Melamine-Functionalized Graphene Oxide as a Multifunctional Modifier for High-Performance Epoxy Nanocomposites with Enhanced Mechanical Properties and Thermal Stability
Abstract
1. Introduction
2. Materials and Methods
2.1. The Materials and Reagents
2.2. Obtaining Functionalized GO
2.3. Characterization of the GO
2.4. Preparation of Epoxy Composites
2.5. Composite Testing
3. Results and Discussion
3.1. Mechanical Properties of Epoxy Composite
3.2. Fractography Analysis of the Obtained Epoxy Composites
3.3. Analysis of the Curing Process of Epoxy Compositions
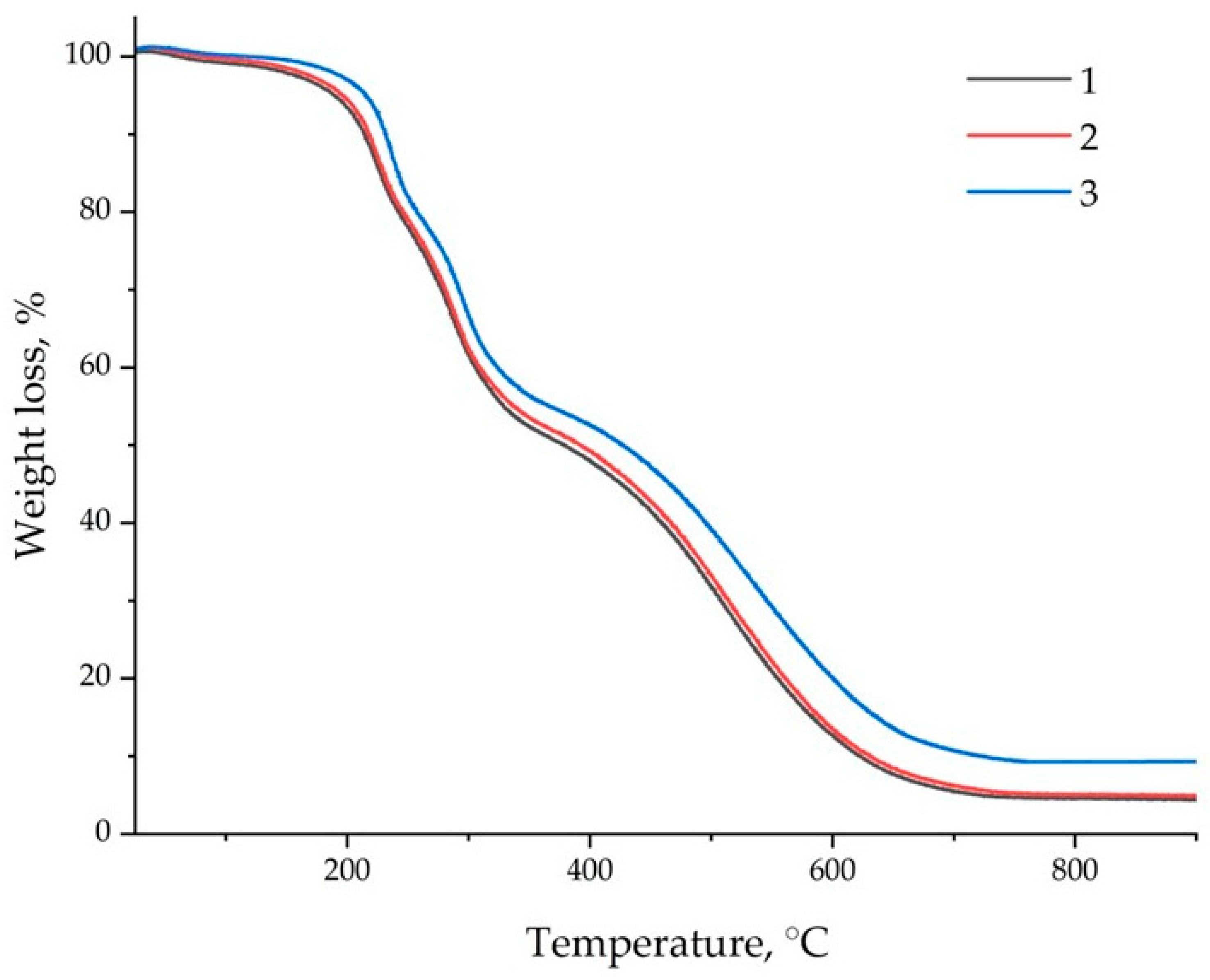
3.4. Analysis of TGA Data of Epoxy Composites
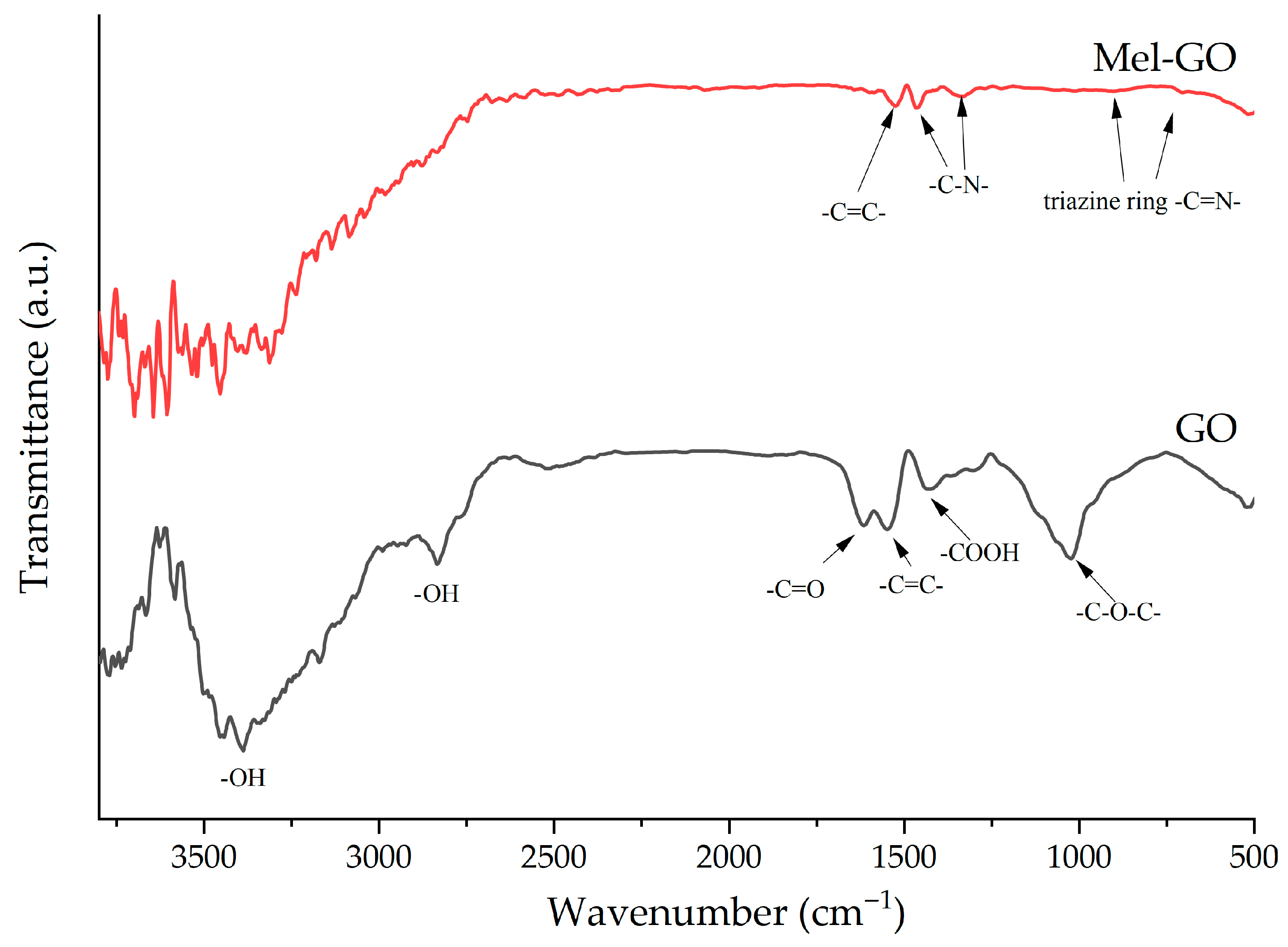
3.5. Structure and Chemistry of GO and Mel-GO
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fekiač, J.J.; Krbata, M.; Kohutiar, M.; Janík, R.; Kakošová, L.; Breznická, A.; Eckert, M.; Mikuš, P. Comprehensive Review: Optimization of Epoxy Composites, Mechanical Properties, & Technological Trends. Polymers 2025, 17, 271. [Google Scholar] [CrossRef]
- Osman, A.; Elhakeem, A.; Kaytbay, S.; Ahmed, A. A Comprehensive Review on the Thermal, Electrical, and Mechanical Properties of Graphene-Based Multi-Functional Epoxy Composites. Adv. Compos. Hybrid Mater. 2022, 5, 547–605. [Google Scholar] [CrossRef]
- Amberkar, T.; Mahanwar, P. Thermal Energy Management in Buildings and Constructions with Phase Change Material-Epoxy Composites: A Review. Energy Sources Part A Recovery Util. Environ. Eff. 2023, 45, 727–761. [Google Scholar] [CrossRef]
- Girish, S.; Vinay, M.S.; Bavan, D.S.; Ahmed, R.; Asdaque, P.M.G.B.; Revanasiddappa, M.; P., D.; Ravitej, Y.P. Evaluation of Thermal Properties of Epoxy Composites Filled by Nanofiller Materials. Discov. Appl. Sci. 2025, 7, 1269. [Google Scholar] [CrossRef]
- Zaghloul, M.M.Y.; Zaghloul, M.M.Y.; Fuseini, M. Recent Progress in Epoxy Nanocomposites: Corrosion, Structural, Flame Retardancy and Applications—A Comprehensive Review. Polym. Adv. Technol. 2023, 34, 3438–3472. [Google Scholar] [CrossRef]
