Functionalization of Photopolymer with Laser-Ablated Copper NPs: A Comprehensive Study of ROS Generation, Antimicrobial Activity and Cytotoxic Profile
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Copper Nanoparticles (Cu NPs) Colloid and Incorporation into Photopolymer Resin
2.2. Additive Manufacturing of Specimens from Modified Composite Materials
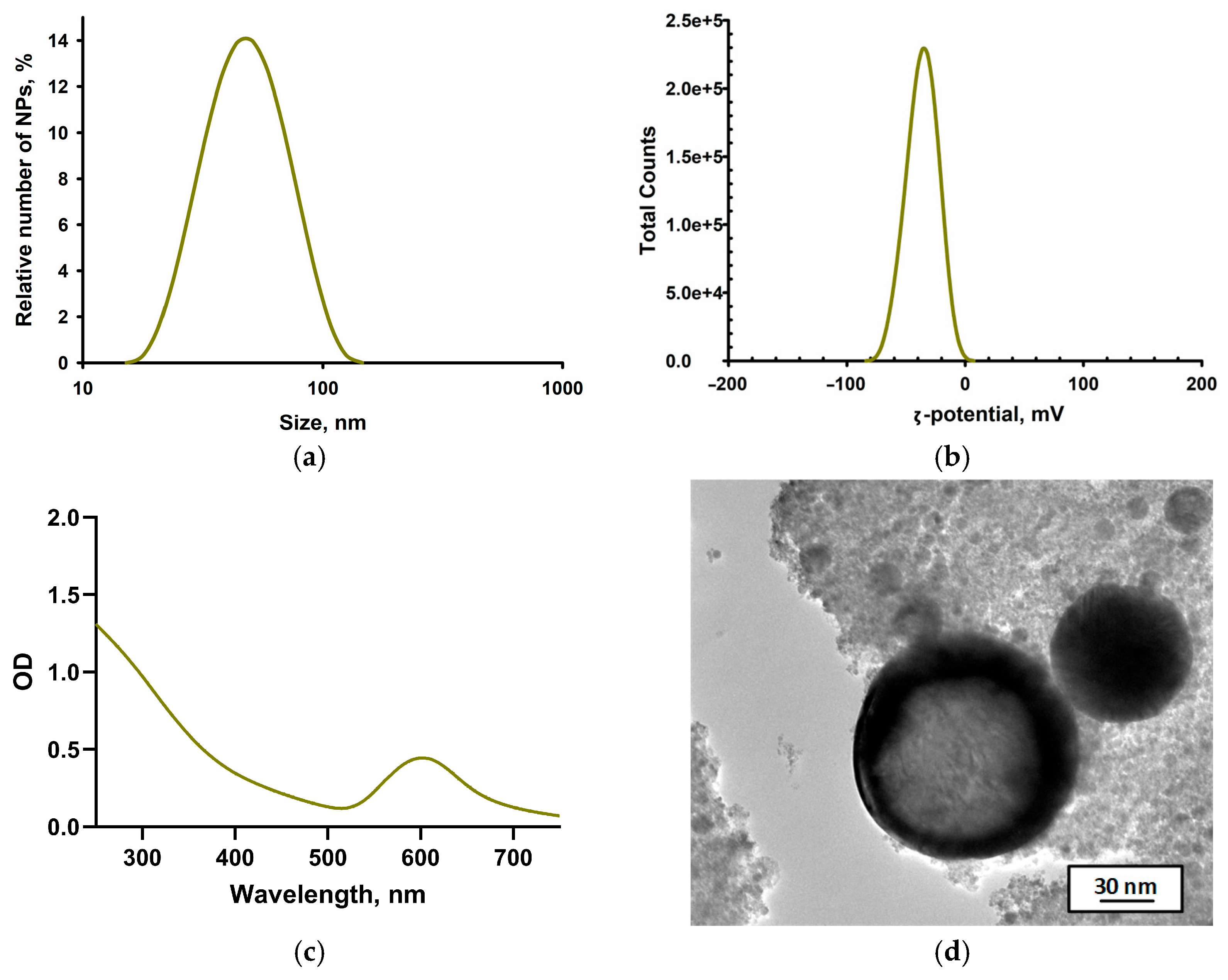
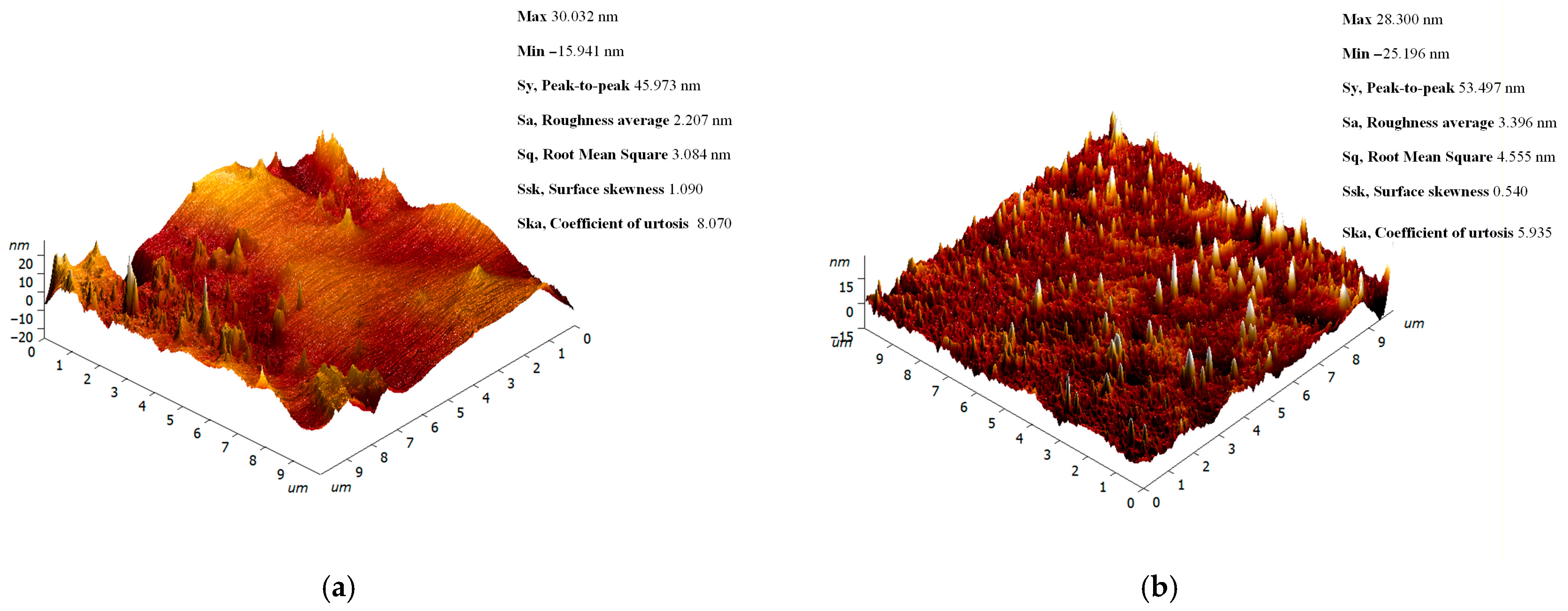
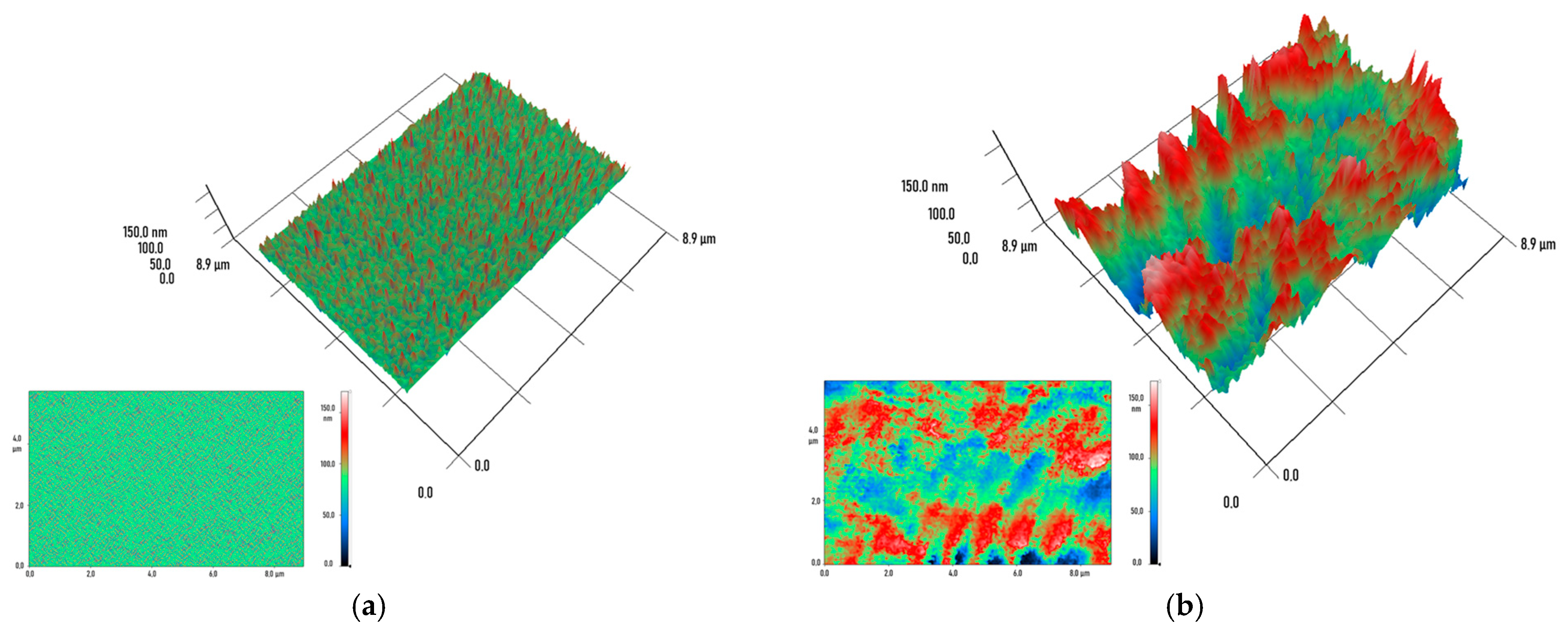
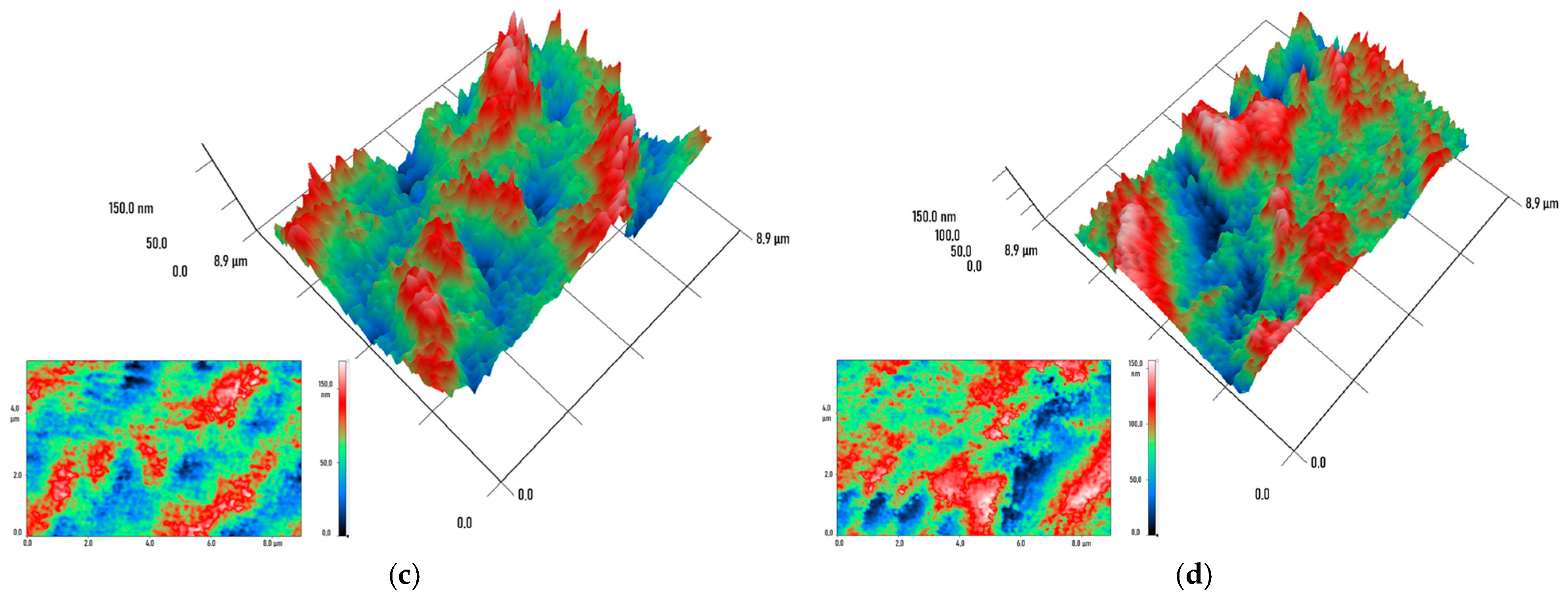
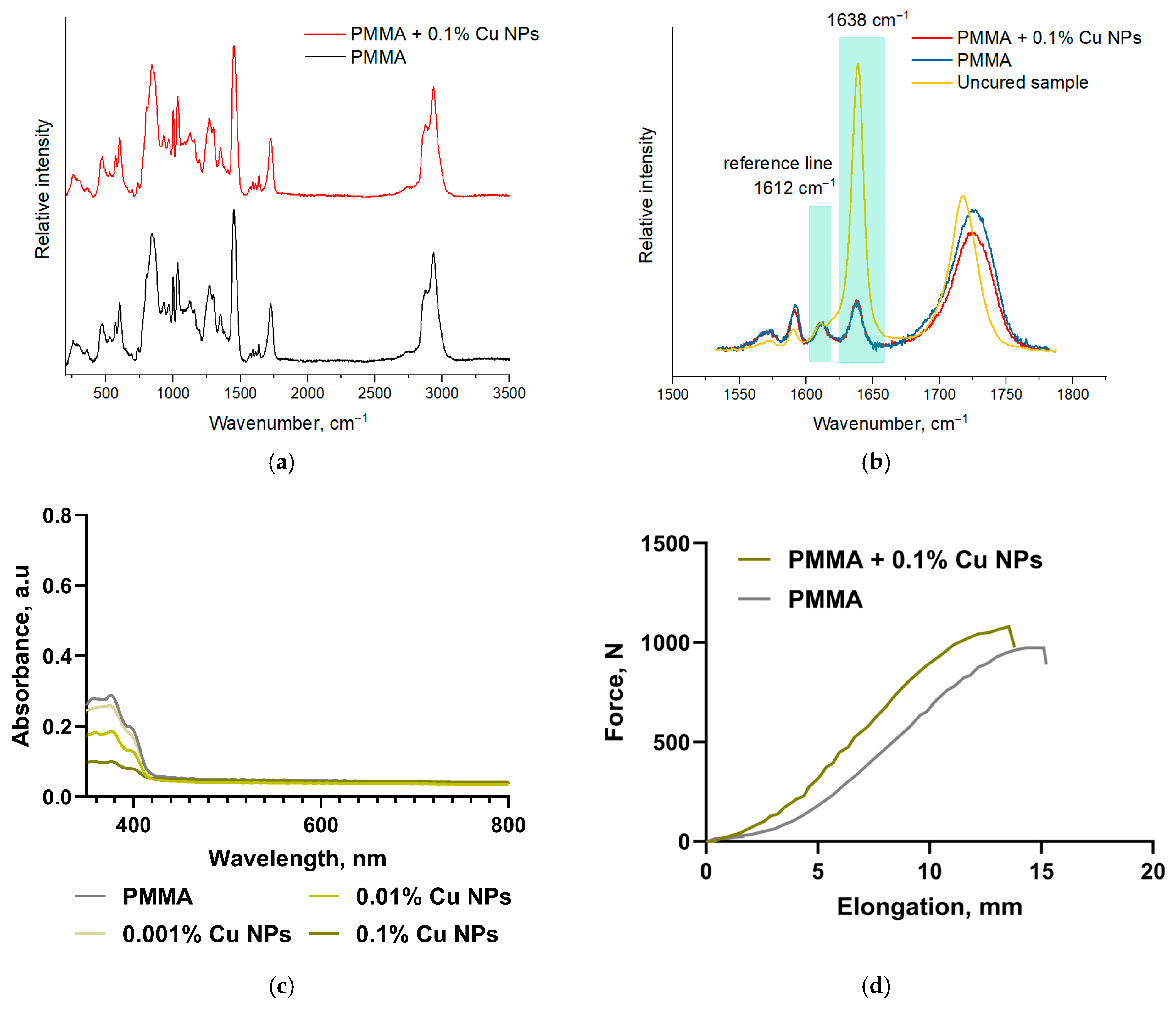
2.3. Physicochemical Characterization of Specimens Printed from Modified Resins
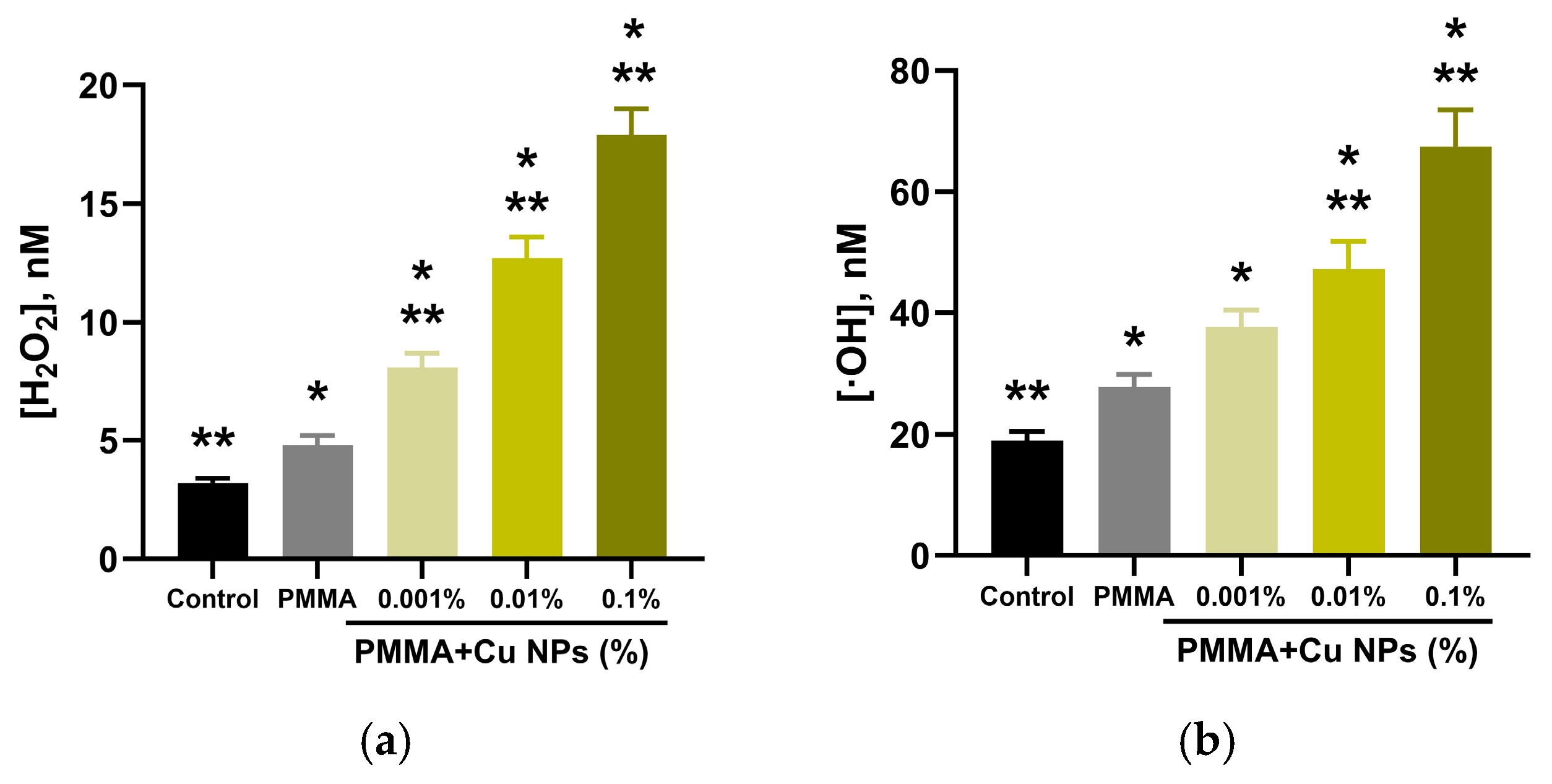
2.4. Quantitative Assessment of Reactive Oxygen Species (ROS) Generation in Aqueous Solutions
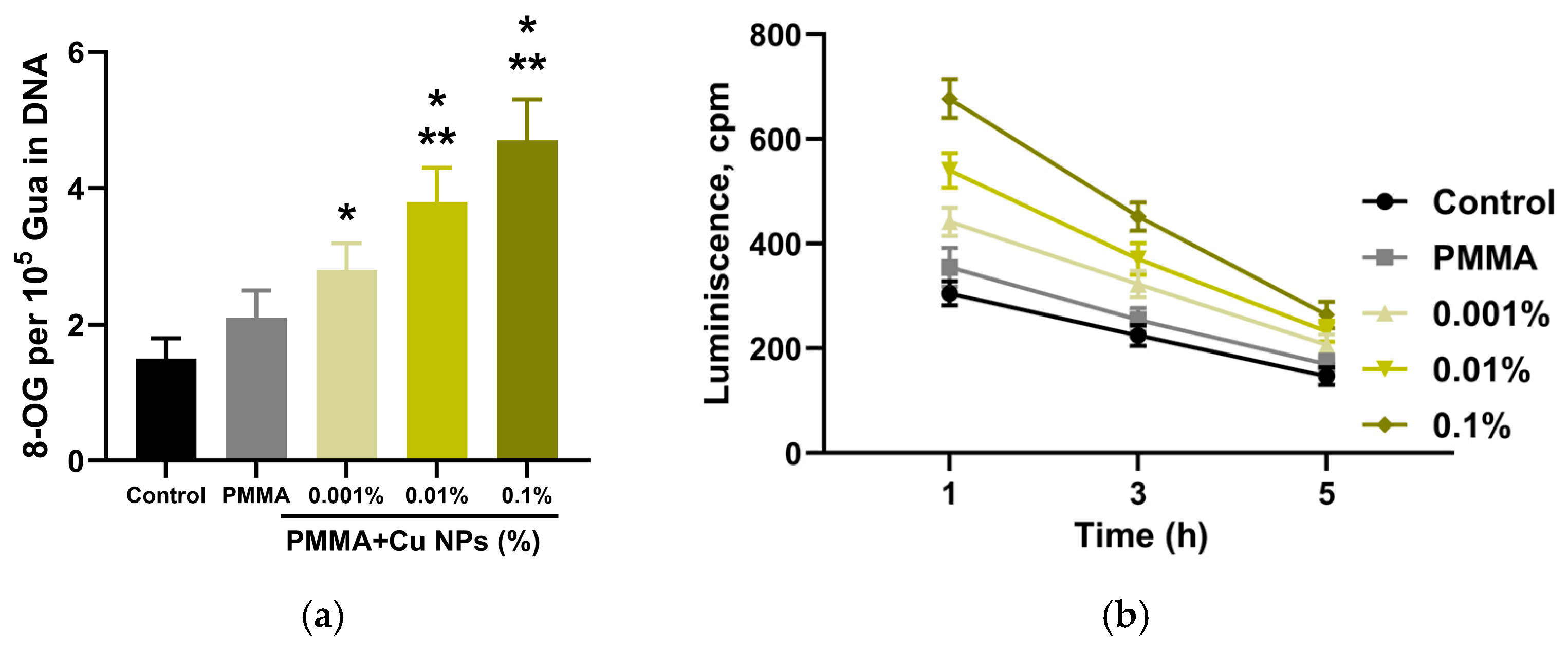
2.5. Quantitative Determination of 8-Oxoguanine in DNA
2.6. Detection of Long-Lived Reactive Protein Species (LRPS)
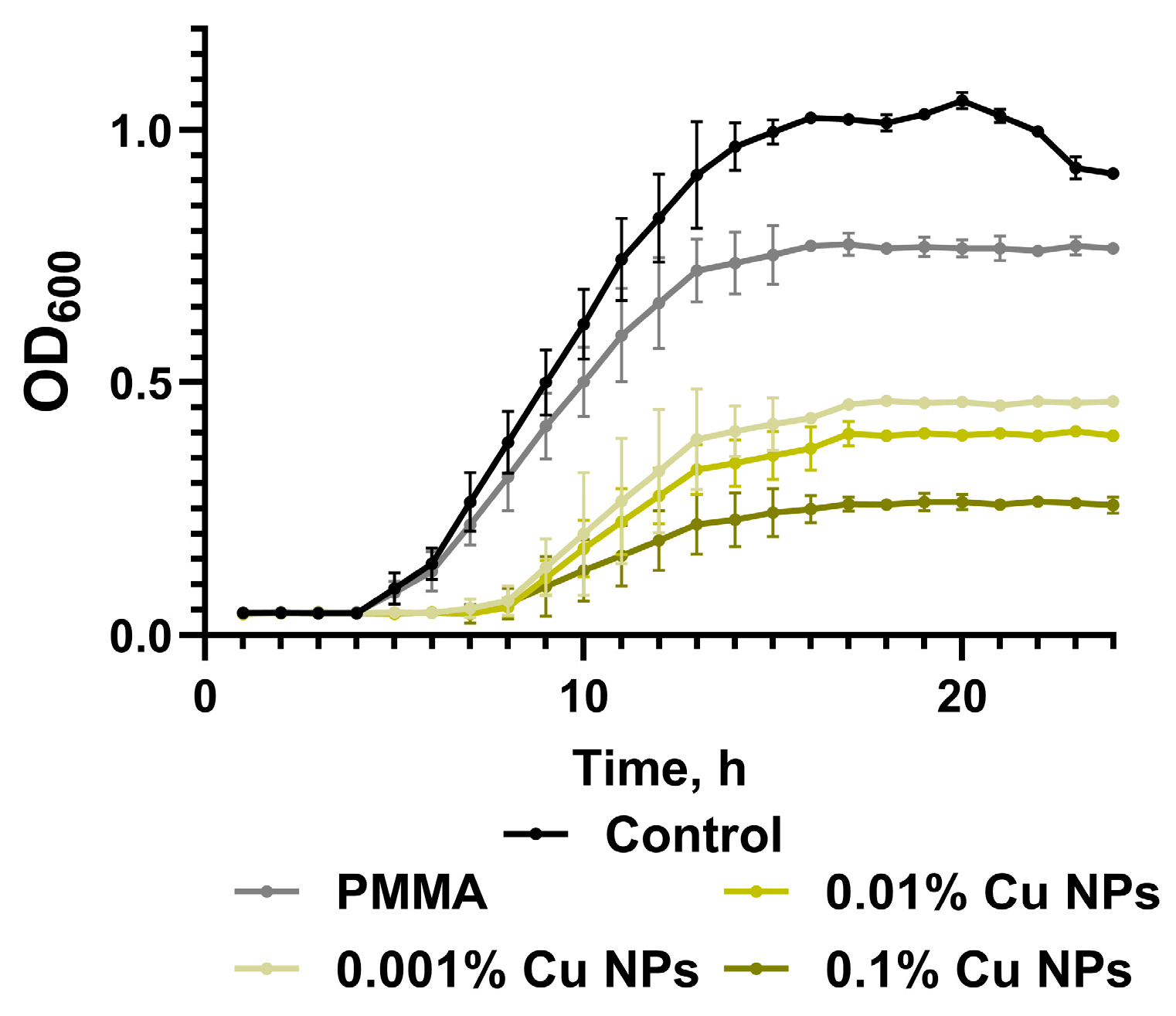
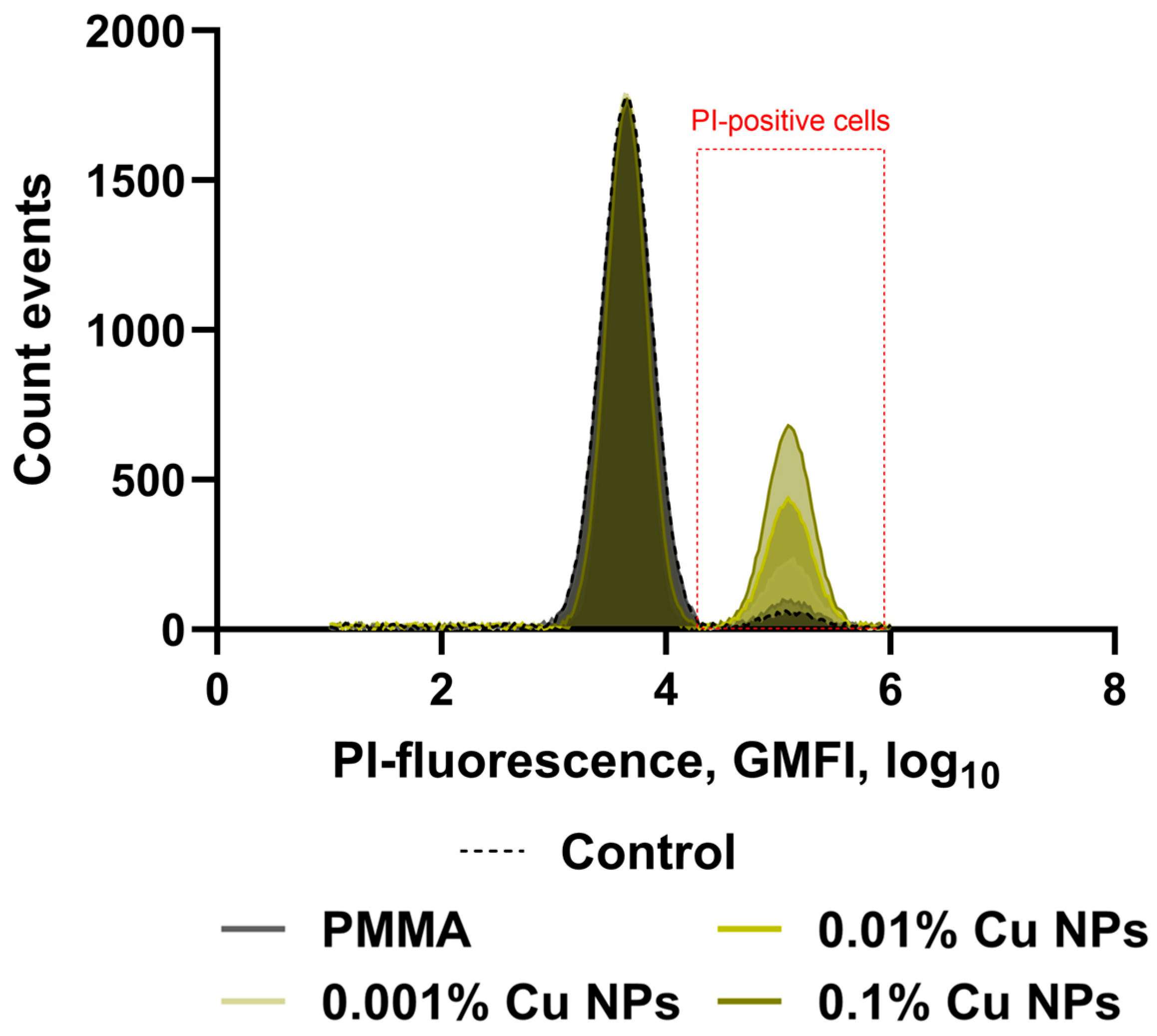
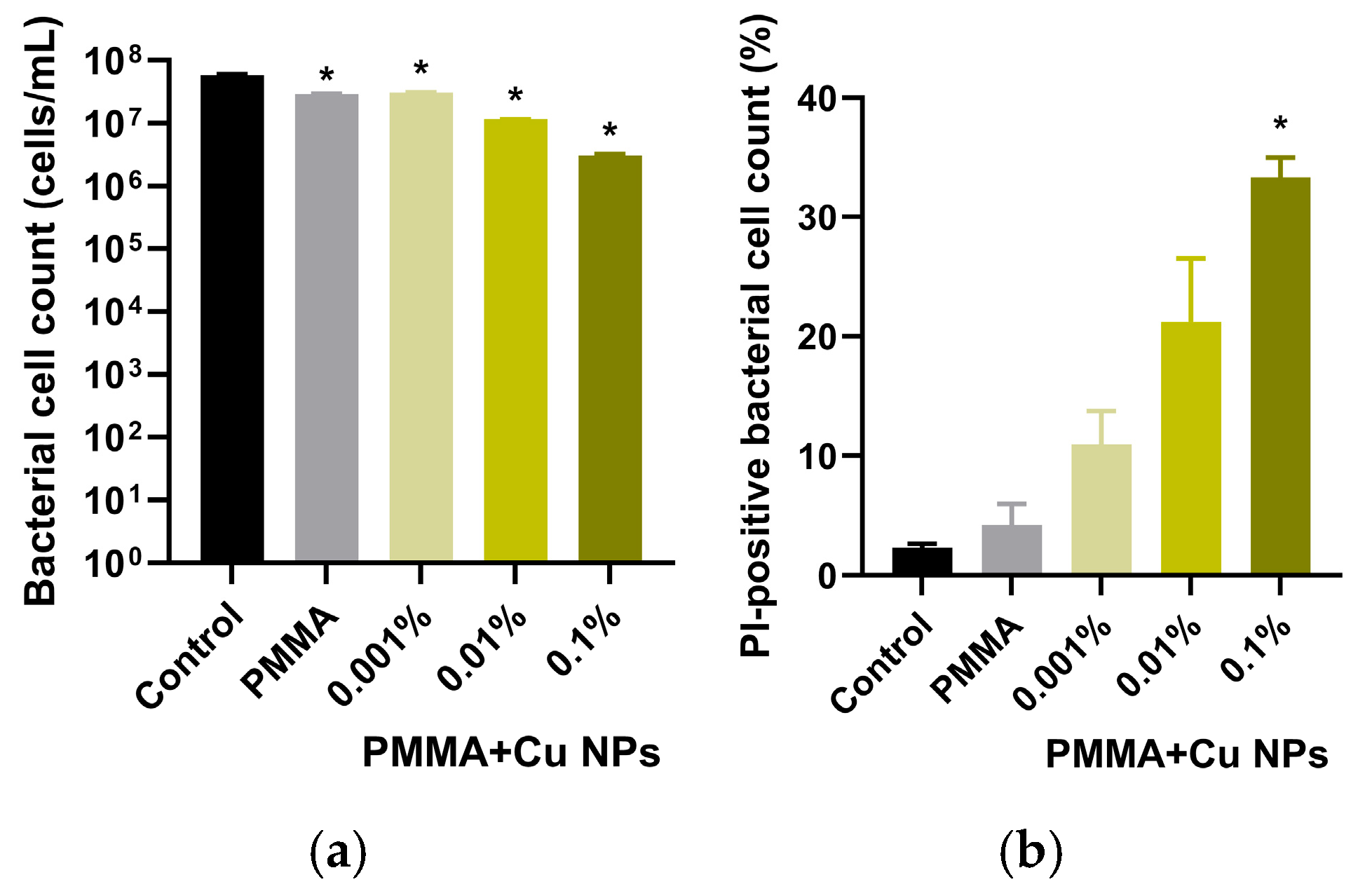
2.7. Microbiological Studies of Antibacterial Activity
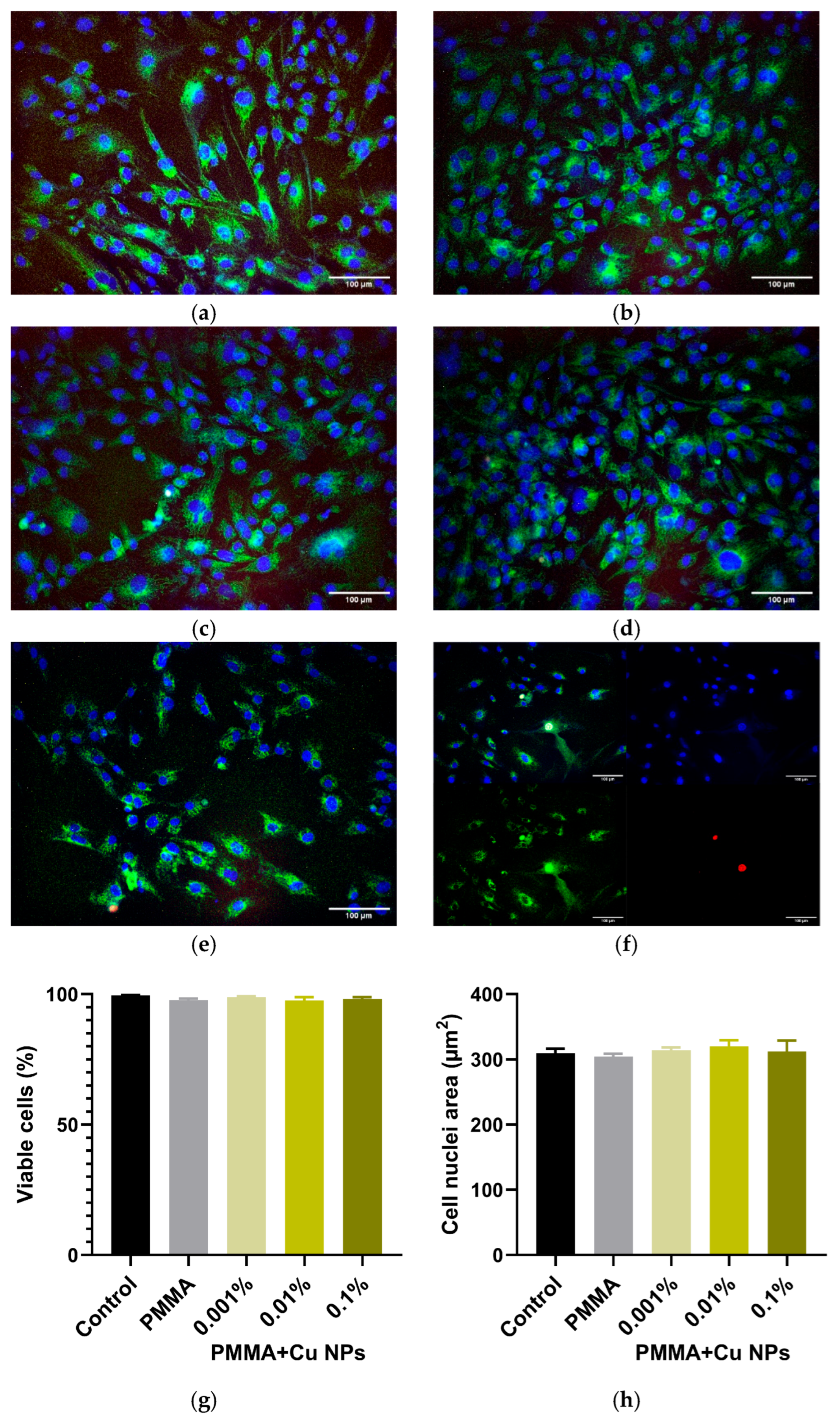
2.8. In Vitro Cytotoxicity Assessment of Composite Materials
2.9. Statistical Processing and Visualization of Experimental Data
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, Y.; Song, X.; Li, X.; Chen, Z.; Zhou, C.; Zhou, Q.; Chen, Y. Recent progress in biomimetic additive manufacturing technology: From materials to functional structures. Adv. Mater. 2018, 30, e1706539. [Google Scholar] [CrossRef]
- Abbasi, M.; Váz, P.; Silva, J.; Martins, P. Head-to-head evaluation of FDM and SLA in additive manufacturing: Performance, cost, and environmental perspectives. Appl. Sci. 2025, 15, 2245. [Google Scholar] [CrossRef]
- Xiao, M.; Zhang, J.; Feng, Y. A review on recent application of acrylate emulsion/resin in cement additives. Polym. Adv. Technol. 2025, 36, 70218. [Google Scholar] [CrossRef]
- Mohd Shuhairi, D.N.; Abdul Malek, N.S.; Mohd Ghazali, M.I.; Md Sin, N.D.; Bonnia, N.N.; Yamada, K.; Rosman, N.; Hajar, N.; Omar, H.; Asli, N.A. Nanofillers in vat photopolymerisation additive manufacturing: Mechanical properties of graphene and acrylate-based composites. Prog. Addit. Manuf. 2025, 10, 11319–11341. [Google Scholar] [CrossRef]
- Natarajan, P.; Kumar, S.M.; Natarajan, S.; Sridharan, D.K.S.; Narayana Kalkura, D.S. Nano-particle coated or impregnated acrylic resins in dental applications: A systematic review of in vivo evidence on mechanical properties, biocompatibility and clinical performance. J. Oral Biol. Craniofacial Res. 2025, 15, 1190–1199. [Google Scholar] [CrossRef]
- Ji, S.; Ren, J.; Zhang, J.; Zhou, W.; Liu, Z. Effects of silanized nano-Al2O3 fillers on the mechanical properties and printing accuracy of photopolymer resins for 3D printing dental applications. Mater. Res. Express 2025, 12, 085401. [Google Scholar] [CrossRef]
- Narongdej, P.; Alterman, N.; Vazquez, M.; Tehrani, M.; Barjasteh, E. Vat photopolymerization (VP) of solvent-free carbon nanoparticle-acrylic nanocomposites. Compos. Part A Appl. Sci. Manuf. 2025, 190, 108628. [Google Scholar] [CrossRef]
