From Design to Application: Advanced Cellulose Scaffolds for Engineered Tissue Regeneration
Abstract
1. Introduction
2. Material Sources and Engineering Strategies
2.1. Cellulose Sources for Tissue Engineering: From Macro to Nano
2.2. Key Design Strategies for Functional Enhancement
2.3. Fabrication Techniques for Structural Control
3. Applications in Tissue Regeneration
3.1. Hard Tissues: Bone and Osteochondral Repair
3.2. Soft Tissues: Skin Wound Healing and Nerve Regeneration
3.3. Specialized Tissues: Cartilage, Ligament, and Cardiovascular Applications
4. Perspective
4.1. Performance Optimization: Matching Material to Biology
4.2. Product Manufacturing: From Scalability to Personalization
4.3. Intelligent Responsive Design and Translation
5. Conclusions
6. Methodology for Literature Search and Selection
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ghilan, A.; Nicu, R.; Ciolacu, D.E.; Ciolacu, F. Insight into the latest medical applications of nanocellulose. Materials 2023, 16, 4447. [Google Scholar] [CrossRef] [PubMed]
- Dar, M.A.; Xie, R.R.; Liu, J.; Ali, S.; Pawar, K.D.; Sudiana, I.M.; Sun, J.Z. Current paradigms and future challenges in harnessing nanocellulose for advanced applications in tissue engineering: A critical state-of-the-art review for biomedicine. Int. J. Mol. Sci. 2025, 26, 1449. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Ao, R.; Xu, M.; Jin, M.; Han, M.; Wang, Z.; Dang, W.; Wu, H.; Lin, W.; Zhen, Y.; et al. Bifunctional adECM bioscaffold with STIM1-ASCs and IGF-2 promotes functional masseter VML repair via myogenesis and fibrosis suppression. Bioact. Mater. 2025, 54, 466–491. [Google Scholar] [CrossRef] [PubMed]
- Guan, Q.; Hou, S.; Wang, K.; Li, L.; Cheng, Y.; Zheng, M.; Liu, C.; Zhao, X.; Zhou, J.; Li, P.; et al. Micropore structure engineering of injectable granular hydrogels via controlled liquid-liquid phase separation facilitates regenerative wound healing in mice and pigs. Biomaterials 2025, 318, 123192. [Google Scholar] [CrossRef]
- Hou, R.Q.; Xie, Y.Y.; Song, R.; Bao, J.K.; Shi, Z.Q.; Xiong, C.X.; Yang, Q.L. Nanocellulose/Polypyrrole hydrogel scaffolds with mechanical strength and electrical activity matching native cardiac tissue for myocardial tissue engineering. Cellulose 2024, 31, 4523–4532. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Z.; Chen, X.; Gao, Y.; Wang, W.; Zhuang, X.; Zhang, H.; Dong, X. Bone tissue engineering scaffold materials: Fundamentals, advances, and challenges. Chin. Chem. Lett. 2024, 35, 109197. [Google Scholar] [CrossRef]
- Ao, C.; Niu, Y.; Zhang, X.; He, X.; Zhang, W.; Lu, C. Fabrication and characterization of electrospun cellulose/nano-hydroxyapatite nanofibers for bone tissue engineering. Int. J. Biol. Macromol. 2017, 97, 568–573. [Google Scholar] [CrossRef]
- Dsouza, M.V.; Dodamani, S.; Kurangi, B.; Kumbar, V.; Hussain, M. Functionalized nanoscaffolds for drug delivery using biomaterials: A glimmer of hope in diabetic wound management. Regen. Eng. Transl. Med. 2025. [Google Scholar] [CrossRef]
- Iwasaki, A.; Hatakeyama, M.; Liu, Q.; Orimoto, A.; Fukuda, T.; Kitaoka, T. Proliferation and differentiation of human dental pulp stem cells on phosphorylated cellulose nanofiber scaffolds. Carbohydr. Polym. 2025, 359, 123593. [Google Scholar] [CrossRef]
- Won, T.; Goh, M.; Lim, C.; Moon, J.; Lee, K.; Park, J.; Chung, K.; Kim, Y.; Lee, S.; Hong, H.; et al. Recent progress in cellulose nanofibril hydrogels for biomedical applications. Polymers 2025, 17, 2272. [Google Scholar] [CrossRef]
- Usala, E.; Gonzalez, Z.; Campillo, N.; Baena, J.; Rincón, E.; Ferrari, B.; Rodríguez, A.; Espinosa, E. Development of 3D printable conductive cellulose-based hydrogel with incorporation of rGO for neural tissue engineering. J. Colloid Interface Sci. 2026, 703, 139285. [Google Scholar] [CrossRef]
- Radakisnin, R.; Majid, M.S.A.; Jamir, M.R.M.; Tahir, M.F.M.; Meng, C.E.; Al Alshahrani, H. Physical, thermal, and mechanical properties of highly porous polylactic acid/cellulose nanofibre scaffolds prepared by salt leaching technique. Nanotechnol. Rev. 2021, 10, 1469–1483. [Google Scholar] [CrossRef]
- Lv, Q.; Tian, Z.; Li, W.; Duan, G.; Han, X.; Zhang, C.; He, S.; Mao, H.; Ma, C.; Jiang, S. Porous carbon derived from biomass-based polymers: Innovative applications in supercapacitors. Chin. Chem. Lett. 2025, 37, 110860. [Google Scholar] [CrossRef]
- Jabbari, F.; Babaeipour, V.; Bakhtiari, S. Bacterial cellulose-based composites for nerve tissue engineering. Int. J. Biol. Macromol. 2022, 217, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Boyetey, M.J.B.; Torgbo, S.; Sukyai, P. Bio-scaffold for bone tissue engineering with focus on bacterial cellulose, biological materials for hydroxyapatite synthesis and growth factors. Eur. Polym. J. 2023, 194, 112168. [Google Scholar] [CrossRef]
- Nishiguchi, A.; Taguchi, T.; Thixotropic, A. Cell-infiltrative nanocellulose hydrogel that promotes in vivo tissue remodeling. ACS Biomater. Sci. Eng. 2020, 6, 946–958. [Google Scholar] [CrossRef]
- Mohammadi, S.; Ghasemi, F.; Alavi, G.S.A.; Alemzadeh, E. Investigate the in vitro biocompatibility, biodegradation, cytotoxicity, and differentiation potential of 3-D gelatin-nanocellulose composite scaffolds loaded with nanohydroxyapatite and simvastatin. Tissue Cell 2024, 91, 102536. [Google Scholar] [CrossRef]
- Karmati, S.K.; Janmohammadi, M.; Nazemi, Z.; Nourbakhsh, M.S.; Bahraminasab, M. Cellulose nanocrystal-reinforced bioactive glass-tragacanth gum nanocomposite scaffold for bone tissue regeneration: A multi-objective optimization. Mater. Chem. Phys. 2026, 347, 131478. [Google Scholar] [CrossRef]
- Banaeyan, R.; Nourany, M.; Hosseini, S.; Galefi, A.; Alipour, A.; Jahanfar, M.; Wang, P.Y.; Homaeigohar, S.; Shahsavarani, H. Polydopamine-based surface functionalization of watermelon rind as a 3D nanofibrous cellulose scaffold for osteogenesis. Cellulose 2023, 31, 443–461. [Google Scholar] [CrossRef]
- Pang, X.N.; Zhang, T.L.; Li, J.Z.; Yu, L.Q.; Liu, Z.B.; Liu, Y.C.; Li, L.; Cheng, L.M.; Zhu, R.R. LDH nanoparticles-doped cellulose nanofiber scaffolds with aligned microchannels direct high-efficiency neural regeneration and organized neural circuit remodeling through RhoA/Rock/Myosin II pathway. Biomaterials 2025, 314, 122873. [Google Scholar] [CrossRef]
- Wu, Y.D.; Wang, Y.F.; Wang, F.; He, J.M.; Huang, Y.D. Preparation and properties study of polylactic acid/bacterial cellulose composite scaffolds by solvent removal. J. Mater. Res. 2022, 37, 1602–1611. [Google Scholar] [CrossRef]
- Liu, G.; Ma, M.; Meng, H.; Liu, J.; Zheng, Y.; Peng, J.; Wei, S.; Sun, Y.; Wang, Y.; Xie, Y.; et al. In-situ self-assembly of bacterial cellulose/poly(3,4-ethylenedioxythiophene)-sulfonated nanofibers for peripheral nerve repair. Carbohydr. Polym. 2022, 281, 119044. [Google Scholar] [CrossRef] [PubMed]
- Hickey, R.J.; Modulevsky, D.J.; Cuerrier, C.M.; Pelling, A.E. Customizing the shape and microenvironment biochemistry of biocompatible macroscopic plant-derived cellulose scaffolds. ACS Biomater. Sci. Eng. 2018, 4, 3726–3736. [Google Scholar] [CrossRef] [PubMed]
- Tamo, A.K. Nanocellulose-based hydrogels as versatile materials with interesting functional properties for tissue engineering applications. J. Mater. Chem. B 2024, 12, 7692–7759. [Google Scholar] [CrossRef]
- Liang, Q.; Chen, S.; Hua, S.; Jiang, W.; Zhan, J.; Pu, C.; Lin, R.; He, Y.; Hou, H.; Qiu, X. Biomimetic versatile anisotropic, electroactive cellulose hydrogel scaffolds tailored from fern stem serving as nerve conduit and cardiac patch. Adv. Sci. 2024, 12, e2400002. [Google Scholar] [CrossRef]
- Nagpal, L.; Arora, S. Engineering cellulose for regenerative medicine: Bacterial cellulose and beyond. Regen. Eng. Transl. Med. 2025, 1, 1–18. [Google Scholar] [CrossRef]
- Seddiqi, H.; Oliaei, E.; Honarkar, H.; Jin, J.; Geonzon, L.C.; Bacabac, R.G.; Klein-Nulend, J. Cellulose and its derivatives: Towards biomedical applications. Cellulose 2021, 28, 1893–1931. [Google Scholar] [CrossRef]
- Kitaoka, T. Nanocellulose targets regenerative medicine. Bioresources 2025, 20, 2464–2467. [Google Scholar] [CrossRef]
- Takayama, G.; Kondo, T. Quantitative evaluation of fiber network structure-property relationships in bacterial cellulose hydrogels. Carbohydr. Polym. 2023, 321, 121311. [Google Scholar] [CrossRef]
- Ul-Islam, M.; Khan, S.; Ullah, M.W.; Park, J.K. Comparative study of plant and bacterial cellulose pellicles regenerated from dissolved states. Int. J. Biol. Macromol. 2019, 137, 247–252. [Google Scholar] [CrossRef]
- Rostamabadi, H.; Bist, Y.; Kumar, Y.; Yildirim-Yalcin, M.; Ceyhan, T.; Falsafi, S.R. Cellulose nanofibers, nanocrystals, and bacterial nanocellulose: Fabrication, characterization, and their most recent applications. Future Postharvest Food 2024, 1, 5–33. [Google Scholar] [CrossRef]
- Rebello, S.; Deepak, A.; Chandrababu, K.; Aneesh, E.M.; Chakrapani, P.S.B.; Jisha, M.S. Microbial consortia-derived cellulose biomaterial: Synthesis, characterization, and utility in neural tissue regeneration. Int. J. Biol. Macromol. 2025, 288, 138719. [Google Scholar] [CrossRef] [PubMed]
- Kamal, T.; Ul-Islam, M.; Khan, S.B.; Bakhsh, E.M.; Chani, M.T.S. Preparation, Characterization, and biological features of cactus coated bacterial cellulose hydrogels. Gels 2022, 8, 88. [Google Scholar] [CrossRef] [PubMed]
- Błażyńska-Spychalska, A.; Kur, M.; Brzeski, T.; Zając, W.; Pankiewicz, T.; Bielecki, S.; Woliński, J.; Jankau, J. Potential of bacterial cellulose in reconstructive surgery of body integumentary system: Preliminary studies in animals. J. Funct. Biomater. 2023, 14, 397. [Google Scholar] [CrossRef] [PubMed]
- Mihhels, K.; Yousefi, N.; Blomster, J.; Solala, I.; Solhi, L.; Kontturi, E. Assessment of the alga cladophora glomerata as a source for cellulose nanocrystals. Biomacromolecules 2023, 24, 4672–4679. [Google Scholar] [CrossRef]
- Broaders, K.E.; Kuehne, E.; Bowden, A.C.; Fraser, M.R. Ketal-modified cellulose as a biodegradable bioplastic. ACS Omega 2025, 10, 56025–56031. [Google Scholar] [CrossRef]
- Xu, J.; Liu, X.; Zhang, Q. The biosynthesis of bacterial cellulose composites accompanied by spray feeding of biomasses. Polymers 2024, 16, 2541. [Google Scholar] [CrossRef]
