Enhancing Devulcanizing Degree and Efficiency of Reclaimed Rubber by Using Alcoholic Amines as the Devulcanizing Agent in Low-Temperature Mechano–Chemical Process
Abstract
1. Introduction
2. Materials and Methods
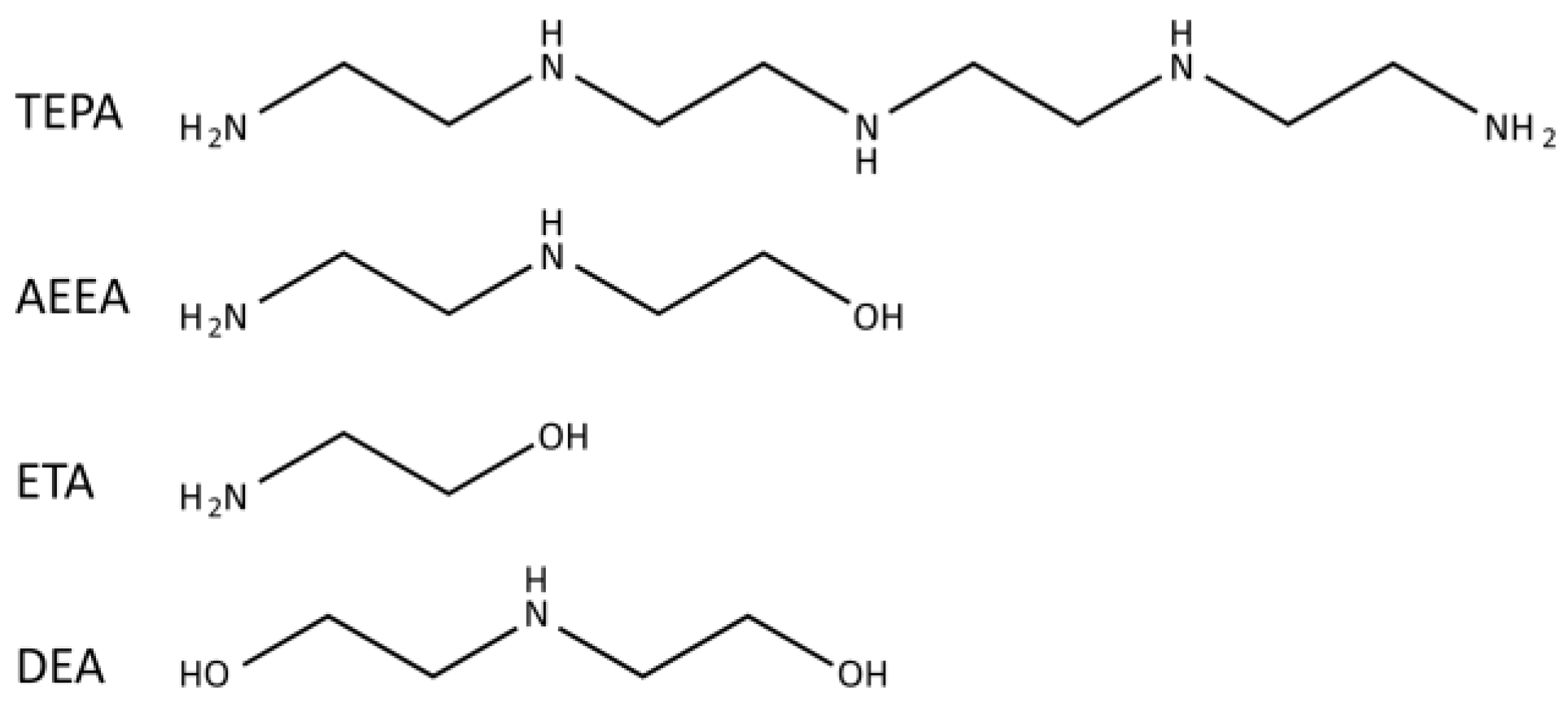
2.1. Materials
2.2. Experimental Procedure
2.2.1. Devulcanization
2.2.2. Vulcanization of RRs
2.3. Characterization
3. Results and Discussion
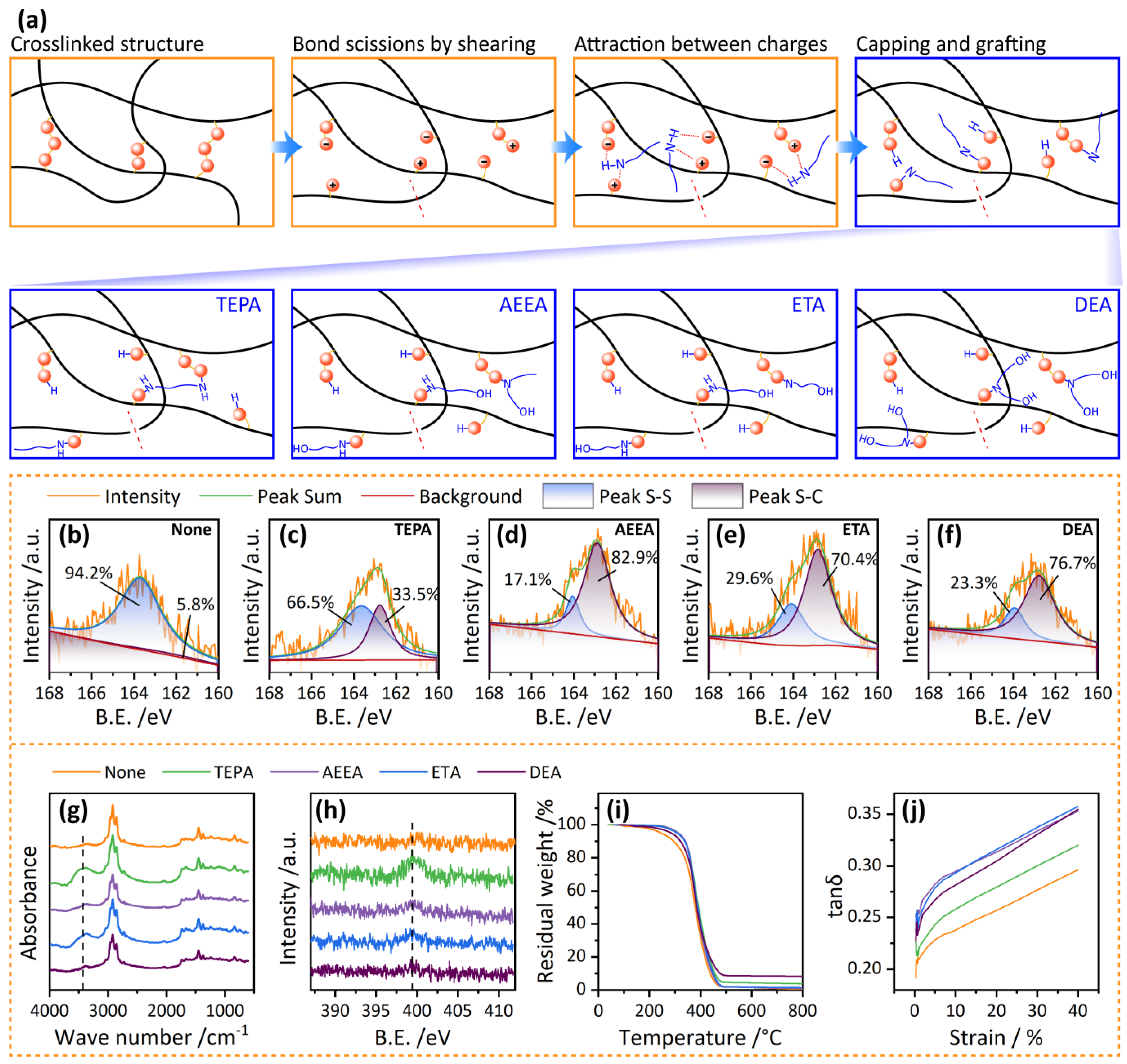
3.1. Mechanism Analysis
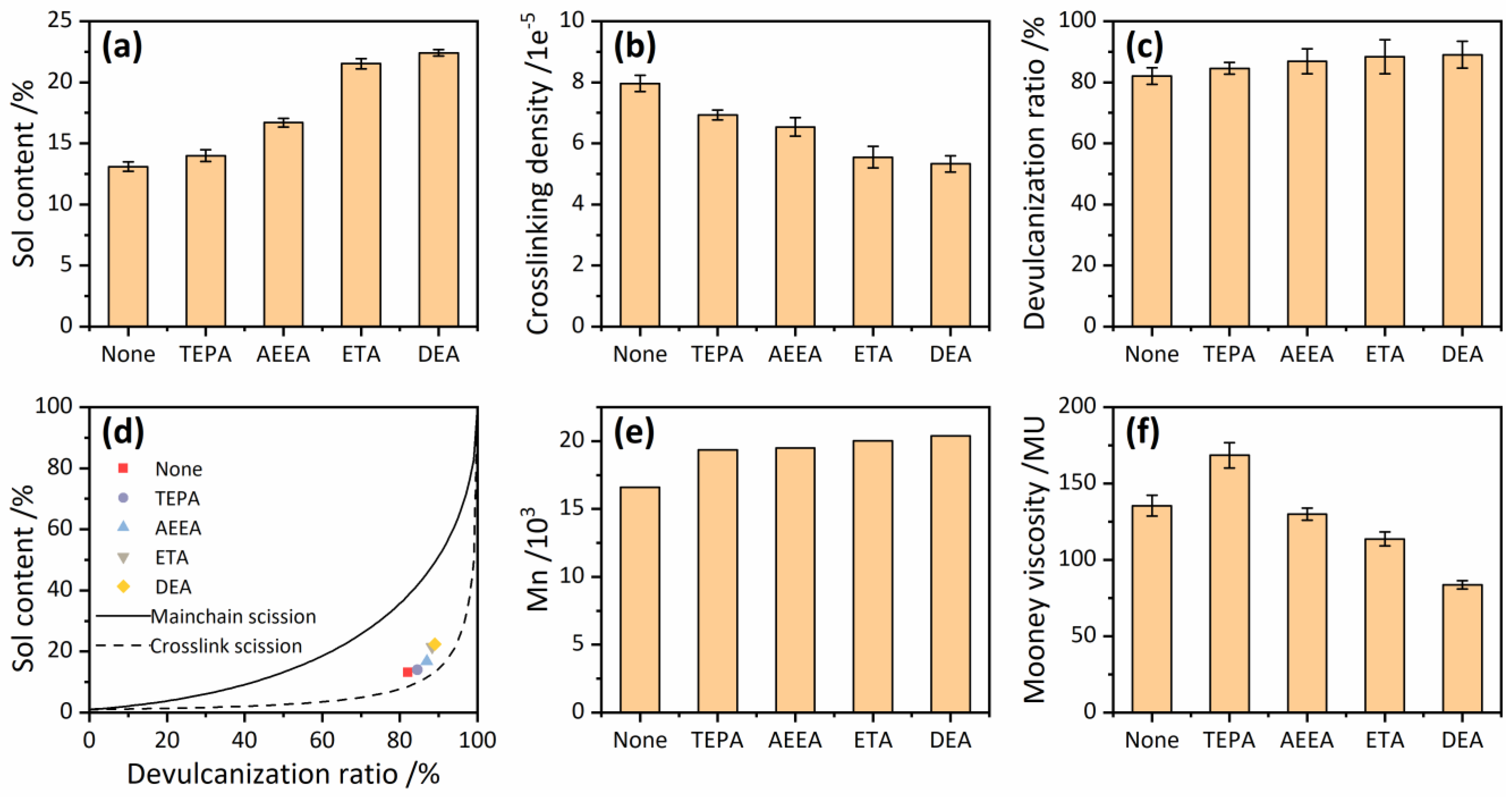
3.2. Devulcanization Degree
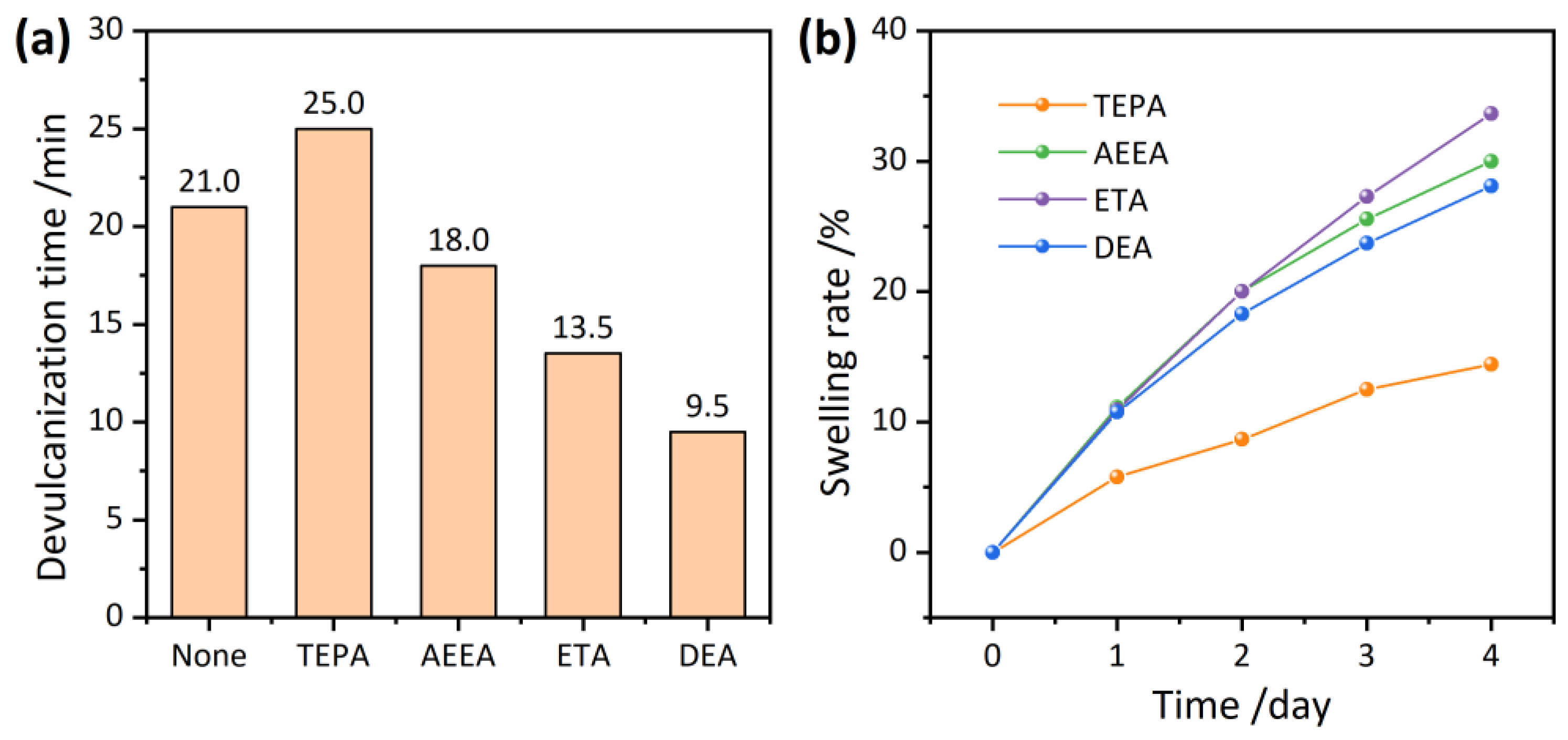
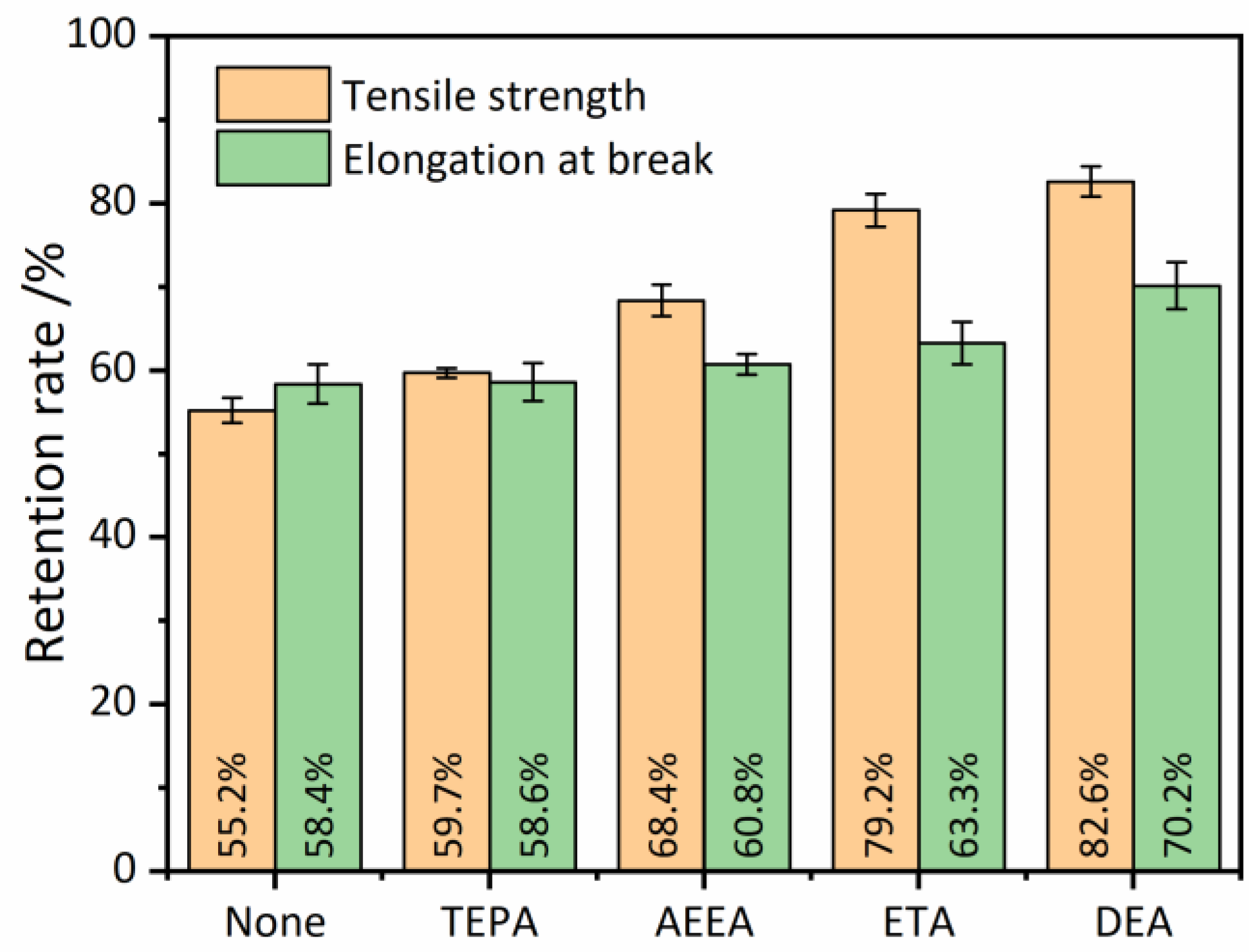
3.3. Devulcanizing Efficiency
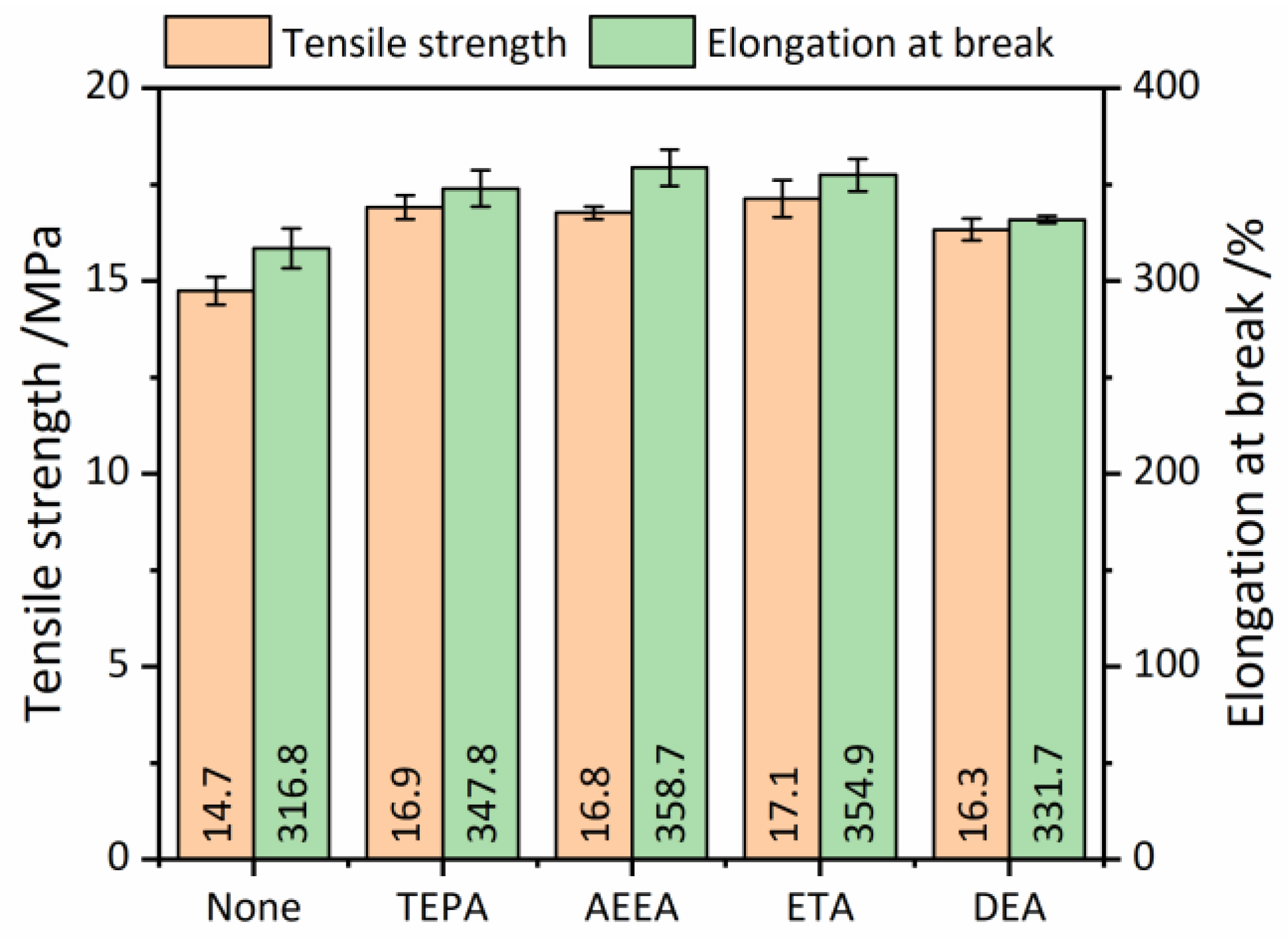
3.4. Properties of RRs
3.4.1. Curing Characteristics
3.4.2. Tensile Properties
3.4.3. Anti-Aging Property
3.4.4. Dynamic Mechanical Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Abbas-Abadi, M.S.; Kusenberg, M.; Shirazi, H.M.; Goshayeshi, B.; Van Geem, K.M. Towards full recyclability of end-of-life tires: Challenges and opportunities. J. Clean Prod. 2022, 374, 134036. [Google Scholar] [CrossRef]
- Advincula, P.A.; Luong, D.X.; Chen, W.; Raghuraman, S.; Shahsavari, R.; Tour, J.M. Flash graphene from rubber waste. Carbon 2021, 178, 649–656. [Google Scholar] [CrossRef]