- Vakili, N.; Ashengroph, M.; Sharifi, A.; Zorab, M.M. Eco-friendly synthesis of copper nanoparticles by using Ralstonia sp. and their antibacterial, anti-biofilm, and antivirulence activities. Biochem. Biophys. Rep. 2025, 42, 101978. [Google Scholar] [CrossRef]
- Muniasamy, S.; Angelina, M.; Ponmanickam, P.; Al-Ansari, M.M.; Mythili, A.; Dixit, S.; Raguvaran, K. Green synthesis of copper nanoparticles using panchagavya: Nanomaterials for antibacterial, anticancer, and environmental applications. Luminescence 2025, 40, e70117. [Google Scholar] [CrossRef]
- Woźniak-Budych, M.J.; Staszak, K.; Staszak, M. Copper and copper-based nanoparticles in medicine—Perspectives and challenges. Molecules 2023, 28, 6687. [Google Scholar] [CrossRef]
- Mamonova, I.A.; Matasov, M.D.; Babushkina, I.V.; Losev, O.E.; Chebotareva, Y.G.; Gladkova, E.V.; Borodulina, Y.V. Study of physical properties and biological activity of copper nanoparticles. Nanotechnol. Russ. 2013, 8, 303–308. [Google Scholar] [CrossRef]
- Malec, W.; Kulasa, J.; Brudny, A.; Hury, A.; Adamczyk, B.; Rzepecki, R.; Sekula, R.; Kmita, G.; Rybak, A. Comparative studies of the properties of copper components: Conventional vs. additive manufacturing technologies. Metals 2024, 14, 975. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, J.; Niu, R.; Bayat, M.; Zhou, Y.; Yin, Y.; Tan, Q.; Liu, S.; Hattel, J.H.; Li, M.; et al. Manufacturing of high strength and high conductivity copper with laser powder bed fusion. Nat. Commun. 2024, 15, 1283. [Google Scholar] [CrossRef]
- Kumar, V.; Pandita, S.; Singh Sidhu, G.P.; Sharma, A.; Khanna, K.; Kaur, P.; Bali, A.S.; Setia, R. Copper bioavailability, uptake, toxicity and tolerance in plants: A comprehensive review. Chemosphere 2021, 262, 127810. [Google Scholar] [CrossRef]
- Guerra, J.P.L.; Penas, D.; Tavares, P.; Pereira, A.S. Influence of cupric (Cu2+) ions on the iron oxidation mechanism by DNA-binding protein from starved cells (Dps) from Marinobacter nauticus. Int. J. Mol. Sci. 2023, 24, 10256. [Google Scholar] [CrossRef]
- Tsang, T.; Davis, C.I.; Brady, D.C. Copper biology. Curr. Biol. 2021, 31, R421–R427. [Google Scholar] [CrossRef]
- Peers, G.; Price, N.M. Copper-containing plastocyanin used for electron transport by an oceanic diatom. Nature 2006, 441, 341–344. [Google Scholar] [CrossRef]
- Fu, Y.; Hou, L.; Han, K.; Zhao, C.; Hu, H.; Yin, S. The physiological role of copper: Dietary sources, metabolic regulation, and safety concerns. Clin. Nutr. 2025, 48, 161–179. [Google Scholar] [CrossRef]
- Georgopoulos, G.; Roy, A.; Yonone-Lioy, M.J.; Opiekun, R.E.; Lioy, P.J. Environmental copper: Its dynamics and human exposure issues. J. Toxicol. Environ. Health Part B 2001, 4, 341–394. [Google Scholar] [CrossRef]
- Conforti, R.A.; Delsouc, M.B.; Zorychta, E.; Telleria, C.M.; Casais, M. Copper in gynecological diseases. Int. J. Mol. Sci. 2023, 24, 17578. [Google Scholar] [CrossRef]
- Guzman Borda, F.L.; Ribeiro de Oliveira, S.J.; Seabra Monteiro Lazaro, L.M.; Kalab Leiróz, A.J. Experimental investigation of the tribological behavior of lubricants with additive containing copper nanoparticles. Tribol. Int. 2018, 117, 52–58. [Google Scholar] [CrossRef]
