Revealing Commercial Epoxy Resins’ Antimicrobial Activity: A Combined Chemical–Physical, Mechanical, and Biological Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Epoxy Resins
2.3. Characterization Techniques
3. Results
3.1. Characterization of the Epoxy Resins and Precursors
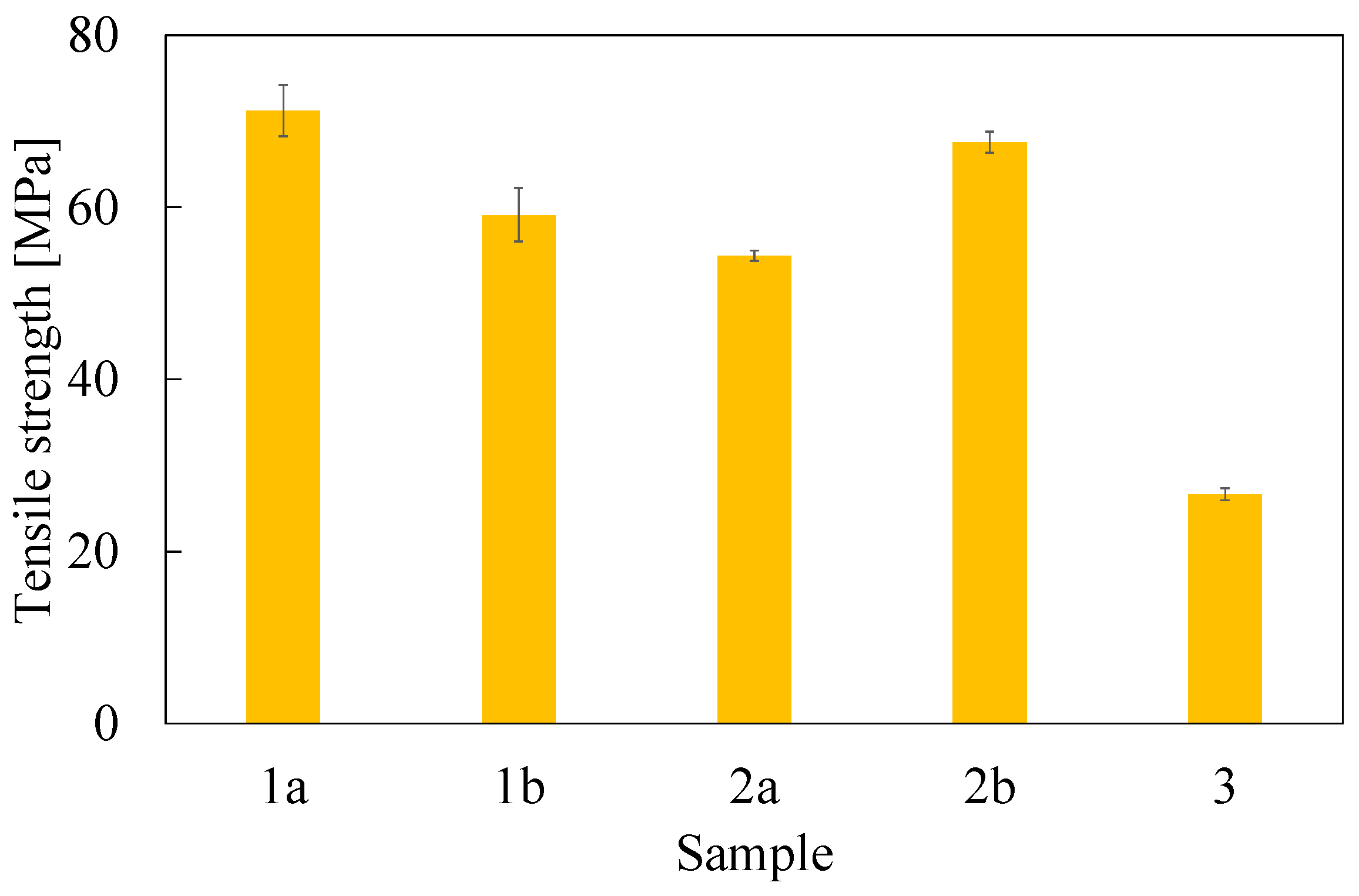