- Liu, X.K.; Hu, H.R.; Ma, J.H.; Wang, B.X. Mineralized cellulose nanofibers reinforced bioactive hydrogel remodels the osteogenic and angiogenic microenvironment for enhancing bone regeneration. Carbohydr. Polym. 2025, 357, 123480. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Liu, Z.; Shao, X.; Lin, Y.; Song, W.; Xu, D.; Gao, Y.; Han, J. Modification methods’ effects on the characteristics of carboxylated cellulose fibers: Carboxyl group introduction method versus physical properties. Bioresources 2024, 19, 1590–1601. [Google Scholar] [CrossRef]
- Yoshikawa, Y.; Yamato, K.; Ishida, A.; Yoshida, Y.; Kumamoto, Y.; Isogai, A. Amidation of carboxy groups in TEMPO-oxidized cellulose for improving surface hydrophobization and thermal stability of TEMPO-CNCs. Carbohydr. Polym. 2025, 347, 122654. [Google Scholar] [CrossRef]
- Elf, P.; Larsson, P.A.; Larsson, A.; Wågberg, L.; Hedenqvist, M.S.; Nilsson, F. Effects of ring opening and chemical modification on the properties of dry and moist cellulose-predictions with molecular dynamics simulations. Biomacromolecules 2024, 25, 7581–7593. [Google Scholar] [CrossRef]
- Rashad, A.; Grøndahl, M.; Heggset, E.B.; Mustafa, K.; Syverud, K. Responses of rat mesenchymal stromal cells to nanocellulose with different functional groups. ACS Appl. Bio Mater. 2023, 6, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Matari, I.A.I.; Han, S.S. 3D printable carboxylated cellulose nanocrystal-reinforced hydrogel inks for tissue engineering. Biofabrication 2020, 12, 025029. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Jiang, Q.; Ren, X.; Xie, Z.; Huang, T.-S. Electrospun non-leaching biocombatible antimicrobial cellulose acetate nanofibrous mats. J. Ind. Eng. Chem. 2015, 27, 315–321. [Google Scholar] [CrossRef]
- Haider, M.K.; Ullah, A.; Sarwar, M.N.; Saito, Y.; Sun, L.; Park, S.; Kim, I.S. Lignin-mediated in-situ synthesis of CuO nanoparticles on cellulose nanofibers: A potential wound dressing material. Int. J. Biol. Macromol. 2021, 173, 315–326. [Google Scholar] [CrossRef]
- Cosimi, A.; Stöbener, D.D.; Nickl, P.; Schusterbauer, R.; Donskyi, I.S.; Weinhart, M. Interfacial nanoengineering of hydrogel surfaces via block copolymer self-assembly. ACS Appl. Mater. Interfaces 2025, 17, 10073–10086. [Google Scholar] [CrossRef]
- Singh, N.; Cui, X.; Boland, T.; Husson, S.M. The role of independently variable grafting density and layer thickness of polymer nanolayers on peptide adsorption and cell adhesion. Biomaterials 2007, 28, 763–771. [Google Scholar] [CrossRef]
- Chen, J.; Liu, M.; Zhang, N.; Dai, P.; Gao, C.; Ma, L.; Liu, H. Influence of the grafted chain length on responsive behaviors of the grafted poly(DEA-co-DMAEMA) hydrogel. Sens. Actuators B Chem. 2010, 149, 34–43. [Google Scholar] [CrossRef]
- Huang, W.; Zulkifli, M.Y.B.; Chai, M.; Lin, R.; Wang, J.; Chen, Y.; Chen, V.; Hou, J. Recent advances in enzymatic biofuel cells enabled by innovative materials and techniques. Exploration 2023, 3, 20220145. [Google Scholar] [CrossRef]
- Leisz, S.; Trutschel, M.-L.; Mäder, K.; Scheller, C.; Strauss, C.; Simmermacher, S. Tabotamp®, respectively, surgicel®, increases the cell death of neuronal and glial cells in vitro. Materials 2020, 13, 2453. [Google Scholar] [CrossRef]
- Park, J.; Lee, D.; Hwang, K.; Lee, J.; Lee, T.-J.; Kim, Y.; Kim, J.H.; Lee, J.; Youe, W.-J.; Chun, S.-J.; et al. Effect of catalyst and oxidant concentrations in a tempo oxidation system on the production of cellulose nanofibers. RSC Adv. 2024, 14, 32852–32862. [Google Scholar] [CrossRef]
- Chen, Y.X.; Hong, G.H.; Li, L.; Qu, Q.; Li, G.; Teng, K.; Ge, L.; Wu, J.J.; Zhang, H.X. Revolutionizing thermal energy harvesting: Bio-flex gel empowered by deep eutectic solvent with exceptional thermopower and electromagnetic wave shielding. Compos. Part B-Eng. 2025, 295, 112166. [Google Scholar] [CrossRef]
- Li, J.; He, K.Y.; Xu, Q. Tissue regeneration with gelatine/polysaccharide derived hydrogel scaffolds: From formulation to in vivo efficacy. Gels 2023, 9, 744. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.Y.; Kang, L.; Li, Z.M.; Tseng, S.L.; Wang, L.Q.; Li, T.H.; Li, Z.J.; Huang, J.Z.; Yu, N.Z.; Long, X. Biological scaffold as potential platforms for stem cells: Current development and applications in wound healing. World J. Stem Cells 2024, 16, 334–352. [Google Scholar] [CrossRef] [PubMed]
- Kheradmandi, R.; Farahani, M.K.; Alizadeh, M.; Rezaei, N.; Zamani, S.; Ehterami, A.; Salehi, M. Enhanced wound healing through hydrogel: Arthrospira platensis and chlorella vulgaris in carboxymethyl cellulose/ carboxymethyl chitosan/gelatin matrix with decellularized amniotic membrane in male wistar rats. Iran. J. Basic. Med. Sci. 2025, 28, 1548–1562. [Google Scholar]
- Ning, X.Z.; Du, R.; Zhang, M.H.; Yang, Y.T.; Yu, F.; Xu, X.M.; Meng, B.Y.; Yan, K. Three-dimensional bioprinted silk fibroin-hydroxypropyl cellulose scaffold loaded with tendon stem/progenitor cells for the prevention of heterotopic ossification following Achilles tendon injury. Int. J. Bioprint. 2025, 11, 297–314. [Google Scholar]
- Ziblim, Z.; Liu, Z.L.; Thapsukhon, B.; Jaikang, C.; Konguthaithip, G.; Choommongkol, V.; Boonyawan, D.; Ruangsuriya, J. The lysine degradation pathway analyzed with 1H-NMR-targeted metabolomics of MG63 cells on poly(l-lactide)-based scaffolds. RSC Adv. 2025, 15, 42614–42629. [Google Scholar] [CrossRef]
- Mohammadalipour, M.; Alihosseini, F.; Toloue, E.B.; Karbasi, S.; Mohammadalipour, Z. Evaluation of PHB-chitosan/CNC scaffolds’ applicability for bone tissue engineering via MG-63 osteoblastic cell cultivation and osteogenic markers gene expression. Int. J. Biol. Macromol. 2025, 320, 146041. [Google Scholar] [CrossRef]
- Tiomnova, O.T.; Coelho, F.; Pellizaro, T.A.G.; Chanfrau, J.E.R.; Capote, T.S.D.; Basmaji, P.; Pantoja, Y.V.; Guastaldi, A.C. Preparation of scaffolds of amorphous calcium phosphate and bacterial cellulose for use in tissue regeneration by freeze-drying process. Biointerface Res. Appl. Chem. 2021, 11, 7357–7367. [Google Scholar] [CrossRef]
- Ma, J.G.; Wu, C.T. Bioactive inorganic particles-based biomaterials for skin tissue engineering. Exploration 2022, 2, 20210083. [Google Scholar] [CrossRef]
- Park, M.; Lee, D.; Shin, S.; Hyun, J. Effect of negatively charged cellulose nanofibers on the dispersion of hydroxyapatite nanoparticles for scaffolds in bone tissue engineering. Colloids Surf. B Biointerfaces 2015, 130, 222–228. [Google Scholar] [CrossRef]
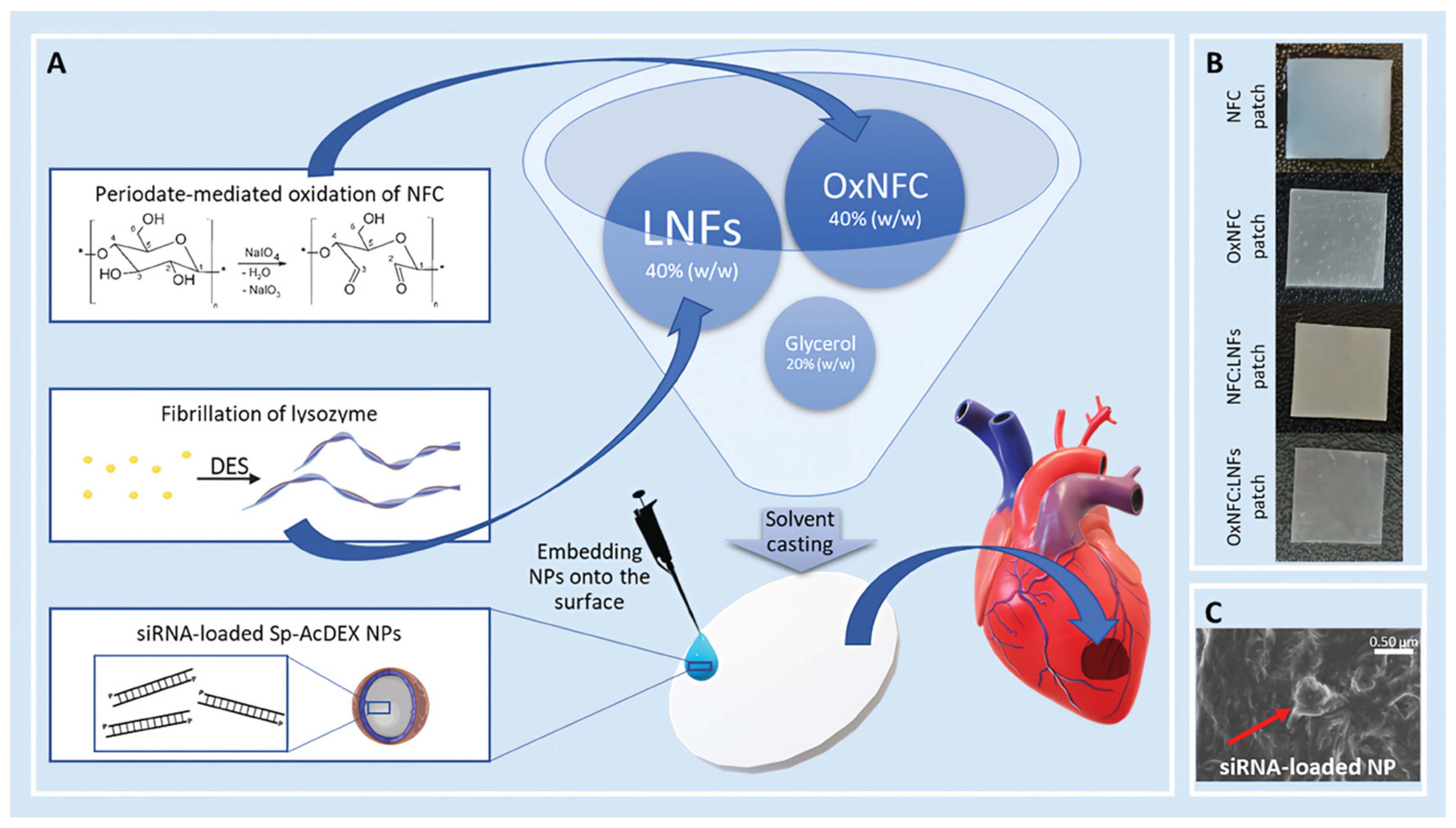
- Carvalho, T.; Bártolo, R.; Correia, A.; Vilela, C.; Wang, S.; Santos, H.A.; Freire, C.S.R. Implantable patch of oxidized nanofibrillated cellulose and lysozyme amyloid nanofibrils for the regeneration of infarcted myocardium tissue and local delivery of rna-loaded nanoparticles. Macromol. Rapid Commun. 2024, 45, e2400129. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, L.; Dong, C.; Liu, J.; Cui, G.; Gao, S.; Liu, Z. Exploring the potential and advancements of circular RNA therapeutics. Exploration 2025, 5, e20240044. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Zhang, Z.; Tang, W.; Dai, Y. Gel/hydrogel-based in situ biomaterial platforms for cancer postoperative treatment and recovery. Exploration 2023, 3, 20220173. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.Y.; Zhang, B.Y.; Wang, Z.Q.; Han, Q.X.; An, M.; Ueda, M.; Ito, Y. Natural polyphenol tannin-immobilized composites: Rational design and versatile applications. J. Mater. Chem. B 2023, 11, 4619–4660. [Google Scholar] [CrossRef]
- Nada, A.A.; Abdellatif, F.H.H.; Soliman, A.A.F.; Shen, J.; Hudson, S.M.; Abou-Zeid, N.Y. Fabrication and bioevaluation of a medicated electrospun mat based on azido-cellulose acetate via click chemistry. Cellulose 2019, 26, 9721–9736. [Google Scholar] [CrossRef]
- De Taillac, L.B.; Porté-Durrieu, M.C.; Labrugère, C.; Bareille, R.; Amédée, J.; Baquey, C. Grafting of rgd peptides to cellulose to enhance human osteoprogenitor cells adhesion and proliferation. Compos. Sci. Technol. 2004, 64, 827–837. [Google Scholar] [CrossRef]
- Hao, L.; Zhang, Y.; Sun, Y.; Han, Y.; Ye, J.; Wang, H.; Gao, L.; Wang, D.; Gao, Y.; Yang, Y.; et al. Advances in nano-delivery systems for targeted natural product delivery to specific skin layers in disease therapy and skin care. Chin. Chem. Lett. 2025, 111597. [Google Scholar] [CrossRef]
- Namazi, H.; Rakhshaei, R.; Hamishehkar, H.; Kafil, H.S. Antibiotic loaded carboxymethylcellulose/MCM-41 nanocomposite hydrogel films as potential wound dressing. Int. J. Biol. Macromol. 2016, 85, 327–334. [Google Scholar] [CrossRef]
- Zhu, Z.; Zhao, J.; Ji, X.; Hu, W.; Leng, W.; Xu, C.; Li, X.; Yang, K.; Li, X.; Zheng, Y.; et al. Bacterial cellulose-based scaffold with in-situ cationic micelle modification for urethral stricture disease: Sustained drug components release, cytokines recruitment, and bacterial microenvironment regulation. Bioact. Mater. 2025, 51, 306–317. [Google Scholar] [CrossRef]
- Dutta, S.D.; Patil, T.V.; Ganguly, K.; Randhawa, A.; Acharya, R.; Moniruzzaman, M.; Lim, K.T. Trackable and highly fluorescent nanocellulose-based printable bio-resins for image-guided tissue regeneration. Carbohydr. Polym. 2023, 320, 121232. [Google Scholar] [CrossRef]
- Deng, Y.; Xi, J.; Meng, L.; Lou, Y.; Seidi, F.; Wu, W.; Xiao, H. Stimuli-responsive nanocellulose hydrogels: An overview. Eur. Polym. J. 2022, 180, 111591. [Google Scholar] [CrossRef]
- Dong, D.; Chen, R.; Jia, J.; Zhao, C.; Chen, Z.; Lu, Q.; Sun, Y.; Huang, W.; Wang, C.; Li, Y.; et al. Tailoring and application of a multi-responsive cellulose nanofibre-based 3D nanonetwork wound dressing. Carbohydr. Polym. 2023, 305, 120542. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, V.; Kumar, P.S.; Albert, A.A.; Krishnasamy, S.; Chandrasekar, M. Recent progress in nanocellulose-based biocomposites for bone tissue engineering and wound healing applications. Carbohydr. Polym. 2025, 357, 123455. [Google Scholar] [CrossRef] [PubMed]
- Mahendiran, B.; Muthusamy, S.; Selvakumar, R.; Rajeswaran, N.; Sampath, S.; Jaisankar, S.N.; Krishnakumar, G.S. Decellularized natural 3D cellulose scaffold derived from Borassus flabellifer (Linn.) as extracellular matrix for tissue engineering applications. Carbohydr. Polym. 2021, 272, 118494. [Google Scholar] [CrossRef]
- Bhardwaj, D.; Singhmar, R.; Garg, M.; Gupta, D.; Dhiman, A.; Han, S.S.; Agrawal, G. Designing advanced hydrogel inks with direct ink writing based 3D printability for engineered biostructures. Eur. Polym. J. 2024, 205, 112736. [Google Scholar] [CrossRef]
