Investigation on Mechanical Properties of Functional Graded Hybrid TPMS Structures Inspired Bone Scaffolds
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Specimen Fabrication
2.2. Compression Tests
2.3. Statistical Methods and Optimization
2.4. Energy Absorption Calculations
3. Results and Discussion
3.1. Deformation Analysis
3.2. Compression Test Results
3.3. Energy Absorption Capacity and Surface/Volume Ratio Results
3.4. Taguchi Analysis Results
4. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bouakaz, I.; Sadeghian Dehkord, E.; Meille, S.; Schrijnemakers, A.; Boschini, F.; Preux, N.; Hocquet, S.; Geris, L.; Nolens, G.; Grossin, D.; et al. 3D Printed Triply Periodic Minimal Surfaces Calcium Phosphate Bone Substitute: The Effect of Porosity Design on Mechanical Properties. Ceram. Int. 2024, 50, 2623–2636. [Google Scholar] [CrossRef]
- Fitzpatrick, V.; Martín-Moldes, Z.; Deck, A.; Torres-Sanchez, R.; Valat, A.; Cairns, D.; Li, C.; Kaplan, D.L. Functionalized 3D-Printed Silk-Hydroxyapatite Scaffolds for Enhanced Bone Regeneration with Innervation and Vascularization. Biomaterials 2021, 276, 120995. [Google Scholar] [CrossRef]
- Zhou, X.; Feng, Y.; Zhang, J.; Shi, Y.; Wang, L. Recent Advances in Additive Manufacturing Technology for Bone Tissue Engineering Scaffolds. Int. J. Adv. Manuf. Technol. 2020, 108, 3591–3606. [Google Scholar] [CrossRef]
- Shi, J.; Zhu, L.; Li, L.; Li, Z.; Yang, J.; Wang, X. A TPMS-Based Method for Modeling Porous Scaffolds for Bionic Bone Tissue Engineering. Sci. Rep. 2018, 8, 7395. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Johansson, C. Osteoinduction, Osteoconduction and Osseointegration. Eur. Spine J. 2001, 10, S96–S101. [Google Scholar] [CrossRef]
- Cheah, C.W.; Al-Namnam, N.M.; Lau, M.N.; Lim, G.S.; Raman, R.; Fairbairn, P.; Ngeow, W.C. Synthetic Material for Bone, Periodontal, and Dental Tissue Regeneration: Where Are We Now, and Where Are We Heading Next? Materials 2021, 14, 6123. [Google Scholar] [CrossRef]
- Zhao, R.; Yang, R.; Cooper, P.R.; Khurshid, Z.; Shavandi, A.; Ratnayake, J. Bone Grafts and Substitutes in Dentistry: A Review of Current Trends and Developments. Molecules 2021, 26, 3007. [Google Scholar] [CrossRef]
- Richter, R.F.; Ahlfeld, T.; Gelinsky, M.; Lode, A. Composites Consisting of Calcium Phosphate Cements and Mesoporous Bioactive Glasses as a 3D Plottable Drug Delivery System. Acta Biomater. 2023, 156, 146–157. [Google Scholar] [CrossRef]
- Ahmed Ramadan, M.; Sabour, H.A.; EL-Shenawy, E. Tribological Properties of 3D Printed Polymers: PCL, ABS, PLA and Co Polyester. Tribol. Ind. 2023, 45, 161–167. [Google Scholar] [CrossRef]
- Zaaba, N.F.; Jaafar, M.; Ismail, H. Tensile and Morphological Properties of Nanocrystalline Cellulose and Nanofibrillated Cellulose Reinforced Bionanocomposites: A Review. Polym. Eng. Sci. 2021, 61, 22–38. [Google Scholar] [CrossRef]
- Nanda, H.S.; Yang, L.; Hu, J.; Jiang, S.; Mao, H. Editorial: Biodegradable Polymers for Biomedical Applications-Volume II. Front. Mater. 2023, 10, 1231445. [Google Scholar] [CrossRef]
- Sheikh, Z.; Najeeb, S.; Khurshid, Z.; Verma, V.; Rashid, H.; Glogauer, M. Biodegradable Materials for Bone Repair and Tissue Engineering Applications. Materials 2015, 8, 5744–5794. [Google Scholar] [CrossRef]
- Paiva, J.C.C.; Oliveira, L.; Vaz, M.F.; Costa-de-Oliveira, S. Biodegradable Bone Implants as a New Hope to Reduce Device-Associated Infections—A Systematic Review. Bioengineering 2022, 9, 409. [Google Scholar] [CrossRef]