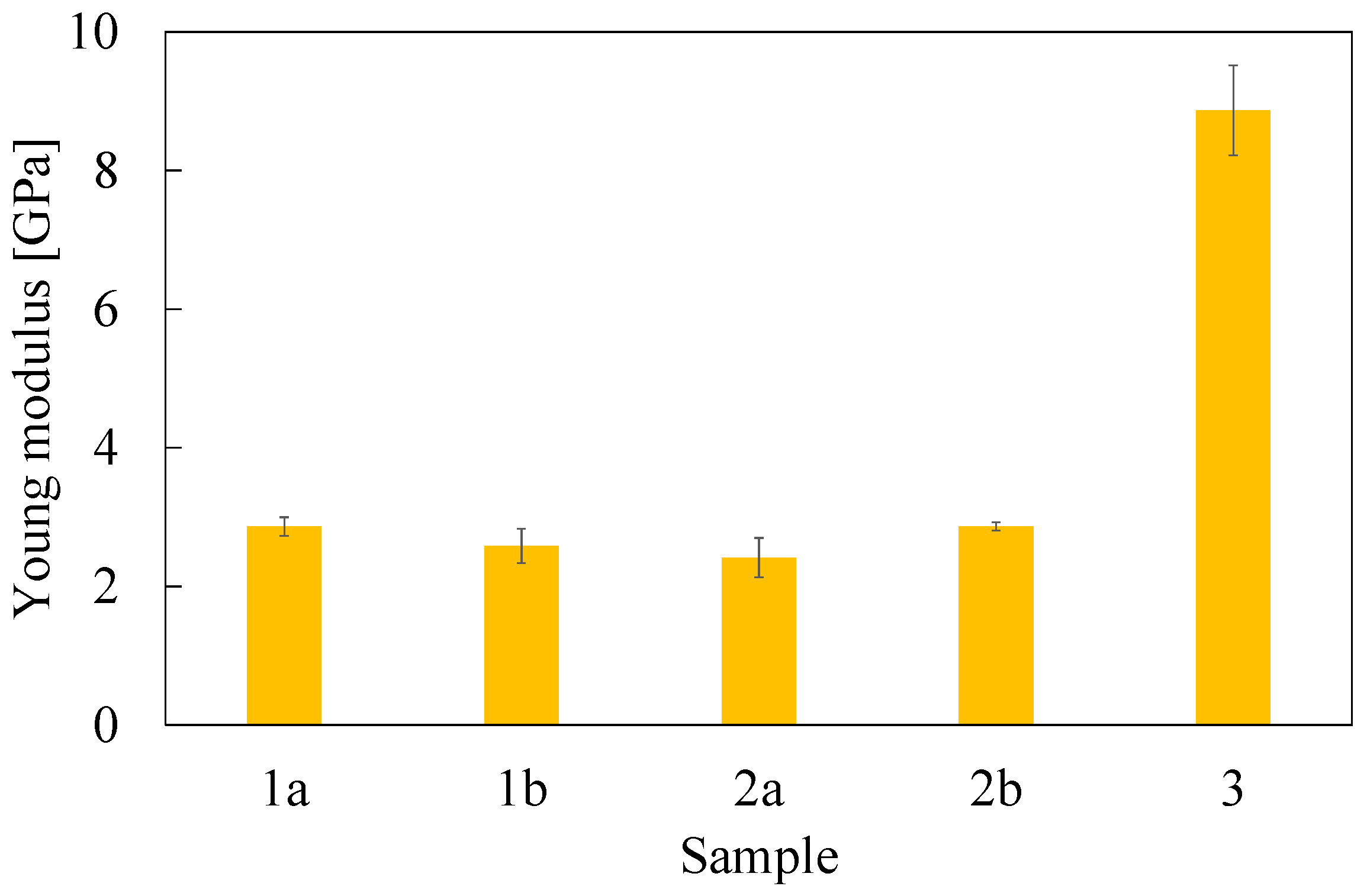
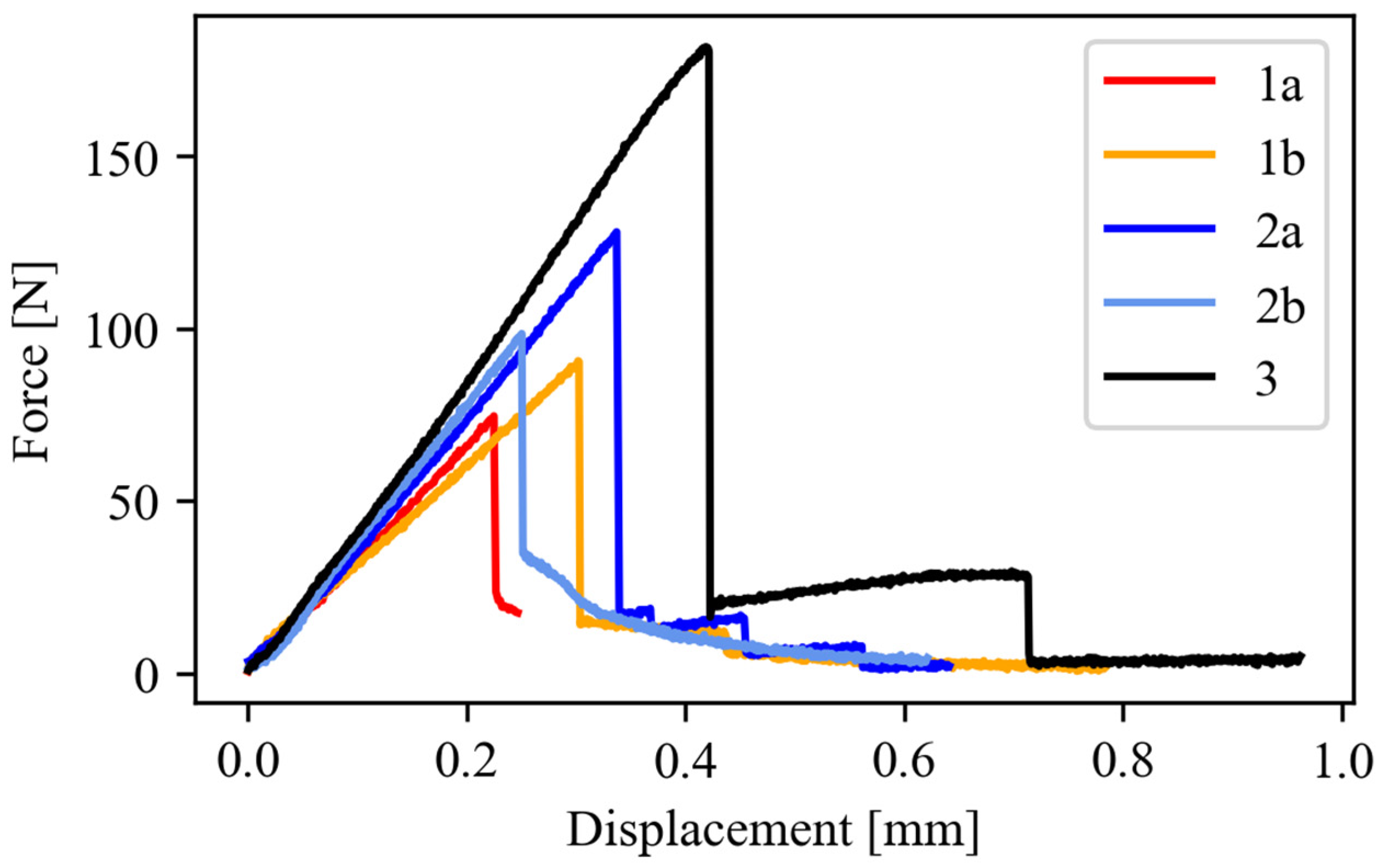
3.2. Mechanical Properties
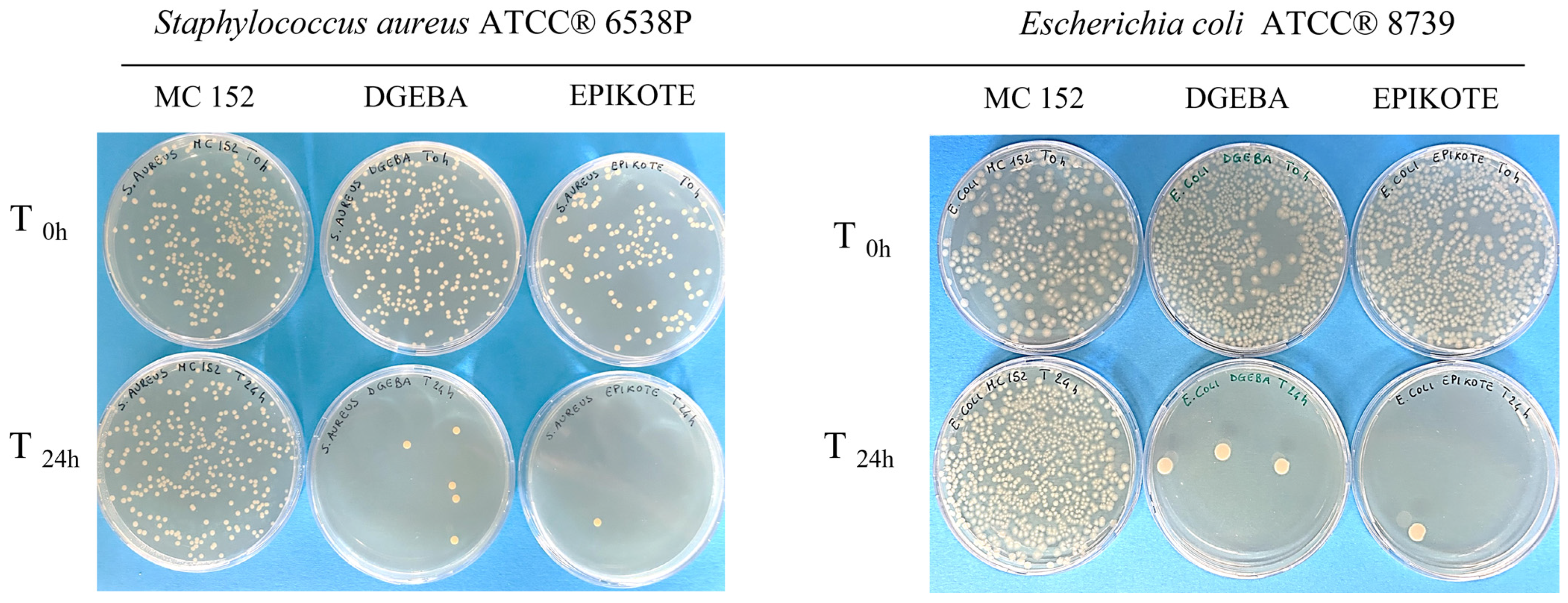
3.3. Antibacterial Activity
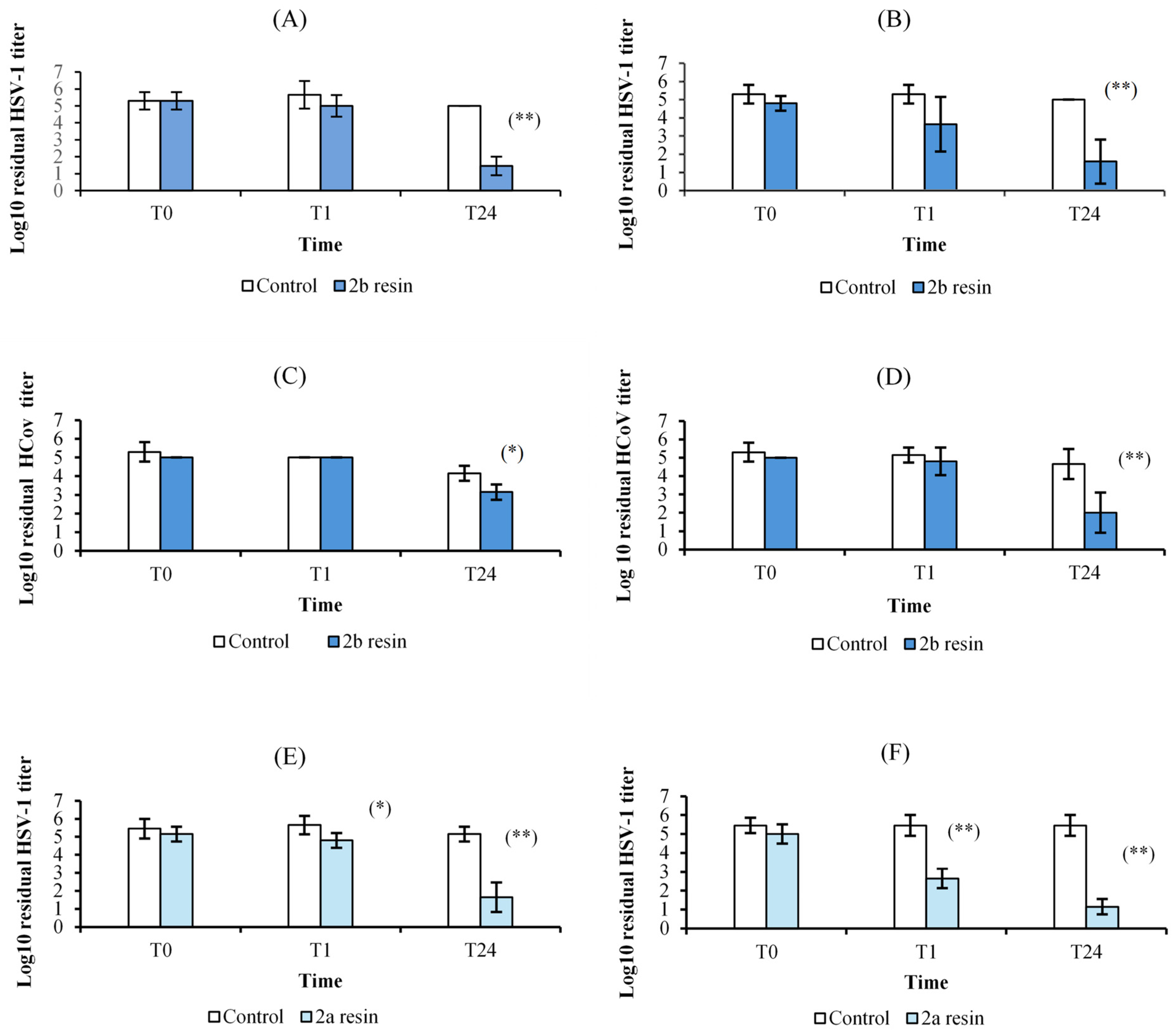
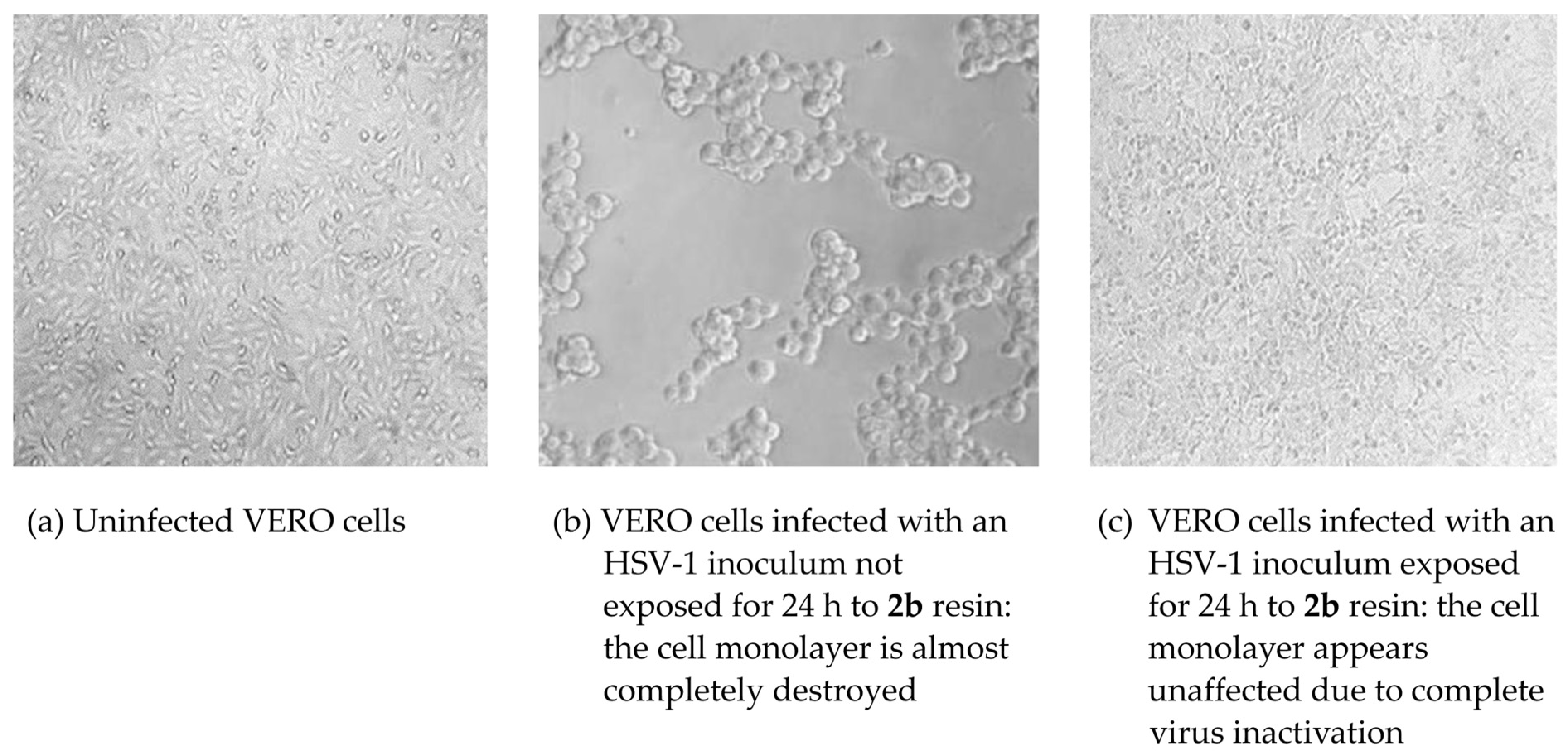
3.4. Antiviral Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Thandar, M.M.; Rahman, M.O.; Haruyama, R.; Matsuoka, S.; Okawa, S.; Moriyama, J.; Yokobori, Y.; Matsubara, C.; Nagai, M.; Ota, E.; et al. Effectiveness of Infection Control Teams in Reducing Healthcare-Associated Infections: A Systematic Review and Meta Analysis. Int. J. Environ. Res. Public Health 2022, 19, 17075. [Google Scholar] [CrossRef] [PubMed]
- Point Prevalence Survey of Healthcare Associated Infections and Antimicrobial Use in European Acute Care Hospitals. European Centre for Disease Prevention and Control. 2024. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/healthcare-associated-point-prevalence-survey-acute-care-hospitals-2022-2023.pdf (accessed on 2 August 2024).
