Promising Agromaterials Based on Biodegradable Polymers: Polylactide and Poly-3-Hydroxybutyrate
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Seed Germination Test
2.3. Analysis of Crystallization
2.4. Morphology of the Sample
2.5. Testing of Mechanical Properties
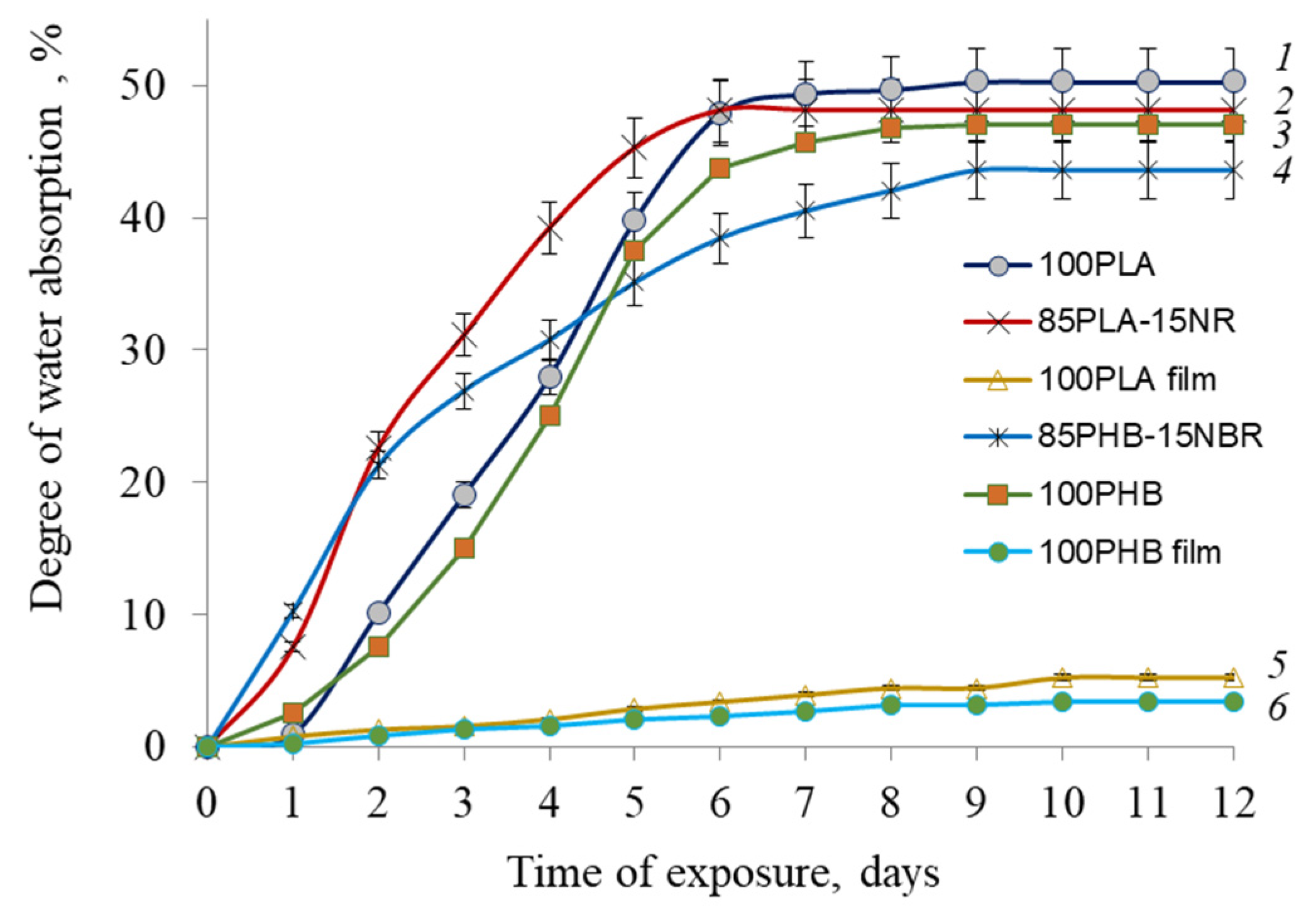
2.6. Water Uptake
2.7. FTIR-ATR Spectroscopy
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khalid, M.Y.; Arif, Z.U. Novel biopolymer-based sustainable composites for food packaging applications: A narrative review. Food Packag. Shelf Life 2022, 33, 100892. [Google Scholar] [CrossRef]
- Abdel-Bari, E.M. Polymer Films; Zaikov, G.E., Ed.; Professiya: St. Petersburg, Russia, 2005; p. 352. [Google Scholar]
- Thakur, M.; Kumar, R. Mulching: Boosting crop productivity and improving soil environment in herbal plants. J. Appl. Res. Med. Aromat. Plants 2020, 20, 100287. [Google Scholar] [CrossRef]
- Zhirmunskaya, N.M. Environmentally Friendly Farming in the Garden (with the Basics of Biodynamics); Marketing: Moscow, Russia, 1996; pp. 93–97. [Google Scholar]
- Stenberg, J.A. A Conceptual Framework for Integrated Pest Management. Trends Plant Sci. 2017, 22, 759–769. [Google Scholar] [CrossRef]
- Briassoulis, D.; Babou, E.; Hiskakis, M.; Kyrikou, I. Analysis of long-term degradation behaviour of polyethylene mulching films with pro-oxidants under real cultivation and soil burial conditions. Environ. Sci. Pollut. Res. 2015, 22, 2584–2598. [Google Scholar] [CrossRef] [PubMed]
- Portillo, F.; Yashchuk, O.; Hermida, É. Evaluation of the rate of abiotic and biotic degradation of oxo-degradable polyethylene. Polym. Test. 2016, 53, 58–69. [Google Scholar] [CrossRef]
- Siwek, P.; Domagala-Swiatkiewicz, I.; Bucki, P.; Puchalski, M. Biodegradable agroplastics in 21st century horticulture. Polimery 2019, 64, 480–486. [Google Scholar] [CrossRef]
- Yang, Y.; Li, P.; Jiao, J.; Yang, Z.; Lv, M.; Li, Y.; Zhou, C.; Wang, C.; He, Z.; Liu, Y.; et al. Renewable sourced biodegradable mulches and their environment impact. Sci. Hortic. 2020, 268, 109375. [Google Scholar] [CrossRef]
- Rogovina, S.; Zhorina, L.; Gatin, A.; Prut, E.; Kuznetsova, O.; Yakhina, A.; Olkhov, A.; Samoylov, N.; Grishin, M.; Iordanskii, A.; et al. Biodegradable Polylactide–Poly(3-Hydroxybutyrate) Compositions Obtained via Blending under Shear Deformations and Electrospinning: Characterization and Environmental Application. Polymers 2020, 12, 1088. [Google Scholar] [CrossRef]