- Wisniewska, P.; Wang, S.F.; Formela, K. Waste tire rubber devulcanization technologies: State-of-the-art, limitations and future perspectives. Waste Manag. 2022, 150, 174–184. [Google Scholar] [CrossRef]
- Hittini, W.; Mourad, A.-H.I.; Abu-Jdayil, B. Utilization of devulcanized waste rubber tire in development of heat insulation composite. J. Clean Prod. 2021, 280, 124492. [Google Scholar] [CrossRef]
- Hong, Y.J.; Jeong, K.M.; Saha, P.; Suh, J.; Kim, J.K. Processing and characterization of microwave and ultrasonically treated waste-EPDM/LDPE polymer composites. Polym. Eng. Sci. 2015, 55, 533–540. [Google Scholar] [CrossRef]
- Li, X.; Deng, X.Q.; Dong, C. Effect of Temperature on Devulcanization of Waste Sidewall Rubber by Supercritical Ethanol. J. Braz. Chem. Soc. 2018, 29, 2169–2179. [Google Scholar] [CrossRef]
- Chittella, H.; Yoon, L.W.; Ramarad, S.; Lai, Z.W. Rubber waste management: A review on methods, mechanism, and prospects. Polym. Degrad. Stab. 2021, 194, 22. [Google Scholar] [CrossRef]
- Zhao, X.P.; Hu, H.; Zhang, D.K.; Zhang, Z.; Peng, S.X.; Sun, Y.M. Curing behaviors, mechanical properties, dynamic mechanical analysis and morphologies of natural rubber vulcanizates containing reclaimed rubber. e-Polymers 2019, 19, 482–488. [Google Scholar] [CrossRef]
