk silica in treating nitrate-rich wastewater via adsorption. J. Environ. Chem. Eng. 2019, 7, 103235. [Google Scholar] [CrossRef]
- Khademian, M.; Sarrafi, Y.; Tajbakhsh, M.; Hasantabar, V. Nitrate removal from aqueous solutions with sodium alginate/quaternary branched polyethylenimine/carboxylated multiwall carbon nanotube. Preprint 2023. [Google Scholar] [CrossRef]



















| Time (t) | Cf | R | MRC | qt |
|---|---|---|---|---|
| (h) | (mg L−1) | (%) | (mg g−1) | (mg g−1) |
| 0.5 | 0.61 | 53.25 | 0.138 | 0.14 |
| 1 | 0.56 | 57.11 | 0.148 | 0.15 |
| 2 | 0.05 | 96.36 | 0.250 | 0.25 |
| 3 | 0.04 | 96.92 | 0.251 | 0.25 |
| Model | Parameters | R2 | |
|---|---|---|---|
| Pseudo-first order | k1 (h−1) | 3.37 | 0.9031 |
| qe (mg g−1) | 1.16 | ||
| Pseudo-second order | qe | 0.29 | 0.9434 |
| k2 (g mg−1 h−1) | 4.27 | ||
| Intraparticle diffusion | kid mg g−1 h−1/2) | 0.1304 | 0.8790 |
| C (mg g−1) | 0.04 | ||
| Ci | MRC | R |
|---|---|---|
| (mg L−1) | (mg g−1) | (%) |
| 1.3 | 0.148 | 57.1 |
| 1.9 | 0.165 | 43.7 |
| 3.6 | 0.323 | 44.4 |
| 4.3 | 0.344 | 42.3 |
| pH | IS | MRC | R |
|---|---|---|---|
| (mmol L−1) | (mg g−1) | (%) | |
| 4 | 6.35 × 10−6 | 0.234 | 76.7 |
| 7 | 6.35 × 10−6 | 0.239 | 66.8 |
| 9 | 6.35 × 10−6 | 0.181 | 53.9 |
| 7 | 6.35 × 10−6 | 0.239 | 66.8 |
| 7 | 0.01 | 0.199 | 54.4 |
| 7 | 0.7 | 0.023 | 6.4 |
| pH | Amax | [AR-27] | R |
|---|---|---|---|
| (524 nm) | (mgL−1) | (%) | |
| S0 | 0.1561 | 3.34 | - |
| AD1 | 0.0818 | 1.75 | 47.6 |
| AD2 | 0.0997 | 2.13 | 36.2 |
| pH | MRC | R |
|---|---|---|
| (mg/g) | (%) | |
| 10 | 0.0063 ± 0.0005 | 26.25 ± 0.61 |
| 7 | 0.0156 ± 0.0002 | 70.83 ± 1.23 |
| 4 | 0.0230 ± 0.0001 | 100.00 ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Martínez, J.M.; Rincón, M.; Palencia, M. Effect of Inorganic Salts on Synthesis of Poly(glycidyl methacrylate) Microspheres, Their Functionalization with Poly(ethyleneimine) and Evaluation of Its Use for Removal of Acid Red 27, Acetaminophen and Nitrites. Polymers 2026, 18, 835. https://doi.org/10.3390/polym18070835
Martínez JM, Rincón M, Palencia M. Effect of Inorganic Salts on Synthesis of Poly(glycidyl methacrylate) Microspheres, Their Functionalization with Poly(ethyleneimine) and Evaluation of Its Use for Removal of Acid Red 27, Acetaminophen and Nitrites. Polymers. 2026; 18(7):835. https://doi.org/10.3390/polym18070835
Chicago/Turabian StyleMartínez, Jina M., Marisol Rincón, and Manuel Palencia. 2026. "Effect of Inorganic Salts on Synthesis of Poly(glycidyl methacrylate) Microspheres, Their Functionalization with Poly(ethyleneimine) and Evaluation of Its Use for Removal of Acid Red 27, Acetaminophen and Nitrites" Polymers 18, no. 7: 835. https://doi.org/10.3390/polym18070835
APA StyleMartínez, J. M., Rincón, M., & Palencia, M. (2026). Effect of Inorganic Salts on Synthesis of Poly(glycidyl methacrylate) Microspheres, Their Functionalization with Poly(ethyleneimine) and Evaluation of Its Use for Removal of Acid Red 27, Acetaminophen and Nitrites. Polymers, 18(7), 835. https://doi.org/10.3390/polym18070835







