3.2. Experiment-2: Surfactant-1 (Vorasurf™ 5906) Screen
Seven formulations were screened in experiment-2 with surfactant-1 loading being the only factor. The response variables measured were the same kinetic responses as in experiment-1 as well as the percentage sag or “sigh back”, calculated using height data, an indicator of cell opening of a PU foam [
40]. Cell size, one of the targeted cell morphology responses, is also measured. The full formulation data for experiment-2 is available in
Supplementary Table S1.
Figure 4A shows the -NCO group conversion with the dashed line indicating the predicted -NCO conversion at a catalyst loading of 0.8 PPHP (X
NCO = 0.95). At a loading of 0 PPHP surfactant-1 the foam collapsed and never reached the temperature probe, hence the low value of X
NCO. At loadings between 0.25 PPHP and 0.75 PPHP, conversion is lower than expected, ranging between 0.87 and 0.91. Above 0.75 PPHP, X
NCO is within 0.03 of the expected conversion, which is within the precision of the FoamPi temperature probe and therefore not significant.
Figure 4B shows the normalised maximum height data, which exhibits exponential behaviour with regard to surfactant-1 loading, and the fitted model predicts a maximum normalised height of 3.40 mm g
−1 using this surfactant in this specific formulation. At loadings above 1 PPHP surfactant-1, maximum normalised height does not increase.
Percentage sag or “sigh back” data is shown in
Figure 4C. The percentage sag decreases from 30% at 0 PPHP surfactant-1 to 7.3% at 0.75 PPHP surfactant-1. This decrease indicates the stabilising effect of the surfactant, reducing the “collapse” of the foams whilst the remaining foam is left with an open cell morphology (as there is still some sag). Above 0.75 PPHP, the sag increases slightly to a value of 12% at a loading of 4 PPHP surfactant-1, which is unexpected and could indicate shrinkage due to closed cells. However, this would need to be confirmed by microscopy/airflow measurement.
Figure 5A shows the effect of surfactant-1 concentration on the cell size of the foam with the error bars indicating one standard error of the cell size as calculated from more than 200 cells.
Figure 5B–G show optical images of the stained foam surfaces at increasing surfactant-1 loading. The cell size decreases, from a cell size of 1200 ± 76 µm at 0.25 PPHP loading surfactant-1, until it plateaus at a loading of 1 PPHP surfactant. An exponential fit of the data shows that the predicted minimum cell size with this surfactant in this formulation is 678 µm. The error bars also reduce in size, indicating that cell size distribution becomes more uniform with an increase in surfactant-1 loading. The images on the right show optical microscopy images of the surface marked foam.
From the results of experiment-2, an understanding of the kinetic effects as well as some information on cell morphology have been gathered for sequential experiments. The minimum amount of surfactant-1 required to produce a stable foam is 0.5 PPHP. As we are interested in reducing the cell size of PU foam to increase capillary action, the range of interest is that which produces the smallest cell sizes. For experiment-3 a loading of 0.8 PPHP is selected, where the predicted cell size is expected to be 753 µm. The range selected for experiment-4 is between 0.8 PPHP (stable foam with reduced cell size) and 2 PPHP (highly stabilised foam with small and uniform cell size). These seven relatively quick screening formulations (20 min experimental time per formulation) give particularly useful information into the effect of surfactant-1 on PU foam cell size.
3.3. Experiment-3: Surfactant-2 (Tegostab® 8476) Screen
Experiment-3 screened surfactant-2, Tegostab
® 8476, a silicone surfactant that promotes extremely small and closed cell morphology. This was the only factor in this screen. The response variables were the same as those in experiment-2 with the addition of open cell content, calculated using airflow data, the second targeted cell morphology response. The full formulation data for experiment-3 is available in
Supplementary Table S1.
