Self-Assembled CNF/rGO/Tannin Composite: Study of the Physicochemical and Wound Healing Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Production of Cellulose Nanofibrils (CNF)
2.2. Mechanical-Enzymatic Pretreatment
2.3. Synthesis of Graphene Oxide (GO)
2.4. Pinus Radiata Bark Extract Production
2.5. Synthesis of Reduced Graphene Oxide/Nanocellulose (rGO/CNF) and Reduced Graphene Oxide/Nanocellulose/Tannin (rGO/CNF/TA) Composites
2.6. Physicochemical Characterization of rGO/CNF and rGO/CNF/TA Composites
2.7. Conductivity Measurements
2.8. Swelling Behavior of rGO/CNF and rGO/CNF/TA Composites
2.9. Mechanical Properties
2.10. Cytotoxicity Assay
2.11. In Vitro Wound Healing Assay (Scratch Test)
2.12. Statistical Analysis
3. Results and Discussion
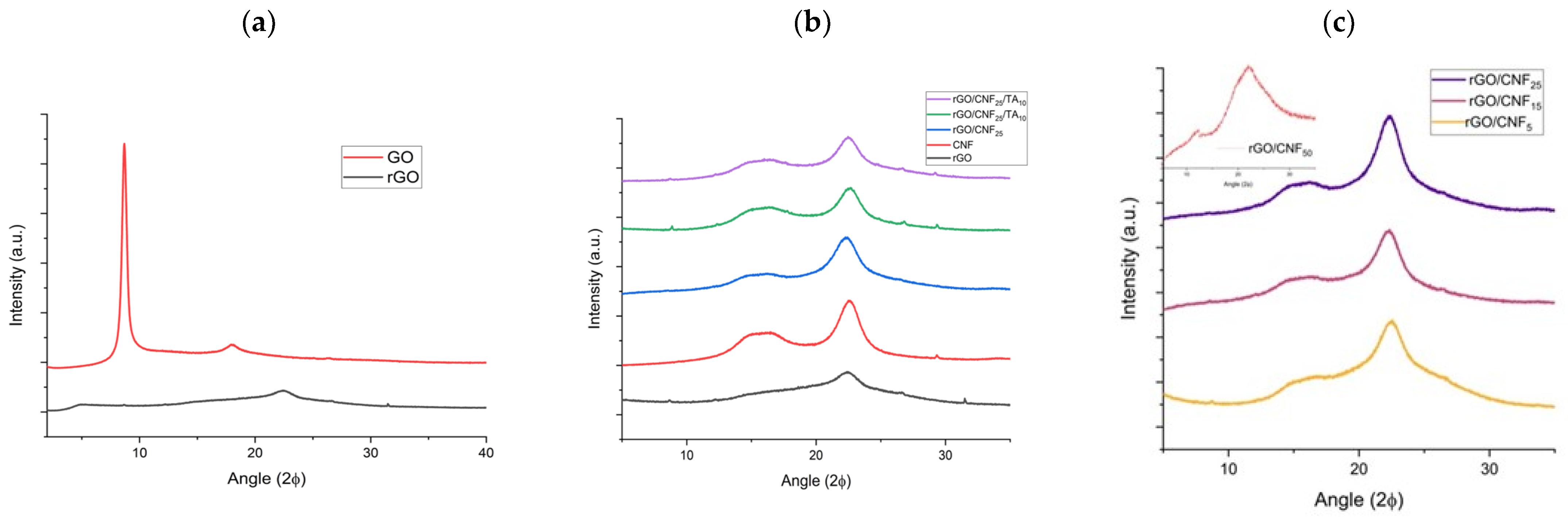
3.1. Morphological Characterization of rGO/CNF and rGO/CNF/TA Composites
3.2. Spectroscopical Characterization of rGO/CNF and rGO/CNF/TA Composites
3.3. Thermal Stability of the Composite Materials
3.4. Conductivity, Surface Charge and Wettability of the Composite Materials
3.5. Swelling Behavior of Composite Materials
3.6. Tension and Deformation of Composite Materials
3.7. Cytotoxicity Assays
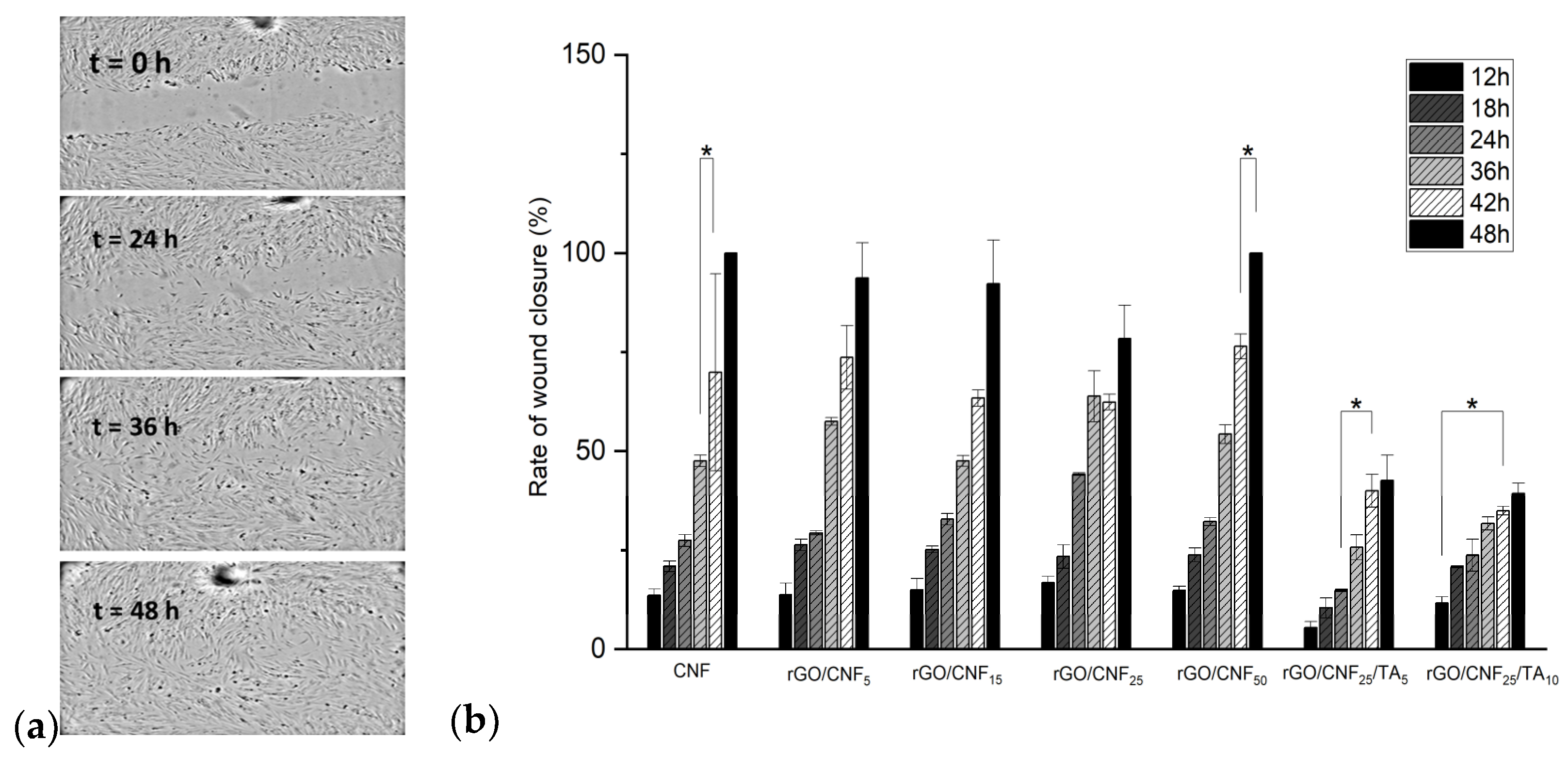
3.8. In Vitro Wound Healing Assay (Scratch Test)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Payne, P.A. Measurement of properties and function of skin. Clin. Phys. Physiol. Meas. 1991, 12, 105–129. [Google Scholar] [CrossRef] [PubMed]
- Chua, A.W.C.; Khoo, Y.C.; Tan, B.K.; Tan, K.C.; Foo, C.L.; Chong, S.J. Skin tissue engineering advances in severe burns: Review and therapeutic applications. Burn. Trauma 2016, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.P.; Zhang, Y.Z.; Lim, C.T. Tissue scaffolds for skin wound healing and dermal reconstruction. WIREs Nanomed. Nanobiotechnol. 2010, 2, 510–525. [Google Scholar] [CrossRef]
- Balasubramani, M.; Kumar, T.; Babu, M. Skin substitutes: A review. Burns 2001, 27, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.; Catanzano, O. Advanced Therapeutic Dressings for Effective Wound Healing—A Review. J. Pharm. Sci. 2015, 104, 3653–3680. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.; Soni, S.; Mittal, G.; Bhatnagar, A. Role of Polymeric Biomaterials as Wound Healing Agents. Int. J. Low. Extrem. Wounds 2014, 13, 180–190. [Google Scholar] [CrossRef]