- Umscheid, C.A.; Mitchell, M.D.; Doshi, J.A.; Agarwal, R.; Williams, K.; Brennan, P.J. Estimating the proportion of healthcare-associated infections that are reasonably preventable and the related mortality and costs. Infect. Control Hosp. Epidemiol. 2011, 32, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Otter, J.A.; Yezli, S.; Salkeld, J.A.; French, G.L. Evidence that contaminated surfaces contribute to the transmission of hospital pathogens and an overview of strategies to address contaminated surfaces in hospital settings. Am. J. Infect. Control 2013, 41, S6–S11. [Google Scholar] [CrossRef] [PubMed]
- Global Report on Infection Prevention and Control; World Health Organization: Geneva, Switzerland, 2022; Available online: https://www.who.int/publications/i/item/9789240051164 (accessed on 2 August 2024).
- Dancer, S.J. Hospital cleaning: Past, present, and future. Antimicrob. Resist. Infect. Control 2023, 12, 80. [Google Scholar] [CrossRef]
- Rutala, W.A.; Weber, D.J. Disinfection and Sterilization in Health Care Facilities: An Overview and Current Issues. Infect. Dis. Clin. N. Am. 2021, 35, 575–607. [Google Scholar] [CrossRef]
- Weber, D.J.; Rutala, W.A.; Anderson, D.J.; Sickbert-Bennett, E.E. “No touch” methods for health care room disinfection: Focus on clinical trials. Am. J. Infect. Control 2023, 51, A134–A143. [Google Scholar] [CrossRef]
- Querido, M.M.; Aguiar, L.; Neves, P.; Pereira, C.C.; Teixeira, J.P. Self-disinfecting surfaces and infection control. Colloids Surf. B Biointerfaces 2019, 178, 8–21. [Google Scholar] [CrossRef]
- Haktaniyan, M.; Bradley, M. Polymers showing intrinsic antimicrobial activity. Chem. Soc. Rev. 2022, 51, 8584–8611. [Google Scholar] [CrossRef]
- Vlad-Bubulac, T.; Hamciuc, C.; Serbezeanu, D.; Macsim, A.M.; Lisa, G.; Anghel, I.; Preda, D.M.; Kalvachev, Y.; Rîmbu, C.M. Simultaneous Enhancement of Flame Resistance and Antimicrobial Activity in Epoxy Nanocomposites Containing Phosphorus and Silver-Based Additives. Molecules 2023, 28, 5650. [Google Scholar] [CrossRef]
- Jain, A.; Duvvuri, L.S.; Farah, S.; Beyth, N.; Domb, A.J.; Khan, W. Antimicrobial Polymers. Adv. Healthc. Mater. 2014, 3, 1969–1985. [Google Scholar] [CrossRef]
- Siedenbiedel, F.; Tiller, J.C. Antimicrobial polymers in solution and on surfaces: Overview and functional principles. Polymers 2012, 4, 46–71. [Google Scholar] [CrossRef]
- Mahira, S.; Jain, A.; Khan, W.; Domb, A.J. Chapter 1 Antimicrobial Materials—An Overview in Biomaterials Science Series No. 5 Antimicrobial Materials for Biomedical Applications; Domb, A.J., Reddy Kunduru, K., Farahr, S., Eds.; The Royal Society of Chemistry: London, UK, 2019. [Google Scholar]
- Shundo, A.; Yamamoto, S.; Tanaka, K. Network formation and physical properties of epoxy resins for future practical applications. J. Am. Chem. Soc. 2022, 2, 1522–1542. [Google Scholar] [CrossRef] [PubMed]
- Ellis, B. Chemistry and Technology of Epoxy Resins; Springer: Dordrecht, The Netherlands, 1993. [Google Scholar]
- Faltyonowicz, H.; Kulazynski, M.; Goodman, S.H. Epoxies, Chapter 8. In Handbook of Thermoset Plastics; Dodiuk, H., Ed.; Elsevier: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Miturska, I.; Rudawska, A.; Muller, M.; Hromasova, M. The influence of mixing methods of epoxy composition ingredients on selected mechanical properties of modified epoxy construction materials. Materials 2021, 14, 411. [Google Scholar] [CrossRef] [PubMed]
