Electrospun Polymer Nanofibers: Processing, Properties, and Applications
Abstract
1. Introduction
2. The Electrospinning Process
2.1. A Brief History
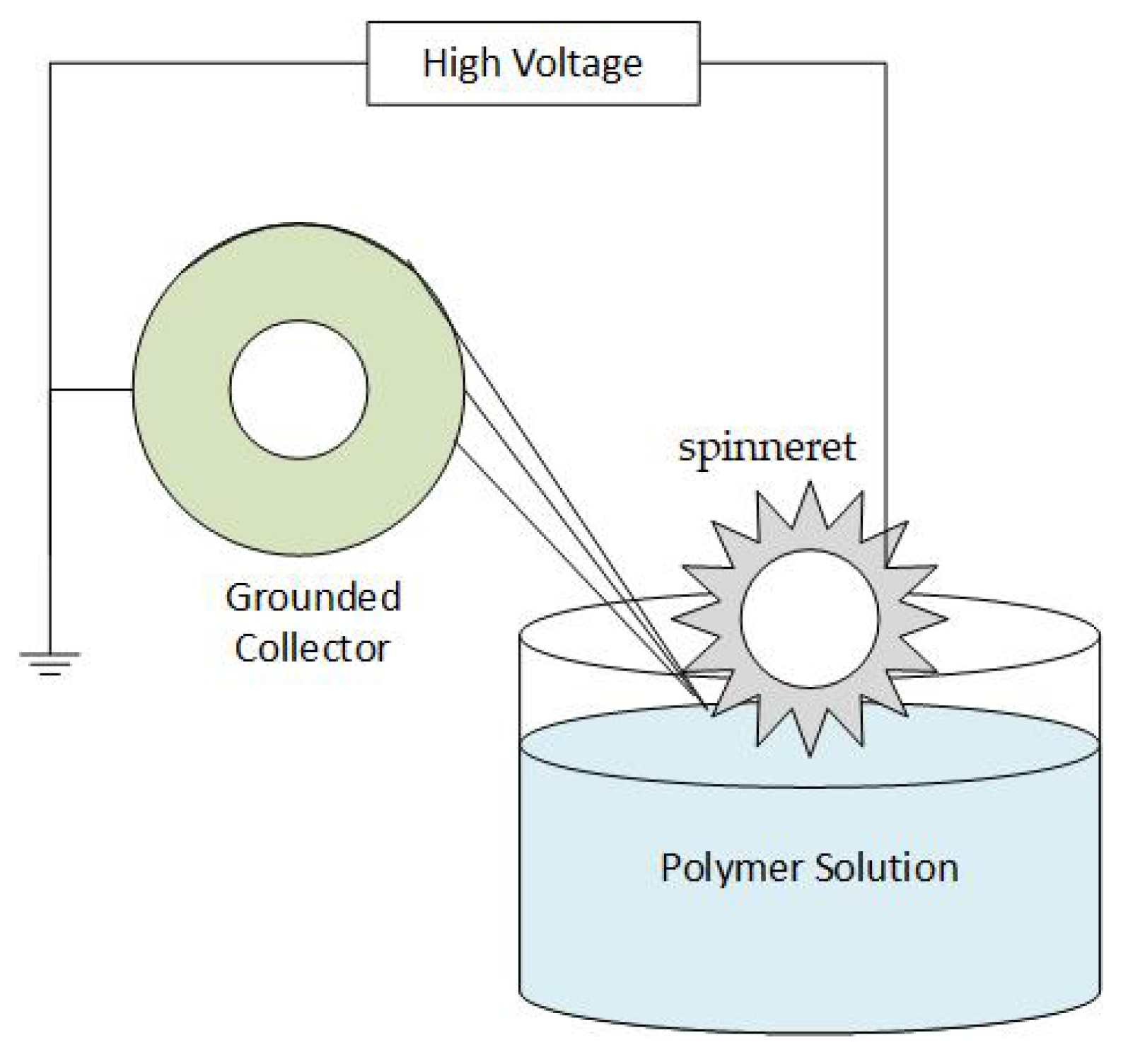
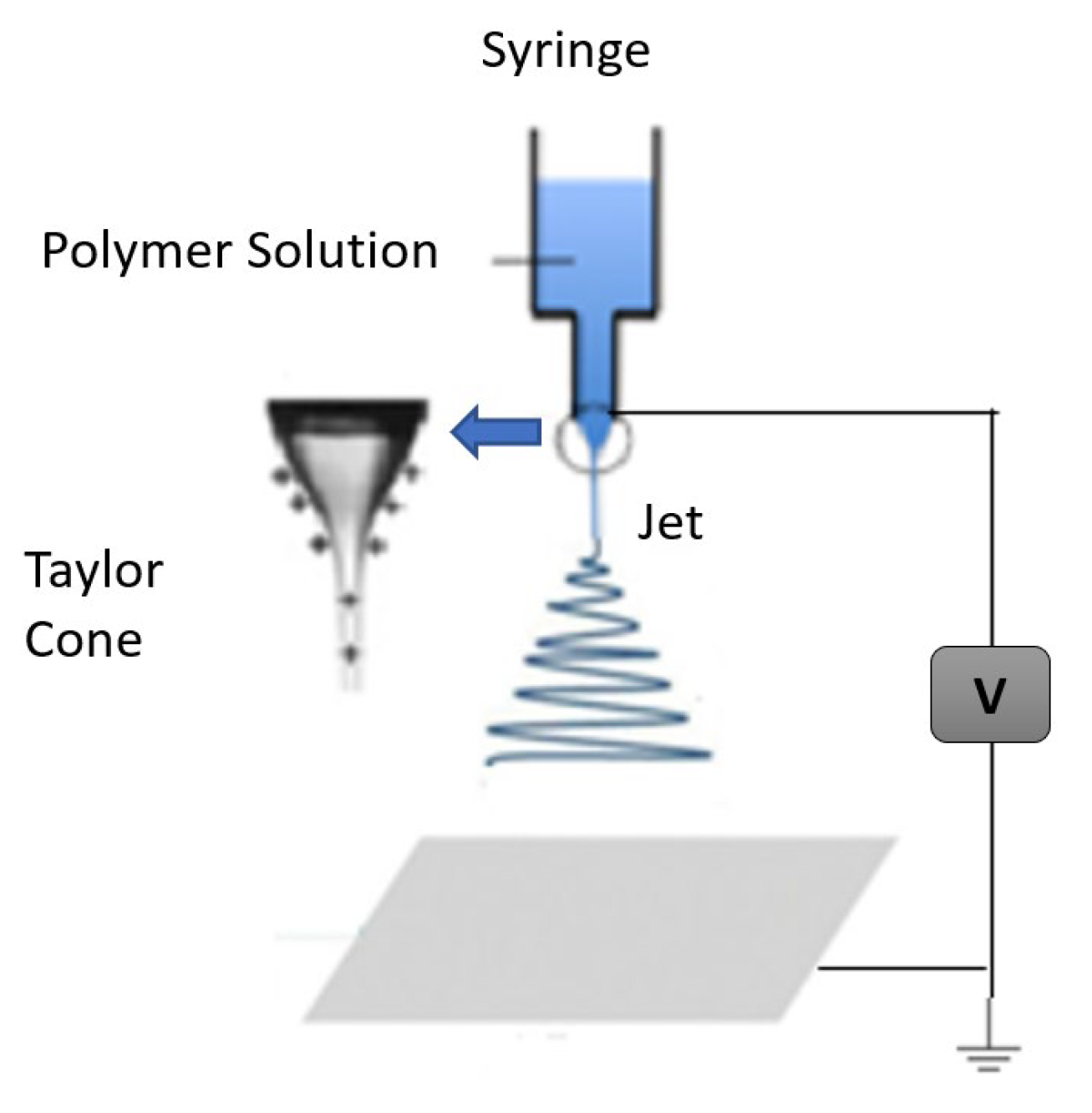
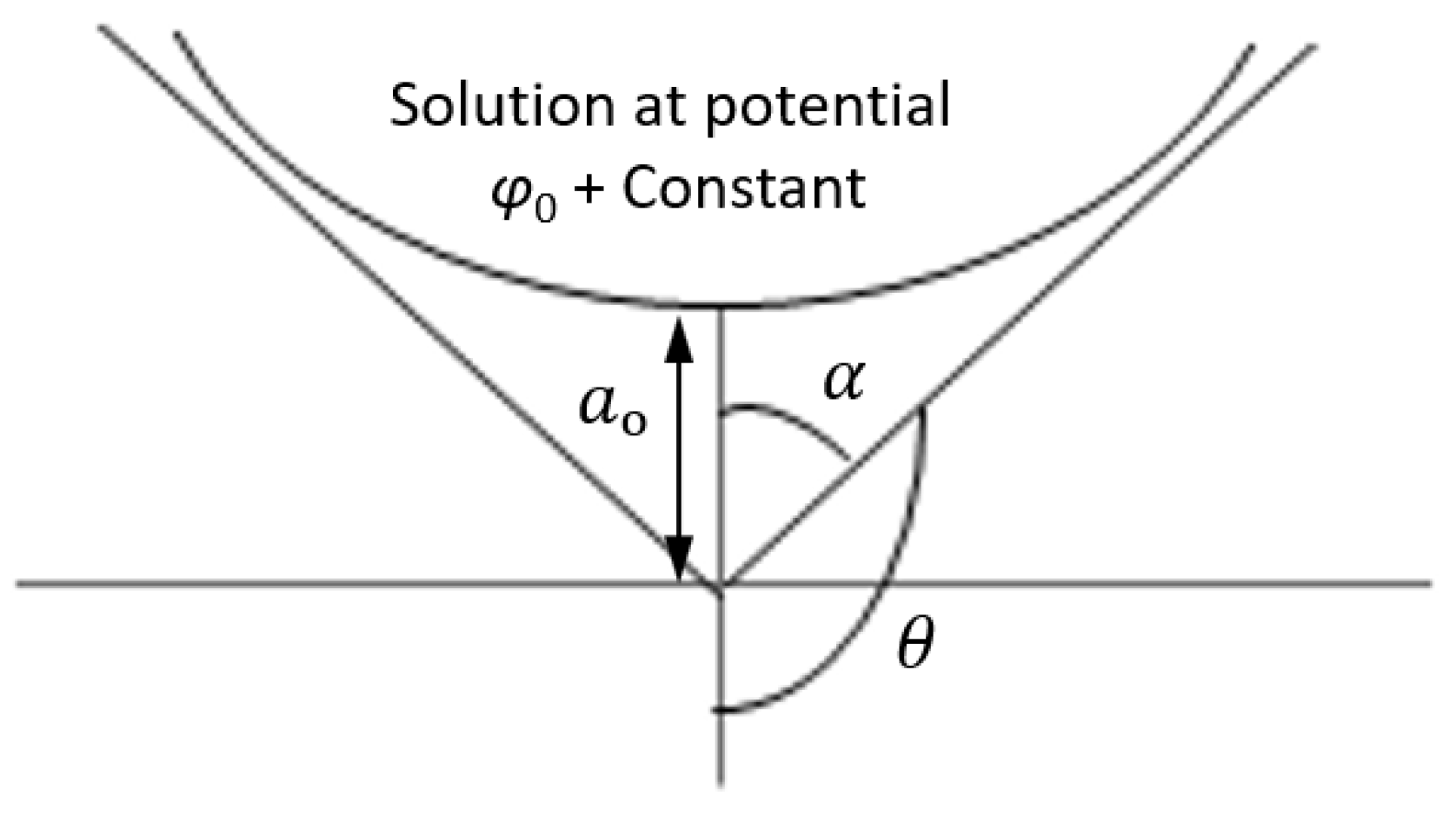
2.2. The Electrospinning Process
2.3. Electrospinning Techniques
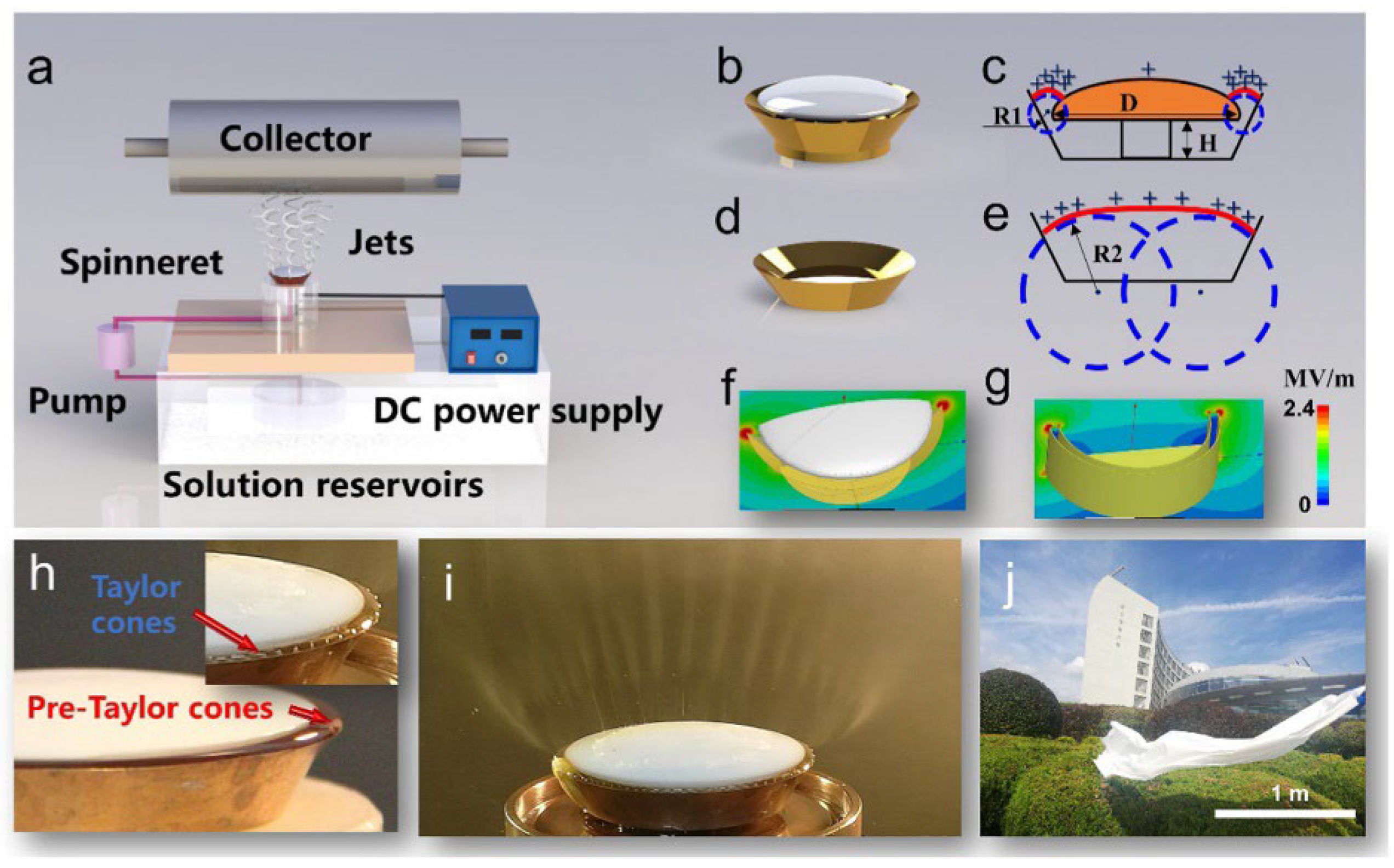
2.4. Scalability of Electrospinning
2.5. Portable Electrospinning
3. Polymers Used in Electrospinning
3.1. Natural and Synthetic Polymers
3.2. Composite Polymers/Copolymers
4. Electrospinning Process Parameters
- Parameters related to the solution, such as polymer molecular weight, polymer concentration, surface tension, conductivity, solvent volatility, and viscosity.
- Parameters related to processing such field strength, flow rate, tip-to-collector separation, applied voltage, placement and design of the needle tip, composition and geometry of the collector, and take-up velocity of the collector.
- Properties related to environmental factors including temperature, humidity, and pressure.
