Shedding Light on Degradation Gradients in Celluloid: An ATR-FTIR Study of Artificially and Naturally Aged Specimens
Abstract
1. Introduction
2. Materials and Methods
2.1. Reference Materials
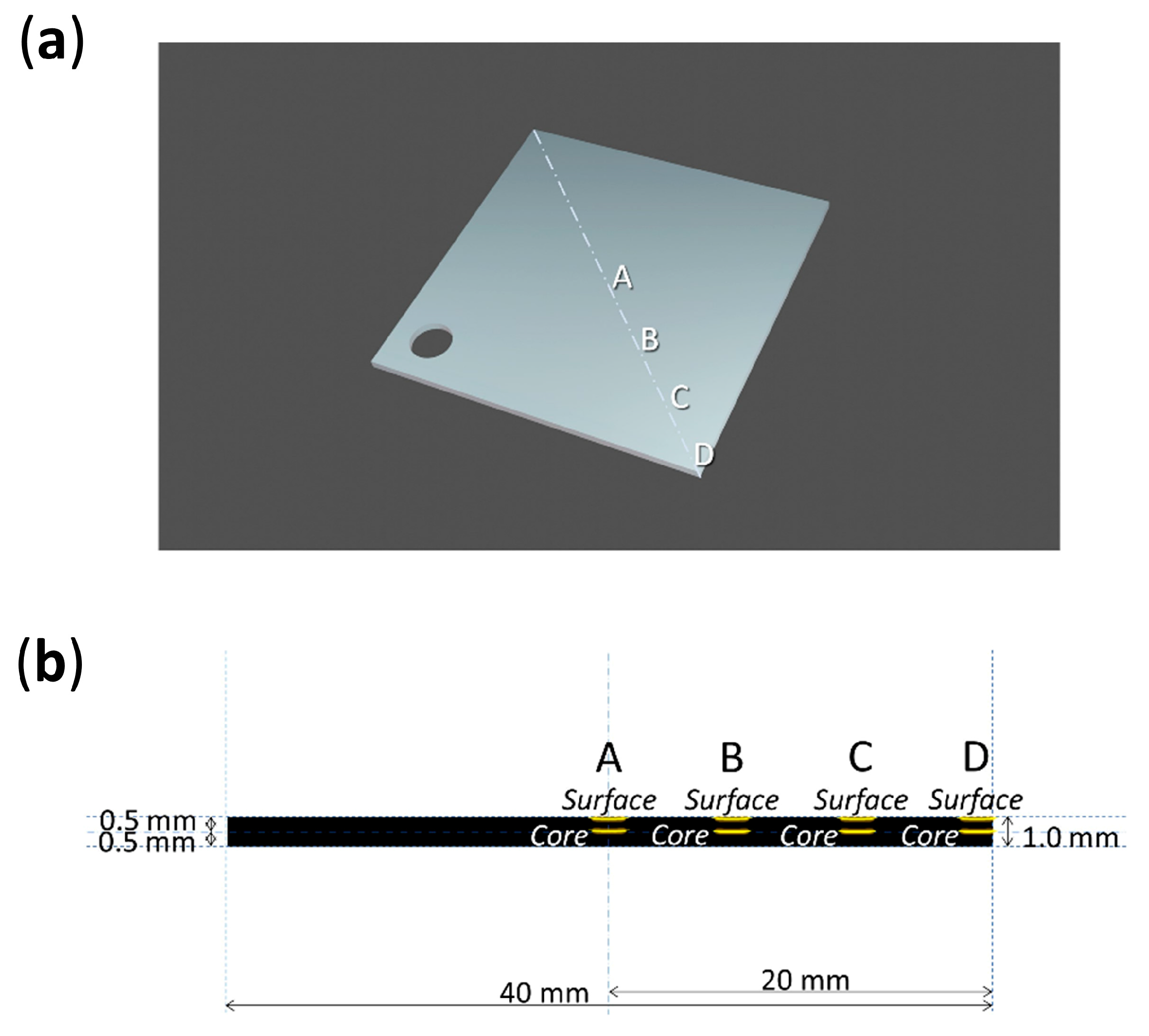
2.2. Celluloid
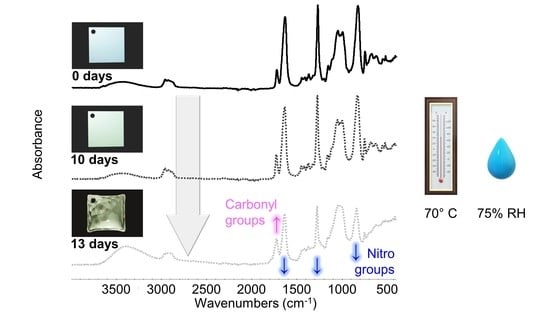
2.3. Artificial Aging of Celluloid Sheets
2.4. Naturally Aged Objects
2.5. Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy (ATR-FTIR)
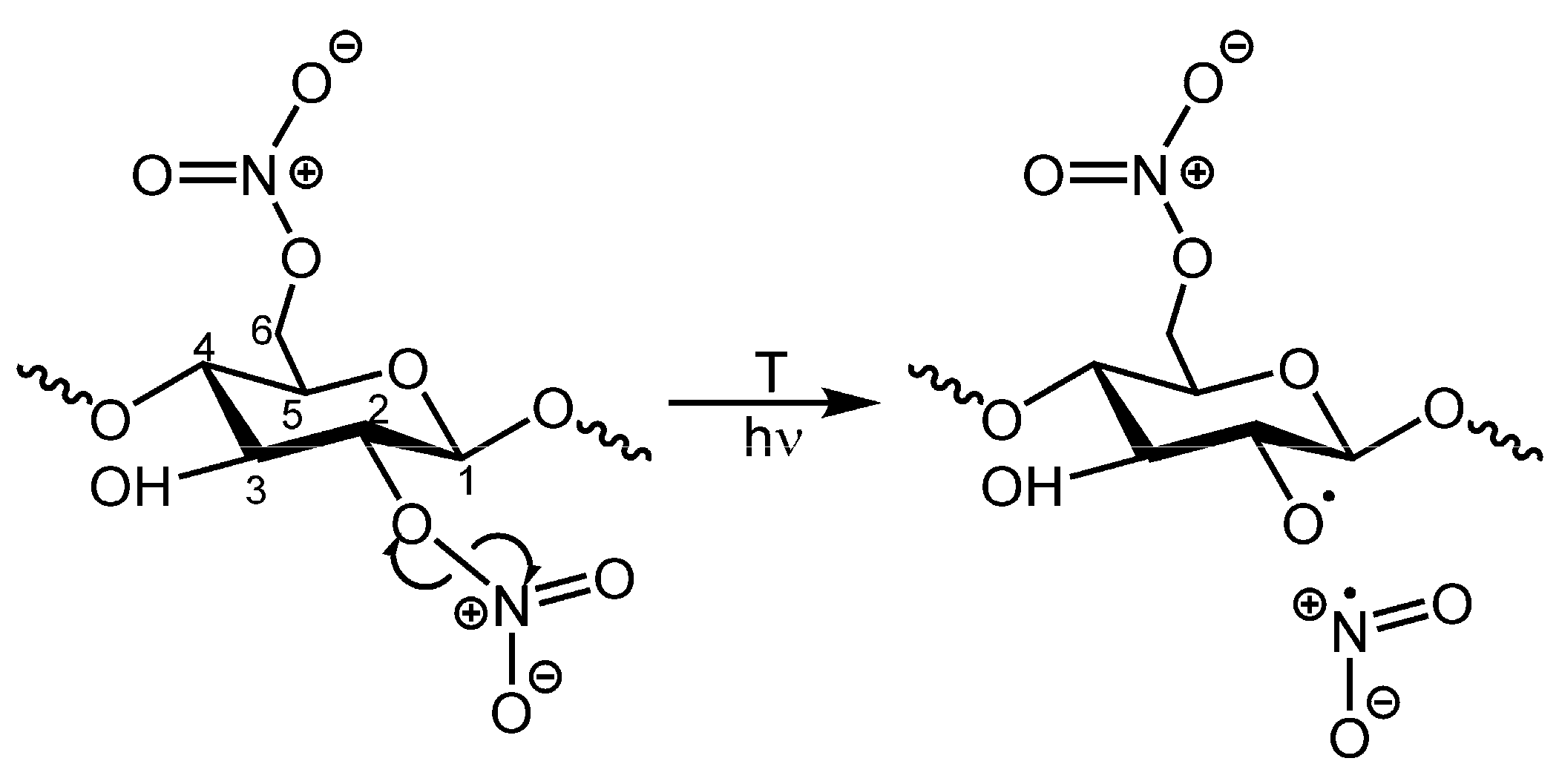
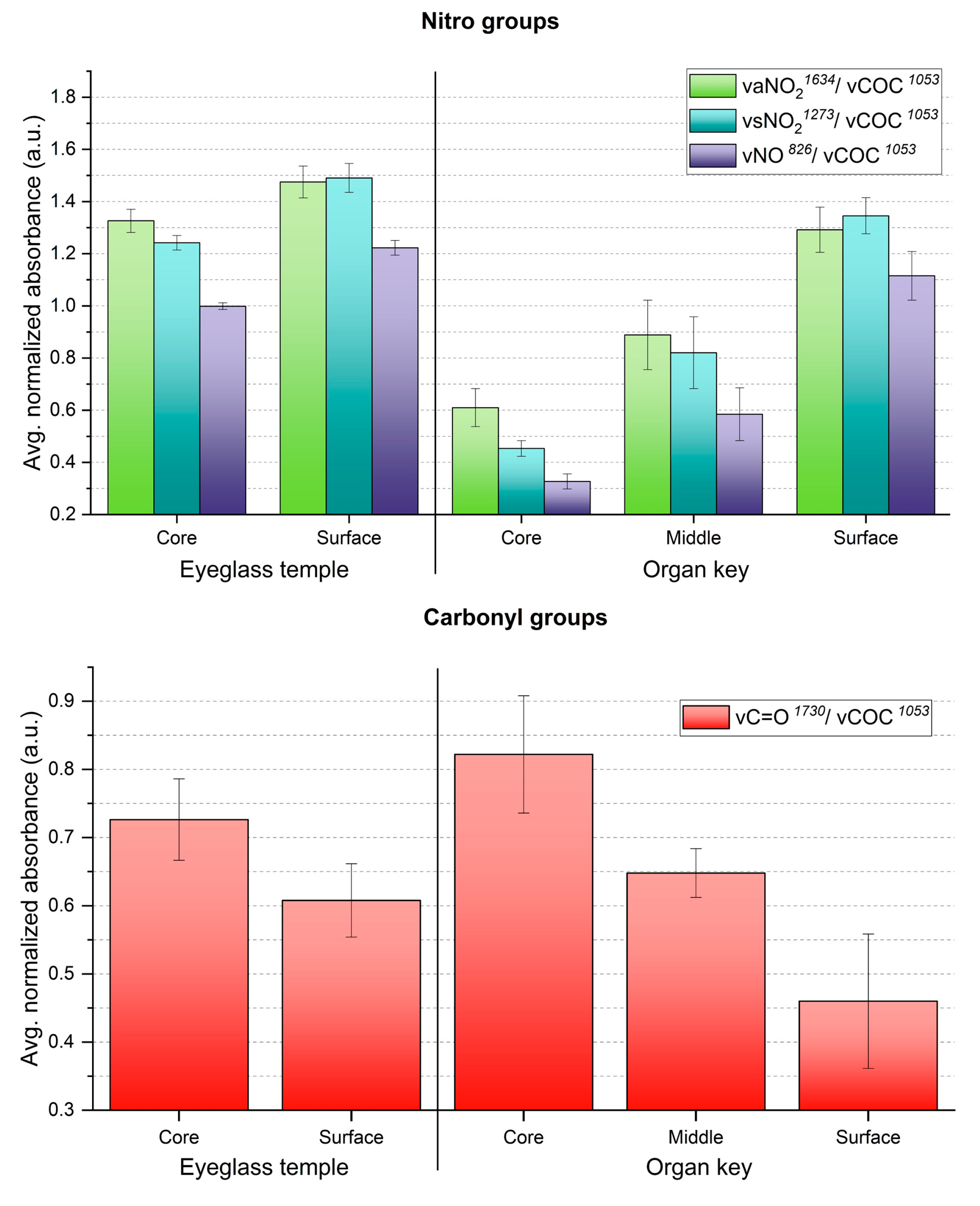
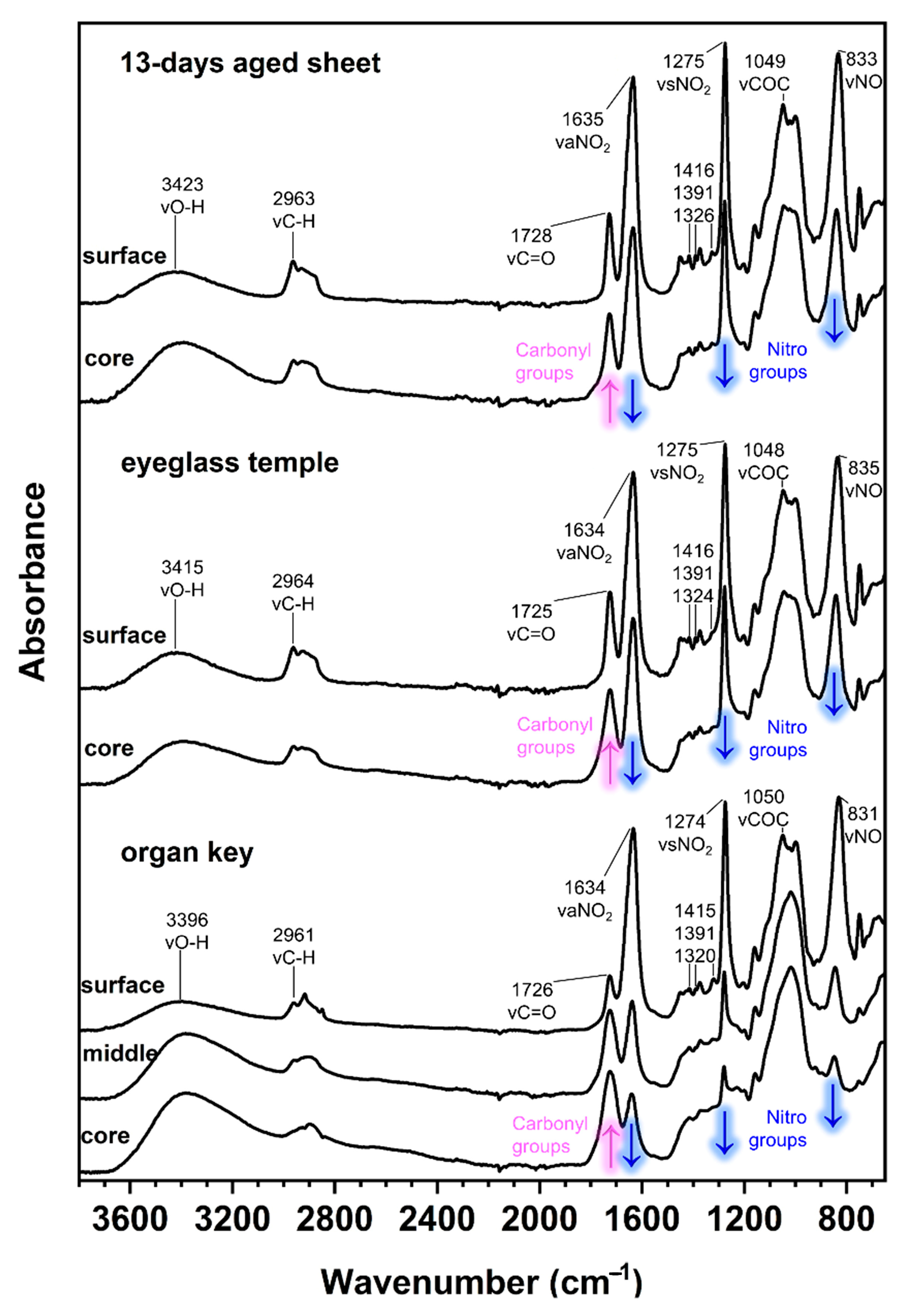
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brill, T.B.; Gongwer, P.E. Thermal Decomposition of Energetic Materials 69. Analysis of the Kinetics of Nitrocellulose at 50 °C–500 °C. Propellants Explos. Pyrotech. 1997, 22, 38–44. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Bellerby, J.M.; Bohn, M.A. Activation Energies for the Decomposition of Nitrate Ester Groups at the Anhydroglucopyranose Ring Positions C2, C3 and C6 of Nitrocellulose Using the Nitration of a Dye as Probe. Polym. Degrad. Stab. 2014, 102, 49–58. [Google Scholar] [CrossRef]
- Trache, D.; Tarchoun, A.F. Differentiation of Stabilized Nitrocellulose during Artificial Aging: Spectroscopy Methods Coupled with Principal Component Analysis. J. Chemom. 2019. [Google Scholar] [CrossRef]
- Bussiere, P.-O.; Gardette, J.-L.; Therias, S. Photodegradation of Celluloid Used in Museum Artifacts. Polym. Degrad. Stab. 2014, 107, 246–254. [Google Scholar] [CrossRef]
- Reilly, J.A. Celluloid Objects: Their Chemistry and Preservation. J. Am. Inst. Conserv. 1991, 30, 145–162. [Google Scholar] [CrossRef]
- Catelli, E.; Sciutto, G.; Prati, S.; Chavez Lozano, M.V.; Gatti, L.; Lugli, F.; Silvestrini, S.; Benazzi, S.; Genorini, E.; Mazzeo, R. A New Miniaturised Short-Wave Infrared (SWIR) Spectrometer for on-Site Cultural Heritage Investigations. Talanta 2020, 218, 121112. [Google Scholar] [CrossRef] [PubMed]