- Mostovoi, A.S.; Yakovlev, E.A.; Burmistrov, I.N.; Panova, L.G. Use of Modified Nanoparticles of Potassium Polytitanate and Physical Methods of Modification of Epoxy Compositions for Improving Their Operational Properties. Russ. J. Appl. Chem. 2015, 88, 129–137. [Google Scholar] [CrossRef]
- Burmistrov, I.N.; Mostovoi, A.S.; Shatrova, N.V.; Panova, L.G.; Kuznetsov, D.V.; Gorokhovskii, A.V.; Il’inykh, I.A. Influence of Surface Modification of Potassium Polytitanates on the Mechanical Properties of Polymer Composites Thereof. Russ. J. Appl. Chem. 2013, 86, 765–771. [Google Scholar] [CrossRef]
- Martinez, P.; Nutt, S. Flax–Reinforced Vitrimer Epoxy Composites Produced via RTM. J. Compos. Sci. 2024, 8, 275. [Google Scholar] [CrossRef]
- Wang, J.; Khan, M.; Tiehu, L.; Javed, E.; Hussain, A.; Zada, A.; Wahab, Z.; Kashif, M.; Abid, S.U.; Raza, A.; et al. Preparation and Structural Investigations of the Graphite and Nanodiamonds Dispersed Epoxy Hybrid Composites for Enhanced Mechanical and Thermal Properties. Bull. Mater. Sci. 2022, 45, 160. [Google Scholar] [CrossRef]
- Yoo, S.C.; Kim, J.; Kim, S.; Lee, D. Enhanced Mechanical Properties of Melamine-Functionalized Boron Nitride Nanosheets Reinforced with Epoxy Nanocomposites for Dental Applications. J. Mater. Sci. 2022, 57, 18205–18219. [Google Scholar] [CrossRef]
- Matykiewicz, D. Hybrid Epoxy Composites with Both Powder and Fiber Filler: A Review of Mechanical and Thermomechanical Properties. Materials 2020, 13, 1802. [Google Scholar] [CrossRef]
- Varghese, A.S.; Sreekanth, M.S. Evaluation of Improved Tribo-Performance Characteristics of MoS2 Functionalized MWCNTs Hybrid Reinforced Epoxy Composites for Severe Erosion Conditions. Tribol. Int. 2024, 197, 109766. [Google Scholar] [CrossRef]
- Zhang, D.; Huang, Y.; Chia, L. Effects of Carbon Nanotube (CNT) Geometries on the Dispersion Characterizations and Adhesion Properties of CNT Reinforced Epoxy Composites. Compos. Struct. 2022, 296, 115942. [Google Scholar] [CrossRef]
