Subcritical Water as Pretreatment Technique for Bioethanol Production from Brewer’s Spent Grain within a Biorefinery Concept
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Subcritical Water Hydrolysis
2.3. Enzymatic Hydrolysis
2.4. Microorganism
2.5. Pre-Inoculum and Inoculum
2.6. Fermentation
2.6.1. Separate Hydrolysis and Fermentation Assays (SHF)—1st Configuration
2.6.2. Simultaneous Saccharification and Fermentation (SSF)—2nd Configuration
Batch Strategy
Fed-Batch Strategy
2.7. Analytical Methods
2.8. Calculations
2.9. Statistical Analysis
3. Results and Discussion
3.1. Subcritical Water Pretreatment
3.2. Enzymatic Hydrolysis of Untreated BSG and Subcritical Water-Pretreated BSG
3.2.1. Untreated BSG
3.2.2. Subcritical Water-Pretreated BSG
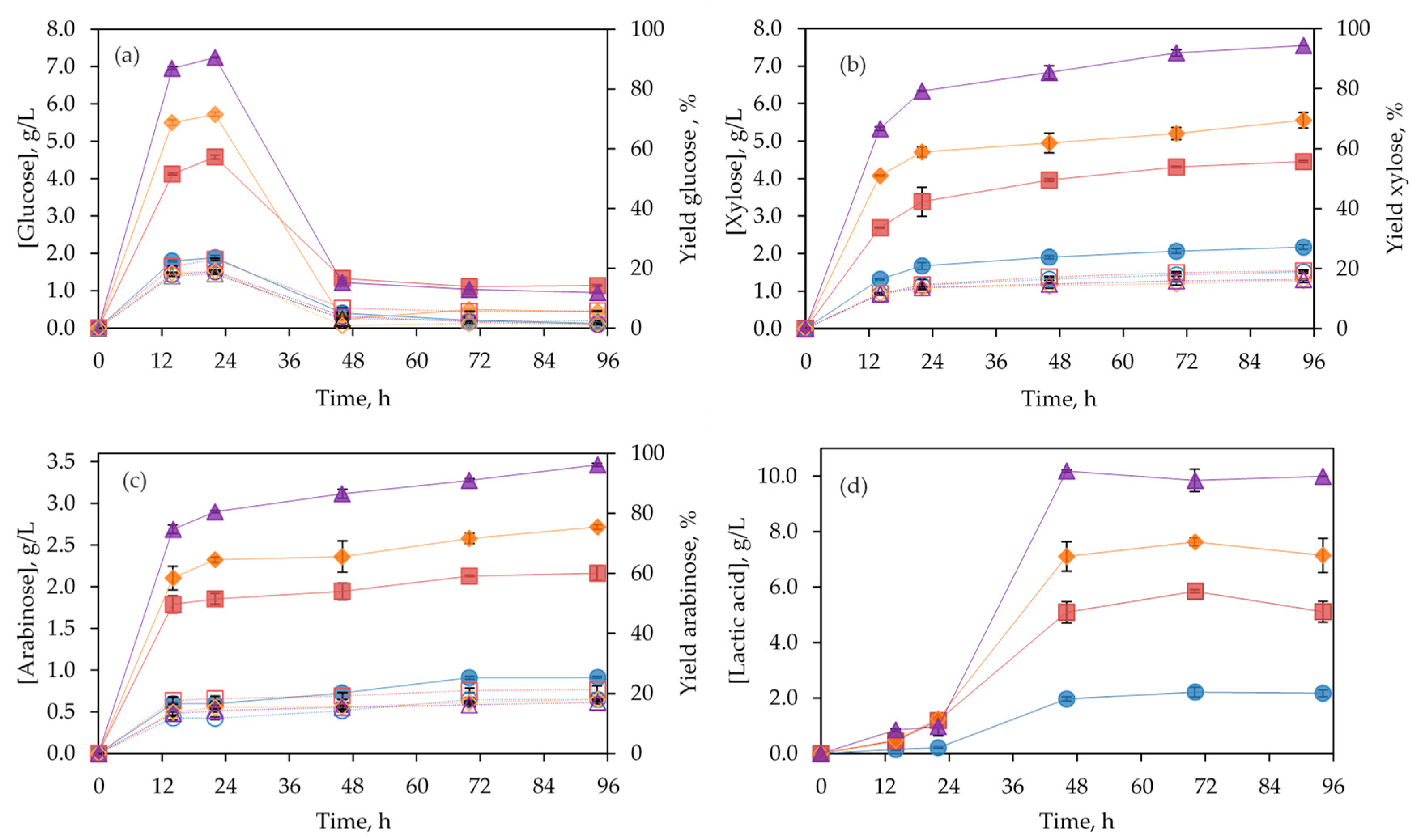
3.3. Separate Hydrolysis and Fermentation (SHF)
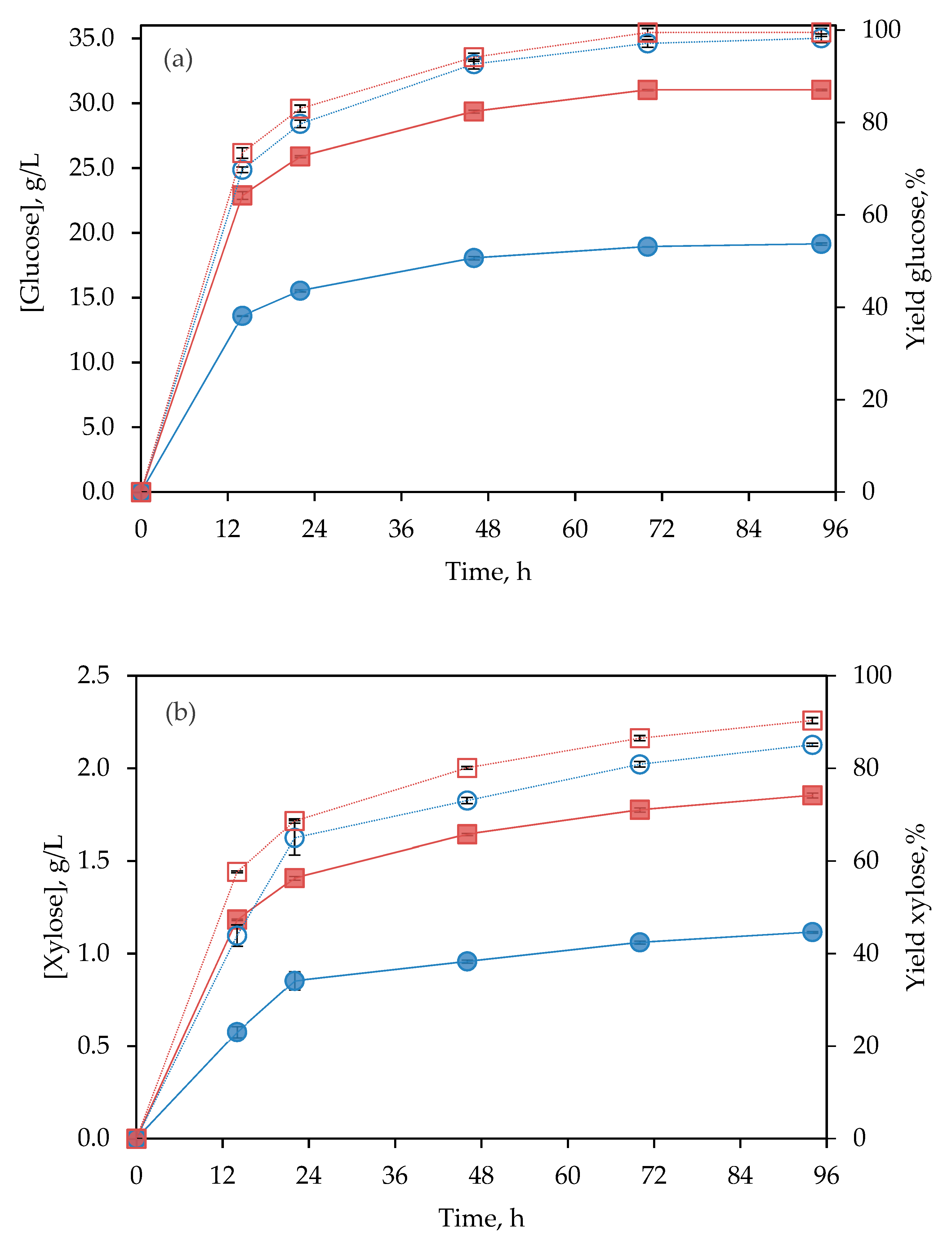
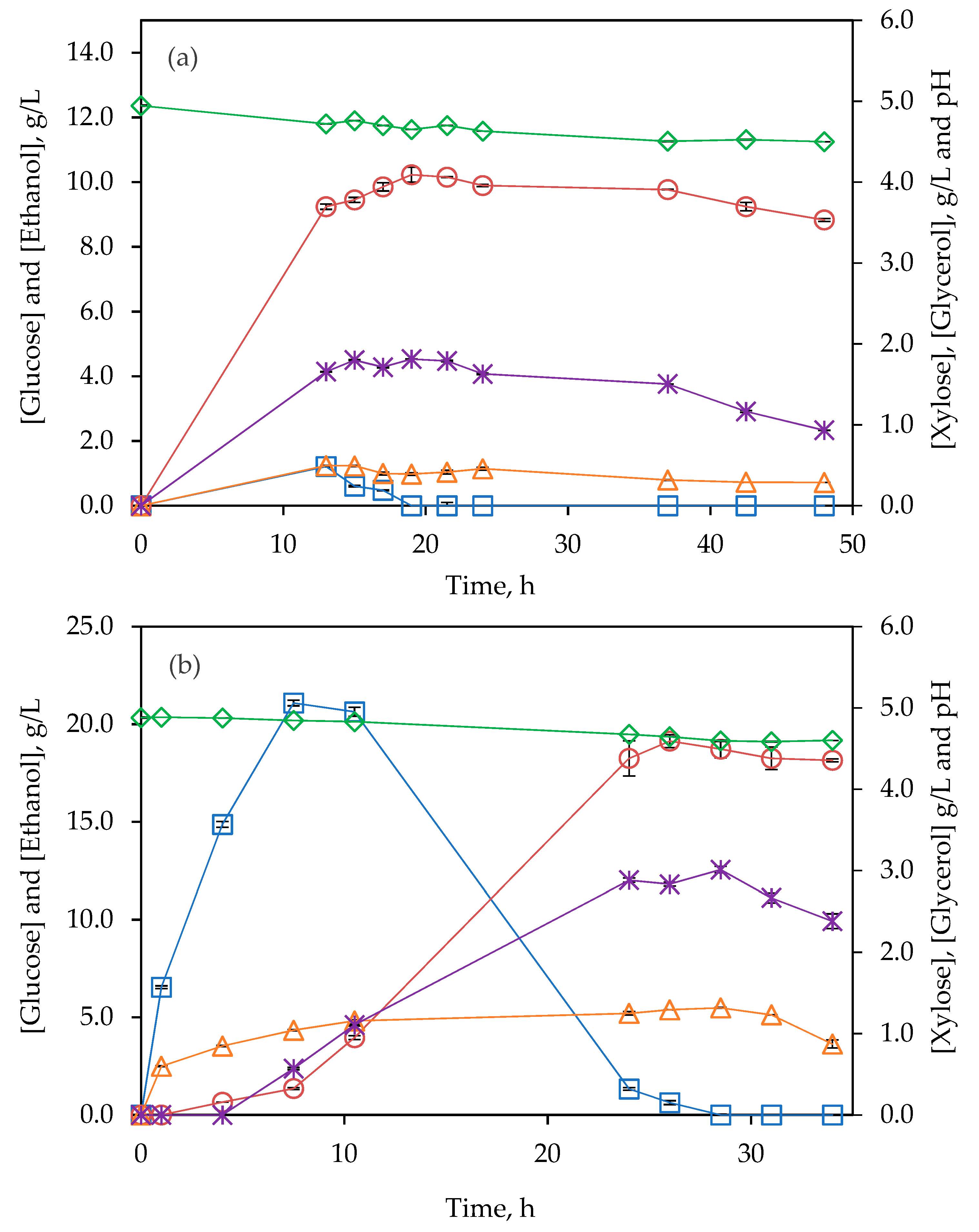
3.4. Simultaneous Saccharification and Fermentation (SSF)
3.4.1. Batch Strategy
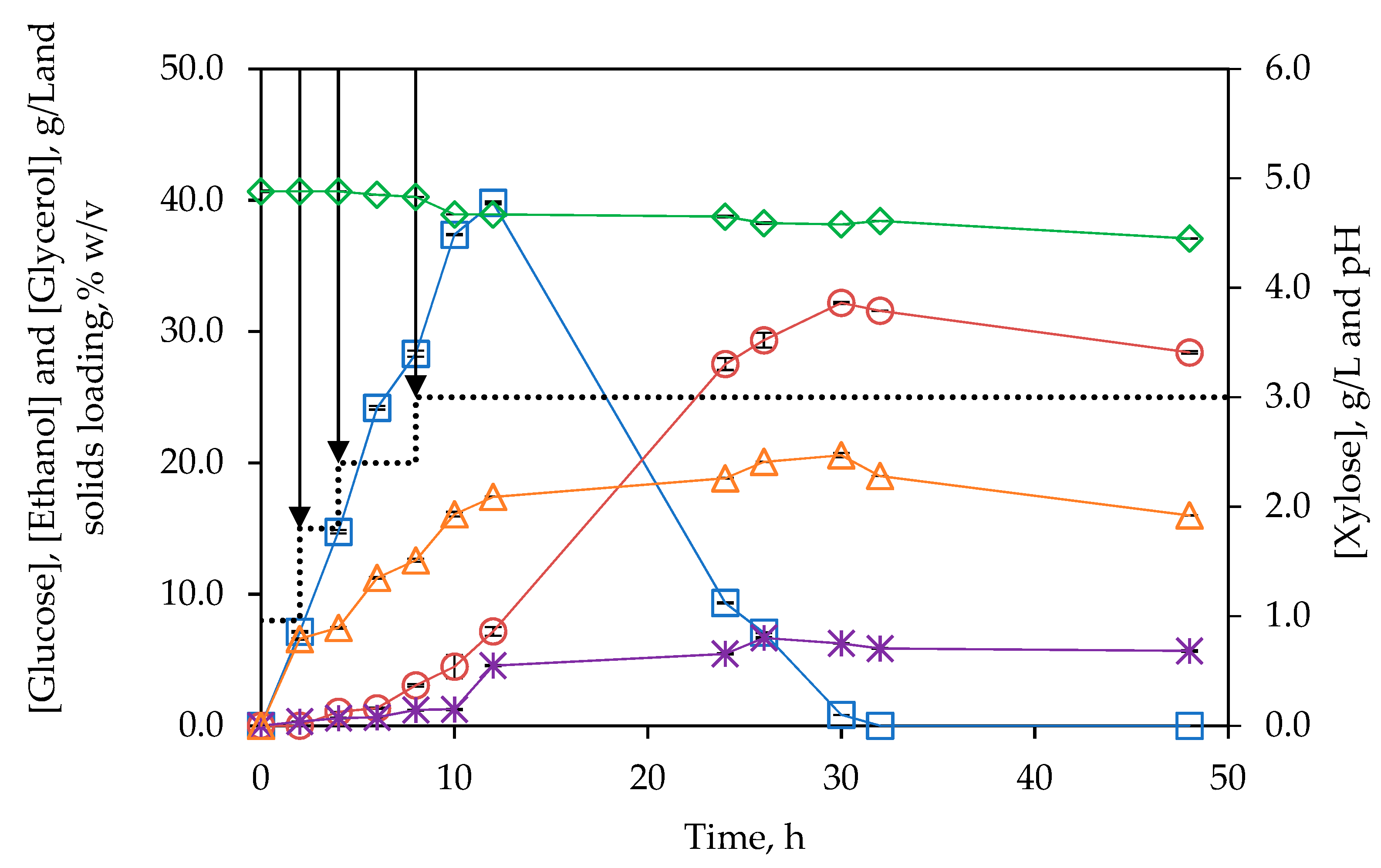
3.4.2. Fed-Batch Strategy
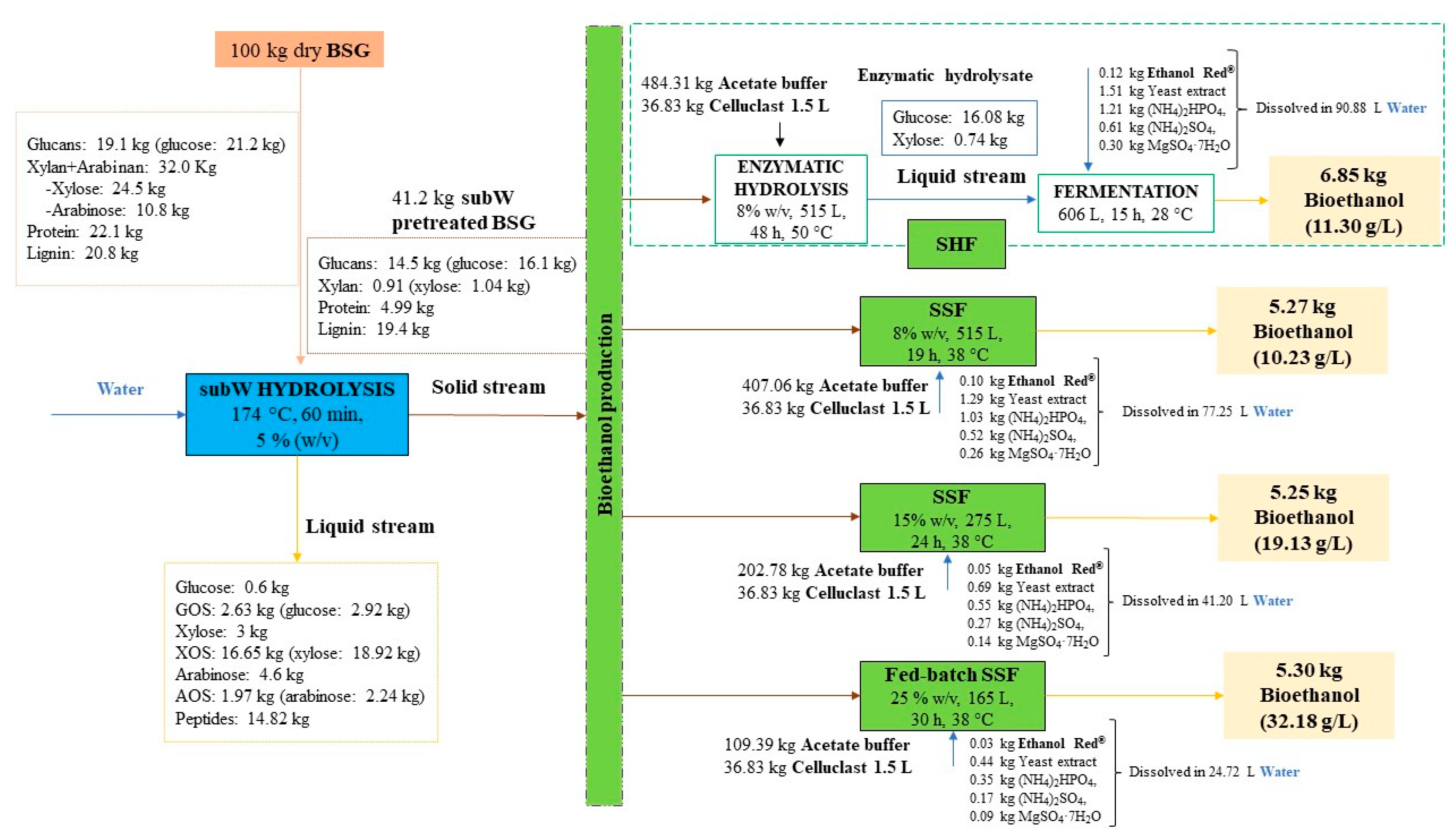
3.5. Process Overview
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dragone, G.; Kerssemakers, A.A.J.; Driessen, J.L.S.P.; Yamakawa, C.K.; Brumano, L.P.; Mussatto, S.I. Innovation and Strategic Orientations for the Development of Advanced Biorefineries. Bioresour. Technol. 2020, 302, 122847. [Google Scholar] [CrossRef] [PubMed]
- Sganzerla, W.G.; Ampese, L.C.; Mussatto, S.I.; Forster-Carneiro, T. A Bibliometric Analysis on Potential Uses of Brewer’s Spent Grains in a Biorefinery for the Circular Economy Transition of the Beer Industry. Biofuels Bioprod. Biorefining 2021, 15, 1965–1988. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Dragone, G.; Roberto, I.C. Brewers’ Spent Grain: Generation, Characteristics and Potential Applications. J. Cereal Sci. 2006, 43, 1–14. [Google Scholar] [CrossRef]
