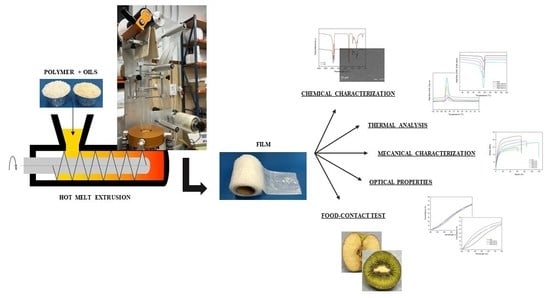
Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
Polymer and Oils
2.2. Preparation
2.2.1. Extrusion
2.2.2. Film Manufacturing
2.3. Chemico-Physical and Mechanical Charaterization
2.3.1. Thermal Characterization
2.3.2. Chemical and Morphological Characterization
2.3.3. Mechanical Characterization
2.3.4. Optical Properties
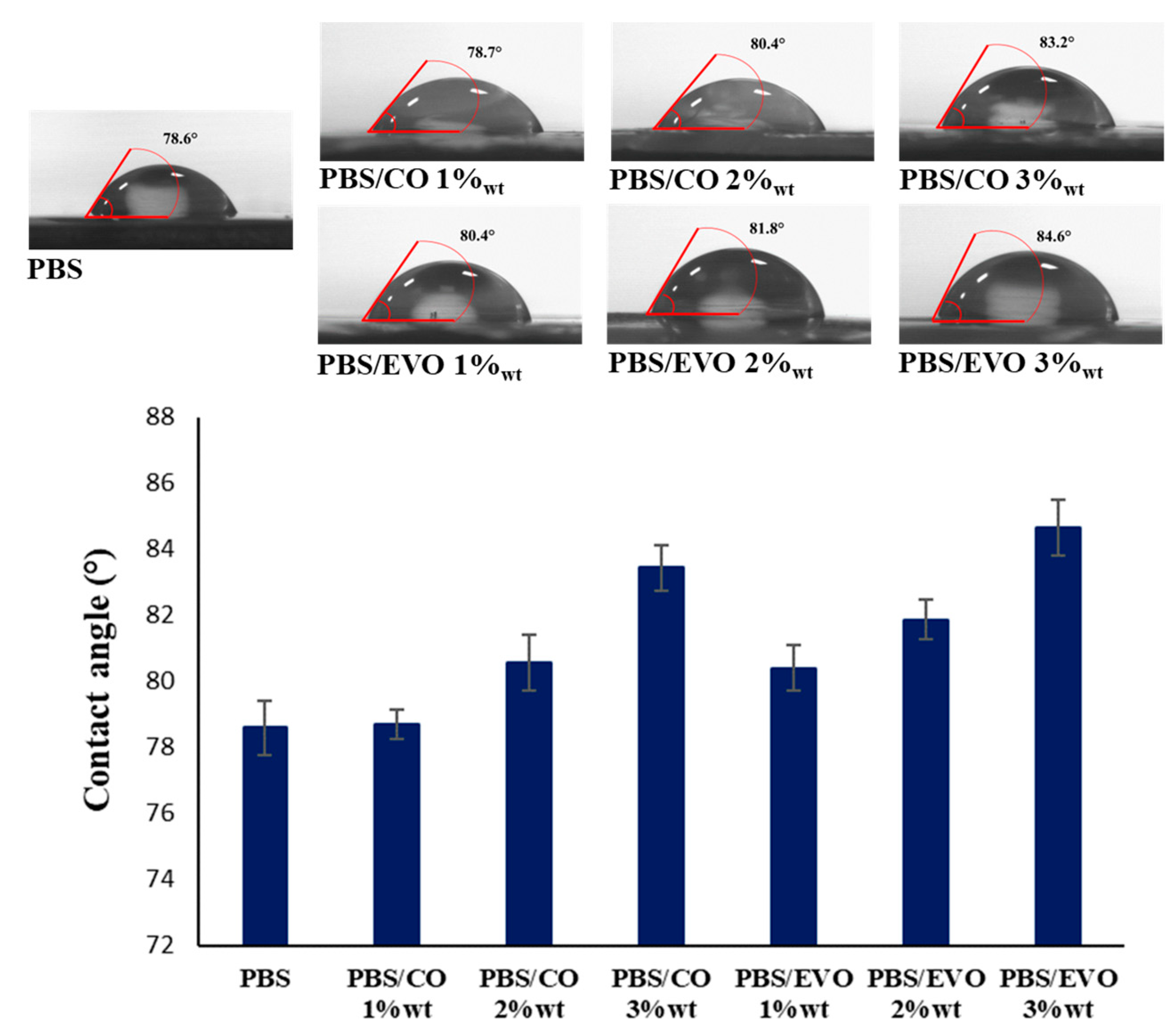
2.3.5. Static Water Contact-Angle Measurements
2.3.6. Food-Contact Test
3. Results
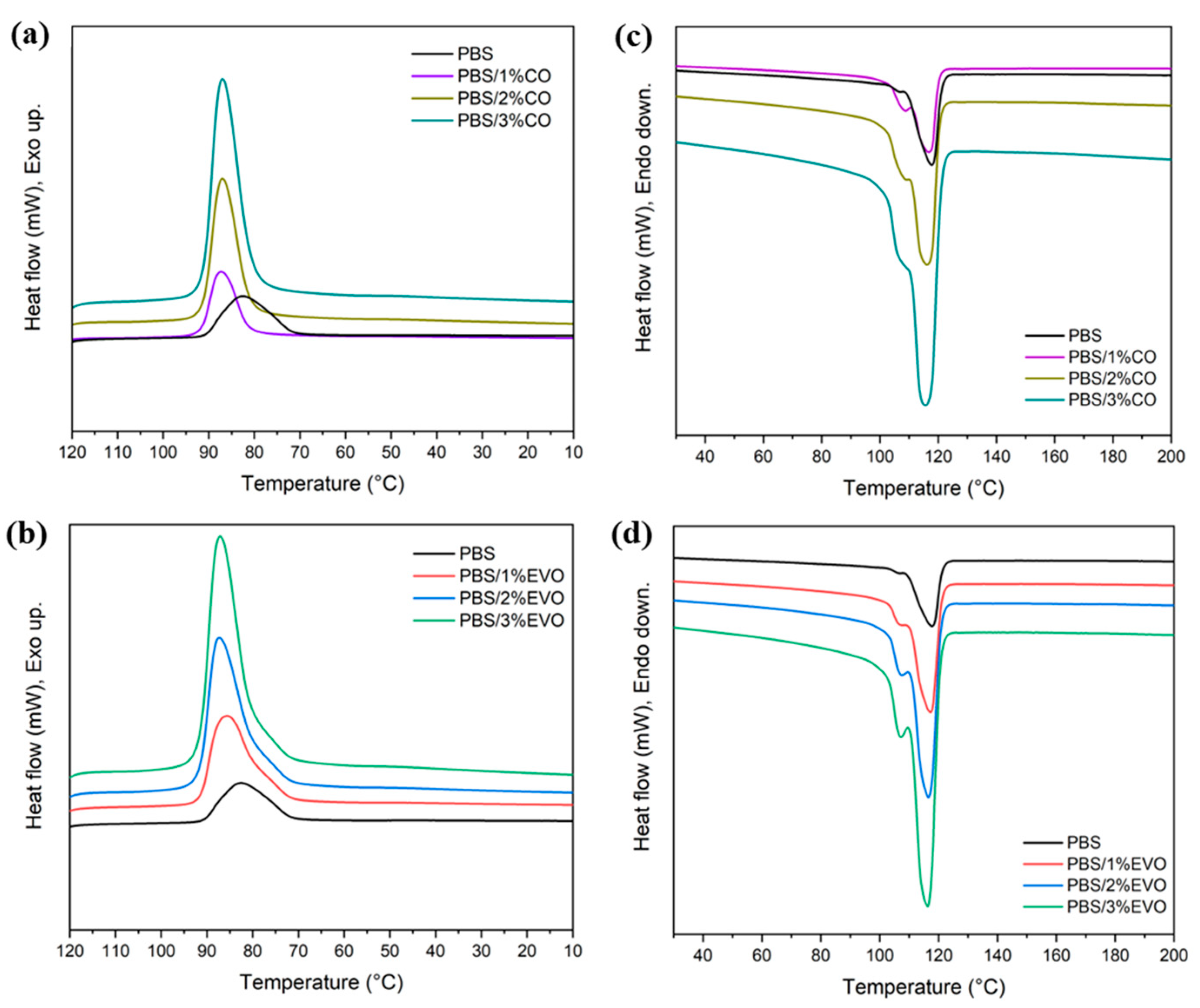
3.1. Thermal Characterization
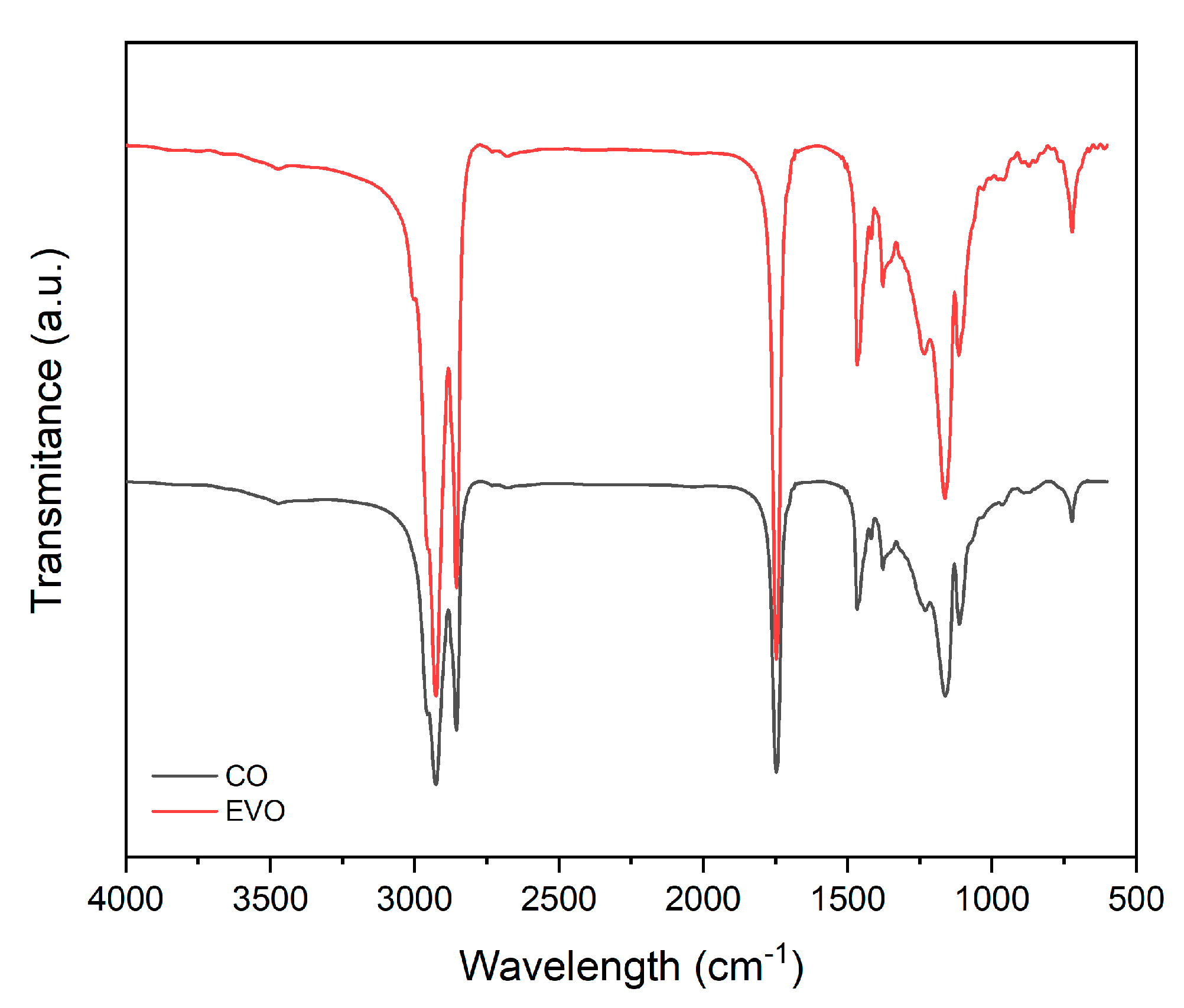
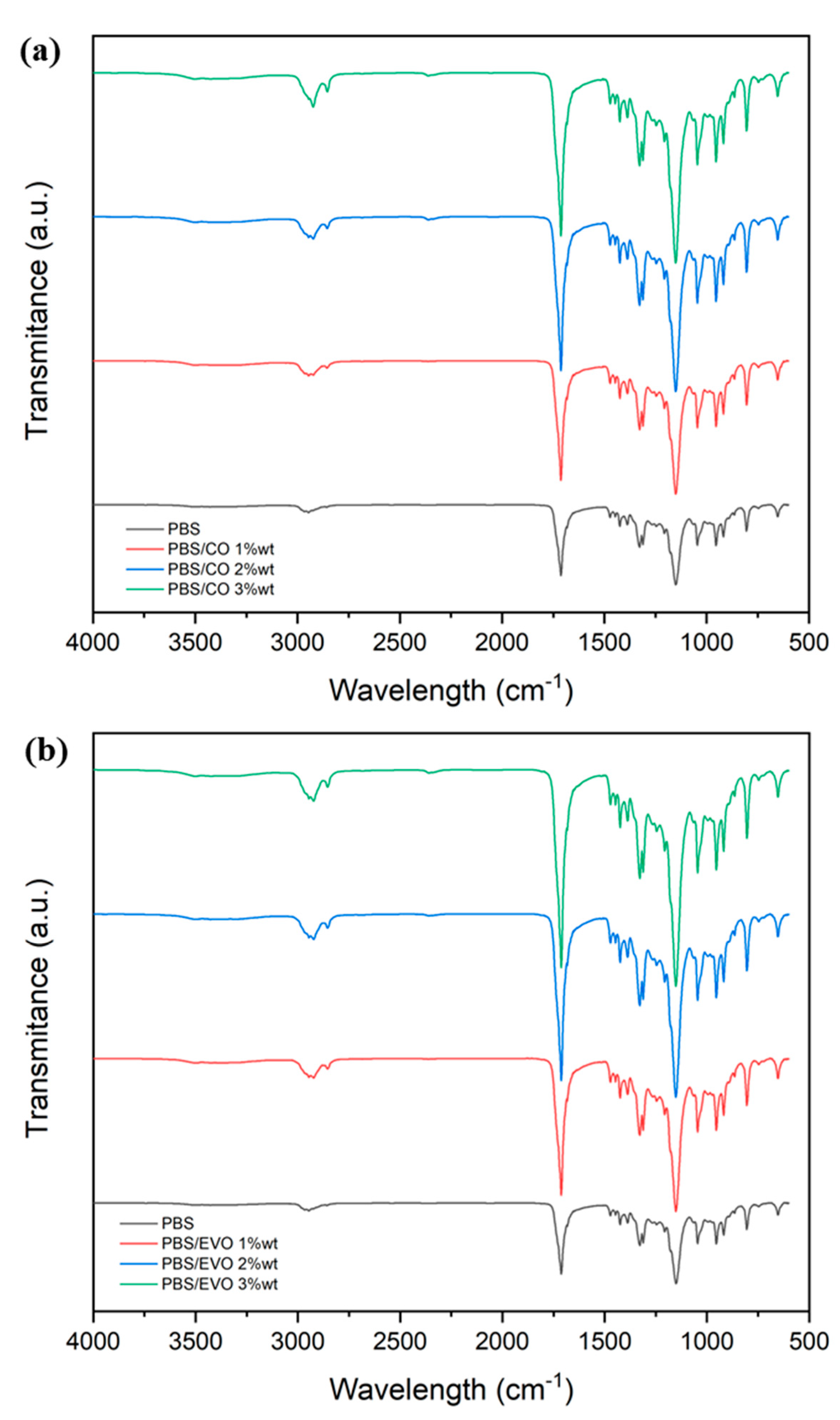
3.2. Chemical and Morphological Characterization
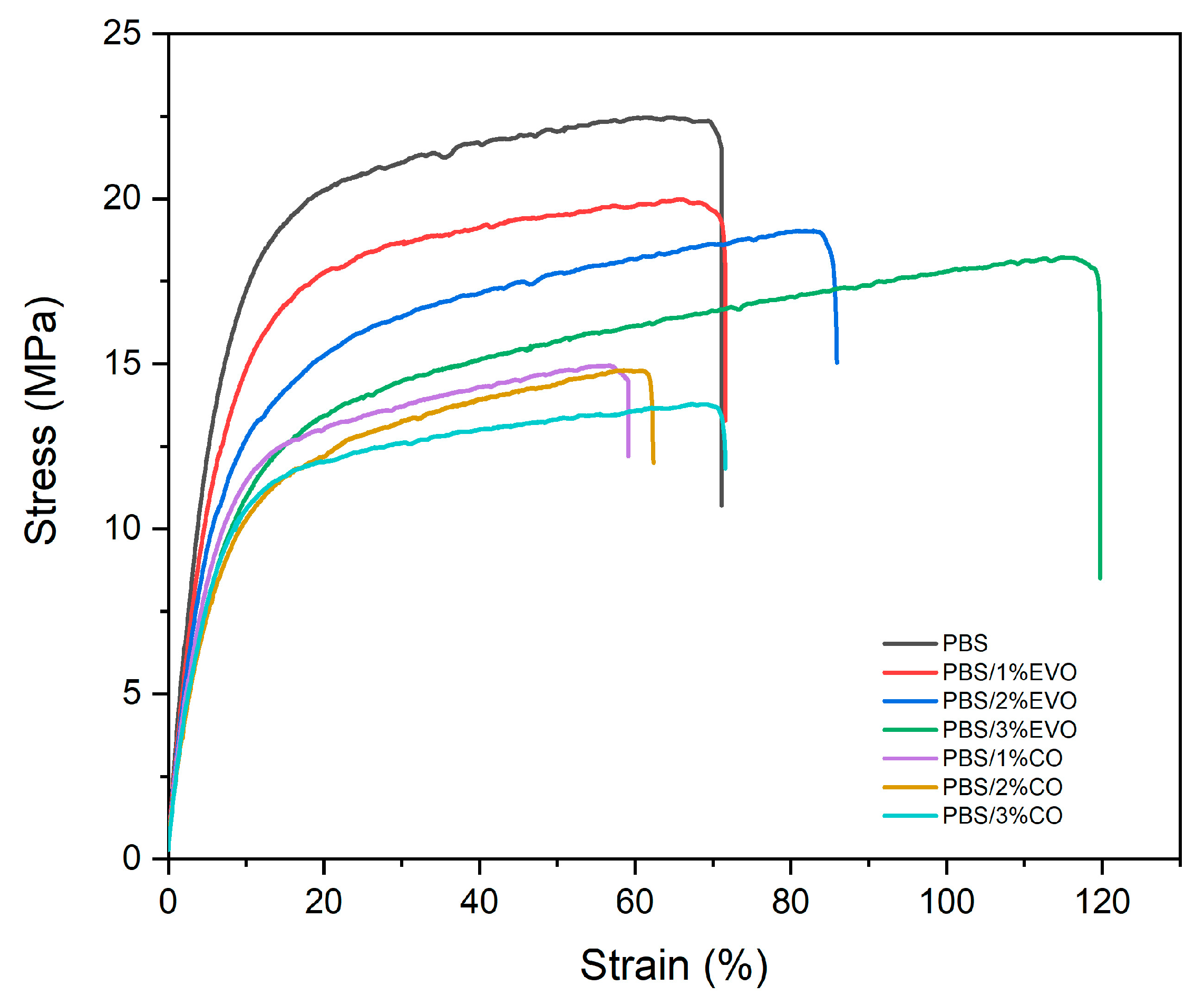
3.3. Mechanical Charaterization
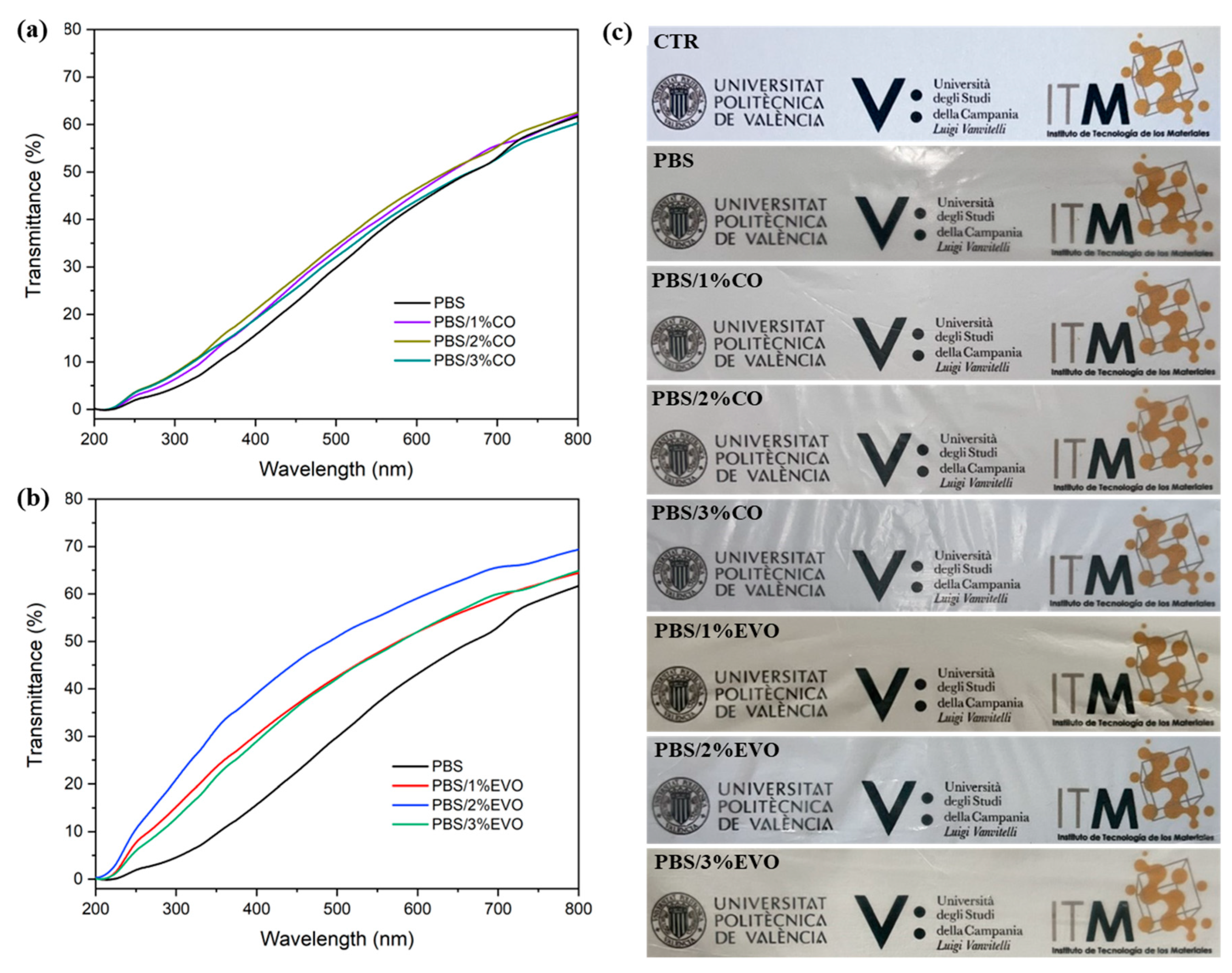
3.4. Optical Properties
3.5. Water-Absorption Analysis
3.6. Food-Contact Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kummu, M.; De Moel, H.; Porkka, M.; Siebert, S.; Varis, O.; Ward, P.J. Lost food, wasted resources: Global food supply chain losses and their impacts on freshwater, cropland, and fertiliser use. Sci. Total Environ. 