- Jutier, J.-J.; Harrison, Y.; Premont, S.; Prud’homme, R.E. A Nonisothermal Fourier Transform Infrared Degradation Study of Nitrocelluloses Derived from Wood and Cotton. J. Appl. Polym. Sci. 1987, 33, 1359–1375. [Google Scholar] [CrossRef]
- Wolfrom, M.L.; Frazer, J.H.; Kuhn, L.P.; Dickey, E.E.; Olin, S.M.; Hoffman, D.O.; Bower, R.S.; Chaney, A.; Carpenter, E.; McWain, P. The Controlled Thermal Decomposition of Cellulose Nitrate. I1,2. J. Am. Chem. Soc. 1955, 77, 6573–6580. [Google Scholar] [CrossRef]
- Chin, A.; Ellison, D.S.; Poehlein, S.K.; Ahn, M.K. Investigation of the Decomposition Mechanism and Thermal Stability of Nitrocellulose/Nitroglycerine Based Propellants by Electron Spin Resonance. Prop. Explos. Pyrotech. 2007, 32, 117–126. [Google Scholar] [CrossRef]
- Shashoua, Y.; Bradley, S.M.; Daniels, V.D. Degradation of Cellulose Nitrate Adhesive. Stud. Conserv. 1992, 37, 113–119. [Google Scholar] [CrossRef]
- Neves, A.; Ramos, A.M.; Callapez, M.E.; Friedel, R.; Réfrégiers, M.; Thoury, M.; Melo, M.J. Novel Markers to Early Detect Degradation on Cellulose Nitrate-Based Heritage at the Submicrometer Level Using Synchrotron UV–VIS Multispectral Luminescence. Sci. Rep. 2021, 11, 20208. [Google Scholar] [CrossRef] [PubMed]
- Neves, A.; Angelin, E.M.; Roldão, É.; Melo, M.J. New Insights into the Degradation Mechanism of Cellulose Nitrate in Cinematographic Films by Raman Microscopy. J. Raman Spectrosc. 2018, 50, 11. [Google Scholar] [CrossRef]
- Berthumeyrie, S.; Collin, S.; Bussiere, P.-O.; Therias, S. Photooxidation of Cellulose Nitrate: New Insights into Degradation Mechanisms. J. Hazard. Mater. 2014, 272, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Hon, D.N.-S.; Gui, T.L. Photodegradation of Cellulose Nitrate. Polym. Photochem. 1986, 7, 299–310. [Google Scholar] [CrossRef]