- Cioffi, N.; Torsi, L.; Ditaranto, N.; Tantillo, G.; Ghibelli, L.; Sabbatini, L.; Bleve-Zacheo, T.; D’Alessio, M.; Zambonin, P.G.; Traversa, E. Copper nanoparticle/polymer composites with antifungal and bacteriostatic properties. Chem. Mater. 2005, 17, 5255–5262. [Google Scholar] [CrossRef]
- Palza, H.; Galarce, N.; Bejarano, J.; Beltran, M.; Caviedes, P. Effect of copper nanoparticles on the cell viability of polymer composites. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 462–468. [Google Scholar] [CrossRef]
- Ameh, T.; Sayes, C.M. The potential exposure and hazards of copper nanoparticles: A review. Environ. Toxicol. Pharmacol. 2019, 71, 103220. [Google Scholar] [CrossRef]
- Chatterjee, A.K.; Chakraborty, R.; Basu, T. Mechanism of antibacterial activity of copper nanoparticles. Nanotechnology 2014, 25, 135101. [Google Scholar] [CrossRef]
- Lai, M.-J.; Huang, Y.-W.; Chen, H.-C.; Tsao, L.-I.; Chang Chien, C.-F.; Singh, B.; Liu, B.R. Effect of size and concentration of copper nanoparticles on the antimicrobial activity in Escherichia coli through multiple mechanisms. Nanomaterials 2022, 12, 3715. [Google Scholar] [CrossRef]
- Piya, S.; Gopali, J.; Joshi, D.R.; Shrestha, R. Oligodynamic action of silver, copper and brass on enteric bacteria isolated from water of Kathmandu Valley. Nepal J. Sci. Technol. 1970, 10, 189–193. [Google Scholar] [CrossRef]
- Oetiker, N.; Salinas, D.; Lucero-Mora, J.; Orellana, R.; Quiroz-Muñoz, M.; Bravo, D.; Pérez-Donoso, J.M. Antimicrobial effect of copper nanoparticles on relevant supragingival oral bacteria. Microorganisms 2024, 12, 624. [Google Scholar] [CrossRef]
- Pourmadadi, M.; Holghoomi, R.; Shamsabadipour, A.; Maleki-baladi, R.; Rahdar, A.; Pandey, S. Copper nanoparticles from chemical, physical, and green synthesis to medicinal application: A review. Plant Nano Biol. 2024, 8, 100070. [Google Scholar] [CrossRef]
- Ashok Sharma, R. Copper-based nanomaterials: Properties and emerging applications. Int. J. Sci. Res. (IJSR) 2025, 14, 862–870. [Google Scholar] [CrossRef]
- Baimler, I.V.; Simakin, A.V.; Uvarov, O.V.; Volkov, M.Y.; Gudkov, S.V. Generation of hydroxyl radicals during laser breakdown of aqueous solutions in the presence of Fe and Cu nanoparticles of different sizes. Phys. Wave Phenom. 2020, 28, 107–110. [Google Scholar] [CrossRef]
- Kozlov, V.A.; Shabalina, A.V.; Fomina, P.A.; Popov, I.A.; Bulygin, E.A.; Oloviannikov, G.A.; Rebezov, M.; Sergienko, K.V.; Sevostyanov, M.A.; Bunkin, N.F. Comprehensive study of material for additive technologies based on polymethyl methacrylate-like photopolymer resin modified with metallic Ti nanoparticles. Opera Medica Physiol. 2025, 12, 58–67. [Google Scholar] [CrossRef]
- Burmistrov, D.E.; Serov, D.A.; Baimler, I.V.; Gritsaeva, A.V.; Chapala, P.; Simakin, A.V.; Astashev, M.E.; Karmanova, E.E.; Dubinin, M.V.; Nizameeva, G.R.; et al. Polymethyl methacrylate-like photopolymer resin with titanium metal nanoparticles is a promising material for biomedical applications. Polymers 2025, 17, 1830. [Google Scholar] [CrossRef]