- Alonso-Riaño, P.; Diez, M.T.S.; Blanco, B.; Beltrán, S.; Trigueros, E.; Benito-Román, O. Water Ultrasound-Assisted Extraction of Polyphenol Compounds from Brewer’s Spent Grain: Kinetic Study, Extract Characterization, and Concentration. Antioxidants 2020, 9, 265. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, D.L.; Pereira, S.R.; Serafim, L.S.; Evtuguin, D.V.; Xavier, A.M.R.B. Second Generation Bioethanol from Lignocellulosics: Processing of Hardwood Sulphite Spent Liquor. In Bioethanol; InTech: London, UK, 2012. [Google Scholar] [CrossRef]
- Hamdy, A.; Abd Elhafez, S.; Hamad, H.; Ali, R. The Interplay of Autoclaving with Oxalate as Pretreatment Technique in the View of Bioethanol Production Based on Corn Stover. Polymers 2021, 13, 3762. [Google Scholar] [CrossRef]
- Alonso-Riaño, P.; Benito-Román, O.; Beltrán, S.; Trigueros, E.; Sanz, M.T. Subcritical Water as Hydrolytic Medium to Recover and Fractionate the Protein Fraction and Phenolic Compounds from Craft Brewer’s Spent Grain. Food Chem. 2021, 351, 129264. [Google Scholar] [CrossRef]
- Gomes, D.G.; Michelin, M.; Romaní, A.; Domingues, L.; Teixeira, J.A. Co-Production of Biofuels and Value-Added Compounds from Industrial Eucalyptus globulus Bark Residues Using Hydrothermal Treatment. Fuel 2021, 285, 119265. [Google Scholar] [CrossRef]
- de Camargos, A.B.; da Fonseca, Y.A.; Silva, N.C.S.; da Silva Barreto, E.; Adarme, O.F.H.; de Oliveira Paranhos, A.G.; Gurgel, L.V.A.; Baêta, B.E.L. Production of Biogas and Fermentable Sugars from Spent Brewery Grains: Evaluation of One- and Two-Stage Thermal Pretreatment in an Integrated Biorefinery. J. Environ. Chem. Eng. 2021, 9, 105960. [Google Scholar] [CrossRef]
- Chen, J.; Wang, X.; Zhang, B.; Yang, Y.; Song, Y.; Zhang, F.; Liu, B.; Zhou, Y.; Yi, Y.; Shan, Y.; et al. Integrating Enzymatic Hydrolysis into Subcritical Water Pretreatment Optimization for Bioethanol Production from Wheat Straw. Sci. Total Environ. 2021, 770, 145321. [Google Scholar] [CrossRef]
- Phojaroen, J.; Jiradechakorn, T.; Kirdponpattara, S.; Sriariyanun, M.; Junthip, J.; Chuetor, S. Performance Evaluation of Combined Hydrothermal-Mechanical Pretreatment of Lignocellulosic Biomass for Enzymatic Enhancement. Polymers 2022, 14, 2313. [Google Scholar] [CrossRef]
- Cocero, M.J.; Cabeza, Á.; Abad, N.; Adamovic, T.; Vaquerizo, L.; Martínez, C.M.; Pazo-Cepeda, M.V. Understanding Biomass Fractionation in Subcritical & Supercritical Water. J. Supercrit. Fluids 2018, 133, 550–565. [Google Scholar] [CrossRef]