- Edge, M.; Allen, N.S.; Hayes, M.; Riley, P.N.K.; Horie, C.V.; Luc-Gardette, J. Mechanisms of Deterioration in Cellulose Nitrate Base Archival Cinematograph Film. Eur. Polym. J. 1990, 26, 623–630. [Google Scholar] [CrossRef]
- Adelstein, P.Z.; Reilly, J.M.; Nishimura, D.W.; Erbland, C.J. Stability of Cellulose Ester Base Photographic Film: Part IV—Behavior of Nitrate Base Film. SMPTE J. 1995, 104, 359–369. [Google Scholar] [CrossRef]
- Quye, A.; Littlejohn, D.; Pethrick, R.A.; Stewart, R.A. Accelerated Ageing to Study the Degradation of Cellulose Nitrate Museum Artefacts. Polym. Degrad. Stab. 2011, 96, 1934–1939. [Google Scholar] [CrossRef]
- Nunes, S.; Ramacciotti, F.; Neves, A.; Angelin, E.M.; Ramos, A.M.; Roldão, É.; Wallaszkovits, N.; Armijo, A.A.; Melo, M.J. A Diagnostic Tool for Assessing the Conservation Condition of Cellulose Nitrate and Acetate in Heritage Collections: Quantifying the Degree of Substitution by Infrared Spectroscopy. Herit. Sci. 2020, 8, 33. [Google Scholar] [CrossRef]
- Manelis, G.B.; Nazin, G.M.; Rubtsov, Y.I.; Strunin, V.A. Thermal Decomposition and Combustion of Explosives and Propellants; Taylor & Francis: London, UK, 2003; ISBN 978-0-415-29984-8. [Google Scholar]
- Pfeil, A.; Krause, H.H.; Eisenreich, N. The Consequences of Beginning Slow Thermal Decomposition on the Molecular Weight of Nitrated Cellulose. Thermochim. Acta 1985, 85, 399–402. [Google Scholar] [CrossRef]
- Thérias, S.; Bussière, P.-O.; Gardette, M.; Gardette, J.-L.; Lattuati-Derieux, A.; Lavédrine, B.; Quôc Khôi, T.; Barabant, G.; Balcar, N.; Colombini, A. Altération des objets en celluloïd dans les collections: Étude des mécanismes de vieillissement. In Proceedings of the Actes du Colloque Sciences des Matériaux du Patrimoine Culturel; Ministère de la Culture et de la Communication: Paris, France, 2012; Volume 2, p. 6. [Google Scholar]
- Selwitz, C. Cellulose Nitrate in Conservation; Research in Conservation; Getty Conservation Institute: Marina del Rey, CA, USA, 1988; ISBN 978-0-89236-098-7. [Google Scholar]