- Esranur, Y.; Osman, E.; Hanzade, H.A.; Serdar, Y. Mechanical properties of a carbon fiber reinforced epoxy resin composite improved by interacting multi-walled carbon nanotubes and graphene nanoplatelets. J. Compos. Mater. 2024, 58, 911–921. [Google Scholar]
- Pramodkumar, B.; Budhe, S. The effect of graphene oxide on thermal, electrical, and mechanical properties of carbon/epoxy composites: Towards multifunctional composite materials. Polym. Compos. 2024, 45, 6374–6384. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, Y.; Wang, Z.; Cui, X.; Liu, G.; Zhang, L.; Zhu, Y.; Chen, J.; Sun, S.; Zhang, K.; et al. A new composite material with energy storage, electro/photo-thermal and robust super-hydrophobic properties for high-efficiency anti-icing/de-icing. Small 2024, 20, 2311435. [Google Scholar] [CrossRef]
- Demirel, M.O.; Ozturkmen, M.B.; Savas, M.; Mutlugun, E.; Erdem, T.; Oz, Y. Effects of silver nanowires and their surface modification on electromagnetic interference, transport and mechanical properties of an aerospace grade epoxy. J. Compos. Mater. 2024, 58, 1267–1277. [Google Scholar] [CrossRef]
- Romero-Fierro, D.; Bustamante-Torres, M.; Bravo-Plascencia, F.; Esquivel-lozano, A.; Ruiz, J.C.; Bucio, E. Recent trends in magnetic polymer nanocomosites for aerospace applications: A review. Polymers 2022, 14, 4084. [Google Scholar] [CrossRef]
- Han, R.; Ma, X.; Cai, L.; Zhang, Z.; Fang, Y.; Wang, J. Low viscosity and low temperature curing reactive POSS/epoxy hybrid resin with enhanced toughness and comprehensive thermal performance. RCS Adv. 2024, 14, 7263–7275. [Google Scholar] [CrossRef]
- Murtaza, H.; Zhao, J.; Tabish, M.; Wang, J.; Mubeen, M.; Zhang, J.; Zhang, S.; Fan, B. Protective and flame-retardant bifunctional epoxy-based nanocomposite coating by intercomponent synergy between modified CaAl-LDH and rGO. Appl. Mater. Interfaces 2024, 16, 13114–13131. [Google Scholar] [CrossRef]
- Ahangaran, F.; Hayaty, M.; Navarchian, A.H.; Pei, Y.; Picchioni, F. Development of self-healing epoxy composites via incorporation of microencapsulated epoxy and mercaptan in poly(methyl methacrylate) shell. Polym. Test. 2019, 73, 395–403. [Google Scholar] [CrossRef]
- Bertani, R.; Bartolozzi, A.; Pontefisso, A.; Quaresimin, M.; Zappalorto, M. Improving the antimicrobial and mechanical properties of epoxy resins via nanomodification: An Overview. Molecules 2021, 26, 5426. [Google Scholar] [CrossRef] [PubMed]
- Komabayashi, T.; Colmenar, D.; Cvach, N.; Bhat, A.; Primus, C.; Imai, Y. Comprehensive review of current endodontics sealers. Dent. Mater. J. 2020, 39, 703–720. [Google Scholar] [CrossRef]
- ISO 11358-1; Plastics—Thermogravimetry (TG) of Polymers—Part 1: General Principles. ISO: Geneva, Switzerland, 2022. Available online: https://www.iso.org/standard/79999.html (accessed on 8 April 2024).
- ISO 527-2; Plastics—Determination of Tensile Properties—Part 2: Test Conditions for Moulding and Extrusion Plastics. ISO: Geneva, Switzerland, 2012. Available online: https://www.iso.org/standard/56046.html (accessed on 8 April 2024).
- ASTM D5045-99; Standard Test Methods for Plane-Strain Fracture Toughness and Strain Energy Release Rate of Plastic Materials. ASTM International: West Conshohocken, PA, USA, 2017. Available online: https://cdn.standards.iteh.ai/samples/6161/2b8009c6e4bb4d028ccc2f23c709277a/ASTM-D5045-99.pdf (accessed on 14 May 2024).
- ISO 22196:2011 (en); Measurement of Antibacterial Activity on Plastics and Other Non-Porous Surfaces. ISO: Geneva, Switzerland, 2011. Available online: https://www.iso.org/standard/54431.html (accessed on 8 April 2024).