- Farjaminejad, S.; Farjaminejad, R.; Hasani, M.; Garcia-Godoy, F.; Abdouss, M.; Marya, A.; Harsoputranto, A.; Jamilian, A. Advances and Challenges in Polymer-Based Scaffolds for Bone Tissue Engineering: A Path Towards Personalized Regenerative Medicine. Polymers 2024, 16, 3303. [Google Scholar] [CrossRef] [PubMed]
- Batoni, E.; Bonatti, A.F.; De Maria, C.; Dalgarno, K.; Naseem, R.; Dianzani, U.; Gigliotti, C.L.; Boggio, E.; Vozzi, G. A Computational Model for the Release of Bioactive Molecules by the Hydrolytic Degradation of a Functionalized Polyester-Based Scaffold. Pharmaceutics 2023, 15, 815. [Google Scholar] [CrossRef] [PubMed]
- Myakinin, A.; Turlybekuly, A.; Pogrebnjak, A.; Mirek, A.; Bechelany, M.; Liubchak, I.; Oleshko, O.; Husak, Y.; Korniienko, V.; Leśniak-Ziółkowska, K.; et al. In Vitro Evaluation of Electrochemically Bioactivated Ti6Al4V 3D Porous Scaffolds. Mater. Sci. Eng. C 2021, 121, 111870. [Google Scholar] [CrossRef]
- Li, Y.; Han, X.; Ma, Z. Research on the Mechanical Properties of PEEK Material Artificial Bone Implants Fabricated by High-Temperature Air-Assisted 3D Printing. J. Mech. Behav. Biomed. Mater. 2026, 173, 107207. [Google Scholar] [CrossRef]
- Jariwala, S.H.; Lewis, G.S.; Bushman, Z.J.; Adair, J.H.; Donahue, H.J. 3D Printing of Personalized Artificial Bone Scaffolds. 3D Print. Addit. Manuf. 2015, 2, 56–64. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, G.; Liang, H.; Gao, C.; Peng, S.; Shen, L.; Shuai, C. Additive Manufacturing of Bone Scaffolds. Int. J. Bioprinting 2018, 5, 148. [Google Scholar] [CrossRef]
- de Carvalho, A.B.G.; Rahimnejad, M.; Oliveira, R.L.M.S.; Sikder, P.; Saavedra, G.S.F.A.; Bhaduri, S.B.; Gawlitta, D.; Malda, J.; Kaigler, D.; Trichês, E.S.; et al. Personalized Bioceramic Grafts for Craniomaxillofacial Bone Regeneration. Int. J. Oral Sci. 2024, 16, 62. [Google Scholar] [CrossRef]
- Pugliese, R.; Graziosi, S. Biomimetic Scaffolds Using Triply Periodic Minimal Surface-Based Porous Structures for Biomedical Applications. SLAS Technol. 2023, 28, 165–182. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Cui, H.; Zhang, H.; Wang, F.; Zhan, X.; Mayer, F.; Nestler, B.; Wegener, M.; Levkin, P.A. 3D Printing of Inherently Nanoporous Polymers via Polymerization-Induced Phase Separation. Nat. Commun. 2021, 12, 247. [Google Scholar] [CrossRef]
- Schoen, A.H. Infinite Periodic Minimal Surfaces Without Self-Intersections; National Aeronautics and Space Administration: Washington, DC, USA, 1970; Volume 5541.
- Shaikh, M.; Kahwash, F.; Lu, Z.; Alkhreisat, M.; Mohammad, A.; Shyha, I. Revolutionising Orthopaedic Implants—A Comprehensive Review on Metal 3D Printing with Materials, Design Strategies, Manufacturing Technologies, and Post-Process Machining Advancements. Int. J. Adv. Manuf. Technol. 2024, 134, 1043–1076. [Google Scholar] [CrossRef]
- Zou, S.; Mu, Y.; Pan, B.; Li, G.; Shao, L.; Du, J.; Jin, Y. Mechanical and Biological Properties of Enhanced Porous Scaffolds Based on Triply Periodic Minimal Surfaces. Mater. Des. 2022, 219, 110803. [Google Scholar] [CrossRef]
- Górnicki, T.; Lambrinow, J.; Golkar-Narenji, A.; Data, K.; Domagała, D.; Niebora, J.; Farzaneh, M.; Mozdziak, P.; Zabel, M.; Antosik, P.; et al. Biomimetic Scaffolds—A Novel Approach to Three Dimensional Cell Culture Techniques for Potential Implementation in Tissue Engineering. Nanomaterials 2024, 14, 531. [Google Scholar] [CrossRef]