- Zlotin, B.L.; Garaev, A.I. Seeding Tape. RF Patent No. 4802221/15, 30 September 1994. [Google Scholar]
- Izmailov, A.Y.; Yelizarov, V.P.; Lobachevsky, Y.P.; Shaikhov, M.K.; Pyshkin, V.K.; Tsarkova, T.V.; Shaikhov, M.M. The unit for seeding the seed material in the tape. RF Patent No. 2013151476/13, 10 April 2015. [Google Scholar]
- Volova, T.G.; Vinogradova, O.N.; Zhila, N.O.; Kiselev, E.G.; Peterson, I.V.; Vasil’Ev, A.D.; Sukovatyi, A.G.; Shishatskaya, E. Physicochemical properties of multicomponent polyhydroxyalkanoates: Novel aspects. Polym. Sci. Ser. A 2017, 59, 98–106. [Google Scholar] [CrossRef]
- Tertyshnaya, Y.V.; Shibryaeva, L.S. Degradation of poly(3-hydroxybutyrate) and its blends during treatment with UV light and water. Polym. Sci. Ser. B 2013, 55, 164–168. [Google Scholar] [CrossRef]
- Podzorova, M.V.; Tertyshnaya, Y.V.; Popov, A.A. Environmentally friendly films based on poly(3-hydroxybutyrate) and poly(lactic acid): A review. Russ. J. Phys. Chem. B 2014, 8, 726–732. [Google Scholar] [CrossRef]
- Arif, Z.U.; Khalid, M.Y.; Zolfagharian, A.; Bodaghi, M. Reactive and Functional Polymers Review 4D bioprinting of smart polymers for biomedical applications: Recent progress, challenges, and future perspectives. React. Funct. Polym. 2022, 179, 105374. [Google Scholar] [CrossRef]
- Umapathi, R.; Ghoreishian, S.M.; Sonwal, S.; Rani, G.M.; Huh, Y.S. Portable electrochemical sensing methodologies for on-site detection of pesticide residues in fruits and vegetables. Rev. Co-ord. Chem. Rev. 2022, 453, 214305. [Google Scholar] [CrossRef]
- Nampoothiri, K.M.; Nair, N.R.; John, R.P. An overview of the recent developments in polylactide (PLA) research. Bioresour. Technol. 2010, 101, 8493–8501. [Google Scholar] [CrossRef]
- Garlotta, D. Literature Review of Poly(Lactic Acid) A Literature Review of Poly(Lactic Acid). J. Polym. Environ. 2001, 9, 63–84. [Google Scholar] [CrossRef]
- Tertyshnaya, Y.V.; Podzorova, M.V. Effect of UV Irradiation on the Structural and Dynamic Characteristics of Polylactide and Its Blends with Polyethylene. Russ. J. Phys. Chem. B 2020, 14, 167–175. [Google Scholar] [CrossRef]
- Bocchini, S.; Fukushima, K.; Di Blasio, A.; Fina, A.; Frache, A.; Geobaldo, F. Polylactic Acid and Polylactic Acid-Based Nanocomposite Photooxidation. Biomacromolecules 2010, 11, 2919–2926. [Google Scholar] [CrossRef] [PubMed]