4.1. Parameters Related to the Polymer Solution
4.1.1. Concentration of the Polymer
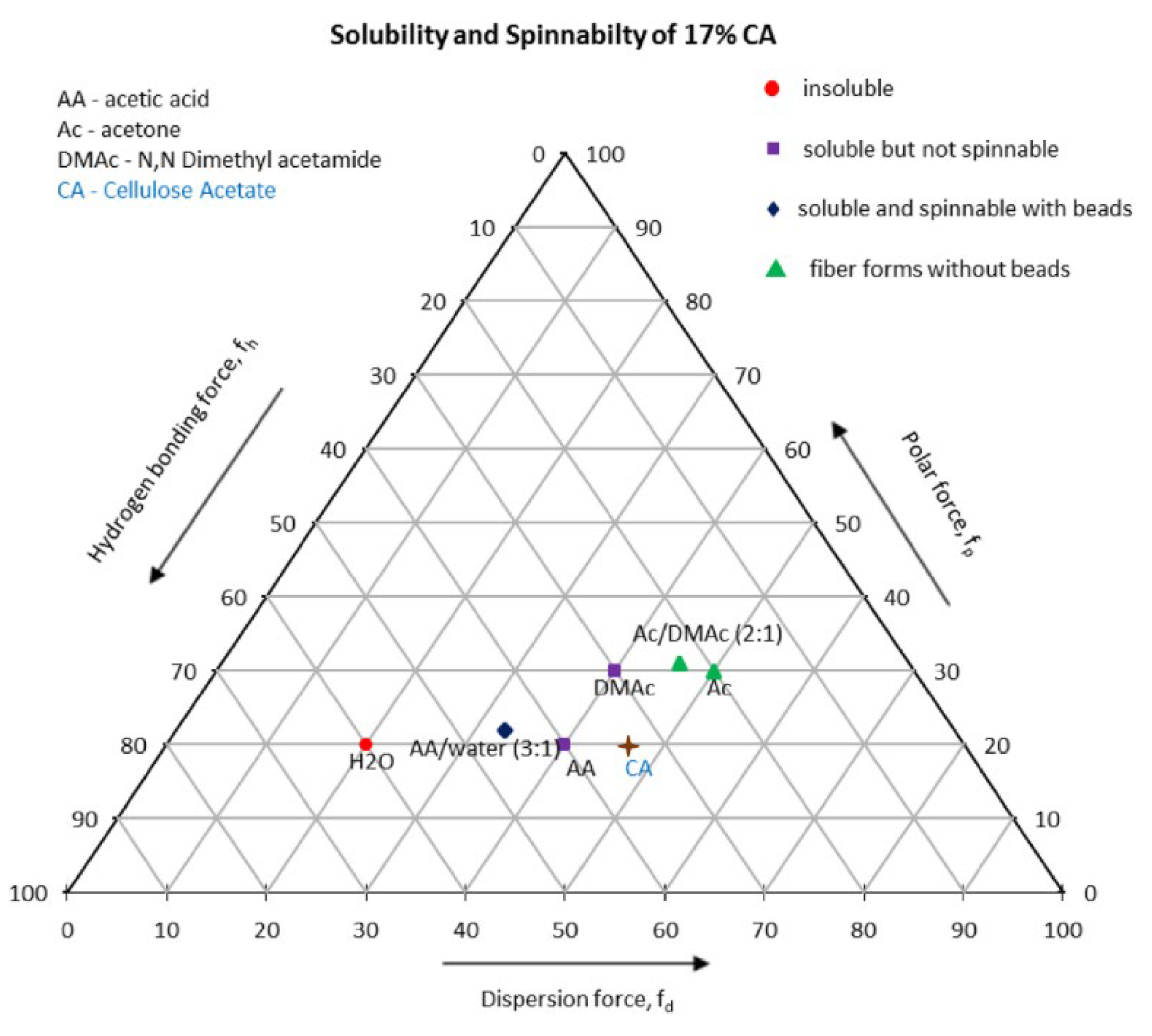
4.1.2. The Solvent
4.1.3. Electrical Conductivity
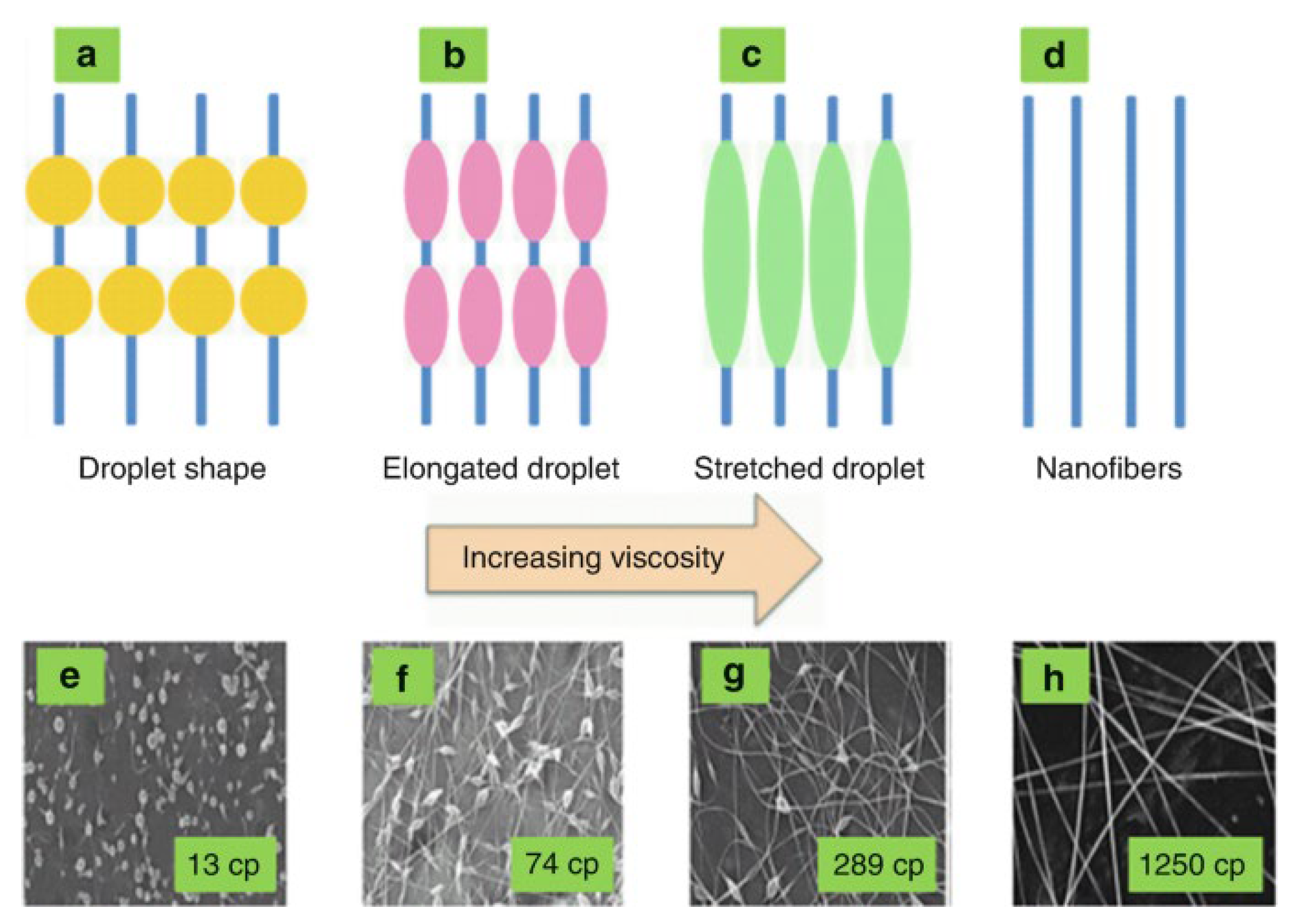
4.1.4. Viscosity
4.1.5. Molecular Weight
4.2. Parameters Related Electrospinning Equipment
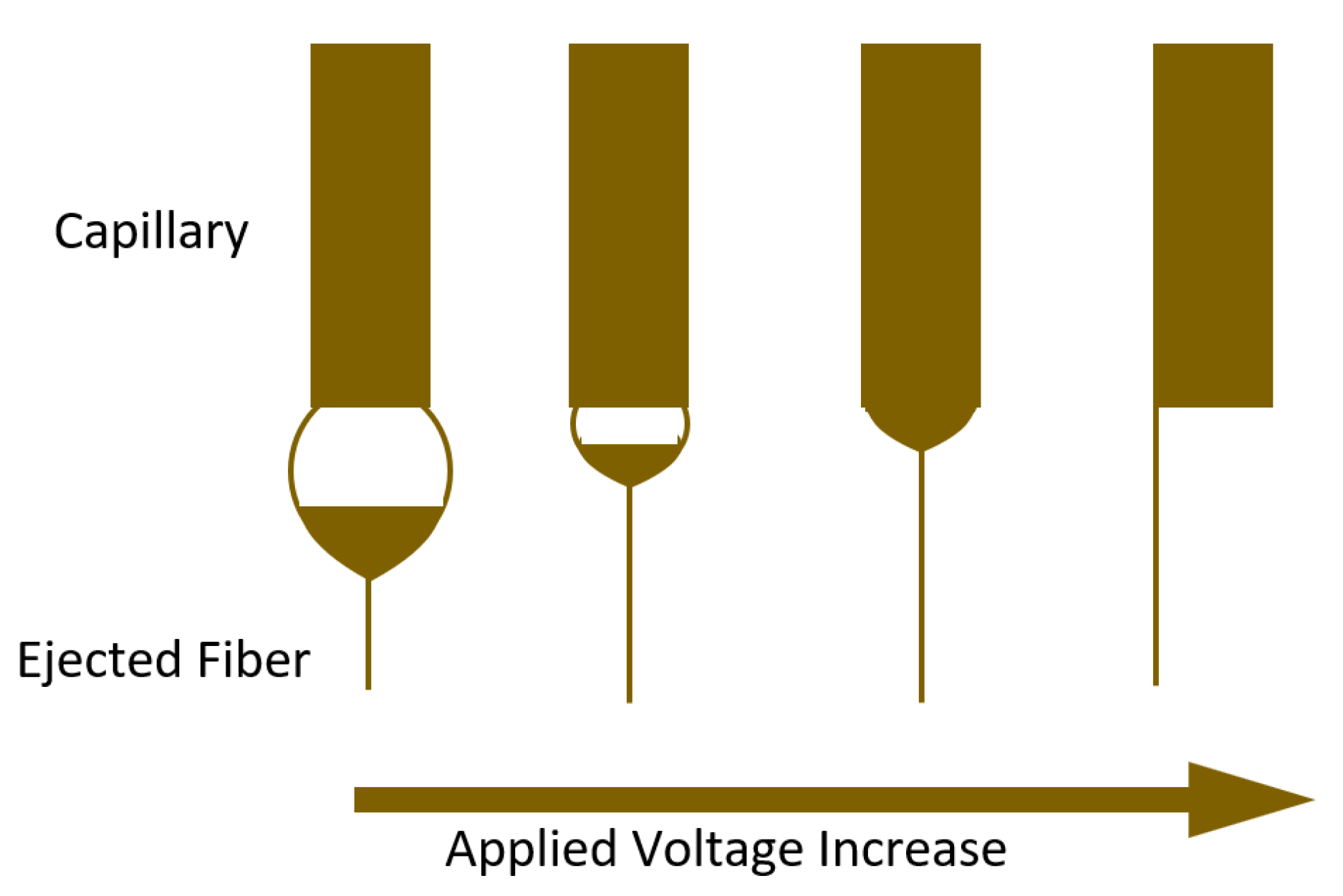
4.2.1. Applied Voltage
4.2.2. Feed Rate
4.2.3. Distance between Tip and Collector
4.3. Parameters Related to Environment
5. Properties of Polymer Nanofibers
5.1. Mechanical Properties
- A manipulation system that precisely isolates, aligns, and grasps a single nanofiber on a frame without slipping or damaging.
- A proper monitoring system to verify that nanofibers are not harmed by characterization tools such as scanning electron microscopes or transmission electron microscopes.
- A sensitive force transducer having a range of nano- to micro-Newton range (n/μN range) resolution that can measure applied force in the n/μN range.
- An actuator that is capable to load nanofibers until fracture, with high resolution (load unit: μN).
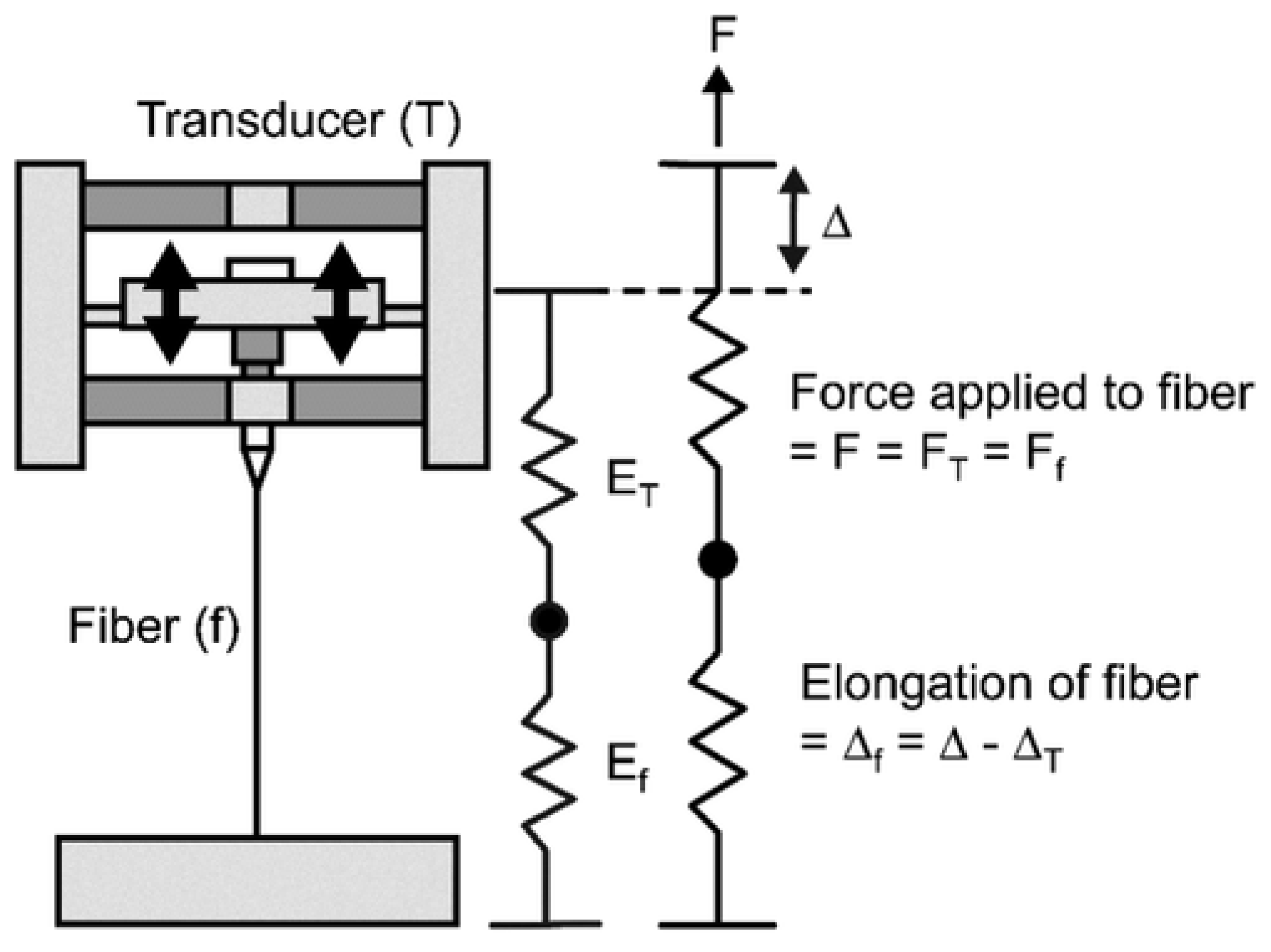
5.1.1. The Tensile Test Method
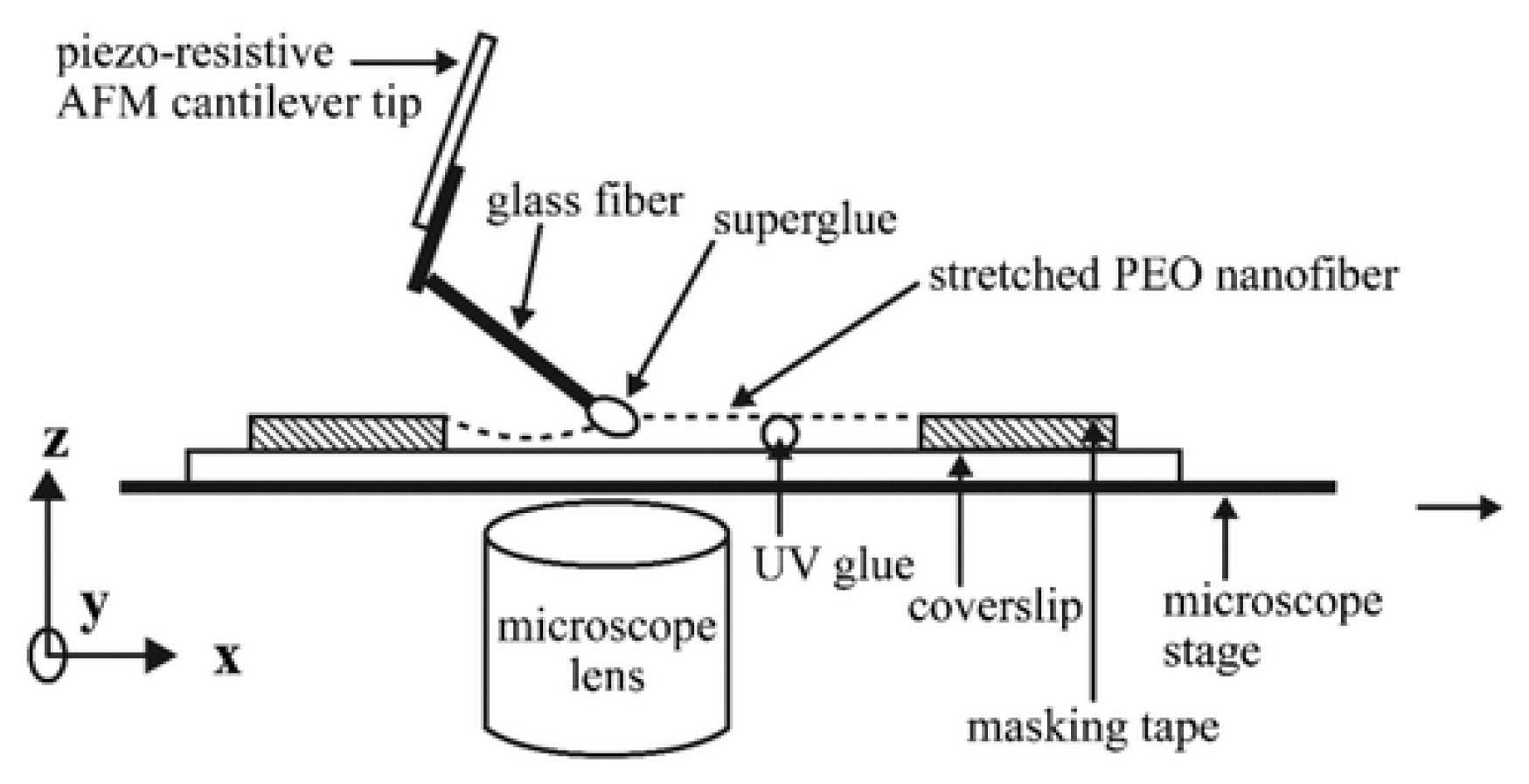
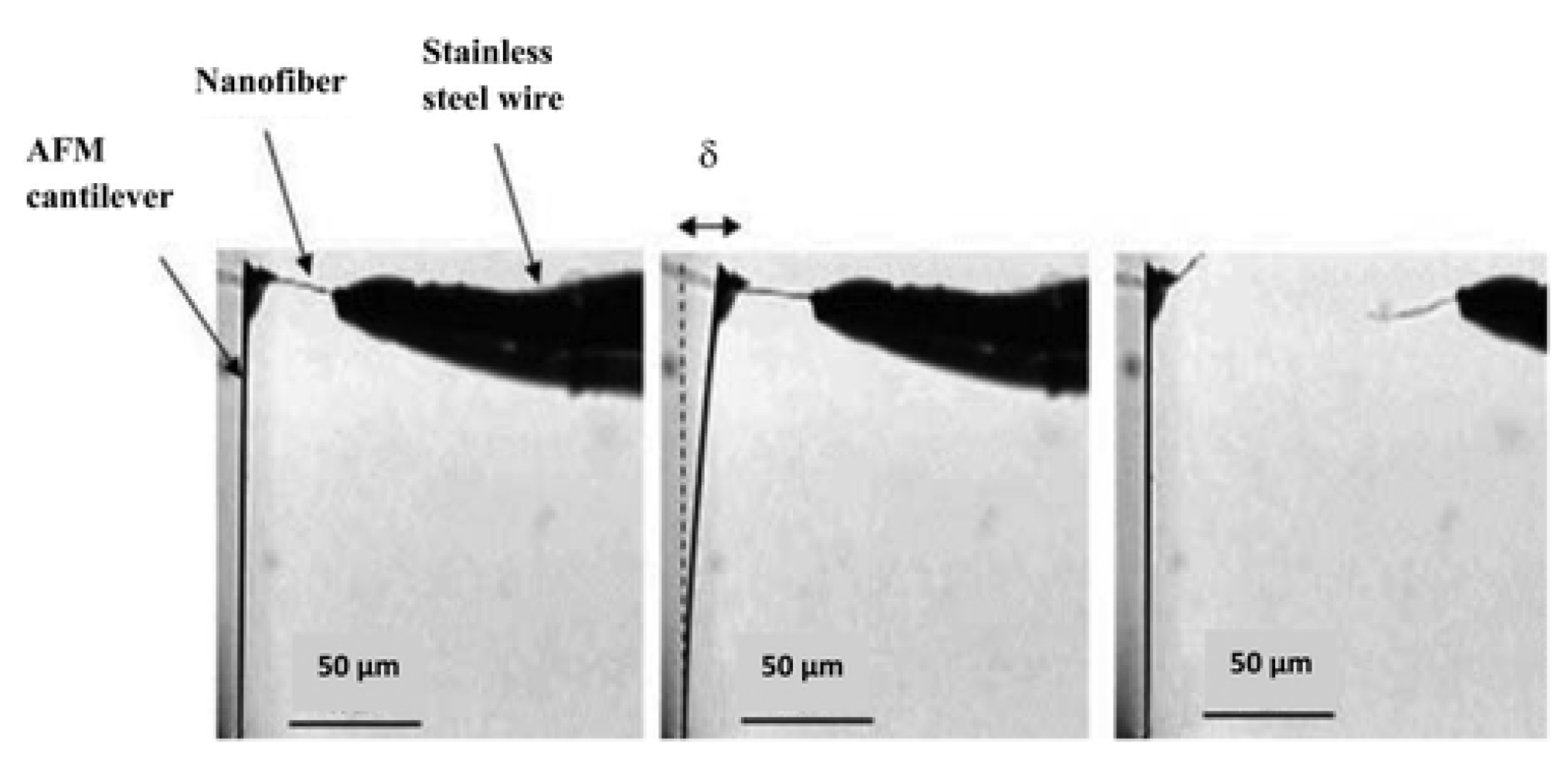
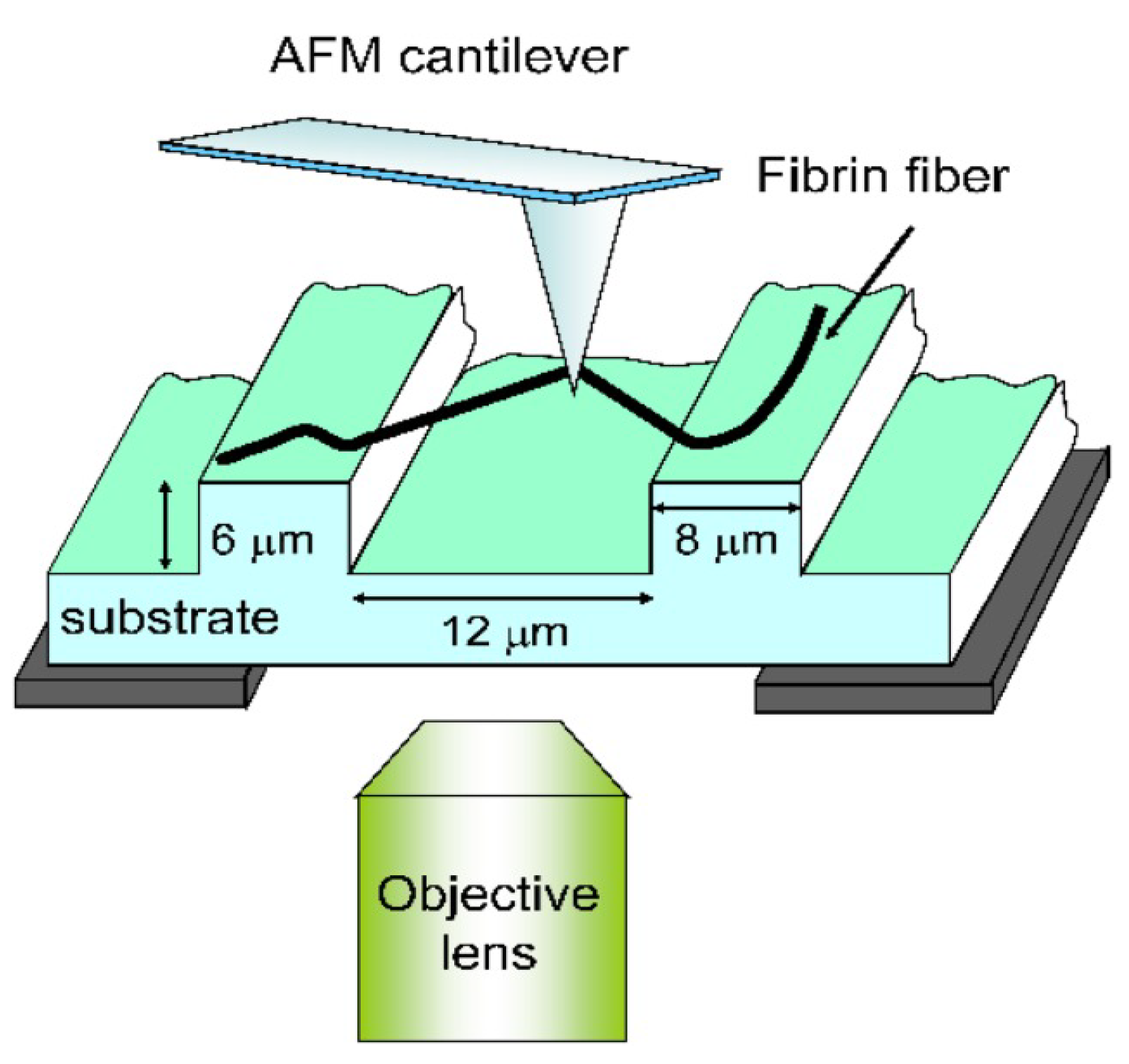
5.1.2. Atomic Force Microscopy Method for Tension Test
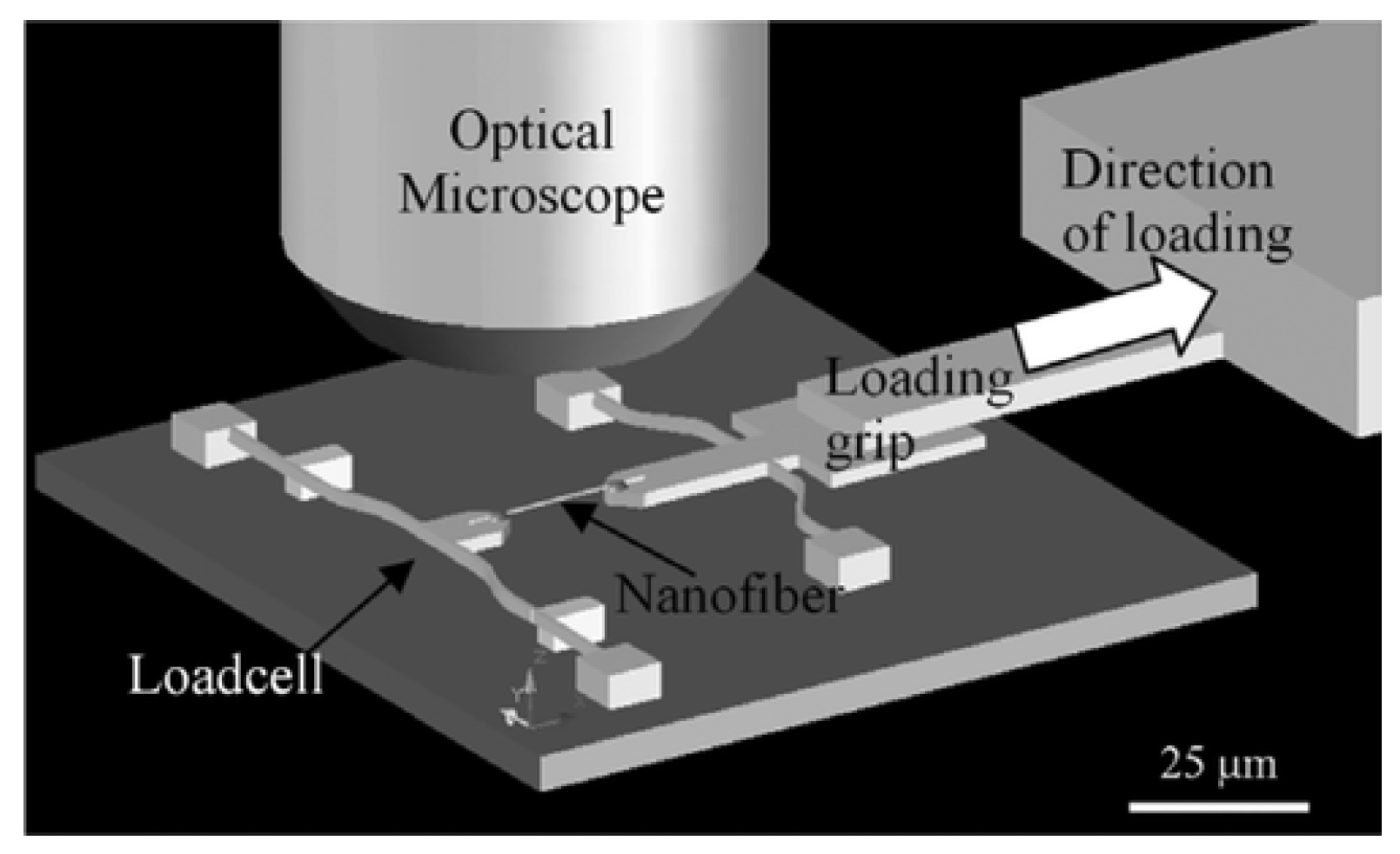
5.1.3. Microelectromechanical System
5.1.4. Nano Indentation Method
5.1.5. Shear Modulation Force Microscopy Method
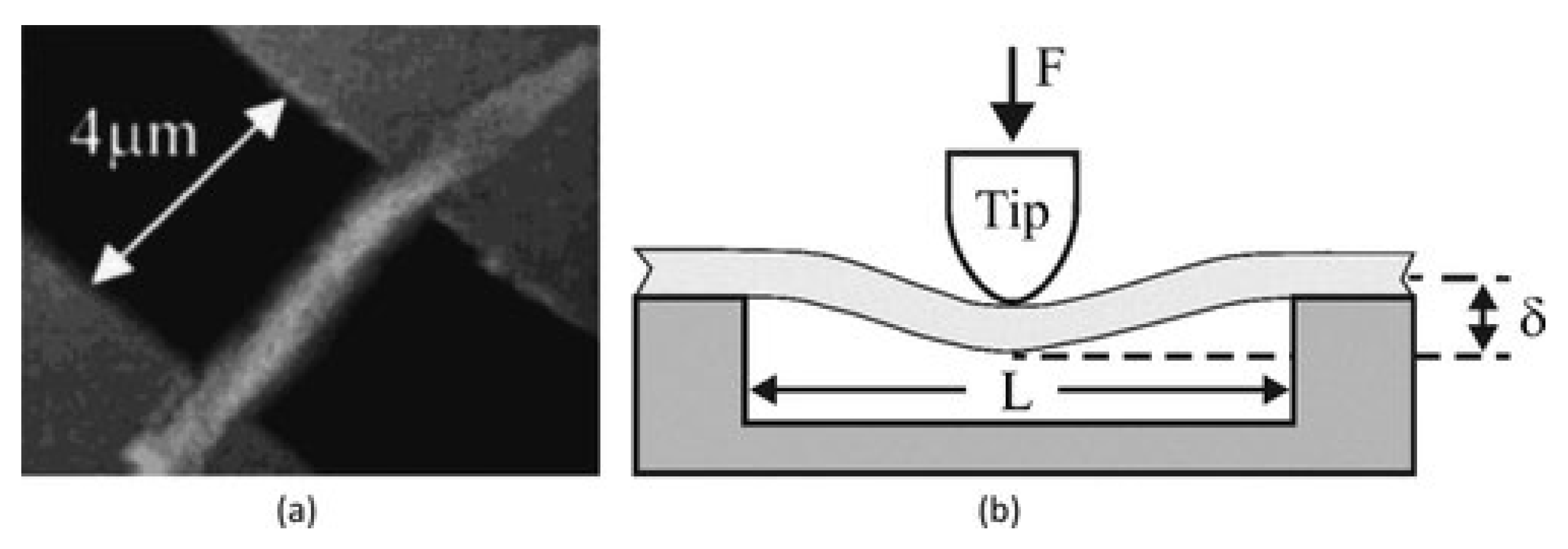
5.1.6. Bending Test Method
Two Point Bending Test Method
Three-Point Bending Test Method
5.1.7. Stretching Method
5.2. Chemical Properties
5.3. Thermal Properties
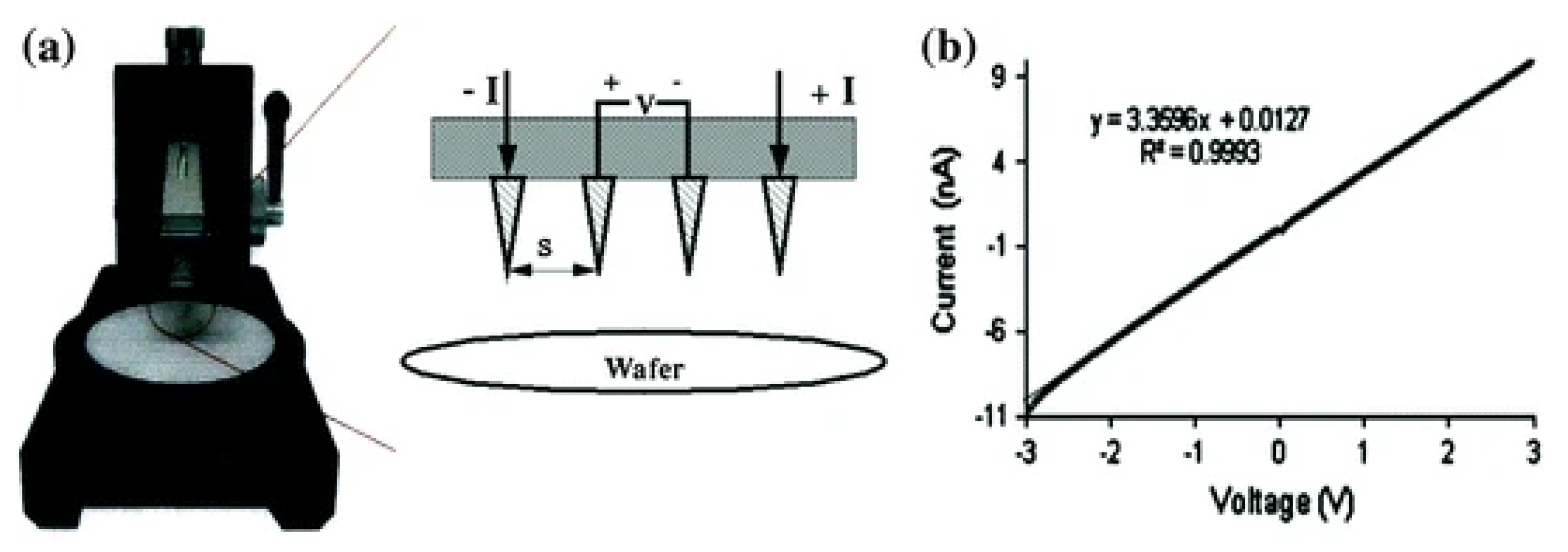
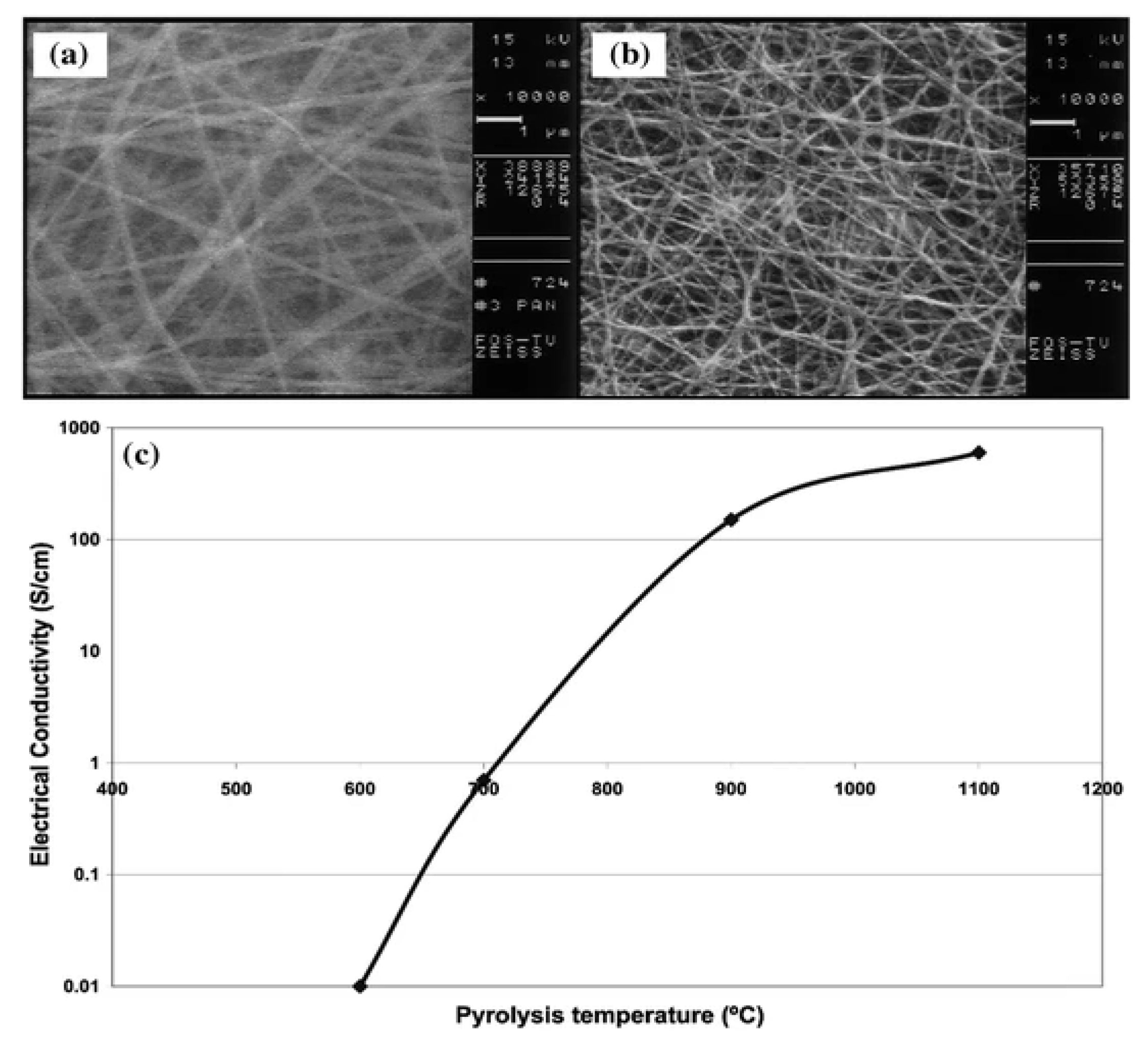
5.4. Electrical Properties
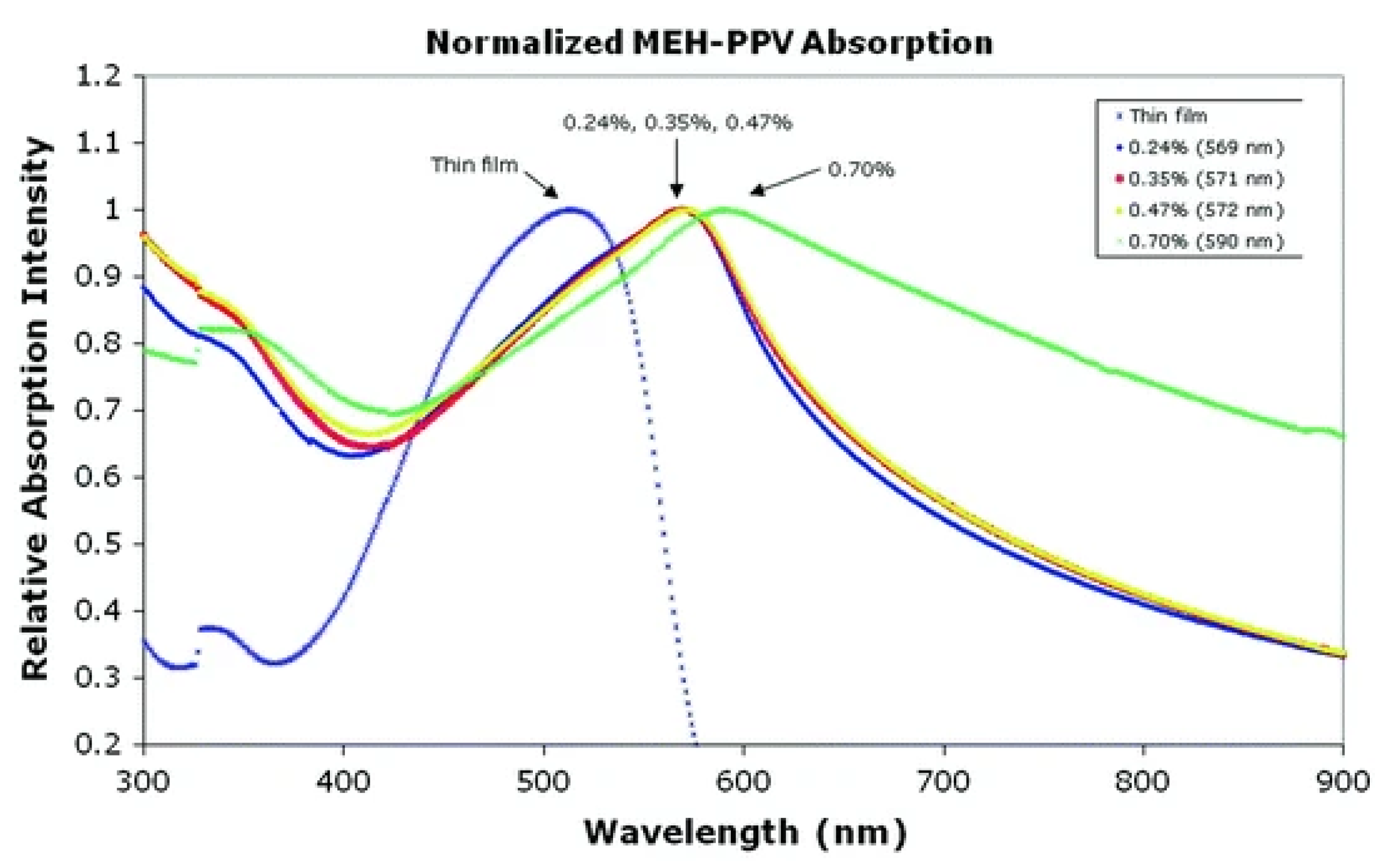
5.5. Optical Properties
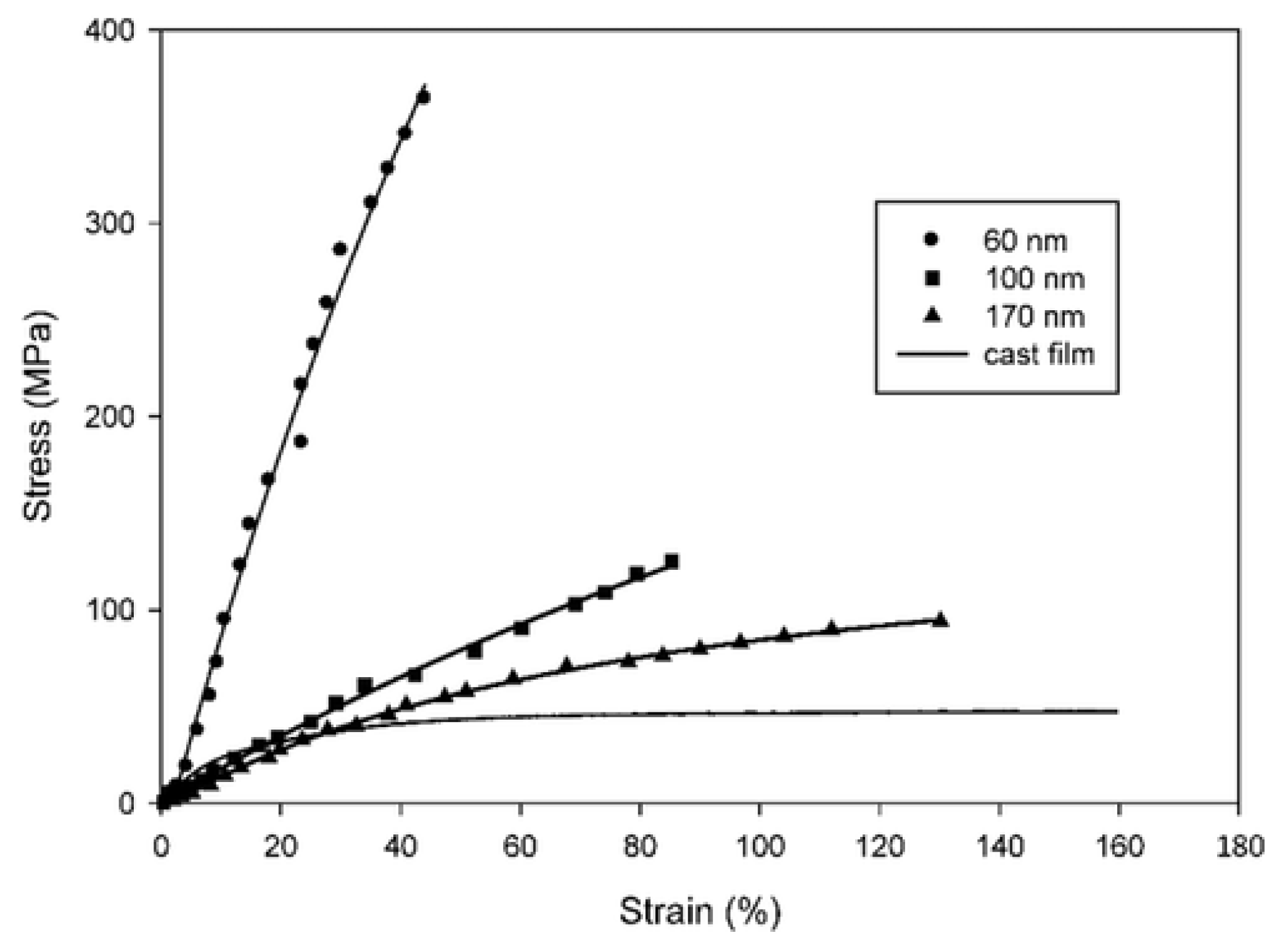
5.6. Effect of Geometrical Properties and Processing Parameters
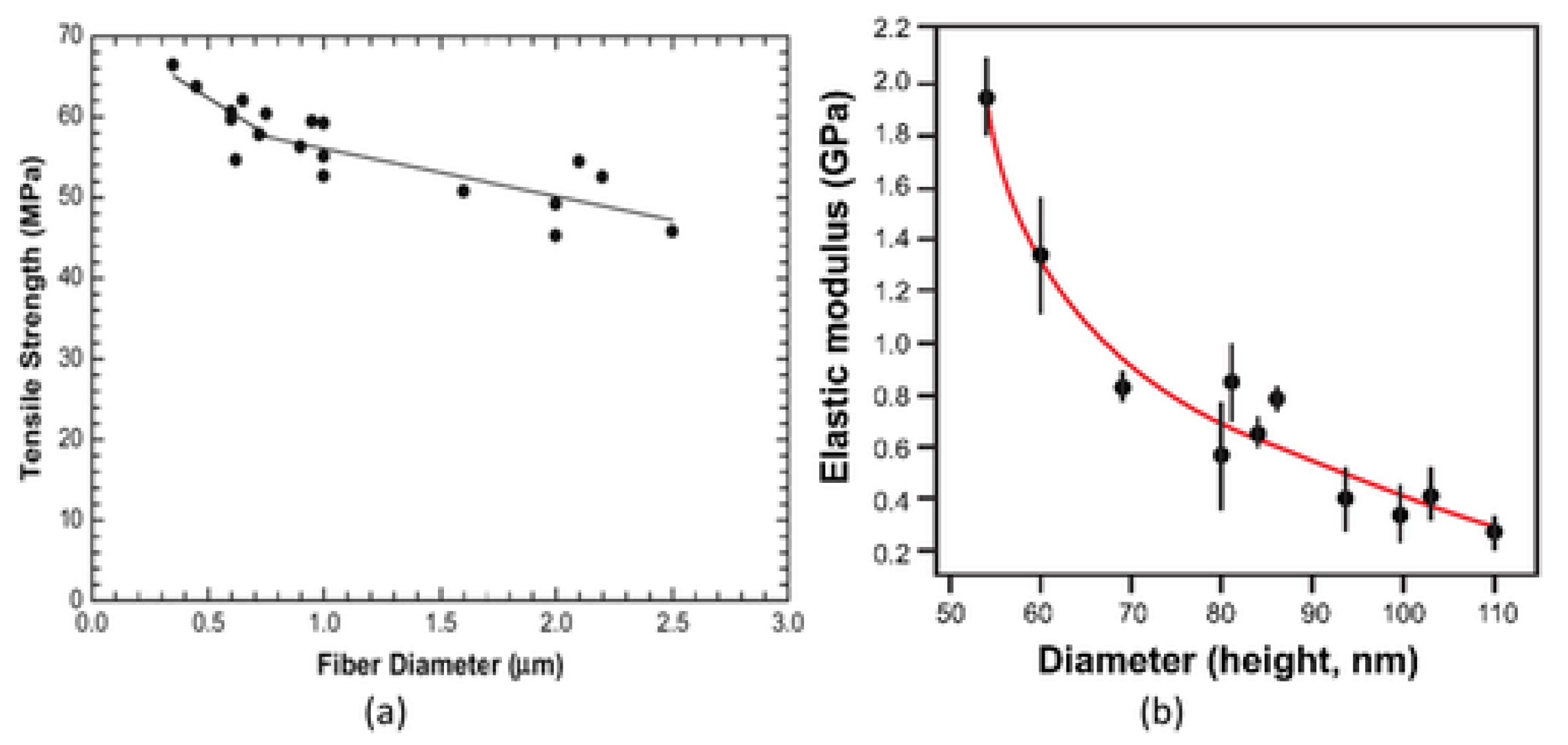
5.6.1. Effect of Diameter
5.6.2. Effect of Collector Type and Take-up Velocity
6. Modeling of Electrospun Nanofibers
6.1. One-Dimensional Steady State Model
6.2. Spivak Dzenis Model
6.3. Wan–Guo–Pan Model
6.4. Allometric Model
7. Applications
7.1. Biomedical Applications
7.1.1. Tissue Engineering
7.1.2. Wound Healing and Dressing
7.1.3. Drug Delivery Systems
7.2. Sensors and Biosensors
7.3. Air Filtration
7.4. Defence Applications
7.5. Energy Devices
7.6. Commercialization
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ko, F.K. Nanofiber technology: Bridging the gap between nano and macro world. Nanoengin. Nanofib. Mater. 2004, 169, 1–18. [Google Scholar]
- Roco, M.C.; Williams, R.S.; Alivisatos, P. Nanotechnology Research Directions: Iwgn Workshop Report. Vision for Nanotechnology r&d in the Next Decade; National Science and Technology Councilarlington VA; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999. [Google Scholar]
- Frenot, A.; Chronakis, I.S. Polymer nanofibers assembled by electrospinning. Curr. Opin. Colloid Interf. Sci. 2003, 8, 64–75. [Google Scholar] [CrossRef]
- Haseeb, B. Controlled deposition and alignment of electrospun PMMA-g-PDMS nanofibers by novel electrospinning setups. Master of Science’s Thesis, KTH School of Chemical Science and Engineering (CHE), Stockholm, Sweden, 2011. [Google Scholar]
- Yang, B.; Wang, L.; Zhang, M.; Luo, J.; Lu, Z.; Ding, X. Fabrication, applications, and prospects of aramid nanofiber. Adv. Funct. Mater. 2020, 30, 2000186. [Google Scholar] [CrossRef]
- Zahmatkeshan, M.; Adel, M.; Bahrami, S.; Esmaeili, F.; Rezayat, S.M.; Saeedi, Y.; Mehravi, B.; Jameie, S.B.; Ashtari, K. Polymer-based nanofibers: Preparation, fabrication, and applications. In Handbook of Nanofibers; Springer: Berlin/Heidelberg, Germany, 2019; pp. 215–261. [Google Scholar]
- Ramesh Kumar, P.; Khan, N.; Vivekanandhan, S.; Satyanarayana, N.; Mohanty, A.; Misra, M. Nanofibers: Effective generation by electrospinning and their applications. J. Nanosci. Nanotechnol. 2012, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Liow, S.S.; Loh, X.J. Biodegradable polymers for electrospinning: Towards biomedical applications. Mater. Sci. Eng. C 2014, 45, 659–670. [Google Scholar] [CrossRef]
- Teo, W.E.; Ramakrishna, S. A review on electrospinning design and nanofibre assemblies. Nanotechnology 2006, 17, R89. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Meghana, B.; Umesh, D.; Abhay, S.; Vilasrao, K. Electrospinning Nanotechnology-A Robust Method for Preparation of Nanofibers for Medicinal and Pharmaceutical Application. Asian J. Pharm. Res. Dev. 2020, 8, 176–184. [Google Scholar] [CrossRef]
- Liu, C.; Tan, Y.; Liu, Y.; Shen, K.; Peng, B.; Niu, X.; Ran, F. Microporous carbon nanofibers prepared by combining electrospinning and phase separation methods for supercapacitor. J. Energy Chem. 2016, 25, 587–593. [Google Scholar] [CrossRef]
- DeFrates, K.; Markiewicz, T.; Xue, Y.; Callaway, K.; Gough, C.; Moore, R.; Bessette, K.; Mou, X.; Hu, X. Air-jet spinning corn zein protein nanofibers for drug delivery: Effect of biomaterial structure and shape on release properties. Mater. Sci. Eng. C 2021, 118, 111419. [Google Scholar] [CrossRef]
- Nain, A.S.; Phillippi, J.A.; Sitti, M.; MacKrell, J.; Campbell, P.G.; Amon, C. Control of cell behavior by aligned micro/nanofibrous biomaterial scaffolds fabricated by spinneret-based tunable engineered parameters (STEP) technique. Small 2008, 4, 1153–1159. [Google Scholar] [CrossRef]
- Yin, Z.; Wu, F.; Zheng, Z.; Kaplan, D.L.; Kundu, S.C.; Lu, S. Self-assembling silk-based nanofibers with hierarchical structures. ACS Biomater. Sci. Eng. 2017, 3, 2617–2627. [Google Scholar] [CrossRef]
- Cheng, K.C.; Bedolla-Pantoja, M.A.; Kim, Y.-K.; Gregory, J.V.; Xie, F.; De France, A.; Hussal, C.; Sun, K.; Abbott, N.L.; Lahann, J. Templated nanofiber synthesis via chemical vapor polymerization into liquid crystalline films. Science 2018, 362, 804–808. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, F.; Yu, Y.; Li, P.; Yang, X.; Lu, J.; Ryu, S. Preparation of PAN-based carbon nanofibers by hot-stretching. Compos. Interf. 2008, 15, 671–677. [Google Scholar] [CrossRef]
- Bera, B. Literature review on electrospinning process (a fascinating fiber fabrication technique). Imper. J. Interdiscip. Res. 2016, 2, 972–984. [Google Scholar]
- Wang, G.; Yu, D.; Kelkar, A.D.; Zhang, L. Electrospun nanofiber: Emerging reinforcing filler in polymer matrix composite materials. Progr. Polymer Sci. 2017, 75, 73–107. [Google Scholar] [CrossRef]
- Agarwal, S.; Greiner, A.; Wendorff, J.H. Functional materials by electrospinning of polymers. Progr. Polymer Sci. 2013, 38, 963–991. [Google Scholar] [CrossRef]
- Ingavle, G.C.; Leach, J.K. Advancements in electrospinning of polymeric nanofibrous scaffolds for tissue engineering. Tiss. Eng. Part B Rev. 2014, 20, 277–293. [Google Scholar] [CrossRef]
- Baji, A.; Mai, Y.-W.; Wong, S.-C.; Abtahi, M.; Chen, P. Electrospinning of polymer nanofibers: Effects on oriented morphology, structures and tensile properties. Compos. Sci. Technol. 2010, 70, 703–718. [Google Scholar] [CrossRef]
- Chronakis, I.S. Novel nanocomposites and nanoceramics based on polymer nanofibers using electrospinning process—A review. J. Mater. Proc. Technol. 2005, 167, 283–293. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Fakirov, S. Synthetic Polymer-Polymer Composites; Carl Hanser Verlag GmbH Co KG: Munich, Germany, 2012. [Google Scholar]
- Hwang, K.Y.; Kim, S.-D.; Kim, Y.-W.; Yu, W.-R. Mechanical characterization of nanofibers using a nanomanipulator and atomic force microscope cantilever in a scanning electron microscope. Polym. Test. 2010, 29, 375–380. [Google Scholar] [CrossRef]
- Yarin, A.L.; Koombhongse, S.; Reneker, D.H. Bending instability in electrospinning of nanofibers. J. Appl. Phys. 2001, 89, 3018–3026. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L.; Fong, H.; Koombhongse, S. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J. Appl. Phys. 2000, 87, 4531–4547. [Google Scholar] [CrossRef]
- Ji, Y.; Li, B.; Ge, S.; Sokolov, J.C.; Rafailovich, M.H. Structure and nanomechanical characterization of electrospun PS/clay nanocomposite fibers. Langmuir 2006, 22, 1321–1328. [Google Scholar] [CrossRef]
