Electrically Conductive Natural Rubber Composite Films Reinforced with Graphite Platelets
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
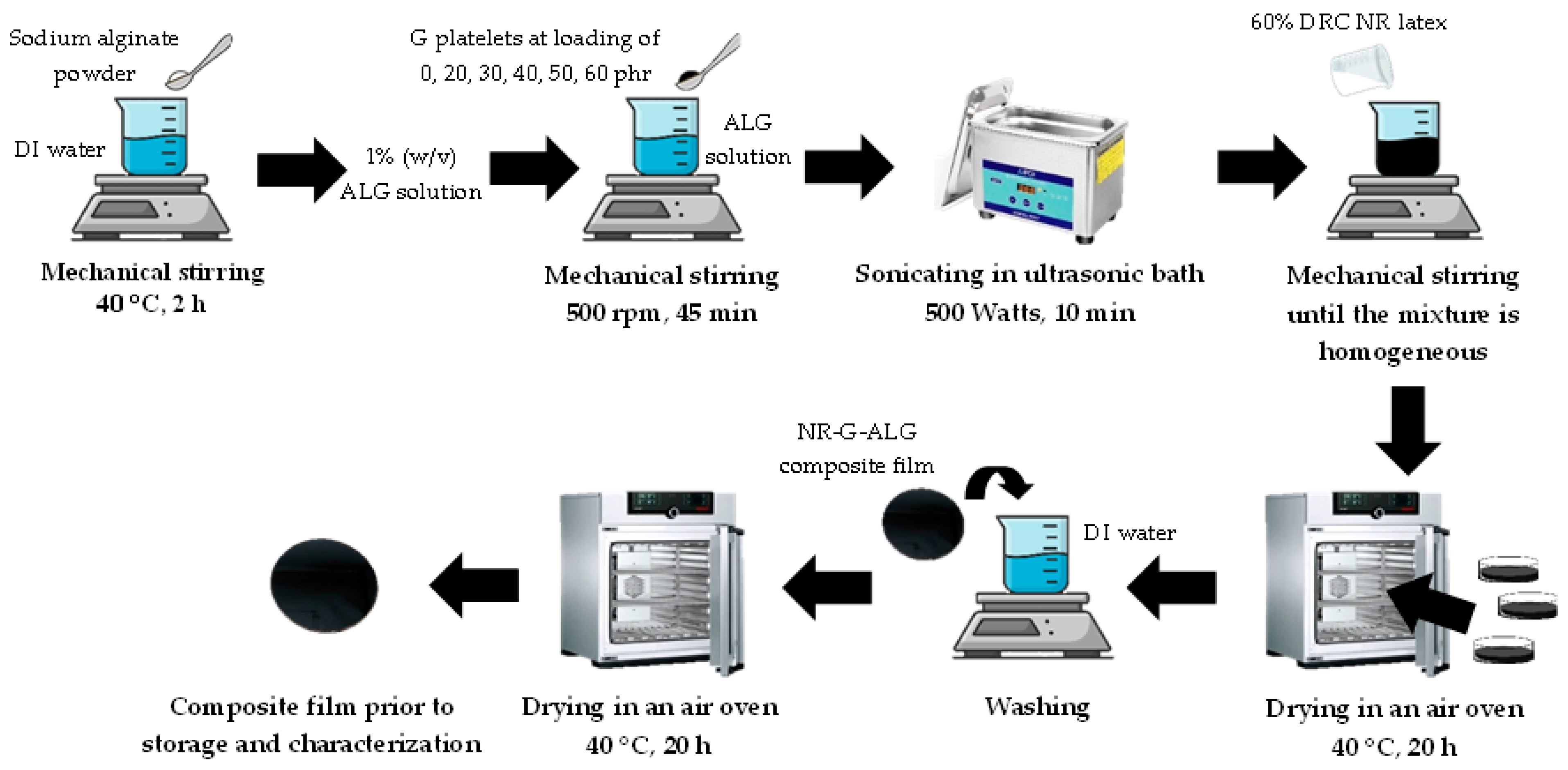
2.2. Fabrication of Natural Rubber, Graphite, and Alginate (NR-G-ALG) Composite Films
2.3. Characterization
3. Results and Discussion
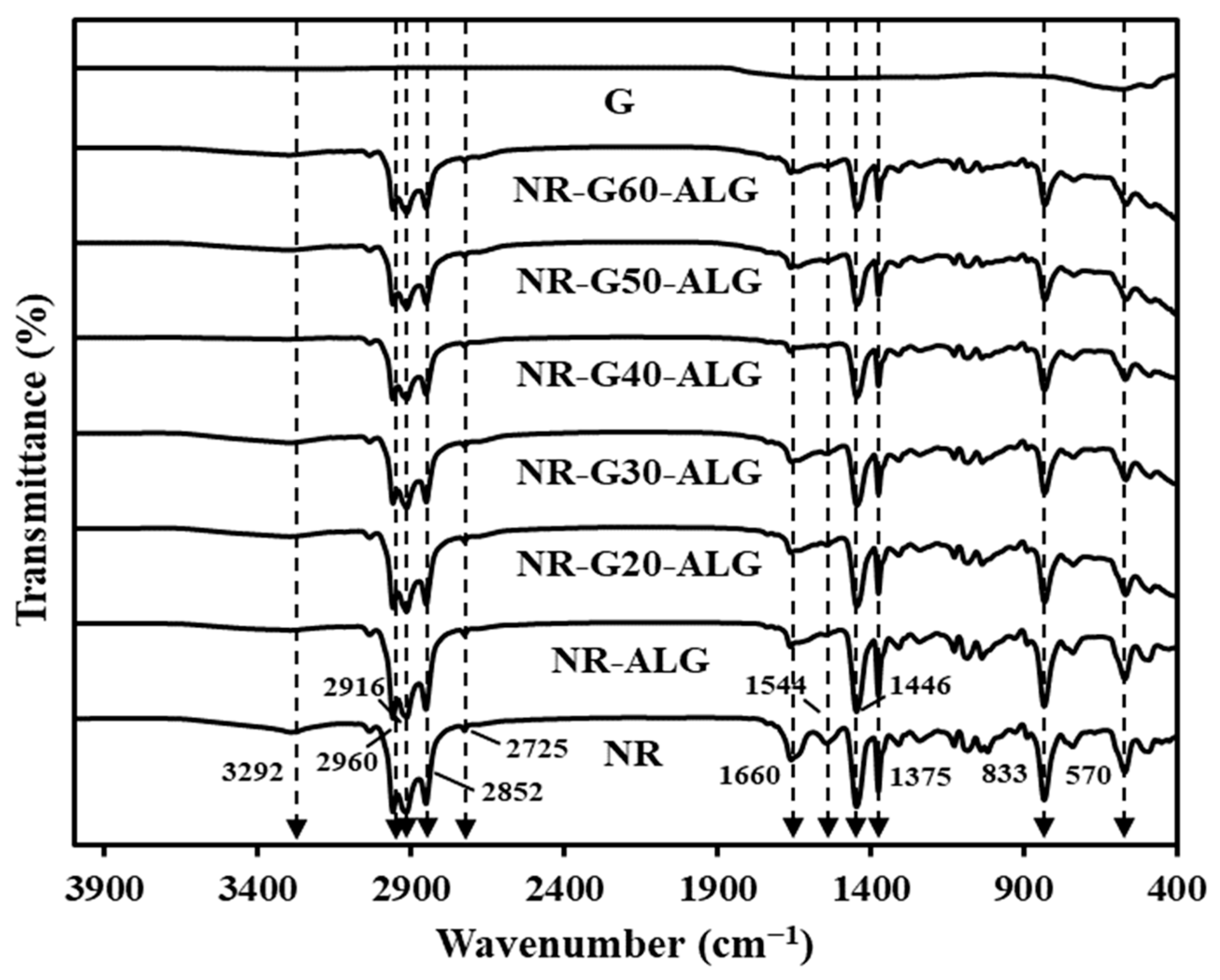
3.1. Fourier Transform Infrared (FTIR) Spectroscopy
3.2. Field Emission-Scanning Electron Microscope (FE-SEM)
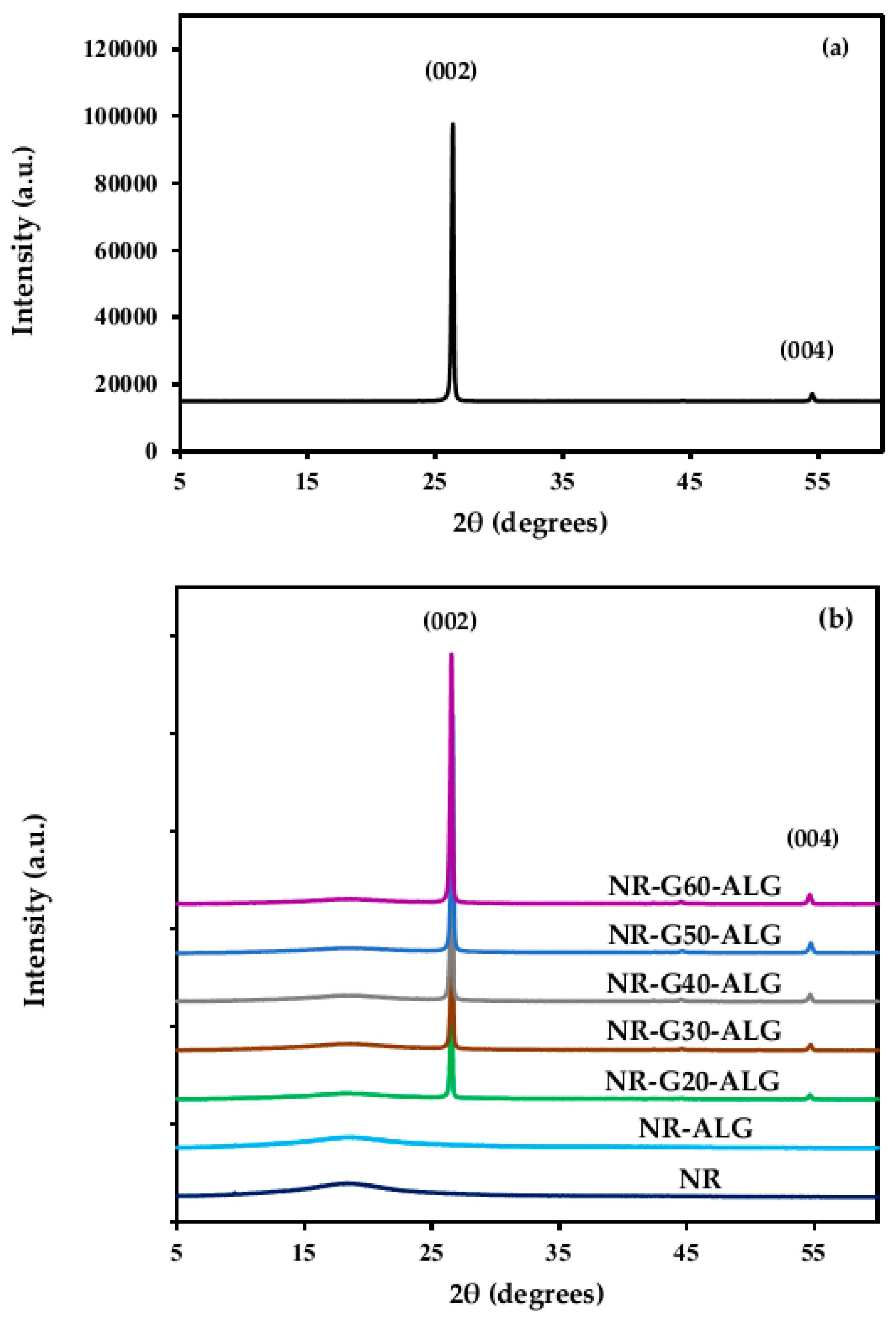