- Ravanbakhsh, H.; Bao, G.; Luo, Z.; Mongeau, L.G.; Zhang, Y.S. Composite inks for extrusion printing of biological and biomedical constructs. ACS Biomater. Sci. Eng. 2020, 7, 4009–4026. [Google Scholar] [CrossRef]
- Luo, H.; Cha, R.; Li, J.; Hao, W.; Zhang, Y.; Zhou, F. Advances in tissue engineering of nanocellulose-based scaffolds: A review. Carbohydr. Polym. 2019, 224, 115144. [Google Scholar] [CrossRef]
- De France, K.J.; Cranston, E.D.; Hoare, T. Mechanically reinforced injectable hydrogels. ACS Appl. Polym. Mater. 2019, 2, 1016–1030. [Google Scholar] [CrossRef]
- Read, S.A.; Go, C.S.; Ferreira, M.J.S.; Ligorio, C.; Kimber, S.J.; Dumanli, A.G.; Domingos, M.A.N. Nanocrystalline cellulose as a versatile engineering material for extrusion-based bioprinting. Pharmaceutics 2023, 15, 2432. [Google Scholar] [CrossRef]
- Wang, Q.; Backman, O.; Nuopponen, M.; Xu, C.; Wang, X. Rheological and printability assessments on biomaterial inks of nanocellulose/photo-crosslinkable biopolymer in light-aided 3D printing. Front. Chem. Eng. 2021, 3, 723429. [Google Scholar] [CrossRef]
- Elango, J.; Zamora-Ledezma, C. Rheological, structural, and biological trade-offs in bioink design for 3D bioprinting. Gels 2025, 11, 659. [Google Scholar] [CrossRef]
- Alibabaei-Omran, F.; Zabihi, E.; Seifalian, A.M.; Javanmehr, N.; Samadikuchaksaraei, A.; Gholipourmalekabadi, M.; Asghari, M.H.; Nouri, H.R.; Pourbagher, R.; Bouzari, Z.; et al. Bilateral crosslinking with glutaraldehyde and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide: An optimization strategy for the application of decellularized human amniotic membrane in tissue engineering. J. Tissue Eng. Regen. Med. 2024, 2024, 8525930. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.; Cheung, D.Y.; Butcher, J.T. Incorporating nanocrystalline cellulose into a multifunctional hydrogel for heart valve tissue engineering applications. J. Biomed. Mater. Res. Part A 2021, 110, 76–91. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.M.; Wang, L.W.; Zhai, T.L.; Wang, X.C.; Dan, Y.; Turng, L.S. The surface grafting of graphene oxide with poly (ethylene glycol) as a reinforcement for poly(lactic acid) nanocomposite scaffolds for potential tissue engineering applications. J. Mech. Behav. Biomed. Mater. 2016, 53, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Zhao, Y.; Li, Z.; Li, L. Emerging polymeric electrospun fibers: From structural diversity to application in flexible bioelectronics and tissue engineering. Exploration 2022, 2, 20210029. [Google Scholar] [CrossRef]
- Petre, D.G.; Leeuwenburgh, S.C.G. The use of fibers in bone tissue engineering. Tissue Eng. Part B Rev. 2022, 28, 141–159. [Google Scholar] [CrossRef]
- Altun, E.; Aydogdu, M.O.; Togay, S.O.; Sengil, A.Z.; Ekren, N.; Haskoylu, M.E.; Oner, E.T.; Altuncu, N.A.; Ozturk, G.; Crabbe-Mann, M.; et al. Bioinspired scaffold induced regeneration of neural tissue. Eur. Polym. J. 2019, 114, 98–108. [Google Scholar] [CrossRef]
- Tabassum, A.; Rajasekaran, S.P.; Kumar, K.K.; Deng, M.; Hu, Z.Q. Influence of electrospinning parameters on polycaprolactone fiber alignment for the differentiation of embryonic stem cells into neuronal lineage—A systematic study. Biomater. Adv. 2026, 179, 214485. [Google Scholar] [CrossRef]
- Nasalapure, A.V.; Pattadkal, S.; Ghatti, V.; Chapi, S.; Kumarswamy, Y.K.; Kasai, D.R.; Chalannavar, R.K. Design of cellulose nanocrystal-integrated poly(vinyl alcohol)/polyethylene glycol electrospun nanofiber scaffolds for biomedical applications. J. Appl. Polym. Sci. 2025, 142, e56463. [Google Scholar] [CrossRef]
- Chen, H.; Jia, P.; Kang, H.; Zhang, H.; Liu, Y.; Yang, P.; Yan, Y.; Zuo, G.; Guo, L.; Jiang, M.; et al. Upregulating hif-1α by hydrogel nanofibrous scaffolds for rapidly recruiting angiogenesis relative cells in diabetic wound. Adv. Healthc. Mater. 2016, 5, 907–918. [Google Scholar] [CrossRef]
- Huang, Z.; Tong, A.; Xing, T.; He, A.; Luo, Y.; Zhang, Y.; Wang, M.; Qiao, S.; Shi, Z.; Chen, F.; et al. A triple-crosslinking strategy for high-performance regenerated cellulose fibers derived from waste cotton textiles. Int. J. Biol. Macromol. 2024, 264, 130779. [Google Scholar] [CrossRef]
- Bade, R.; Gebert, B.; Tsarkova, L.; Bahners, T.; Gutmann, J.S.; Koch, D.; Murshed, M.M.; Müssig, J. Towards coupling agent-free composites made from regenerated cellulose/HDPE by UV radiation-induced cross-linking. Compos. Interfaces 2025, 32, 1011–1031. [Google Scholar] [CrossRef]
- Sivasankar, M.V.; Parcha, S.R. Chitosan/microfibrillated cellulose/bioceramic scaffolds for bone tissue engineering. Cellulose 2025, 32, 6067–6082. [Google Scholar] [CrossRef]
- Wang, B.X.; Lv, X.G.; Li, Z.; Zhang, M.H.; Yao, J.J.; Sheng, N.; Lu, M.J.; Wang, H.P.; Chen, S.Y. Urethra-inspired biomimetic scaffold: A therapeutic strategy to promote angiogenesis for urethral regeneration in a rabbit model. Acta Biomater. 2020, 102, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Stokols, S.; Tuszynski, M.H. Freeze-dried agarose scaffolds with uniaxial channels stimulate and guide linear axonal growth following spinal cord injury. Biomaterials 2006, 27, 443–451. [Google Scholar] [CrossRef]
- Dudaryeva, O.Y.; Cousin, L.; Krajnovic, L.; Gröbli, G.; Sapkota, V.; Ritter, L.; Deshmukh, D.; Cui, Y.; Style, R.W.; Levato, R.; et al. Tunable Bicontinuous macroporous cell culture scaffolds via kinetically controlled phase separation. Adv. Mater. 2025, 37, e2410452. [Google Scholar] [CrossRef]
- Thai, T.H.; Nguyen, T.M.; Truong, M.-D.; Phan, T.T.T.; Le, D.T.; Doan, H.N.; Nguyen, T.-H. Fabrication of 3D PCL/PVP scaffolds using monosodium glutamate as porogen by solvent casting/particulate leaching method for oral and maxillofacial bone tissue engineering. Biomed. Mater. 2024, 19, 055030. [Google Scholar] [CrossRef]
- Doustdar, F.; Olad, A.; Ghorbani, M. Effect of glutaraldehyde and calcium chloride as different crosslinking agents on the characteristics of chitosan/cellulose nanocrystals scaffold. Int. J. Biol. Macromol. 2022, 208, 912–924. [Google Scholar] [CrossRef]
- Ojagh, S.M.A.; Vahabzadeh, F.; Fallah, N.; Lan, X.; Adesida, A.B.; Kamkar, M.; Van De Ven, T.G.M.; Koshani, R. Highly functionalized all-cellulose nanocomposites via bacteria-enabled in-situ modifications. Chem. Eng. J. 2024, 498, 155409. [Google Scholar] [CrossRef]
- Pogorelova, N.; Parshin, D.; Lipovka, A.; Besov, A.; Digel, I.; Larionov, P. Structural and viscoelastic properties of bacterial cellulose composites: Implications for prosthetics. Polymers 2024, 16, 3200. [Google Scholar] [CrossRef]
- Schaffner, M.; Rühs, P.A.; Coulter, F.; Kilcher, S.; Studart, A.R. 3D printing of bacteria into functional complex materials. Sci. Adv. 2017, 3, eaao6804. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, M.; Mahmoudi, N.; Anaran, F.R.; Simchi, A. Electrospinning of nanodiamond-modified polysaccharide nanofibers with physico-mechanical properties close to natural skins. Mar. Drugs 2016, 14, 128. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.Y.; Wang, J.; Chen, H.X.; Shi, X.L.; Wang, X.C.; Zhu, Y.H.; Tan, Z.K. TThe topography of fibrous scaffolds modulates the paracrine function of Ad-MSCs in the regeneration of skin tissues. Biomater. Sci. 2019, 7, 4248–4259. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, N.; Buang, F.; Lazim, A.M.; Ahmad, N.; Martin, C.; Amin, M.C.I.M. Characterization and biocompatibility evaluation of bacterial cellulose-based wound dressing hydrogel: Effect of electron beam irradiation doses and concentration of acrylic acid. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 105, 2553–2564. [Google Scholar] [CrossRef]
- D A, G.; Adhikari, J.; Debnath, P.; Ghosh, S.; Ghosh, P.; Thomas, S.; Ghandilyan, E.; Gorbatov, P.; Kuchukyan, E.; Gasparyan, S.; et al. 3D printing of bacterial cellulose for potential wound healing applications: Current trends and prospects. Int. J. Biol. Macromol. 2024, 279, 135213. [Google Scholar] [CrossRef]
- Dutta, S.D.; Patel, D.K.; Lim, K.-T. Functional cellulose-based hydrogels as extracellular matrices for tissue engineering. J. Biol. Eng. 2019, 13, 55. [Google Scholar] [CrossRef]
- Qin, C.; Sun, B.; Chen, X.; Ping, D.; Bai, W.; Shi, L.; Nazir, A.; Chen, C.; Sun, D. Schiff base-grafted bacterial cellulose-PCL composites: Humidity/temperature-driven hydrogen bond reconfiguration for mechanically adaptive nanofiber. Chem. Eng. J. 2025, 525, 170132. [Google Scholar] [CrossRef]
- Xu, Y.; Shi, Y.; Lei, F.; Dai, L. A novel and green cellulose-based Schiff base-Cu (II) complex and its excellent antibacterial activity. Carbohydr. Polym. 2020, 230, 115671. [Google Scholar] [CrossRef]
- Li, M.; Hu, X.; Liu, X.; Zhao, L.; Zhao, W.; Li, Y.; Bie, X.; Jiao, H.; Zhao, Y.; Ma, C. 3D bioprinted piezoelectric hydrogel synergized with LIPUS to promote bone regeneration. Mater. Today Bio 2025, 31, 101604. [Google Scholar] [CrossRef]
- Yan, J.; Qi, S.; Zhao, Y.; Tian, P.; Kong, N.; Ma, W.; Yan, P.; Zhang, J.; Gao, X.; Guan, H.; et al. 3D-printed triply periodic minimal surface ceramic scaffold loaded with bone morphogenetic protein-2 and zoledronic for cranium defect repairment. J. Tissue Eng. Regen. Med. 2025, 2025, 9964384. [Google Scholar] [CrossRef]
- Ruccolo, L.; Evangelista, A.; Benazzo, M.; Conti, B.; Pisani, S. Electrospun bio-scaffolds for mesenchymal stem cell-mediated neural differentiation: Systematic review of advances and future directions. Int. J. Mol. Sci. 2025, 26, 9528. [Google Scholar] [CrossRef] [PubMed]
- Fan, N.; Song, D.; Ding, H.; Yang, H.; Xu, C.; Wang, C.; Yang, Y. E-jet 3D printed aligned nerve guidance conduits incorporated with decellularized extracellular matrix hydrogel encapsulating extracellular vesicles for peripheral nerve repair. Acta Biomater. 2025, 194, 122–139. [Google Scholar] [CrossRef] [PubMed]
- Moazzam, M. From waste to wealth and regeneration: A prisma review of sustainable biomaterials for scalable tissue engineering. Biotechnol. Sustain. Mater. 2025, 2, 22. [Google Scholar] [CrossRef]
