Electrochemically Enhanced Delivery of Pemetrexed from Electroactive Hydrogels
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis
2.1.1. Preparation of Polymerizable CS Derivatives
2.1.2. Preparation of Non-Conductive Hydrogels
2.1.3. Preparation of Conductive Hydrogels
2.2. Characterization
2.2.1. UV-Vis Spectroscopy
2.2.2. Attenuated Total Reflectance Fourier Transform Infrared (ATR-FTIR) Spectroscopy
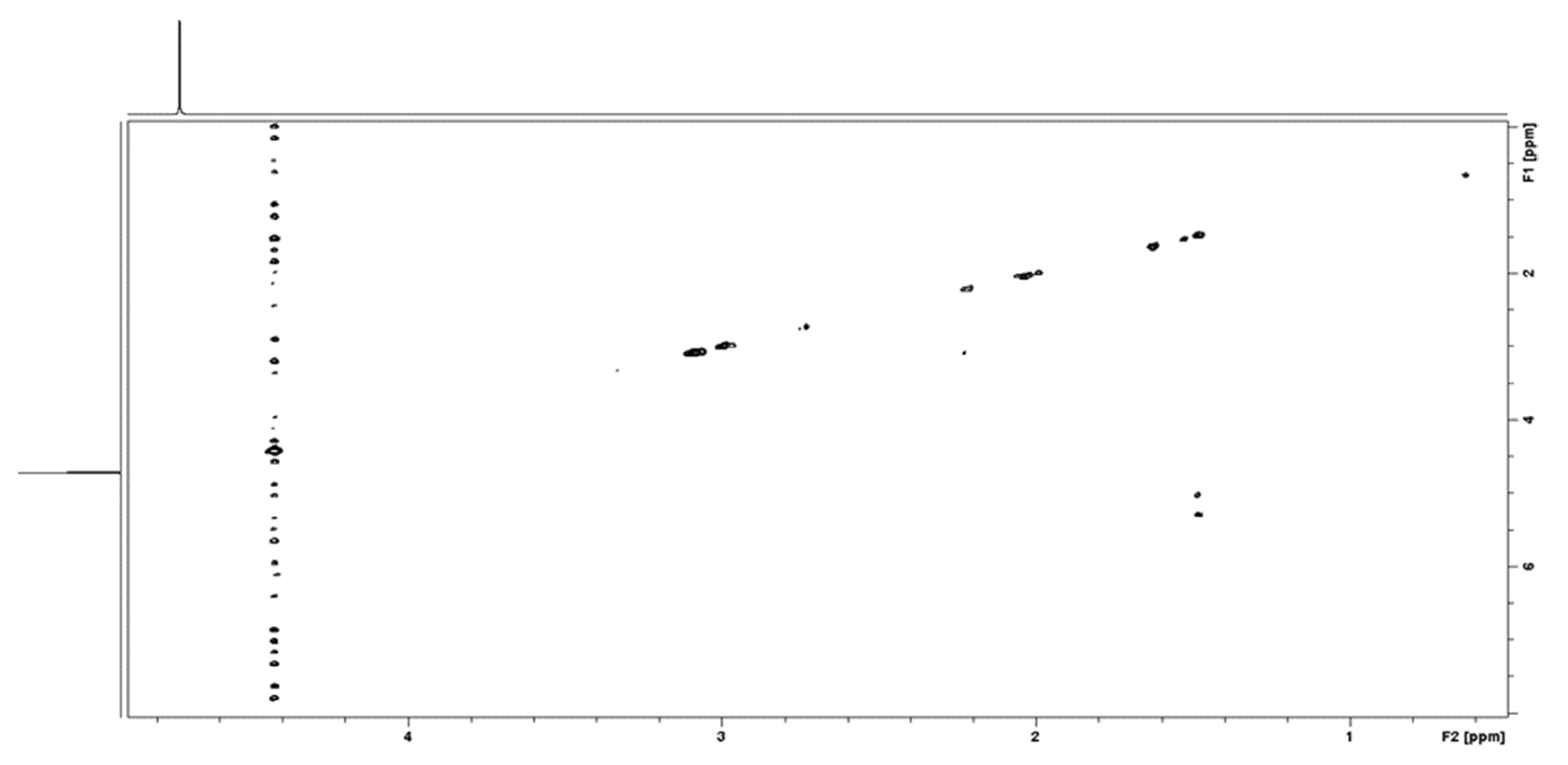
2.2.3. Solution State 1H NMR Spectroscopy
2.2.4. Solid State 13C NMR Spectroscopy
2.2.5. Scanning Electron Microscopy (SEM)
2.2.6. Conductivity Determination
2.2.7. X-ray Diffraction (XRD)
2.2.8. Thermogravimetric Analysis (TGA)
2.2.9. Swelling Studies
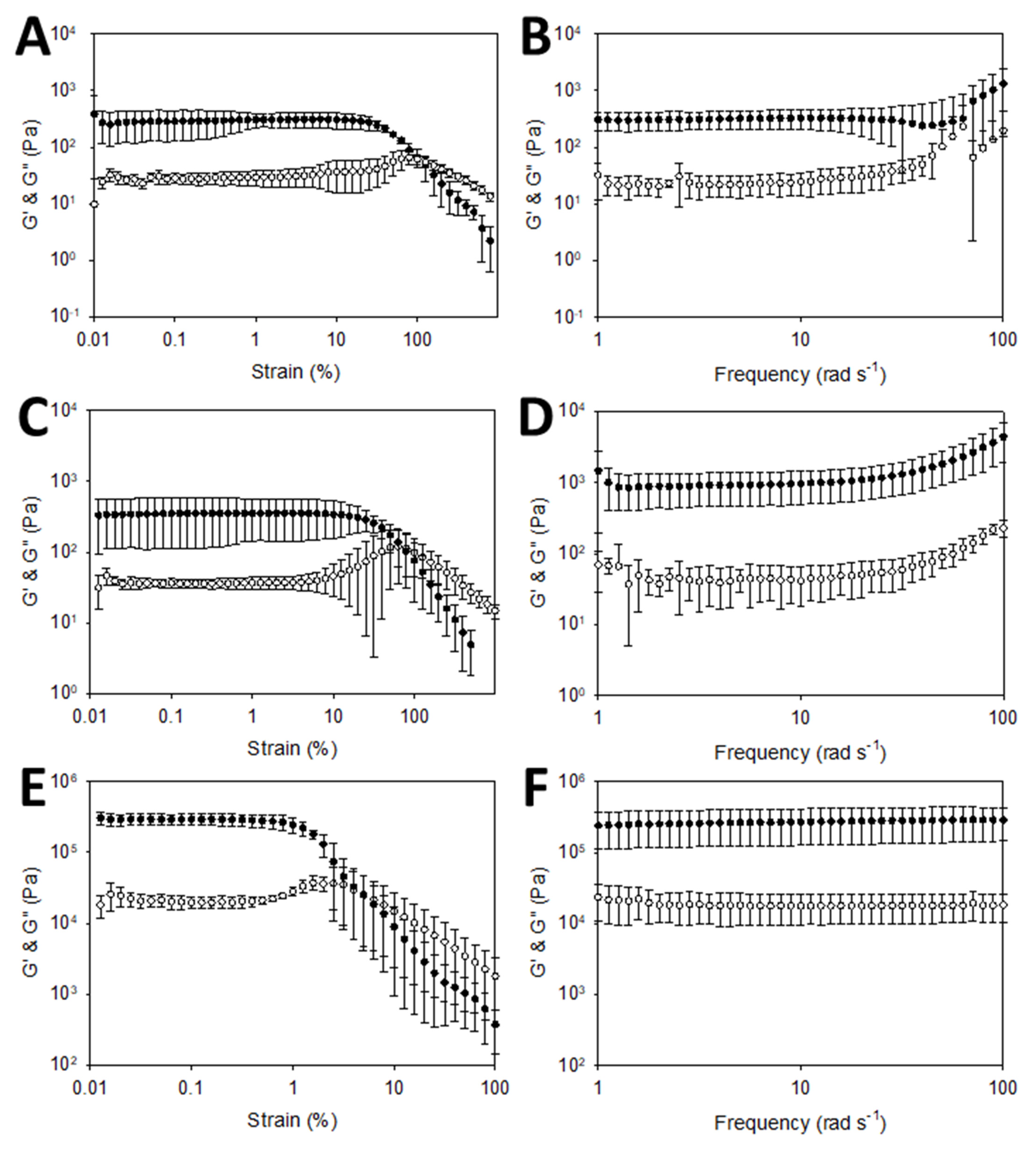
2.2.10. Rheological Characterization
2.3. In Silico and In Vitro Validation
2.3.1. In Silico Toxicity Screening Studies
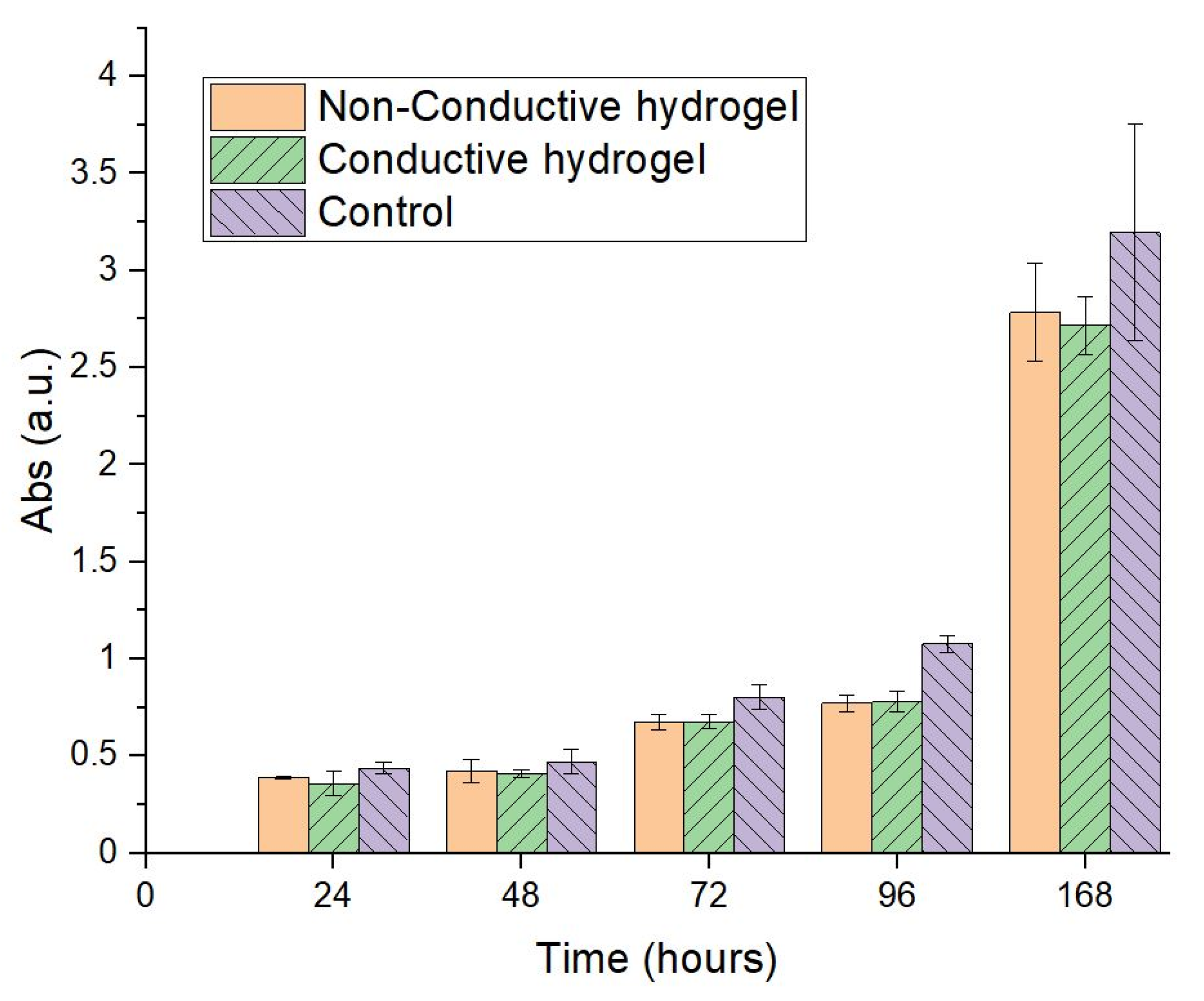
2.3.2. In Vitro Cell Culture Studies
2.3.3. In Vitro Drug Loading
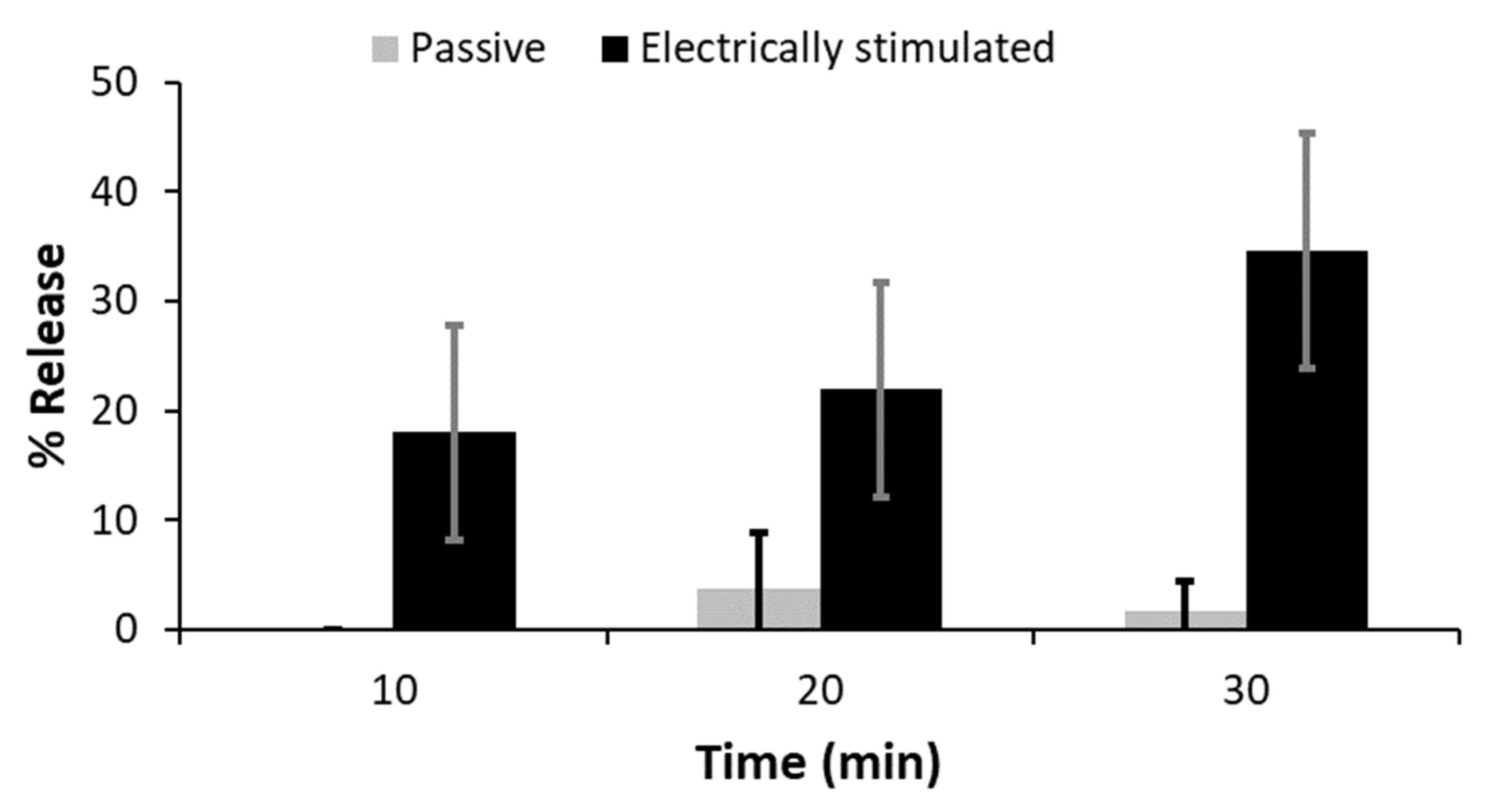
2.3.4. In Vitro Drug Delivery Studies
3. Results and Discussion
3.1. CS Modification and Characterization
3.2. Hydrogel Preparation and Characterization
3.3. In Silico and In Vitro Validation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mignani, S.; El Kazzouli, S.; Bousmina, M.; Majoral, J.P. Dendrimer space concept for innovative nanomedicine: A futuristic vision for medicinal chemistry. Prog. Polym. Sci. 2013, 38, 993–1008. [Google Scholar] [CrossRef]
- Samuelsen, A.B. The traditional uses, chemical constituents and biological activities of Plantago major L. A review. J. Ethnopharmacol. 2000, 71, 1–21. [Google Scholar] [CrossRef]
- Li, W.Q.; Guo, H.F.; Li, L.Y.; Zhang, Y.F.; Cui, J.W. The promising role of antibody drug conjugate in cancer therapy: Combining targeting ability with cytotoxicity effectively. Cancer Med. 2021, 10, 4677–4696. [Google Scholar] [CrossRef] [PubMed]
- Grainger, D.W. Controlled-release and local delivery of therapeutic antibodies. Expert Opin. Biol. Ther. 2004, 4, 1029–1044. [Google Scholar] [CrossRef] [PubMed]
- Kuang, T.R.; Liu, Y.R.; Gong, T.T.; Peng, X.F.; Hu, X.L.; Yu, Z.Q. Enzyme-responsive Nanoparticles for Anticancer Drug Delivery. Curr. Nanosci. 2016, 12, 38–46. [Google Scholar] [CrossRef]
- Linsley, C.S.; Wu, B.M. Recent advances in light-responsive on-demand drug- delivery systems. Ther. Deliv. 2017, 8, 89–107. [Google Scholar] [CrossRef]
- Yoshida, T.; Lai, T.C.; Kwon, G.S.; Sako, K. pH-and ion-sensitive polymers for drug delivery. Expert Opin. Drug Deliv. 2013, 10, 1497–1513. [Google Scholar] [CrossRef]
- Bikram, M.; West, J.L. Thermo-responsive systems for controlled drug delivery. Expert Opin. Drug Deliv. 2008, 5, 1077–1091. [Google Scholar] [CrossRef]
- Szunerits, S.; Teodorescu, F.; Boukherroub, R. Electrochemically triggered release of drugs. Eur. Polym. J. 2016, 83, 467–477. [Google Scholar] [CrossRef]
- Guo, J.; Fan, D. Electrically Controlled Biochemical Release from Micro/Nanostructures for in vitro and in vivo Applications: A Review. ChemNanoMat 2018, 4, 1023–1038. [Google Scholar] [CrossRef]
- Zhang, Z.N.; Zhang, R.; Rajagopalan, R.; Tang, Z.; Sun, D.; Wang, H.Y.; Tang, Y.G. A high-capacity self-sacrificial additive based on electroactive sodiated carbonyl groups for sodium-ion batteries. Chem. Commun. 2022, 58, 8702–8705. [Google Scholar] [CrossRef]
- Zhao, Y.; Tavaresa, A.C.; Gauthier, M.A. Nano-engineered electro-responsive drug delivery systems. J. Mater. Chem. B 2016, 4, 3019–3030. [Google Scholar] [CrossRef]
- Zinger, B.; Miller, L.L. Timed release of chemicals from polypyrrole films. J. Am. Chem. Soc. 1984, 106, 6861–6863. [Google Scholar] [CrossRef]