2012, 438, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Stenmarck, Â.; Jensen, C.; Quested, T.; Moates, G.; Buksti, M.; Cseh, B.; Juul, S.; Parry, A.; Politano, A.; Redlingshofer, B.; et al. Estimates of European Food Waste Levels; IVL Swedish Environmental Research Institute: Stockholm, Sweden, 2016; p. 79. Available online: https://research.wur.nl/en/publications/estimates-of-european-food-waste-levels (accessed on 1 January 2023).
- Anastasopoulou, A.; Fortibuoni, T. Impact of plastic pollution on marine life in the Mediterranean Sea. In Plastics in the Aquatic Environment—Part I; Springer: Cham, Switzerland, 2019; pp. 135–196. [Google Scholar] [CrossRef]
- Nazrin, A.; Sapuan, S.M.; Zuhri, M.Y.M.; Ilyas, R.A.; Syafiq, R.S.F.K.S.; Sherwani, S.F.K. Nanocellulose reinforced thermoplastic starch (TPS), polylactic acid (PLA), and polybutylene succinate (PBS) for food packaging applications. Front. Chem. 2020, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Fetting, C. The European Green Deal. In ESDN Report; ESDN Office: Vienna, Austria, 2020. [Google Scholar]
- Zamri, G.B.; Azizal, N.K.A.; Nakamura, S.; Okada, K.; Nordin, N.H.; Othman, N.; Akhir, F.N.M.D.; Kaida, N.; Hara, H. Delivery, impact and approach of household food waste reduction campaigns. J. Clean. Prod. 2020, 246, 118969. [Google Scholar] [CrossRef]