- Shashoua, Y. Conservation of Plastics: Materials Science, Degradation and Preservation, 1st ed.; Elsevier: Oxford, UK, 2008; ISBN 978-0-7506-6495-0. [Google Scholar]
- Shrotri, A.; Kobayashi, H.; Fukuoka, A. Chapter Two—Catalytic Conversion of Structural Carbohydrates and Lignin to Chemicals. In Advances in Catalysis; Song, C., Ed.; Academic Press: Cambridge, MA, USA, 2017; Volume 60, pp. 59–123. [Google Scholar]
- Lai, A.J. Determining the Dominant Degradation Mechanisms in Nitrocellulose. Doctoral Thesis, UCL (University College London), London, UK, 2020. [Google Scholar]
- Cold Storage of Museum Objects Made of Cellulose Nitrate. Available online: https://www.deutsches-museum.de/en/forschung/forschungsinstitut/projekte-und-forschungsbereiche/projects/detail-page/kaltlagerung-von-musealen-objekten-aus-cellulosenitrat (accessed on 28 October 2022).
- Elsässer, C.; Micheluz, A.; Pamplona, M.; Kavda, S.; Montag, P. Selection of Thermal, Spectroscopic, Spectrometric, and Chromatographic Methods for Characterizing Historical Celluloid. J. Appl. Polym. Sci. 2021, 138. [Google Scholar] [CrossRef]
- Elsässer, C.; Pamplona, M.; Mayr, V.; Donner, T.; Grießbach, S. Lower Temperature, Longer Lifetime: Practice at the Deutsches Museum and Research Perspectives for Storing 3D Cellulose Nitrate Objects. Z. Kunsttechnol. Konserv. 2021, 34, 17–26. [Google Scholar]
- Coughlin, M.; Seeger, A.M. You Collected What?! The Risks and Rewards of Acquiring Cellulose Nitrate. In Plastics: Looking at the Future and Learning from the Past; Keneghan, B., Louise, E., Eds.; Archetype Publications: London, UK, 2008; pp. 119–124. [Google Scholar]
- Keßler, K. Celluloid–Ein Werkstoff Zersetzt Sich. In Kunststoff als Werkstoff Celluloid und Polyurethan-Weichschaum, Material—Eigenschaften—Erhaltung; Siegl: München, Germany, 2001; p. 139. ISBN 978-3-935643-01-6. [Google Scholar]