- Mirzapour, M.; Cousin, P.; Robert, M.; Benmokrane, B. Dispersion Characteristics, the Mechanical, Thermal Stability, and Durability Properties of Epoxy Nanocomposites Reinforced with Carbon Nanotubes, Graphene, or Graphene Oxide. Polymers 2024, 16, 1836. [Google Scholar] [CrossRef]
- Naresh, K.; Khan, K.A.; Umer, R. Experimental Characterization and Modeling Multifunctional Properties of Epoxy/Graphene Oxide Nanocomposites. Polymers 2021, 13, 2831. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, L.; Lin, J.; Cui, P.; Li, M.; Du, X. Thermal Conductivity of Epoxy Composites Filled by Thermally Reduced Graphite Oxide with Different Reduction Degree. J. Compos. Mater. 2017, 51, 1743–1752. [Google Scholar] [CrossRef]
- Monteserín, C.; Blanco, M.; Aranzabe, E.; Aranzabe, A.; Laza, J.M.; Larrañaga-Varga, A.; Vilas, J.L. Effects of Graphene Oxide and Chemically-Reduced Graphene Oxide on the Dynamic Mechanical Properties of Epoxy Amine Composites. Polymers 2017, 9, 449. [Google Scholar] [CrossRef] [PubMed]
- Bekeshev, A.; Mostovoy, A.; Shcherbakov, A.; Tastanova, L.; Akhmetova, M.; Apendina, A.; Orynbassar, R.; Lopukhova, M. The Influence of Pristine and Aminoacetic Acid-Treated Aluminum Nitride on the Structure, Curing Processes, and Properties of Epoxy Nanocomposites. J. Compos. Sci. 2023, 7, 482. [Google Scholar] [CrossRef]
- Singh, B.; Mohanty, A. Nano-Mechanical Approach to Study the Behavior of Annealed Nanodiamond Reinforced Epoxy Nanocomposites. Polym. Compos. 2023, 44, 2997–3006. [Google Scholar] [CrossRef]
- Wei, Y.; Hu, X.; Jiang, Q.; Sun, Z.; Wang, P.; Qiu, Y.; Liu, W. Influence of Graphene Oxide with Different Oxidation Levels on the Properties of Epoxy Composites. Compos. Sci. Technol. 2018, 161, 74–84. [Google Scholar] [CrossRef]
- Zhao, H.; Ding, J.; Yu, H. Variation of Mechanical and Thermal Properties in Sustainable Graphene Oxide/Epoxy Composites. Sci. Rep. 2018, 8, 16560. [Google Scholar] [CrossRef]
- Rhili, K.; Chergui, S.; ElDouhaibi, A.S.; Siaj, M. Hexachlorocyclotriphosphazene Functionalized Graphene Oxide as a Highly Efficient Flame Retardant. ACS Omega 2021, 6, 6252–6260. [Google Scholar] [CrossRef]