- Mangara, S.; Yadav, A.; Bal, L.M.; Dash, S.K.; Mahanti, N.K. Application of biodegradable polymers in food packaging industry: A comprehensive review. J. Package Technol. Res. 2019, 3, 77–96. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Dalla Rosa, M. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Rafiqah, S.A.; Khalina, A.; Harmaen, A.S.; Tawakkal, I.A.; Zaman, K.; Asim, M.; Nurrazi, M.N.; Lee, C.H. A review on properties and application of bio-based poly (butylene succinate). Polymers 2021, 13, 1436. [Google Scholar] [CrossRef] [PubMed]
- Pennacchio, A.; Ventorino, V.; Cimini, D.; Pepe, O.; Schiraldi, C.; Inverso, M.; Faraco, V. Isolation of new cellulase and xylanase producing strains and application to lignocellulosic biomasses hydrolysis and succinic acid production. Bioresour. Technol. 2018, 259, 325–333. [Google Scholar] [CrossRef]
- Mohamad, N.; Mohamad Mazlan, M.; Tawakkal, I.S.M.A.; Talib, R.A.; Kian, L.K.; Jawaid, M. Characterization of active polybutylene succinate films filled essential oils for food packaging application. J. Polym. Environ. 2022, 30, 585–596. [Google Scholar] [CrossRef]
- Stoleru, E.; Brebu, M. Stabilization techniques of essential oils by incorporation into biodegradable polymeric materials for food packaging. Molecules 2021, 26, 6307. [Google Scholar] [CrossRef]
- Nikolic, M.S.; Djonlagic, J. Synthesis and characterization of biodegradable poly (butylene succinate-co-butylene adipate) s. Polym. Degrad. Stab. 2001, 74, 263–270. [Google Scholar] [CrossRef]