- Morgan, J. Conservation of Plastics: An Introduction to Their History, Manufacture, Deterioration, Identification and Care/John Morgan; Plastics Historical Society: Conservation Unit, Museums & Galleries Commission: London, UK, 1991; ISBN 978-0-948630-14-9. [Google Scholar]
- Chavez Lozano, M.V.; Sciutto, G.; Prati, S.; Mazzeo, R. Deep Eutectic Solvents: Green Solvents for the Removal of Degraded Gelatin on Cellulose Nitrate Cinematographic Films. Herit. Sci. 2022, 10, 114. [Google Scholar] [CrossRef]
- Kimura, J. Chemiluminescence Study on Thermal Decomposition of Nitrate Esters (PETN and NC). Propellants Explos. Pyrotech. 1989, 14, 89–92. [Google Scholar] [CrossRef]
- Kimura, J. Kinetic Mechanism on Thermal Degradation of a Nitrate Ester Propellant. Propellants Explos. Pyrotech. 1988, 13, 8–12. [Google Scholar] [CrossRef]
- Quye, A.; Littlejohn, D.; Pethrick, R.A.; Stewart, R.A. Investigation of Inherent Degradation in Cellulose Nitrate Museum Artefacts. Polym. Degrad. Stab. 2011, 96, 1369–1376. [Google Scholar] [CrossRef]
- Mitchell, G.; France, F.; Nordon, A.; Tang, P.; Gibson, L.T. Assessment of Historical Polymers Using Attenuated Total Reflectance-Fourier Transform Infra-Red Spectroscopy with Principal Component Analysis. Herit. Sci. 2013, 1, 28. [Google Scholar] [CrossRef]
- Moore, D.S.; McGrane, S.D. Comparative Infrared and Raman Spectroscopy of Energetic Polymers. J. Mol. Struct. 2003, 661–662, 561–566. [Google Scholar] [CrossRef]
- Pereira, A.; Candeias, A.; Cardoso, A.; Rodrigues, D.; Vandenabeele, P.; Caldeira, A.T. Non-Invasive Methodology for the Identification of Plastic Pieces in Museum Environment—A Novel Approach. Microchem. J. 2016, 124, 846–855. [Google Scholar] [CrossRef]