- Huang, D.; Li, Z.; Li, G.; Zhou, F.; Wang, G.; Ren, X.; Su, J. Biomimetic Structural Design in 3D-Printed Scaffolds for Bone Tissue Engineering. Mater. Today Bio 2025, 32, 101664. [Google Scholar] [CrossRef]
- Viet, N.V.; Waheed, W.; Alazzam, A.; Zaki, W. Effective Compressive Behavior of Functionally Graded TPMS Titanium Implants with Ingrown Cortical or Trabecular Bone. Compos. Struct. 2023, 303, 116288. [Google Scholar] [CrossRef]
- Karamanli, İ.A.; Temiz, A. Tailoring the Compressive Strength of the Triply Periodic Minimal Surface Lattice with Nozzle Temperature Grade. J. Braz. Soc. Mech. Sci. Eng. 2025, 47, 146. [Google Scholar] [CrossRef]
- Shi, Y.; Hou, X.; Na, Z.; Zhou, J.; Yu, N.; Liu, S.; Xin, L.; Gao, G.; Liu, Y. Bio-Inspired Attachment Mechanism of Dynastes Hercules: Vertical Climbing for On-Orbit Assembly Legged Robots. J. Bionic Eng. 2024, 21, 137–148. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, F.; Zhai, W.; Cheng, S.; Li, J.; Wang, Y. Unraveling of Advances in 3D-Printed Polymer-Based Bone Scaffolds. Polymers 2022, 14, 566. [Google Scholar] [CrossRef]
- Han, C.; Li, Y.; Wang, Q.; Wen, S.; Wei, Q.; Yan, C.; Hao, L.; Liu, J.; Shi, Y. Continuous Functionally Graded Porous Titanium Scaffolds Manufactured by Selective Laser Melting for Bone Implants. J. Mech. Behav. Biomed. Mater. 2018, 80, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D. New Paradigms in Hierarchical Porous Scaffold Design for Tissue Engineering. Mater. Sci. Eng. C 2013, 33, 1759–1772. [Google Scholar] [CrossRef]
- Pehlivan, F.; Karamanlı, İ.A.; Temiz, A.; Öztürk, F.H.; Karaca, M.M. Hierarchical Cellular Structures Based on TPMS Mimicking Cancellous Bone. J. Mech. Behav. Biomed. Mater. 2025, 168, 107037. [Google Scholar] [CrossRef]
- Demircioğlu, C.; Öztürk, F.H. Optimization of Energy Absorption in Bio-Inspired Cylindrical Lattice Structures: An Experimental Investigation with Box–Behnken Design. Rapid Prototyp. J. 2025. [Google Scholar] [CrossRef]
- Demir, S.; Temiz, A.; Pehlivan, F. The Investigation of Printing Parameters Effect on Tensile Characteristics for Triply Periodic Minimal Surface Designs by Taguchi. Polym. Eng. Sci. 2024, 64, 1209–1221. [Google Scholar] [CrossRef]
- Temiz, A.; Pehlivan, F.; Öztürk, F.H.; Demir, S. Compression Behavior of Sheet-Network Triply Periodic Minimal Surface Metamaterials as a Function of Density Grading. J. Reinf. Plast. Compos. 2024, 43, 1430–1443. [Google Scholar] [CrossRef]
- Ejeh, C.J.; Barsoum, I.; Abou-Ali, A.M.; Abu Al-Rub, R.K. Combining Multiple Lattice-Topology Functional Grading Strategies for Enhancing the Dynamic Compressive Behavior of TPMS-Based Metamaterials. J. Mater. Res. Technol. 2023, 27, 6076–6093. [Google Scholar] [CrossRef]
- Qureshi, Z.A.; Al-Omari, S.A.B.; Elnajjar, E.; Al-Ketan, O.; Al-Rub, R.A. On the Effect of Porosity and Functional Grading of 3D Printable Triply Periodic Minimal Surface (TPMS) Based Architected Lattices Embedded with a Phase Change Material. Int. J. Heat Mass Transf. 2022, 183, 122111. [Google Scholar] [CrossRef]
- ASTM D1621; Standard Test Method for Compressive Properties of Rigid Cellular Plastics. ASTM: West Conshohocken, PA, USA, 2000.
- Anycubic Anycubic Bio Resin Chemical Composition and Mechanical Properties. Available online: https://www.anycubic.au/products/bio-resin-sale-for-resin-3d-printer (accessed on 27 December 2025).
- Browning, M.B.; Cereceres, S.N.; Luong, P.T.; Cosgriff-Hernandez, E.M. Determination of the in Vivo Degradation Mechanism of PEGDA Hydrogels. J. Biomed. Mater. Res. Part A 2014, 102, 4244–4251. [Google Scholar] [CrossRef]