- American Public Health Association. Diagnostic Procedures for Viral and Rickettsial Diseases; Subcommittee on Diagnostic Procedures for Viral and Rickettsial Diseases; Lennette, E.H., Schmidt, N.J., Eds.; American Public Health Association, Incorporated: New York City, NY, USA, 1964. [Google Scholar]
- Gonzalez, M.G.; Cabanelas, J.C.; Baselga, J. Applications of FIR on epoxy Resins -Identification, Monitoring the Curing Process, Phase Separation and WaterUptake. In Infrared Spectroscopy-Material Science, Engineering and Technology; Theophanides, T., Ed.; IntechOpen: London, UK, 2012. [Google Scholar]
- Nakagawa, H.; Tsuge, S.; Koyama, T. Studies on thermal degradation of epoxy resins by high-resolution pyrolysis-gas chromatography. J. Anal. Appl. Pyrolysis 1987, 12, 97–113. [Google Scholar] [CrossRef]
- McAninch, I.M.; Palmese, G.R.; Lenhart, J.L.; La Scala, J. DMA testing of epoxy resins: The importance of dimensions. Polym. Eng. Sci. 2015, 55, 2761–2774. [Google Scholar] [CrossRef]
- Mahanta, U.; Khandelwal, M.; Deshpande, A.S. Antimicrobial surfaces: A review of synthetic approaches, applicability and outlook. J. Mater. Sci. 2021, 56, 17915–17941. [Google Scholar] [CrossRef]
- Bazaka, K.; Jacob, M.V.; Crawford, R.J.; Ivanova, E.P. Efficient surface modification of biomaterials to prevent biofilm formation and the attachment of microorganisms. Appl. Microbiol. Biotechnol. 2012, 95, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Cunliffe, A.J.; Askew, P.D.; Stephan, I.; Iredale, G.; Cosemans, P.; Simmons, L.M.; Verran, J.; Redfern, J. How Do We Determine the Efficacy of an Antibacterial Surface? A Review of Standardised Antibacterial Material Testing Methods. Antibiotics 2021, 10, 1069. [Google Scholar] [CrossRef]
- Atkinson, B.; Petersen, E. SARS-CoV-2 shedding and infectivity. Lancet 2020, 395, 1339–1340. [Google Scholar] [CrossRef]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect Dis. 2006, 6, 130–137. [Google Scholar] [CrossRef]
- Long, Q.; Zhou, W.; Zhou, H.; Tang, Y.; Chen, W.; Liu, Q.; Bian, X. Polyamine-containing natural products: Structure, bioactivity, and biosynthesis. Nat. Prod. Rep. 2024, 41, 525–564. [Google Scholar] [CrossRef] [PubMed]
- Zarafu, I.; Turcu, I.; Culita, D.C.; Petrescu, S.; Popa, M.; Chifiriuc, M.C.; Limban, C.; Telehoiu, A.; Ionita, P. Antimicrobial Features of Organic Functionalized Graphene-Oxide with Selected Amines. Materials 2018, 11, 1704. [Google Scholar] [CrossRef] [PubMed]
- Spivak, A.Y.; Khalitova, R.R.; Nedopekina, D.A.; Gubaidullin, R.R. Antimicrobial properties of amine- and guanidine-functionalized derivatives of betulinic, ursolic and oleanolic acids: Synthesis and structure/activity evaluation. Steroids 2020, 154, 108530. [Google Scholar] [CrossRef] [PubMed]
- Glushkov, V.A.; P’yankova, O.S.; Anikina, L.V.; Vikharev, Y.B.; Feshina, E.V.; Shklyaev, Y.V.; Tolstikov, A.G. Synthesis and biological activity of cyclohexylamine derivatives. Pharm. Chem. J. 2006, 40, 298–302. [Google Scholar] [CrossRef]
- Marzano, C.; Sbovata, S.M.; Gandin, V.; Colavito, D.; del Giudice, E.; Michelin, R.A.; Venzo, A.; Seraglia, R.; Benetollo, F.; Schiavon, M.; et al. A new class of antitumor trans-amine-amidine-Pt(II) cationic complexes: Influence of chemical structure and solvent on in vitro and in vivo tumor cell proliferation. J. Med. Chem. 2010, 53, 6210–6227. [Google Scholar] [CrossRef]
- Wei, T.; Tang, Z.; Yu, Q.; Chen, H. Smart antibacterial surfaces with switchable bacteria-killing and bacteria-releasing capabilities. ACS Appl. Mater. Interfaces 2017, 9, 37511–37523. [Google Scholar] [CrossRef]
- Ding, X.; Duan, S.; Ding, X.; Liu, R.; Xu, F.J. Versatile antibacterial materials: An emerging arsenal for combatting bacterial pathogens. Adv. Funct. Mater. 2018, 28, 1802140. [Google Scholar] [CrossRef]
- Hoque, J.; Akkapeddi, P.; Yadav, V.; Manjunath, G.B.; Uppu, D.S.S.M.; Konai, M.M.; Yarlagadda, V.; Sanyal, K.; Haldar, J. Broad spectrum antibacterial and antifungal polymeric paint meterials: Synthesis, structure-activity relationship, and membrane-active mode of action. ACS Appl. Mater. Interfaces 2015, 7, 1804–1815. [Google Scholar] [CrossRef]
- Hernando-Perez, M.; Cartagena-Rivera, A.X.; Losdorfer Bozic, A.; Carrillo, P.J.P.; San Martin, C.; Mateu, M.G.; Raman, A.; Podgornik, R.; de Pablo, P.J. Quantitative nanoscale electrostatics of viruses. Nanoscale 2015, 7, 17289–17298. [Google Scholar] [CrossRef]