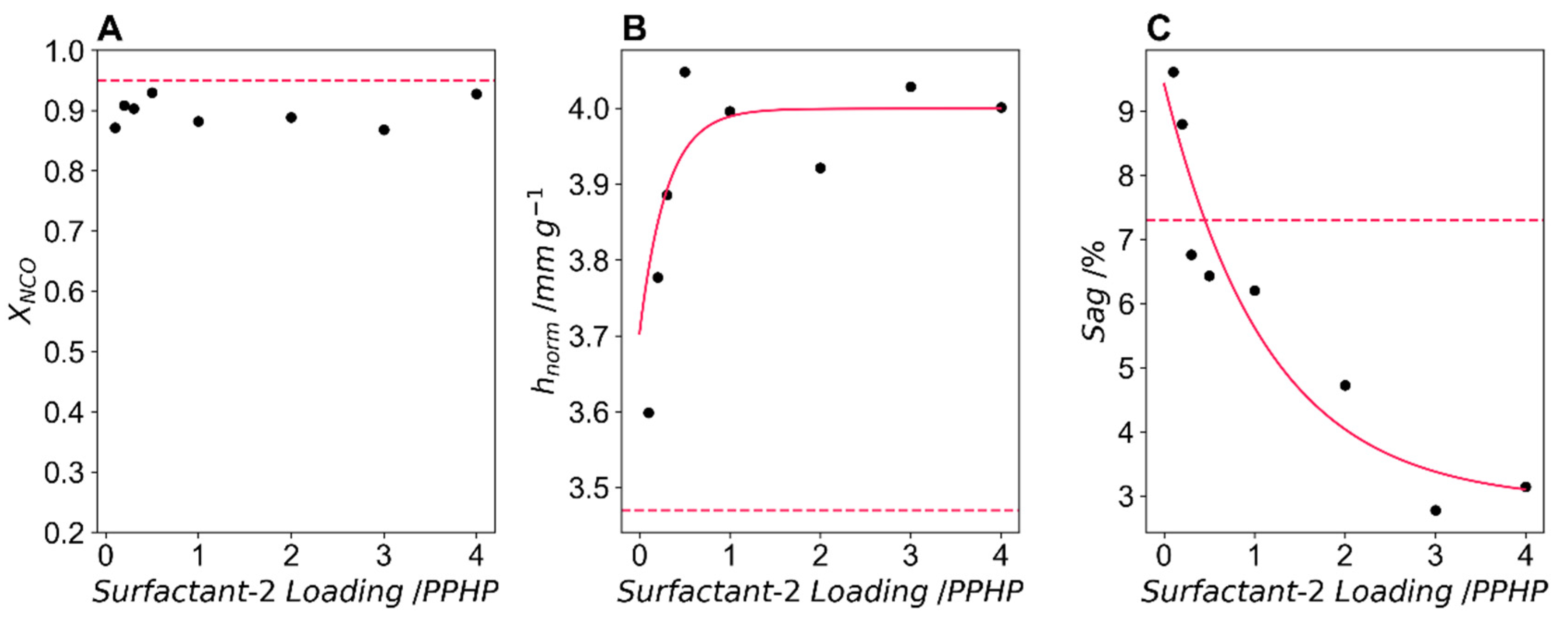
Figure 6A shows X
NCO, the isocyanate conversion at varying surfactant-2 loadings. The conversion is reduced at all loadings of surfactant-2, X
NCO varied between 0.872 and 0.929, and although this reduction is slight, it may indicate an antagonistic effect between surfactant-2 and the catalyst, which can be further explored in the DoE experiment-4.
Figure 6B shows the normalised maximum height, which increases with any addition of surfactant-2 above the baseline of 3.47 mm g
−1. This reaches a plateau at a loading of ~1 PPHP and a normalised maximum height of 3.99 mm g
−1 is predicted to be the greatest value of this formulation using an exponential fit of the data. This increase in normalised height shows the greater stabilising ability of surfactant-2 when compared to surfactant-1.
Sag is shown in
Figure 6C, which reduces with increased surfactant-2 loading. Interestingly, at low levels of surfactant-2 sag is greater than the formulation with none of this surfactant. This may indicate that at low loadings of surfactant-2 there is an antagonistic effect due to incompatibility between the two surfactants. At all loadings above 1 PPHP surfactant-2 the sag is reduced below that of the formulation with no surfactant-2. An exponential fit of the data predicts a minimum sag of 2.90%.