- Kathuria, N.; Tripathi, A.; Kar, K.; Kumar, A. Synthesis and characterization of elastic and macroporous chitosan-gelatin cryogels for tissue engineering. Acta Biomater. 2009, 5, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhu, Z.; Liu, Y.; Zheng, Y.; Xie, Y.; Lin, J.; Cai, T. Double-modified bacterial cellulose/soy protein isolate composites by laser hole forming and selective oxidation used for urethral repair. Biomacromolecules 2021, 23, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Ding, H.; Tan, J.; Zhang, Y.; Guo, Y.; Kang, R.; Zhu, X.; Wang, H.; Baughman, R.; Yin, Z.; et al. Multifaceted mechanical responsive metamaterials: Mechanisms, fabrications, and applications. Innovation 2026, 7, 101070. [Google Scholar] [CrossRef]
- Bracchi, M.; Nicotra, F.; Russo, L. 3D Bioprintable hydrogels via enzymatic crosslinking of hyaluronic acid and phenol-functionalized gelatin to mimic extracellular (ECM) network. Carbohydr. Polym. Technol. Appl. 2025, 11, 100928. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, T.; Xu, J.; Yan, S.; Qi, B. Soybean protein isolate/sodium alginate double-network hydrogels with high mechanical strength via synergistic cross-linking of imine and ionic bonds. Food Chem. 2025, 494, 146233. [Google Scholar] [CrossRef]
- Jing, R.; Pennisi, C.P.; Nielsen, T.T.; Larsen, K.L. Advanced supramolecular hydrogels and their applications in the formulation of next-generation bioinks for tissue engineering: A review. Int. J. Biol. Macromol. 2025, 311, 143461. [Google Scholar] [CrossRef]
- Shen, T.; Dong, H.; Wang, P. Research progress on nanocellulose and its composite materials as orthopedic implant biomaterials. Alex. Eng. J. 2024, 87, 575–590. [Google Scholar] [CrossRef]
- Sreedharan, M.; Vijayamma, R.; Liyaskina, E.; Revin, V.V.; Ullah, M.W.; Shi, Z.J.; Yang, G.; Grohens, Y.; Kalarikkal, N.; Khan, K.A.; et al. Nanocellulose-based hybrid scaffolds for skin and bone tissue engineering: A 10-year overview. Biomacromolecules 2024, 25, 2136–2155. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wang, J.; Zhou, S.; Pan, P.; Chen, T.; Wang, X.; Liu, W. Biodegradable cellulose scaffolds embedding kartogenin-loaded microspheres for cartilage regeneration. Int. J. Biol. Macromol. 2025, 306, 141344. [Google Scholar] [CrossRef] [PubMed]
- Von Witzleben, M.; Gasiūnaitė, A.; Ihle, M.; Akkineni, A.R.; Schütz, K.; Ahlfeld, T.; Gelinsky, M.; Lode, A.; Duin, S. Uniting 4D printing and melt electrowriting for the enhancement of regenerative small diameter vascular grafts. Adv. Healthc. Mater. 2025, 14, e02380. [Google Scholar] [CrossRef] [PubMed]
- Kurtuluş, O.Ç.; Ondaral, S.; Emin, N.; Kadak, A.E. Bioaerogels produced from tempo oxidized nano cellulose with chitosan, gelatin, and alginate: General performances for wound dressing application. Cellulose 2024, 32, 295–311. [Google Scholar] [CrossRef]
- Adhikari, J.; Dasgupta, S.; Das, P.; Gouripriya, D.A.; Barui, A.; Basak, P.; Ghosh, M.; Saha, P. Bilayer regenerated cellulose/quaternized chitosan-hyaluronic acid/collagen electrospun scaffold for potential wound healing applications. Int. J. Biol. Macromol. 2024, 261, 129661. [Google Scholar] [CrossRef]
- Xun, X.; Li, Y.; Zhu, X.; Zhang, Q.; Lu, Y.; Yang, Z.; Wan, Y.; Yao, F.; Deng, X.; Luo, H. Fabrication of robust, shape recoverable, macroporous bacterial cellulose scaffolds for cartilage tissue engineering. Macromol. Biosci. 2021, 21, e2100167. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, W.; Wu, X.; Xie, C.; Zhang, Y.; Li, L.; Gu, Y.; Hu, Z.; Zhai, X.; Liang, R.; et al. Divide-and-conquer strategy with engineered ossification center organoids for rapid bone healing through developmental cell recruitment. Nat. Commun. 2025, 16, 6200. [Google Scholar] [CrossRef]
- Ling, T.X.; Zha, X.J.; Zhou, K.; Zhao, X.; Jia, J.; Pan, K.Q.; Chen, A.J.; Yang, W.; Zhou, Z.K. A facile strategy toward hierarchically porous composite scaffold for osteosarcoma ablation and massive bone defect repair. Compos. Part B-Eng. 2022, 234, 109660. [Google Scholar] [CrossRef]
- Li, X.; Ding, J.; Wang, J.; Zhuang, X.; Chen, X. Biomimetic biphasic scaffolds for osteochondral defect repair. Regen. Biomater. 2015, 2, 221–228. [Google Scholar] [CrossRef]
- Younus, Z.M.; Ahmed, I.; Roach, P.; Forsyth, N.R. A phosphate glass reinforced composite acrylamide gradient scaffold for osteochondral interface regeneration. Biomater. Biosyst. 2024, 15, 100099. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, X.; Shu, H.; Tan, X.; Fu, J.; Sun, G.; Jiang, X.; Sun, D.; Chen, C.; Sun, L.; et al. In situ grown bacterial cellulose-based integrated biphasic biomimetic scaffold for synchronous repair of critical-sized osteochondral defects. Mater. Today Adv. 2025, 28, 100671. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, D.; Liu, Y.; Peng, L.; Lu, D.; Wang, P.; Ke, D.; Yang, H.; Zhu, X.; Ruan, C. 3d-bioprinted anisotropic bicellular living hydrogels boost osteochondral regeneration via reconstruction of cartilage-bone interface. Innovation 2024, 5, 100542. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.; Sun, B.; Shen, W.; Gai, Y.; Shi, L.; Chen, C.; Sun, D. Oriented bacterial cellulose microfibers with tunable mechanical performance fabricated via green reassembly avenue. Int. J. Biol. Macromol. 2024, 254, 127990. [Google Scholar] [CrossRef] [PubMed]
- Utoiu, E.; Manoiu, V.S.; Oprita, E.I.; Craciunescu, O. Bacterial cellulose: A sustainable source for hydrogels and 3D-printed scaffolds for tissue engineering. Gels 2024, 10, 387. [Google Scholar] [CrossRef]
- Cetin, F.S.; Avci, T.; Uygur, E.; Ilhan, E.; Kaya, E.; Tinaz, G.B.; Duta, L.; Dogan, C.; Gunduz, O. Development and characterization of antimicrobial chitosan/polyethylene oxide/bacterial cellulose nanofibers. Polymers 2025, 17, 693. [Google Scholar] [CrossRef]
- Zhong, M.L.; Li, J.S.; Tang, A.Q.; Zhang, Q.; Ji, D.H.; Peng, M.X.; Zhang, R.C.; Xiong, G.Y.; Wan, Y.Z.; Fan, H.S. A facile green approach for fabricating bacterial cellulose scaffold with macroporous structure and cell affinity. J. Bioact. Compat. Polym. 2019, 34, 442–452. [Google Scholar] [CrossRef]
- Davlet, M.; Smyrnova, K.; Pogrebnjak, A. Advanced biomaterials in tissue engineering: A critical review of nanocomposites based on bacterial cellulose, mxenes, hydroxyapatite, and metal particles for regenerative medicine. Adv. Colloid Interface Sci. 2025, 345, 103634. [Google Scholar] [CrossRef]
- Deng, J.; Song, Q.; Liu, S.; Pei, W.; Wang, P.; Zheng, L.; Huang, C.; Ma, M.; Jiang, Q.; Zhang, K. Advanced applications of cellulose-based composites in fighting bone diseases. Compos. Part B Eng. 2022, 245, 110221. [Google Scholar] [CrossRef]
- Liu, H.; Xing, F.; Yu, P.Y.; Zhe, M.; Shakya, S.; Liu, M.; Xiang, Z.; Duan, X.; Ritz, U. Multifunctional aerogel: A unique and advanced biomaterial for tissue regeneration and repair. Mater. Des. 2024, 243, 113091. [Google Scholar] [CrossRef]
- Garai, S.; Sinha, A. Biomimetic nanocomposites of carboxymethyl cellulose-hydroxyapatite: Novel three dimensional load bearing bone grafts. Colloids Surf. B Biointerfaces 2014, 115, 182–190. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Prabakaran, M.; Ke, M.; Gang, X.; Chung, I.M.; Um, I.C.; Gopiraman-S, M.; Kim, I.S. Highly dispersed nanoscale hydroxyapatite on cellulose nanofibers for bone regeneration. Mater. Lett. 2016, 168, 56–61. [Google Scholar] [CrossRef]
- Feng, Y.H.Z.; Cölfen, H.; Xiong, R. Organized mineralized cellulose nanostructures for biomedical applications. J. Mater. Chem. B 2023, 11, 5321–5349. [Google Scholar] [CrossRef] [PubMed]
- Sadathojai, M.; Asadnia, M.; Zarei-Fard, N.; Arvaneh, A.-R. Electrospinning of liquefied banana stem residue in conjugation with hydroxyapatite nanocrystals: Towards new scaffolds for bone tissue engineering. Cellulose 2022, 29, 4039–4056. [Google Scholar] [CrossRef]
- Li, L.; Lu, P.; Liu, Y.; Yang, J.; Li, S. Three-dimensional-bioprinted bioactive glass/cellulose composite scaffolds with porous structure towards bone tissue engineering. Polymers 2023, 15, 2226. [Google Scholar] [CrossRef] [PubMed]
- Monfared, M.H.; Ranjbar, F.E.; Torbati, M.; Poursamar, S.A.; Lotfibakhshaiesh, N.; Ai, J.; Ebrahimi-Barough, S.; Azami, M. Preparation and characterization of 3D nanocomposite scaffold from bioactive glass/β-tricalcium phosphate via Robocasting method for bone tissue engineering. J. Non-Cryst. Solids 2022, 593, 121769. [Google Scholar] [CrossRef]
- Zhao, J.; Pan, X.; Sun, Y.; Xu, F.; Fang, Y. Harnessing stereomicrostructural engineering for next-generation biodegradable adhesives. Innovation 2025, 6, 100936. [Google Scholar] [CrossRef]
- Song, S.-H.; Yun, Y.-P.; Kim, H.-J.; Park, K.; Kim, S.E.; Song, H.-R. Bone formation in a rat tibial defect model using carboxymethyl cellulose/BioC/bone morphogenic protein-2 hybrid materials. Biomed Res. Int. 2014, 2014, 230152. [Google Scholar] [CrossRef]
- Simpson, C.R.; Kelly, H.M.; Murphy, C.M. The design and development of an injectable thermoresponsive hydrogel for controlled simvastatin release in bone repair applications. Gels 2025, 11, 995. [Google Scholar] [CrossRef]
- Wang, D.Q.; Xu, H.; Liu, J.M.; Chen, Z.X.; Li, Y.Y.; Hu, B.H.; Zhang, D.Y.; Li, J.S.; Chu, H.T. Bio-inspired cellulose reinforced anisotropic composite hydrogel with zone-dependent complex mechanical adaptability and cell recruitment characteristics. Compos. Part B-Eng. 2020, 202, 108418. [Google Scholar] [CrossRef]
- Xu, B.; Luo, Z.Y.; Wang, D.; Huang, Z.Y.; Zhou, Z.K.; Wang, H.Y. In vitro and in vivo repair effects of the ncf-col-nha aerogel scaffold loaded with sost monoclonal antibody and sdf-1 in steroid-induced osteonecrosis. Front. Bioeng. Biotechnol. 2022, 10, 825231. [Google Scholar] [CrossRef]
- Chen, S.; Shi, Y.; Zhang, X.; Ma, J. Evaluation of BMP-2 and VEGF loaded 3D printed hydroxyapatite composite scaffolds with enhanced osteogenic capacity in vitro and in vivo. Mater. Sci. Eng. C 2020, 112, 110893. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhang, M.; Guo, J.; Wei, H.; Li, W.; Luo, Y. Alginate/bacterial cellulose/GelMA scaffolds with aligned nanopatterns and hollow channel networks for vascularized bone repair. Int. J. Biol. Macromol. 2025, 308, 142578. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yang, X.; Xiao, X.; Li, X.; Chen, C.; Sun, D. Biomimetic design of platelet-rich plasma controlled release bacterial cellulose/hydroxyapatite composite hydrogel for bone tissue engineering. Int. J. Biol. Macromol. 2024, 269, 132124. [Google Scholar] [CrossRef] [PubMed]