- Baimler, I.V.; Gudkov, S.V.; Matveeva, T.A.; Simakin, A.V.; Shcherbakov, I.A. Generation of reactive oxygen species during water drops fall on a solid surface. Phys. Wave Phenom. 2024, 32, 187–189. [Google Scholar] [CrossRef]
- Shtarkman, I.N.; Gudkov, S.V.; Chernikov, A.V.; Bruskov, V.I. Effect of amino acids on X-ray-induced hydrogen peroxide and hydroxyl radical formation in water and 8-oxoguanine in DNA. Biochemistry 2008, 73, 470–478. [Google Scholar] [CrossRef]
- Ivanov, V.E.; Usacheva, A.M.; Chernikov, A.V.; Bruskov, V.I.; Gudkov, S.V. Formation of long-lived reactive species of blood serum proteins induced by low-intensity irradiation of helium-neon laser and their involvement in the generation of reactive oxygen species. J. Photochem. Photobiol. B Biol. 2017, 176, 36–43. [Google Scholar] [CrossRef]
- Sevostyanov, M.A.; Kolmakov, A.G.; Sergiyenko, K.V.; Kaplan, M.A.; Baikin, A.S.; Gudkov, S.V. Mechanical, physical–chemical and biological properties of the new Ti–30Nb–13Ta–5Zr alloy. J. Mater. Sci. 2020, 55, 14516–14529. [Google Scholar] [CrossRef]
- Chaikov, L.L.; Kirichenko, M.N.; Krivokhizha, S.V.; Kupov, M.R.; Lobanov, A.N.; Sverbil, P.P.; Nagaev, E.I.; Shkirin, A.V. Two convenient methods for detection of non-dairy fats in butter by dynamic light scattering and luminescence spectroscopy. Appl. Sci. 2023, 13, 8563. [Google Scholar] [CrossRef]
- Ignat’ev, P.S.; Kol’ner, L.S.; Indukaev, K.V.; Teleshevskii, V.I. Laser modulation interference microscopy as a means of controlling the form and roughness of optical surfaces. Meas. Tech. 2015, 58, 772–776. [Google Scholar] [CrossRef]
- Gudkov, S.V.; Sarimov, R.M.; Astashev, M.E.; Pishchalnikov, R.Y.; Yanykin, D.V.; Simakin, A.V.; Shkirin, A.V.; Serov, D.A.; Konchekov, E.M.; Gusein-zade, N.G.; et al. Modern physical methods and technologies in agriculture. Phys.-Uspekhi 2024, 67, 194–210. [Google Scholar] [CrossRef]
- Sakane, S.; Akimoto, K.; Konishi, K.; Takaoka, K.; Iwatsuki, H.; Akutsu, M.; Sugai, T.; Tanaka, H. Catalytic activity of nonaggregating Cu nanoparticles supported in pores of zeolite for aerobic oxidation of benzyl alcohol. ACS Omega 2023, 9, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Satyvaldiev, A.S.; Zhasnakunov, Z.K.; Omurzak, E.; Doolotkeldieva, T.D.; Bobusheva, S.T.; Orozmatova, G.T.; Kelgenbaeva, Z. Copper nanoparticles: Synthesis and biological activity. IOP Conf. Ser. Mater. Sci. Eng. 2018, 302, 012075. [Google Scholar] [CrossRef]
- Dement’eva, O.V.; Rudoy, V.M. Copper nanoparticles synthesized by the polyol method and their oxidation in polar dispersion media. The influence of chloride and acetate ions. Colloid J. 2012, 74, 668–674. [Google Scholar] [CrossRef]
- Ali, M.; Sajjad, U.; Ali, A.; Ali, H.M.; Hamid, K. Metallic nanoparticles-based additive manufacturing: A review of recent advances on progress, prospects, and challenges. Mater. Des. 2025, 257, 114464. [Google Scholar] [CrossRef]
- Gil, L.D.; Monteiro, S.N.; Colorado, H.A. Polymer matrix nanocomposites fabricated with copper nanoparticles and photopolymer resin via vat photopolymerization additive manufacturing. Polymers 2024, 16, 2434. [Google Scholar] [CrossRef]