- Winther, B.; McCue, K.; Ashe, K.; Rubino, J.R.; Hendley, J.O. Environmental contamination with rhinovirus and transfer to fingers of healthy individuals by daily life activity. J. Med. Virol. 2007, 79, 1606–1610. [Google Scholar] [CrossRef]
- Kramer, A.; Assadian, O. Survival of microorganisms on inanimate surfaces. In Use of Biocidal Surfaces for Reduction of Healthcare Acquired Infections; Borkoe, G., Ed.; Springer International Publishing: Cham, Switzerland, 2014; Chapter 2; pp. 7–26. [Google Scholar]
- Drelich, J.; Chibowski, E.; Desheng Meng, D.; Terpilowski, K. Hydrophilic and superhydrophilic surfaces and materials. Soft Matter 2011, 7, 9804–9828. [Google Scholar] [CrossRef]
- Hasan, J.; Xu, Y.; Yarlagadda, T.; Schuetz, M.; Spann, K.; Yarlagadda, P.K.D.V. Antiviral and antibacterial nanostructured surfaces with excellent mechanical properties for hospital applications. ACS Biomater. Sci. Eng. 2020, 6, 3608–3618. [Google Scholar] [CrossRef]
- Fernandes, S.C.M.; Sadocco, P.; Alonso-Verona, A.; Palomares, T.; Ceiza, A.; Silvestre, A.J.D.; Mondragon, I.; Freire, C.S.R. Bioinspired antimicrobial and biocompatible bacterial cellulose membranes obtained by surface functionalization with aminoalkyl groups. ACS Appl. Mater. Interfaces 2013, 5, 3290–3297. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718. [Google Scholar] [CrossRef]
- Wei, T.; Yu, Q.; Chen, H. Responsive and synergistic antibacterial coatings: Fighting against bacteria in a smart and effective way. Adv. Healthc. Mater. 2019, 8, 1801381. [Google Scholar] [CrossRef] [PubMed]
- Mamba, S.M.; Mishra, A.K.; Mamba, B.B.; Njobeh, P.B.; Dutton, M.F.; Fosso-Kankeu, E. Spectral, Thermal and in vitro antimicrobial studies of cyclohexylamine-N-dithiocarbamate transition metal complexes. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 77, 579–587. [Google Scholar] [CrossRef]
- Butler, J.; Handy, R.D.; Upton, M.; Besinis, A. Review of antimicrobial nanocoatings in medicine and dentistry: Mechanisms of action, biocompatibility performance, safety, and benefits compared to antibiotics. ACS Nano 2013, 17, 7064–7092. [Google Scholar] [CrossRef]






| Sample | Epoxy Resin | Resin Precursor Amount (g) | Curing Agent Amount (g) | Density (g/cm3) 1 | Tg (°C) 2 | TGA (°C) 3 |
|---|---|---|---|---|---|---|
| 1a | EC157/W152LR | 120 | 40 | 1.153 | 79.9 (74) | 332 |
| 1b | EC157/W152LR | 120 | 45 | 1.148 | 82.9 (84) | 330 |
| 2a | MGS/RIMH235 | 100 | 34 | 1.156 | 79.9 (85) | 338 |
| 2b | MGS/RIMH235 | 100 | 38 | 1.160 | 80.9 (80) | 333 |
| 3 | MC152/W101 | 100 | 12 | 1.797 | 48.6 (53) | 319 4 |
| Sample | Tensile Strength (MPa) | Young Modulus (GPa) | Fracture Toughness (MPa m0.5) |
|---|---|---|---|
| 1a | 71.2 ± 3.0 | 2.86 ± 0.13 | 0.90 ± 0.09 |
| 1b | 59.1 ± 3.1 | 2.59 ± 0.25 | 1.17 ± 0.14 |
| 2a | 54.4 ± 0.6 | 2.42 ± 0.29 | 1.31 ± 0.13 |
| 2b | 67.6 ± 1.2 | 2.87 ± 0.06 | 1.04 ± 0.11 |
| 3 | 26.7 ± 0.7 | 8.87 ± 0.65 | 2.60 ± 0.18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rigo, M.; Khatami, H.; Mansi, A.; Marcelloni, A.M.; Proietto, A.R.; Chiominto, A.; Amori, I.; Bargellini, A.; Marchesi, I.; Frezza, G.; et al. Revealing Commercial Epoxy Resins’ Antimicrobial Activity: A Combined Chemical–Physical, Mechanical, and Biological Study. Polymers 2024, 16, 2571. https://doi.org/10.3390/polym16182571
Rigo M, Khatami H, Mansi A, Marcelloni AM, Proietto AR, Chiominto A, Amori I, Bargellini A, Marchesi I, Frezza G, et al. Revealing Commercial Epoxy Resins’ Antimicrobial Activity: A Combined Chemical–Physical, Mechanical, and Biological Study. Polymers. 2024; 16(18):2571. https://doi.org/10.3390/polym16182571
Chicago/Turabian StyleRigo, Mario, Hamoun Khatami, Antonella Mansi, Anna Maria Marcelloni, Anna Rita Proietto, Alessandra Chiominto, Ilaria Amori, Annalisa Bargellini, Isabella Marchesi, Giuseppina Frezza, and et al. 2024. "Revealing Commercial Epoxy Resins’ Antimicrobial Activity: A Combined Chemical–Physical, Mechanical, and Biological Study" Polymers 16, no. 18: 2571. https://doi.org/10.3390/polym16182571
APA StyleRigo, M., Khatami, H., Mansi, A., Marcelloni, A. M., Proietto, A. R., Chiominto, A., Amori, I., Bargellini, A., Marchesi, I., Frezza, G., Lipani, F., Cermelli, C., Rossini, A., Quaresimin, M., Zappalorto, M., Pontefisso, A., Pastrello, M., Rossetto, D., Modesti, M., ... Bertani, R. (2024). Revealing Commercial Epoxy Resins’ Antimicrobial Activity: A Combined Chemical–Physical, Mechanical, and Biological Study. Polymers, 16(18), 2571. https://doi.org/10.3390/polym16182571