- Rohman, A.; Che Man, Y.B. Potential use of FTIR-ATR spectroscopic method for determination of virgin coconut oil and extra virgin olive oil in ternary mixture systems. Food Anal. Methods 2011, 4, 155–162. [Google Scholar] [CrossRef]
- Lerma-García, M.J.; Ramis-Ramos, G.; Herrero-Martínez, J.M.; Simó-Alfonso, E.F. Authentication of extra virgin olive oils by Fourier-transform infrared spectroscopy. Food Chem. 2010, 118, 78–83. [Google Scholar] [CrossRef]
- Abderrahim, B.; Abderrahman, E.; Mohamed, A.; Fatima, T.; Abdesselam, T.; Krim, O. Kinetic thermal degradation of cellulose, polybutylene succinate and a green composite: Comparative study. World J. Environ. Eng. 2015, 3, 95–110. [Google Scholar] [CrossRef]
- Qiu, Z.; Komura, M.; Ikehara, T.; Nishi, T. DSC and TMDSC study of melting behaviour of poly (butylene succinate) and poly (ethylene succinate). Polymer 2003, 44, 7781–7785. [Google Scholar] [CrossRef]
- Fenni, S.E.; Wang, J.; Haddaoui, N.; Favis, B.D.; Müller, A.J.; Cavallo, D. Crystallization and self-nucleation of PLA, PBS and PCL in their immiscible binary and ternary blends. Thermochim. Acta 2019, 677, 117–130. [Google Scholar] [CrossRef]
- Blanco, I.; Poggetto, G.D.; Morrone, B.; Tranquillo, E.; Barrino, F.; Catauro, M. Fly Ash Filled Geopolymers: Preparation and Thermal Study. Macromol. Symp. 2020, 389, 1900052. [Google Scholar] [CrossRef]