- Tan, X.; Li, X.; Geng, Y.; Yin, Z.; Wang, L.; Wang, W.; Deng, Y. Polymer microbubble-based fabry–perot fiber interferometer and sensing applications. IEEE Photonics Technol. Lett. 2015, 27, 2035–2038. [Google Scholar] [CrossRef]
- Johnson, P.B.; Christy, R.W. Optical constants of the noble metals. Phys. Rev. B 1972, 6, 4370–4379. [Google Scholar] [CrossRef]
- Delgado, K.; Quijada, R.; Palma, R.; Palza, H. Polypropylene with embedded copper metal or copper oxide nanoparticles as a novel plastic antimicrobial agent. Lett. Appl. Microbiol. 2011, 53, 50–54. [Google Scholar] [CrossRef]
- Jardón-Maximino, N.; Cadenas-Pliego, G.; Ávila-Orta, C.A.; Comparán-Padilla, V.E.; Lugo-Uribe, L.E.; Pérez-Alvarez, M.; Tavizón, S.F.; Santillán, G.d.J.S. Antimicrobial property of polypropylene composites and functionalized copper nanoparticles. Polymers 2021, 13, 1694. [Google Scholar] [CrossRef] [PubMed]
- Tamayo, L.A.; Zapata, P.A.; Vejar, N.D.; Azócar, M.I.; Gulppi, M.A.; Zhou, X.; Thompson, G.E.; Rabagliati, F.M.; Páez, M.A. Release of silver and copper nanoparticles from polyethylene nanocomposites and their penetration into Listeria monocytogenes. Mater. Sci. Eng. C 2014, 40, 24–31. [Google Scholar] [CrossRef]
- Liu, F.; Hu, C.-Y.; Zhao, Q.; Shi, Y.-J.; Zhong, H.-N. Migration of copper from nanocopper/LDPE composite films. Food Addit. Contam. Part A 2016, 33, 1741–1749. [Google Scholar] [CrossRef]
- Berghaus, E.; Klocke, T.; Maletz, R.; Petersen, S. Degree of conversion and residual monomer elution of 3D-printed, milled and self-cured resin-based composite materials for temporary dental crowns and bridges. J. Mater. Sci. Mater. Med. 2023, 34, 23. [Google Scholar] [CrossRef]
- Kirby, S.; Pesun, I.; Nowakowski, A.; França, R. Effect of different post-curing methods on the degree of conversion of 3D-printed resin for models in dentistry. Polymers 2024, 16, 549. [Google Scholar] [CrossRef] [PubMed]
- Šimunović, L.; Jurela, A.; Sudarević, K.; Bačić, I.; Haramina, T.; Meštrović, S. Influence of post-processing on the degree of conversion and mechanical properties of 3D-printed polyurethane aligners. Polymers 2023, 16, 17. [Google Scholar] [CrossRef] [PubMed]
- Bürgers, R.; Schubert, A.; Müller, J.; Krohn, S.; Rödiger, M.; Leha, A.; Wassmann, T. Cytotoxicity of 3D-printed, milled, and conventional oral splint resins to L929 cells and human gingival fibroblasts. Clin. Exp. Dent. Res. 2022, 8, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Rus, F.; Neculau, C.; Imre, M.; Duica, F.; Popa, A.; Moisa, R.M.; Voicu-Balasea, B.; Radulescu, R.; Ripszky, A.; Ene, R.; et al. Polymeric materials used in 3DP in dentistry—Biocompatibility testing challenges. Polymers 2024, 16, 3550. [Google Scholar] [CrossRef]
- LoPachin, R.M.; Gavin, T. Molecular mechanism of acrylamide neurotoxicity: Lessons learned from organic chemistry. Environ. Health Perspect. 2012, 120, 1650–1657. [Google Scholar] [CrossRef]
- Huchthausen, J.; Escher, B.I.; Grasse, N.; König, M.; Beil, S.; Henneberger, L. Reactivity of acrylamides causes cytotoxicity and activates oxidative stress response. Chem. Res. Toxicol. 2023, 36, 1374–1385. [Google Scholar] [CrossRef]