- Ramakrishna, S. An Introduction to Electrospinning and Nanofibers; World Scientific: Singapore, 2005. [Google Scholar]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Anton, F. Process and Apparatus for Preparing Artificial Threads. U.S. Patent No. 1,975,504, 2 October 1934. [Google Scholar]
- Baumgarten, P.K. Electrostatic spinning of acrylic microfibers. J. Colloid Interf. Sci. 1971, 36, 71–79. [Google Scholar] [CrossRef]
- Teo, W.-E.; Ramakrishna, S. Electrospun nanofibers as a platform for multifunctional, hierarchically organized nanocomposite. Compos. Sci. Technol. 2009, 69, 1804–1817. [Google Scholar] [CrossRef]
- Zhang, Y.; Venugopal, J.; Huang, Z.-M.; Lim, C.T.; Ramakrishna, S. Crosslinking of the electrospun gelatin nanofibers. Polymer 2006, 47, 2911–2917. [Google Scholar] [CrossRef]
- Kulkarni, A.; Bambole, V.; Mahanwar, P. Electrospinning of polymers, their modeling and applications. Polym. Plastics Technol. Eng. 2010, 49, 427–441. [Google Scholar] [CrossRef]
- Hu, X.; Liu, S.; Zhou, G.; Huang, Y.; Xie, Z.; Jing, X. Electrospinning of polymeric nanofibers for drug delivery applications. J. Control. Release 2014, 185, 12–21. [Google Scholar] [CrossRef]
- Dzenis, Y. Spinning continuous fibers for nanotechnology. Science 2004, 304, 1917–1919. [Google Scholar] [CrossRef]
- Taylor, S.R. Abundance of chemical elements in the continental crust: A new table. Geochim. Cosmochim. Acta 1964, 28, 1273–1285. [Google Scholar] [CrossRef]
- Reneker, D.; Yarin, A.; Zussman, E.; Xu, H. Electrospinning of nanofibers from polymer solutions and melts. Adv. Appl. Mechan. 2007, 41, 43–346. [Google Scholar]
- Matarrese, S.; Pantano, O.; Saez, D. General relativistic dynamics of irrotational dust: Cosmological implications. Phys. Rev. Lett. 1994, 72, 320. [Google Scholar] [CrossRef]
- Brown, T.D.; Dalton, P.D.; Hutmacher, D.W. Melt electrospinning today: An opportune time for an emerging polymer process. Progr. Polymer Sci. 2016, 56, 116–166. [Google Scholar] [CrossRef]
- Yu, M.; Dong, R.H.; Yan, X.; Yu, G.F.; You, M.H.; Ning, X.; Long, Y.Z. Recent advances in needleless electrospinning of ultrathin fibers: From academia to industrial production. Macromol. Mater. Eng. 2017, 302, 1700002. [Google Scholar] [CrossRef]
- El-Sayed, H.; Vineis, C.; Varesano, A.; Mowafi, S.; Carletto, R.A.; Tonetti, C.; Abou Taleb, M. A critique on multi-jet electrospinning: State of the art and future outlook. Nanotechnol. Rev. 2019, 8, 236–245. [Google Scholar] [CrossRef]
- Kim, Y.; Ahn, K.; Sung, Y. A Manufacturing Device and the Method of Preparing for the Nanofibers via Electro-Blown Spinning Process. KR Patent Registration No. 1005434890000, 23 January 2006. [Google Scholar]
- Wang, L.; Ahmad, Z.; Huang, J.; Li, J.-S.; Chang, M.-W. Multi-compartment centrifugal electrospinning based composite fibers. Chem. Eng. J. 2017, 330, 541–549. [Google Scholar] [CrossRef]
- He, X.-X.; Zheng, J.; Yu, G.-F.; You, M.-H.; Yu, M.; Ning, X.; Long, Y.-Z. Near-field electrospinning: Progress and applications. J. Phys. Chem. C 2017, 121, 8663–8678. [Google Scholar] [CrossRef]
- Han, D.; Steckl, A.J. Coaxial electrospinning formation of complex polymer fibers and their applications. ChemPlusChem 2019, 84, 1453–1497. [Google Scholar] [CrossRef] [PubMed]
- Buzgo, M.; Mickova, A.; Rampichova, M.; Doupnik, M. Blend electrospinning, coaxial electrospinning, and emulsion electrospinning techniques. In Core-Shell Nanostructures for Drug Delivery and Theranostics; Woodhead Publishing Series in Biomaterials; Elsevier: London, UK, 2018; pp. 325–347. [Google Scholar]
- Persano, L.; Camposeo, A.; Tekmen, C.; Pisignano, D. Industrial upscaling of electrospinning and applications of polymer nanofibers: A review. Macromol. Mater. Eng. 2013, 298, 504–520. [Google Scholar] [CrossRef]
- Zheng, Y.; Gong, R.H.; Zeng, Y. Multijet motion and deviation in electrospinning. RSC Adv. 2015, 5, 48533–48540. [Google Scholar] [CrossRef]
- Wang, X.; Wang, X.; Lin, T. Electric field analysis of spinneret design for needleless electrospinning of nanofibers. J. Mater. Res. 2012, 27, 3013–3019. [Google Scholar] [CrossRef]
- Niu, H.; Lin, T.; Wang, X. Needleless electrospinning. I. A comparison of cylinder and disk nozzles. J. Appl. Polym. Sci. 2009, 114, 3524–3530. [Google Scholar] [CrossRef]
- Niu, H.; Zhou, H.; Yan, G.; Wang, H.; Fu, S.; Zhao, X.; Shao, H.; Lin, T. Enhancement of coil electrospinning using two-level coil structure. Indust. Eng. Chem. Res. 2018, 57, 15473–15478. [Google Scholar] [CrossRef]
- Thoppey, N.M.; Bochinski, J.R.; Clarke, L.I.; Gorga, R.E. Edge electrospinning for high throughput production of quality nanofibers. Nanotechnology 2011, 22, 345301. [Google Scholar] [CrossRef]
- Jiang, G.; Zhang, S.; Qin, X. High throughput of quality nanofibers via one stepped pyramid-shaped spinneret. Mater. Lett. 2013, 106, 56–58. [Google Scholar] [CrossRef]
- Wei, L.; Sun, R.; Liu, C.; Xiong, J.; Qin, X. Mass production of nanofibers from needleless electrospinning by a novel annular spinneret. Mater. Design 2019, 179, 107885. [Google Scholar] [CrossRef]
- Xiong, J.; Liu, Y.; Li, A.; Wei, L.; Wang, L.; Qin, X.; Yu, J. Mass production of high-quality nanofibers via constructing pre-Taylor cones with high curvature on needleless electrospinning. Mater. Design 2021, 197, 109247. [Google Scholar] [CrossRef]
- Yan, X.; Yu, M.; Ramakrishna, S.; Russell, S.J.; Long, Y.-Z. Advances in portable electrospinning devices for in situ delivery of personalized wound care. Nanoscale 2019, 11, 19166–19178. [Google Scholar] [CrossRef]
- Hekmati, A.H.; Rashidi, A.; Ghazisaeidi, R.; Drean, J.-Y. Effect of needle length, electrospinning distance, and solution concentration on morphological properties of polyamide-6 electrospun nanowebs. Textile Res. J. 2013, 83, 1452–1466. [Google Scholar] [CrossRef]
- De, B.; Banerjee, S.; Verma, K.D.; Pal, T.; Manna, P.; Kar, K.K. Carbon nanofiber as electrode materials for supercapacitors. In Handbook of Nanocomposite Supercapacitor Materials II; Series in Materials Science, Springer Nature Switzerland Springer; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2020; pp. 149–181, 179–200. [Google Scholar]
- Aleisa, R. Electrospinning. In Handbook of Synthetic Methodologies and Protocols of Nanomaterials: Volume 3: Unconventional Methods for Nanostructure Fabrication; World Scientific Publishing Co. Pte. Ltd.: Singapore, 2020; pp. 149–181. [Google Scholar]
- Gu, S.; Ren, J.; Vancso, G. Process optimization and empirical modeling for electrospun polyacrylonitrile (PAN) nanofiber precursor of carbon nanofibers. Eur. Polymer J. 2005, 41, 2559–2568. [Google Scholar] [CrossRef]
- Gu, X.; Li, N.; Luo, J.; Xia, X.; Gu, H.; Xiong, J. Electrospun polyurethane microporous membranes for waterproof and breathable application: The effects of solvent properties on membrane performance. Polymer Bullet. 2018, 75, 3539–3553. [Google Scholar] [CrossRef]
- Liu, F.; Li, M.; Shao, W.; Yue, W.; Hu, B.; Weng, K.; Chen, Y.; Liao, X.; He, J. Preparation of a polyurethane electret nanofiber membrane and its air-filtration performance. J. Colloid Interf. Sci. 2019, 557, 318–327. [Google Scholar] [CrossRef]
- Ding, B.; Kim, H.Y.; Lee, S.C.; Shao, C.L.; Lee, D.R.; Park, S.J.; Kwag, G.B.; Choi, K.J. Preparation and characterization of a nanoscale poly (vinyl alcohol) fiber aggregate produced by an electrospinning method. J. Polymer Sci. Part Polymer Phys. 2002, 40, 1261–1268. [Google Scholar] [CrossRef]
- Maslakci, N.N.; Ulusoy, S.; Uygun, E.; Çevikbaş, H.; Oksuz, L.; Can, H.K.; Uygun Oksuz, A. Ibuprofen and acetylsalicylic acid loaded electrospun PVP-dextran nanofiber mats for biomedical applications. Polymer Bullet. 2017, 74, 3283–3299. [Google Scholar] [CrossRef]
- Karakaş, H. Electrospinning of nanofibers and their applications. J. Algebr. Statistic 2015, 13, 1447–1454. [Google Scholar]
- Maurmann, N.; Sperling, L.-E.; Pranke, P. Electrospun and electrosprayed scaffolds for tissue engineering. Cut. Edge Enabling Technol. Regen. Med. 2018, 10, 79–100. [Google Scholar]
- Shin, M.K.; Kim, Y.J.; Kim, S.I.; Kim, S.-K.; Lee, H.; Spinks, G.M.; Kim, S.J. Enhanced conductivity of aligned PANi/PEO/MWNT nanofibers by electrospinning. Sens. Actuat. Chem. 2008, 134, 122–126. [Google Scholar] [CrossRef]