- Peñas, M.I.; Pérez-Camargo, R.A.; Hernández, R.; Müller, A.J. A Review on Current Strategies for the Modulation of Thermomechanical, Barrier, and Biodegradation Properties of Poly (Butylene Succinate)(PBS) and Its Random Copolymers. Polymers 2022, 14, 1025. [Google Scholar] [CrossRef]
- De La Rosa-Ramírez, H.; Aldas, M.; Ferri, J.M.; López-Martínez, J.; Samper, M.D. Modification of poly (lactic acid) through the incorporation of gum rosin and gum rosin derivative: Mechanical performance and hydrophobicity. J. Appl. Polym. Sci. 2020, 137, 49346. [Google Scholar] [CrossRef]
- Rohman, A. Infrared spectroscopy for quantitative analysis and oil parameters of olive oil and virgin coconut oil: A review. Int. J. Food Prop. 2017, 20, 1447–1456. [Google Scholar] [CrossRef]
- Tranquillo, E.; Barrino, F.; Dal Poggetto, G.; Blanco, I. Sol–gel synthesis of silica-based materials with different percentages of PEG or PCL and high chlorogenic acid content. Materials 2019, 12, 155. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Pacifico, S.; Piccolella, S.; Lancellotti, I.; Leonelli, C. Synthesis of WEEE-based geopolymers and their cytotoxicity. Mater. Today Proc. 2021, 34, 121–124. [Google Scholar] [CrossRef]