- Kim, D.H.; Richardson-Burns, S.M.; Hendricks, J.L.; Sequera, C.; Martin, D.C. Effect of Immobilized Nerve Growth Factor on Conductive Polymers: Electrical Properties and Cellular Response. Adv. Funct. Mater. 2007, 17, 79–86. [Google Scholar] [CrossRef]
- Minisy, I.M.; Salahuddin, N.A.; Ayad, M.M. In vitro release study of ketoprofen-loaded chitosan/polyaniline nanofibers. Polym. Bull. 2020, 78, 5609–5622. [Google Scholar] [CrossRef]
- Bolla, P.K.; Rodriguez, V.A.; Kalhapure, R.S.; Kolli, C.S.; Andrews, S.; Renukuntla, J. A review on pH and temperature responsive gels and other less explored drug delivery systems. J. Drug Deliv. Sci. Technol. 2018, 46, 416–435. [Google Scholar] [CrossRef]
- Ruiz, A.L.; Ramirez, A.; McEnnis, K. Single and Multiple Stimuli-Responsive Polymer Particles for Controlled Drug Delivery. Pharmaceutics 2022, 14, 421. [Google Scholar] [CrossRef]
- Shen, M.Y.; Yuran, S.; Aviv, Y.; Ayalew, H.; Luo, C.H.; Tsai, Y.H.; Reches, M.; Yu, H.H.; Shenhar, R. Electrically Responsive, Nanopatterned Surfaces for Triggered Delivery of Biologically Active Molecules into Cells. ACS Appl. Mater. Interfaces 2019, 11, 1201–1208. [Google Scholar] [CrossRef]
- Peters, J.T.; Wechsler, M.E.; Peppas, N.A. Advanced biomedical hydrogels: Molecular architecture and its impact on medical applications. Regen. Biomater. 2021, 8, rbab060. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Yu, J.C.; Kahkoska, A.R.; Wang, J.Q.; Buse, J.B.; Gu, Z. Advances in transdermal insulin delivery. Adv. Drug Deliv. Rev. 2019, 139, 51–70. [Google Scholar] [CrossRef]
- Taghipour, Y.D.; Hokmabad, V.R.; Del Bakhshayesh, A.R.; Asadi, N.; Salehi, R.; Nasrabadi, H.T. The Application of Hydrogels Based on Natural Polymers for Tissue Engineering. Curr. Med. Chem. 2020, 27, 2658–2680. [Google Scholar]
- Ghosh, S. Recent research and development in synthetic polymer-based drug delivery systems. J. Chem. Res. 2004, 4, 241–246. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Gull, N.; Khan, S.M.; Islam, A.; Butt, M.T.Z. Hydrogels used for Biomedical Applications. In Bio Monomers for Green Polymeric Composite Materials, 1st ed.; Visakh, P.M., Bayraktar, O., Menon, G., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2019; pp. 175–199. [Google Scholar]
- Cascone, S.; Lamberti, G. Hydrogel-based commercial products for biomedical applications: A review. Int. J. Pharm. 2020, 573, 118803. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.Y.; Yu, L.; Ding, J.D. PEG-based thermosensitive and biodegradable hydrogels. Acta Biomater. 2021, 128, 42–59. [Google Scholar] [CrossRef]
- Alexander, A.; Ajazuddin, B.; Khan, J.; Saraf, S.; Saraf, S. Polyethylene glycol (PEG)-Poly(N-isopropylacrylamide) (PNIPAAm) based thermosensitive injectable hydrogels for biomedical applications. Eur. J. Pharm. Biopharm. 2014, 88, 575–585. [Google Scholar] [CrossRef]
- Bashir, S.; Hina, M.; Iqbal, J.; Rajpar, A.H.; Mujtaba, M.A.; Alghamdi, N.A.; Wageh, S.; Ramesh, K.; Ramesh, S. Fundamental Concepts of Hydrogels: Synthesis, Properties, and Their Applications. Polymers 2020, 12, 2702. [Google Scholar] [CrossRef]
- Li, Z.L.; Lin, Z.Q. Recent advances in polysaccharide-based hydrogels for synthesis and applications. Aggregate 2021, 2, e21. [Google Scholar] [CrossRef]
- Liu, Z.H.; Jiao, Y.P.; Wang, Y.F.; Zhou, C.R.; Zhang, Z.Y. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, C.A.; Alnaief, M.; Smirnova, I. Polysaccharide-based aerogels-Promising biodegradable carriers for drug delivery systems. Carbohydr. Polym. 2011, 86, 1425–1438. [Google Scholar] [CrossRef]
- Lei, K.; Li, Z.; Zhu, D.D.; Sun, C.Y.; Sun, Y.L.; Yang, C.C.; Zheng, Z.; Wang, X.L. Polysaccharide-based recoverable double-network hydrogel with high strength and self-healing properties. J. Mater. Chem. B 2020, 8, 794–802. [Google Scholar] [CrossRef]