- Kucharczyk, M.; Słowik-Rylska, M.; Cyran-Stemplewska, S.; Gieroń, M.; Nowak-Starz, G.; Kręcisz, B. Acrylates as a significant causes of allergic contact dermatitis—New sources of exposure. Adv. Dermatol. Allergol. 2021, 38, 555–560. [Google Scholar] [CrossRef]
- Pemberton, M.A.; Kimber, I. Methyl methacrylate and respiratory sensitisation: A comprehensive review. Crit. Rev. Toxicol. 2022, 52, 139–166. [Google Scholar] [CrossRef]
- De Santis, A.; Baldi, M. Photo-polymerisation of composite resins measured by micro-Raman spectroscopy. Polymer 2004, 45, 3797–3804. [Google Scholar] [CrossRef]
- Kuznetsov, S.M.; Novikov, V.S.; Sagitova, E.A.; Ustynyuk, L.Y.; Glikin, A.A.; Prokhorov, K.A.; Nikolaeva, G.Y.; Pashinin, P.P. Raman spectra of n-pentane, n-hexane, and n-octadecane: Experimental and density functional theory (DFT) study. Laser Phys. 2019, 29, 085701. [Google Scholar] [CrossRef]
- Cheng, H.-L.; Chu, N.; Jin, F.-L.; Park, S.-J. Role of Copper Nanoparticles in the Thermal and Mechanical Properties of Expanded Graphite-Reinforced Epoxy Hybrids. ACS Omega 2024, 9, 17533–17540. [Google Scholar] [CrossRef]
- Necati, A. The effect of Cu nanoparticle adding on to epoxy-based adhesive and adhesion properties. Sci. Rep. 2020, 10, 11038. [Google Scholar] [CrossRef]
- Bazan, P.; Gajda, M.; Nosal, P.; Bąk, A.; Setlak, K.; Łach, M. The Influence of Copper Oxide Particle Size on the Properties of Epoxy Resin. Appl. Sci. 2024, 14, 2534. [Google Scholar] [CrossRef]
- Rahal, M.; Noirbent, G.; Graff, B.; Toufaily, J.; Hamieh, T.; Gigmes, D.; Dumur, F.; Lalevée, J. Novel Copper complexes as visible light photoinitiators for the synthesis of interpenetrating polymer networks (IPNs). Polymers 2022, 14, 1998. [Google Scholar] [CrossRef]
- Zhou, H.; Zha, H.; Zhang, Z.; Huang, J.; Liu, M. Advanced mechanical polymer nanocomposites: From confined interphase to structural synergy. Chem. Commun. 2025, 61, 16132–16149. [Google Scholar] [CrossRef] [PubMed]
- El Samad, S.; Marhaba, S. Localized surface plasmon resonance of copper nanoparticles using finite element method. BAU J.-Sci. Technol. 2022, 3, 2. [Google Scholar] [CrossRef]
- Chudal, L.; Pandey, N.K.; Phan, J.; Johnson, O.; Lin, L.; Yu, H.; Shu, Y.; Huang, Z.; Xing, M.; Liu, J.P.; et al. Copper-cysteamine nanoparticles as a heterogeneous fenton-like catalyst for highly selective cancer treatment. ACS Appl. Bio Mater. 2020, 3, 1804–1814. [Google Scholar] [CrossRef]
- Cao, W.; Jin, M.; Yang, K.; Chen, B.; Xiong, M.; Li, X.; Cao, G. Fenton/Fenton-like metal-based nanomaterials combine with oxidase for synergistic tumor therapy. J. Nanobiotechnol. 2021, 19, 325. [Google Scholar] [CrossRef]
- Grinberg, M.; Nemtsova, Y.; Ageyeva, M.; Brilkina, A.; Vodeneev, V. Effect of low-dose ionizing radiation on spatiotemporal parameters of functional responses induced by electrical signals in tobacco plants. Photosynth. Res. 2023, 157, 119–132. [Google Scholar] [CrossRef]
- de Jager, T.L.; Cockrell, A.E.; Du Plessis, S.S. Ultraviolet light induced generation of reactive oxygen species. In Ultraviolet Light in Human Health, Diseases and Environment; Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2017; pp. 15–23. [Google Scholar]