- Catauro, M.; Barrino, F.; Dal Poggetto, G.; Milazzo, M.; Blanco, I.; Ciprioti, S.V. Structure, drug absorption, bioactive and antibacterial properties of sol-gel SiO2/ZrO2 materials. Ceram. Int. 2020, 46, 29459–29465. [Google Scholar] [CrossRef]
- Ferri, J.M.; Garcia-Garcia, D.; Sánchez-Nacher, L.; Fenollar, O.; Balart, R. The effect of maleinized linseed oil (MLO) on mechanical performance of poly (lactic acid)-thermoplastic starch (PLA-TPS) blends. Carbohyd. Polym. 2016, 147, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Ferri, J.M.; Samper, M.D.; García-Sanoguera, D.; Reig, M.J.; Fenollar, O.; Balart, R. Plasticizing effect of biobased epoxidized fatty acid esters on mechanical and thermal properties of poly (lactic acid). J. Mater. Sci. 2016, 51, 5356–5366. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Samper, M.D.; Jiménez-López, M.; Aldas, M.; López, J. Combined effect of linseed oil and gum rosin as natural additives for PVC. Ind. Crop. Prod. 2017, 99, 196–204. [Google Scholar] [CrossRef]
- Luzi, F.; Del Buono, D.; Orfei, B.; Moretti, C.; Bounaurio, R.; Torre, L.; Puglia, D. Lemna minor aqueous extract as a natural ingredient incorporated in poly (vinyl alcohol)-based films for active food packaging systems. Food Packag. Shelf Life 2022, 32, 100822. [Google Scholar] [CrossRef]




| Label | Composition | ||
|---|---|---|---|
| Polymer (wt%) | Olive Oil EVO (wt%) | Coconut Oil (wt%) | |
| PBS | 100 | --- | --- |