- Mondal, D.; Haghpanah, Z.; Huxman, C.J.; Tanter, S.; Sun, D.; Gorbet, M.; Willett, T.L. MSLA-Based 3D Printing of Acrylated Epoxidized Soybean Oil—Nano-Hydroxyapatite Composites for Bone Repair. Mater. Sci. Eng. C 2021, 130, 112456. [Google Scholar] [CrossRef]
- Overbeck, M.; Heimbs, S.; Kube, J.; Hühne, C. Energy Absorption Properties of 3D-Printed Polymeric Gyroid Structures for an Aircraft Wing Leading Edge. Aerospace 2024, 11, 801. [Google Scholar] [CrossRef]
- Maskery, I.; Aboulkhair, N.T.; Aremu, A.O.; Tuck, C.J.; Ashcroft, I.A. Compressive Failure Modes and Energy Absorption in Additively Manufactured Double Gyroid Lattices. Addit. Manuf. 2017, 16, 24–29. [Google Scholar] [CrossRef]
- Sharma, D.; Hiremath, S.S. Bio-Inspired Repeatable Lattice Structures for Energy Absorption: Experimental and Finite Element Study. Compos. Struct. 2022, 283, 115102. [Google Scholar] [CrossRef]
- Li, Y.; Feng, Z.; Hao, L.; Huang, L.; Xin, C.; Wang, Y.; Bilotti, E.; Essa, K.; Zhang, H.; Li, Z.; et al. A Review on Functionally Graded Materials and Structures via Additive Manufacturing: From Multi-Scale Design to Versatile Functional Properties. Adv. Mater. Technol. 2020, 5, 1900981. [Google Scholar] [CrossRef]
- Aghajani, S.; Wu, C.; Li, Q.; Fang, J. Additively Manufactured Composite Lattices: A State-of-the-Art Review on Fabrications, Architectures, Constituent Materials, Mechanical Properties, and Future Directions. Thin-Walled Struct. 2024, 197, 111539. [Google Scholar] [CrossRef]
- Dave, H.K.; Karumuri, R.T.; Prajapati, A.R.; Rajpurohit, S.R. Specific Energy Absorption During Compression Testing of ABS and FPU Parts Fabricated Using LCD-SLA Based 3D Printer. Rapid Prototyp. J. 2022, 28, 1530–1540. [Google Scholar] [CrossRef]
- Dara, A.; Bahubalendruni, M.A.R.; Johnney Mertens, A.; Balamurali, G. Numerical and Experimental Investigations of Novel Nature Inspired Open Lattice Cellular Structures for Enhanced Stiffness and Specific Energy Absorption. Mater. Today Commun. 2022, 31, 103286. [Google Scholar] [CrossRef]
- Lal Lazar, P.J.; Subramanian, J.; Natarajan, E.; Markandan, K.; Ramesh, S. Anisotropic Structure-Property Relations of FDM Printed Short Glass Fiber Reinforced Polyamide TPMS Structures under Quasi-Static Compression. J. Mater. Res. Technol. 2023, 24, 9562–9579. [Google Scholar] [CrossRef]
- Saleh, M.; Anwar, S.; Al-Ahmari, A.M.; Alfaify, A. Compression Performance and Failure Analysis of 3D-Printed Carbon Fiber/PLA Composite TPMS Lattice Structures. Polymers 2022, 14, 4595. [Google Scholar] [CrossRef]
- Kumar, R.; Ramkumar, J.; Balani, K. Design and Parametrization of TPMS Lattice Using Computational and Experimental Approach. Eng. Res. Express 2024, 6, 035556. [Google Scholar] [CrossRef]
- Poltue, T.; Karuna, C.; Khrueaduangkham, S.; Seehanam, S.; Promoppatum, P. Design Exploration of 3D-Printed Triply Periodic Minimal Surface Scaffolds for Bone Implants. Int. J. Mech. Sci. 2021, 211, 106762. [Google Scholar] [CrossRef]
- Guo, X.; Ding, J.; Li, X.; Qu, S.; Hsi Fuh, J.Y.; Lu, W.F.; Song, X.; Zhai, W. Interpenetrating Phase Composites with 3D Printed Triply Periodic Minimal Surface (TPMS) Lattice Structures. Compos. Part B Eng. 2023, 248, 110351. [Google Scholar] [CrossRef]
- Novak, N.; Borovinšek, M.; Al-Ketan, O.; Ren, Z.; Vesenjak, M. Impact and Blast Resistance of Uniform and Graded Sandwich Panels with TPMS Cellular Structures. Compos. Struct. 2022, 300, 116174. [Google Scholar] [CrossRef]