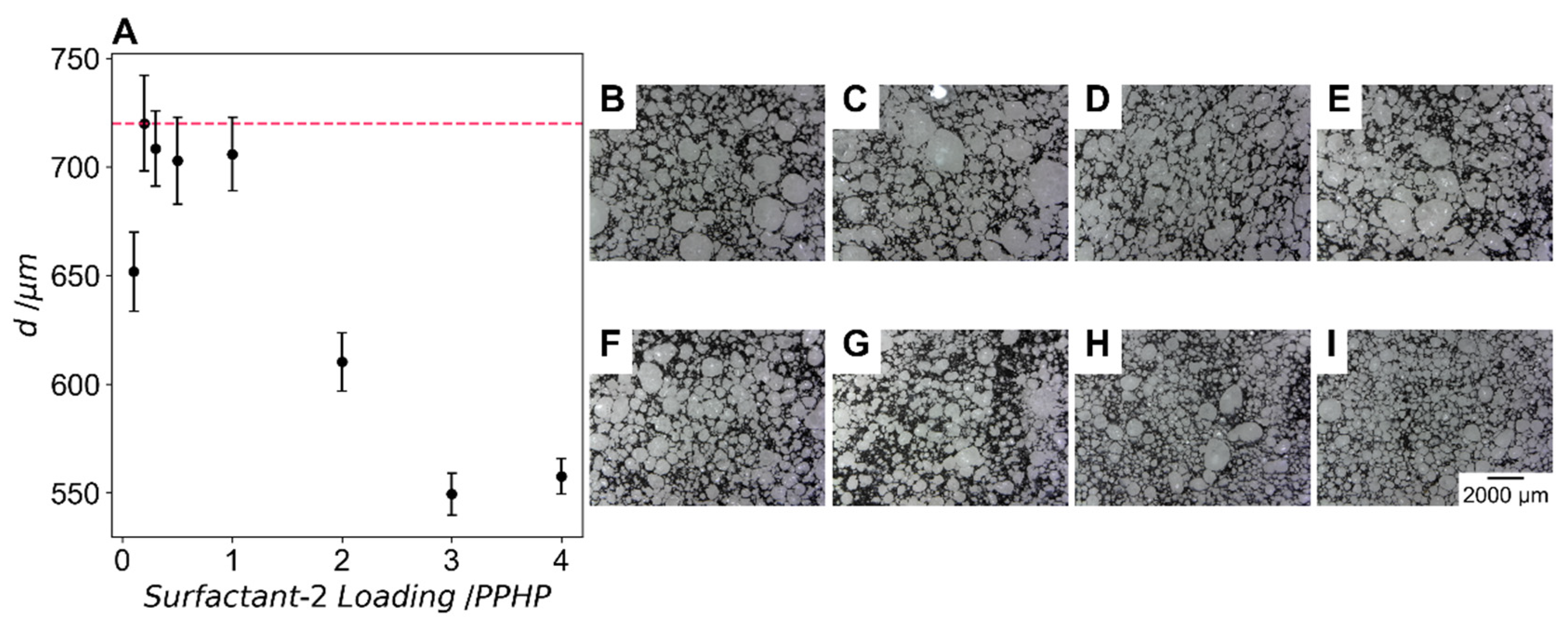
The effect of surfactant-2 loading on cell size of the foam is shown in
Figure 7A with error bars indicating one standard error with more than 200 cells counted for each sample.
Figure 7B–I shows optical images of the stained PU foam surface at increasing surfactant-2 loading. At 0.1 PPHP surfactant-2 we see a reduction in cell size from the formulation with no surfactant-2. However, at loadings between 0.2 PPHP and 1 PPHP cell size is not significantly different to the foam without this surfactant. At loadings of 2 PPHP, 3 PPHP and 4 PPHP the cell size reduces to 610 ± 13.4 µm, 549 ± 9.69 µm and 558 ± 8.10 µm, respectively. These results indicate that the cell size does not reduce between 3 PPHP and 4 PPHP and that these values likely represent the smallest cell size that can be produced using this surfactant in this foam formulation. The low standard error values indicate that foams with highly homogenous cells are produced using this surfactant, particularly at higher loadings.
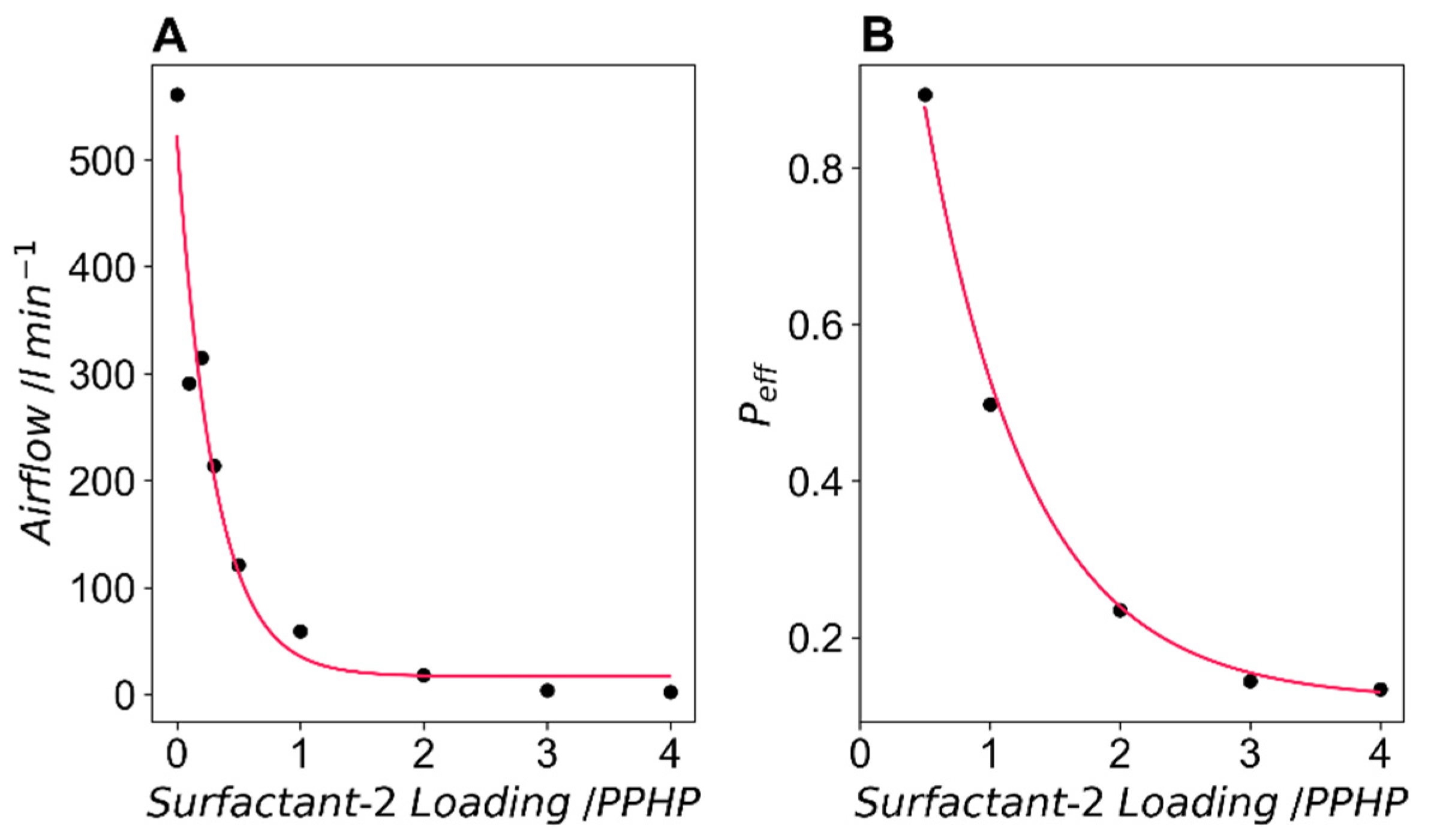
Airflow data is shown in
Figure 8A. Airflow decreases with any increase of surfactant-2 and reaches a plateau at surfactant-2 loadings above 2 PPHP. From previous work (data not shown), airflow values above 130 L min
−1 indicate a foam with an open cell ratio exceeding 0.95. All formulations with a loading of surfactant-2 below 0.5 PPHP fall into this category.
Figure 8B shows the effective open cell ratio (p