| PBS/CO 1 wt% | 99 | --- | 1 |
| PBS/CO 2 wt% | 98 | --- | 2 |
| PBS/CO 3 wt% | 97 | --- | 3 |
| PBS/EVO 1 wt% | 99 | 1 | --- |
| PBS/EVO 2 wt% | 98 | 2 | --- |
| PBS/EVO 3 wt% | 97 | 3 | --- |
| Samples | DSC (at a Stream Rate of 10 °C min−1) | ||||
|---|---|---|---|---|---|
| Tcc (°C) 1 | ΔHcc (Jg−1) 2 | Tm (°C) 3 | ΔHm (Jg−1) 4 | Xc (%) 5 | |
| PBS | 82.6 | 69.2 | 117.8 | 74.1 | 0.04 |
| PBS/CO 1 wt% | 87.2 | 66.9 | 116.8 | 74.9 | 0.07 |
| PBS/CO 2 wt% | 86.7 | 64.1 | 114.9 | 68.1 | 0.04 |
| PBS/CO 3 wt% | 86.7 | 66.3 | 114.4 | 71.6 | 0.05 |
| PBS/EVO 1 wt% | 86.9 | 70.8 | 116.7 | 76.2 | 0.05 |
| PBS/EVO 2 wt% | 87.9 | 63.3 | 116.3 | 68.8 | 0.05 |
| PBS/EVO 3 wt% | 86.8 | 66.7 | 115.2 | 72.1 | 0.05 |
| Peak (cm−1) | Assignment of Bonds | Mode of Vibration |
|---|---|---|
| 3006 | =C–H | Stretching |
| 2930 | =C–H (CH2) | Stretching (asymmetrical) |
| 2855 | =C–H (CH2) | Stretching (symmetrical) |
| 1747 | C=O | Stretching |
| 1466 | C–H (CH2) | Bending (scissoring) |
| 1415 | =C–H | Bending (rocking) |
| 1377 | C–H (CH2) | Bending (symmetrical) |
| 1161 | CH2 | Bending |
| 1113, 1095, 1028 | C–O | Stretching |
| 965 | CH=CH | Bending out of plane |
| 858 | =CH2 | Wagging |
| 722 | CH=CH | Bending out of plane |
| Sample | Tensile Strength (Mpa) | Tensile Modulus (Mpa) | Elongation at Break (%) |
|---|---|---|---|