- Alonso-Riaño, P.; Ramos, C.; Trigueros, E.; Beltrán, S.; Sanz, M.T. Study of Subcritical Water Scale-up from Laboratory to Pilot System for Brewer’s Spent Grain Valorization. Ind. Crops Prod. 2023, 191, 115927. [Google Scholar] [CrossRef]
- Rojas-Chamorro, J.A.; Romero, I.; Ruiz, E.; Cara, C.; Castro, E. Comparison of Fermentation Strategies for Ethanol Production from Pretreated Brewers Spent Grain. Chem. Eng. Trans. 2017, 61, 637–642. [Google Scholar] [CrossRef]
- Sluiter, J.B.; Ruiz, R.O.; Scarlata, C.J.; Sluiter, A.D.; Templeton, D.W. Compositional Analysis of Lignocellulosic Feedstocks. 1. Review and Description of Methods. J. Agric. Food Chem. 2010, 58, 9043–9053. [Google Scholar] [CrossRef] [PubMed]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Sugars, Byproducts, and Degradation Products in Liquid Fraction Process Samples; Issue Date: 12/08/2006; Laboratory Analytical Procedure (LAP): Golden, CO, USA, 2006. [Google Scholar]
- Adney, B.; Nrel, J.B. Measurement of Cellulase Activities: Laboratory Analytical Procedure (LAP); Issue Date: 08/12/1996; Laboratory Analytical Procedure (LAP): Golden, CO, USA, 2008. [Google Scholar]
- Lv, S.; Yu, Q.; Zhuang, X.; Yuan, Z.; Wang, W.; Wang, Q.; Qi, W.; Tan, X. The Influence of Hemicellulose and Lignin Removal on the Enzymatic Digestibility from Sugarcane Bagasse. BioEnergy Res. 2013, 6, 1128–1134. [Google Scholar] [CrossRef]
- Kashaninejad, M.; Blanco, B.; Benito-Román, O.; Beltrán, S.; Niknam, S.M.; Sanz, M.T. Maximizing the Freeze-Dried Extract Yield by Considering the Solvent Retention Index: Extraction Kinetics and Characterization of Moringa Oleifera Leaves Extracts. Food Bioprod. Process. 2021, 130, 132–142. [Google Scholar] [CrossRef]
- Friedl, A.; Padouvas, E.; Rotter, H.; Varmuza, K. Prediction of Heating Values of Biomass Fuel from Elemental Composition. Anal. Chim. Acta 2005, 544, 191–198. [Google Scholar] [CrossRef]
- Yuan, Y.; Jiang, B.; Chen, H.; Wu, W.; Wu, S.; Jin, Y.; Xiao, H. Recent Advances in Understanding the Effects of Lignin Structural Characteristics on Enzymatic Hydrolysis. Biotechnol. Biofuels 2021, 14, 205. [Google Scholar] [CrossRef]