eff) calculated from the airflow data for formulations with surfactant-2 loadings above 0.5 PPHP. In the range between 0.5 PPHP and 4 PPHP surfactant-2 we can produce a range of foams where p
eff ranges between 0.13 and 0.85. The exponential fit of this data predicts a minimum open cell ratio of 0.12.
In experiment-3 eight screening formulations with varying loadings of surfactant-2 were trialled. These eight formulations required roughly 30 min each to react and measure the resulting foam morphology properties. This set of formulations produced a wide range of PU foam cell morphologies. There was little effect on the cell size of the foams at surfactant-2 loadings below 0.5 PPHP and there was possibly some antagonism between the two surfactants at low loadings as sag increased from the formulation with no surfactant-2. Surfactant-2 reduced airflow and increased the closed cell content as expected from this surfactant. Considering the range of catalyst (0.5–1 PPHP) and the range of surfactant-1 (0.8 PPHP–2 PPHP) suggested for experiment-4, and the catalyst and surfactant loadings used in experiment-3 (0.8 PPHP and 1 PPHP, respectively), the range of surfactant-2 needs to be carefully selected for experiment-4. The lower bound is selected as 1 PPHP surfactant-2. To account for the case where high blowing catalyst and surfactant-1 loading would tend to a high open cell content, this value would lead to a foam with mostly open cells. The upper range was selected to be 3 PPHP as any further increase in surfactant-2 had no effect on cell size and resulted in a mostly closed cell foam.