- Miao, T.X.; Wang, J.Q.; Zeng, Y.; Liu, G.; Chen, X.Y. Polysaccharide-Based Controlled Release Systems for Therapeutics Delivery and Tissue Engineering: From Bench to Bedside. Adv. Sci. 2018, 5, 1700513. [Google Scholar] [CrossRef]
- Coviello, T.; Matricardi, P.; Marianecci, C.; Alhaique, F. Polysaccharide hydrogels for modified release formulations. J. Cont. Release 2007, 119, 5–24. [Google Scholar] [CrossRef]
- Raveendran, S.; Yoshida, Y.; Maekawa, T.; Kumar, S. Pharmaceutically versatile sulfated polysaccharide based bionano platforms. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 605–626. [Google Scholar] [CrossRef]
- Cheung, R.C.F.; Ng, T.B.; Wong, J.H.; Chan, W.Y. Chitosan: An Update on Potential Biomedical and Pharmaceutical Applications. Mar. Drugs 2015, 13, 5156–5186. [Google Scholar] [CrossRef]
- Zhang, Z.X.; Liow, S.S.; Xue, K.; Zhang, X.K.; Li, Z.B.; Loh, X.J. Autonomous Chitosan-Based Self-Healing Hydrogel Formed through Noncovalent Interactions. ACS Appl. Polym. Mat. 2019, 1, 1769–1777. [Google Scholar] [CrossRef]
- Ahmadi, F.; Oveisi, Z.; Samani, S.M.; Amoozgar, Z. Chitosan based hydrogels: Characteristics and pharmaceutical applications. Res. Pharm. Sci. 2015, 10, 1–16. [Google Scholar]
- Pella, M.C.G.; Lima-Tenorio, M.K.; Neto, E.T.T.; Guilherme, M.R.; Muniz, E.C.; Rubira, A.F. Chitosan-based hydrogels: From preparation to biomedical applications. Carbohydr. Polym. 2018, 196, 233–245. [Google Scholar] [CrossRef]
- Racine, L.; Texier, I.; Auzely-Velty, R. Chitosan-based hydrogels: Recent design concepts to tailor properties and functions. Polym. Int. 2017, 66, 981–998. [Google Scholar] [CrossRef]
- Wang, J.; Vermerris, W. Antimicrobial Nanomaterials Derived from Natural Products-A Review. J. Mater. 2016, 9, 255. [Google Scholar] [CrossRef]
- DellaGreca, M.; Zuppolini, S.; Zarrelli, A. Isolation of lignans as seed germination and plant growth inhibitors from Mediterranean plants and chemical synthesis of some analogues. Phytochem. Rev. 2013, 12, 717–731. [Google Scholar] [CrossRef]
- Kim, S.; Jang, L.K.; Jang, M.; Lee, S.; Hardy, J.G.; Lee, J.Y. Electrically Conductive Polydopamine-Polypyrrole as High Performance Biomaterials for Cell Stimulation in Vitro and Electrical Signal Recording in Vivo. ACS Appl. Mater. Interfaces 2018, 10, 33032–33042. [Google Scholar] [CrossRef] [PubMed]
- Hendricks, J.L.; Chikar, J.A.; Crumling, M.A.; Raphael, Y.; Martin, D.C. Localized cell and drug delivery for auditory prostheses. Hear. Res. 2008, 242, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Chikar, J.A.; Hendricks, J.L.; Richardson-Burns, S.M.; Raphael, Y.; Pfings, B.E.; Martin, D.C. The use of a dual PEDOT and RGD-functionalized alginate hydrogel coating to provide sustained drug delivery and improved cochlear implant function. Biomaterials 2012, 33, 1982–1990. [Google Scholar] [CrossRef] [PubMed]
- Ashton, M.D.; Cooper, P.A.; Municoy, S.; Desimone, M.F.; Cheneler, D.; Shnyder, S.D.; Hardy, J.G. Controlled Bioactive Delivery Using Degradable Electroactive Polymers. Biomacromolecules 2022, 23, 3031–3040. [Google Scholar] [CrossRef]
- Molino, P.J.; Innis, P.C.; Higgins, M.J.; Kapsa, R.M.I.; Wallace, G.G. Influence of biopolymer loading on the physiochemical and electrochemical properties of inherently conducting polymer biomaterials. Synth. Met. 2015, 200, 40–47. [Google Scholar] [CrossRef]
- Higgins, M.J.; Molino, P.J.; Yue, Z.; Wallace, G.G. Organic Conducting Polymer–Protein Interactions. Chem. Mater. 2012, 24, 828–839. [Google Scholar] [CrossRef]