- Cai, P.; Lu, S.; Yu, J.; Xiao, L.; Wang, J.; Liang, H.; Huang, L.; Han, G.; Bian, M.; Zhang, S.; et al. Injectable nanofiber-reinforced bone cement with controlled biodegradability for minimally-invasive bone regeneration. Bioact. Mater. 2023, 21, 267–283. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, P.; Li, M.; Li, J.; Lin, Y. Remedying infectious bone defects via 3D printing technology. Chin. Chem. Lett. 2025, 36, 110686. [Google Scholar] [CrossRef]
- Iglesias-Mejuto, A.; Malandain, N.; Ferreira-Gonçalves, T.; Ardao, I.; Reis, C.P.; Laromaine, A.; Roig, A.; García-González, C.A. Cellulose-in-cellulose 3D-printed bioaerogels for bone tissue engineering. Cellulose 2023, 31, 515–534. [Google Scholar] [CrossRef]
- Diaz, F.; Zimmermann, L.; Dale, T.P.; Forsyth, N.R.; Boccaccini, A.R. Tuning the properties of all natural polymeric scaffolds for tendon repair with cellulose microfibers. Carbohydr. Polym. Technol. Appl. 2024, 7, 100447. [Google Scholar] [CrossRef]
- Jin, Q.F.; Xinga, S.; Ju, S.; Cao, Y.; Duan, K.; Jin, L.; Zhang, Y.; He, Y.; Chen, J.Y.; Zhang, J.W. Anisotropic robust poly(vinyl alcohol) hydrogels inspired by bio-tissue. Mater. Des. 2025, 250, 113613. [Google Scholar] [CrossRef]
- Tang, R.; Pan, Z.-Z.; Liu, M.; Ohwada, M.; Nishihara, H. Critical impact of nanocellulose on the synthesis of porous cellulose monolith with oriented microchannels: Structure control, mechanics, and mass transport. Nano Res. 2023, 16, 8018–8024. [Google Scholar] [CrossRef]
- Wang, X.; Wu, D.; Liao, W.; Liu, Y.; Pei, W.; Wang, J.; Gu, J.; Wang, P.; Lan, K.; Huang, C. Constructing osteo-inductive bio-ink for 3D printing through hybridization of gelatin with maleic acid modified bacterial cellulose by regulating addition volumes of maleic acid solution. J. Bioresour. Bioprod. 2024, 9, 336–350. [Google Scholar] [CrossRef]
- Salihu, R.; Razak, S.I.A.; Sani, M.H.; Wsoo, M.A.; Zawawi, N.A.; Shahir, S. Citrate-modified bacterial cellulose as a potential scaffolding material for bone tissue regeneration. PLoS ONE 2024, 19, e0312396. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, A.B.D.S.M.; Sivadas, V.P.P.D.; Nair, P.D.P.D. 3D-printed biphasic scaffolds for the simultaneous regeneration of osteochondral tissues. Biomed. Mater. 2021, 16, 054102. [Google Scholar] [CrossRef] [PubMed]
- Kumbhar, J.; Jadhav, S.; Bodas, D.; Barhanpurkar-Naik, A.; Wani, M.; Paknikar, K.; Rajwade, J. In vitro and in vivo studies of a novel bacterial cellulose-based acellular bilayer nanocomposite scaffold for the repair of osteochondral defects. Int. J. Nanomed. 2017, 12, 6437–6459. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, Y.; Kong, H.; Zhang, J.; Chan, H.F.; Wang, J.; Shao, D.; Tao, Y.; Li, M. Microneedle system for tissue engineering and regenerative medicine. Exploration 2023, 3, 20210170. [Google Scholar] [CrossRef]
- Kowalska-Ludwicka, K.; Cala, J.; Grobelski, B.; Sygut, D.; Jesionek-Kupnicka, D.; Kolodziejczyk, M.; Bielecki, S.; Pasieka, Z. Modified bacterial cellulose tubes for regeneration of damaged peripheral nerves. Arch. Med. Sci. 2013, 3, 527–534. [Google Scholar] [CrossRef]
- Bar-Shai, N.; Mann, D.; Shalom, A.; Nyska, A.; Golberg, A. Biocompatibility and structural insights of seaweed-derived cellulose scaffolds in a subcutaneous implantation model. Sci. Rep. 2025, 15, 35667. [Google Scholar] [CrossRef]
- Cherng, J.H.; Chou, S.C.; Chen, C.L.; Wang, Y.W.; Chang, S.J.; Fan, G.Y.; Leung, F.S.; Meng, E. Bacterial cellulose as a potential bio-scaffold for effective re-epithelialization therapy. Pharmaceutics 2021, 13, 1592. [Google Scholar] [CrossRef]
- Bogadi, S.; Uddin, M.E.; Rahman, M.H.; Karri, V.V.S.R.; Begum, R.; Udeabor, S.E. Wound healing in the modern era: Emerging research, biomedical advances, and transformative clinical approaches. J. Drug Deliv. Sci. Technol. 2025, 110, 107058. [Google Scholar] [CrossRef]
- Ye, M.; Mei, N.; Zheng, H.; Wu, B.; Wei, W.; Dong, W.; Song, D.; Ding, W.; Ye, N.; Tremblay, P.-L.; et al. Production of bacterial cellulose from diverse food wastes: Effects on physical properties, structure, and performance. Cellulose 2025, 32, 8845–8861. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.W.; Park, S.; Lim, K.T.; Seonwoo, H.; Kim, Y.; Hong, B.H.; Choung, Y.H.; Chung, J.H. Bacterial cellulose nanofibrillar patch as a wound healing platform of tympanic membrane perforation. Adv. Healthc. Mater. 2013, 2, 1525–1531. [Google Scholar] [CrossRef]
- Liu, D.; Cao, Y.; Qu, R.; Gao, G.; Chen, S.; Zhang, Y.; Wu, M.; Ma, T.; Li, G. Production of bacterial cellulose hydrogels with tailored crystallinity from Enterobacter sp. FY-07 by the controlled expression of colanic acid synthetic genes. Carbohydr. Polym. 2019, 207, 563–570. [Google Scholar] [CrossRef]
- Wei, B.; Yang, G.; Hong, F. Preparation and evaluation of a kind of bacterial cellulose dry films with antibacterial properties. Carbohydr. Polym. 2011, 84, 533–538. [Google Scholar] [CrossRef]
- Sun, Z.; Chen, X.Y.; Ma, X.M.; Cui, X.X.; Yi, Z.; Li, X.D. Cellulose/keratin-catechin nanocomposite hydrogel for wound hemostasis. J. Mater. Chem. B 2018, 6, 6133–6141. [Google Scholar] [CrossRef] [PubMed]
- Dong, D.; Mao, L.; Qin, Z.; Guo, Y.; Yu, J.; Hu, X.; He, J.; Feng, S.; Zhang, M.; Liu, Y.; et al. A simple and effective hydrogel dressing for advanced management of full-thickness skin wound by multi-functional strategies. J. Nanobiotechnol. 2025, 23, 745. [Google Scholar] [CrossRef] [PubMed]
- Joorabloo, A.; Liu, T. Recent advances in reactive oxygen species scavenging nanomaterials for wound healing. Exploration 2024, 4, 20230066. [Google Scholar] [CrossRef]
- Barjasteh, M.; Dehnavi, S.M.; Seyedkhani, S.A.; Rahnamaee, S.Y.; Golizadeh, M. Improved biological activities of dual nanofibrous chitosan/bacterial cellulose wound dressing by a novel silver-based metal-organic framework. Surf. Interfaces 2023, 36, 102631. [Google Scholar] [CrossRef]
- Li, L.; Cheng, X.; Huang, Q.; Cheng, Y.; Xiao, J.; Hu, J. Sprayable antibacterial hydrogels by simply mixing of aminoglycoside antibiotics and cellulose nanocrystals for the treatment of infected wounds. Adv. Healthc. Mater. 2022, 11, e2201286. [Google Scholar] [CrossRef]
- Da, L.C.; Huang, Y.Z.; Xie, H.Q. Progress in development of bioderived materials for dermal wound healing. Regen. Biomater. 2017, 4, 325–334. [Google Scholar] [CrossRef]
- Du, Y.; Liu, Y.; Zhang, Y.; Nie, Y.; Xu, Z.; Qin, L.; Zhang, W.; Lai, Y. Structurally and Functionally Adaptive Biomimetic Periosteum: Materials, Fabrication, and Construction Strategies. Exploration 2025, 5, 70005. [Google Scholar] [CrossRef]
- Liu, J.; Shi, Y.; Cheng, L.; Sun, J.; Yu, S.; Lu, X.; Biranje, S.; Xu, W.; Zhang, X.; Song, J.; et al. Growth factor functionalized biodegradable nanocellulose scaffolds for potential wound healing application. Cellulose 2021, 28, 5643–5656. [Google Scholar] [CrossRef]
- Hong, G.H.; Li, J.W.; Wei, W.Q.; Wu, Y.; Li, L.; Chen, Y.B.; Xie, D.L.; Qu, Q.; Rojas, O.J.; Hu, G.Z.; et al. Starfish-inspired synergistic reinforced hydrogel wound dressing: Dual responsiveness and enhanced bioactive compound delivery for advanced skin regeneration and management. ACS Nano 2025, 19, 10180–10198. [Google Scholar] [CrossRef]
- Zhao, S.; Zhao, W.; Wang, N.; Ling, J.; Ouyang, X.-K. A sustained H2S-releasing nanocellulose-based hydrogel with anti-inflammatory and antibacterial properties for promoting infected wound healing. Carbohydr. Polym. 2025, 355, 123424. [Google Scholar] [CrossRef] [PubMed]
- Kamali, A.; Shamloo, A. Fabrication and evaluation of a bilayer hydrogel-electrospinning scaffold prepared by the freeze-gelation method. J. Biomech. 2020, 98, 109466. [Google Scholar] [CrossRef] [PubMed]
- Maurer, K.; Renkert, M.; Duis, M.; Weiss, C.; Wessel, L.M.; Lange, B. Application of bacterial nanocellulose-based wound dressings in the management of thermal injuries: Experience in 92 children. Burns 2022, 48, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Anton-Sales, I.; Beekmann, U.; Laromaine, A.; Roig, A.; Kralisch, D. Opportunities of bacterial cellulose to treat epithelial tissues. Curr. Drug Targets 2019, 20, 808–822. [Google Scholar] [CrossRef]
- Li, M.; Sun, L.; Liu, Z.X.; Shen, Z.Z.; Cao, Y.Y.; Han, L.; Sang, S.B.; Wang, J.M. 3D bioprinting of heterogeneous tissue-engineered skin containing human dermal fibroblasts and keratinocytes. Biomater. Sci. 2023, 11, 2461–2477. [Google Scholar] [CrossRef]
- Dun, X.-P.; Parkinson, D. Classic axon guidance molecules control correct nerve bridge tissue formation and precise axon regeneration. Neural Regener. Res. 2020, 15, 6–9. [Google Scholar] [CrossRef]
- Zheng, S.; Wei, H.; Cheng, H.; Qi, Y.; Gu, Y.; Ma, X.; Sun, J.; Ye, F.; Guo, F.; Cheng, C. Advances in nerve guidance conduits for peripheral nerve repair and regeneration. Am. J. Stem Cells 2023, 12, 112–123. [Google Scholar]
- Riva, E.R.; Özkan, M.; Contreras, E.; Pawar, S.; Zinno, C.; Escarda-Castro, E.; Kim, J.; Wieringa, P.; Stellacci, F.; Micera, S.; et al. Beyond the limiting gap length: Peripheral nerve regeneration through implantable nerve guidance conduits. Biomater. Sci. 2024, 12, 1371–1404. [Google Scholar] [CrossRef]
- Zhong, C. Industrial-scale production and applications of bacterial cellulose. Front. Bioeng. Biotechnol. 2020, 8, 605374. [Google Scholar] [CrossRef]
- Hou, Y.J.; Wang, X.Y.; Yang, J.; Zhu, R.; Zhang, Z.R.; Li, Y. Development and biocompatibility evaluation of biodegradable bacterial cellulose as a novel peripheral nerve scaffold. J. Biomed. Mater. Res. Part A 2018, 106, 1288–1298. [Google Scholar] [CrossRef]
- Shea, G.-H.; Mok, F. Optimization of nanofiber scaffold properties towards nerve guidance channel design. Neural Regener. Res. 2018, 13, 1179–1180. [Google Scholar] [CrossRef] [PubMed]
- Pawelec, K.M.; Van Boxtel, H.A.; Kluijtmans, S.G.J.M. Ice-templating of anisotropic structures with high permeability. Mater. Sci. Eng. C 2017, 76, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.-H.; Ma, T.-L.; Tan, D.-H.; Su, A.-J.A.; Washington, K.M.; Wang, C.-C.; Huang, Y.-C.; Wu, M.-C.; Su, W.-F. Injectable hydrogel guides neurons growth with specific directionality. Int. J. Mol. Sci. 2023, 24, 7952. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, K.; Xia, B.; Ma, W.; Li, Y.; Zhang, J.; Wang, L.; Lei, M.; Li, B.; Wang, Q.; et al. Piezotronic effect for in situ electrostimulation of neural stem cell therapy for nerve injury. Nano Energy 2024, 120, 109181. [Google Scholar] [CrossRef]