- Wu, H.; Liu, C.; Cheng, L.; Yu, Y.; Zhao, H.; Wang, L. Enhancing the Mechanical and Tribological Properties of Epoxy Composites via Incorporation of Reactive Bio-Based Epoxy Functionalized Graphene Oxide. RSC Adv. 2020, 10, 40148–40156. [Google Scholar] [CrossRef]
- Li, Y.; Yang, H.; Wang, F.; Huang, Y. Superior Anticorrosion Performance of Epoxy-Based Composites with Well-Dispersed Melamine Modified Graphene Oxide. J. Appl. Polym. Sci. 2021, 138, 49866. [Google Scholar] [CrossRef]
- Qin, J.; Zhang, W.; Yang, R. Direct Diazotization of Graphite Nanoplatelets with Melamine and Their Favorable Application in Epoxy Resins. Polym. Adv. Technol. 2020, 31, 1300–1311. [Google Scholar] [CrossRef]
- Xue, B.; Guo, Y.; Li, R.; Zhou, Q.; Cao, J.; Xie, G.; Wang, Z.; Gu, S.; Yuan, R. Enhanced Thermal and Mechanical Performance of Epoxy Composites via Three-Dimensional Boron Nitride Nanostructures. J. Mater. Res. Technol. 2025, 39, 5822–5831. [Google Scholar] [CrossRef]
- Bifulco, A.; Parida, D.; Salmeia, K.A.; Nazir, R.; Lehner, S.; Stämpfli, R.; Markus, H.; Malucelli, G.; Branda, F.; Gaan, S. Fire and Mechanical Properties of DGEBA-Based Epoxy Resin Cured with a Cycloaliphatic Hardener: Combined Action of Silica, Melamine and DOPO-Derivative. Mater. Des. 2020, 193, 108862. [Google Scholar] [CrossRef]
- Yakovlev, A.V.; Yakovleva, E.V.; Tseluikin, V.N.; Krasnov, V.V.; Mostovoy, A.S.; Rakhmetulina, L.A.; Frolov, I.N. Electrochemical Synthesis of Multilayer Graphene Oxide by Anodic Oxidation of Disperse Graphite. Russ. J. Electrochem. 2019, 55, 1196–1202. [Google Scholar] [CrossRef]
- Li, Y.; Xu, H.; Zhang, Y.; Dong, L.; Liu, C.; Xie, H.; Yu, W. Paving 3D Interconnected Cring-C3N4@rGO Skeleton for Polymer Composites with Efficient Thermal Management Performance yet High Electrical Insulation. Int. Commun. Heat Mass Transf. 2022, 135, 106147. [Google Scholar] [CrossRef]
- Politano, G.G.; Versace, C. Recent Advances in the Raman Investigation of Structural and Optical Properties of Graphene and Other Two-Dimensional Materials. Crystals 2023, 13, 1357. [Google Scholar] [CrossRef]