- Sabzekar, M.; Zohuri, G.; Chenar, M.P.; Mortazavi, S.M.; Kariminejad, M.; Asadi, S. A new approach for reclaiming of waste automotive EPDM rubber using waste oil. Polym. Degrad. Stab. 2016, 129, 56–62. [Google Scholar] [CrossRef]
- Costamagna, M.; Brunella, V.; Luda, M.P.; Romagnolli, U.; Muscato, B.; Girotto, M.; Baricco, M.; Rizzi, P. Environmental assessment of rubber recycling through an innovative thermo-mechanical devulcanization process using a co-rotating twin-screw extruder. J. Clean Prod. 2022, 348, 131352. [Google Scholar] [CrossRef]
- Sripornsawat, B.; Saiwari, S.; Pichaiyut, S.; Nakason, C. Influence of ground tire rubber devulcanization conditions on properties of its thermoplastic vulcanizate blends with copolyester. Eur. Polym. J. 2016, 85, 279–297. [Google Scholar] [CrossRef]
- Liu, H.L.; Wang, X.P.; Jia, D.M. Recycling of waste rubber powder by mechano-chemical modification. J. Clean Prod. 2020, 245, 118716. [Google Scholar] [CrossRef]
- Formela, K.; Hejna, A.; Zedler, L.; Colom, X.; Canavate, J. Microwave treatment in waste rubber recycling-recent advances and limitations. Express Polym. Lett. 2019, 13, 565–588. [Google Scholar] [CrossRef]
- Vahdatbin, M.; Jamshidi, M. Using chemical agent in microwave assisted devulcanization of NR/SBR blends: An effective recycling method. Resour. Conserv. Recy. 2022, 179, 106045. [Google Scholar] [CrossRef]
- Karabork, F.; Pehlivan, E.; Akdemir, A. Characterization of styrene butadiene rubber and microwave devulcanized ground tire rubber composites. J. Polym. Eng. 2014, 34, 543–554. [Google Scholar] [CrossRef]
- Molanorouzi, M.; Mohaved, S.O. Reclaiming waste tire rubber by an irradiation technique. Polym. Degrad. Stab. 2016, 128, 115–125. [Google Scholar] [CrossRef]
- Gumede, J.I.; Hlangothi, B.G.; Woolard, C.D.; Hlangothi, S.P. Organic chemical devulcanization of rubber vulcanizates in supercritical carbon dioxide and associated less eco-unfriendly approaches: A review. Waste Manag. Res. 2022, 40, 490–503. [Google Scholar] [CrossRef]
- Sabzekar, M.; Chenar, M.P.; Mortazavi, S.M.; Kariminejad, M.; Asadi, S.; Zohuri, G. Influence of process variables on chemical devulcanization of sulfur-cured natural rubber. Polym. Degrad. Stab. 2015, 118, 88–95. [Google Scholar] [CrossRef]