- Kim, S.; Jang, Y.; Jang, M.; Lim, A.; Hardy, J.G.; Park, H.S.; Lee, J.Y. Versatile biomimetic conductive polypyrrole films doped with hyaluronic acid of different molecular weights. Acta Biomater. 2018, 80, 258–268. [Google Scholar] [CrossRef]
- Svirskis, D.; Travas-Sejdic, J.; Rodgers, A.; Garg, S. Electrochemically controlled drug delivery based on intrinsically conducting polymers. J. Control. Release 2010, 146, 6–15. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, X.; Guo, B.; Ma, P.X. Multifunctional Interpenetrating Polymer Network Hydrogels Based on Methacrylated Alginate for the Delivery of Small Molecule Drugs and Sustained Release of Protein. Biomacromolecules 2014, 15, 3246–3252. [Google Scholar] [CrossRef]
- Qu, J.; Liang, Y.; Shi, M.; Guo, B.; Gao, Y.; Yin, Z. Biocompatible conductive hydrogels based on dextran and aniline trimer as electro-responsive drug delivery system for localized drug release. Int. J. Biol. Macromol. 2019, 140, 255–264. [Google Scholar] [CrossRef]
- Qu, J.; Zhao, X.; Ma, P.X.; Guo, B. Injectable antibacterial conductive hydrogels with dual response to an electric field and pH for localized “smart” drug release. Acta Biomater. 2018, 72, 55–69. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, L.; Guo, B.; Ma, P.X. Cytocompatible injectable carboxymethyl chitosan/N-isopropylacrylamide hydrogels for localized drug delivery. Carbohydr. Polym. 2014, 103, 110–118. [Google Scholar] [CrossRef]
- Catt, K.; Li, D.X.; Hoang, V.; Beard, R.; Cui, X.T. Self-powered therapeutic release from conducting polymer/graphene oxide films on magnesium. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2495–2503. [Google Scholar] [CrossRef]
- Sundararajan, R. Electroporation-Based Therapies for Cancer, From Basics to Clinical Applications, 1st ed.; Woodhead Publishing: Cambridge, UK, 2014. [Google Scholar]
- Mir, L.M.; Orlowski, S. The Basis of Electrochemotherapy. In Electrochemotherapy, Electrogenetherapy, and Transdermal Drug Delivery. Methods in Molecular Medicine; Jaroszeski, M.J., Heller, R., Gilbert, R., Eds.; Humana Press: Totowa, NJ, USA, 2000; Volume 37, pp. 99–117. [Google Scholar]
- Miklavčič, D.; Mali, B.; Kos, B.; Heller, R.; Serša, G. Electrochemotherapy: From the drawing board into medical practice. BioMed. Eng. Online 2014, 13, 29. [Google Scholar] [CrossRef]
- Esmaeili, N.; Friebe, M. Electrochemotherapy: A Review of Current Status, Alternative IGP Approaches, and Future Perspectives. J. Healthc. Eng. 2019, 2019, 2784516. [Google Scholar] [CrossRef]
- Tellado, M.; Mir, L.M.; Maglietti, F. Veterinary Guidelines for Electrochemotherapy of Superficial Tumors. Front. Vet. Sci. 2022, 9, 868989. [Google Scholar] [CrossRef]
- Jamsek, C.; Sersa, G.; Bosnjak, M.; Groselj, A. Long term response of electrochemotherapy with reduced dose of bleomycin in elderly patients with head and neck non-melanoma skin cancer. Radiol. Oncol. 2020, 54, 79–85. [Google Scholar] [CrossRef]
- McMillan, A.; McElroy, L.; O’Toole, L.; Matteucci, P.; Totty, J.P. Electrochemotherapy vs radiotherapy in the treatment of primary cutaneous malignancies or cutaneous metastases from primary solid organ malignancies: A protocol for a systematic review and meta-analysis. J. Surg. Protoc. Res. Methodol. 2022, 2022, snac005. [Google Scholar] [CrossRef]
- Sersa, G.; Ursica, K.; Cemazar, M.; Heller, R.; Bosnjak, M.; Campana, L.G. Biological factors of the tumour response to electrochemotherapy: Review of the evidence and a research roadmap. Eur. J. Surg. Oncol. 2021, 47, 1836–1846. [Google Scholar] [CrossRef]
- Sundararajan, R.; Mittal, L.; Camarillo, I.G. Electrochemotherapy Modulates Mammary Tumor Growth in Rats on a Western Diet Supplemented with Curcumin. Biomedicines 2020, 8, 498. [Google Scholar] [CrossRef] [PubMed]