3.4. Experiment-4: DoE Screen
Experiment-4 focused on mapping the experimental formulation space defined by the three screening experiments that preceded it and fully characterising the resulting PU foam physical properties. The DoE factors are the catalyst, Dabco
® T (range 0.5–1 PPHP), surfactant-1, Vorasurf™ 5906 (range 0.8–2 PPHP), and surfactant-2, Tegostab
® 8476 (range 1–3 PPHP). The responses are isocyanate conversion (X
NCO), density (ρ), cell size (d
cell), open cell fraction (p
eff), water holding capacity (WHC), water drop penetration time (WDPT), maximum capillary rise (α
1), and rate of water uptake (α
2). 16 formulations were screened. For modelling the three factors are labelled as x
1 (catalyst), x
2 (surfactant-1) and x
3 (surfactant-2). The full formulation data for experiment-4 is available in
Supplementary Table S1.
The three screening experiments led to all 16 formulations producing stable foams, which would have been highly unlikely if an iterative DoE approach had not been taken.
Figure 9 shows model parameters for each of the kinetic and cell morphology responses. The top part of each subplot shows the significant model parameters from the generalised model used to predict each of the responses. The horizontal bars indicate the value and sign of the result of the t-ratio for each model parameter. The t-ratio is defined as the estimate of the coefficient divided by the standard error of the estimate. Those shown in pink have a t-ratio > 2 which indicates the parameter has
p < 0.05. The bars in blue indicate that the value of the t-ratio < 2 and
p > 0.05. The plot below the horizontal bars shows the actual response as a function of the predicted response, with the fit shown in the solid pink line and the dashed black line indicating the y = x line. The r
2 is inset as is the k-fold r
2 (k = 5) for each of the models.
The t-ratio and actual/predicted isocyanate conversion (X
NCO) are shown in
Figure 9A. The random error parameter, ε, is the parameter that explains the most variance, indicating that at the lower limit of factor ranges (x
1 = 0.5 PPHP, x
2 = 0.8 PPHP, and x
3 = 1 PPHP), there is an isocyanate conversion of 0.849. This is lower than the expected conversion at a catalyst loading of 0.5 PPHP (0.900) and may be explained by the value of the surfactant-2 coefficient (x
3) as well as the negative value of the interaction parameter between x
1 and x
3. These show that x
3 reduces isocyanate conversion and that catalyst loadings may need to be increased slightly in the presence of x
3. This confirms the X
NCO results from experiment-3, where conversion was lower in all formulations than expected. Furthermore, the X
NCO model, whilst adequate, underpredicts at low conversions, as can be seen from the deviation from the x = y curve in
Figure 9A. The model has four fitting parameters as well as the random error parameter and is only influenced by the loading of the catalyst and surfactant-2. The r
2 value of the model, 0.746, is low, and the model is not very robust with a low k-fold r
2 of 0.403. These low values can be explained by the fact that all formulations in experiments 2, 3, and 4 were at values of catalyst loading that should have led to X
NCO values above 0.9. At these values, the response is more sensitive to any experimental errors, either in weighing of components or errors introduced by the FoamPi apparatus used in measuring temperature rise. The FoamPi thermocouple had a relative standard deviation of 3.33% in terms of maximum temperature, and this may explain the variation, which is unexplained by the model.
The t-ratio and actual/predicted density (ρ) are shown in
Figure 9B. The random error parameters, ε, explains the greatest amount of variation in the density model. The density only varies between 25.4 kg m
−3 and 29.6 kg m
−3, and this low variation, much like the X
NCO result, increases the significance of any experimental error. The model has four fitting parameters as well as the random error parameter and is only influenced by the loading of the two surfactants. The density model has the lowest r
2 value of all modelled responses (0.699) and a low k-fold r
2 of 0.481. x
3, surfactant-2 loading, explains the most variation after the random error parameter and is negative, reducing the foam density with increased loading. x
2 is also negative, showing that increasing the loading of either surfactant reduces density (however, this falls below a t-ratio of 2, indicating
p > 0.05 of this coefficient), and the interaction parameter indicates that there is a synergism for decreasing density between the two surfactants. The model underpredicts at low density and overpredicts at high density, observable in the deviation from the x = y curve in
Figure 9B. Although some insights can be taken from this model, it shows the importance of formulating for the generation of foams with a large range of targeted physical properties.