3.3. X-ray Diffraction (XRD)
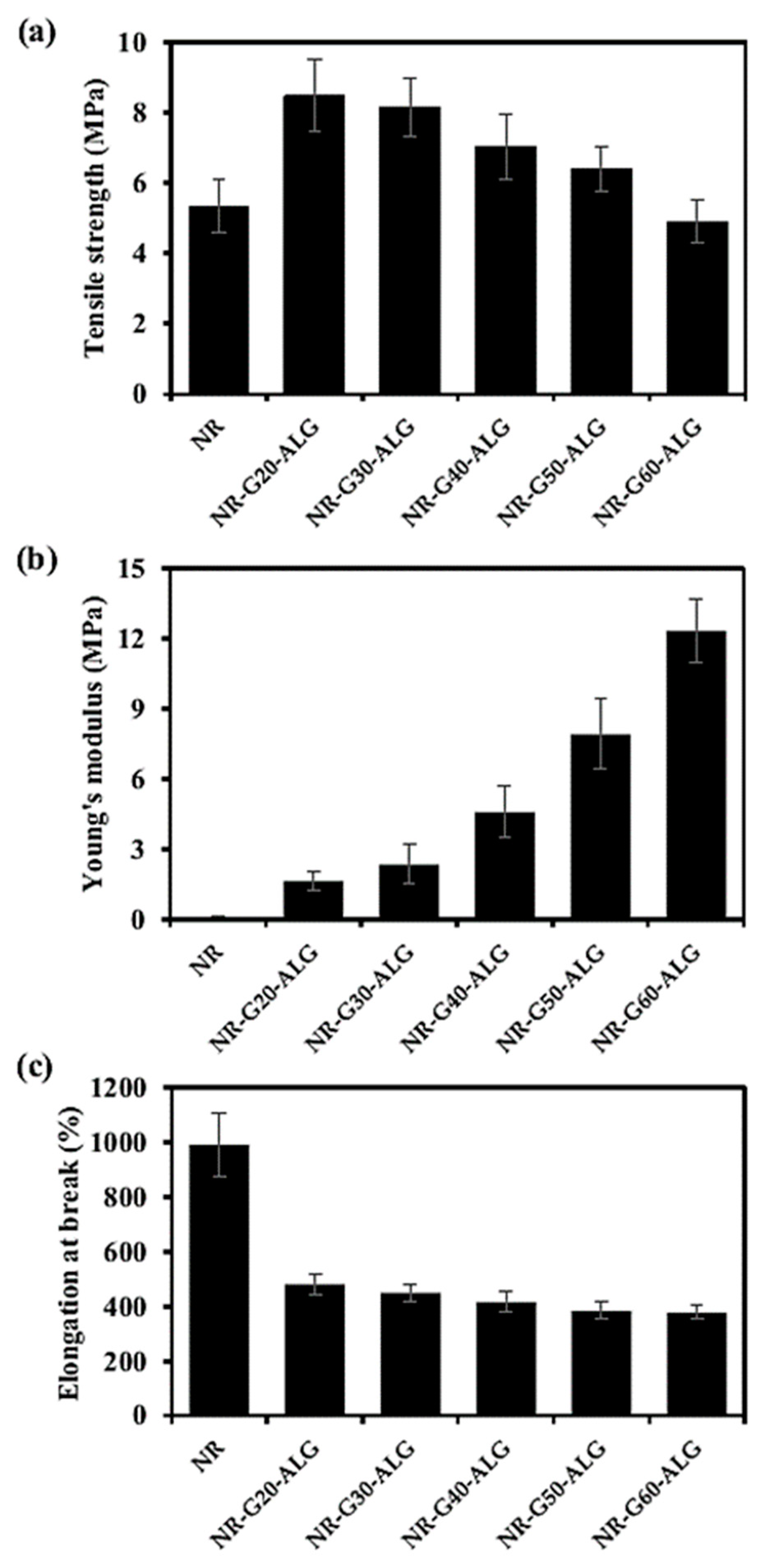
3.4. Mechanical Properties
3.5. Thermal Properties
3.6. Water Absorption Capacity (WAC)
3.7. Toluene Uptake
3.8. Biodegradation in Soil
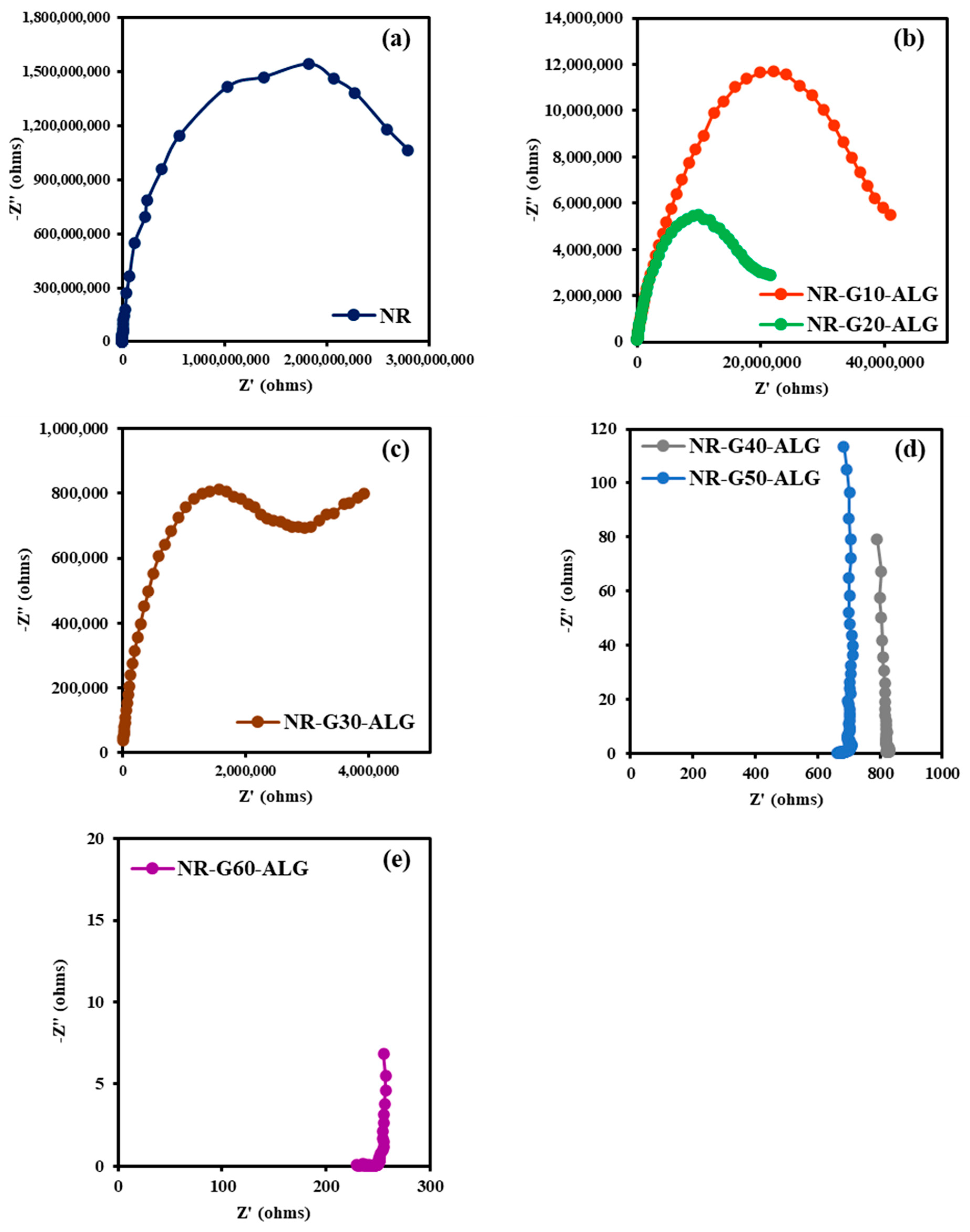
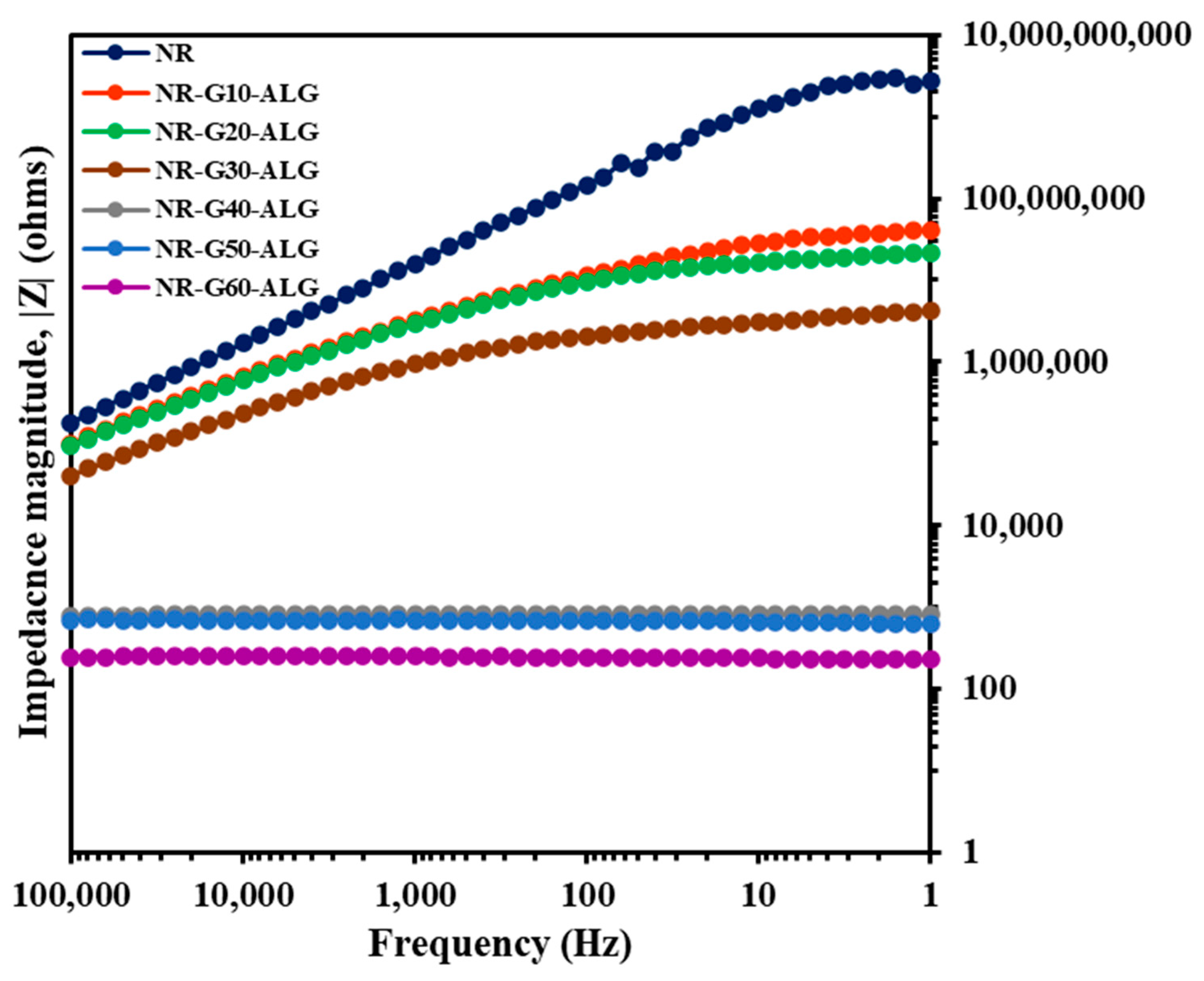
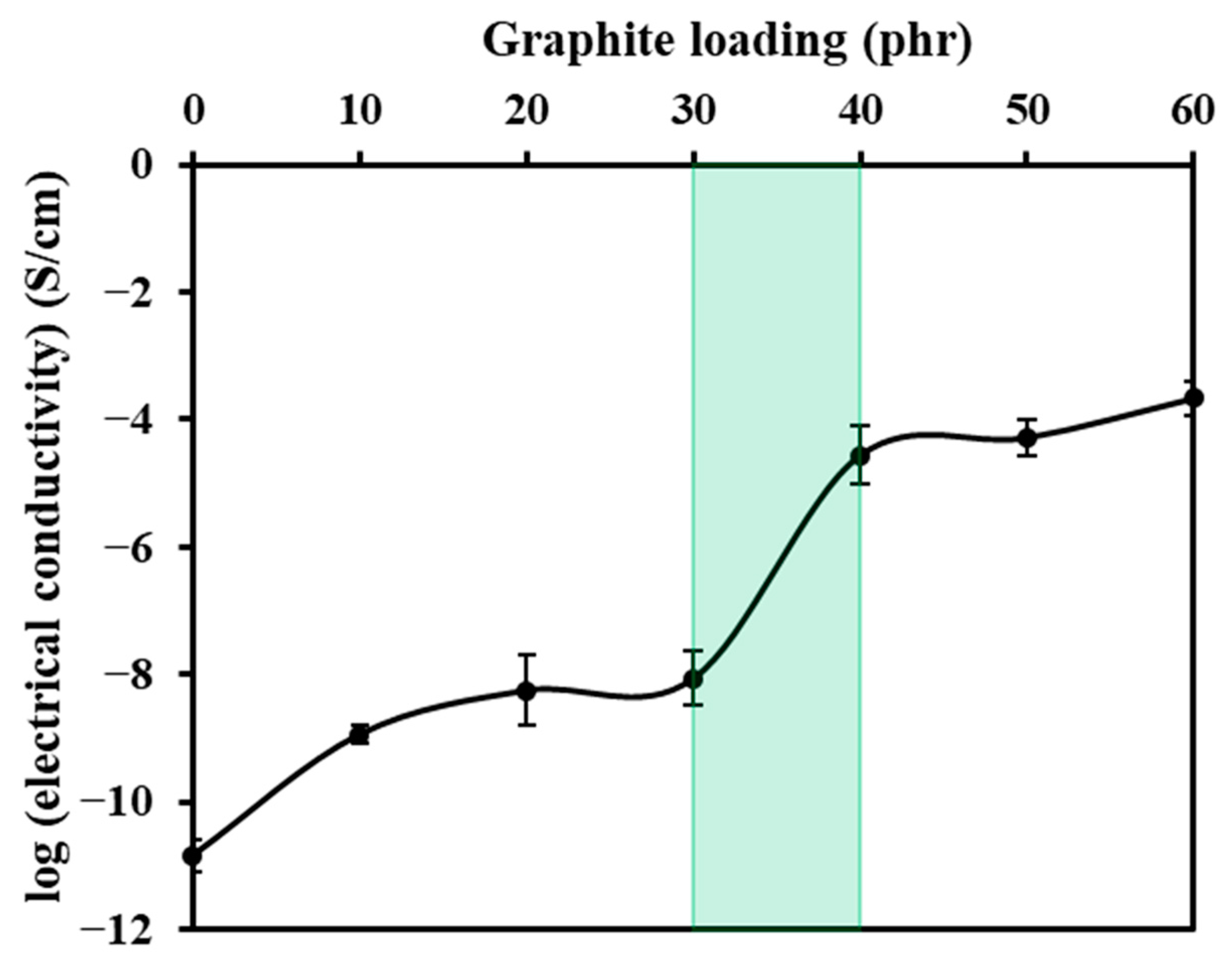
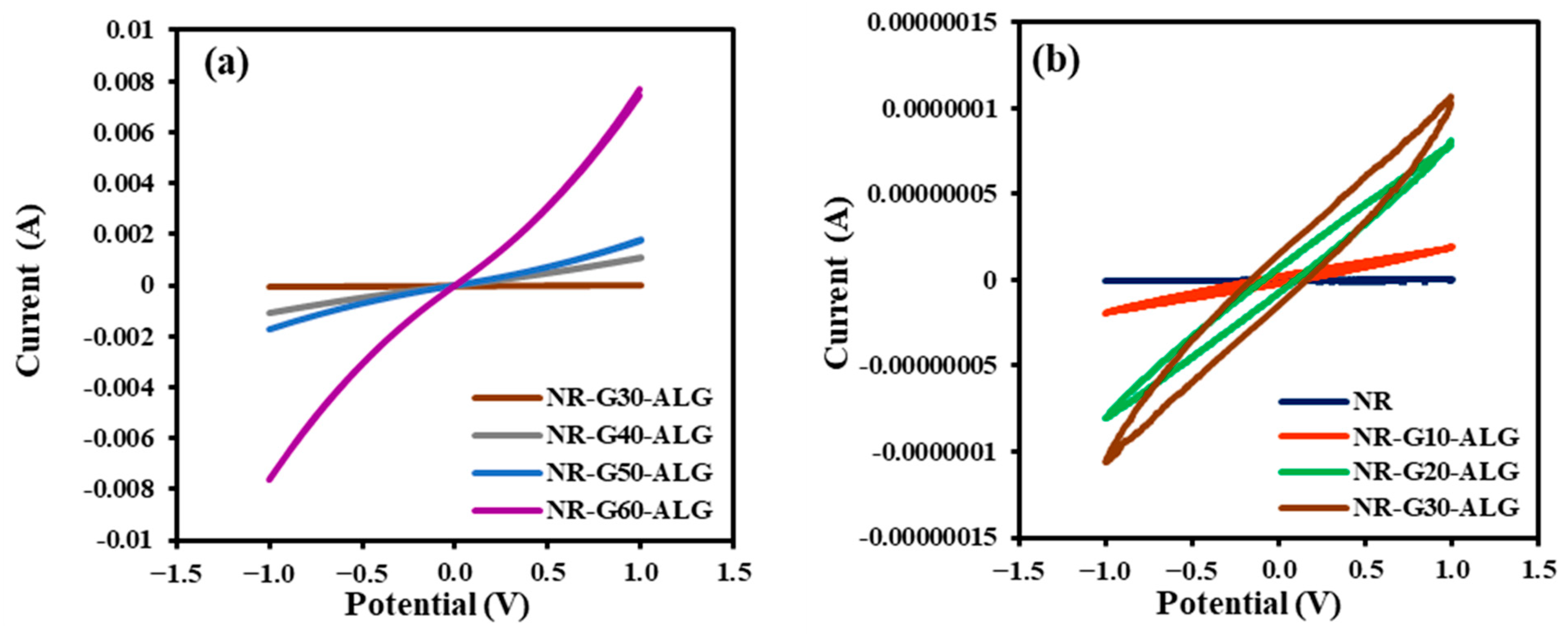
3.9. Electrical Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arif, Z.U.; Khalid, M.Y.; Sheikh, M.F.; Zolfagharian, A.; Bodaghi, M. Biopolymeric sustainable materials and their emerging applications. J. Environ. Chem. Eng. 2022, 10, 108159. [Google Scholar] [CrossRef]
- Herculano, R.; Tzu, L.; Silva, C.; Brunello, C.; De Queiroz, A.; Kinoshita, A.; Graeff, C. Nitric Oxide Release Using Natural Rubber Latex as Matrix. Mater. Res.-Ibero-Am. J. Mater. 2011, 14, 355–359. [Google Scholar] [CrossRef]
- Tran, T.N.; Pasetto, P.; Pichon, C.; Bruant, D.; Brotons, G.; Nourry, A. Natural rubber based films integrating Zosteric acid analogues as bioactive monomers. React. Funct. Polym. 2019, 144, 104343. [Google Scholar] [CrossRef]
- Sowcharoensuk, C. Industry Outlook 2022–2024: Natural Rubber. Available online: https://www.krungsri.com/en/research/industry/industry-outlook/agriculture/rubber/io/rubber-2022 (accessed on 23 November 2023).
- Doner, R. The Risks of Unbalanced Rubber Sector Development. Available online: https://www.bangkokpost.com/opinion/opinion/369082/the-risks-of-unbalanced-rubber-sector-development (accessed on 11 September 2023).