- Farber, P.L.; Isoldi, F.C.; Ferreira, L.M. Electric Factors in Wound Healing. Adv. Wound Care 2021, 10, 461–476. [Google Scholar] [CrossRef]
- Zhao, M.; Song, B.; Pu, J.; Wada, T.; Reid, B.; Tai, G.; Wang, F.; Guo, A.; Walczysko, P.; Gu, Y.; et al. Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-γ and PTEN. Nature 2006, 442, 457–460. [Google Scholar] [CrossRef]
- Fan, Z.; Liu, B.; Wang, J.; Zhang, S.; Lin, Q.; Gong, P.; Ma, L.; Yang, S. A Novel Wound Dressing Based on Ag/Graphene Polymer Hydrogel: Effectively Kill Bacteria and Accelerate Wound Healing. Adv. Funct. Mater. 2014, 24, 3933–3943. [Google Scholar] [CrossRef]
- Tang, P.; Han, L.; Li, P.; Jia, Z.; Wang, K.; Zhang, H.; Tan, H.; Guo, T.; Lu, X. Mussel-Inspired Electroactive and Antioxidative Scaffolds with Incorporation of Polydopamine-Reduced Graphene Oxide for Enhancing Skin Wound Healing. ACS Appl. Mater. Interfaces 2019, 11, 7703–7714. [Google Scholar] [CrossRef]
- Fall, A.B.; Lindström, S.B.; Sundman, O.; Ödberg, L.; Wågberg, L. Colloidal Stability of Aqueous Nanofibrillated Cellulose Dispersions. Langmuir 2011, 27, 11332–11338. [Google Scholar] [CrossRef]
- Brakat, A.; Zhu, H. Nanocellulose-Graphene Derivative Hybrids: Advanced Structure-Based Functionality from Top-down Synthesis to Bottom-up Assembly. ACS Appl. Bio Mater. 2021, 4, 7366–7401. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, F.; Chen, X.; Wang, X.-W.; Zhang, W.-B.; Peng, J.; Li, J.; Zhai, M. Stretchable, Conductive, and Self-Healing Hydrogel with Super Metal Adhesion. Chem. Mater. 2018, 30, 4289–4297. [Google Scholar] [CrossRef]
- Shin, S.R.; Li, Y.-C.; Jang, H.L.; Khoshakhlagh, P.; Akbari, M.; Nasajpour, A.; Zhang, Y.S.; Tamayol, A.; Khademhosseini, A. Graphene-based materials for tissue engineering. Adv. Drug Deliv. Rev. 2016, 105, 255–274. [Google Scholar] [CrossRef]