- Fiorentzis, M.; Katopodis, P.; Kalirai, H.; Seitz, B.; Viestenz, A.; Coupland, S.E. Image Analysis of 3D Conjunctival Melanoma Cell Cultures Following Electrochemotherapy. Biomedicines 2020, 8, 158. [Google Scholar] [CrossRef] [PubMed]
- Esposito, E.; Siani, C.; Pace, U.; Costanzo, R.; di Giacomo, R. Debulking mastectomy with electrochemotherapy: A case report of no surgery approach to recurrent breast cancer. Transl. Cancer Res. 2021, 10, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Sersa, G.; Stabuc, B.; Cemazar, M.; Miklavcic, D.; Rudolf, Z. Electrochemotherapy with cisplatin: Clinical experience in malignant melanoma patients. Clin. Cancer Res. 2000, 6, 863–867. [Google Scholar]
- Šatkauskas, S.; Batiuškaitė, D.; Šalomskaitė-Davalgienė, S.; Venslauskas, M.S. Effectiveness of tumor electrochemotherapy as a function of electric pulse strength and duration. Bioelectrochemestry 2005, 65, 105–111. [Google Scholar] [CrossRef]
- Riva, G.; Salonia, L.; Fassone, E.; Sapino, S.; Piano, F.; Pecorari, G. Quality of Life in Electrochemotherapy for Cutaneous and Mucosal Head and Neck Tumors. J. Clin. Med. 2021, 10, 4366. [Google Scholar] [CrossRef]
- Rega, D.; Granata, V.; Petrillo, A.; Pace, U.; Di Marzo, M.; Fusco, R.; D’Alessio, V.; Nasti, G.; Romano, C.; Avallone, A.; et al. Electrochemotherapy of Primary Colon Rectum Cancer and Local Recurrence: Case Report and Prospective Analysis. J. Clin. Med. 2022, 11, 2745. [Google Scholar] [CrossRef]
- Coriat, R.; Pellat, A. Updates on the Treatment of Pancreatic Diseases: Focus on Surgery, Electrochemotherapy and Rituximab. J. Clin. Med. 2022, 11, 239. [Google Scholar] [CrossRef]
- Izzo, F.; Granata, V.; Fusco, R.; D’Alessio, V.; Petrillo, A.; Lastoria, S.; Piccirillo, M.; Albino, V.; Belli, A.; Nasti, G.; et al. A Multicenter Randomized Controlled Prospective Study to Assess Efficacy of Laparoscopic Electrochemotherapy in the Treatment of Locally Advanced Pancreatic Cancer. J. Clin. Med. 2021, 10, 4011. [Google Scholar] [CrossRef]
- Izzo, F.; Granata, V.; Fusco, R.; D’Alessio, V.; Petrillo, A.; Lastoria, S.; Piccirillo, M.; Albino, V.; Belli, A.; Tafuto, S.; et al. Clinical Phase I/II Study: Local Disease Control and Survival in Locally Advanced Pancreatic Cancer Treated with Electrochemotherapy. J. Clin. Med. 2021, 10, 1305. [Google Scholar] [CrossRef]
- Kaur, G.; Adhikari, R.; Cass, P.; Bown, M.; Gunatillake, P. Electrically conductive polymers and composites for biomedical applications. RSC Adv. 2015, 5, 37553–37567. [Google Scholar] [CrossRef]
- Zhan, C.X.; Yu, G.Q.; Lu, Y.; Wang, L.Y.; Wujcik, E.; Wei, S.Y. Conductive polymer nanocomposites: A critical review of modern advanced devices. J. Mater. Chem. C 2017, 5, 1569–1585. [Google Scholar] [CrossRef]
- Antezana, P.E.; Municoy, S.; Pérez, C.J.; Desimone, M.F. Collagen Hydrogels Loaded with Silver Nanoparticles and Cannabis Sativa Oil. Antibiotics 2021, 10, 142. [Google Scholar] [CrossRef]
- Echazú, M.I.A.; Olivetti, C.E.; Peralta, I.; Alonso, M.R.; Anesini, C.; Perez, C.J.; Alvarez, G.S.; Desimone, M.F. Development of pH-responsive biopolymer-silica composites loaded with Larrea divaricata Cav. extract with antioxidant activity. Colloids Surf. B Biointerfaces 2018, 169, 82–91. [Google Scholar] [CrossRef]
- Municoy, S.; Antezana, P.E.; Pérez, C.J.; Bellino, M.G.; Desimone, M.F. Tuning the antimicrobial activity of collagen biomaterials through a liposomal approach. J. Appl. Polym. Sci. 2021, 138, 50330. [Google Scholar] [CrossRef]
- Milosevic, M. Internal Reflection and ATR Spectroscopy; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012. [Google Scholar]