- Zhao, F.; Liu, G.; Guan, Y.; Li, J.; Wang, T.; Zhao, J.; He, W.; Zhang, L.; Meng, H.; Xu, W.; et al. An electromechanical converted bacterial cellulose based composite film for repairing peripheral nerve injury through mimicking physiological electrical signal. Adv. Fiber Mater. 2025, 7, 1929–1948. [Google Scholar] [CrossRef]
- Ding, Z.; Tang, Y.; Zhu, P. Reduced graphene oxide/cellulose nanocrystal composite films with high specific capacitance and tensile strength. Int. J. Biol. Macromol. 2022, 200, 574–582. [Google Scholar] [CrossRef]
- Fraser, S.A.; Van Zyl, W.E. In situpolymerization and electrical conductivity of polypyrrole/cellulose nanocomposites using schweizer’s reagent. RSC Adv. 2022, 12, 22031–22043. [Google Scholar] [CrossRef]
- Carrascosa, A.; Sánchez, J.S.; Morán-Aguilar, M.G.; Gabriel, G.; Vilaseca, F. Advanced flexible wearable electronics from hybrid nanocomposites based on cellulose nanofibers, pedot: Pss and reduced graphene oxide. Polymers 2024, 16, 3035. [Google Scholar] [CrossRef]
- Chang, H.-T.; Heuer, R.A.; Oleksijew, A.M.; Coots, K.S.; Roque, C.B.; Nella, K.T.; Mcguire, T.L.; Matsuoka, A.J. An engineered three-dimensional stem cell niche in the inner ear by applying a nanofibrillar cellulose hydrogel with a sustained-release neurotrophic factor delivery system. Acta Biomater. 2020, 108, 111–127. [Google Scholar] [CrossRef]
- Liu, C.; Li, X.; Zhao, Q.; Xie, Y.; Yao, X.; Wang, M.; Cao, F. Nanofibrous bicomponent scaffolds for the dual delivery of NGF and GDNF: Controlled release of growth factors and their biological effects. J. Mater. Sci. Mater. Med. 2021, 32, 9. [Google Scholar] [CrossRef]
- Qiao, D.; Yang, N.; Shi, L.; Chen, L.; Gu, J.; Peng, X.; Chen, C.; Sun, D. Interface modifiable bacterial cellulose hydrogel based nerve guidance conduit effectively rehabilitate sciatic nerve injured rat. Biomacromolecules 2025, 26, 7434–7446. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.K.; Ray, J.B.; El-Sayed, M.H.; Alalawy, A.I.; Omer, N.; Abdelaziz, M.A.; Abouzeid, R. Functionalization of bacterial cellulose: Exploring diverse applications and biomedical innovations: A review. Int. J. Biol. Macromol. 2024, 264, 130454. [Google Scholar] [CrossRef] [PubMed]
- Asakura, A.; Iwai, T.; Ikeguchi, R.; Aoyama, T.; Noguchi, T.; Yoshimoto, K.; Sakamoto, D.; Fujita, K.; Miyazaki, Y.; Akieda, S.; et al. Nerve regeneration using a Bio 3D conduit derived from umbilical cord-Derived mesenchymal stem cells in a rat sciatic nerve defect model. PLoS ONE 2024, 19, e0310711. [Google Scholar]
- Moutos, F.T.; Guilak, F. Functional properties of cell-seeded three-dimensionally woven poly(epsilon-caprolactone) scaffolds for cartilage tissue engineering. Tissue Eng. Part A 2010, 16, 1291–1301. [Google Scholar] [CrossRef]
- Govindharaj, M.; Al Hashimi, N.; Soman, S.S.; Kanwar, S.; Vijayavenkataraman, S. 3D Bioprinting of human Mesenchymal Stem Cells in a novel tunic decellularized ECM bioink for Cartilage Tissue Engineering. Materialia 2022, 23, 101457. [Google Scholar] [CrossRef]
- Guo, X.; Ma, Y.; Min, Y.; Sun, J.; Shi, X.; Gao, G.; Sun, L.; Wang, J. Progress and prospect of technical and regulatory challenges on tissue-engineered cartilage as therapeutic combination product. Bioact. Mater. 2023, 20, 501–518. [Google Scholar] [CrossRef]
- Phatchayawat, P.P.; Khamkeaw, A.; Yodmuang, S.; Phisalaphong, M. 3D bacterial cellulose-chitosan-alginate-gelatin hydrogel scaffold for cartilage tissue engineering. Biochem. Eng. J. 2022, 184, 108476. [Google Scholar] [CrossRef]
- Andersson, J.; Stenhamre, H.; Bäckdahl, H.; Gatenholm, P. Behavior of human chondrocytes in engineered porous bacterial cellulose scaffolds. J. Biomed. Mater. Res. A 2010, 94, 1124–1132. [Google Scholar] [CrossRef]
- Li, Y.; Xun, X.; Xu, Y.; Zhan, A.; Gao, E.; Yu, F.; Wang, Y.; Luo, H.; Yang, C. Hierarchical porous bacterial cellulose scaffolds with natural biomimetic nanofibrous structure and a cartilage tissue-specific microenvironment for cartilage regeneration and repair. Carbohydr. Polym. 2022, 276, 118790. [Google Scholar] [CrossRef]
- Jia, Y.Y.; Zhu, W.J.; Zheng, M.M.; Huo, M.M.; Zhong, C. Bacterial cellulose/hyaluronic acid composite hydrogels with improved viscoelastic properties and good thermodynamic stability. Plast. Rubber Compos. 2018, 47, 165–175. [Google Scholar] [CrossRef]
- Hammad, M.; Dugué, J.; Maubert, E.; Baugé, C.; Boumédiene, K. Decellularized apple hypanthium as a plant-based biomaterial for cartilage regeneration in vitro: A comparative study of progenitor cell types and environmental conditions. J. Biol. Eng. 2025, 19, 38. [Google Scholar] [CrossRef] [PubMed]
- Shanto, P.C.; Park, S.; Fahad, M.A.A.; Park, M.; Lee, B.-T. 3D bio-printed proteinaceous bioactive scaffold loaded with dual growth factor enhanced chondrogenesis and in situ cartilage regeneration. Bioact. Mater. 2025, 46, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Monzón, M.; Donate, R.; Liu, C.; Tamaddon, M.; Oliveira, J.M. Bamos project: Osteochondral scaffold innovation applied to osteoarthritis. Vitr. Models 2022, 1, 209–211. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Peng, X.; Xi, L.J.; Luo, Y.H.; Wang, Y.H.; Zhou, Y.F.; Yu, X.X. Feasibility study of oxidized naringin as a novel crosslinking agent for crosslinking decellularized porcine achilles tendon and its potential application for anterior cruciate ligament repair. J. Biomed. Mater. Res. A 2023, 111, 170–184. [Google Scholar] [CrossRef]
- Domingues, R.M.A.; Chiera, S.; Gershovich, P.; Motta, A.; Reis, R.L.; Gomes, M.E. Enhancing the biomechanical performance of anisotropic nanofibrous scaffolds in tendon tissue engineering: Reinforcement with cellulose nanocrystals. Adv. Healthc. Mater. 2016, 5, 1364–1375. [Google Scholar] [CrossRef]
- Silva, N.H.C.S.; Garrido-Pascual, P.; Moreirinha, C.; Almeida, A.; Palomares, T.; Alonso-Varona, A.; Vilela, C.; Freire, C.S.R. Multifunctional nanofibrous patches composed of nanocellulose and lysozyme nanofibers for cutaneous wound healing. Int. J. Biol. Macromol. 2020, 165, 1198–1210. [Google Scholar] [CrossRef]
- Panicker, P.S.; Kim, H.C.; Agumba, D.O.; Muthoka, R.M.; Kim, J. Electric field-assisted wet spinning to fabricate strong, tough, and continuous nanocellulose long fibers. Cellulose 2022, 29, 3499–3511. [Google Scholar] [CrossRef]
- Atarbashi-Moghadam, F.; Azadi, A.; Nokhbatolfoghahaei, H.; Taghipour, N. Effect of simultaneous and sequential use of TGF-β1 and TGF-β3 with FGF-2 on teno/ligamentogenic differentiation of periodontal ligament stem cells. Arch. Oral Biol. 2024, 162, 105956. [Google Scholar] [CrossRef]
- Liang, Z.; Xiao, Q.; Wu, Y.; Song, D.; Li, Y.; Chen, J.; Sun, Q.; Yang, Z.; Peng, T.; Wang, Y.; et al. Multidimensional biomimetic strategy integrating structural, functional and biological cues for enhanced tendon-to-bone interface regeneration. Mater. Today Bio 2025, 35, 102510. [Google Scholar] [CrossRef]
- Andrade, F.K.; Costa, R.; Domingues, L.; Soares, R.; Gama, M. Improving bacterial cellulose for blood vessel replacement: Functionalization with a chimeric protein containing a cellulose-binding module and an adhesion peptide. Acta Biomater. 2010, 6, 4034–4041. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.C.; Meng, Q.S.; Hu, J.G. Bacterial nanocellulose hydrogel: A promising alternative material for the fabrication of engineered vascular grafts. Polymers 2023, 15, 3812. [Google Scholar] [CrossRef] [PubMed]
- Klemm, D.; Schumann, D.; Udhardt, U.; Marsch, S. Bacterial synthesized cellulose-artificial blood vessels for microsurgery. Prog. Polym. Sci. 2001, 26, 1561–1603. [Google Scholar] [CrossRef]
- Zahedmanesh, H.; Mackle, J.N.; Sellborn, A.; Drotz, K.; Bodin, A.; Gatenholm, P.; Lally, C. Bacterial cellulose as a potential vascular graft: Mechanical characterization and constitutive model development. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 97B, 105–113. [Google Scholar] [CrossRef]
- Fink, H.; Faxälv, L.; Molnár, G.F.; Drotz, K.; Risberg, B.; Lindahl, T.L.; Sellborn, A. Real-time measurements of coagulation on bacterial cellulose and conventional vascular graft materials. Acta Biomater. 2010, 6, 1125–1130. [Google Scholar] [CrossRef]
- Tohidi, H.; Maleki, N.; Simchi, A. Conductive, injectable, and self-healing collagen-hyaluronic acid hydrogels loaded with bacterial cellulose and gold nanoparticles for heart tissue engineering. Int. J. Biol. Macromol. 2024, 280, 135749. [Google Scholar] [CrossRef]
- Kiliç, S.C.; Özdal, M.; Kiliç, N.; Ceylan, O.; Koç, G. Dispersed bacterial cellulose (DBC) alone graft material and adding DBC or the boric acid-absorbed DBC to xenograft increases the new bone formation during rabbit maxillary sinus augmentation- a pilot study. J. Stomatol. Oral Maxillofac. Surg. 2025, 126, 102321. [Google Scholar] [CrossRef]
- Singh, J.; Steele, T.W.J.; Lim, S. Fibrillated bacterial cellulose liquid carbene bioadhesives for mimicking and bonding oral cavity surfaces. J. Mater. Chem. B 2022, 10, 2570–2583. [Google Scholar] [CrossRef]
- Maia, A.L.; Lins, E.M.; Rocha, F.A.; Mahnke, L.C.; Pinto, F.C.M.; De Oliveira Pereira, T.; Neves, M.V.; Palácio, S.B.; Hodel, K.V.S.; Barbosa, J.D.V.; et al. Application of bacterial cellulose film as a wound dressing in varicose vein surgery: A randomized clinical trial. J. Vasc. Surg. Venous Lymphat. Disord. 2025, 13, 102302. [Google Scholar] [CrossRef]
- Maia, A.L.; Lins, E.M.; Aguiar, J.L.A.; Pinto, F.C.M.; Rocha, F.A.; Batista, L.L.; Fernandes, W.R.D.M.A. Bacterial cellulose biopolymer film and gel dressing for the treatment of ischemic wounds after lower limb revascularization. Rev. Col. Bras. Cir. 2019, 46, e20192260. [Google Scholar] [CrossRef]
- Stanescu, P.-O.; Radu, I.-C.; Alexa, R.L.; Hudita, A.; Tanasa, E.; Ghitman, J.; Stoian, O.; Tsatsakis, A.; Ginghina, O.; Zaharia, C.; et al. Novel chitosan and bacterial cellulose biocomposites tailored with polymeric nanoparticles for modern wound dressing development. Drug Deliv. 2021, 28, 1932–1950. [Google Scholar] [CrossRef]
- Tang, S.; Chi, K.; Xu, H.; Yong, Q.; Yang, J.; Catchmark, J.M. A covalently cross-linked hyaluronic acid/bacterial cellulose composite hydrogel for potential biological applications. Carbohydr. Polym. 2021, 252, 117123. [Google Scholar] [CrossRef]
- Qian, H.; Liu, J.; Wang, X.; Pei, W.; Fu, C.; Ma, M.; Huang, C. The state-of-the-art application of functional bacterial cellulose-based materials in biomedical fields. Carbohydr. Polym. 2023, 300, 120252. [Google Scholar] [CrossRef]