- Si, W.-J.; Yuan, W.-Q.; Li, Y.-D.; Chen, Y.-K.; Zeng, J.-B. Tailoring toughness of fully biobased poly(lactic acid)/natural rubber blends through dynamic vulcanization. Polym. Test. 2018, 65, 249–255. [Google Scholar] [CrossRef]
- Maroufkhani, M.; Katbab, A.; Zhang, J. Manipulation of the properties of PLA nanocomposites by controlling the distribution of nanoclay via varying the acrylonitrile content in NBR rubber. Polym. Test. 2018, 65, 313–321. [Google Scholar] [CrossRef]
- Yang, F.; Murugan, R.; Wang, S.; Ramakrishna, S. Electrospinning of nano/micro scale poly(l-lactic acid) aligned fibers and their potential in neural tissue engineering. Biomaterials 2005, 26, 2603–2610. [Google Scholar] [CrossRef]
- Mujica-Garcia, A.; Navarro-Baena, I.; Kenny, J.M.; Peponi, L. Influence of the Processing Parameters on the Electrospinning of Biopolymeric Fibers. J. Renew. Mater. 2014, 2, 23–34. [Google Scholar] [CrossRef]
- Tertyshnaya, Y.; Karpova, S.; Moskovskiy, M.; Dorokhov, A. Electrospun Polylactide/Natural Rubber Fibers: Effect Natural Rubber Content on Fiber Morphology and Properties. Polymers 2021, 13, 2232. [Google Scholar] [CrossRef] [PubMed]
- Nicosia, A.; Gieparda, W.; Foksowicz-Flaczyk, J.; Walentowska, J.; Wesołek, D.; Vazquez, B.; Prodi, F.; Belosi, F. Air filtration and antimicrobial capabilities of electrospun PLA/PHB containing ionic liquid. Sep. Purif. Technol. 2015, 154, 154–160. [Google Scholar] [CrossRef]
- Karpova, S.G.; Tertyshnaya, Y.V.; Podzorova, M.V.; Popov, A.A. Effect of Exposure in Aqueous Medium at Elevated Temperature on the Structure of Nonwoven Materials Based on Polylactide and Natural Rubber. Polym. Sci. Ser. A 2021, 62, 515–525. [Google Scholar] [CrossRef]
- Tertyshnaya, Y.; Podzorova, M.; Moskovskiy, M. Impact of Water and UV Irradiation on Nonwoven Polylactide/Natural Rubber Fiber. Polymers 2021, 13, 461. [Google Scholar] [CrossRef] [PubMed]
- Tertyshnaya, Y.V.; Karpova, S.G.; Podzorova, M.V.; Khvatov, A.V.; Moskovskiy, M.N. Thermal Properties and Dynamic Characteristics of Electrospun Polylactide/Natural Rubber Fibers during Disintegration in Soil. Polymers 2022, 14, 1058. [Google Scholar] [CrossRef]
- GOST 12038-84; Agricultural Seeds. Methods for Determining Seed Germination. Standards Publishing House: Moscow, Russia, 1990; p. 29.