- Gudkov, S.V.; Pustovoy, V.I.; Sarimov, R.M.; Serov, D.A.; Simakin, A.V.; Shcherbakov, I.A. Diversity of effects of mechanical influences on living systems and aqueous solutions. Int. J. Mol. Sci. 2025, 26, 5556. [Google Scholar] [CrossRef] [PubMed]
- Gudkova, V.V.; Razvolyaeva, D.A.; Borzosekov, V.D.; Konchekov, E.M. Features of the FOX and griess method for assessing the biological activity of plasma treated solutions. Plasma Chem. Plasma Process. 2023, 44, 305–334. [Google Scholar] [CrossRef]
- Gapeyev, A.; Lukyanova, N.; Gudkov, S. Hydrogen peroxide induced by modulated electromagnetic radiation protects the cells from DNA damage. Open Life Sci. 2014, 9, 915–921. [Google Scholar] [CrossRef]
- Bruskov, V.I.; Masalimov, Z.K.; Chernikov, A.V. Heat-induced generation of reactive oxygen species in water. Dokl. Biochem. Biophys. 2002, 384, 181–184. [Google Scholar] [CrossRef]
- Lyakhov, G.A.; Man’ko, V.I.; Shcherbakov, I.A.; Suyazov, N.V. Impact of classical vibrations and magnetic fields on quantum objects. Phys. Wave Phenom. 2024, 32, 67–72. [Google Scholar] [CrossRef]
- Ponomarev, V.A.; Sheveyko, A.N.; Kuptsov, K.A.; Sukhanova, E.V.; Popov, Z.I.; Permyakova, E.S.; Slukin, P.V.; Ignatov, S.G.; Ilnitskaya, A.S.; Gloushankova, N.A.; et al. X-ray and UV irradiation-induced reactive oxygen species mediated antibacterial activity in Fe and Pt nanoparticle-decorated Si-doped TiCaCON films. ACS Appl. Mater. Interfaces 2023, 15, 50940–50952. [Google Scholar] [CrossRef]
- Belov, S.V.; Danyleiko, Y.K.; Glinushkin, A.P.; Kalinitchenko, V.P.; Egorov, A.V.; Sidorov, V.A.; Konchekov, E.M.; Gudkov, S.V.; Dorokhov, A.S.; Lobachevsky, Y.P.; et al. An activated potassium phosphate fertilizer solution for stimulating the growth of agricultural plants. Front. Phys. 2021, 8, 618320. [Google Scholar] [CrossRef]
- Gudkov, S.V.; Gudkova, O.Y.; Chernikov, A.V.; Bruskov, V.I. Protection of mice against X-ray injuries by the post-irradiation administration of guanosine and inosine. Int. J. Radiat. Biol. 2009, 85, 116–125. [Google Scholar] [CrossRef]
- Maliki, M.; Ifijen, I.H.; Ikhuoria, E.U.; Jonathan, E.M.; Onaiwu, G.E.; Archibong, U.D.; Ighodaro, A. Copper nanoparticles and their oxides: Optical, anticancer and antibacterial properties. Int. Nano Lett. 2022, 12, 379–398. [Google Scholar] [CrossRef]
- Zhai, Y.; Liang, Z.; Liu, X.; Zhang, W. Employing copper-based nanomaterials to combat multi-drug-resistant bacteria. Microorganisms 2025, 13, 708. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhou, S.; Xu, X.; Du, Q. Copper-containing nanoparticles: Mechanism of antimicrobial effect and application in dentistry—A narrative review. Front. Surg. 2022, 9, 905892. [Google Scholar] [CrossRef]
- Ermini, M.L.; Voliani, V. Antimicrobial nano-agents: The copper age. ACS Nano 2021, 15, 6008–6029. [Google Scholar] [CrossRef]
- Ramos-Zúñiga, J.; Bruna, N.; Pérez-Donoso, J.M. Toxicity mechanisms of copper nanoparticles and copper surfaces on bacterial cells and viruses. Int. J. Mol. Sci. 2023, 24, 10503. [Google Scholar] [CrossRef]