- Zhuang, X.; Wang, W.; Yu, Q.; Qi, W.; Wang, Q.; Tan, X.; Zhou, G.; Yuan, Z. Liquid Hot Water Pretreatment of Lignocellulosic Biomass for Bioethanol Production Accompanying with High Valuable Products. Bioresour. Technol. 2016, 199, 68–75. [Google Scholar] [CrossRef]
- Selig, M.J.; Viamajala, S.; Decker, S.R.; Tucker, M.P.; Himmel, M.E.; Vinzant, T.B. Deposition of Lignin Droplets Produced During Dilute Acid Pretreatment of Maize Stems Retards Enzymatic Hydrolysis of Cellulose. Biotechnol. Prog. 2007, 23, 1333–1339. [Google Scholar] [CrossRef]
- Williams, D.L.; Hodge, D.B. Impacts of Delignification and Hot Water Pretreatment on the Water Induced Cell Wall Swelling Behavior of Grasses and Its Relation to Cellulolytic Enzyme Hydrolysis and Binding. Cellulose 2014, 21, 221–235. [Google Scholar] [CrossRef]
- Trigueros, E.; Sanz, M.T.; Filipigh, A.; Beltrán, S.; Riaño, P. Enzymatic Hydrolysis of the Industrial Solid Residue of Red Seaweed after Agar Extraction: Extracts Characterization and Modelling. Food Bioprod. Process. 2021, 126, 356–366. [Google Scholar] [CrossRef]
- Di Cagno, R.; Filannino, P.; Gobbetti, M. Vegetable and Fruit Fermentation by Lactic Acid Bacteria. In Biotechnology of Lactic Acid Bacteria: Novel Applications, 2nd ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2015; pp. 216–230. [Google Scholar] [CrossRef]
- Michelin, M.; Teixeira, J.A. Liquid Hot Water Pretreatment of Multi Feedstocks and Enzymatic Hydrolysis of Solids Obtained Thereof. Bioresour. Technol. 2016, 216, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Riaño, P.; Melgosa, R.; Trigueros, E.; Illera, A.E.; Beltrán, S.; Sanz, M.T. Valorization of Brewer’s Spent Grain by Consecutive Supercritical Carbon Dioxide Extraction and Enzymatic Hydrolysis. Food Chem. 2022, 396, 133493. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Dragone, G.; Fernandes, M.; Milagres, A.M.F.; Roberto, I.C. The Effect of Agitation Speed, Enzyme Loading and Substrate Concentration on Enzymatic Hydrolysis of Cellulose from Brewer’s Spent Grain. Cellulose 2008, 15, 711–721. [Google Scholar] [CrossRef]