- Reowdecha, M.; Dittanet, P.; Sae-oui, P.; Loykulnant, S.; Prapainainar, P. Film and latex forms of silica-reinforced natural rubber composite vulcanized using electron beam irradiation. Heliyon 2021, 7, e07176. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.-C.; Zhu, D.; Xie, W.-Y.; Xia, J.-H.; He, M.-F.; Liao, S. In-situ observation of spatial organization of natural rubber latex particles and exploring the relationship between particle size and mechanical properties of natural rubber. Ind. Crops Prod. 2022, 180, 114737. [Google Scholar] [CrossRef]
- Phetarporn, V.; Loykulnant, S.; Kongkaew, C.; Seubsai, A.; Prapainainar, P. Composite properties of graphene-based materials/natural rubber vulcanized using electron beam irradiation. Mater. Today Commun. 2019, 19, 413–424. [Google Scholar] [CrossRef]
- Mohammad, H.; Stepashkin, A.A.; Tcherdyntsev, V.V. Effect of Graphite Filler Type on the Thermal Conductivity and Mechanical Behavior of Polysulfone-Based Composites. Polymers 2022, 14, 399. [Google Scholar] [CrossRef]
- Khan, A.; Savi, P.; Quaranta, S.; Rovere, M.; Giorcelli, M.; Tagliaferro, A.; Rosso, C.; Jia, C.Q. Low-Cost Carbon Fillers to Improve Mechanical Properties and Conductivity of Epoxy Composites. Polymers 2017, 9, 642. [Google Scholar] [CrossRef]
- Qin, M.; Xu, Y.; Cao, R.; Feng, W.; Chen, L. Efficiently Controlling the 3D Thermal Conductivity of a Polymer Nanocomposite via a Hyperelastic Double-Continuous Network of Graphene and Sponge. Adv. Funct. Mater. 2018, 28, 1805053. [Google Scholar] [CrossRef]
- Ray, M.; George, J.J.; Chakraborty, A.; Bhowmick, A.K. An Investigation of the Electromagnetic Shielding Effectiveness of Ethylene Vinyl Acetate Elastomer Reinforced with Carbon Nanofillers. Polym. Polym. Compos. 2010, 18, 59–65. [Google Scholar] [CrossRef]
- Kasgoz, A.; Akın, D.; Ayten, A.İ.; Durmus, A. Effect of different types of carbon fillers on mechanical and rheological properties of cyclic olefin copolymer (COC) composites. Compos. Part B Eng. 2014, 66, 126–135. [Google Scholar] [CrossRef]
- Charlier, J.C.; Gonze, X.; Michenaud, J.P. Graphite Interplanar Bonding: Electronic Delocalization and van der Waals Interaction. Europhys. Lett. 1994, 28, 403. [Google Scholar] [CrossRef]
- Alofi, A.; Srivastava, G.P. Thermal conductivity of graphene and graphite. Phys. Rev. B 2013, 87, 115421. [Google Scholar] [CrossRef]
- Jin, J.; Leesirisan, S.; Song, M. Electrical conductivity of ion-doped graphite/polyethersulphone composites. Compos. Sci. Technol. 2010, 70, 1544–1549. [Google Scholar] [CrossRef][Green Version]
- Sadasivuni, K.K.; Ponnamma, D.; Thomas, S.; Grohens, Y. Evolution from graphite to graphene elastomer composites. Prog. Polym. Sci. 2014, 39, 749–780. [Google Scholar] [CrossRef]
- Li, Y.; Liu, G.; Wang, L.; Zhang, J.; Xu, M.; Shi, S.Q. Multifunctional conductive graphite/cellulosic microfiber-natural rubber composite sponge with ultrasensitive collision-warning and fire-waring. Chem. Eng. J. 2022, 431, 134046. [Google Scholar] [CrossRef]
- Han, Y.; Lai, K.C.; Lii-Rosales, A.; Tringides, M.C.; Evans, J.W.; Thiel, P.A. Surface energies, adhesion energies, and exfoliation energies relevant to copper-graphene and copper-graphite systems. Surf. Sci. 2019, 685, 48–58. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Kozbial, A.; Shenoy, G.; Zhou, F.; McGinley, R.; Ireland, P.; Morganstein, B.; Kunkel, A.; Surwade, S.P.; et al. Effect of airborne contaminants on the wettability of supported graphene and graphite. Nat. Mater. 2013, 12, 925–931. [Google Scholar] [CrossRef]
- Martínez-Cano, B.; Mendoza-Meneses, C.J.; García-Trejo, J.F.; Macías-Bobadilla, G.; Aguirre-Becerra, H.; Soto-Zarazúa, G.M.; Feregrino-Pérez, A.A. Review and Perspectives of the Use of Alginate as a Polymer Matrix for Microorganisms Applied in Agro-Industry. Molecules 2022, 27, 4248. [Google Scholar] [CrossRef]
- Reddy, S.G. Alginates—A Seaweed Product: Its Properties and Applications. In Properties and Applications of Alginates; Irem, D., Esra, I., Tugba, K.-G., Eds.; IntechOpen: Rijeka, Croatia, 2021; Chapter 2. [Google Scholar]
- Jiang, Y.; Pang, X.; Deng, Y.; Sun, X.; Zhao, X.; Xu, P.; Shao, P.; Zhang, L.; Li, Q.; Li, Z. An Alginate Hybrid Sponge with High Thermal Stability: Its Flame Retardant Properties and Mechanism. Polymers 2019, 11, 1973. [Google Scholar] [CrossRef]
- Shen, Q.; Wu, M.; Xu, C.; Wang, Y.; Wang, Q.; Liu, W. Sodium alginate crosslinked oxidized natural rubber supramolecular network with rapid self-healing at room temperature and improved mechanical properties. Compos. Part A Appl. Sci. Manuf. 2021, 150, 106601. [Google Scholar] [CrossRef]
- Panitchakarn, P.; Wikranvanich, J.; Phisalaphong, M. Synthesis and characterization of natural rubber/coal fly ash composites via latex aqueous microdispersion. J. Mater. Cycles Waste Manag. 2018, 21, 134–144. [Google Scholar] [CrossRef]
- Sintharm, P.; Phisalaphong, M. Green Natural Rubber Composites Reinforced with Black/White Rice Husk Ashes: Effects of Reinforcing Agent on Film’s Mechanical and Dielectric Properties. Polymers 2021, 13, 882. [Google Scholar] [CrossRef] [PubMed]
- Supanakorn, G.; Varatkowpairote, N.; Taokaew, S.; Phisalaphong, M. Alginate as Dispersing Agent for Compounding Natural Rubber with High Loading Microfibrillated Cellulose. Polymers 2021, 13, 468. [Google Scholar] [CrossRef] [PubMed]
- Krainoi, A.; Boonkerd, K. Mechanical/electrical properties and strain sensibility of epoxidized natural rubber nanocomposite filled with carbon nanotube: Effect of sodium alginate as a surfactant on latex technology process. Express Polym. Lett. 2023, 17, 850–866. [Google Scholar] [CrossRef]
- Suppanucroa, N.; Nimpaiboon, A.; Boonchuay, K.; Khamkeaw, A.; Phisalaphong, M. Green composite sponge of natural rubber reinforced with cellulose filer using alginate as a dispersing agent. J. Mater. Res. Technol. 2023, 27, 3119–3130. [Google Scholar] [CrossRef]
- Phomrak, S.; Phisalaphong, M. Reinforcement of Natural Rubber with Bacterial Cellulose via a Latex Aqueous Microdispersion Process. J. Nanomater. 2017, 2017, 4739793. [Google Scholar] [CrossRef]
- Phomrak, S.; Phisalaphong, M. Lactic Acid Modified Natural Rubber–Bacterial Cellulose Composites. Appl. Sci. 2020, 10, 3583. [Google Scholar] [CrossRef]
- Supanakorn, G.; Taokaew, S.; Phisalaphong, M. Ternary composite films of natural rubber, cellulose microfiber, and carboxymethyl cellulose for excellent mechanical properties, biodegradability and chemical resistance. Cellulose 2021, 28, 8553–8566. [Google Scholar] [CrossRef]
- ASTM D882; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. ASTM International: West Conshohocken, PA, USA, 2018.