The t-ratio and actual/predicted cell size (d
cell) are shown in
Figure 9C. Cell size is the first of the targeted physical properties. The random error parameter, ε, again explains the highest variance. At the minimum loadings of the three factors, the model predicts a cell size of 602 µm. Cell size is predicted using all three factors as well as two interaction parameters, between the catalyst loading and the surfactant loadings. The model has five parameters as well as the random error parameter. The cell size is reduced by increasing the loading of catalyst as well as surfactant-2, which is as expected. Surfactant-1 has a positive coefficient, indicating that an increase in loading increases cell size. However, the low t-ratio value (1.55) means this coefficient has a
p > 0.05. The low significance of this coefficient may be due to the range used for this surfactant-1. In experiment-2, we determined that cell size did not vary drastically above loadings of 1 PPHP. The range of 0.8 PPHP and 2 PPHP in experiment-4 means that surfactant-1 loading had little effect in the ranges used in this experiment. The high negative t-ratio values of the interaction coefficients between catalyst loading and both surfactants indicate that increasing catalyst loading, whilst increasing the loading of either surfactant, acts synergistically and decreases the cell size. The r
2 value of 0.868 shows a good fit of the model, and the k-fold r
2 = 0.664 shows that the model is reasonably robust. Finally, the model fits well to the x = y curve shown in
Figure 9C, indicating that the model does not over or under predict cell size at any of the values in this experiment.
The t-ratio and actual/predicted open cell fraction (p
eff) are shown in
Figure 9D. The random error, ε, explains most of the variance, and the model predicts that at the minimum range of loadings for the three factors, there is an open cell fraction of 0.655. Open cell fraction is the second of the targeted cell morphology properties and the response is a function of all three of the factors as well as the curvature parameter in x
3. The model has a total of four fitting parameters as well as the random error parameter. The surfactant-2 explains the second highest amount of variance and has a negative coefficient, reducing the open cell fraction of the foams. This is expected as this surfactant is an excellent cell stabiliser used in the production of rigid foams. Surfactant-1 is also an important parameter in the model, with a positive coefficient, indicating that an increase in surfactant-1 increases the open cell content. Again, this is not unexpected as it is a surfactant that promotes open cells. The curvature parameter in surfactant-2 means that as you increase the loading of surfactant-2, the rate of reduction of the number of open cells decreases. This is also expected since there is a hard limit to the fraction of closed cells. An open cell ratio below zero or above one cannot exist in a foam. The negative value of the catalyst coefficient is unexpected as this catalyst promotes the blowing reaction and it would be expected that increasing the rate of blowing reaction would increase the number of open cells. The r
2 of the model is good (0.931) and the k-fold r
2 = 0.900 indicates that this model is highly robust in this dataset. The model also predicts values well within the range of responses, as shown by the overlap between the model fit and the x = y line in
Figure 9D.
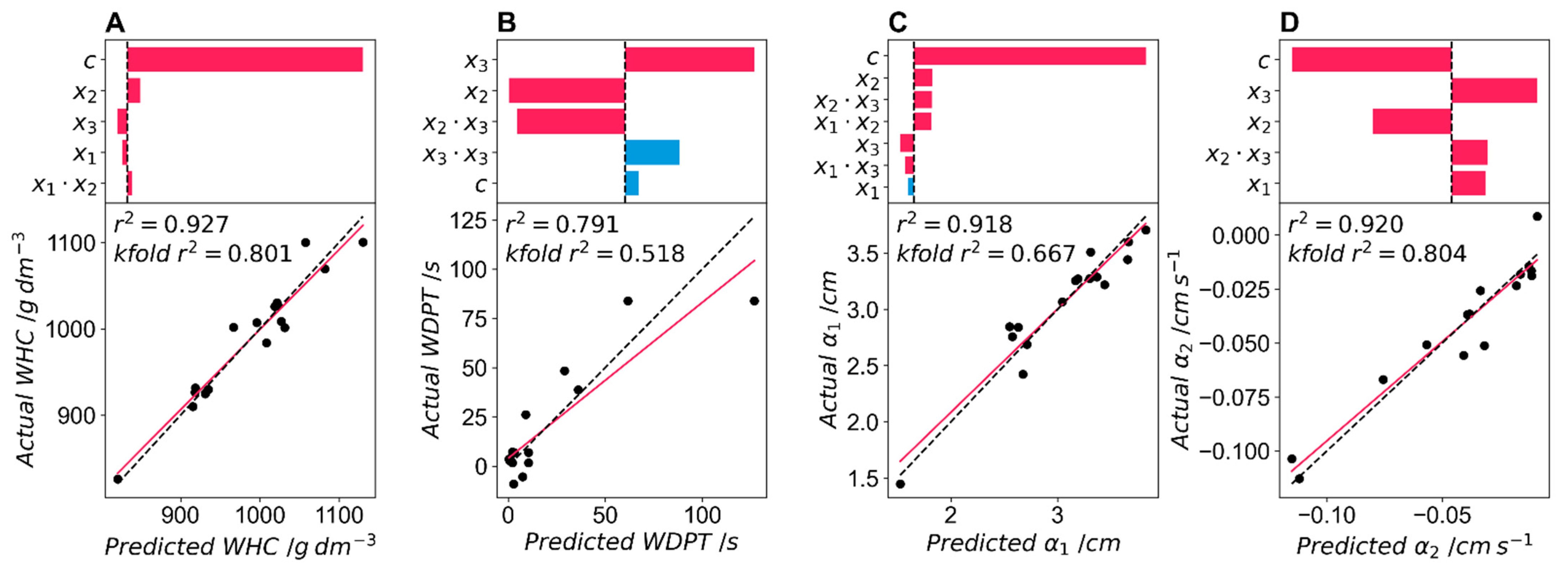
Like
Figure 9,
Figure 10 shows model parameters for each of the hydrodynamic property responses.