- Gatenholm, P.; Klemm, D. Bacterial nanocellulose as a renewable material for biomedical applications. MRS Bull. 2010, 35, 208–213. [Google Scholar] [CrossRef]
- Jankau, J.; Błażyńska-Spychalska, A.; Kubiak, K.; Jędrzejczak-Krzepkowska, M.; Pankiewicz, T.; Ludwicka, K.; Dettlaff, A.; Pęksa, R. Bacterial cellulose properties fulfilling requirements for a biomaterial of choice in reconstructive surgery and wound healing. Front. Bioeng. Biotechnol. 2022, 9, 805053. [Google Scholar] [CrossRef]
- Liu, G.; Zou, F.; He, W.; Li, J.; Xie, Y.; Ma, M.; Zheng, Y. The controlled degradation of bacterial cellulose in simulated physiological environment by immobilization and release of cellulase. Carbohydr. Polym. 2023, 314, 120906. [Google Scholar] [CrossRef]
- Avery, D.; Morandini, L.; Celt, N.; Bergey, L.; Simmons, J.; Martin, R.K.; Donahue, H.J.; Olivares-Navarrete, R. Immune cell response to orthopedic and craniofacial biomaterials depends on biomaterial composition. Acta Biomater. 2023, 161, 285–297. [Google Scholar] [CrossRef]
- Kiziloz, S.; Ward, E.J.; Hawthorne, D.; Sinha, A.; Cooksley, G.; Sarker, D.; Crua, C.; Lloyd, A.; Shuck, C.E.; Gogotsi, Y.; et al. Ti3C2TX mxene augments osmo-adaptive repression of the inflammatory stress response for improved wound repair. Nanoscale 2025, 17, 12758–12774. [Google Scholar] [CrossRef]
- Mauney, J.R.; Adam, R.M. Dynamic reciprocity in cell-scaffold interactions. Adv. Drug Deliv. Rev. 2015, 82–83, 77–85. [Google Scholar] [CrossRef]
- Öz, Y.E.; Bingül, N.D.; Morçimen, Z.G.; Şendemir, A.; Hameş, E.E. Fabrication of porous bone scaffolds using degradable and mouldable bacterial cellulose. Cellulose 2024, 31, 2921–2935. [Google Scholar] [CrossRef]
- Potthast, A.; Kostic, M.; Schiehser, S.; Kosma, P.; Rosenau, T. Studies on oxidative modifications of cellulose in the periodate system: Molecular weight distribution and carbonyl group profiles. Holzforschung 2007, 61, 662–667. [Google Scholar] [CrossRef]
- Busuioc, C.; Isopencu, G.; Banciu, A.; Banciu, D.D.; Oprea, O.; Mocanu, A.; Deleanu, I.; Zaulet, M.; Popescu, L.; Tanasuica, R.; et al. Bacterial cellulose hybrid composites with calcium phosphate for bone tissue regeneration. Int. J. Mol. Sci. 2022, 23, 16180. [Google Scholar] [CrossRef]
- Rogovina, S.; Aleksanyan, K.; Prut, E.; Gorenberg, A. Biodegradable blends of cellulose with synthetic polymers and some other polysaccharides. Eur. Polym. J. 2013, 49, 194–202. [Google Scholar] [CrossRef]
- Mndlovu, H.; Kumar, P.; Toit, L.C.D.; Choonara, Y.E. A review of biomaterial degradation assessment approaches employed in the biomedical field. npj Mater. Degrad. 2024, 8, 66. [Google Scholar] [CrossRef]
- Rzhepakovsky, I.; Piskov, S.; Avanesyan, S.; Sizonenko, M.; Timchenko, L.; Anfinogenova, O.; Nagdalian, A.; Blinov, A.; Denisova, E.; Kochergin, S.; et al. Composite of bacterial cellulose and gelatin: A versatile biocompatible scaffold for tissue engineering. Int. J. Biol. Macromol. 2024, 256, 128369. [Google Scholar] [CrossRef]
- Lee, J.; Kim, H.J.; Song, M.; Kim, M.; Eom, Y.; Lee, Y.; Lee, H. Membrane emulsification-derived cellulose microbeads encapsulating vitamin C with pH-sensitive release via chitosan nanowhisker coating. Carbohydr. Polym. 2026, 372, 124526. [Google Scholar] [CrossRef]
- Bappy, M.M.; Van Epps, E.; Priddy, L.B.; Tian, W. Parameter optimization for accurate and repeatable strut width in the 3D printing of composite bone scaffolds. J. Manuf. Process. 2024, 131, 1631–1641. [Google Scholar] [CrossRef]
- Lee, J.; Liao, H.; Wang, Q.; Han, J.; Han, J.H.; Shin, H.E.; Ge, M.; Park, W.; Li, F. Exploration of nanozymes in viral diagnosis and therapy. Exploration 2022, 2, 20210086. [Google Scholar] [CrossRef]
- Patel, D.K.; Ganguly, K.; Dutta, S.D.; Patil, T.V.; Lim, K.-T. Cellulose nanocrystals vs. cellulose nanospheres: A comparative study of cytotoxicity and macrophage polarization potential. Carbohydr. Polym. 2023, 303, 120464. [Google Scholar] [CrossRef]
- Shi, X.; Zhang, J.; Guan, W.; Li, C.; Chen, W.; Yu, G.; Yu, H. Sustainable synthesis of amino-cellulose nanofibers for biomaterial platforms. Sci. Adv. 2025, 11, eadx4556. [Google Scholar] [CrossRef]
- Christo, S.N.; Bachhuka, A.; Diener, K.R.; Mierczynska, A.; Hayball, J.D.; Vasilev, K. The role of surface nanotopography and chemistry on primary neutrophil and macrophage cellular responses. Adv. Healthc. Mater. 2016, 5, 956–965. [Google Scholar] [CrossRef]
- Vidakis, N.; Petousis, M.; Mountakis, N.; Spyridaki, M.; Gkagkanatsiou, K.; Stratakis, E. Optimization of main process control parameters of PHA bio-polymer in material extrusion additive manufacturing: Experimental design and predictive models. Int. J. Adv. Manuf. Technol. 2025, 139, 1607–1623. [Google Scholar] [CrossRef]
- Yong, X.; Liu, Y.-X.; Chaparro, F.J.; Tian, Z.; Jia, Y.; Gosser, J.; Gaumer, J.; Ross, L.; Tafreshi, H.; Lannutti, J.J. Visualization of porosity and pore size gradients in electrospun scaffolds using laser metrology. PLoS ONE 2023, 18, e0282903. [Google Scholar]
- Sivan, M.; Madheswaran, D.; Hauzerova, S.; Novotny, V.; Hedvicakova, V.; Jencova, V.; Kostakova, E.K.; Schindler, M.; Lukas, D. AC electrospinning: Impact of high voltage and solvent on the electrospinnability and productivity of polycaprolactone electrospun nanofibrous scaffolds. Mater. Today Chem. 2022, 26, 101025. [Google Scholar] [CrossRef]
- Bliley, J.M.; Shiwarski, D.J.; Feinberg, A.W. 3D-bioprinted human tissue and the path toward clinical translation. Sci. Transl. Med. 2022, 14, eabo7047. [Google Scholar] [CrossRef]
- Furlan, L.; Zamuner, A.; Riccioni, A.; Sabbadin, G.; Russo, T.; Gallicchio, V.; D’auria, G.; Falcigno, L.; Manni, L.; Ballarin, L.; et al. Comparative analysis of tunicate vs. plant-based cellulose in chitosan hydrogels for bone regeneration. Gels 2025, 11, 102. [Google Scholar] [CrossRef]
- Huang, J.H.; Huang, Z.W.; Xiong, J.Y.; Xia, J.; Wang, Y.Q.; Yang, L.; Liang, Y.J. 3D bioprinted scaffolds of polysaccharide hydrogels in osteochondral and cartilage tissue engineering. Des. Monomers Polym. 2023, 26, 258–272. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, S.; Gu, Z.; Zhao, Y. A bibliometric analysis: Research progress and prospects on transition metal dichalcogenides in the biomedical field. Chin. Chem. Lett. 2021, 32, 3762–3770. [Google Scholar] [CrossRef]
- Mahmud, M.A.P.; Tat, T.; Xiao, X.; Adhikary, P.; Chen, J. Advances in 4d-printed physiological monitoring sensors. Exploration 2021, 1, 20210033. [Google Scholar] [CrossRef]
- Dusim, G.A.H.; Muhamad, F.; Lai, K.W. Enhancing calcium phosphate cements: A review of bacterial cellulose (bc) and other biopolymer reinforcements for biomedical applications. Biomater. Adv. 2025, 172, 214245. [Google Scholar] [CrossRef]
- Sekar, M.P.; Budharaju, H.; Zennifer, A.; Sethuraman, S.; Vermeulen, N.; Sundaramurthi, D.; Kalaskar, D.M. Current standards and ethical landscape of engineered tissues-3D bioprinting perspective. J. Tissue Eng. 2021, 12, 20417314211027677. [Google Scholar] [CrossRef]
- Armengol, E.S.; Hock, N.; Saribal, S.; To, D.; Summonte, S.; Veider, F.; Kali, G.; Bernkop-Schnuerch, A.; Laffleur, F. Unveiling the potential of biomaterials and their synergistic fusion in tissue engineering. Eur. J. Pharm. Sci. 2024, 196, 106761. [Google Scholar] [CrossRef]
- Behbehani, M.; Glen, A.; Taylor, C.S.; Schuhmacher, A.; Claeyssens, F.; Haycock, J.W. Pre-clinical evaluation of advanced nerve guide conduits using a novel 3D in vitro testing model. Int. J. Bioprint. 2024, 4, 123. [Google Scholar] [CrossRef]
- Deumens, R.; Bozkurt, A.; Meek, M.F.; Marcus, M.A.E.; Joosten, E.A.J.; Weis, J.; Brook, G.A. Repairing injured peripheral nerves: Bridging the gap. Prog. Neurobiol. 2010, 92, 245–276. [Google Scholar] [CrossRef]
- Wang, D.; Marom, N.; Coxe, F.R.; Kalia, V.; Burge, A.J.; Jones, K.J.; Rodeo, S.A.; Williams, R.J. Preoperative grades of osteoarthritis and meniscus volume correlate with clinical outcomes of osteochondral graft treatment for cartilage defects in the knee. Cartilage 2019, 12, 344–353. [Google Scholar] [CrossRef]










| Cellulose Source/Type | Functionalization Strategy | Fabrication Technique | Key Structural/ Properties | Tissue Regeneration | Ref. |
|---|---|---|---|---|---|
| TEMPO-CNCs/Carbon Dots | Fluorescent functionalization (for tracking) | DLP 3D printing | Photocurable resin, precise microstructures, fluorescence | Skin and vascular regeneration | [71] |
| Carboxylated CNCs | Nano-reinforcement and crosslinking site | Extrusion 3D printing | Enhanced shear-thinning, mechanical strength, shape fidelity | General tissue engineering | [43] |
| Bacterial Cellulose (BC) | Oxidation, cationic PU micelle and CD grafting | In situ biosynthesis + chemical crosslinking | Dual-network, sustained drug release, antibacterial, cytokine recruitment | Urethral regeneration | [70] |
| Cellulose Nanofibers (CNFs) | Phosphorylation | Casting | Reduced fiber size/crystallinity, tuned wettability for protein/cell adhesion | Dental pulp stem cell culture | [9] |
| Gelatin/Nanocellulose/nHA/Simvastatin | Composite formulation and drug loading | Freeze-drying + chemical crosslinking (glutaraldehyde) | Highly porous sponge, osteoconductive and osteoinductive | Bone regeneration | [17] |
| TOBC (m-TOBC)/GelMA/DMOG-loaded MSNs | Enzyme mineralization and bioactive loading | Extrusion 3D printing | Biomimicked bone ECM, improved rheology/mechanics, angiogenic drug release | Bone regeneration | [38] |
| CNF/Mg-Fe LDH/RA and SHH | Composite formulation and growth factor loading | Anisotropic freeze-drying | Aligned microchannels, sustained factor release | Neural regeneration | [20] |
| TOCNF/rGO (PEI-modified) | Composite formulation and surface charge modification | Extrusion 3D printing | Conductive, good printability and mechanical recovery, supports neural cell growth | Neural regeneration | [11] |
| BC/PCL | Polymer blending | Electrospinning | Nanofibrous mesh with hollow microbeads, enhanced cell adhesion/proliferation | Neural regeneration | [88] |
| BC/Poly(3,4-ethylenedioxythiophene)-SNFs | Sulfonation and in situ polymerization | In situ assembly + rolling/coating | Conductive composite membrane, layered structure, suitable for nerve conduits | Peripheral nerve repair | [22] |