- Ghorai, S.; Bhunia, S.; Roy, M.; De, D. Mechanochemical devulcanization of natural rubber vulcanizate by dual function disulfide chemicals. Polym. Degrad. Stab. 2016, 129, 34–46. [Google Scholar] [CrossRef]
- El-Nemr, K.F.; Raslan, H.A.; Ali, M.A.M.; Hasan, M.M. Innovative gamma rays irradiated styrene butadiene rubber/reclaimed waste tire rubber blends: A comparative study using mechano-chemical and microwave devulcanizing methods. J. Polym. Eng. 2020, 40, 267–277. [Google Scholar] [CrossRef]
- Seghar, S.; Asaro, L.; Rolland-Monnet, M.; Hocine, N.A. Thermo-mechanical devulcanization and recycling of rubber industry waste. Resour. Conserv. Recycl. 2019, 144, 180–186. [Google Scholar] [CrossRef]
- Guo, L.; Lv, D.; Ren, D.; Qu, L.; Wang, W.; Hao, K.; Guo, X.; Chen, T.; Sun, J.; Wang, C.; et al. Effectiveness of original additives in waste rubbers for revulcanization after reclamation with a low-temperature mechanochemical devulcanization method. J. Clean Prod. 2021, 297, 126620. [Google Scholar] [CrossRef]
- Guo, L.; Wang, C.; Lv, D.; Ren, D.; Zhai, T.; Sun, C.; Liu, H. Rubber reclamation with high bond-breaking selectivity using a low-temperature mechano-chemical devulcanization method. J. Clean Prod. 2021, 279, 123266. [Google Scholar] [CrossRef]