- Rojas-Chamorro, J.A.; Cara, C.; Romero, I.; Ruiz, E.; Romero-García, J.M.; Mussatto, S.I.; Castro, E. Ethanol Production from Brewers’ Spent Grain Pretreated by Dilute Phosphoric Acid. Energy Fuels 2018, 32, 5226–5233. [Google Scholar] [CrossRef]
- Qing, Q.; Yang, B.; Wyman, C.E. Xylooligomers Are Strong Inhibitors of Cellulose Hydrolysis by Enzymes. Bioresour. Technol. 2010, 101, 9624–9630. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Liu, Y.; Uchida, S.; Kawarada, K.; Ukagami, Y.; Ichinose, H.; Kaneko, S.; Fukuda, K. Effects of Cellulose Crystallinity, Hemicellulose, and Lignin on the Enzymatic Hydrolysis of Miscanthus sinensis to Monosaccharides. Biosci. Biotechnol. Biochem. 2008, 72, 805–810. [Google Scholar] [CrossRef]
- Berlin, A.; Maximenko, V.; Gilkes, N.; Saddler, J. Optimization of Enzyme Complexes for Lignocellulose Hydrolysis. Biotechnol. Bioeng. 2007, 97, 287–296. [Google Scholar] [CrossRef]
- Branco, R.H.R.; Amândio, M.S.T.; Serafim, L.S.; Xavier, A.M.R.B. Ethanol Production from Hydrolyzed Kraft Pulp by Mono- And Co-Cultures of Yeasts- And Challenge of C6 and C5 Sugars Consumption. Energies 2020, 13, 744. [Google Scholar] [CrossRef]
- Amândio, M.S.T.; Rocha, J.M.S.; Serafim, L.S.; Xavier, A.M.R.B. Cellulosic Bioethanol from Industrial Eucalyptus globulus Bark Residues Using Kraft Pulping as a Pretreatment. Energies 2021, 14, 2185. [Google Scholar] [CrossRef]
- Rojas-Chamorro, J.A.; Romero, I.; López-Linares, J.C.; Castro, E. Brewer’s Spent Grain as a Source of Renewable Fuel through Optimized Dilute Acid Pretreatment. Renew. Energy 2020, 148, 81–90. [Google Scholar] [CrossRef]
- Liguori, R.; Soccol, C.R.; de Souza Vandenberghe, L.P.; Woiciechowski, A.L.; Faraco, V. Second Generation Ethanol Production from Brewers’ Spent Grain. Energies 2015, 8, 2575–2586. [Google Scholar] [CrossRef]
- Wilkinson, S.; Smart, K.A.; Cook, D.J. Optimisation of Alkaline Reagent Based Chemical Pre-Treatment of Brewers Spent Grains for Bioethanol Production. Ind. Crops Prod. 2014, 62, 219–227. [Google Scholar] [CrossRef]
- Bedő, S.; Rozbach, M.; Nagy, L.; Fehér, A.; Fehér, C. Optimised Fractionation of Brewer’s Spent Grain for a Biorefinery Producing Sugars, Oligosaccharides, and Bioethanol. Processes 2021, 9, 366. [Google Scholar] [CrossRef]