- Aussawasathien, D.; Sahasithiwat, S.; Menbangpung, L. Electrospun camphorsulfonic acid doped poly (o-toluidine)–polystyrene composite fibers: Chemical vapor sensing. Synthetic Metals 2008, 158, 259–263. [Google Scholar] [CrossRef]
- Janani, G.; Kumar, M.; Chouhan, D.; Moses, J.C.; Gangrade, A.; Bhattacharjee, S.; Mandal, B.B. Insight into silk-based biomaterials: From physicochemical attributes to recent biomedical applications. ACS Appl. Bio Mater. 2019, 2, 5460–5491. [Google Scholar] [CrossRef] [PubMed]
- Bognitzki, M.; Czado, W.; Frese, T.; Schaper, A.; Hellwig, M.; Steinhart, M.; Greiner, A.; Wendorff, J.H. Nanostructured fibers via electrospinning. Adv. Mater. 2001, 13, 70–72. [Google Scholar] [CrossRef]
- Guibo, Y.; Qing, Z.; Yahong, Z.; Yin, Y.; Yumin, Y. The electrospun polyamide 6 nanofiber membranes used as high efficiency filter materials: Filtration potential, thermal treatment, and their continuous production. J. Appl. Polym. Sci. 2013, 128, 1061–1069. [Google Scholar] [CrossRef]
- Jin, H.-J.; Fridrikh, S.V.; Rutledge, G.C.; Kaplan, D.L. Electrospinning Bombyx mori silk with poly (ethylene oxide). Biomacromolecules 2002, 3, 1233–1239. [Google Scholar] [CrossRef]
- Yao, C.; Li, X.; Neoh, K.; Shi, Z.; Kang, E. Surface modification and antibacterial activity of electrospun polyurethane fibrous membranes with quaternary ammonium moieties. J. Membran. Sci. 2008, 320, 259–267. [Google Scholar] [CrossRef]
- Wei, N.; Sun, C.; Wang, J.; Huang, L.Q. Research on electrospinning of cellulose acetate prepared by acetone/DMAc solvent. In Proceedings of Applied Mechanics and Materials; Trans Tech Publications: Bäch, Switzerland, 2014; Volume 469, pp. 126–129. [Google Scholar]
- Wang, X.; Lee, S.-H.; Drew, C.; Senecal, K.J.; Kumar, J.; Samuelson, L.A. Highly sensitive optical sensors using electrospun polymeric nanofibrous membranes. MRS Online Proc. Library (OPL) 2001, 708, 1400. [Google Scholar] [CrossRef]
- Behtaj, S.; Karamali, F.; Masaeli, E.; Anissimov, Y.G.; Rybachuk, M. Electrospun PGS/PCL, PLLA/PCL, PLGA/PCL and pure PCL scaffolds for retinal progenitor cell cultivation. BioChem. Eng. J. 2021, 166, 107846. [Google Scholar] [CrossRef]
- Dong, B.; Arnoult, O.; Smith, M.E.; Wnek, G.E. Electrospinning of collagen nanofiber scaffolds from benign solvents. Macromol. Rapid Commun. 2009, 30, 539–542. [Google Scholar] [CrossRef]
- Koombhongse, S.; Liu, W.; Reneker, D.H. Flat polymer ribbons and other shapes by electrospinning. J. Polymer Sci. Part Polymer Phys. 2001, 39, 2598–2606. [Google Scholar] [CrossRef]
- Jayaraman, K.; Kotaki, M.; Zhang, Y.; Mo, X.; Ramakrishna, S. Recent advances in polymer nanofibers. J. Nanosci. Nanotechnol. 2004, 4, 52–65. [Google Scholar]
- Sobolčiak, P.; Tanvir, A.; Popelka, A.; Moffat, J.; Mahmoud, K.A.; Krupa, I. The preparation, properties and applications of electrospun co-polyamide 6, 12 membranes modified by cellulose nanocrystals. Mater. Design 2017, 132, 314–323. [Google Scholar] [CrossRef]
- Matysiak, W.; Tański, T.; Smok, W.; Gołombek, K.; Schab-Balcerzak, E. Effect of conductive polymers on the optical properties of electrospun polyacrylonitryle nanofibers filled by polypyrrole, polythiophene and polyaniline. Appl. Surf. Sci. 2020, 509, 145068. [Google Scholar] [CrossRef]
- Joseph, B.; Augustine, R.; Kalarikkal, N.; Thomas, S.; Seantier, B.; Grohens, Y. Recent advances in electrospun polycaprolactone based scaffolds for wound healing and skin bioengineering applications. Mater. Today Commun. 2019, 19, 319–335. [Google Scholar] [CrossRef]
- Ashammakhi, N.; Wimpenny, I.; Nikkola, L.; Yang, Y. Electrospinning: Methods and development of biodegradable nanofibres for drug release. J. Biomed. Nanotechnol. 2009, 5, 19. [Google Scholar] [CrossRef]
- Sill, T.J.; Von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef]
- Stojanovska, E.; Canbay, E.; Pampal, E.S.; Calisir, M.D.; Agma, O.; Polat, Y.; Simsek, R.; Gundogdu, N.S.; Akgul, Y.; Kilic, A. A review on non-electro nanofibre spinning techniques. RSC Adv. 2016, 6, 83783–83801. [Google Scholar] [CrossRef]
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrost. 1995, 35, 151–160. [Google Scholar] [CrossRef]
- Bae, H.-S.; Haider, A.; Selim, K.; Kang, D.-Y.; Kim, E.-J.; Kang, I.-K. Fabrication of highly porous PMMA electrospun fibers and their application in the removal of phenol and iodine. J. Polym. Res. 2013, 20, 1–7. [Google Scholar] [CrossRef]
- Qian, Y.-F.; Su, Y.; Li, X.-Q.; Wang, H.; He, C.L. Electrospinning of polymethyl methacrylate nanofibres in different solvents. Iran. Polym. J. 2010, 19, 79–87. [Google Scholar]
- Haider, A.H.; Kang, S. A Comprehensive Review Summarizing the Effect of Electrospinning Parameters and Potential Applications of Nanofibers in Biomedical and Biotechnology. Arab. J. Chem. 2015, 10, 24. [Google Scholar] [CrossRef]
- Agarwal, S.; Wendorff, J.H.; Greiner, A. Use of electrospinning technique for biomedical applications. Polymer 2008, 49, 5603–5621. [Google Scholar] [CrossRef]
- Majumder, S.; Matin, M.A.; Sharif, A.; Arafat, M.T. Understanding solubility, spinnability and electrospinning behaviour of cellulose acetate using different solvent systems. Bullet. Mater. Sci. 2019, 42, 9. [Google Scholar] [CrossRef]
- Abbasi, N.; Soudi, S.; Hayati-Roodbari, N.; Dodel, M.; Soleimani, M. The effects of plasma treated electrospun nanofibrous poly (ε-caprolactone) scaffolds with different orientations on mouse embryonic stem cell proliferation. Cell J. 2014, 16, 245. [Google Scholar]
- Hodge, J.; Quint, C. The improvement of cell infiltration in an electrospun scaffold with multiple synthetic biodegradable polymers using sacrificial PEO microparticles. J. Biomed. Mater. Res. Part 2019, 107, 1954–1964. [Google Scholar] [CrossRef]
- Santoro, M.; Shah, S.R.; Walker, J.L.; Mikos, A.G. Poly (lactic acid) nanofibrous scaffolds for tissue engineering. Adv. Drug Deliv. Rev. 2016, 107, 206–212. [Google Scholar] [CrossRef]
- Zafar, M.; Najeeb, S.; Khurshid, Z.; Vazirzadeh, M.; Zohaib, S.; Najeeb, B.; Sefat, F. Potential of electrospun nanofibers for biomedical and dental applications. Materials 2016, 9, 73. [Google Scholar] [CrossRef]
- Nagam Hanumantharao, S.; Rao, S. Multi-functional electrospun nanofibers from polymer blends for scaffold tissue engineering. Fibers 2019, 7, 66. [Google Scholar] [CrossRef]
- Tan, E.; Lim, C. Mechanical characterization of nanofibers—A review. Compos. Sci. Technol. 2006, 66, 1102–1111. [Google Scholar] [CrossRef]
- Bazbouz, M.B.; Stylios, G.K. The tensile properties of electrospun nylon 6 single nanofibers Part B Polymer physics. J. Polymer Science Part Polym. Phys. 2010, 48, 1719–1731. [Google Scholar] [CrossRef]
- Zhou, X.; Ding, C.; Cheng, C.; Liu, S.; Duan, G.; Xu, W.; Liu, K.; Hou, H. Mechanical and thermal properties of electrospun polyimide/rGO composite nanofibers via in-situ polymerization and in-situ thermal conversion. Eur. Polymer J. 2020, 141, 110083. [Google Scholar] [CrossRef]
- Hang, F.; Lu, D.; Bailey, R.J.; Jimenez-Palomar, I.; Stachewicz, U.; Cortes-Ballesteros, B.; Davies, M.; Zech, M.; Bödefeld, C.; Barber, A.H. In situ tensile testing of nanofibers by combining atomic force microscopy and scanning electron microscopy. Nanotechnology 2011, 22, 365708. [Google Scholar] [CrossRef]
- Gu, S.Y.; Wu, Q.L.; Ren, J.; Vancso, G.J. Mechanical properties of a single electrospun fiber and its structures. Macromolecul. Rapid Commun. 2005, 26, 716–720. [Google Scholar] [CrossRef]
- Yang, L.; Fitie, C.F.; Van Der Werf, K.O.; Bennink, M.L.; Dijkstra, P.J.; Feijen, J. Mechanical properties of single electrospun collagen type I fibers. Biomaterials 2008, 29, 955–962. [Google Scholar] [CrossRef]
- Lee, K.; Kim, H.; Khil, M.; Ra, Y.; Lee, D. Characterization of nano-structured poly (ε-caprolactone) nonwoven mats via electrospinning. Polymer 2003, 44, 1287–1294. [Google Scholar] [CrossRef]
- Hansen, L.M.; Smith, D.J.; Reneker, D.H.; Kataphinan, W. Water absorption and mechanical properties of electrospun structured hydrogels. J. Appl. Polym. Sci. 2005, 95, 427–434. [Google Scholar] [CrossRef]
- Wei, X.; Xia, Z.; Wong, S.-C.; Baji, A. Modelling of mechanical properties of electrospun nanofibre network. Int. J. ExpeR. Comput. Biomechan. 2009, 1, 45–57. [Google Scholar] [CrossRef]
- Domaschke, S.; Zündel, M.; Mazza, E.; Ehret, A.E. A 3D computational model of electrospun networks and its application to inform a reduced modelling approach. Int. J. Solids Struct. 2019, 158, 76–89. [Google Scholar] [CrossRef]
