Programmable Transition between Adhesive/Anti-Adhesive Performances on Porous PVDF Spheres Supported by Shape Memory PLLA
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of Sample
2.3. Microstructure Characterization
2.4. Drop Shape Analysis (DSA)
2.5. Dynamic Mechanical Analysis (DMA)
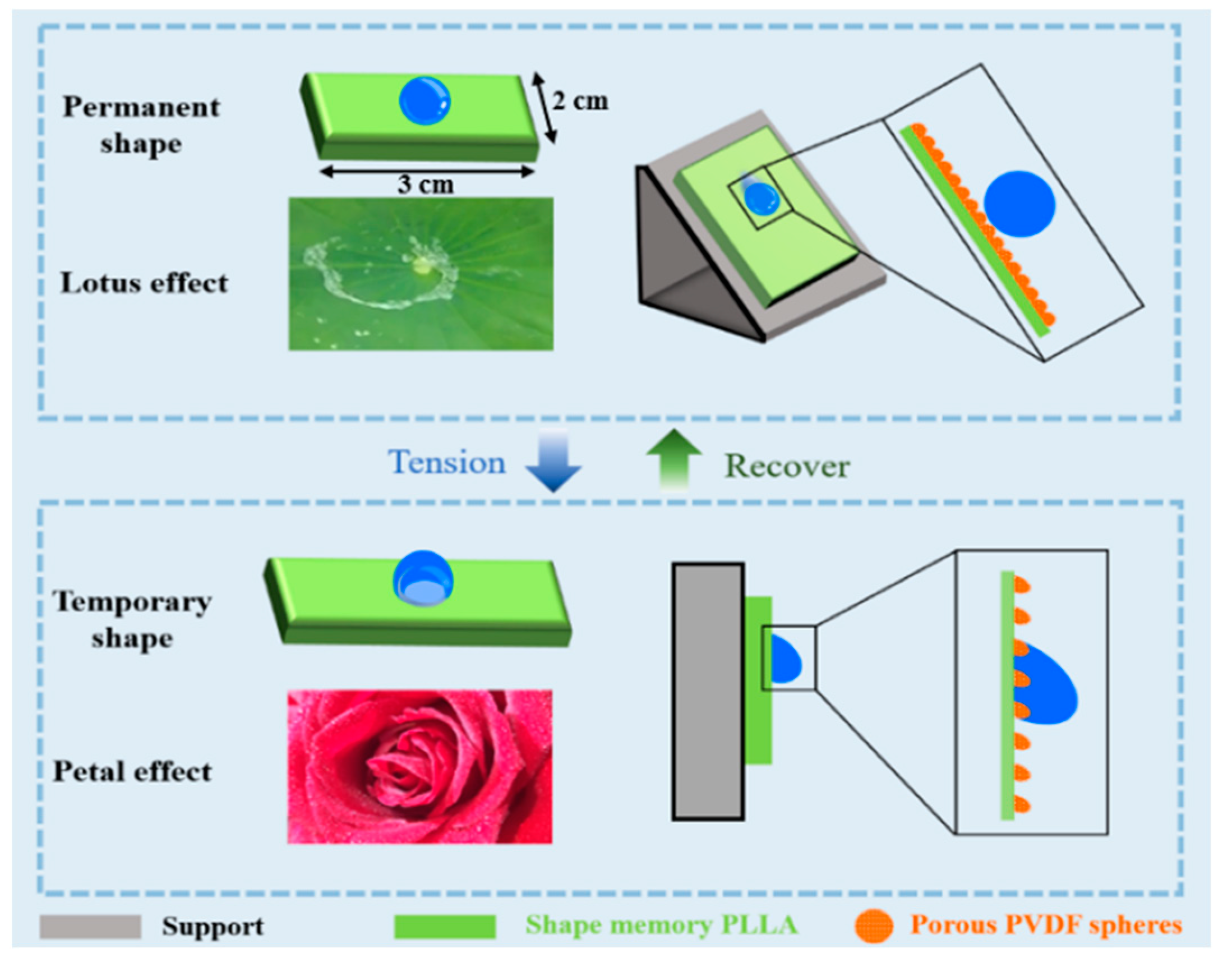
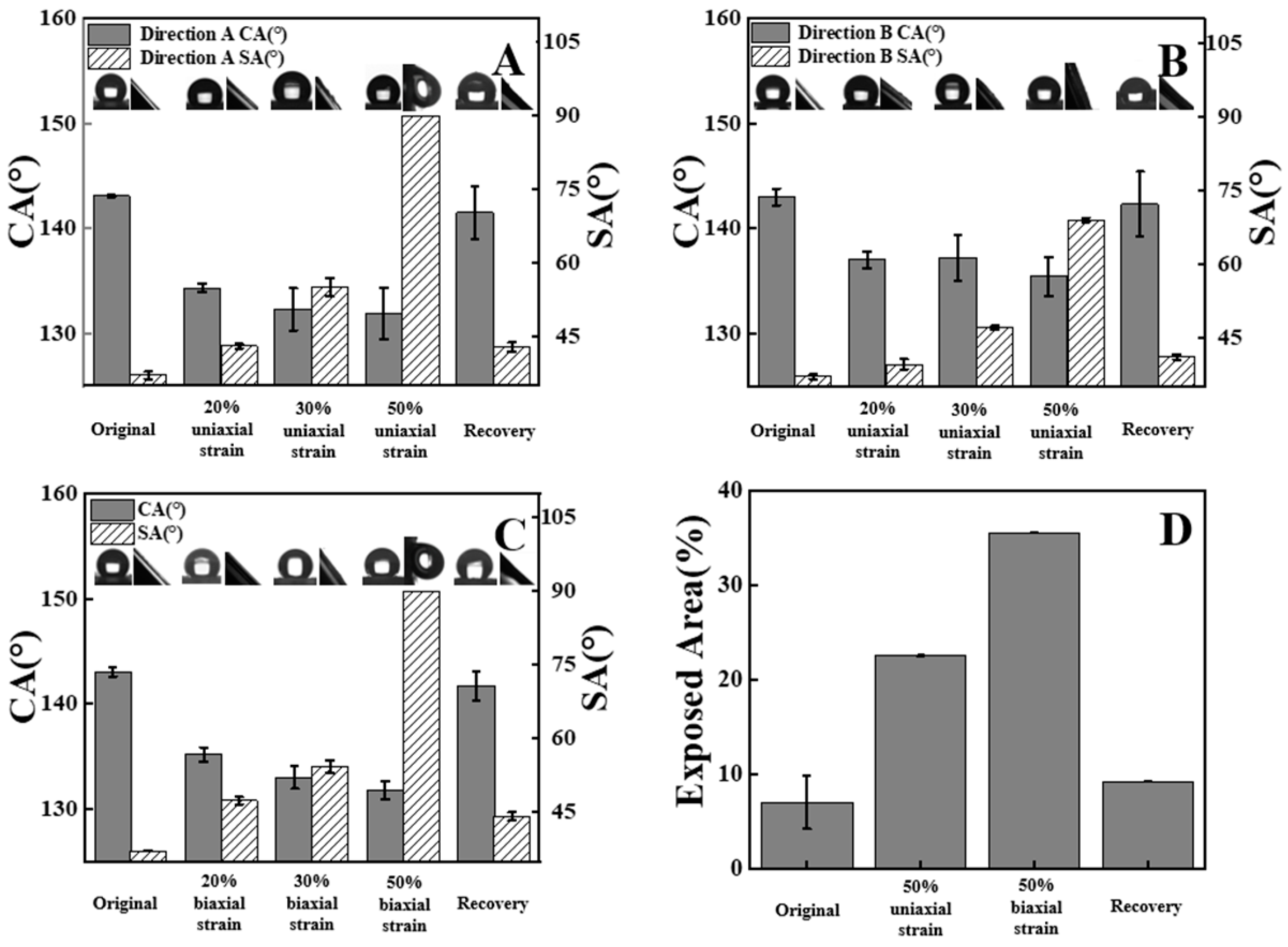
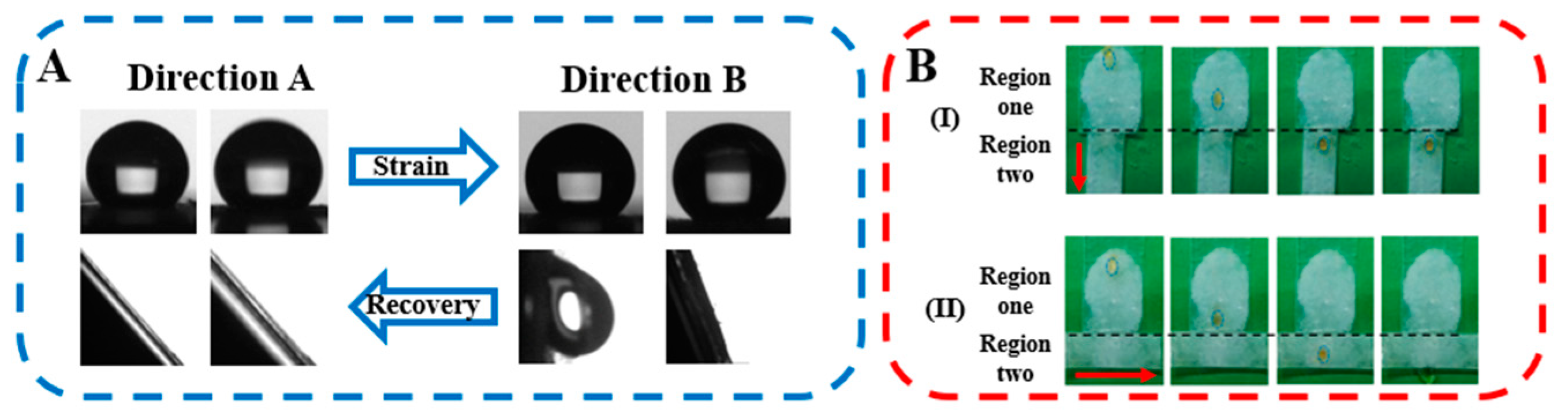
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, Y.; Gao, Y.; Zhang, C.; Zhao, X.; Ma, Y.; Du, F. Static and dynamic wetting behavior of TX-100 solution on superhydrophobic rice (oryzasativa.) leaf surfaces. Colloids Surfaces A 2018, 547, 148–156. [Google Scholar] [CrossRef]
- Fang, Y.; Sun, G.; Bi, Y.; Zhi, H. Multiple-dimensional micro/nano structural models for hydrophobicity of butterfly wing surfaces and coupling mechanism. Sci. Bull. 2015, 60, 256–263. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Yang, X.; Yang, Z. Study on micro and nano structure and superhydrophobicity for several typical planning animals legs. Appl. Mech. Mater. 2014, 459, 551–554. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, F.; Niu, J.; Jiang, Y.; Wang, Z. Superhydrophobic surfaces: From structural control to functional application. J. Mater. Chem. 2008, 18, 621–633. [Google Scholar] [CrossRef]
- Shao, Y.; Zhao, J.; Fan, Y.; Wan, Z.; Lu, L.; Zhang, Z.; Ming, W.; Ren, L. Shape memory superhydrophobic surface with switchable transition between “Lotus Effect” to “rose petals effect”. Chem. Eng. J. 2020, 382, 122989. [Google Scholar] [CrossRef]
- Farhadi, S.; Farzaneh, M.; Kulinich, S.A. Anti-icing performance of superhydrophobic surfaces. Appl. Surf. Sci. 2011, 257, 6264–6269. [Google Scholar] [CrossRef]