- Gagol, M.; Boczkaj, G.; Haponiuk, J.; Formela, K. Investigation of volatile low molecular weight compounds formed during continuous reclaiming of ground tire rubber. Polym. Degrad. Stab. 2015, 119, 113–120. [Google Scholar] [CrossRef]
- Shi, J.; Jiang, K.; Ren, D.; Zou, H.; Wang, Y.; Lv, X.; Zhang, L. Structure and performance of reclaimed rubber obtained by different methods. J. Appl. Polym. Sci. 2013, 129, 999–1007. [Google Scholar] [CrossRef]
- Ghosh, J.; Ghorai, S.; Bhunia, S.; Roy, M.; De, D. The Role of Devulcanizing Agent for Mechanochemical Devulcanization of Styrene Butadiene Rubber Vulcanizate. Polym. Eng. Sci. 2018, 58, 74–85. [Google Scholar] [CrossRef]
- Azarabtin, S.; Mousavi, S.R.; Khameneh, R.J.; Mortazavi, S.M.M.; Ehsani, M.; Ranjbar, H.; Khonakdar, H.A. Effect of different devulcanization agents on the mechano-chemical devulcanization process of waste tires. Mater. Today Commun. 2022, 33, 104416. [Google Scholar] [CrossRef]
- Bockstal, L.; Berchem, T.; Schmetz, Q.; Richel, A. Devulcanisation and reclaiming of tires and rubber by physical and chemical processes: A review. J. Clean Prod. 2019, 236, 16. [Google Scholar] [CrossRef]