- Scanes, K.T.; Hohrnann, S.; Prior, B.A. Glycerol Production by the Yeast Saccharomyces Cerevisiae and Its Relevance to Wine: A Review. S. Afr. J. Enol. Vitic. 1998, 19, 17–24. [Google Scholar] [CrossRef]
- Zacchi, G.; Axelsson, A. Economic Evaluation of Preconcentration in Production of Ethanol from Dilute Sugar Solutions. Biotechnol. Bioeng. 1989, 34, 223–233. [Google Scholar] [CrossRef]
- Rojas-Chamorro, J.A.; Romero-García, J.M.; Cara, C.; Romero, I.; Castro, E. Improved Ethanol Production from the Slurry of Pretreated Brewers’ Spent Grain through Different Co-Fermentation Strategies. Bioresour. Technol. 2020, 296, 122367. [Google Scholar] [CrossRef]
- Pinheiro, T.; Coelho, E.; Romaní, A.; Domingues, L. Intensifying Ethanol Production from Brewer’s Spent Grain Waste: Use of Whole Slurry at High Solid Loadings. New Biotechnol. 2019, 53, 1–8. [Google Scholar] [CrossRef]
- Lynch, K.M.; Steffen, E.J.; Arendt, E.K. Brewers’ Spent Grain: A Review with an Emphasis on Food and Health. J. Inst. Brew. 2016, 122, 553–568. [Google Scholar] [CrossRef]
- George, A.; Simet, K.; Carradorini, A. Brewer’s Spent Grain to Xylitol & Polylactic Acid; Senior Design Reports (CBE); Penn Libraries: Philadelphia, PA, USA, 2017; pp. 1–368. Available online: https://repository.upenn.edu/cbe_sdr/90 (accessed on 15 September 2022).
- Steinbach, D.; Kruse, A.; Sauer, J. Pretreatment Technologies of Lignocellulosic Biomass in Water in View of Furfural and 5-Hydroxymethylfurfural Production—A Review. Biomass Convers. Biorefinery 2017, 7, 247–274. [Google Scholar] [CrossRef]
- Benito-Román, Ó.; Alonso-Riaño, P.; de Cerio, E.D.; Sanz, M.T.; Beltrán, S. Semi-Continuous Hydrolysis of Onion Skin Wastes with Subcritical Water: Pectin Recovery and Oligomers Identification. J. Environ. Chem. Eng. 2022, 10, 107439. [Google Scholar] [CrossRef]
- Ranganathan, P. Preliminary Techno-Economic Evaluation of 2G Ethanol Production with Co-Products from Rice Straw. Biomass Convers. Biorefinery 2022, 12, 3673–3686. [Google Scholar] [CrossRef]
- Engineering ToolBox. Fuels–Higher and Lower Calorific Values. Available online: https://www.engineeringtoolbox.com/fuels-higher-calorific-values-d_169.html (accessed on 15 September 2022).