- Wang, Z.; Yuan, L.; Wang, L.; Wu, T. Stretchable superlyophobic surfaces for nearly-lossless droplet transfer. Sens. Actuators B 2017, 244, 649–654. [Google Scholar] [CrossRef]
- Fürstner, R.; Barthlott, W. Wetting and self-cleaning properties of artificial superhydrophobic surfaces. Langmuir 2005, 21, 956–961. [Google Scholar] [CrossRef] [PubMed]
- Lv, T.; Cheng, Z.; Zhang, D.; Zhang, E.; Zhao, Q.; Liu, Y.; Jiang, L. Superhydrophobic surface with shape memory micro/nanostructure and its application rewritable chip for droplet storage. ACS Nano 2016, 10, 9379–9386. [Google Scholar] [CrossRef]
- Cho, E.C.; Chang-Jian, C.W.; Chen, H.C.; Chuang, K.S.; Zheng, J.H.; Hsiao, Y.S.; Lee, K.C.; Huang, J.H. Robust multifunctional superhydrophobic coatings with enhanced water/oil separation, self-cleaning, anti-corrosion, and anti-biological adhesion. Chem. Eng. J. 2017, 314, 347–357. [Google Scholar] [CrossRef]
- Wu, B.; Cui, X.; Jiang, H.; Wu, N.; Peng, C.; Hu, Z.; Liang, X.; Yan, Y.; Huang, J.; Li, D. A superhydrophobic coating harvesting mechanical robustness, passive anti-icing and active deicingper formances. J. Colloid Interface Sci. 2021, 590, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Gao, Y.; Liu, N.; Feng, L.; Jiang, L. Special wettable materials for oil/water separation. J. Mater. Chem. A 2014, 2, 2445–2460. [Google Scholar] [CrossRef]
- Sun, T.; Feng, L.; Gao, X.; Jiang, L. Bioinspired surfaces with special wettability. Acc. Chem. Res. 2005, 38, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.; Wang, Y.; Lv, T.; Li, L.; Cheng, Z.; Liu, Y. Bio-inspired design of hierarchical PDMS microstructures with tunable adhesive superhydrophobicity. Nanoscale 2015, 7, 6151–6158. [Google Scholar] [CrossRef]