- Mathioudakis, G.N.; Visvini, G.A.; Sygellou, L.; Soto Beobide, A.; Voyiatzis, G.A. Comparative In-Depth Investigation of Benchmark Graphene Oxides in the Perspective of Their Integration into Industrial Production Processes. Nanomaterials 2025, 15, 980. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Ibrahim, S.; Chakraborty, K.; Ghosh, S.; Pal, T. Stepwise Reduction of Graphene Oxide and Studies on Defect-Controlled Physical Properties. Sci. Rep. 2024, 14, 294. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-H.; De Silva, K.K.H.; Kumara, G.R.A.; Yoshimura, M. Structural Evolution of Hydrothermally Derived Reduced Graphene Oxide. Sci. Rep. 2018, 8, 6849. [Google Scholar] [CrossRef] [PubMed]
- Mousavi-Zadeh, S.H.; Poursalehi, R.; Yourdkhani, A. Photocatalytic Activity of Self-Heterojunctioned Intermediate Phases in HCl Protonated and HNO3 Deconjugated g-C3N4 Nanostructures. Heliyon 2024, 10, e38025. [Google Scholar] [CrossRef]
- Wan, W.; Sun, J.-Y.; Ye, S.; Zhang, Q. Confining the Polymerization Degree of Graphitic Carbon Nitride in Porous Zeolite-Y and Its Luminescence. RSC Adv. 2018, 8, 25057–25064. [Google Scholar] [CrossRef]
- Bekeshev, A.; Mostovoy, A.; Shcherbakov, A.; Zhumabekova, A.; Serikbayeva, G.; Vikulova, M.; Svitkina, V. Effect of Phosphorus and Chlorine Containing Plasticizers on the Physicochemical and Mechanical Properties of Epoxy Composites. J. Compos. Sci. 2023, 7, 178. [Google Scholar] [CrossRef]
- Mostovoy, A.S.; Yakovlev, A.V. Effect of Additions of Electrochemically Oxidized Graphite on the Physicochemical and Mechanical Properties of Modified Epoxy Composites. Russ. J. Appl. Chem. 2019, 92, 1439–1446. [Google Scholar] [CrossRef]
- Parvizi, P.; Jalilian, M.; Dearn, K.D. Epoxy Composites Reinforced with Nanomaterials and Fibres: Manufacturing, Properties, and Applications. Polym. Test. 2025, 146, 108761. [Google Scholar] [CrossRef]
- ISO 527-1:2019; Plastics—Determination of Tensile Properties—Part 1: General Principles. ISO: Geneva, Switzerland, 2019.