- Dong, Y. Nanostructures: Properties, Production Methods and Applications; Nova Science Publishers, Inc.: New York, NY, USA, 2013. [Google Scholar]
- Uslu, E.; Gavgali, M.; Erdal, M.O.; Yazman, Ş.; Gemi, L. Determination of mechanical properties of polymer matrix composites reinforced with electrospinning N66, PAN, PVA and PVC nanofibers: A comparative study. Materials Today Commun. 2021, 26, 101939. [Google Scholar] [CrossRef]
- Duan, G.; Liu, S.; Hou, H. Synthesis of polyacrylonitrile and mechanical properties of its electrospun nanofibers. e-Polymers 2018, 18, 569–573. [Google Scholar] [CrossRef]
- Mathew, G.; Hong, J.; Rhee, J.; Lee, H.; Nah, C. Preparation and characterization of properties of electrospun poly (butylene terephthalate) nanofibers filled with carbon nanotubes. Polym. Test. 2005, 24, 712–717. [Google Scholar] [CrossRef]
- Tan, E.; Goh, C.; Sow, C.; Lim, C. Tensile test of a single nanofiber using an atomic force microscope tip. Appl. Phys. Lett. 2005, 86, 073115. [Google Scholar] [CrossRef]
- Zussman, E.; Burman, M.; Yarin, A.; Khalfin, R.; Cohen, Y. Tensile deformation of electrospun nylon-6, 6 nanofibers. J. Polym. Sci. Part B Polym. Phys 2006, 44, 1482–1489. [Google Scholar] [CrossRef]
- Zussman, E.; Chen, X.; Ding, W.; Calabri, L.; Dikin, D.; Quintana, J.; Ruoff, R. Mechanical and structural characterization of electrospun PAN-derived carbon nanofibers. Carbon 2005, 43, 2175–2185. [Google Scholar] [CrossRef]
- Lim, C.; Tan, E.; Ng, S. Effects of crystalline morphology on the tensile properties of electrospun polymer nanofibers. Appl. Phys. Lett. 2008, 92, 141908. [Google Scholar] [CrossRef]
- Tan, E.; Lim, C. Novel approach to tensile testing of micro-and nanoscale fibers. Rev. Sci. Inst. 2004, 75, 2581–2585. [Google Scholar] [CrossRef]
- Naraghi, M.; Chasiotis, I.; Kahn, H.; Wen, Y.; Dzenis, Y. Novel method for mechanical characterization of polymeric nanofibers. Rev. Sci. Instrum. 2007, 78, 085108. [Google Scholar] [CrossRef]
- Naraghi, M.; Chasiotis, I.; Kahn, H.; Wen, Y.; Dzenis, Y. Mechanical deformation and failure of electrospun polyacrylonitrile nanofibers as a function of strain rate. Appl. Phys. Lett. 2007, 91, 151901. [Google Scholar] [CrossRef]
- Naraghi, M.; Arshad, S.; Chasiotis, I. Molecular orientation and mechanical property size effects in electrospun polyacrylonitrile nanofibers. Polymer 2011, 52, 1612–1618. [Google Scholar] [CrossRef]
- Oshida, K.; Murata, M.; Takizawa, Y. Analysis of the dispersion state of pitch particles in polymers for nanofiber fabrication by optical microscopy and image processing. J. Phys. Chem. Solids 2022, 163, 110585. [Google Scholar] [CrossRef]
- Jaeger, D.; Schischka, J.; Bagdahn, J.; Jaeger, R. Tensile testing of individual ultrathin electrospun poly (l-lactic acid) fibers. J. Appl. Polym. Sci. 2009, 114, 3774–3779. [Google Scholar] [CrossRef]
- Samuel, B.; Haque, M.A.; Yi, B.; Rajagopalan, R.; Foley, H. Mechanical testing of pyrolysed poly-furfuryl alcohol nanofibres. Nanotechnology 2007, 18, 115704. [Google Scholar] [CrossRef]
- Jee, A.-Y.; Lee, M. Comparative analysis on the nanoindentation of polymers using atomic force microscopy. Polym. Test. 2010, 29, 95–99. [Google Scholar] [CrossRef]
- Kang, S.-K.; Kim, J.-H.; Lee, Y.-H.; Kim, J.-Y.; Kwon, D. Correlation between the plastic strain and the plastic pileup of the instrumented indentation by utilizing the interrupted tensile test. Mater. Sci. Eng. 2012, 535, 197–201. [Google Scholar] [CrossRef]
- Wang, W.; Barber, A.H. Measurement of size-dependent glass transition temperature in electrospun polymer fibers using AFM nanomechanical testing. J. Polym. Sci. Part Polym. Phys. 2012, 50, 546–551. [Google Scholar] [CrossRef]
- Yarin, A.; Zussman, E. Electrospinning of nanofibers from polymer solutions. XXI ICTAM 2004, 15–21. [Google Scholar]
- Shakil, U.A.; Hassan, S.B.; Yahya, M.Y.; Nauman, S. Mechanical properties of electrospun nanofiber reinforced/interleaved epoxy matrix composites—A review. Polym. Compos. 2020, 41, 2288–2315. [Google Scholar] [CrossRef]
- Tan, E.P.; Lim, C. Effects of annealing on the structural and mechanical properties of electrospun polymeric nanofibres. Nanotechnology 2006, 17, 2649. [Google Scholar] [CrossRef]
- Tan, E.; Lim, C. Physical properties of a single polymeric nanofiber. Appl. Phys. Lett. 2004, 84, 1603–1605. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Najafabadi, M.A.; Saghafi, H.; Mohammadi, R. Considering damages to open-holed composite laminates modified by nanofibers under the three-point bending test. Polym. Test. 2018, 70, 363–377. [Google Scholar] [CrossRef]
- Bauchau, O.A.; Craig, J.I. Structural Analysis: With Applications to Aerospace Structures; Springer Science & Business Media: Dordrecht, The Netherlands; Heidelberg, Germany; London, UK; New York, NY, USA, 2009; Volume 163. [Google Scholar]
- Tan, E.; Ng, S.; Lim, C. Tensile testing of a single ultrafine polymeric fiber. Biomaterials 2005, 26, 1453–1456. [Google Scholar] [CrossRef] [PubMed]
- Hua, L.; Hu, C.; Li, J.; Huang, B.; Du, J. Mechanical characterization of TiO2 nanowires flexible scaffold by nano-indentation/scratch. J. Mechan. Behav. Biomed. Mater. 2022, 126, 105069. [Google Scholar] [CrossRef] [PubMed]
- Janković, B.; Pelipenko, J.; Škarabot, M.; Muševič, I.; Kristl, J. The design trend in tissue-engineering scaffolds based on nanomechanical properties of individual electrospun nanofibers. Int. J. Pharm. 2013, 455, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Zhang, P.; Jiang, Y.; Xu, F.; Yin, J.; Zuo, Y. Mechanical properties of nylon-6/SiO2 nanofibers prepared by electrospinning. Mater. Lett. 2009, 63, 34–36. [Google Scholar] [CrossRef]
- Liao, C.-C.; Wang, C.-C.; Chen, C.-Y.; Lai, W.-J. Stretching-induced orientation of polyacrylonitrile nanofibers by an electrically rotating viscoelastic jet for improving the mechanical properties. Polymer 2011, 52, 2263–2275. [Google Scholar] [CrossRef]
- Wu, N.; Chen, L.; Wei, Q.; Liu, Q.; Li, J. Nanoscale three-point bending of single polymer/inorganic composite nanofiber. J. Textile Inst. 2012, 103, 154–158. [Google Scholar] [CrossRef]
- Guhados, G.; Wan, W.; Hutter, J.L. Measurement of the elastic modulus of single bacterial cellulose fibers using atomic force microscopy. Langmuir 2005, 21, 6642–6646. [Google Scholar] [CrossRef]
- Croisier, F.; Duwez, A.-S.; Jérôme, C.; Léonard, A.; Van Der Werf, K.; Dijkstra, P.J.; Bennink, M.L. Mechanical testing of electrospun PCL fibers. Acta Biomater. 2012, 8, 218–224. [Google Scholar] [CrossRef]
- Carlisle, C.R.; Coulais, C.; Namboothiry, M.; Carroll, D.L.; Hantgan, R.R.; Guthold, M. The mechanical properties of individual, electrospun fibrinogen fibers. Biomaterials 2009, 30, 1205–1213. [Google Scholar] [CrossRef]
- Baker, S.; Sigley, J.; Helms, C.C.; Stitzel, J.; Berry, J.; Bonin, K.; Guthold, M. The mechanical properties of dry, electrospun fibrinogen fibers. Mater. Sci. Eng. C 2012, 32, 215–221. [Google Scholar] [CrossRef]
- Carlisle, C.R.; Coulais, C.; Guthold, M. The mechanical stress–strain properties of single electrospun collagen type I nanofibers. Acta Biomater. 2010, 6, 2997–3003. [Google Scholar] [CrossRef]
- Sadrjahani, M.; Hoseini, S.; Mottaghitalab, V.; Haghi, A. Development and characterization of highly oriented pan nanofiber. Braz. J. Chem. Eng. 2010, 27, 583–589. [Google Scholar] [CrossRef]
- Ratner, B.; Chilkoti, A.; Castner, D. Contemporary methods for characterizing complex biomaterial surfaces. In Biologically Modified Polymeric Biomaterial Surfaces; Springer: Dordrecht, The Netherlands, 1992; pp. 25–36. [Google Scholar]
- Zhang, Y.; Huang, Z.-M.; Xu, X.; Lim, C.T.; Ramakrishna, S. Preparation of core− shell structured PCL-r-gelatin bi-component nanofibers by coaxial electrospinning. Chem. Mater. 2004, 16, 3406–3409. [Google Scholar] [CrossRef]
- Li, H.; Ke, Y.; Hu, Y. Polymer nanofibers prepared by template melt extrusion. J. Appl. Polym. Sci. 2006, 99, 1018–1023. [Google Scholar] [CrossRef]
- Unser, A.M.; Xie, Y. Electrospinning of nanofibers. The Nanobiotechnology Handbook; CRC Press: Boca Raton, FL, USA, 2012; pp. 293–320. [Google Scholar]
- Ma, P.X.; Zhang, R. Synthetic nano-scale fibrous extracellular matrix. J. Biomed. Mater. Res. Off. J. Soc. Biomater. Japan. Soc. Biomater. Aust. Soc. Biomater. 1999, 46, 60–72. [Google Scholar] [CrossRef]