- Huang, N.; Zhang, S.; Yang, L.; Liu, M.; Li, H.; Zhang, Y.; Yao, S. Multifunctional Electrochemical Platforms Based on the Michael Addition/Schiff Base Reaction of Polydopamine Modified Reduced Graphene Oxide: Construction and Application. ACS Appl. Mater. Interfaces 2015, 7, 17935–17946. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Deng, Y.; Lyu, B.; Zhang, R.; Zhang, H.; Ma, H.; Lyu, Y.; Wei, S. Rapidly-Deposited Polydopamine Coating via High Temperature and Vigorous Stirring: Formation, Characterization and Biofunctional Evaluation. PLoS ONE 2014, 9, e113087. [Google Scholar] [CrossRef] [PubMed]
- Schofield, P.; Mbugua, D.; Pell, A. Analysis of condensed tannins: A review. Anim. Feed. Sci. Technol. 2001, 91, 21–40. [Google Scholar] [CrossRef]
- Maugeri, A.; Lombardo, G.E.; Cirmi, S.; Süntar, I.; Barreca, D.; Laganà, G.; Navarra, M. Pharmacology and toxicology of tannins. Arch. Toxicol. 2022, 96, 1257–1277. [Google Scholar] [CrossRef]
- Su, X.; Liu, X.; Wang, S.; Li, B.; Pan, T.; Liu, D.; Wang, F.; Diao, Y.; Li, K. Wound-healing promoting effect of total tannins from Entada phaseoloides (L.) Merr. in rats. Burns 2017, 43, 830–838. [Google Scholar] [CrossRef]
- Li, K.; Diao, Y.; Zhang, H.; Wang, S.; Zhang, Z.; Yu, B.; Huang, S.; Yang, H. Tannin extracts from immature fruits of Terminalia chebula Fructus Retz. promote cutaneous wound healing in rats. BMC Complement. Altern. Med. 2011, 11, 86. [Google Scholar] [CrossRef]
- Sileika, T.S.; Barrett, D.G.; Zhang, R.; Lau, K.H.A.; Messersmith, P.B. Colorless Multifunctional Coatings Inspired by Polyphenols Found in Tea, Chocolate, and Wine. Angew. Chem. Int. Ed. 2013, 52, 10766–10770. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, Z.; Yin, J. Facile Synthesis of Soluble Graphene via a Green Reduction of Graphene Oxide in Tea Solution and Its Biocomposites. ACS Appl. Mater. Interfaces 2011, 3, 1127–1133. [Google Scholar] [CrossRef]
- Zeng, J.; Zeng, Z.; Cheng, Z.; Wang, Y.; Wang, X.; Wang, B.; Gao, W. Cellulose nanofibrils manufactured by various methods with application as paper strength additives. Sci. Rep. 2021, 11, 1–16. [Google Scholar] [CrossRef]
- Andrade, A.; Henríquez-Gallegos, S.; Albornoz-Palma, G.; Pereira, M. Effect of the chemical and structural characteristics of pulps of Eucalyptus and Pinus on the deconstruction of the cell wall during the production of cellulose nanofibrils. Cellulose 2021, 28, 5387–5399. [Google Scholar] [CrossRef]