- Feng, L.; Zhang, Y.; Xi, J.; Zhu, Y.; Wang, N.; Xia, F.; Jiang, L. Petal effect: A superhydrophobic state with high adhesive force. Langmuir 2008, 24, 4114–4119. [Google Scholar] [CrossRef] [PubMed]
- Marmur, A. The Lotus Effect: Superhydrophobicity and metastability. Langmuir 2004, 20, 3517–3519. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Xi, Y.; Jiang, L. Recent developments in bio-inspired special wettability. Chem. Soc. Rev. 2010, 39, 3240–3255. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wei, J.; Su, Z. Fabrication of surfaces with extremely high contact angle hysteresis from polyelectrolyte multilayer. Langmuir 2011, 27, 15299–15304. [Google Scholar] [CrossRef]
- Gao, D.; Gao, J.; Guo, Z. Underwater manipulation of oil droplets and bubbles on superhydrophobic surfaces via switchable adhesion. Chem. Commun. 2019, 55, 3394–3397. [Google Scholar] [CrossRef]
- Wang, Y.; Lai, H.; Cheng, Z.; Zhang, H.; Liu, Y.; Jiang, L. Smart superhydrophobic shape memory adhesive surface toward selective capture/release of microdroplets. ACS Appl. Mater. Interfaces 2019, 11, 10988–10997. [Google Scholar] [CrossRef]
- Cheng, Z.; Zhang, D.; Lv, T.; Lai, H.; Zhang, E.; Kang, H.; Wang, Y.; Liu, P.; Liu, Y.; Du, Y.; et al. Superhydrophobic shape memory polymer arrays with switchable isotropic/anisotropic wetting. Adv. Funct. Mater. 2018, 28, 1705002. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Shen, J.; Ye, L.; Xie, K.; You, J.; Li, Y. Reversible transition between adhesive and antiadhesive performances by stretching/recovery on superhydrophobic TPU/CNTs composite membrane surface. Appl. Surf. Sci. 2019, 471, 900–903. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, X.; Li, Y.; You, J. Microsphere with narrow nanopores: Fabrication in PVDF/PMMA/PLLA blend and enhanced adsorption/separation performances. Appl. Surf. Sci. 2021, 566, 150673. [Google Scholar] [CrossRef]