- Izzo, F.C.; Carrieri, A.; Bartolozzi, G.; van Keulen, H.; Lorenzon, I.; Balliana, E.; Cucci, C.; Grazzi, F.; Picollo, M. Elucidating the Composition and the State of Conservation of Nitrocellulose-Based Animation Cells by Means of Non-Invasive and Micro-Destructive Techniques. J. Cult. Herit. 2018, 35, 254–262. [Google Scholar] [CrossRef]
- Abbate, S.; Burgi, L.F.; Gangemi, F.; Gangemi, R.; Lebon, F.; Longhi, G.; Pultz, V.M.; Lightner, D.A. Comparative Analysis of IR and Vibrational Circular Dichroism Spectra for a Series of Camphor-Related Molecules. J. Phys. Chem. A 2009, 113, 11390–11405. [Google Scholar] [CrossRef]
- Wypych, G. Handbook of Polymers; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 978-1-927885-11-6. [Google Scholar]
- Yang, Y. Cellulose Nitrate. In Polymer Data Handbook; Mark, J.E., Ed.; Oxford University Press: New York, NY, USA, 1999; ISBN 978-0-19-510789-0. [Google Scholar]
- Hamrang, A. Degradation and Stabilisation of Cellulose Based Plastics & Artifacts. Ph.D. Thesis, Manchester Metropolitan University, Manchester, UK, 1994. [Google Scholar]
- Touris, A.; Turcios, A.; Mintz, E.; Pulugurtha, S.R.; Thor, P.; Jolly, M.; Jalgaonkar, U. Effect of Molecular Weight and Hydration on the Tensile Properties of Polyamide 12. Results Mater. 2020, 8, 100149. [Google Scholar] [CrossRef]
- Gardner, R.J.; Martin, J.R. Humid Aging of Plastics: Effect of Molecular Weight on Mechanical Properties and Fracture Morphology of Polycarbonate. J. Appl. Polym. Sci. 1979, 24, 1269–1280. [Google Scholar] [CrossRef]
- Van Oosten, T.B. Properties of Plastics: A Guide for Conservators; Getty Publications: Los Angeles, CA, USA, 2022; ISBN 978-1-60606-693-5. [Google Scholar]
- Steward, R.A. Analytical Studies of the Degradation of Cellulose Nitrate Artefacts. Ph.D. Thesis, University of Strathclyde, Glasgow, UK, 1997. [Google Scholar]
- Steward, R.; Litteljohn, D.; Pethrick, R.A.; Tennen, N.H.; Quye, A. The use of accelerated ageing tests for studying the degradation of cellulose nitrate. In Proceedings of the ICOM-CC 11th Triennial Meeting Preprints, Edinburgh, UK, 1–6 September 1996; James & James Ltd.: Northampton, UK, 1996; pp. 967–970. [Google Scholar]