- Sarasua, J.-R.; Balerdi, P.; Maiza, I. Crystallization and thermal behaviour of optically pure polylactides and their blends. J. Mater. Sci. 2005, 40, 1855–1862. [Google Scholar] [CrossRef]
- Bao, Q.; Wong, W.; Liu, S.; Tao, X. Accelerated Degradation of Poly(lactide acid)/Poly(hydroxybutyrate) (PLA/PHB) Yarns/Fabrics by UV and O2 Exposure in South China Seawater. Polymers 2022, 14, 1216. [Google Scholar] [CrossRef]
- GOST 4650-2014; Plastics—Methods for Determination of Water Absorption. ISS 83.080 OKSTU 2209; Standards Publishing House: Moscow, Russia, 1 March 2015.
- Obroucheva, N.V. Transition from hormonal to nonhormonal regulation as exemplified by seed dormancy release and germination triggering. Russ. J. Plant Physiol. 2012, 59, 546–555. [Google Scholar] [CrossRef]
- Tertyshnaya, Y.U.V.; Levina, N.S.; Popov, A.A.; Moskovskii, M.N.; Izmailov, A.Y.U. Hydrolytic Destruction of Agrofiber Made of Natural Polymers. Fibre Chem. 2019, 51, 117–120. [Google Scholar] [CrossRef]
- Metlitsky, L.V. Biochemistry on Guard of the Harvest; Nauka: Moscow, Russia, 1965; p. 183. [Google Scholar]
- Obroucheva, N.V.; Sinkevich, I.A.; Lityagina, S.V.; Novikova, G.V. Water relations in germinating seeds. Russ. J. Plant Physiol. 2017, 64, 625–633. [Google Scholar] [CrossRef]
- Tomihata, K.; Suzuki, M.; Ikada, Y. The pH dependence of monofilament sutures on hydrolytic degradation. J. Biomed. Mater. Res. 2001, 58, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Olewnik-Kruszkowska, E. Influence of the type of buffer solution on thermal and structural properties of polylactide-based composites. Polym. Degrad. Stab. 2016, 129, 87–95. [Google Scholar] [CrossRef]
- Tertyshnaya, Y.V.; Karpova, S.G.; Popov, A.A. Effect of aqueous medium on the molecular mobility of polylactide. Russ. J. Phys. Chem. B 2017, 11, 531–537. [Google Scholar] [CrossRef]
- Jordá-Vilaplana, A.; Fombuena, V.; García-García, D.; Samper, M.; Sánchez-Nácher, L. Surface modification of polylactic acid (PLA) by air atmospheric plasma treatment. Eur. Polym. J. 2014, 58, 23–33. [Google Scholar] [CrossRef]
- Gorrasi, G.; Pantani, R. Hydrolysis and Biodegradation of Poly(lactic acid). In Synthesis, Structure and Properties of Poly(lactic acid); Di Lorenzo, M.L., Androsch, R., Eds.; Springer: Cham, Switzerland, 2018; pp. 119–151. [Google Scholar] [CrossRef]
- Maharana, T.; Mohanty, B.; Negi, Y. Melt–solid polycondensation of lactic acid and its biodegradability. Prog. Polym. Sci. 2009, 34, 99–124. [Google Scholar] [CrossRef]
- Boots, B.; Russell, C.W.; Green, D.S. Effects of Microplastics in Soil Ecosystems: Above and Below Ground. Environ. Sci. Technol. 2019, 53, 11496–11506. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, X.; Zhang, S.; Zhang, S.; Sun, Y. Interactions of microplastics and cadmium on plant growth and arbuscular mycorrhizal fungal communities in an agricultural soil. Chemosphere 2020, 254, 126791. [Google Scholar] [CrossRef]