- Kojima, M.; Ogawa, K.; Mizoshima, H.; Tosaka, M.; Kohjiya, S.; Ikeda, Y. Devulcanization of sulfur-cured isoprene rubber in supercritical carbon dioxide. Rubber Chem. Technol. 2003, 76, 957–968. [Google Scholar] [CrossRef]
- Jiang, C.; Zhang, Y.S.; Ma, L.; Zhou, L.; He, H. Tailoring the properties of ground tire rubber/high-density polyethylene blends by combining surface devulcanization and in-situ grafting technology. Mater. Chem. Phys. 2018, 220, 161–170. [Google Scholar] [CrossRef]
- Guo, L.; Ren, D.; Wang, W.; Hao, K.; Guo, X.; Liu, F.; Xu, Y.; Liu, M.; Liu, H. Low-Temperature Mechano-Chemical Rubber Reclamation Using Terpinene as a Swelling Agent to Enhance Bond-Breaking Selectivity. Polymers 2021, 13, 4272. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hao, K.; Guo, X.; Liu, F.; Xu, Y.; Guo, S.; Bai, L.; Liu, G.; Qu, L.; Liu, M.; et al. Mechano-chemical rubber reclamation using aminolysis products of waste flexible polyurethane foams as the devulcanizing agent. J. Clean Prod. 2023, 384, 135421. [Google Scholar] [CrossRef]
- ASTM D5289-2007a; Standard Test Method for Rubber Property-Vulcanization Using Rotorless Cure Meters. ASTM: Conshohocken, PA, USA, 2007.