- Sintharm, P.; Nimpaiboon, A.; Liao, Y.-C.; Phisalaphong, M. Bacterial cellulose reinforced with skim/fresh natural rubber latex for improved mechanical, chemical and dielectric properties. Cellulose 2022, 29, 1739–1758. [Google Scholar] [CrossRef]
- Krainoi, A.; Boonkerd, K. Novel hybrid natural rubber nanocomposites with carbon nanotube and cellulose nanofiber for strain-sensitive sensor. Ind. Crops Prod. 2022, 187, 115455. [Google Scholar] [CrossRef]
- Rolere, S.; Liengprayoon, S.; Vaysse, L.; Sainte-Beuve, J.; Bonfils, F. Investigating natural rubber composition with Fourier Transform Infrared (FT-IR) spectroscopy: A rapid and non-destructive method to determine both protein and lipid contents simultaneously. Polym. Test. 2015, 43, 83–93. [Google Scholar] [CrossRef]
- Kundi, A.H.W.; Waseem, I. Synthesis of Graphene Nano Sheets by the Rapid Reduction of Electrochemically Exfoliated Graphene Oxide Induced by Microwaves. J. Chem. Soc. Pak. 2016, 38, 11. [Google Scholar]
- Bera, M.; Yadav, C.; Gupta, P.; Maji, P. Facile One-Pot Synthesis of Graphene Oxide by Sonication Assisted Mechanochemical Approach and Its Surface Chemistry. J. Nanosci. Nanotechnol. 2018, 18, 902–912. [Google Scholar] [CrossRef]
- Wipatkrut, P.; Poompradub, S. Exfoliation approach for preparing high conductive reduced graphite oxide and its application in natural rubber composites. Mater. Sci. Eng. B 2017, 218, 74–83. [Google Scholar] [CrossRef]
- Hu, M.; Zhou, Y.; Nie, W.; Chen, P. Functionalized Graphene Nanosheets with Fewer Defects Prepared via Sodium Alginate Assisted Direct Exfoliation of Graphite in Aqueous Media for Lithium-Ion Batteries. ACS Appl. Nano Mater. 2018, 1, 1985–1994. [Google Scholar] [CrossRef]
- Al-Hartomy, O.A.; Gupta, R.K.; Al-Ghamdi, A.A.; Yakuphanoglu, F. High performance organic-on-inorganic hybrid photodiodes based on organic semiconductor-graphene oxide blends. Synth. Met. 2014, 195, 217–221. [Google Scholar] [CrossRef]
- Chimupala, Y.; Junploy, P.; Hardcastle, T.; Westwood, A.; Scott, A.; Johnson, B.; Brydson, R. Universal synthesis method for mixed phase TiO2(B)/anatase TiO2 thin films on substrates via a modified low pressure chemical vapour deposition (LPCVD) route. J. Mater. Chem. A 2016, 4, 5685–5699. [Google Scholar] [CrossRef]
- Mohammad, H.; Stepashkin, A.A.; Laptev, A.I.; Tcherdyntsev, V.V. Mechanical and Conductive Behavior of Graphite Filled Polysulfone-Based Composites. Appl. Sci. 2023, 13, 542. [Google Scholar] [CrossRef]
- Alshammari, B.A.; Wilkinson, A.N.; AlOtaibi, B.M.; Alotibi, M.F. Influence of Carbon Micro- and Nano-Fillers on the Viscoelastic Properties of Polyethylene Terephthalate. Polymers 2022, 14, 2440. [Google Scholar] [CrossRef]
- Alshammari, B.A.; Al-Mubaddel, F.S.; Karim, M.R.; Hossain, M.; Al-Mutairi, A.S.; Wilkinson, A.N. Addition of Graphite Filler to Enhance Electrical, Morphological, Thermal, and Mechanical Properties in Poly (Ethylene Terephthalate): Experimental Characterization and Material Modeling. Polymers 2019, 11, 1411. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Sinturel, C.; Bahloul, O.; Saboungi, M.-L.; Thomas, S.; Salvetat, J.-P. Improving reinforcement of natural rubber by networking of activated carbon nanotubes. Carbon 2008, 46, 1037–1045. [Google Scholar] [CrossRef]