The t-ratio and actual/predicted water holding content (WHC) are shown in
Figure 10A. WHC is the first of the targeted hydrodynamic properties. The random error parameter again explains the greatest amount of variance. This translates to a WHC of 1030 g dm
−3. This value is above the theoretical maximum (1000 g dm
−3) and indicates that the foams are likely swelling to a volume above their dry volume (the polymer itself swells in water and the foam struts expand). Therefore, this response WHC is likely acting as a combination response, combining the maximum water holding as well as the maximum swelling of the foam, which may be of interest. Therefore, it is reported as is (g water per dm
−3 dry foam), allowing for values above the threshold of 1000 g dm
−3. The model consists of four fitting parameters as well as a random error parameter. The WHC ranges between 820 g dm
−3 and 1130 g dm
−3, indicating that over 24 h, all formulations, even those with a high number of closed cells, can absorb a large amount of water. The factor explaining the highest amount of variance is surfactant-1 loading, which has a positive coefficient indicating that an increase in surfactant-1 increases WHC. The surfactant-2 coefficient is negative, as is that for the catalyst. This result is similar to the model for open cell fraction and may indicate that open cell fraction may be a good predictor for WHC. The r
2 for the model is very good (0.927) and the k-fold r
2 = 0.801 shows that this model is robust in this dataset. The model also predicts values well within the range of responses, as shown by the overlap between the model fit and the x = y line in
Figure 10A.
The t-ratio and actual/predicted water drop penetration test (WDPT) are shown in
Figure 10B. The WDPT is the second target response for the hydrodynamic properties. It is only a factor of the two surfactant loadings, with the loading of surfactant-2 explaining the most variance. The positive value of this coefficient indicates that increasing surfactant-2 loading increases the WDPT. This is not unexpected as this surfactant increases the number of closed cells, which should increase the time taken for water to penetrate the foam. Surfactant-1 loading has the second highest significance and a positive value, as expected for this surfactant that promotes open cells. The interaction parameter between the two surfactants shows that at high loadings of surfactant-1, surfactant-2 has a lesser effect on the WDPT. The last two factors, the curvature of the surfactant-2 coefficient and the random error parameter, have a t-ratio of less than 2 and therefore a
p > 0.05. This model has the worst fit of the targeted responses, with an r
2 of 0.791, and is the least robust, with a k-fold r
2 of 0.518. This poor fit may be due to the nature of the WDPT, which has a large variation between samples due to the subjectivity of determining the endpoint of the experiment, particularly with formulations with longer WDPT times. For example, one formulation had a mean WDPT of 61.5 s but varied between 14.9 s and 126.7 s in the five repeat droplets. For future experiments, it may be required to increase the number of repeat droplets to reduce this error or use a more robust test. The model also overpredicts the WDPT at high WDPT times, as shown in the deviation from the x = y line in
Figure 10B.
Figure 10C,D show the t-ratio and actual/predicted values for α
1 and α
2 coefficients, the two fitting parameters for the capillarity test. These two parameters are expected to be the most important in predicting plant growth in hydroponic experiments and are the responses of most interest here. The maximum capillary rise required the most complex model to fit the data. This model has six fitting parameters as well as a random error parameter and is influenced by all three of the factors. The random error parameter explains the largest amount of variance and predicts a maximum capillary rise of 3.60 cm at the minimum loadings of the three factors. The two surfactants had the same effects as those seen in the previous hydrodynamic tests, with an increase in surfactant-1 increasing the maximum capillary rise and an increase in surfactant-2 reducing the maximum capillary rise. This again suggests that open cell fraction may influence the maximum capillary rise. The effect of catalyst loading had a t-ratio below 2 and therefore a
p > 0.05, so it had little effect on the maximum capillary rise. The interaction coefficient between all three factors was significant, indicating that at high catalyst loadings, the two surfactants have a larger effect on maximum capillary rise, while surfactant-2 loadings have a smaller effect at high surfactant-1 loadings. The fit for maximum capillary rise is good with a r
2 of 0.918 and is reasonably robust with a k-fold r
2 of 0.667. The fit predicts values of the maximum capillary rise with the fit having little deviation from the y = x curve
Figure 10C.