- Crmarić, D.; Bura-Nakić, E. Interaction between Cu and thiols of biological and environmental importance: Case study using combined spectrophotometric/bathocuproine sulfonate disodium salt hydrate (BCS) assay. Molecules 2023, 28, 5065. [Google Scholar] [CrossRef]
- Sajjad, H.; Sajjad, A.; Haya, R.T.; Khan, M.M.; Zia, M. Copper oxide nanoparticles: In vitro and in vivo toxicity, mechanisms of action and factors influencing their toxicology. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2023, 271, 109682. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, B.M.; Ali, S.F.; Murdock, R.C.; Hussain, S.M.; Srivatsan, M. Copper nanoparticles exert size and concentration dependent toxicity on somatosensory neurons of rat. Nanotoxicology 2009, 4, 150–160. [Google Scholar] [CrossRef]
- Na, I.; Kennedy, D.C. Size-specific copper nanoparticle cytotoxicity varies between human cell lines. Int. J. Mol. Sci. 2021, 22, 1548. [Google Scholar] [CrossRef]
- Rodhe, Y.; Skoglund, S.; Odnevall Wallinder, I.; Potácová, Z.; Möller, L. Copper-based nanoparticles induce high toxicity in leukemic HL60 cells. Toxicol. Vitr. 2015, 29, 1711–1719. [Google Scholar] [CrossRef]
- Fahmy, H.M.; Ebrahim, N.M.; Gaber, M.H. In-vitro evaluation of copper/copper oxide nanoparticles cytotoxicity and genotoxicity in normal and cancer lung cell lines. J. Trace Elem. Med. Biol. 2020, 60, 126481. [Google Scholar] [CrossRef] [PubMed]
- Valodkar, M.; Rathore, P.S.; Jadeja, R.N.; Thounaojam, M.; Devkar, R.V.; Thakore, S. Cytotoxicity evaluation and antimicrobial studies of starch capped water soluble copper nanoparticles. J. Hazard. Mater. 2012, 201–202, 244–249. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Burmistrov, D.E.; Serov, D.A.; Sizov, L.R.; Astashev, M.E.; Karmanova, E.E.; Baimler, I.V.; Simakin, A.V.; Ignatenko, D.N.; Yanbaev, F.M.; Kuzmin, E.V.; et al. Functionalization of Photopolymer with Laser-Ablated Copper NPs: A Comprehensive Study of ROS Generation, Antimicrobial Activity and Cytotoxic Profile. Polymers 2026, 18, 238. https://doi.org/10.3390/polym18020238
Burmistrov DE, Serov DA, Sizov LR, Astashev ME, Karmanova EE, Baimler IV, Simakin AV, Ignatenko DN, Yanbaev FM, Kuzmin EV, et al. Functionalization of Photopolymer with Laser-Ablated Copper NPs: A Comprehensive Study of ROS Generation, Antimicrobial Activity and Cytotoxic Profile. Polymers. 2026; 18(2):238. https://doi.org/10.3390/polym18020238
Chicago/Turabian StyleBurmistrov, Dmitriy E., Dmitriy A. Serov, Lev R. Sizov, Maxim E. Astashev, Ekaterina E. Karmanova, Ilya V. Baimler, Alexander V. Simakin, Dmitriy N. Ignatenko, Fatikh M. Yanbaev, Evgeny V. Kuzmin, and et al. 2026. "Functionalization of Photopolymer with Laser-Ablated Copper NPs: A Comprehensive Study of ROS Generation, Antimicrobial Activity and Cytotoxic Profile" Polymers 18, no. 2: 238. https://doi.org/10.3390/polym18020238
APA StyleBurmistrov, D. E., Serov, D. A., Sizov, L. R., Astashev, M. E., Karmanova, E. E., Baimler, I. V., Simakin, A. V., Ignatenko, D. N., Yanbaev, F. M., Kuzmin, E. V., & Gudkov, S. V. (2026). Functionalization of Photopolymer with Laser-Ablated Copper NPs: A Comprehensive Study of ROS Generation, Antimicrobial Activity and Cytotoxic Profile. Polymers, 18(2), 238. https://doi.org/10.3390/polym18020238