- Feng, C.; Ni, H.; Chen, J.; Yang, W. Facile Method to Fabricate Highly Thermally Conductive Graphite/PP Composite with Network Structures. ACS Appl. Mater. Interfaces 2016, 8, 19732–19738. [Google Scholar] [CrossRef] [PubMed]
- Huq, T.; Salmieri, S.; Khan, A.; Khan, R.A.; Le Tien, C.; Riedl, B.; Fraschini, C.; Bouchard, J.; Uribe-Calderon, J.; Kamal, M.R.; et al. Nanocrystalline cellulose (NCC) reinforced alginate based biodegradable nanocomposite film. Carbohydr. Polym. 2012, 90, 1757–1763. [Google Scholar] [CrossRef] [PubMed]
- Abulateefeh, S.; Taha, M. Enhanced drug encapsulation and extended release profiles of calcium–alginate nanoparticles by using tannic acid as a bridging cross-linking agent. J. Microencapsul. 2014, 32, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.; Santos, J.; Chierice, G.; Cavalheiro, É. Thermal behavior of alginic acid and its sodium salt. Eclet. Quim. 2004, 29, 57–63. [Google Scholar] [CrossRef]
- Deng, H.; Reynolds, C.T.; Cabrera, N.O.; Barkoula, N.M.; Alcock, B.; Peijs, T. The water absorption behaviour of all-polypropylene composites and its effect on mechanical properties. Compos. Part B Eng. 2010, 41, 268–275. [Google Scholar] [CrossRef]
- Turmanova, S.; Genieva, S.; Vlaev, L. Obtaining Some Polymer Composites Filled with Rice Husks Ash-A Review. Int. J. Chem. 2012, 4, 62–89. [Google Scholar] [CrossRef]
- Phuangngamphan, M.; Okhawilai, M.; Hiziroglu, S.; Rimdusit, S. Development of highly conductive graphite-/graphene-filled polybenzoxazine composites for bipolar plates in fuel cells. J. Appl. Polym. Sci. 2019, 136, 47183. [Google Scholar] [CrossRef]
- Sheshmani, S.; Ashori, A.; Arab Fashapoyeh, M. Wood plastic composite using graphene nanoplatelets. Int. J. Biol. Macromol. 2013, 58, 1–6. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Xu, C.; Zhang, M.; Shang, X. Preparation of graphene/poly(vinyl alcohol) nanocomposites with enhanced mechanical properties and water resistance. Polym. Int. 2011, 60, 816–822. [Google Scholar] [CrossRef]
- Alo, O.A.; Otunniyi, I.O. Comparative study of flexural and physical properties of graphite-filled immiscible polypropylene/epoxy and high-density polyethylene/epoxy blends. Polym. Polym. Compos. 2021, 29, S1103–S1112. [Google Scholar] [CrossRef]
- Prolongo, S.G.; Moriche, R.; Jiménez-Suárez, A.; Sánchez, M.; Ureña, A. Advantages and disadvantages of the addition of graphene nanoplatelets to epoxy resins. Eur. Polym. J. 2014, 61, 206–214. [Google Scholar] [CrossRef]
- Gautam, R.K.; Kar, K.K. Synergistic Effects of Carbon Fillers of Phenolic Resin Based Composite Bipolar Plates on the Performance of PEM Fuel Cell. Fuel Cells 2016, 16, 179–192. [Google Scholar] [CrossRef]
- Nie, J.; Huang, J.; Fan, J.; Cao, L.; Xu, C. Strengthened, Self-Healing, and Conductive ENR-Based Composites Based on Multiple Hydrogen Bonding Interactions. ACS Sustain. Chem. Eng. 2020, 8, 13724–13733. [Google Scholar] [CrossRef]
- Ismail, M.N.; Khalaf, A.I. Styrene–butadiene rubber/graphite powder composites: Rheometrical, physicomechanical, and morphological properties. J. Appl. Polym. Sci. 2011, 120, 298–304. [Google Scholar] [CrossRef]