- Gibson, L.J. Cellular Solids. MRS Bull. 2003, 28, 270–274. [Google Scholar] [CrossRef]
- Shevchenko, V.; Balabanov, S.; Sychov, M.; Karimova, L. Prediction of Cellular Structure Mechanical Properties with the Geometry of Triply Periodic Minimal Surfaces (TPMS). ACS Omega 2023, 8, 26895–26905. [Google Scholar] [CrossRef]
- Al Rifaie, M.; Mian, A.; Srinivasan, R. Compression Behavior of Three-Dimensional Printed Polymer Lattice Structures. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 2018, 233, 1574–1584. [Google Scholar] [CrossRef]
- Qiu, N.; Wan, Y.; Shen, Y.; Fang, J. Experimental and Numerical Studies on Mechanical Properties of TPMS Structures. Int. J. Mech. Sci. 2024, 261, 108657. [Google Scholar] [CrossRef]
- Kumar, R.; Ramkumar, J.; Balani, K. Uniform and Multi-Morphology Graded TPMS Structures: Design Strategies, 3D Printing and Mechanical Properties. J. Mech. Behav. Biomed. Mater. 2026, 173, 107208. [Google Scholar] [CrossRef] [PubMed]
- Zhong, M.; Zhou, W.; Xi, H.; Liang, Y.; Wu, Z. Double-Level Energy Absorption of 3D Printed TPMS Cellular Structures via Wall Thickness Gradient Design. Materials 2021, 14, 6262. [Google Scholar] [CrossRef] [PubMed]
- Justino Netto, J.; Sardinha, M.; Leite, M. Influence of the Cell Size and Wall Thickness on the Compressive Behaviour of Fused Filament Fabricated PLA Gyroid Structures. Mech. Mater. 2024, 195, 105051. [Google Scholar] [CrossRef]
- Feng, G.; Li, S.; Xiao, L.; Song, W. Mechanical Properties and Deformation Behavior of Functionally Graded TPMS Structures under Static and Dynamic Loading. Int. J. Impact Eng. 2023, 176, 104554. [Google Scholar] [CrossRef]
- Song, J.; Wang, M.; Li, D.; Zhang, J. Deformation and Energy Absorption Performance of Functionally Graded TPMS Structures Fabricated by Selective Laser Melting. Appl. Sci. 2024, 14, 2064. [Google Scholar] [CrossRef]
- Rohani Nejad, S.; Hosseinpour, M.; Mirbagheri, S.M.H. Experimental and Simulation Investigation on Energy Absorption of Cu Casting Lattice Structure during Compression. Adv. Eng. Mater. 2023, 25, 2300447. [Google Scholar] [CrossRef]
- Şimşek, U.; Gülcan, O.; Günaydın, K.; Tamer, A. Prediction of Compressive Behavior of Laser-Powder-Bed Fusion-Processed TPMS Lattices by Regression Analysis. J. Manuf. Mater. Process. 2024, 8, 16. [Google Scholar] [CrossRef]
- Lu, C.; Ding, J.; Jiang, X.; Wen, P.; Zhang, C.; Shen, Q.; Chen, F. Enhancing Mechanical Properties and Damage Tolerance of Additive Manufactured Ceramic TPMS Lattices by Hybrid Design. J. Am. Ceram. Soc. 2024, 107, 6524–6531. [Google Scholar] [CrossRef]
- de Aquino, D.A.; Maskery, I.; Longhitano, G.A.; Jardini, A.L.; del Conte, E.G. Investigation of Load Direction on the Compressive Strength of Additively Manufactured Triply Periodic Minimal Surface Scaffolds. Int. J. Adv. Manuf. Technol. 2020, 109, 771–779. [Google Scholar] [CrossRef]
- Sivakumar, N.K.; Palaniyappan, S.; Vishal, K.; Alibrahim, K.A.; Alodhayb, A.; A, M.K. Crushing Behavior Optimization of Octagonal Lattice-structured Thin-walled 3D Printed Carbon Fiber Reinforced PETG (CF/PETG) Composite Tubes under Axial Loading. Polym. Compos. 2024, 45, 1228–1249. [Google Scholar] [CrossRef]
- Rivera, J.; Yang, Q.; Bustillos, C.G.; Chandrasekaran, S.; Wat, A.; Sobalvarro, E.M.; Worsley, M.A.; Pascall, A.J.; Kuntz, J.D. Mechanical Responses of Architected Boron Carbide-Aluminum Lattice Composites Fabricated via Reactive Metallic Infiltration of Hierarchical Pore Structures. Mater. Today Commun. 2023, 37, 107550. [Google Scholar] [CrossRef]