- ISO 178:2019; International Organization for Standardization—Plastics—Determination of Flexural Properties. ISO: Geneva, Switzerland, 2019.
- ISO 179-1:2010; Plastics—Determination of Charpy Impact Properties—Part 1: Non-Instrumented Impact Test. ISO: Geneva, Switzerland, 2010.
- ASTM D2240; Standard Test Method for Rubber Property—Durometer Hardness. ASTM: Philadelphia, PA, USA, 2021.
- Saeb, M.R.; Bakhshandeh, E.; Khonakdar, H.A.; Mäder, E.; Scheffler, C.; Heinrich, G. Cure Kinetics of Epoxy Nanocomposites Affected by MWCNTs Functionalization: A Review. Sci. World J. 2013, 2013, 703708. [Google Scholar] [CrossRef]
- Ipate, A.-M.; Serbezeanu, D.; Iftimie, I.-A.; Lisa, G.; Rîmbu, C.-M.; Vlad-Bubulac, T. Study on the Structure, Thermal Properties and Antibacterial Properties of Phosphorus-Modified PVA/TiO2 Composite Films. Gels 2025, 11, 1020. [Google Scholar] [CrossRef]
- Carrasco, F.; Pagès, P. Thermal Degradation and Stability of Epoxy Nanocomposites: Influence of Montmorillonite Content and Cure Temperature. Polym. Degrad. Stab. 2008, 93, 1000–1007. [Google Scholar] [CrossRef]
- Ackermann, A.C.; Fischer, M.; Wick, A.; Carosella, S.; Fox, B.L.; Middendorf, P. Mechanical, Thermal and Electrical Properties of Epoxy Nanocomposites with Amine-Functionalized Reduced Graphene Oxide via Plasma Treatment. J. Compos. Sci. 2022, 6, 153. [Google Scholar] [CrossRef]
- Uthaman, A.; Lal, H.M.; Li, C.; Xian, G.; Thomas, S. Mechanical and Water Uptake Properties of Epoxy Nanocomposites with Surfactant-Modified Functionalized Multiwalled Carbon Nanotubes. Nanomaterials 2021, 11, 1234. [Google Scholar] [CrossRef]
- Halim, W.Z.A.W.A.; Junid, R.; Sazali, N.; Siregar, J.P.; Razak, J.A.; Fitriyana, D.F.; Cionita, T.; Jaafar, J. Enhancement of Flexural Modulus and Strength of Epoxy Nanocomposites with the Inclusion of Functionalized GNPs Using Tween 80. J. Eng. Appl. Sci. 2024, 71, 69. [Google Scholar] [CrossRef]
- Gharieh, A.; Seyed Dorraji, M.S. A Systematic Study on the Synergistic Effects of MWCNTs and Core–Shell Particles on the Physicomechanical Properties of Epoxy Resin. Sci. Rep. 2021, 11, 20789. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yang, S.; Liu, H.; Shao, Q.; Chen, Q.; Lu, C.; Jiang, Y.; Liu, C.; Guo, Z. Reinforced Carbon Fiber Laminates with Oriented Carbon Nanotube Epoxy Nanocomposites: Magnetic Field Assisted Alignment and Cryogenic Temperature Mechanical Properties. J. Colloid Interface Sci. 2018, 517, 40–51. [Google Scholar] [CrossRef]
- Mali, A.; Agbo, P.; Mantripragada, S.; Zhang, L. Surface-Modified Electrospun Glass Nanofibers from Silane Treatment and Their Use for High-Performance Epoxy-Based Nanocomposite Materials. Materials 2023, 16, 6817. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Zeng, K.Y.; He, C.B. Morphology, Tensile and Fracture Characteristics of Epoxy-Alumina Nanocomposites. Mater. Sci. Eng. A 2010, 527, 5670–5676. [Google Scholar] [CrossRef]