- Ghose, T.K. Measurement of cellulase activities. Pure Appl. Chem. 1987, 59, 257–268. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.S.; Alemany, L.B.; Lu, W.; Tour, J.M. Correction to Improved Synthesis of Graphene Oxide. ACS Nano 2018, 12, 2078. [Google Scholar] [CrossRef]
- Bocalandro, C.; Sanhueza, V.; Gómez-Caravaca, A.M.; González-Álvarez, J.; Fernández, K.; Roeckel, M.; Rodríguez-Estrada, M.T. Comparison of the composition of Pinus radiata bark extracts obtained at bench- and pilot-scales. Ind. Crop. Prod. 2012, 38, 21–26. [Google Scholar] [CrossRef]
- Wei, J.; Xie, J.; Zhang, P.; Zou, Z.; Ping, H.; Wang, W.; Xie, H.; Shen, J.Z.; Lei, L.; Fu, Z. Bioinspired 3D Printable, Self-Healable, and Stretchable Hydrogels with Multiple Conductivities for Skin-like Wearable Strain Sensors. ACS Appl. Mater. Interfaces 2021, 13, 2952–2960. [Google Scholar] [CrossRef]
- Schuhladen, K.; Raghu, S.N.V.; Liverani, L.; Neščáková, Z.; Boccaccini, A.R. Production of a novel poly(ɛ-caprolactone)-methylcellulose electrospun wound dressing by incorporating bioactive glass and Manuka honey. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 109, 180–192. [Google Scholar] [CrossRef]
- Li, M.; Wang, X.; Zhao, R.; Miao, Y.; Liu, Z. A novel graphene-based micro/nano architecture with high strength and conductivity inspired by multiple creatures. Sci. Rep. 2021, 11, 1–15. [Google Scholar] [CrossRef]
- Hsu, H.H.; Khosrozadeh, A.; Li, B.; Luo, G.; Xing, M.M.; Zhong, W. An Eco-Friendly, Nanocellulose/RGO/in Situ Formed Polyaniline for Flexible and Free-Standing Supercapacitors. ACS Sustain. Chem. Eng. 2019, 7, 4766–4776. [Google Scholar] [CrossRef]
- Faniyi, I.O.; Fasakin, O.; Olofinjana, B.; Adekunle, A.S.; Oluwasusi, T.V.; Eleruja, M.A.; Ajayi, E.O.B. The comparative analyses of reduced graphene oxide (RGO) prepared via green, mild and chemical approaches. SN Appl. Sci. 2019, 1. [Google Scholar] [CrossRef]
- Wang, J.; Ran, R.; Sunarso, J.; Yin, C.; Zou, H.; Feng, Y.; Li, X.; Zheng, X.; Yao, J. Nanocellulose-assisted low-temperature synthesis and supercapacitor performance of reduced graphene oxide aerogels. J. Power Sources 2017, 347, 259–269. [Google Scholar] [CrossRef]