- Nezha Tahri, J.; Wifak, B.; Hanane, S.; Naïma El, G. Biodegradation: Involved Microorganisms and Genetically Engineered Microorganisms. In Biodegradation; Rolando, C., Francisca, R., Eds.; IntechOpen: Rijeka, Croatia, 2013; Chapter 11. [Google Scholar]
- Manikandan, S.; Vickram, S.; Sirohi, R.; Subbaiya, R.; Krishnan, R.Y.; Karmegam, N.; Sumathijones, C.; Rajagopal, R.; Chang, S.W.; Ravindran, B.; et al. Critical review of biochemical pathways to transformation of waste and biomass into bioenergy. Bioresour. Technol. 2023, 372, 128679. [Google Scholar] [CrossRef]
- Bosco, F.; Mollea, C. Biodegradation of Natural Rubber: Microcosm Study. Water Air Soil Pollut. 2021, 232, 227. [Google Scholar] [CrossRef]
- Pantani, R.; Sorrentino, A. Influence of crystallinity on the biodegradation rate of injection-moulded poly(lactic acid) samples in controlled composting conditions. Polym. Degrad. Stab. 2013, 98, 1089–1096. [Google Scholar] [CrossRef]
- Barbosa, R.; Gonçalves, R.; Blanco, G.E.d.O.; Saccardo, M.C.; Tozzi, K.A.; Zuquello, A.G.; Scuracchio, C.H. Multi-sensing properties of hybrid filled natural rubber nanocomposites using impedance spectroscopy. Electrochim. Acta 2022, 435, 141341. [Google Scholar] [CrossRef]
- Chai, K.L.; Aung, M.M.; Noor, I.M.; Lim, H.N.; Abdullah, L.C. Observation of ionic conductivity on PUA-TBAI-I2 gel polymer electrolyte. Sci. Rep. 2022, 12, 124. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Adhikari, R.; Cass, P.; Bown, M.; Gunatillake, P. Electrically conductive polymers and composites for biomedical applications. RSC Adv. 2015, 5, 37553–37567. [Google Scholar] [CrossRef]


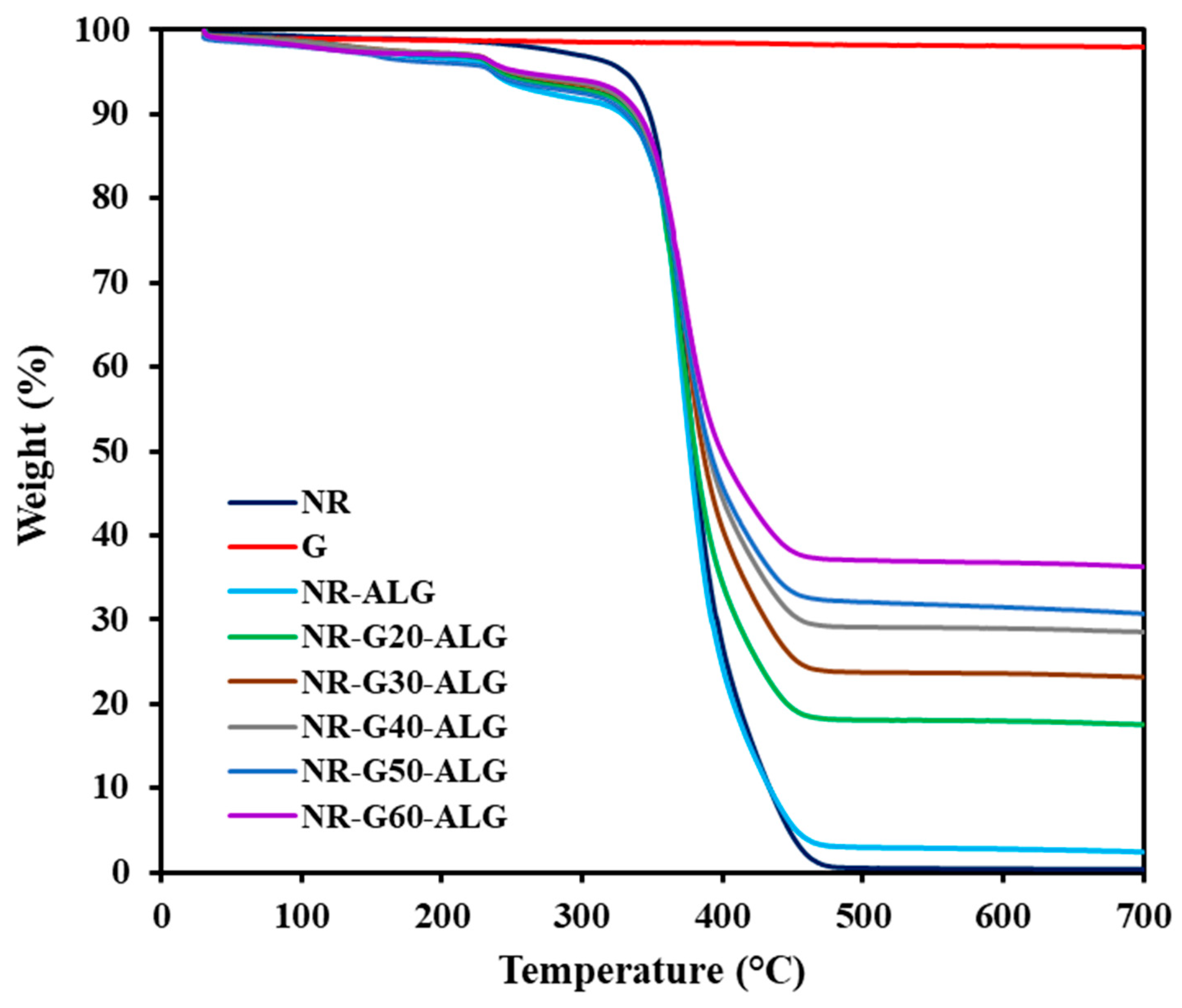
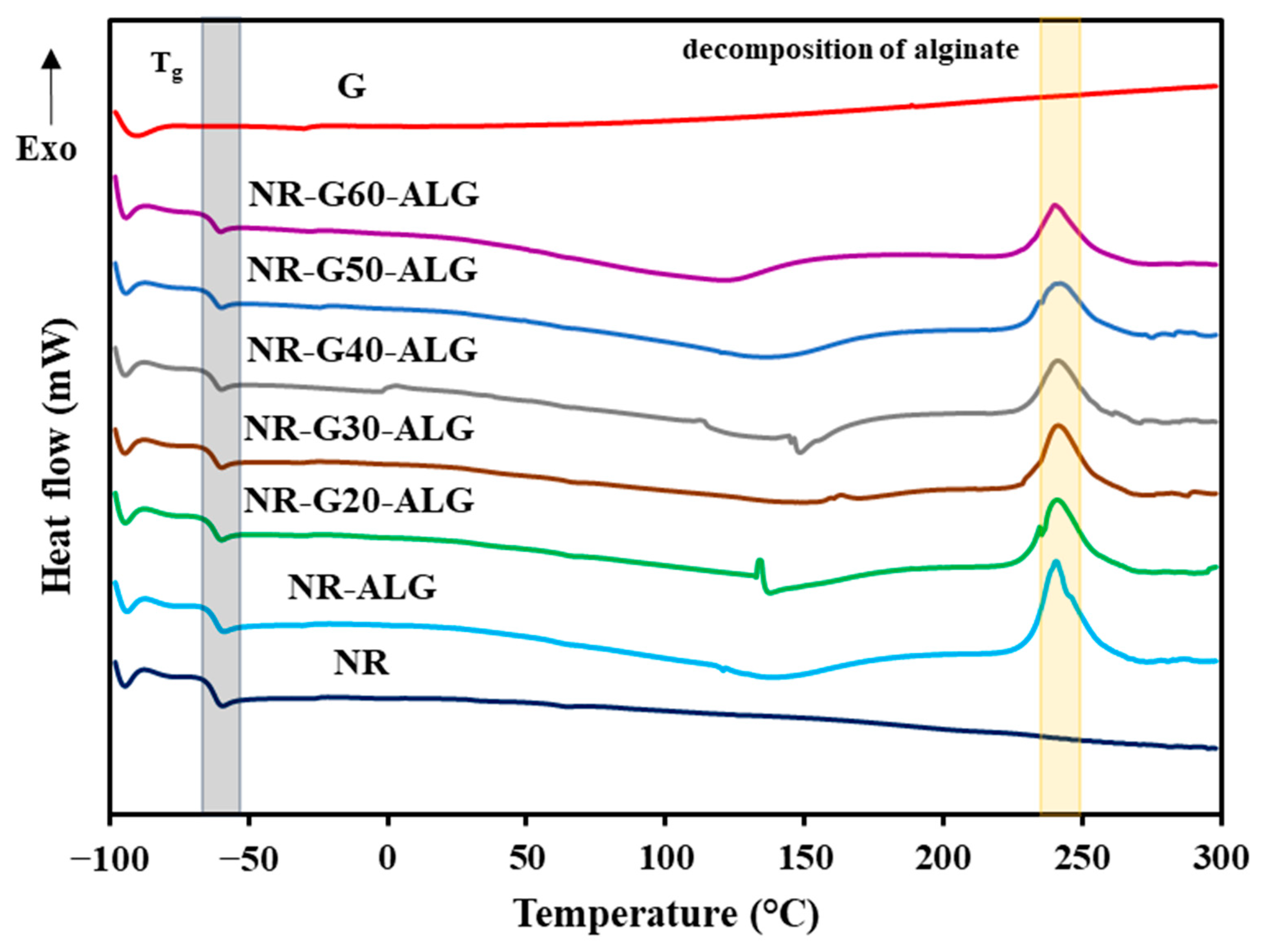
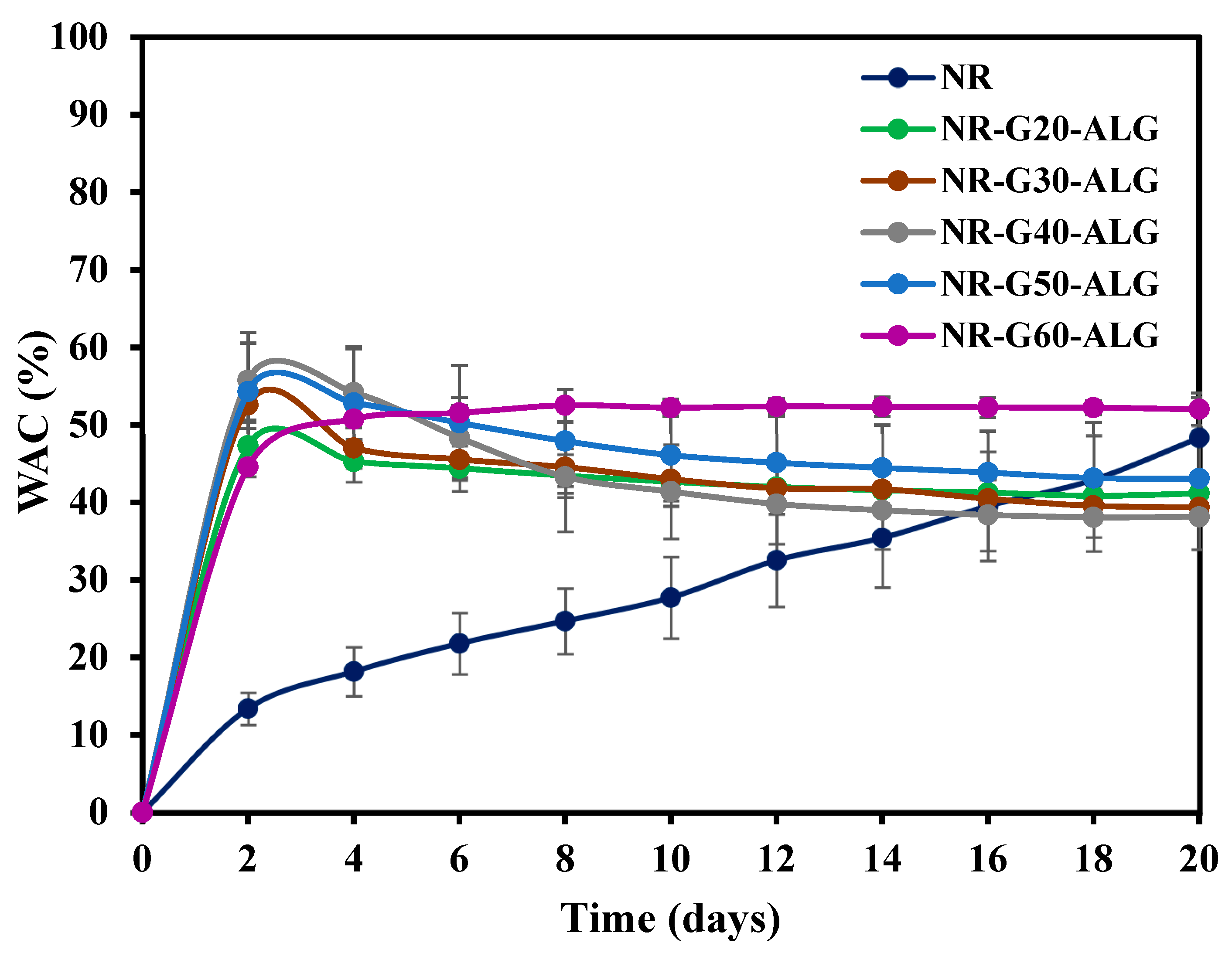

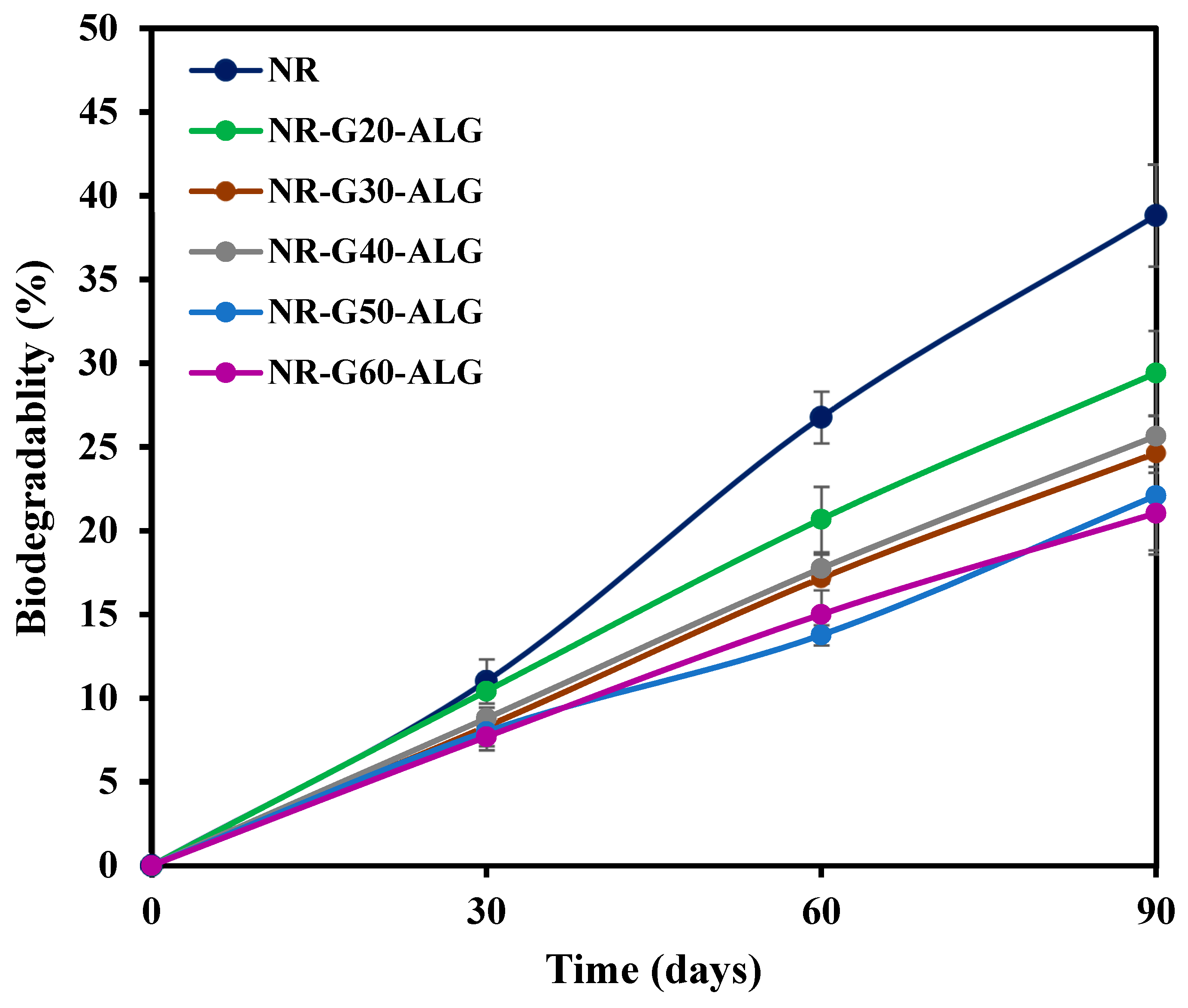

| Sample | Graphite Loading (phr) | T5% (°C) | T10% (°C) | T50% (°C) | Residual Mass at 700 °C (wt.%) |
|---|---|---|---|---|---|
| NR | - | 330.1 | 347.8 | 379.3 | 0.37 |
| NR-ALG | - | 236.7 | 328.8 | 376.7 | 2.41 |
| NR-G20-ALG | 20 | 242.5 | 335.6 | 380.3 | 17.54 |
| NR-G30-ALG | 30 | 248.2 | 338.3 | 385.9 | 23.18 |
| NR-G40-ALG | 40 | 250.2 | 337.7 | 389.7 | 28.54 |
| NR-G50-ALG | 50 | 246.6 | 331.2 | 391 | 30.70 |
| NR-G60-ALG | 60 | 257.2 | 340.4 | 399.1 | 36.27 |
| G | 100 | - | - | - | 97.98 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kitsawat, V.; Siri, S.; Phisalaphong, M. Electrically Conductive Natural Rubber Composite Films Reinforced with Graphite Platelets. Polymers 2024, 16, 288. https://doi.org/10.3390/polym16020288
Kitsawat V, Siri S, Phisalaphong M. Electrically Conductive Natural Rubber Composite Films Reinforced with Graphite Platelets. Polymers. 2024; 16(2):288. https://doi.org/10.3390/polym16020288
Chicago/Turabian StyleKitsawat, Veerapat, Saranrat Siri, and Muenduen Phisalaphong. 2024. "Electrically Conductive Natural Rubber Composite Films Reinforced with Graphite Platelets" Polymers 16, no. 2: 288. https://doi.org/10.3390/polym16020288
APA StyleKitsawat, V., Siri, S., & Phisalaphong, M. (2024). Electrically Conductive Natural Rubber Composite Films Reinforced with Graphite Platelets. Polymers, 16(2), 288. https://doi.org/10.3390/polym16020288