- Warren, R.C. Transitions and relaxations in plasticised nitrocellulose. Polymer 1988, 29, 919–923. [Google Scholar] [CrossRef]
- Schilling, M.; Bouchard, M.; Khanjian, H.; Learner, T.; Phenix, A.; Rivenc, R. Application of Chemical and Thermal Analysis Methods for Studying Cellulose Ester Plastics. Acc. Chem. Res. 2010, 43, 888–896. [Google Scholar] [CrossRef] [PubMed]
- Bauer, C.L.; Deacon, P.R.; Garman, R.N.; Emsley, A.M.; Jones, J.R. Low Temperature Non-Isothermal Aging of Nitrocellulose. Propellants Explos. Pyrotech. 2005, 30, 231–236. [Google Scholar] [CrossRef]
- Kavda, S.; Micheluz, A.; Elsässer, C.; Pamplona, M. Development of a Gel Permeation Chromatography Method for Analysing Cellulose Nitrate in Museums. J. Sep. Sci. 2021, 44, 1795–1804. [Google Scholar] [CrossRef]
- Elsässer, C.; Pamplona, M.; Trischler, H. Kaltlagerung von Musealen, Anthropogenen Objekten auf der Basis von Cellulosenitrat—Methoden zur Untersuchung des Chemischen Abbaus; DBU Reports; Objekt- und Restaurierungsforschung, Deutsches Museum von Meisterwerken der Naturwissenschaft und Technik: Munich, Germany, 2022; p. 35. [Google Scholar]







| Wavenumber (cm−1) | Assignment | ||
|---|---|---|---|
| CN Membrane (Reference) | Unaged CN Coupon | Racemic Camphor (Reference) | |
| 3657 | |||
| 3448 | 3468 | νO-H (bound) | |
| 2966 | 2963 | 2958 | νC-H [35,36] |
| 2923 | 2927 | 2931 | νsC-H [35,37] |
| 2908 | |||
| 2887 | 2872 | ||
| 1728 | 1738 | νC=O [37], from camphor [35,38,39] or CN degradation products [11,13,15,36] | |
| 1637 | 1635 | νaO-NO2 [35,39] | |
| 1454 | 1452 | 1447 | δCH2 in CN [37] δaCH3 and δsCH2 in camphor [40] |
| 1427 | 1428 | δCH2 [35] | |
| 1417 | 1416 | δsCH2 at position C3 in camphor [40] | |
| 1391 | 1390 | δsCH3 in camphor [40] | |