- Ye, C.; Zhao, J.; Ye, L.; Jiang, Z.; You, J.; Li, Y. Precise inter-lamellar/inter-fibrillar localization and consequent fabrication of porous membranes with crystallization-modulated pore-size. Polymers 2018, 142, 48–51. [Google Scholar] [CrossRef]
- Wang, J.; Ding, M.; Cheng, X.; Ye, C.; Li, F.; Li, Y.; You, J. Hierarchically porous membranes with isolated-round-pores connected by narrow-nanopores: A novel solution for trade-off effect in separation. J. Polym. Sci. 2020, 604, 118040. [Google Scholar] [CrossRef]
- Ye, C.; Cao, X.; Wang, H.; Wang, J.; Wang, T.; Wang, Z.; Li, Y.; You, J. Inter-spherulitic/inner-spherulitic localization of PBSU during crystallization of PVDF in PVDF/PBSU blend. J. Polym. Sci. 2020, 58, 1699–1706. [Google Scholar] [CrossRef]
- Zhao, D.; Zuo, J.; Lu, K.; Chung, T. Fluorographite modified PVDF membranes for seawater desalination via direct contact membrane distillation. Desalination 2017, 413, 119–126. [Google Scholar] [CrossRef]
- Kusumaa, N.; Purwantoa, M.; Sudrajata, M.; Jaafarb, J.; Othmanb, M.M.; Azizb, Y.; Raharjoc, M. Qtaishat, Fabrication and characterization of modified PVDF hollow fiber membrane coated with hydrophobic surface modifying macromolecules for desalination application. J. Environ. Chem. Eng. 2021, 9, 105582. [Google Scholar] [CrossRef]
- Li, H.; Shi, W.; Zeng, X.; Hung, S.; Zhang, H.; Qin, X. Improved desalination properties of hydrophobic GO-incorporated PVDF electrospun nanofibrous composites for vacuum membrane distillation. Sep. Purif. Technol. 2020, 230, 115889. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Q.; Wang, T.; Wang, L.; You, J.; Li, Y. Micro-pore geometry manipulation by macroscopic deformation based on shape memory effect in porous PLLA membrane and its enhanced separation performance. ACS Appl. Mater. Interfaces 2017, 9, 43415–43419. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Ye, C.; Zhao, J.; Chen, D.; Weng, B.; You, J.; Liu, Y. Shape memory polymers with interconnected nanopores and high mechanical strength. J. Polym. Sci. B Polym. Phys. 2018, 56, 125–130. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, J.; Xu, J.; Zhao, J.; Zhu, Y.; Li, Y.; You, J. Switchable isotropic/anisotropic wettability and programmable droplet transportation on a shape-memory honeycomb. ACS Appl. Mater. Interfaces 2020, 12, 42314–42320. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Zhang, L.; Cheng, X.; Wang, J.; Li, Y.; You, J. Programmable Transition between Adhesive/Anti-Adhesive Performances on Porous PVDF Spheres Supported by Shape Memory PLLA. Polymers 2022, 14, 374. https://doi.org/10.3390/polym14030374
Zhao J, Zhang L, Cheng X, Wang J, Li Y, You J. Programmable Transition between Adhesive/Anti-Adhesive Performances on Porous PVDF Spheres Supported by Shape Memory PLLA. Polymers. 2022; 14(3):374. https://doi.org/10.3390/polym14030374
Chicago/Turabian StyleZhao, Jiaqin, Liang Zhang, Xiong Cheng, Jiayao Wang, Yongjin Li, and Jichun You. 2022. "Programmable Transition between Adhesive/Anti-Adhesive Performances on Porous PVDF Spheres Supported by Shape Memory PLLA" Polymers 14, no. 3: 374. https://doi.org/10.3390/polym14030374
APA StyleZhao, J., Zhang, L., Cheng, X., Wang, J., Li, Y., & You, J. (2022). Programmable Transition between Adhesive/Anti-Adhesive Performances on Porous PVDF Spheres Supported by Shape Memory PLLA. Polymers, 14(3), 374. https://doi.org/10.3390/polym14030374