- Song, J.; Wang, X.; Chang, C.-T. Preparation and Characterization of Graphene Oxide. J. Nanomater. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Soto, R. Evidence of chemical reactions between di- and poly-glycidyl ether resins and tannins isolated from Pinus radiata D. Don bark. Bioresour. Technol. 2005, 96, 95–101. [Google Scholar] [CrossRef]
- Zhu, Y.-H.; Zhang, Q.; Sun, G.-T.; Chen, C.-Z.; Zhu, M.-Q.; Huang, X.-H. The synthesis of tannin-based graphene aerogel by hydrothermal treatment for removal of heavy metal ions. Ind. Crop. Prod. 2021, 176, 114304. [Google Scholar] [CrossRef]
- Gu, R.; Xu, W.Z.; Charpentier, P.A. Synthesis of polydopamine-coated graphene-polymer nanocomposites via RAFT polymerization. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 3941–3949. [Google Scholar] [CrossRef]
- Liao, J.; He, S.; Guo, S.; Luan, P.; Mo, L.; Li, J. Antibacterial Performance of a Mussel-Inspired Polydopamine-Treated Ag/Graphene Nanocomposite Material. Materials 2019, 12, 3360. [Google Scholar] [CrossRef]
- Mohaiyiddin, M.S.; Lin, O.H.; Owi, W.T.; Chan, C.H.; Chia, C.H.; Zakaria, S.; Villagracia, A.R.; Akil, H.M. Characterization of nanocellulose recovery from Elaeis guineensis frond for sustainable development. Clean Technol. Environ. Policy 2016, 18, 2503–2512. [Google Scholar] [CrossRef]
- Liu, X.; Gu, W.; Wang, K.; Zhang, W.; Xia, C.; Shi, S.Q.; Li, J. Thiol-branched graphene oxide and polydopamine-induced nanofibrillated cellulose to strengthen protein-based nanocomposite films. Cellulose 2019, 26, 7223–7236. [Google Scholar] [CrossRef]
- Li, M.; Miao, Y.; Zhai, X.; Yin, Y.; Zhang, Y.; Jian, Z.; Wang, X.; Sun, L.; Liu, Z. Preparation of and research on bioinspired graphene oxide/nanocellulose/polydopamine ternary artificial nacre. Mater. Des. 2019, 181. [Google Scholar] [CrossRef]
- Gan, P.G.; Sam, S.T.; Bin Abdullah, M.F.; Omar, M.F. Thermal properties of nanocellulose-reinforced composites: A review. J. Appl. Polym. Sci. 2019, 137. [Google Scholar] [CrossRef]
- Zhao, Y.; Meng, Y.; Zhu, F.; Su, J.; Han, J. Mechanical Reinforcement in Nylon 6 Nanocomposite Fiber Incorporated with Dopamine Reduced Graphene Oxide. Materials 2022, 15, 5095. [Google Scholar] [CrossRef]
- Dang, L.N.; Seppälä, J. Electrically conductive nanocellulose/graphene composites exhibiting improved mechanical properties in high-moisture condition. Cellulose 2015, 22, 1799–1812. [Google Scholar] [CrossRef]