- Grdadolnik, J. ATR-FTIR Spectroscopy: Its advantages and limitations. Acta Chim. Slov. 2002, 49, 631–642. [Google Scholar]
- Kaur, H.; Rana, B.; Tomar, D.; Kaur, S.; Jena, K.C. Fundamentals of ATR-FTIR Spectroscopy and Its Role for Probing In-Situ Molecular-Level Interactions. In Modern Techniques of Spectroscopy. Progress in Optical Science and Photonics; Singh, D.K., Pradhan, M., Materny, A., Eds.; Springer: Singapore, 2021; Volume 13. [Google Scholar]
- Shah, S.A.S.; Firlak, M.; Berrow, S.R.; Halcovitch, N.R.; Baldock, S.J.; Yousafzai, B.M.; Hathout, R.M.; Hardy, J.G. Electrochemically Enhanced Drug Delivery Using Polypyrrole Films. Materials 2018, 11, 1123. [Google Scholar] [CrossRef]
- Kasaai, M.R. Determination of the degree of N-acetylation for chitin and chitosan by various NMR spectroscopy techniques: A review. Carbohydr. Polym. 2010, 79, 801–810. [Google Scholar] [CrossRef]
- Lavertu, M.; Xia, Z.; Serreqi, A.N.; Berrada, M.; Rodrigues, A.; Wang, D.; Buschmann, M.D.; Gupta, A. A validated 1H NMR method for the degree of deacetylation of chitosan. J. Pharm. Biomed. Anal. 2003, 32, 1149–1158. [Google Scholar] [CrossRef]
- Kumirska, J.; Czerwicka, M.; Kaczyński, Z.; Bychowska, A.; Brzozowski, K.; Thöming, J.; Stepnowski, P. Application of Spectroscopic Methods for Structural Analysis of Chitin and Chitosan. Mar. Drugs 2010, 8, 1567–1636. [Google Scholar] [CrossRef]
- Ma, G.; Zhang, X.; Han, J.; Song, G.; Nie, J. Photo-polymeriable chitosan derivative prepared by Michael reaction of chitosan and polyethylene glycol diacrylate (PEGDA). Int. J. Biol. Macromol. 2009, 45, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Capitani, D.; De Angelis, A.A.; Crescenzi, V.; Masci, G.; Segre, A.L. NMR study of a novel chitosan-based hydrogel. Carbohydr. Polym. 2001, 45, 245–252. [Google Scholar] [CrossRef]
- Capitani, D.; Crescenzi, V.; Segre, A.L. Water in Hydrogels. An NMR Study of Water/Polymer Interactions in Weakly Cross-Linked Chitosan Networks. Macromolecules 2001, 34, 4136–4144. [Google Scholar] [CrossRef]
- Li, Q.; Yang, D.; Ma, G.; Xu, Q.; Chen, X.; Lu, F.; Nie, J. Synthesis and characterization of chitosan-based hydrogels. Int. J. Biol. Macromol. 2009, 44, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Nokab, M.E.H.E.; van der Wel, P.C.A. Use of solid-state NMR spectroscopy for investigating polysaccharide-based hydrogels: A review. Carbohydr. Polym. 2020, 240, 116276. [Google Scholar] [CrossRef]
- Štiglic, A.D.; Kargl, R.; Beaumont, M.; Strauss, C.; Makuc, D.; Egger, D.; Plavec, J.; Rojas, O.J.; Kleinschek, K.S.; Mohan, T. Influence of Charge and Heat on the Mechanical Properties of Scaffolds from Ionic Complexation of Chitosan and Carboxymethyl Cellulose. ACS Biomater. Sci. Eng. 2021, 7, 3618–3632. [Google Scholar] [CrossRef]
- Gartner, C.; Löpez, B.L.; Sierra, L.; Graf, R.; Spiess, H.W.; Gaborieau, M. Interplay between Structure and Dynamics in Chitosan Films Investigated with Solid-State NMR, Dynamic Mechanical Analysis, and X-ray Diffraction. Biomacromolecules 2011, 12, 1380–1386. [Google Scholar] [CrossRef]
- Heux, L.; Brugnerotto, J.; Desbrières, J.; Versali, M.F.; Rinaudo, M. Solid state NMR for determination of degree of acetylation of chitin and chitosan. Biomacromolecules 2000, 1, 746–751. [Google Scholar] [CrossRef]
- Devreux, F.; Bidan, G.; Syed, A.A.; Tsintavis, C. Solid state 13C NMR in conducting polymers. J. Phys. 1985, 46, 1595–1601. [Google Scholar] [CrossRef]
- de Alvarenga, E.S. Characterization and Properties of Chitosan. In Biotechnology of Biopolymers, 1st ed.; Elnashar, M., Ed.; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Kasaai, M.R. The Use of Various Types of NMR and IR Spectroscopy for Structural Characterization of Chitin and Chitosan. In Chitin, Chitosan, Oligosaccharides and Their Derivatives, 1st ed.; Kim, S.-K., Ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar] [CrossRef]