| CMC/CMCS/Gelatin/Algal Extracts | Composite formulation and bioactive incorporation | Freeze-drying + chemical crosslinking (EDAC) | Porous hydrogel sponge, enhanced wound healing properties | Skin wound healing | [55] |
| Cellulose Acetate (CA) | N-halamine compound grafting (MDI) | Electrospinning | Antimicrobial nanofibrous mats, durable, non-leaching | Antimicrobial wound dressing | [44] |
| PLA/CNF (from Pennisetum purpureum) | Composite formulation | Solvent casting and salt leaching | Highly porous (>76%), improved compressive strength and hydrophilicity | Bone regeneration | [12] |
| PHB/Chitosan/CNC | Composite formulation | Electrospinning | Enhanced tensile strength/modulus; Osteogenic marker upregulation | Bone regeneration | [58] |
| BC/BAM (Bladder Acellular Matrix) | Composite formulation | Freeze-drying + chemical crosslinking (EDC/NHS) | Biomimetic composition (collagen, GAGs, VEGF), enhanced mechanical strength | Urethral regeneration/Angiogenesis | [95] |
| Oxidized BC (OBC)/Soy Protein Isolate (SPI) | Selective oxidation and protein composite | Laser perforation + oxidation + compositing | Improved biocompatibility, cell guidance, degradability | Urethral regeneration | [116] |
| Scaffold Composition | Target Tissue | Mechanical Property | Porosity (%) | Pore Size (μm) | Degradation Time | Performance Highlight | Ref. |
|---|---|---|---|---|---|---|---|
| Gelatin, Bacterial Nanocellulose, Nanohydroxyapatite, Simvastatin | Bone tissue | NR | NR | BNC-G group: 211–244 μm average; No pores > 300 μm | ~21% mass loss in 4 weeks for BNC-G-nHA-Sim scaffold (in vitro, PBS) | Excellent biocompatibility; Significantly enhanced osteogenic differentiation of BMSCs; Sustained simvastatin release over 216 h. | [17] |
| GelMA/m-TOBC/DMSN hydrogel | Bone tissue | Compressive modulus: ~15.6 Kpa; Enhanced storage modulus (G’) | NR | NR | ~20% mass loss after 7 days (in vitro, collagenase II) | Biomimetic bone ECM; Improved printability and mechanics; Synergistic release of osteogenic ions (Ca2+, PO43−) and angiogenic drug (DMOG). | [38] |
| PHB-chitosan/CNC (3 wt% CNC) | Bone tissue | Tensile strength: 4.52 MPa; Modulus: 130.29 MPa | NR | NR | ~35% weight loss after 100 days (PBS) | Significant osteogenic gene upregulation (OPN: 10.7-fold, ALP: 4.1-fold); High cell viability (91.5%); Excellent bioactivity (Ca/P ratio ~ 1.73); Enhanced hydrophilicity and surface roughness. | [58] |
| GelMA, PEGDA, T-CNC@CDs, LAP | Skin, Blood vessel, Muscle | Elastic modulus: ~13 ± 4.2 Kpa (for GPCD hydrogel) | NR | GPCD: 166 ± 20 μm; GM: 82 ± 13 μm; GPD: 40–120 ± 10 μm. | Short-term: ~35–48% weight loss in 5 h (PBS/trypsin) | >95% cell viability; Upregulated gene expression (COL1A: 6.74-fold, KRT1: 4.27-fold); 30-day cell tracking; Stable fluorescence (pH 6.5–9.5); High-resolution DLP printability. | [71] |
| CMC/CMCS/Gelatin hydrogel loaded with 1% Arthrospira platensis (AP) and/or 1% Chlorella vulgaris (CV) extracts. | Skin (Wound healing) | NR | NR | ~37–105 μm (AP reduced pore size more than CV). | ~24–48 h (in PBS; AP/CV groups dissolved within 24 h, base hydrogel degraded 32% in 48 h). | Synergistic effects; Highest wound closure rate (92%) at 14 days in a rat model. | [55] |
| CNF/Chitosan aerogels (CNF1/2 with Ch) | Skin (Wound healing) | NR | pore volume 0.122–0.357 cm3/g | ~0.003 μm (3.0–3.5 nm) | NR | High biocompatibility and strong antibacterial activity against S. aureus and E. coli. | [125] |
| Bilayer: RC/Quaternized CS base layer + Collagen/HA top layer. | Skin (Wound healing) | Ultimate tensile strength (UTS): 2.29 MPa (for crosslinked bilayer scaffold). | >80% | NR | Degradation observed over 10 days in mild acidic pH (4.5–5.6) | Antibacterial (vs. E. coli); Enhanced angiogenesis (9.76-fold increase in VEGF-A gene expression); Promoted cell migration (100% scratch closure); Stimulated collagen secretion. | [126] |
| TOCNF/rGO (PEI-modified) | Neural tissue | 379.94–1267.28 Kpa | NR | NR | 14 days (mass loss decreased with rGO content) | >90% printing fidelity; Conductive (up to 0.21 S/m); Supports astrocyte viability and alignment. | [11] |
| CNF/Mg–Fe LDH loaded with RA/SHH | Neural tissue | Similar modulus to skin tissue (for CNF film); Aligned microchannel structure provides topological guidance | NR | Homogeneously distributed pores; Aligned microchannels present | NR | Promotes aligned axonal growth; Enhances neuron/oligodendrocyte differentiation; Suppresses astrocytes; Restores motor function and electrophysiology via RhoA/Rock/Myosin II pathway. | [20] |
| BC/PCL (50:50 wt.%) blend, electrospun | Neural tissue | Max. tensile strength ~30 MPa (vs. ~14.6 MPa for pure PCL) | NR | Fiber diameter: 70–120 nm; Hollow beads: 100 nm–1.6 μm | NR | Enhanced fibroblast adhesion/proliferation vs. pure PCL; Supported DRG neurite outgrowth and alignment. | [88] |
| BC/PEDOT-SNFs (BPS) composite membrane | Peripheral nerve | Tensile strength: 5.80 ± 0.14 MPa (BPS 10–10); Young’s modulus: ~0.33 MPa | ~97% | NR | NR | High conductivity (up to 10−2 S/cm); Excellent biocompatibility; Promotes ADSCs adhesion; Improves peripheral nerve regeneration in vivo. | [22] |
| BC/BAM composite | Urethral tissue | Compressive stress: 33 → 74 Kpa; Young’s modulus: 12 → 90 Kpa | >85% | 187 ± 59 μm (BC0.5/BAM0.5) | NR | Promoted angiogenesis and epithelialization; Achieved stricture-free repair in rabbit model. | [95] |
| DMBC/SPI (Double-Modified BC with Soy Protein Isolate) | Urethral tissue | ~1.58 ± 0.12 MPa (DMBC/SPI1) | NR | 200–300 μm (laser-drilled macropores) | ~60 days (50.03% mass loss in PBS) | Promotes aligned cell growth; Supports smooth urethral regeneration with mild inflammation. | [116] |
| MCC scaffold embedding PLGA/KGN microspheres (MCC/PKGN) | Cartilage | 4.53 MPa | NR | 200–500 μm | Partial degradation (7.73% at 8 weeks in vitro) | Sustained KGN release promotes BMSC migration; Chondrogenic differentiation; Enhanced cartilage regeneration in vivo. | [123] |
| EDC/NHS-crosslinked macroporous bacterial cellulose (cMP-BC) | Cartilage | Enhanced compressive strength and shape recovery in wet state; Stable 3D network in aqueous environment | NR | 64–195 μm (tunable via BC concentration) | NR | Excellent biocompatibility; Promoted chondrocyte infiltration and cartilage-specific ECM deposition; Mechanically robust and shape-recoverable. | [127] |
| Target Tissue | Core Functional Requirements | Representative Cellulose Scaffold Design | Key Design Rationale and Features | Ref. |
|---|---|---|---|---|
| Bone | Mechanical support and load-bearing; Osteoconductivity and Osteoinductivity; Vascularization support | Composite Scaffold (Gelatin/Nanocellulose/nHA/Simvastatin) | Composite formulation and drug loading: nHA provides osteoconduction; Nanocellulose reinforces; Simvastatin release induces osteogenesis. | [17] |
| Mineralized TOBC/GelMA/DMOG-loaded MSNs | Bioactive mineralization and angiogenic loading: TOBC reinforces and mimics bone ECM; In situ mineralization enhances mechanics; DMOG delivery promotes vascularization. | [38] | ||
| Skin Wound | Moisture management and exudate absorption; Barrier against infection; Promotion of re-epithelialization and angiogenesis | Bilayer RC/Quaternized CS-HA/Collagen Scaffold | Bilayer structure and multifunctional composite: Dense RC/CS layer provides antibacterial barrier; Porous Collagen/HA layer promotes cell migration. | [126] |
| CMC/CMCS/Gelatin/Algal Extracts | Composite hydrogel and bioactive incorporation: Polysaccharide matrix ensures high absorbency; Natural extracts enhance healing. | [55] | ||
| TEMPO-CNF-based Bioaerogels (with Chitosan, Gelatin, Alginate) | Tunable absorption and antibacterial activity: High porosity and carboxyl groups enable exceptional fluid uptake (WAV); CNF/Chitosan composites show strong antibacterial effects. | [125] | ||
| Neural Tissue | Topographical guidance for axonal growth; Support for Schwann cell activity; Neurotrophic factor delivery | CNF/Mg-Fe LDH/RA and SHH Scaffold | Anisotropic structure and growth factor loading: Aligned microchannels direct neurite extension; LDH enables sustained factor release; | [20] |
| BC/PCL (50:50 wt.%) nanofibrous scaffold with embedded hollow micro/nanobeads | Anisotropic fibers guide neurite growth. Hollow beads are drug-loadable; BC provides hydrophilicity and biocompatibility; PCL enhances mechanical strength; Blend enhances biocompatibility and neural growth. | [88] | ||
| Cartilage | Compressive strength and elasticity; Support for chondrogenic phenotype; Lubrication and integration | MCC Scaffold embedding PLGA/KGN Microspheres | Controlled drug delivery: MCC provides support; PLGA microspheres enable sustained release of chondroinductive agent (KGN). | [123] |
| Macroporous BC Scaffold (MP-BC) | Macroporosity and mechanical robustness: Crosslinking enhances compressive strength and shape recovery; Interconnected macropores facilitate cell infiltration and ECM deposition. | [127] | ||
| Cardiovascular (Vascular Graft) | Suture retention and burst pressure; Anti-thrombogenicity; Compliance matching native vessel | BC/Bladder Acellular Matrix (BAM) | Biomimetic composite: BC provides strength; BAM provides natural ECM to enhance endothelialization. | [95] |
| In situ Biosynthesized BC Tubular Graft (BASYC®) | Seamless nanofibrous structure: Native BC network provides high wet strength and a smooth, low-thrombogenicity lumen. | [224] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tong, Y.; Cai, Y.; Wu, Y.; Zhuo, W.; Liao, J. From Design to Application: Advanced Cellulose Scaffolds for Engineered Tissue Regeneration. Polymers 2026, 18, 614. https://doi.org/10.3390/polym18050614
Tong Y, Cai Y, Wu Y, Zhuo W, Liao J. From Design to Application: Advanced Cellulose Scaffolds for Engineered Tissue Regeneration. Polymers. 2026; 18(5):614. https://doi.org/10.3390/polym18050614
Chicago/Turabian StyleTong, Yao, Yong Cai, Yanting Wu, Wenkun Zhuo, and Jinfeng Liao. 2026. "From Design to Application: Advanced Cellulose Scaffolds for Engineered Tissue Regeneration" Polymers 18, no. 5: 614. https://doi.org/10.3390/polym18050614
APA StyleTong, Y., Cai, Y., Wu, Y., Zhuo, W., & Liao, J. (2026). From Design to Application: Advanced Cellulose Scaffolds for Engineered Tissue Regeneration. Polymers, 18(5), 614. https://doi.org/10.3390/polym18050614