- Ding, Z.; Tang, Y.; Zhu, P. Reduced graphene oxide/cellulose nanocrystal composite films with high specific capacitance and tensile strength. Int. J. Biol. Macromol. 2022, 200, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Anikushin, B.M.; Lagutin, P.G.; Kanbetova, A.M.; Novikov, A.A.; Vinokurov, V.A. Zeta Potential of Nanosized Particles of Cellulose as a Function of pH. Chem. Technol. Fuels Oils 2022, 57, 913–916. [Google Scholar] [CrossRef]
- Şimşek, B.; Ultav, G.; Korucu, H.; Yartaşı, A. Improvement of the Graphene Oxide Dispersion Properties with the Use of TOPSIS Based Taguchi Application. Period. Polytech. Chem. Eng. 2018, 62, 323–335. [Google Scholar] [CrossRef]
- Miao, Y.; Wang, X.; Liu, Y.; Liu, Z.; Chen, W. Preparation of Graphene Oxide/Cellulose Composites with Microcrystalline Cellulose Acid Hydrolysis Using the Waste Acids Generated by the Hummers Method of Graphene Oxide Synthesis. Polymers 2021, 13, 4453. [Google Scholar] [CrossRef]
- Guo, Q.; Shi, Z.; Xu, H.; Ma, X.; Yin, J.; Tian, M. Fabrication of Super Extensible and Highly Tough Graphene Composite Hydrogels by Thermal Treatment Strategy for the Mixture of Tannin and Graphene Oxide. Macromol. Chem. Phys. 2017, 218. [Google Scholar] [CrossRef]
- Ejima, H.; Richardson, J.J.; Liang, K.; Best, J.P.; van Koeverden, M.P.; Such, G.K.; Cui, J.; Caruso, F. One-Step Assembly of Coordination Complexes for Versatile Film and Particle Engineering. Science 2013, 341, 154–157. [Google Scholar] [CrossRef]
- Missio, A.L.; Mattos, B.D.; de F. Ferreira, D.; Magalhães, W.L.E.; Bertuol, D.A.; Gatto, D.A.; Petutschnigg, A.; Tondi, G. Nanocellulose-tannin films: From trees to sustainable active packaging. J. Clean. Prod. 2018, 184, 143–151. [Google Scholar] [CrossRef]
- He, F.; Peng, L.; Long, W.; Jiang, X.; Zhang, W. Structure-Dependent Eco-Toxicity of Vegetable Tannin. Processes 2022, 10, 816. [Google Scholar] [CrossRef]
- Schmidt, A.J.; Krieg, J.-C.; Hemmeter, U.M.; Kircher, T.; Schulz, E.; Clement, H.-W.; Heiser, P. Impact of plant extracts tested in attention-deficit/hyperactivity disorder treatment on cell survival and energy metabolism in human neuroblastoma SH-SY5Y cells. Phytother. Res. 2010, 24, 1549–1553. [Google Scholar] [CrossRef]