The greater the absolute value of the rate of water uptake, the greater the curvature in the exponential fit of the water uptake curve and the quicker the time until the maximum capillary rise is reached. The model for the rate of water uptake (α
2) required four fitting parameters as well as a random error and was influenced by all three of the factors. The random error parameter explained the largest part of the variance. The effects of the two surfactants are the same as for the other hydrodynamic properties, with an increase in loading of surfactant-1 increasing the rate of water uptake and an increase in loading of surfactant-2 reducing the rate of water uptake. An increase in catalyst loading decreased the rate of water uptake. The signs of these three coefficients in the model are the same as those that predicted open cell content, suggesting that open cell content may have an important role in predicting the rate of water uptake. The model explains a large fraction of the variance and has an r
2 = 0.920 and a k-fold r
2 = 0.804 suggesting that the model is robust within this dataset. Finally, the model does not over or under predict the rate of water uptake as there is little deviation from the x = y curve and the predicted/actual fit shown in
Figure 10D.
3.5. Effect of Cell Morphology on Hydrodynamic Properties
The results from the modelling of the physical properties showed that the sign and t-ratio of the coefficients in cell morphology models, particularly open cell fraction, and hydrodynamic properties models were often similar. It is therefore worth examining these relationships further.
A generalised model of the form shown in Equation (4) was used to fit the cell morphology parameters (cell size and open cell fraction) to the hydrodynamic properties.
HP is the hydrodynamic parameter of interest (WHC, WDPT, α
1 or α
2) and p
eff and d
cell are the cell morphology parameters. The generalised model was simplified by removing non-significant parameters using the same approach as that used in experiment-4 for modelling foam responses from catalyst and surfactant loadings. This model proved a good fit for the WHC and α
2 properties, but none of the possible models fitted the WDPT and α
1 responses well. On examination, a better fit for those response was an exponential fit of the form shown in Equation (5).
For all models, all the coefficients for cell size (d
cell) were not significant and were dropped from the models. The open cell ratio explained a large proportion of the variation in all four of the hydrodynamic properties. This may be due to the relatively low range of cell sizes in experiment-4 (~550 µm to 700 µm).
Figure 11 shows the four hydrodynamic properties as a function of the open cell fraction.
The WHC is shown in
Figure 11A with a quadratic equation fitted to the data. As the open cell content increases, the WHC also increases. However, at higher open cell content, this reaches a maximum and the WHC does not appreciably increase further. The r
2 of the fit is 0.799. The WDPT as a function of p
eff is shown in
Figure 11B with the exponential fit of the data. The exponential increase in WDPT at low open cell fraction is an important finding showing that there is a rapid increase in WDPT or “hydrophobicity” of the foam as we introduce closed cells that only take up water slowly following swelling of the polymer. The exponential fit had an r
2 of 0.804. Practically, at these high WDPT (>60 s), they would be classed as “slightly hydrophobic” soils [
36] and would likely not be suitable for use as synthetic growing media. We can therefore conclude from the model that at p
eff < 0.146 foams (at which WDPT = 60 s) are not suitable as synthetic growing media.
Figure 11C shows the maximum capillary rise (α
1) data as a function of open cell ratio and shows the exponential fit to the data. This exponential fit with a r
2 of 0.886 outperformed the best fit of the generalised model (Equation (4)) which had an r
2 of 0.599. This model shows that increasing the p
eff value increases the maximum capillary rise to a maximum (3.40 cm) at an open cell fraction of 0.336 and increasing the open cell fraction further does not increase the maximum capillary rise further.
Figure 11D shows the rate of water uptake (α
2) data as a function of open cell ratio and shows a linear fit of the open cell ratio to α
2. This fit explains a significant amount of the variance with an r
2 of 0.882. This finding indicates that any closed cells impede the pathway for capillary action, reducing the rate at which water can be taken up by the foam via capillary action. The significance of the results shown in
Figure 11A,C,D is that there may be a trade-off between the rate of water uptake, maximum capillary rise and water holding content, with all three of these properties being highly influenced by the open cell fraction. It is not known which of these growing media properties is more important in regulating plant growth in hydroponic systems. However, with the models and formulations developed in this work, these experiments can now be done.