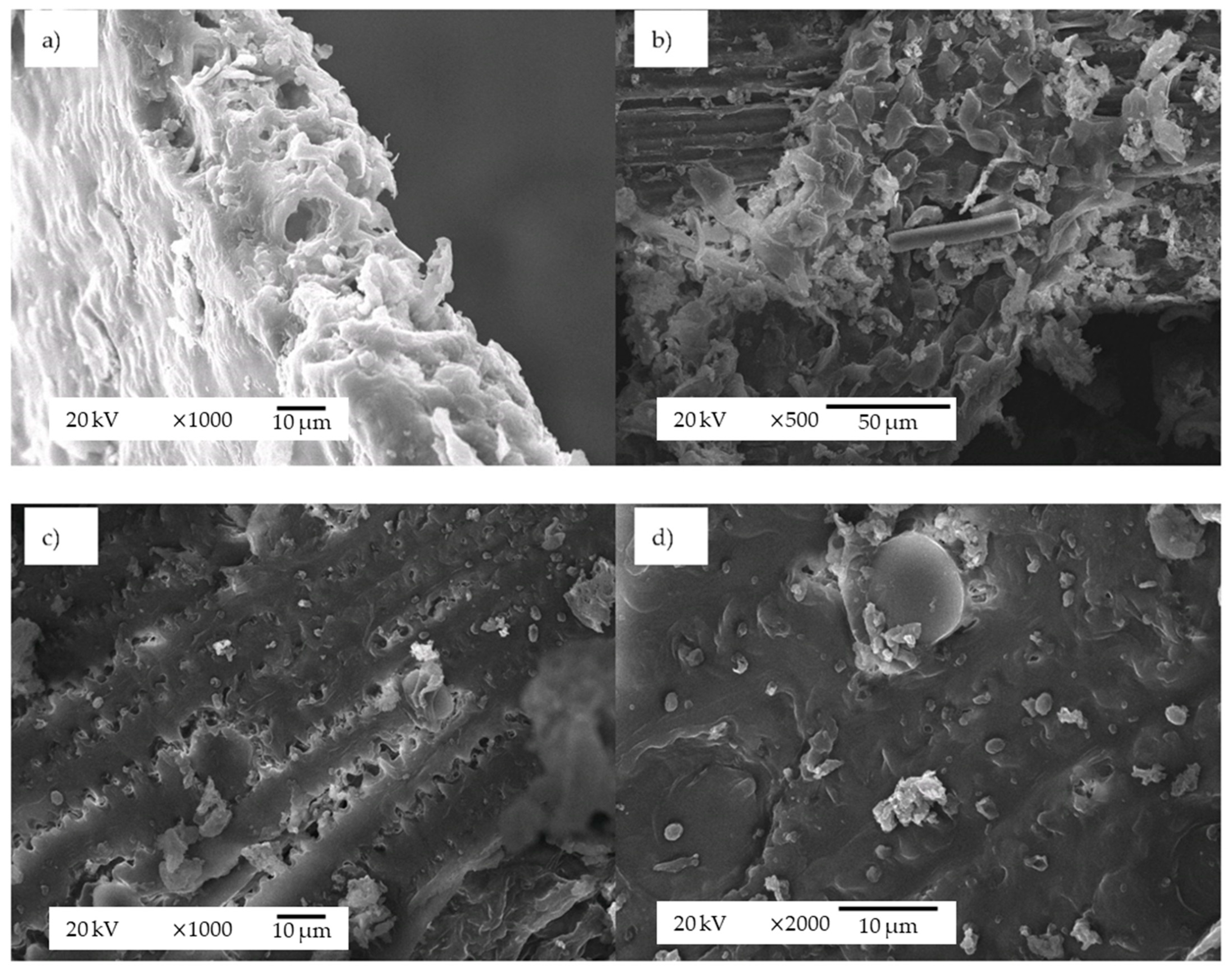

| Compound | Raw BSG (% dwb) | subW-Pretreated BSG (% dwb) | subW Hydrolysate (g/100 gBSG) | E-PBSG (% dwb) |
| Glucans | 19.1 ± 0.2 | 35.1 ± 0.3 | 3.16 ± 0.02 | 1.00 ± 0.01 |
| Starch | 4.11 ± 0.06 | - | - | - |
| Β-glucan | 0.99 ± 0.01 | - | - | - |
| Cellulose | 14.0 ± 0.2 | - | - | - |
| Hemicelluloses | 32.0 ± 0.6 | 2.22 ± 0.04 | 0.150± 0.001 | |
| Xylan | 21.6 ± 0.4 | 2.22 ± 0.04 | 19.28 ± 0.09 | 0.150 ± 0.001 |
| Arabinan | 9.5 ± 0.4 | Not detected | 6.03 ± 0.01 | Not detected |
| Acetate | 0.93 ± 0.05 | Not detected | - | Not detected |
| Lignin | 20.8 ± 0.2 | 47.1 ± 0.5 | - | 87.60 ± 0.05 |
| Acid insoluble | 15.5 ± 0.1 | 42.9 ± 0.4 | - | 85.78 ± 0.04 |
| Acid soluble | 5.3 ± 0.2 | 4.2 ± 0.1 | - | 1.82 ± 0.03 |
| Ash | 3.32 ± 0.06 | 2.7 ± 0.1 | - | 3.1 ± 0.1 |
| Proteins | 22.1 ± 0.7 | 12.1 ± 0.1 | 14.82 ± 0.04 | 19.0 ± 0.9 |
| Pretreatment | Enzyme. | Enzyme Dosage | Conf | Solid Loading (% w/v) | [Glucose] (g/L) | Glucose Yield (%) | [Ethanol] (1) g/L | Ethanol Yield (%) | Productivity (2) (g·L−1·h−1) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| SubW, 174 °C, 50 bar, 60 min. | Celluclast 1.5L | 40 FPU/gBSG. | SHF | 8 | 31.1 | 99.7 | 11.3 | (1) 83 | 0.75 | This work |
| Fed-batch SSF | 25 | - | - | 32.2 | 65 | 1.07 | ||||
| H3PO4 (6.83%), 160 °C | Cellic CTec3 β-glucosidase | 15 FPU/gsubstrate 15 IU/gsubstrate | SHF | 15 | 59.4 | 22.6 | 72 | 0.94 | [14] | |
| SSF | - | - | 18.5 | 62 | 0.60 | |||||
| H2SO4 (1%), 130 °C, 26 min | Cellic CTec3 β-glucosidase | 15 FPU/gsubstrate 15 IU/gsubstrate | SSF | 25 | 27 | 68 | [36] | |||
| 1. H2SO4 (1.25%), 120 °C, 17 min 2.NaOH (2%), 120 °C, 90 min | Cellulase β-glucosidase (Novozymes) | 2.24% (v/v) 1%(v/v) | SHF | 8 | 75 | 97 | 12.8 | 54.8 | 0.53 | [37] |
| NaOH (5%), 50 °C, 12 h | Cellic®CTec2 | 51 FPU/gsolid (calculated) | SHF | 15 | 41.7 | 17.3 | 81 | N.R. | [38] | |
| 1. H2SO4 (1.8 5%), 90 °C, 19.5 min 2. H2SO4 (1%), 120 °C, 30 min | Cellic® CTec3 | 0.04 g/gdry-matter | SHF | 15 | 46.1 | 75.5 | 16.9 | 71.6 | 0.72 | [39] |
| Configuration | Solids Loading (%) | [Ethanol]max (g/L) | Ethanol Yield (%) | Yethanol/solid (g/g) | Productivity (g·L−1·h−1) |
|---|---|---|---|---|---|
| SHF | 8 | 11.30 ± 0.01 b | 83.4 ± 0.4 b | 0.1662 ± 0.0002 b | (1) 0.754 ± 0.001 d (2) 0.1794 ± 0.0002 a |
| SSF | 8 | 10.2 ± 0.2 a | 64.2 ± 0.8 a | 0.128 ± 0.003 a | 0.54 ± 0.01 b |
| SSF | 15 | 19.1 ± 0.3 c | 64.0 ± 0.6 a | 0.128 ± 0.002 a | 0.74 ±0.01 c |