- Sumner, D.R. Long-Term Implant Fixation and Stress-Shielding in Total Hip Replacement. J. Biomech. 2015, 48, 797–800. [Google Scholar] [CrossRef]
- Hutmacher, D.W. Scaffolds in Tissue Engineering Bone and Cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Temiz, A.; Yaşar, M.; Koç, E. Fabrication of Open-Pore Biodegradable Magnesium Alloy Scaffold via Infiltration Technique. Int. J. Met. 2022, 16, 317–328. [Google Scholar] [CrossRef]
- Aerssens, J.; Boonen, S.; Lowet, G.; Dequeker, J. Interspecies Differences in Bone Composition, Density, and Quality: Potential Implications for in Vivo Bone Research. Endocrinology 1998, 139, 663–670. [Google Scholar] [CrossRef]
- Egermann, M.; Goldhahn, J.; Holz, R.; Schneider, E.; Lill, C.A. A Sheep Model for Fracture Treatment in Osteoporosis: Benefits of the Model versus Animal Welfare. Lab. Anim. 2008, 42, 453–464. [Google Scholar] [CrossRef]
- He, L.; Li, T.; Zhong, D.; Tao, H.; Peng, Y.; Chen, S. Blast Resistance in Sandwich Structures Based on TPMS. Buildings 2023, 13, 2835. [Google Scholar] [CrossRef]
- Shi, X.; Liao, W.; Li, P.; Zhang, C.; Liu, T.; Wang, C.; Wu, J. Comparison of Compression Performance and Energy Absorption of Lattice Structures Fabricated by Selective Laser Melting. Adv. Eng. Mater. 2020, 22, 2000453. [Google Scholar] [CrossRef]
- Choy, S.Y.; Sun, C.-N.; Leong, K.F.; Wei, J. Compressive Properties of Ti-6Al-4V Lattice Structures Fabricated by Selective Laser Melting: Design, Orientation and Density. Addit. Manuf. 2017, 16, 213–224. [Google Scholar] [CrossRef]
- Porter, J.H.; Cain, T.M.; Fox, S.L.; Harvey, P.S. Influence of Infill Properties on Flexural Rigidity of 3D-Printed Structural Members. Virtual Phys. Prototyp. 2019, 14, 148–159. [Google Scholar] [CrossRef]
- Ziaie, B.; Velay, X.; Saleem, W. Exploring the Optimal Mechanical Properties of Triply Periodic Minimal Surface Structures for Biomedical Applications: A Numerical Analysis. J. Mech. Behav. Biomed. Mater. 2024, 160, 106757. [Google Scholar] [CrossRef]
- Novak, N.; Al-Ketan, O.; Borovinšek, M.; Krstulović-Opara, L.; Rowshan, R.; Vesenjak, M.; Ren, Z. Development of Novel Hybrid TPMS Cellular Lattices and Their Mechanical Characterisation. J. Mater. Res. Technol. 2021, 15, 1318–1329. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, J.; Niu, Q.; Xu, C.; Huo, R. A Study of Energy Absorption Properties of Heteromorphic TPMS and Multi-Morphology TPMS under Quasi-Static Compression. Thin-Walled Struct. 2024, 205, 112519. [Google Scholar] [CrossRef]
- Song, J.; Huo, Q.; Li, D.; Chen, B.; Zhang, J. Energy-Absorption Behavior of Novel Bio-Inspired Thin-Walled Honeycomb Tubes Filled with TPMS Structure. Coatings 2024, 14, 675. [Google Scholar] [CrossRef]
- Wan, L.; Hu, D.; Zhang, H.; Yang, Z. Energy Absorption of Foam-Filled TPMS-Based Tubular Lattice Structures Subjected to Quasi-Static Lateral Crushing. Eng. Struct. 2024, 316, 118581. [Google Scholar] [CrossRef]
- Zhang, T.; Liu, F.; Deng, X.; Zhao, M.; Zhou, H.; Zhang, D.Z. Experimental Study on the Thermal Storage Performance of Phase Change Materials Embedded with Additively Manufactured Triply Periodic Minimal Surface Architected Lattices. Int. J. Heat Mass Transf. 2022, 199, 123452. [Google Scholar] [CrossRef]