| Type of the Substrate Sample | Weight, g | Root Length, cm | Plant Height, cm | |
|---|---|---|---|---|
| 1 Plant | Roots | |||
| Control sample | 0.156 ± 0.018 | 0.035 ± 0.004 | 8.8 ± 0.7 | 11.4 ± 0.4 |
| PLA | 0.184 ± 0.023 | 0.054 ± 0.005 | 11.0 ± 0.8 | 12.0 ± 0.5 |
| 85PLA/15NR | 0.191 ± 0.022 | 0.052 ± 0.003 | 12.3 ± 0.6 | 12.6 ± 0.5 |
| PHB | 0.182 ± 0.019 | 0.052 ± 0.004 | 13.4 ± 0.6 | 12.8 ± 0.4 |
| 85PHB/15NBR | 0.196 ± 0.020 | 0.050 ± 0.003 | 10.8 ± 0.7 | 12.5 ± 0.3 |
| Type of the Substrate Sample | Tm, °C (Δ ± 0.3 °C) | ΔHm, J/g (Δ ± 0.5 °C) | χc, % (Δ ± 0.5%) |
|---|---|---|---|
| PLA nonwoven fabric (n/w) initial | 165 | 37 | 40 |
| PLA n/w after seed germination | 161 | 35 | 38 |
| PLA n/w after saturation with water | 166 | 43 | 46 |
| 85PLA/15NR nonwoven fabric (n/w) initial | 166 | 36 | 38 |
| 85PLA/15NR n/w after seed germination | 165 | 32 | 34 |
| 85PLA/15NR n/w after saturation with water | 168 | 42 | 45 |
| PHB nonwoven fabric (n/w) initial | 173 | 84 | 58 |
| PHB n/w after seed germination | 163 | 53 | 36 |
| PHB n/w after saturation with water | 171 | 82 | 56 |
| 85PHB/15NBR nonwoven fabric (n/w) initial | 170 | 76 | 52 |
| 85PHB/15NBR n/w after seed germination | 163 | 59 | 40 |
| 85PHB/15NBR n/w after saturation with water | 165 | 60 | 41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tertyshnaya, Y.V.; Podzorova, M.V.; Varyan, I.A.; Tcherdyntsev, V.V.; Zadorozhnyy, M.Y.; Medvedeva, E.V. Promising Agromaterials Based on Biodegradable Polymers: Polylactide and Poly-3-Hydroxybutyrate. Polymers 2023, 15, 1029. https://doi.org/10.3390/polym15041029
Tertyshnaya YV, Podzorova MV, Varyan IA, Tcherdyntsev VV, Zadorozhnyy MY, Medvedeva EV. Promising Agromaterials Based on Biodegradable Polymers: Polylactide and Poly-3-Hydroxybutyrate. Polymers. 2023; 15(4):1029. https://doi.org/10.3390/polym15041029
Chicago/Turabian StyleTertyshnaya, Yulia Victorovna, Maria Victorovna Podzorova, Ivetta Aramovna Varyan, Victor Victorovich Tcherdyntsev, Mikhail Yurievich Zadorozhnyy, and Elena Valerievna Medvedeva. 2023. "Promising Agromaterials Based on Biodegradable Polymers: Polylactide and Poly-3-Hydroxybutyrate" Polymers 15, no. 4: 1029. https://doi.org/10.3390/polym15041029
APA StyleTertyshnaya, Y. V., Podzorova, M. V., Varyan, I. A., Tcherdyntsev, V. V., Zadorozhnyy, M. Y., & Medvedeva, E. V. (2023). Promising Agromaterials Based on Biodegradable Polymers: Polylactide and Poly-3-Hydroxybutyrate. Polymers, 15(4), 1029. https://doi.org/10.3390/polym15041029