- Forsyth, M.; Truong, V.T.; Smith, M.E. Structural characterization of conducting polypyrrole using 13C cross-polarization/magic-angle spinning solid-state nuclear magnetic resonance spectroscopy. Polymer 1994, 35, 1593–1601. [Google Scholar] [CrossRef]
- Souto-Maior, R.M.; Tavares, M.I.B.; Monteiro, E.E.C. Solid State 13C NMR Study of Methyl Methacrylate-Methacrylic Acid Copolymers. Ann. Magn. Reson. 2005, 4, 69–72. [Google Scholar]
- Bu, Y.; Xu, H.X.; Li, X.; Xu, W.J.; Yin, Y.X.; Dai, H.L.; Wang, X.B.; Huang, Z.J.; Xu, P.H. A conductive sodium alginate and carboxymethyl chitosan hydrogel doped with polypyrrole for peripheral nerve regeneration. RSC Adv. 2018, 8, 10806–10817. [Google Scholar] [CrossRef]
- Chalmers, E.; Lee, H.; Zhu, C.; Liu, X. Increasing the Conductivity and Adhesion of Polypyrrole Hydrogels with Electropolymerized Polydopamine. Chem. Mater. 2020, 32, 234–244. [Google Scholar] [CrossRef]
- Distler, T.; Polley, C.; Shi, F.; Schneidereit, D.; Ashton, M.D.; Friedrich, O.; Kolb, J.F.; Hardy, J.G.; Detsch, R.; Seitz, H.; et al. Electrically Conductive and 3D-Printable Oxidized Alginate-Gelatin Polypyrrole: PSS Hydrogels for Tissue Engineering. Adv. Healthc. Mater. 2021, 10, 2001876. [Google Scholar] [CrossRef]
- Manousiouthakis, E.; Park, J.; Hardy, J.G.; Lee, J.Y.; Schmidt, C.E. Towards the translation of electroconductive organic materials for regeneration of neural tissues. Acta Biomater. 2022, 139, 22–42. [Google Scholar] [CrossRef]
- Alemdar, N. Synthesis of chitosan-based hydrogel by using photopolymerization technique. Anadolu Univ. J. Sci. Technol. A-Appl. Sci. Eng. 2016, 17, 391–400. [Google Scholar] [CrossRef]
- Bober, P.; Gavrilov, N.; Kovalcik, A.; Mičušík, M.; Unterweger, C.; Pašti, I.A.; Šeděnková, I.; Acharya, U.; Pfleger, J.; Filippov, S.K.; et al. Electrochemical properties of lignin/polypyrrole composites and their carbonized analogues. Mater. Chem. Phys. 2018, 213, 352–361. [Google Scholar] [CrossRef]
- Uguz, I.; Ganji, M.; Hama, A.; Tanaka, A.; Inal, S.; Youssef, A.; Owens, R.M.; Quilichini, P.P.; Ghestem, A.; Bernard, C.; et al. Autoclave Sterilization of PEDOT:PSS Electrophysiology Devices. Adv. Healthc. Mater. 2016, 5, 3094. [Google Scholar] [CrossRef]
- Hyun, K.; Wilhelm, M.; Klein, C.O.; Cho, K.S.; Nam, J.G.; Ahn, K.H.; Lee, S.J.; Ewoldt, R.H.; McKinley, G.H. A review of nonlinear oscillatory shear tests: Analysis and application of large amplitude oscillatory shear (LAOS). Prog. Polym. Sci. 2011, 36, 1697–1753. [Google Scholar] [CrossRef]
- Singh, G.; Chanda, A. Mechanical properties of whole-body soft human tissues: A review. Biomed. Mater. 2021, 16, 062004. [Google Scholar] [CrossRef]
- Macmillan, D.S.; Chilton, M.L. A defined approach for predicting skin sensitisation hazard and potency based on the guided integration of in silico, in chemico and in vitro data using exclusion criteria. Regul. Toxicol. Pharmacol. 2019, 101, 35–47. [Google Scholar] [PubMed]
- Foster, R.S.; Fowkes, A.; Cayley, A.; Thresher, A.; Werner, A.D.; Barber, C.G.; Kocks, G.; Tennant, R.E.; Williams, R.V.; Kane, S.; et al. The importance of expert review to clarify ambiguous situations for (Q)SAR predictions under ICH M7. Genes Environ. 2020, 42, 27. [Google Scholar] [CrossRef] [PubMed]
- Sigma Aldrich. Available online: https://www.sigmaaldrich.com/GB/en/sds/aldrich/410896 (accessed on 4 September 2022).
- Cellink. Available online: https://www.cellink.com/wp-content/uploads/2019/07/Irgacure-2959-SDS-12-July-2019.pdf (accessed on 4 September 2022).