- Kim, S.H.; Nam, Y.S.; Lee, T.S.; Park, W.H. Silk fibroin nanofiber. Electrospinning, properties, and structure. Polym. J. 2003, 35, 185–190. [Google Scholar] [CrossRef]
- Peresin, M.S.; Habibi, Y.; Zoppe, J.O.; Pawlak, J.J.; Rojas, O.J. Nanofiber composites of polyvinyl alcohol and cellulose nanocrystals: Manufacture and characterization. Biomacromolecules 2010, 11, 674–681. [Google Scholar] [CrossRef]
- Luzio, A.; Canesi, E.V.; Bertarelli, C.; Caironi, M. Electrospun polymer fibers for electronic applications. Materials 2014, 7, 906–947. [Google Scholar] [CrossRef]
- Srivastava, Y.; Marquez, M.; Thorsen, T. Multijet electrospinning of conducting nanofibers from microfluidic manifolds. J. Appl. Polym. Sci. 2007, 106, 3171–3178. [Google Scholar] [CrossRef]
- Prabhakaran, M.P.; Ghasemi-Mobarakeh, L.; Jin, G.; Ramakrishna, S. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells. J. Biosci. Bioeng. 2011, 112, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Nune, M.; Kumaraswamy, P.; Maheswari Krishnan, U.; Sethuraman, S. Self-assembling peptide nanofibrous scaffolds for tissue engineering: Novel approaches and strategies for effective functional regeneration. Curr. Protein Peptide Sci. 2013, 14, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Mohammadzadehmoghadam, S.; Dong, Y.; Barbhuiya, S.; Guo, L.; Liu, D.; Umer, R.; Qi, X.; Tang, Y. Electrospinning: Current status and future trends. Nano-size Polym. 2016, 10, 89–154. [Google Scholar]
- Zhang, Y.; Rutledge, G.C. Electrical conductivity of electrospun polyaniline and polyaniline-blend fibers and mats. Macromolecules 2012, 45, 4238–4246. [Google Scholar] [CrossRef]
- McCullen, S.D.; Stevens, D.R.; Roberts, W.A.; Ojha, S.S.; Clarke, L.I.; Gorga, R.E. Morphological, electrical, and mechanical characterization of electrospun nanofiber mats containing multiwalled carbon nanotubes. Macromolecules 2007, 40, 997–1003. [Google Scholar] [CrossRef]
- Chronakis, I.S.; Grapenson, S.; Jakob, A. Conductive polypyrrole nanofibers via electrospinning: Electrical and morphological properties. Polymer 2006, 47, 1597–1603. [Google Scholar] [CrossRef]
- Nagata, S.; Atkinson, G.M.; Pestov, D.; Tepper, G.C.; Mcleskey, J.T. Electrospun polymer-fiber solar cell. Adv. Mater. Sci. Eng. 2011, 2013, 1000. [Google Scholar] [CrossRef]
- Babel, A.; Li, D.; Xia, Y.; Jenekhe, S.A. Electrospun nanofibers of blends of conjugated polymers: Morphology, optical properties, and field-effect transistors. Macromolecules 2005, 38, 4705–4711. [Google Scholar] [CrossRef]
- Balderas, U.; Falcony, C.; Moggio, I.; Arias, E.; Mondragón, M. A photoluminescence study of electrospun fibers of conjugated poly [2-methoxy-5-(2′-ethylhexyloxy)-1, 4-phenylenevinylene] blended with poly (9-vinylcarbazole). Polymer 2013, 54, 2062–2066. [Google Scholar] [CrossRef]
- Wong, S.-C.; Baji, A.; Leng, S. Effect of fiber diameter on tensile properties of electrospun poly (ε-caprolactone). Polymer 2008, 49, 4713–4722. [Google Scholar] [CrossRef]
- Shin, M.K.; Kim, S.I.; Kim, S.J.; Kim, S.-K.; Lee, H.; Spinks, G.M. Size-dependent elastic modulus of single electroactive polymer nanofibers. Appl. Phys. Lett. 2006, 89, 231929. [Google Scholar] [CrossRef]
- Yuan, B.; Wang, J.; Han, R.P. Capturing tensile size-dependency in polymer nanofiber elasticity. J. Mechan. Behav. Biomed. Mater. 2015, 42, 26–31. [Google Scholar] [CrossRef]
- Wingert, M.C.; Jiang, Z.; Chen, R.; Cai, S. Strong size-dependent stress relaxation in electrospun polymer nanofibers. J. Appl. Phys. 2017, 121, 015103. [Google Scholar] [CrossRef]
- Arinstein, A.; Burman, M.; Gendelman, O.; Zussman, E. Effect of supramolecular structure on polymer nanofibre elasticity. Nat. Nanotechnol. 2007, 2, 59–62. [Google Scholar] [CrossRef]
- Wu, X.-F.; Dzenis, Y.A. Size effect in polymer nanofibers under tension. J. Appl. Phys. 2007, 102, 044306. [Google Scholar] [CrossRef]
- Dingreville, R.; Qu, J.; Cherkaoui, M. Surface free energy and its effect on the elastic behavior of nano-sized particles, wires and films. J. Mechan. Phys. Solids 2005, 53, 1827–1854. [Google Scholar] [CrossRef]
- Cuenot, S.; Frétigny, C.; Demoustier-Champagne, S.; Nysten, B. Surface tension effect on the mechanical properties of nanomaterials measured by atomic force microscopy. Phys. Rev. 2004, 69, 165410. [Google Scholar] [CrossRef]
- Shin, M.K.; Kim, S.H.; Jung, S.-i.; Kim, S.I.; Kim, S.J.; Kim, B.J.; So, I. The effect of DNA on mechanical properties of nanofiber hydrogels. Appl. Phys. Lett. 2008, 93, 171903. [Google Scholar] [CrossRef]
- Stachewicz, U.; Bailey, R.J.; Wang, W.; Barber, A.H. Size dependent mechanical properties of electrospun polymer fibers from a composite structure. Polymer 2012, 53, 5132–5137. [Google Scholar] [CrossRef]
- Boas, M.; Gradys, A.; Vasilyev, G.; Burman, M.; Zussman, E. Electrospinning polyelectrolyte complexes: pH-responsive fibers. Soft Matt. 2015, 11, 1739–1747. [Google Scholar] [CrossRef]
- Zander, N.E. Hierarchically structured electrospun fibers. Polymers 2013, 5, 19–44. [Google Scholar] [CrossRef]
- Huang, C.; Chen, S.; Reneker, D.H.; Lai, C.; Hou, H. High-strength mats from electrospun poly (p-phenylene biphenyltetracarboximide) nanofibers. Adv. Mater. 2006, 18, 668–671. [Google Scholar] [CrossRef]
- Lee, K.-H.; Kim, K.-W.; Pesapane, A.; Kim, H.-Y.; Rabolt, J.F. Polarized FT-IR study of macroscopically oriented electrospun nylon-6 nanofibers. Macromolecules 2008, 41, 1494–1498. [Google Scholar] [CrossRef]
- Moon, S.; Farris, R.J. The morphology, mechanical properties, and flammability of aligned electrospun polycarbonate (PC) nanofibers. Polym. Eng. Sci. 2008, 48, 1848–1854. [Google Scholar]
- Moon, S.; Farris, R.J. Strong electrospun nanometer-diameter polyacrylonitrile carbon fiber yarns. Carbon 2009, 47, 2829–2839. [Google Scholar] [CrossRef]
- Inai, R.; Kotaki, M.; Ramakrishna, S. Structure and properties of electrospun PLLA single nanofibres. Nanotechnology 2005, 16, 208. [Google Scholar] [CrossRef]
- Li, Y.; Lim, C.T.; Kotaki, M. Study on structural and mechanical properties of porous PLA nanofibers electrospun by channel-based electrospinning system. Polymer 2015, 56, 572–580. [Google Scholar] [CrossRef]
- Kongkhlang, T.; Tashiro, K.; Kotaki, M.; Chirachanchai, S. Electrospinning as a new technique to control the crystal morphology and molecular orientation of polyoxymethylene nanofibers. J. Am. Chem. Soc. 2008, 130, 15460–15466. [Google Scholar] [CrossRef]
- Hohman, M.M.; Shin, M.; Rutledge, G.; Brenner, M.P. Electrospinning and electrically forced jets. I. Stability theory. Phys. Fluids 2001, 13, 2201–2220. [Google Scholar] [CrossRef]
- Hohman, M.M.; Shin, M.; Rutledge, G.; Brenner, M.P. Electrospinning and electrically forced jets. II. Applications. Phys. Fluids 2001, 13, 2221–2236. [Google Scholar] [CrossRef]
- Gañán-Calvo, A.M. Cone-jet analytical extension of Taylor’s electrostatic solution and the asymptotic universal scaling laws in electrospraying. Phys. Rev. Lett. 1997, 79, 217. [Google Scholar] [CrossRef]
- Feng, J. The stretching of an electrified non-Newtonian jet: A model for electrospinning. Phys. Fluids 2002, 14, 3912–3926. [Google Scholar] [CrossRef]
- Hea, J.-H. A brief review on mathematical models for electrospinning. In Proceedings of the Third MIT Conference on Computational Fluid and Solid Mechanics, Cambridge, MA, USA, 14–17 June 2005; Elsevier Ltd.: Amsterdam, The Netherlands, 2005; pp. 665–667. [Google Scholar]
- Spivak, A.; Dzenis, Y.; Reneker, D. Model of steady state jet in the electrospinning process. Mechan. Res. Commun. 2000, 27, 37–42. [Google Scholar] [CrossRef]
- Wan, Y.-Q.; Guo, Q.; Pan, N. Thermo-electro-hydrodynamic model for electrospinning process. Int. J. Nonlinear Sci. Num. Simul. 2004, 5, 5–8. [Google Scholar] [CrossRef]
- He, J.H.; Xu, L.; Wu, Y.; Liu, Y. Mathematical models for continuous electrospun nanofibers and electrospun nanoporous microspheres. Polym. Int. 2007, 56, 1323–1329. [Google Scholar] [CrossRef]
- He, J.-H. Allometric scaling law in conductive polymer. Polymer 2004, 45, 9067–9070. [Google Scholar] [CrossRef]
- Oliveira, J.E.; Mattoso, L.H.; Orts, W.J.; Medeiros, E.S. Structural and morphological characterization of micro and nanofibers produced by electrospinning and solution blow spinning: A comparative study. Adv. Mater. Sci. Eng. 2013, 2013, 409572. [Google Scholar] [CrossRef]