- Dargusch, M.S.; Soro, N.; Demir, A.G.; Venezuela, J.; Sun, Q.; Wang, Y.; Abdal-hay, A.; Alali, A.Q.; Ivanovski, S.; Previtali, B.; et al. Optimising Degradation and Mechanical Performance of Additively Manufactured Biodegradable Fe–Mn Scaffolds Using Design Strategies Based on Triply Periodic Minimal Surfaces. Smart Mater. Med. 2024, 5, 127–139. [Google Scholar] [CrossRef]
- Mulhi, A.; Dehgahi, S.; Waghmare, P.; Qureshi, A. Dimensional Assessment of Uniformly Periodic Porosity Primitive TPMS Lattices Using Additive Manufacturing Laser Powder Bed Fusion Technique. Int. J. Adv. Manuf. Technol. 2023, 124, 2127–2148. [Google Scholar] [CrossRef]
- Lerebours, C.; Thomas, C.D.L.; Clement, J.G.; Buenzli, P.R.; Pivonka, P. The Relationship between Porosity and Specific Surface in Human Cortical Bone Is Subject Specific. Bone 2015, 72, 109–117. [Google Scholar] [CrossRef]
- Soysal, H.; Geneci, F.; Ocak, M. Investigation of the Human Maxilla and Mandible Trabecular Microstructure with Micro-Computed Tomography. Harran Üniversitesi Tıp Fakültesi Derg. 2023, 20, 67–73. [Google Scholar] [CrossRef]
- Rodić, D.; Sekulić, M.; Savković, B.; Madić, M.; Trifunović, M. Integration of RSM and Machine Learning for Accurate Prediction of Surface Roughness in Laser Processing. Appl. Sci. 2025, 15, 7064. [Google Scholar] [CrossRef]
- Panjabi, M.M.; White, A.A.I.I.I. Basic Biomechanics of the Spine. Neurosurgery 1980, 7, 76–93. [Google Scholar] [CrossRef]
- Oftadeh, R.; Perez-Viloria, M.; Villa-Camacho, J.C.; Vaziri, A.; Nazarian, A. Biomechanics and Mechanobiology of Trabecular Bone: A Review. J. Biomech. Eng. 2015, 137, 108021–1080215. [Google Scholar] [CrossRef]







| Chemical Composition of Bio Resin | |
|---|---|
| 2-Oxepanone, homopolymer, 2-[(1-oxo-2-propen-1-yl)oxy]ethyl ester | 30–60% |
| Oxirane, 2-methyl-, polymer with oxirane, bis(2-methyl-2-propenoate), block | 20–40% |
| 1,2-Ethanediyl bisacrylate | 25–40% |
| phenyl bis (2,4,6-trimethylbenzoyl)-phosphine oxide | 2–5% |
| Mechanical Properties | |
| Density | 1.13–1.15 kg/mm3 |
| Hardness | 83–85 Shore D |
| Tensile Strength | 35–40 MPa |
| Elongation | 21–28% |
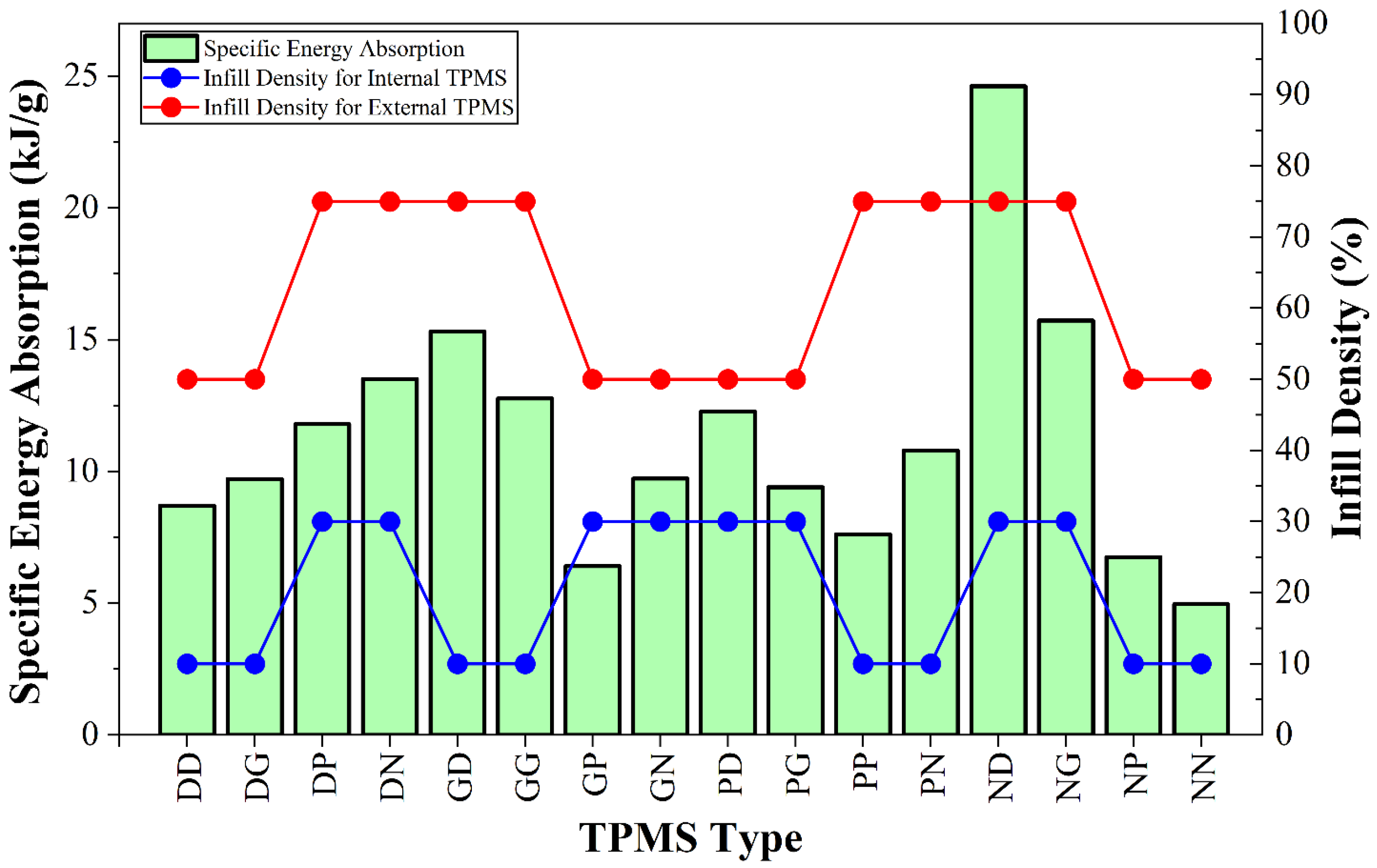
| Specimen Code | Internal TPMS Type | Infill Density for Internal TPMS (%) | External TPMS Type | Infill Density for External TPMS (%) |
|---|---|---|---|---|
| DD | Diamond | 10 | Diamond | 50 |
| DG | Diamond | 10 | Gyroid | 50 |
| DP | Diamond | 30 | Primitive | 75 |
| DN | Diamond | 30 | Neovius | 75 |
| GD | Gyroid | 10 | Diamond | 75 |
| GG | Gyroid | 10 | Gyroid | 75 |
| GP | Gyroid | 30 | Primitive | 50 |
| GN | Gyroid | 30 | Neovius | 50 |
| PD | Primitive | 30 | Diamond | 50 |
| PG | Primitive | 30 | Gyroid | 50 |
| PP | Primitive | 10 | Primitive | 75 |
| PN | Primitive | 10 | Neovius | 75 |
| ND | Neovius | 30 | Diamond | 75 |
| NG | Neovius | 30 | Gyroid | 75 |
| NP | Neovius | 10 | Primitive | 50 |
| NN | Neovius | 10 | Neovius | 50 |
| Specimen Code | First Peak (kN) | Maximum Force (kN) | Energy Absorption (kJ) | Specific Energy Absorption (kJ/g) | Surface/Volume Ratio (1/mm) |
|---|---|---|---|---|---|
| DD | 5.54 ± 0.32 | 6.17 ± 0.43 | 120.15 ± 11.85 | 8.70 ± 0.86 | 5.03 |
| DG | 5.17 ± 0.28 | 5.67 ± 0.37 | 135.63 ± 12.17 | 9.72 ± 0.87 | 4.51 |
| DP | 7.12 ± 0.57 | 7.32 ± 0.46 | 208.16 ± 17.89 | 11.80 ± 1.01 | 3.12 |
| DN | 18.96 ± 1.12 | 19.14 ± 1.23 | 360.13 ± 24.15 | 13.51 ± 0.91 | 1.81 |
| GD | 9.92 ± 0.83 | 13.22 ± 1.09 | 294.56 ± 23.87 | 15.31 ± 1.24 | 3.12 |
| GG | 10.03 ± 0.81 | 13.67 ± 0.89 | 244.70 ± 19.43 | 12.77 ± 1.01 | 2.86 |
| GP | 3.01 ± 0.22 | 3.01 ± 0.22 | 62.82 ± 5.62 | 6.41 ± 0.57 | 5.65 |