- Schmidt, C.A.; Murillo, R.; Bruhn, T.; Bringmann, G.; Goettert, M.; Heinzmann, B.; Brecht, V.; Laufer, S.A.; Merfort, I. Catechin Derivatives from Parapiptadenia rigida with in Vitro Wound-Healing Properties. J. Nat. Prod. 2010, 73, 2035–2041. [Google Scholar] [CrossRef] [PubMed]







| Sample | C–C% | C–OH% | C–O–C% | O–C=O% | C–N% |
|---|---|---|---|---|---|
| TA | 47 | 33 | 16 | 3 | N.D. |
| CNF | 46 | 32 | 17 | 3 | 19 |
| GO | 30 | 23 | 22 | 23 | N.D. |
| rGO/CNF25 | 38 | 2 | 37 | 21 | 13 |
| GO/CNF25/TA10 | 46 | 28 | 25 | 25 | 20 |
| Sample | Ti (°C) 1 | Tmax (°C) 2 | Tb (°C) 3 | M600 (%) 4 |
|---|---|---|---|---|
| CNF | 168.27 | 226.79 | 269.70 | 13.84 |
| rGO | 314.04 | 345.87 | 381.39 | 1.33 |
| rGO/CNF25 | 171.61 | 223.93 | 267.44 | 32.10 |
| rGO/CNF25/TA10 | 153.13 | 202.24 | 248.01 | 47.62 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández, K.; Llanquileo, A.; Bustos, M.; Aedo, V.; Ruiz, I.; Carrasco, S.; Tapia, M.; Pereira, M.; Meléndrez, M.F.; Aguayo, C.; et al. Self-Assembled CNF/rGO/Tannin Composite: Study of the Physicochemical and Wound Healing Properties. Polymers 2023, 15, 2752. https://doi.org/10.3390/polym15122752
Fernández K, Llanquileo A, Bustos M, Aedo V, Ruiz I, Carrasco S, Tapia M, Pereira M, Meléndrez MF, Aguayo C, et al. Self-Assembled CNF/rGO/Tannin Composite: Study of the Physicochemical and Wound Healing Properties. Polymers. 2023; 15(12):2752. https://doi.org/10.3390/polym15122752
Chicago/Turabian StyleFernández, Katherina, Aylen Llanquileo, Monserrat Bustos, Valentina Aedo, Isleidy Ruiz, Sebastián Carrasco, Mauricio Tapia, Miguel Pereira, Manuel F. Meléndrez, Claudio Aguayo, and et al. 2023. "Self-Assembled CNF/rGO/Tannin Composite: Study of the Physicochemical and Wound Healing Properties" Polymers 15, no. 12: 2752. https://doi.org/10.3390/polym15122752
APA StyleFernández, K., Llanquileo, A., Bustos, M., Aedo, V., Ruiz, I., Carrasco, S., Tapia, M., Pereira, M., Meléndrez, M. F., Aguayo, C., & Atanase, L. I. (2023). Self-Assembled CNF/rGO/Tannin Composite: Study of the Physicochemical and Wound Healing Properties. Polymers, 15(12), 2752. https://doi.org/10.3390/polym15122752