| Fed-batch SSF | 25 (8 + 7 + 5 + 5) | 32.18 ± 0.08 d | 64.7 ± 0.3 a | 0.1287 ± 0.0003 a | 1.073 ±0.003 e |
| Compound | mfeed (kg) | msubW-hydrolysate (kg) | msubW-pretreated-BSG (kg) | menzymatic-hydrolysate (kg) | mE-PBSG (kg) | moutlet (kg) | MB (%) |
|---|---|---|---|---|---|---|---|
| Solid | 100 | - | 41.2 | - | 24.5 | - | - |
| Glucose | 21.2 ± 0.2 | 3.51 ± 0.02 | 16.1 ± 0.1 | 16.08 ± 0.26 | 0.27 ± 0.00 | 19.9 ± 0.3 | 94 ± 2 |
| Xylose | 24.5 ± 0.5 | 21.9 ± 0.1 | 1.04 ± 0.02 | 0.74 ± 0.01 | 0.04 ± 0.00 | 22.7 ± 0.1 | 92 ± 2 |
| Arabinose | 10.8 ± 0.5 | 6.85 ± 0.01 | Not detected | Not detected | Not detected | 6.85 ± 0.01 | 64 ± 3 |
| Carbohydrates | 56.5 ± 0.7 | 32.3 ± 0.1 | 17.1 ± 0.1 | 16.8 ± 0.3 | 0.31 ± 0.00 | 49.4 ± 0.3 | 87 ± 2 |
| Protein | 22.1 ± 0.7 | 14.85 ± 0.04 | 4.99 ± 0.06 | Not determined | 4.8 ± 0.2 | 19.6 ± 0.2 | 89 ± 4 |
| Lignin | 20.8 ± 0.2 | Not determined | 19.4 ± 0.2 | Not determined | 21.42 ± 0.01 | 21.42 ± 0.01 | 103 ± 1 |
| Raw BSG | subW-Pretreated BSG | E-PBSG | SSF-PBSG | |
|---|---|---|---|---|
| C (% dwb) | 49.1 ± 0.5 | 53.3 ± 0.7 | 65.5 ± 0.9 | 65.2 ± 0.5 |
| H (% dwb) | 6.64 ± 0.01 | 6.87 ± 0.07 | 8.45 ± 0.04 | 8.45 ± 0.06 |
| N (% dwb) | 3.5 ± 0.1 | 1.94 ± 0.02 | 3.0 ± 0.1 | 3.14 ± 0.01 |
| HHV (MJ/kg) | 20.1 ± 0.3 a | 22.0 ± 0.7 b | 31 ± 2 c | 30.3 ± 0.4 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alonso-Riaño, P.; Amândio, M.S.T.; Xavier, A.M.R.B.; Beltrán, S.; Sanz, M.T. Subcritical Water as Pretreatment Technique for Bioethanol Production from Brewer’s Spent Grain within a Biorefinery Concept. Polymers 2022, 14, 5218. https://doi.org/10.3390/polym14235218
Alonso-Riaño P, Amândio MST, Xavier AMRB, Beltrán S, Sanz MT. Subcritical Water as Pretreatment Technique for Bioethanol Production from Brewer’s Spent Grain within a Biorefinery Concept. Polymers. 2022; 14(23):5218. https://doi.org/10.3390/polym14235218
Chicago/Turabian StyleAlonso-Riaño, Patricia, Mariana S. T. Amândio, Ana M. R. B. Xavier, Sagrario Beltrán, and Maria Teresa Sanz. 2022. "Subcritical Water as Pretreatment Technique for Bioethanol Production from Brewer’s Spent Grain within a Biorefinery Concept" Polymers 14, no. 23: 5218. https://doi.org/10.3390/polym14235218
APA StyleAlonso-Riaño, P., Amândio, M. S. T., Xavier, A. M. R. B., Beltrán, S., & Sanz, M. T. (2022). Subcritical Water as Pretreatment Technique for Bioethanol Production from Brewer’s Spent Grain within a Biorefinery Concept. Polymers, 14(23), 5218. https://doi.org/10.3390/polym14235218