- ASTM-D2240; Standard Test Method for Rubber Property-Durometer Hardness. ASTM: Conshohocken, PA, USA, 2021.
- ASTM D 2084; Standard Test Method for Rubber Property-Vulcanization Using Oscillating Disk Cure Meter. ASTM: Conshohocken, PA, USA, 2019.
- ASTM D 1646; Standard Test Methods for Rubber—Viscosity, Stress Relaxation, and Pre-Vulcanization Characteristics (Mooney Viscometer). ASTM: Conshohocken, PA, USA, 2019.
- Li, Y.; Zhao, S.; Wang, Y. Improvement of the properties of natural rubber/ground tire rubber composites through biological desulfurization of GTR. J. Polym. Res. 2012, 19, 9864. [Google Scholar] [CrossRef]
- Wagner, A.J.; Wolfe, G.M.; Fairbrother, D.H. Reactivity of vapor-deposited metal atoms with nitrogen-containing polymers and organic surfaces studied by in situ XPS. Appl. Surf. Sci. 2003, 219, 317–328. [Google Scholar] [CrossRef]
- Tao, G.L.; He, Q.H.; Xia, Y.P.; Jia, G.C.; Yang, H.C.; Ma, W.Z. The effect of devulcanization level on mechanical properties of reclaimed rubber by thermal-mechanical shearing devulcanization. J. Appl. Polym. Sci. 2013, 129, 2598–2605. [Google Scholar] [CrossRef]

| Samples | MH/N m | ML/N m | ΔM/N m | T10/min | T90/min |
|---|---|---|---|---|---|
| None | 14.67 ± 0.82 | 2.78 ± 0.19 | 11.89 ± 0.74 | 2.94 ± 0.21 | 5.68 ± 0.32 |
| TEPA | 15.78 ± 0.75 | 2.59 ± 0.15 | 13.29 ± 0.83 | 2.49 ± 0.16 | 4.30 ± 0.25 |
| AEEA | 15.64 ± 0.44 | 2.21 ± 0.13 | 13.21 ± 0.54 | 2.42 ± 0.18 | 3.59 ± 0.21 |
| ETA | 15.07 ± 0.66 | 2.14 ± 0.15 | 12.93 ± 0.80 | 2.44 ± 0.10 | 2.49 ± 0.27 |
| DEA | 14.48 ± 0.23 | 2.09 ± 0.09 | 12.39 ± 0.27 | 2.41 ± 0.16 | 3.84 ± 0.29 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, L.; Bai, L.; Zhao, J.; Liu, K.; Jian, X.; Chai, H.; Liu, F.; Guo, S.; Liu, G.; Liu, H. Enhancing Devulcanizing Degree and Efficiency of Reclaimed Rubber by Using Alcoholic Amines as the Devulcanizing Agent in Low-Temperature Mechano–Chemical Process. Polymers 2024, 16, 395. https://doi.org/10.3390/polym16030395
Guo L, Bai L, Zhao J, Liu K, Jian X, Chai H, Liu F, Guo S, Liu G, Liu H. Enhancing Devulcanizing Degree and Efficiency of Reclaimed Rubber by Using Alcoholic Amines as the Devulcanizing Agent in Low-Temperature Mechano–Chemical Process. Polymers. 2024; 16(3):395. https://doi.org/10.3390/polym16030395
Chicago/Turabian StyleGuo, Lei, Lichen Bai, Jinyang Zhao, Kexin Liu, Xingao Jian, Hailin Chai, Fumin Liu, Shouyun Guo, Gongxu Liu, and Haichao Liu. 2024. "Enhancing Devulcanizing Degree and Efficiency of Reclaimed Rubber by Using Alcoholic Amines as the Devulcanizing Agent in Low-Temperature Mechano–Chemical Process" Polymers 16, no. 3: 395. https://doi.org/10.3390/polym16030395
APA StyleGuo, L., Bai, L., Zhao, J., Liu, K., Jian, X., Chai, H., Liu, F., Guo, S., Liu, G., & Liu, H. (2024). Enhancing Devulcanizing Degree and Efficiency of Reclaimed Rubber by Using Alcoholic Amines as the Devulcanizing Agent in Low-Temperature Mechano–Chemical Process. Polymers, 16(3), 395. https://doi.org/10.3390/polym16030395