- Dyachkova, T.P.; Khan, Y.A.; Burakova, E.A.; Galunin, E.V.; Shigabaeva, G.N.; Stolbov, D.N.; Titov, G.A.; Chapaksov, N.A.; Tkachev, A.G. Characteristics of Epoxy Composites Containing Carbon Nanotubes/Graphene Mixtures. Polymers 2023, 15, 1476. [Google Scholar] [CrossRef]
- Maras, M.M.; Kantarci, F. Structural Behavior of RC Beams Strengthened Using Fiber-Reinforced Polymer U-Jackets. Struct. Concr. 2023, 24, 2384–2401. [Google Scholar] [CrossRef]
- Ozdemir, M.F.; Maras, M.M.; Yurtseven, H.B. Flexural Behavior of Laminated Wood Beams Strengthened with Novel Hybrid Composite Systems: An Experimental Study. J. Korean Wood Sci. Technol. 2023, 51, 526–541. [Google Scholar] [CrossRef]
- Ilyin, S.O.; Kotomin, S.V. Effect of Nanoparticles and Their Anisometry on Adhesion and Strength in Hybrid Carbon-Fiber-Reinforced Epoxy Nanocomposites. J. Compos. Sci. 2023, 7, 147. [Google Scholar] [CrossRef]
- Lyn Hor, J.; Wang, H.; Fakhraai, Z.; Lee, D. Effects of Polymer–Nanoparticle Interactions on the Viscosity of Unentangled Polymers under Extreme Nanoconfinement during Capillary Rise Infiltration. Soft Matter 2018, 14, 2438–2446. [Google Scholar] [CrossRef]
- Bandeira de Souza, Z.S.; Araújo do Nascimento, P.L.; Samara, M.; David, É.; Macedo Fechine, G.J.; Alves da Motta Sobrinho, M.; Demarquette, N.R. Influence of Graphene Functionalization on the Curing Kinetics, Dynamical Mechanical Properties and Morphology of Epoxy Nanocomposites. Polymer 2025, 320, 128067. [Google Scholar] [CrossRef]
- Lomakin, S.; Koverzanova, E.; Usachev, S.; Shilkina, N.; Khvatov, A.; Erina, N.; Rogovina, S.; Kuznetsova, O.; Siracusa, V.; Berlin, A.; et al. Specific Impact of the Layered Nanomodifiers—Graphene Nanoplates, and Na+ Montmorillonite on Thermal Degradation of Polylactic Acid: Mechanism and Kinetics. Polymers 2026, 18, 347. [Google Scholar] [CrossRef]
- Corcione, C.E.; Frigione, M. Characterization of Nanocomposites by Thermal Analysis. Materials 2012, 5, 2960–2980. [Google Scholar] [CrossRef]
- Bakry, A.M.; Awad, F.S.; Bobb, J.A.; Ibrahim, A.A.; El-Shall, M.S. Melamine-Based Functionalized Graphene Oxide and Zirconium Phosphate for High Performance Removal of Mercury and Lead Ions from Water. RSC Adv. 2020, 10, 37883–37897. [Google Scholar] [CrossRef] [PubMed]
- Arabi Shamsabadi, A.; Rad, V.; Soroush, M. Graphene Oxide/Melamine/Ionic Liquid Membranes for Selective CO2 Separation. Nano Trends 2024, 6, 100033. [Google Scholar] [CrossRef]