| 1375 | 1374 | 1373 | δC-H in CN [35] δsCH3 and νC-C in camphor [40] |
| 1325 | 1324 | ωCH2, νC-C, and δsCH3 at C1 in camphor [40] | |
| 1278 | 1276 | 1277 | νsNO2 in CN [35,37,38,39] ωCH2, νCC, with minimum ρCH2 and ring deformation in camphor [40] |
| 1160 | 1159 | 1167 | νaO-C-C [38] νCC, ρCH2 and τCH2, in camphor [40] |
| 1115 | 1111 | νCO in ring [35] | |
| 1061 | 1051 | 1045 | νaO-C-C attached to the NO2 group [38], νCOC of the cellulose ring in CN [18] νCC, τCH2, ρCH2 and in-plane δCO in camphor [40] |
| 1022 | 1021 | 1022 | νCO [37] |
| 999 | 999 | νC-O [37] | |
| 947 | 945 | 951 | δsCH [37] |
| 918 | 918 | 914 | δsCH [37] |
| 827 | 828 | 827 | ν-NO in CN [37,38,39] |
| 750 | 750 | 751 | δO-NO2 in CN [35,37,39] |
| 694 | 698 | δO-NO2 [35,37,39] | |
| 681 | 676 | Pyranose [37] | |
| 554 | 553 | ||
| 541 | |||
| 521 | 521 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chavez Lozano, M.V.; Elsässer, C.; Angelin, E.M.; Pamplona, M. Shedding Light on Degradation Gradients in Celluloid: An ATR-FTIR Study of Artificially and Naturally Aged Specimens. Polymers 2023, 15, 522. https://doi.org/10.3390/polym15030522
Chavez Lozano MV, Elsässer C, Angelin EM, Pamplona M. Shedding Light on Degradation Gradients in Celluloid: An ATR-FTIR Study of Artificially and Naturally Aged Specimens. Polymers. 2023; 15(3):522. https://doi.org/10.3390/polym15030522
Chicago/Turabian StyleChavez Lozano, Marco Valente, Christina Elsässer, Eva Mariasole Angelin, and Marisa Pamplona. 2023. "Shedding Light on Degradation Gradients in Celluloid: An ATR-FTIR Study of Artificially and Naturally Aged Specimens" Polymers 15, no. 3: 522. https://doi.org/10.3390/polym15030522
APA StyleChavez Lozano, M. V., Elsässer, C., Angelin, E. M., & Pamplona, M. (2023). Shedding Light on Degradation Gradients in Celluloid: An ATR-FTIR Study of Artificially and Naturally Aged Specimens. Polymers, 15(3), 522. https://doi.org/10.3390/polym15030522