- Rahmati, M.; Mills, D.K.; Urbanska, A.M.; Saeb, M.R.; Venugopal, J.R.; Ramakrishna, S.; Mozafari, M. Electrospinning for tissue engineering applications. Progr. Mater. Sci. 2021, 117, 100721. [Google Scholar] [CrossRef]
- Preethi, G.; Joseph, M.; Unnikrishnan, B.; Shiji, R.; Sreelekha, T. Biomedical applications of natural polymer based nanofibrous scaffolds. Int. J. Med. Nano Res. 2015, 2, 9. [Google Scholar]
- Rim, N.G.; Shin, C.S.; Shin, H. Current approaches to electrospun nanofibers for tissue engineering. Biomed. Mater. 2013, 8, 014102. [Google Scholar] [CrossRef]
- Patel, S.; Kurpinski, K.; Quigley, R.; Gao, H.; Hsiao, B.S.; Poo, M.-M.; Li, S. Bioactive nanofibers: Synergistic effects of nanotopography and chemical signaling on cell guidance. Nano Lett. 2007, 7, 2122–2128. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, K.; Agatemor, C.; Špitálsky, Z.; Thomas, S.; Kny, E. Electrospinning tissue engineering and wound dressing scaffolds from polymer-titanium dioxide nanocomposites. Chem. Eng. J. 2019, 358, 1262–1278. [Google Scholar] [CrossRef]
- Zahmatkeshan, M.; Gheybi, F.; Rezayat, S.M.; Jaafari, M.R. Improved drug delivery and therapeutic efficacy of PEgylated liposomal doxorubicin by targeting anti-HER2 peptide in murine breast tumor model. Eur. J. Pharm. Sci. 2016, 86, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Asyikin binti Abdul Aziz, Z.; Ahmad, A.; Hamidah Mohd-Setapar, S.; Hassan, H.; Lokhat, D.; Amjad Kamal, M. Recent advances in drug delivery of polymeric nano-micelles. Curr. Drug Metabol. 2017, 18, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J. Control. Release 2021, 334, 463–484. [Google Scholar] [CrossRef] [PubMed]
- Zelkó, R.; Lamprou, D.A.; Sebe, I. Recent Development Of Electrospinning For Drug Delivery; MDPI: Basel, Switzerland, 2019; Volume 12, p. 5. [Google Scholar]
- Halicka, K.; Cabaj, J. Electrospun nanofibers for sensing and biosensing applications—A review. Int. J. Mol. Sci. 2021, 22, 6357. [Google Scholar] [CrossRef] [PubMed]
- Aussawasathien, D.; Dong, J.-H.; Dai, L. Electrospun polymer nanofiber sensors. Synth. Met. 2005, 154, 37–40. [Google Scholar] [CrossRef]
- Yilmaz, O.E.; Erdem, R. Evaluating hydrogen detection performance of an electrospun CuZnFe2O4 nanofiber sensor. Int. J. Hydr. Energy 2020, 45, 26402–26412. [Google Scholar] [CrossRef]
- Wang, W.; Huang, H.; Li, Z.; Zhang, H.; Wang, Y.; Zheng, W.; Wang, C. Zinc oxide nanofiber gas sensors via electrospinning. J. Am. Ceramic Soc. 2008, 91, 3817–3819. [Google Scholar] [CrossRef]
- Aliheidari, N.; Aliahmad, N.; Agarwal, M.; Dalir, H. Electrospun nanofibers for label-free sensor applications. Sensors 2019, 19, 3587. [Google Scholar] [CrossRef]
- Ding, B.; Wang, M.; Wang, X.; Yu, J.; Sun, G. Electrospun nanomaterials for ultrasensitive sensors. Mater. Today 2010, 13, 16–27. [Google Scholar] [CrossRef]
- Zhu, M.; Han, J.; Wang, F.; Shao, W.; Xiong, R.; Zhang, Q.; Pan, H.; Yang, Y.; Samal, S.K.; Zhang, F. Electrospun nanofibers membranes for effective air filtration. Macromol. Mater. Eng. 2017, 302, 1600353. [Google Scholar] [CrossRef]
- Kandasubramanian, B.; Ramdayal, M. Advancement in textile technology for defence application. Defence Sci. J. 2013, 63, 331. [Google Scholar] [CrossRef]
- Ramaseshan, R.; Sundarrajan, S.; Liu, Y.; Barhate, R.; Lala, N.L.; Ramakrishna, S. Functionalized polymer nanofibre membranes for protection from chemical warfare stimulants. Nanotechnology 2006, 17, 2947. [Google Scholar] [CrossRef]
- Thenmozhi, S.; Dharmaraj, N.; Kadirvelu, K.; Kim, H.Y. Electrospun nanofibers: New generation materials for advanced applications. Mater. Sci. Eng. 2017, 217, 36–48. [Google Scholar] [CrossRef]
- Shi, X.; Zhou, W.; Ma, D.; Ma, Q.; Bridges, D.; Ma, Y.; Hu, A. Electrospinning of nanofibers and their applications for energy devices. J. Nanomater. 2015, 2015, 124. [Google Scholar] [CrossRef]

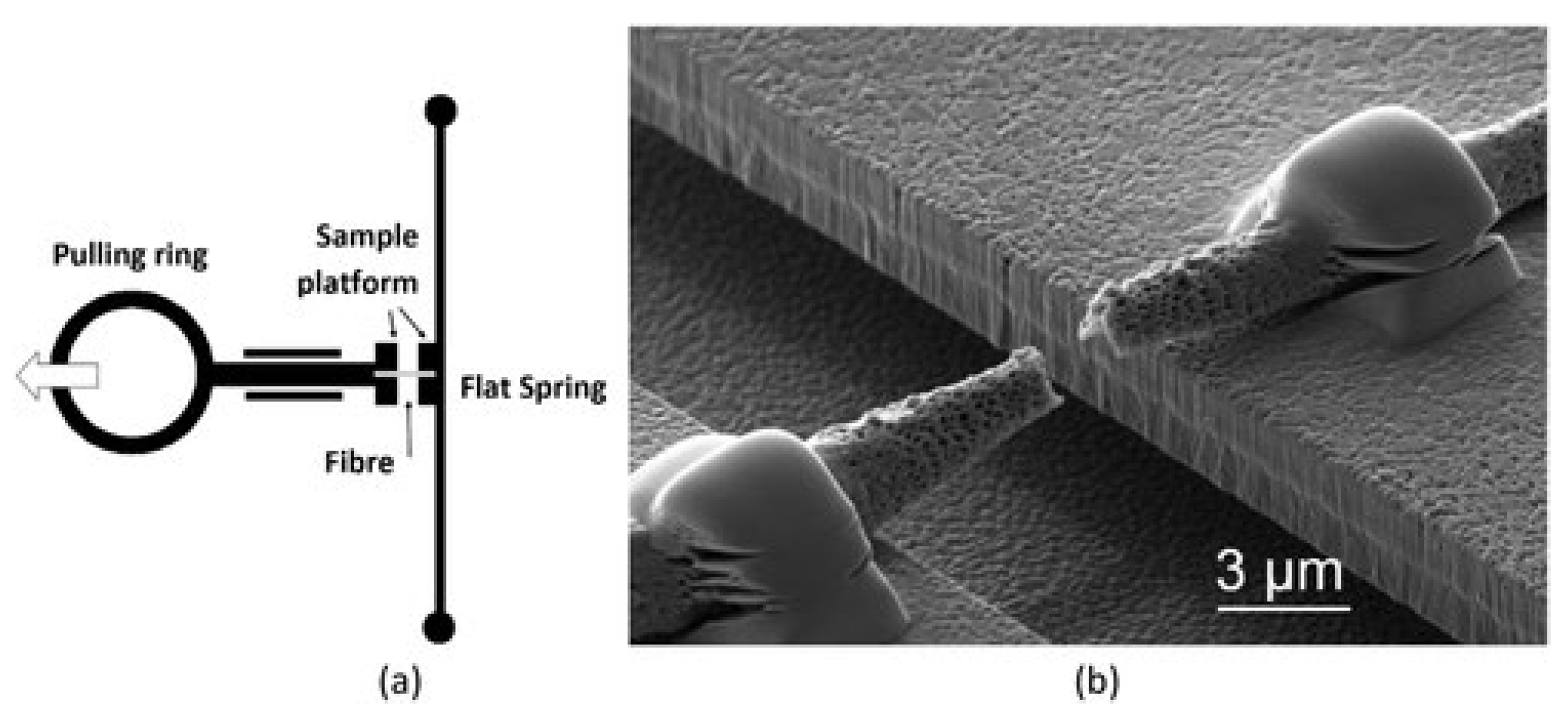
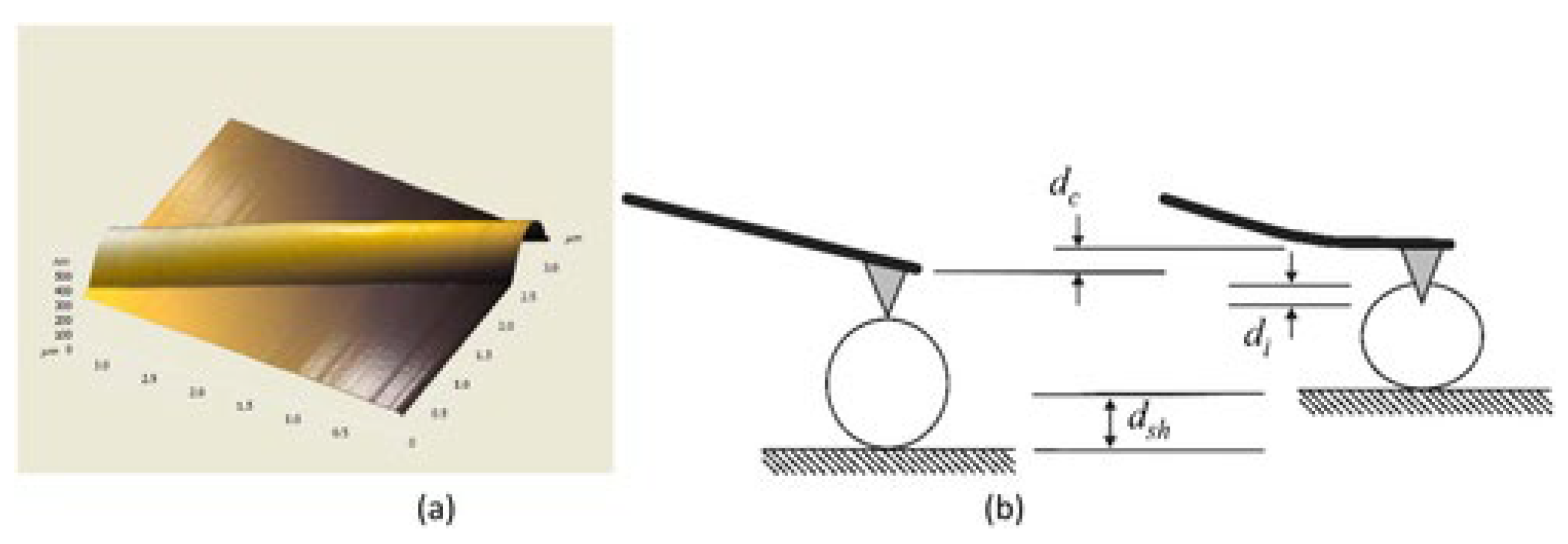
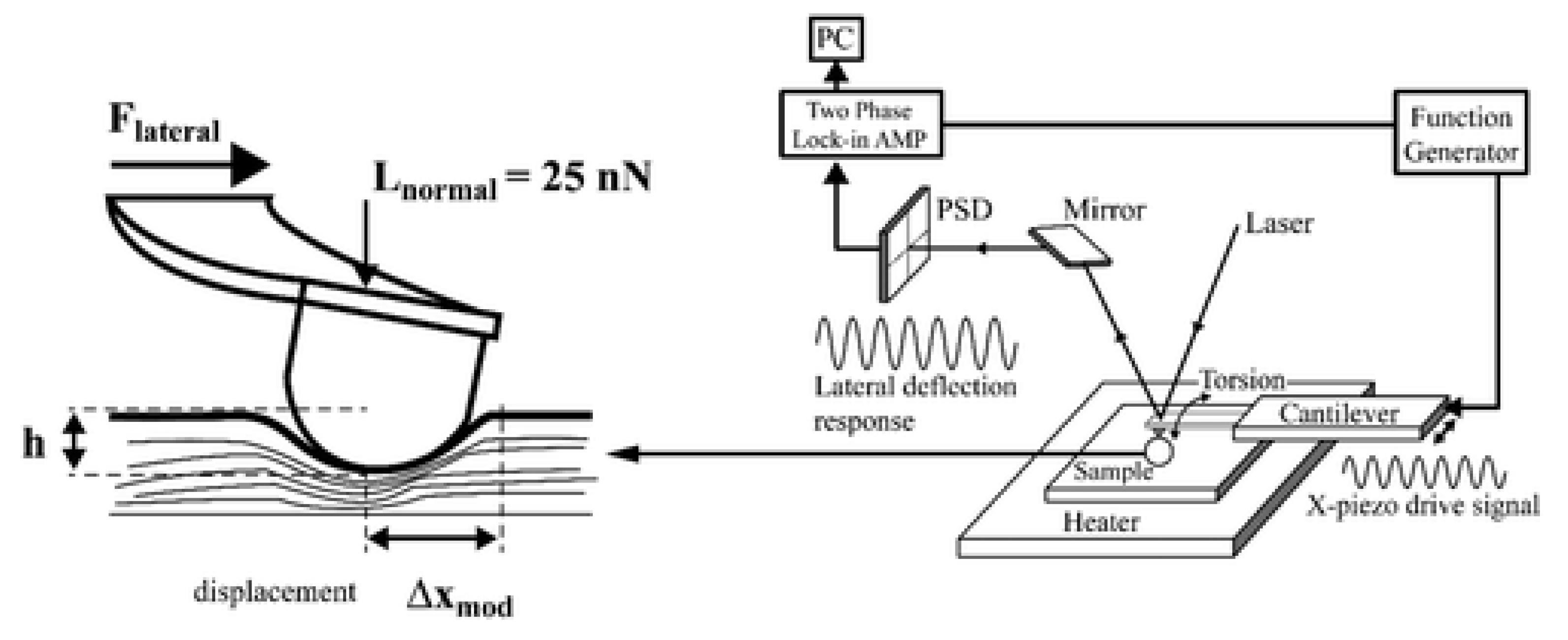
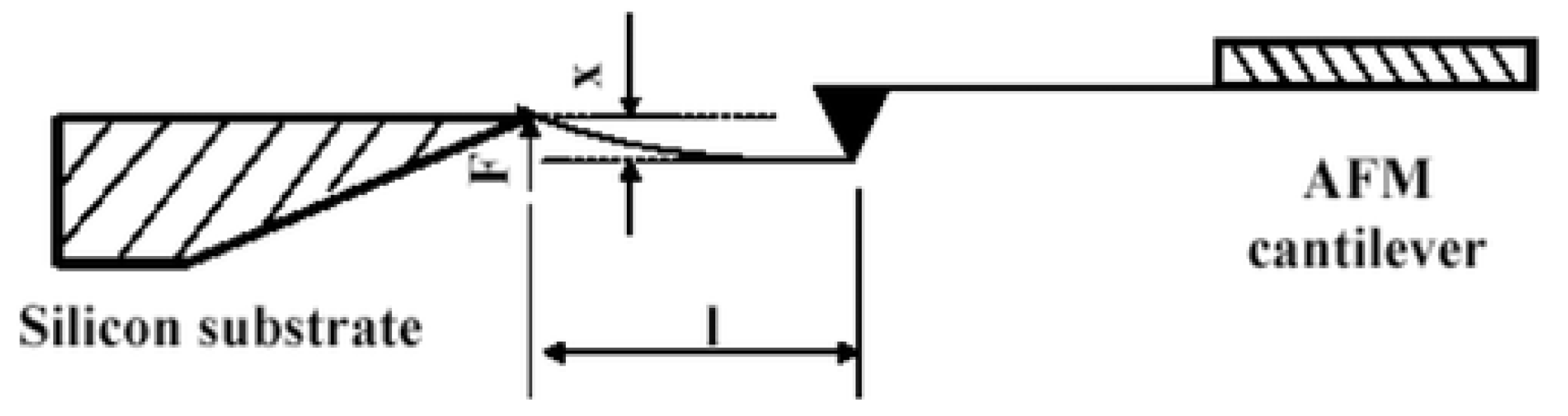






| Technique | Principle | Pot 1 | Rep 2 | FDC 3 | Advantages (+)/Limitations (−) | Refs. |
|---|---|---|---|---|---|---|
| Phase separation | Polymer rich and poor phases are generated, elimination of poor phase leads to generation of nanofibrous structure | Lab | Yes | Yes | + Consistent for batch-to-batch production, can adjust props by varying concentration + Easy − Few polymers can be processed | [13,14] |
| Drawing | Pulling, followed by solidification, of dissolved spun material to solid material | Lab | Yes | No | + Less equipment required for processing + Easy processing − Discontinuous, fibers generated sequentially | [15] |
| Self-assembly | Used to produce peptide nanofibers | Lab | Yes | No | − Few polymers can be processed −complex − Difficult | [16] |
| Template synthesis | Nano-porous membrane is utilized as template with different materials (metals, carbon, conductive polymers etc.) | Lab | Yes | Yes | + Fibers of different diameters can be produced using various templates + Moderately easy − Applicable for few polymers | [17] |
| Electrospinning | Electric field to process micro/nanofibers from polymer in liquid solution of melts | Lab and comm. | Yes | Yes | + Simple, uses various polymers, produces long continuous nanofibers, feasible to generate aligned nanofibers + Moderately easy | [19,20] |
| Technique | Principle | Advantages/Limitations |
|---|---|---|
| Melt Electrospinning | Apparatus is similar to conventional electrospinning setup along with provision of melting of polymer. | + Environmentally friendly without need for ventilation system. + High throughput rate and easily processed polymeric fiber blends. + Suitable to electrospin non-soluble polymers (such as PE and PP). − Require high temperature melting system, electric discharge issues related to melt, and low conductivity of melt. [42] |
| Needleless/free surface electrospinning | Simultaneous generation of various jets from open liquid surface without capillary effect. | Classified into rotating and stationary needleless electrospinning. Morphology and production rates depend on shape of spinneret (conical wire coil, bowel edge, rotary cone, metal plate, splashing, and moving bead chain) + Production rates bet. 2.5–100 g/h are possible, up to 250 times greater than conventional spinning. − Difficult to control the spatial movement of multiple jets and inadequate stability of the free liquid. [43] |
| Multi-jet Electrospinning | Polymer is split up into distinct jets during trajectory. | Needles are arranged linearly or in 2D with triangular, circular, square, elliptical, and hexagonal arrays. + Upscale electrospinning process by utilizing multiple needles. − Interference of electric fields − Clog up of needles with polymer solutions. − Jet deviation causes instability issues, requiring auxiliary plate and extra electrode configurations. [44] |
| Electro-blowing (Gas Jet/Gas Assisted Electrospinning) | Air blowing shear force and electric field simultaneously applied to generation of nanofibers. | A spinneret with air nozzle generates nanofiber web to electrospin thick solutions where surface tension is very high. + Improved productivity + Hot gases can be utilized to reduce solution viscosity and increase the fiber elongation. [45] |
| Centrifugal Electrospinning | Combination of electrical and centrifugal forces in electrospinning process. | + Rotation range: 300–600 rpm, << centrifugal spinning range. + Improved polymer chain orientation and production. + Due to centrifugal effect, projection force on jet is higher, resulting in less flight time and improving the fiber stretching. + High production rate, easy maintenance. − Non continuous fibers, requires expensive equipment. [46] |
| Near-Field Electrospinning | In this technique, distance between substrate and nozzle is reduced few millimeters. | + Utilized for deposition of nanofibers at predictable location, which is not feasible by common electrospinning process. + Lower voltage (~ 200 V) is required to generate electric field to initiate spinning. + Improvement in morphology of nanofibers. [47] |