| GN | 11.08 ± 0.92 | 12.15 ± 1.01 | 184.11 ± 13.78 | 9.73 ± 0.73 | 2.46 |
| PD | 6.02 ± 0.58 | 7.94 ± 0.63 | 194.52 ± 9.75 | 12.27 ± 0.62 | 3.68 |
| PG | 5.73 ± 0.56 | 6.20 ± 0.71 | 150.28 ± 16.02 | 9.40 ± 1.00 | 3.23 |
| PP | 5.99 ± 0.49 | 5.99 ± 0.49 | 99.74 ± 7.93 | 7.61 ± 0.58 | 3.35 |
| PN | 18.20 ± 1.74 | 18.20 ± 1.19 | 238.61 ± 31.87 | 10.79 ± 1.44 | 1.68 |
| ND | 15.39 ± 1.17 | 23.77 ± 1.82 | 674.21 ± 46.30 | 24.62 ± 1.69 | 2.20 |
| NG | 11.59 ± 0.93 | 15.69 ± 1.09 | 429.34 ± 33.14 | 15.72 ± 1.21 | 2.01 |
| NP | 3.12 ± 0.39 | 3.12 ± 0.32 | 75.90 ± 6.92 | 6.75 ± 0.61 | 5.50 |
| NN | 7.61 ± 0.61 | 9.19 ± 0.74 | 101.09 ± 9.82 | 4.97 ± 0.48 | 2.60 |
| Source | DF | Contribution (%) | F-Value | p-Value | |
|---|---|---|---|---|---|
| First Peak (kN) | Internal TPMS Type | 3 | 0.50 | 0.15 | 0.925 |
| Infill Density for Internal TPMS | 1 | 3.01 | 2.75 | 0.141 | |
| External TPMS Type | 3 | 46.62 | 14.23 | 0.002 | |
| Infill Density for External TPMS | 1 | 42.23 | 38.67 | 0.000 | |
| Error | 7 | 7.64 | |||
| Total | 15 | 100 | |||
| Maximum Force (kN) | Internal TPMS Type | 3 | 5.34 | 1.69 | 0.254 |
| Infill Density for Internal TPMS | 1 | 4.41 | 4.19 | 0.080 | |
| External TPMS Type | 3 | 38.35 | 12.16 | 0.004 | |
| Infill Density for External TPMS | 1 | 44.54 | 42.38 | 0.000 | |
| Error | 7 | 7.36 | |||
| Total | 15 | 100 | |||
| Energy Absorption (kJ) | Internal TPMS Type | 3 | 14.06 | 4.04 | 0.049 |
| Infill Density for Internal TPMS | 1 | 15.18 | 13.07 | 0.009 | |
| External TPMS Type | 3 | 23.80 | 6.83 | 0.017 | |
| Infill Density for External TPMS | 1 | 38.84 | 33.46 | 0.001 | |
| Error | 7 | 8.13 | |||
| Total | 15 | 100 | |||
| Specific Energy Absorption (kJ/g) | Internal TPMS Type | 3 | 5.69 | 1.22 | 0.371 |
| Infill Density for Internal TPMS | 1 | 13.44 | 8.65 | 0.022 | |
| External TPMS Type | 3 | 33.56 | 7.20 | 0.015 | |
| Infill Density for External TPMS | 1 | 36.42 | 23.43 | 0.002 | |
| Error | 7 | 10.88 | |||
| Total | 15 | 100 | |||
| Surface/Volume Ratio (1/mm) | Internal TPMS Type | 3 | 4.99 | 2.08 | 0.192 |
| Infill Density for Internal TPMS | 1 | 5.20 | 6.50 | 0.038 | |
| External TPMS Type | 3 | 43.74 | 18.22 | 0.001 | |
| Infill Density for External TPMS | 1 | 40.48 | 50.60 | 0.000 | |
| Error | 7 | 5.60 | |||
| Total | 15 | 100 |
| S | R2 (%) | Adj. R2 (%) | |
|---|---|---|---|
| First Peak (kN) | 2.0068 | 92.36 | 83.62 |
| Maximum Force (kN) | 2.4405 | 92.64 | 84.23 |
| Energy Absorption (kJ) | 65.9120 | 91.87 | 82.59 |
| Specific Energy Absorption (kJ/g) | 2.2826 | 88.12 | 76.69 |
| Surface/Volume Ratio (1/mm) | 0.4398 | 94.40 | 88.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Karamanli, İ.A. Investigation on Mechanical Properties of Functional Graded Hybrid TPMS Structures Inspired Bone Scaffolds. Polymers 2026, 18, 236. https://doi.org/10.3390/polym18020236
Karamanli İA. Investigation on Mechanical Properties of Functional Graded Hybrid TPMS Structures Inspired Bone Scaffolds. Polymers. 2026; 18(2):236. https://doi.org/10.3390/polym18020236
Chicago/Turabian StyleKaramanli, İsmail Aykut. 2026. "Investigation on Mechanical Properties of Functional Graded Hybrid TPMS Structures Inspired Bone Scaffolds" Polymers 18, no. 2: 236. https://doi.org/10.3390/polym18020236
APA StyleKaramanli, İ. A. (2026). Investigation on Mechanical Properties of Functional Graded Hybrid TPMS Structures Inspired Bone Scaffolds. Polymers, 18(2), 236. https://doi.org/10.3390/polym18020236