| PBS | 22.5 ± 1.29 | 361.5 ± 1.91 | 71.1 ± 2.22 |
| PBS/CO 1 wt% | 15.0 ± 2.06 | 291.6 ± 10.5 | 59.2 ± 2.80 |
| PBS/CO 2 wt% | 14.8 ± 1.47 | 226.3 ± 9.44 | 62.4 ± 3.11 |
| PBS/CO 3 wt% | 13.8 ± 0.80 | 249.1 ± 9.99 | 71.6 ± 2.21 |
| PBS/EVO 1 wt% | 20.2 ± 1.71 | 336.4 ± 2.76 | 71.9 ± 2.95 |
| PBS/EVO 2 wt% | 19.0 ± 2.27 | 300.4 ± 4.18 | 85.9 ± 2.28 |
| PBS/EVO 3 wt% | 18.2 ± 1.42 | 244.8 ± 8.60 | 119.8 ± 3.21 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barrino, F.; De La Rosa-Ramírez, H.; Schiraldi, C.; López-Martínez, J.; Samper, M.D. Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS). Polymers 2023, 15, 1212. https://doi.org/10.3390/polym15051212
Barrino F, De La Rosa-Ramírez H, Schiraldi C, López-Martínez J, Samper MD. Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS). Polymers. 2023; 15(5):1212. https://doi.org/10.3390/polym15051212
Chicago/Turabian StyleBarrino, Federico, Harrison De La Rosa-Ramírez, Chiara Schiraldi, Juan López-Martínez, and María Dolores Samper. 2023. "Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS)" Polymers 15, no. 5: 1212. https://doi.org/10.3390/polym15051212
APA StyleBarrino, F., De La Rosa-Ramírez, H., Schiraldi, C., López-Martínez, J., & Samper, M. D. (2023). Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS). Polymers, 15(5), 1212. https://doi.org/10.3390/polym15051212