| Coaxial Electrospinning | Utilized two concentrically align capillaries to generate core shell structure. | + The setup can be configured in both vertical and horizontal direction. + It can generate hollow electrospun nanofibers. + Almost all polymer solutions and their composites’ matrices can be processed into core shell and hollow nanofibers. − Dimensional control is difficult, solvent evaporation issues. [48] |
| Emulsion electrospinning | Similar to solution electrospinning, but solution is substituted by emulsion with oil in water or water in oil. | + Used to generate core-shell fiber, it is a relatively simpler setup than co axial electrospinning. + Utilized for encapsulation of range of bioactive materials with various solubilities into polymeric nanofibers. + User friendly and economical technique. [49] |
| Polymers | Solvents | Applications | Refs. |
|---|---|---|---|
| Nylon6,6, PA-6,6 | Formic acid | Smart clothing | [60] |
| Polylactic acid | Dimethyl formamide Methylene chloride and dimethyl Formamide Dichloromethane | Membrane for medical use, sensor, filter, drug delivery system | [7,11] |
| Polybenzimidazole | Dimethyl accetamide | Protective clothing, nanofiber reinforced composites | [61] |
| Polycarboate | Dimethyl ormamide: tetrahydrofuran (1:1), Dichlormethane, chloroform, tetrahydrofuran Dimethylformamide: tetrahydrofuran (1:1) | Protective clothing, sensor, filter | [62] |
| Polyacrylonitrile | Dimethyl formamide | Carbon nanofiber | [63] |
| Polyurethanes | Dimethyl formamide Dimethylformamide | Protective clothing Electret filter | [64,65] |
| Polyvinil alcohol | Distilled water | [66] | |
| Polyethylene-co-vinyl acetate | Drug delivery system | [67] | |
| Polyethylene oxide | Distilled water Distilled water: ethanol (3:2) Isopropyle alcohol+water | Microelectronic wiring, interconnects Electret filter | [68] |
| Collagen–Polyethylene oxide | Hydrochloric acid Hydrochloric acid (pH = 2.0) | Wound healing, tissue engineering, hemostatic agents | [69] |
| Polyaniline/polyethylene oxide blend | Chloroform, camphorsulfonic acid | Conductive fiber | [70] |
| Polyaniline/polystyrene | Chloroform, camphorsulfonic acid | Conductive fiber | [71] |
| Silk-like polymer with fibronectin functionality | Formic acid | Implantable device | [72] |
| Polyvinylcarbazole | Polyvinylcarbazole | Sensor, filter | [73] |
| Polystyrene | Tetrahydrofuran, dimethylformamide, CS2 (carbon disulfide), toluene, Methylethylketone, Dimethylformamide Tetrahydrofuran | Enzymatic biotransformation, Flat ribbons, catalyst, filter | [11] |
| Polyamide | Dimethylacetamide | Glass fiber filter media | [74] |
| Silk/polyethylene oxide blend | Silk/PEO blend | Biomaterial scaffolds | [75] |
| Poly vinyl phenol | Tetrahydrofuran | Antimicrobial agent | [76] |
| Antimicrobial agent | Acetone, acetic acid, dimethylacetamide | Membrane | [77] |
| Mix of (polyacrylic acid -polypyrene methanol) and polyurethane | Dimethylformamide | Optical sensor | [78] |
| poly(lactic-co-glycolic acid) | Tetrahydrofuran: dimethylformamide (1:1) | Scaffold for tissue engineering | [79] |
| Collagen | Hexafluoro-2-propanol | Scaffold for tissue engineering | [80] |
| Poly (vinylidene fluoride) PVDF | Dimethylformamide: dimethylacetamide (1/1) | Flat ribbons | [81] |
| Nylon-4,6, PA-4,6 | Formic acid | Transparent composite | [82] |
| Transparent composite | Isopropanol/water: 70/30 (%v/v) | Biomedical | [83] |
| Polyacrylnitrile/TiO2 | Photovoltaic and conductive polymers | [84] | |
| Polycaprolactone/metal | ZnO: cosmetic use | [85] |
| Different Parameters | Effect on Morphology |
|---|---|
| ↑ Polymer concentration | ↑ Fiber diameter if > 15 wt.% (with in optimal range) ↓ Bead formation |
| ↑ Molecular weight | ↓ Droplet and bead formation Irregularity in shape with larger pores |
| ↑ Volatility of solvent | Pores generated on surface of fiber (macrotexture) |
| ↑ Solution conductivity | ↓ Fiber diameter (broad distribution of diameter) and uniform bead free fibers |
| ↑ Distance between collector and capillary | ↓ Fiber diameter, for generation of uniform fiber optimal distance, i.e., 30 cm is required, too short or too long cause bead formation |
| ↑ Voltage applied | ↓ Fiber diameter initially, then ↑ (not monotonic) |
| ↑ Feed rate | ↑ Fiber diameter and, if too high, bead formation occurs |
| ↑ Temperature | ↓ Fiber diameter and ↓ viscosity |
| ↑ Air velocity | ↑ Fiber diameter |
| ↑ Humidity | Produce circular pores on fiber |
| Type of Portable Devices | Advantages/Limitations |
|---|---|
| Hand-held spinnerets | + Flexible in operation, precise deposition of fibers, accuracy in flow rate, in situ spinning. − Electricity dependent, expensive. |
| Generator powered | + Power supply, in situ spinning, flexible in operation, affordable. − Unstable voltage, not precise deposition, limited flow rate. |
| Battery powered | + Precise deposition, flexible in operation, precise deposition, affordable. − Battery capacity issues, high voltage limitation. |
| Application | Action/Mechanism |
|---|---|
| Tissue Engineering | NFs act as scaffolding for tissue regeneration and growth. |
| Wound Healing and Dressing | NF porosity provides space for cell migration and regeneration and increases oxygen exchange, in addition to reinforcement. |
| Drug Delivery Systems | Porosity, large aspect ratio, and cross-linking give rise to drug-carrying capacity at minute volumes for directed delivery. |
| Sensors and Biosensors | The NF chemically reacts to the change and activates an energetic signal to be detected and recorded. |
| Air Filtration | NF high cross-linking and spatial structure provide ability to capture microparticles and filter the environment. |
| Defense: protection from warfare toxic gasses | NFs carry catalysts capturing toxic warfare chemical stimulants. Porosity and hydrophilicity of membranes qualify them as filter media. |
| Energy Devices | NFs are used as separators in fuel cells and large photoelectric conversion efficiency due to large specific area and porosity. |
| Product | Utilization | Manufacturer | Description | Websites 1 |
|---|---|---|---|---|
| Aeos | Wound Healing | Zeus Company Inc. | Consist of nonwoven fibrous for sutures | https://www.zeusinc.com/ |
| ResQFoam | Wound Healing | Arsenal Medical | Non-compressible hemorrhage treated by foam contained in core shell fiber | https://arsenalmedical.com/ |
| Rethink | Surgical mask | Stellenbosch Nanofiber Company | Specializing in commercial scale manufacture of advanced biomedical nanofiber materials | https://sncfibers.com/ |
| Spincare | Wound Healing | Nanomedic | Portable electrospinning equipment | https://nanomedic.com/ |
| Nexture | Textile | Lime Nano | Nanofibers membranes | https://limenano.com/ |
| NanoDream | Textile | NanoLayr | Nanofiber clothing | https://www.nanolayr.com/ |
| Wetlaid Fabrics | Textile | Hirose Paper Mfg. Co | Nanofiber Nonwoven Fabrics | https://www.hirose-paper-mfg.co.jp/ |
| BreaSAFE | Air Filtration | Nano4Fibers | Masks are made of nanofibers to protect from microbes, toxic gases, dust, and order. | https://www.nano4fibers.com/ |
| FilterLayr | Air Filtration | Nanolayr | Air filtration unit | https://www.nanolayr.com/ |
| Exceed | Air Filtration | Espin Technologies, Inc. | Nanofiber membranes for air filtrations | https://espintechnologies.com/ |
| ProTura | Water treatment | Parker | Nanofibers cellulose filtration assembly | https://www.parker.com/ |
| Nanofiber Filter | Water Treatment | Astral Pool | Self-cleaning filters | https://www.astralpool.com/ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Abduljabbar, A.; Farooq, I. Electrospun Polymer Nanofibers: Processing, Properties, and Applications. Polymers 2023, 15, 65. https://doi.org/10.3390/polym15010065
Al-Abduljabbar A, Farooq I. Electrospun Polymer Nanofibers: Processing, Properties, and Applications. Polymers. 2023; 15(1):65. https://doi.org/10.3390/polym15010065
Chicago/Turabian StyleAl-Abduljabbar, Abdulhamid, and Irfan Farooq. 2023. "Electrospun Polymer Nanofibers: Processing, Properties, and Applications" Polymers 15, no. 1: 65. https://doi.org/10.3390/polym15010065
APA StyleAl-Abduljabbar, A., & Farooq, I. (2023). Electrospun Polymer Nanofibers: Processing, Properties, and Applications. Polymers, 15(1), 65. https://doi.org/10.3390/polym15010065