- Meng, Q.; Zhu, Y.; Li, S.; Zhao, K.; Araby, S.; Han, S. Aramid Nanofiber-Reinforced Graphene Oxide Frameworks for Durable and Flame-Resistant Fire Warning Sensors. J. Mater. Res. Technol. 2025, 35, 4175–4188. [Google Scholar] [CrossRef]









| Composition | τgel, min | τres, min | Tmax, °C |
|---|---|---|---|
| Pristine epoxy composition without GO | 104 | 146 | 88 |
| Epoxy composition containing GO | 146 | 195 | 94 |
| Epoxy composition containing mel-GO | 48 | 74 | 122 |
| Composition | Tstart, °C | Tend, °C | Tmax, °C | H, J/g |
|---|---|---|---|---|
| Pristine epoxy composition without GO | 59 | 159 | 108 | 536 |
| Epoxy composition containing GO | 71 | 150 | 108 | 446 |
| Epoxy composition containing mel-GO | 52 | 163 | 105 | 507 |
| Samples | T5%, °C | T10%, °C | T30%, °C | T50%, °C | T60%, °C | T80%, °C | Residues at 900 °C, wt.% | THRI | Tv |
|---|---|---|---|---|---|---|---|---|---|
| EP | 190 | 214 | 279 | 385 | 460 | 558 | 4.45 | 119.3 | 114 |
| EP/GO | 195 | 217 | 281 | 392 | 467 | 562 | 4.85 | 120.8 | 142 |
| EP/GO-melamin | 217 | 232 | 294 | 430 | 498 | 604 | 9.29 | 128.9 | 175 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mostovoy, A.; Shcherbakov, A.; Bekeshev, A.; Brudnik, S.; Yakovlev, A.; Zhumabekova, A.; Yakovleva, E.; Ussenkulova, S.; Rastegaev, O.; Lopukhova, M. Melamine-Functionalized Graphene Oxide as a Multifunctional Modifier for High-Performance Epoxy Nanocomposites with Enhanced Mechanical Properties and Thermal Stability. Polymers 2026, 18, 657. https://doi.org/10.3390/polym18050657
Mostovoy A, Shcherbakov A, Bekeshev A, Brudnik S, Yakovlev A, Zhumabekova A, Yakovleva E, Ussenkulova S, Rastegaev O, Lopukhova M. Melamine-Functionalized Graphene Oxide as a Multifunctional Modifier for High-Performance Epoxy Nanocomposites with Enhanced Mechanical Properties and Thermal Stability. Polymers. 2026; 18(5):657. https://doi.org/10.3390/polym18050657
Chicago/Turabian StyleMostovoy, Anton, Andrey Shcherbakov, Amirbek Bekeshev, Sergey Brudnik, Andrey Yakovlev, Arai Zhumabekova, Elena Yakovleva, Sholpan Ussenkulova, Oleg Rastegaev, and Marina Lopukhova. 2026. "Melamine-Functionalized Graphene Oxide as a Multifunctional Modifier for High-Performance Epoxy Nanocomposites with Enhanced Mechanical Properties and Thermal Stability" Polymers 18, no. 5: 657. https://doi.org/10.3390/polym18050657
APA StyleMostovoy, A., Shcherbakov, A., Bekeshev, A., Brudnik, S., Yakovlev, A., Zhumabekova, A., Yakovleva, E., Ussenkulova, S., Rastegaev, O., & Lopukhova, M. (2026). Melamine-Functionalized Graphene Oxide as a Multifunctional Modifier for High-Performance Epoxy Nanocomposites with Enhanced Mechanical Properties and Thermal Stability. Polymers, 18(5), 657. https://doi.org/10.3390/polym18050657