- Calleman, C.J. The metabolism and pharmacokinetics of acrylamide: Implications for mechanisms of toxicity and human risk estimation. Drug Metab. Rev. 1996, 28, 527–590. [Google Scholar] [CrossRef] [PubMed]
- Lehning, E.J.; Persaud, A.; Dyer, K.R.; Jortner, B.S.; LoPachin, R.M. Biochemical and morphologic characterization of acrylamide peripheral neuropathy. Toxicol. Appl. Pharmacol. 1998, 151, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Crofton, K.M.; Padilla, S.; Tilson, H.A.; Anthony, D.C.; Raymer, J.H.; MacPhail, R.C. The impact of dose rate on the neurotoxicity of acrylamide: The interaction of administered dose, target tissue concentrations, tissue damage, and functional effects. Toxicol. Appl. Pharmacol. 1996, 139, 163–176. [Google Scholar] [CrossRef]
- Edwards, P.M. Neurotoxicity of acrylamide and its analogues and effects of these analogues and other agents on acrylamide neuropathy. Br. J. Ind. Med. 1975, 32, 31–38. [Google Scholar]
- Unagolla, J.M.; Jayasuriya, A.C. Hydrogel-based 3D bioprinting: A comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Appl. Mater. Today. 2020, 18, 100479. [Google Scholar] [CrossRef]
- Mancha Sánchez, E.; Gómez-Blanco, J.C.; López Nieto, E.; Casado, J.G.; Macías-García, A.; Díaz Díez, M.A.; Carrasco-Amador, J.P.; Torrejón Martín, D.; Sánchez-Margallo, F.M.; Pagador, J.B. Hydrogels for Bioprinting: A Systematic Review of Hydrogels Synthesis, Bioprinting Parameters, and Bioprinted Structures Behavior. Front. Bioeng. Biotechnol. 2020, 8, 776. [Google Scholar] [CrossRef]
- Zhao, C.; Lv, Q.; Wu, W. Application and Prospects of Hydrogel Additive Manufacturing. Gels 2022, 8, 297. [Google Scholar] [CrossRef]
- Yang, R.; Chen, F.; Guo, J.S.; Zhou, D.F.; Luan, S.F. Recent advances in polymeric biomaterials-based gene delivery for cartilage repair. Bioact. Mater. 2020, 5, 990–1003. [Google Scholar] [CrossRef]
- Minardi, S.; Taraballi, F.; Pandolfi, L.; Tasciotti, E. Patterning Biomaterials for the Spatiotemporal Delivery of Bioactive Molecules. Front. Bioeng. Biotechnol. 2016, 4, 45. [Google Scholar] [CrossRef]
- Patil, S.S.; Shahiwala, A. Patented pulsatile drug delivery technologies for chronotherapy. Expert Opin. Ther. Pat. 2014, 24, 845–856. [Google Scholar] [CrossRef]
- Albuquerque, T.; Neves, A.R.; Quintela, T.; Costa, D. Exploring the link between chronobiology and drug delivery: Effects on cancer therapy. J. Mol. Med. 2021, 99, 1349–1371. [Google Scholar] [CrossRef] [PubMed]
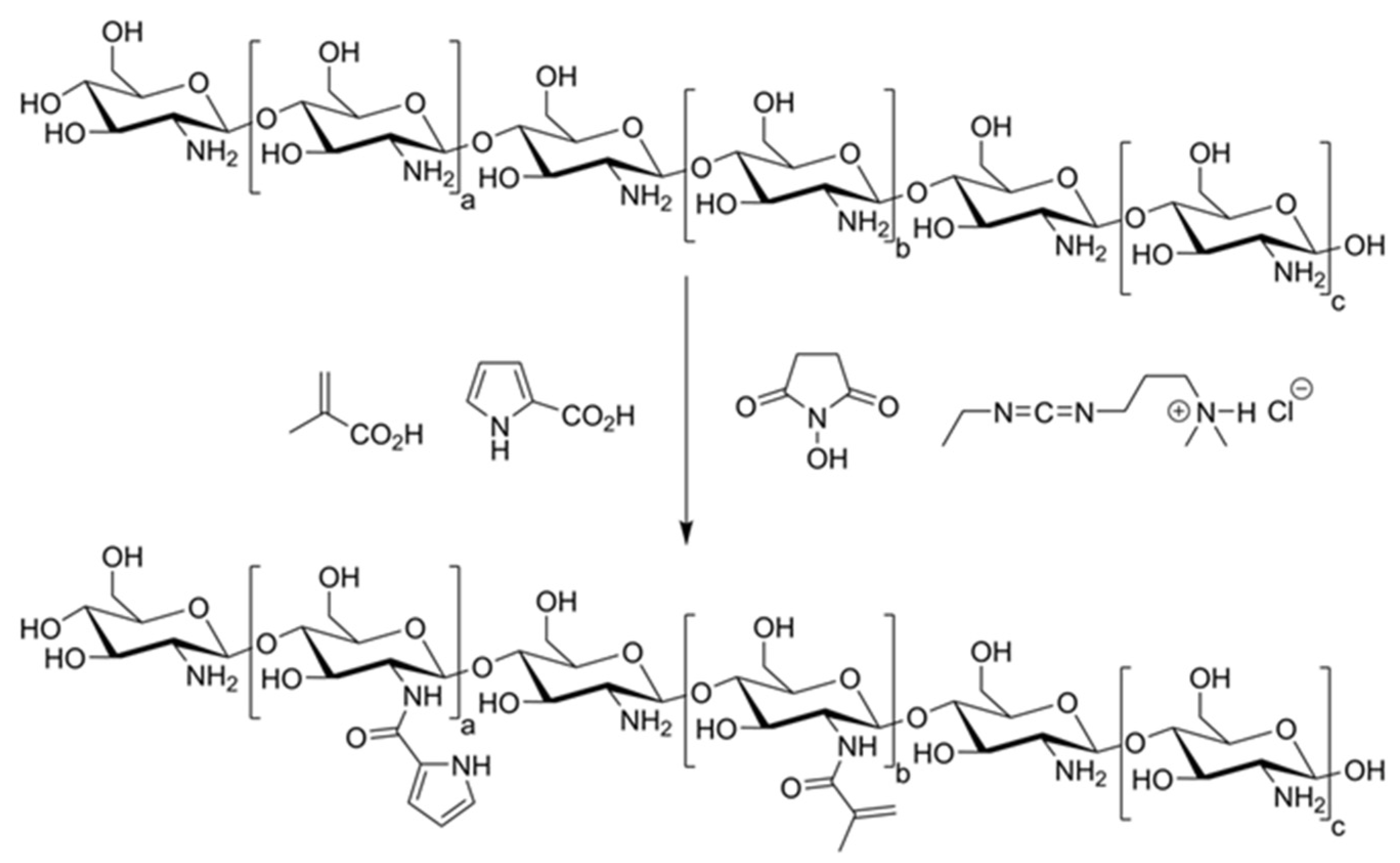
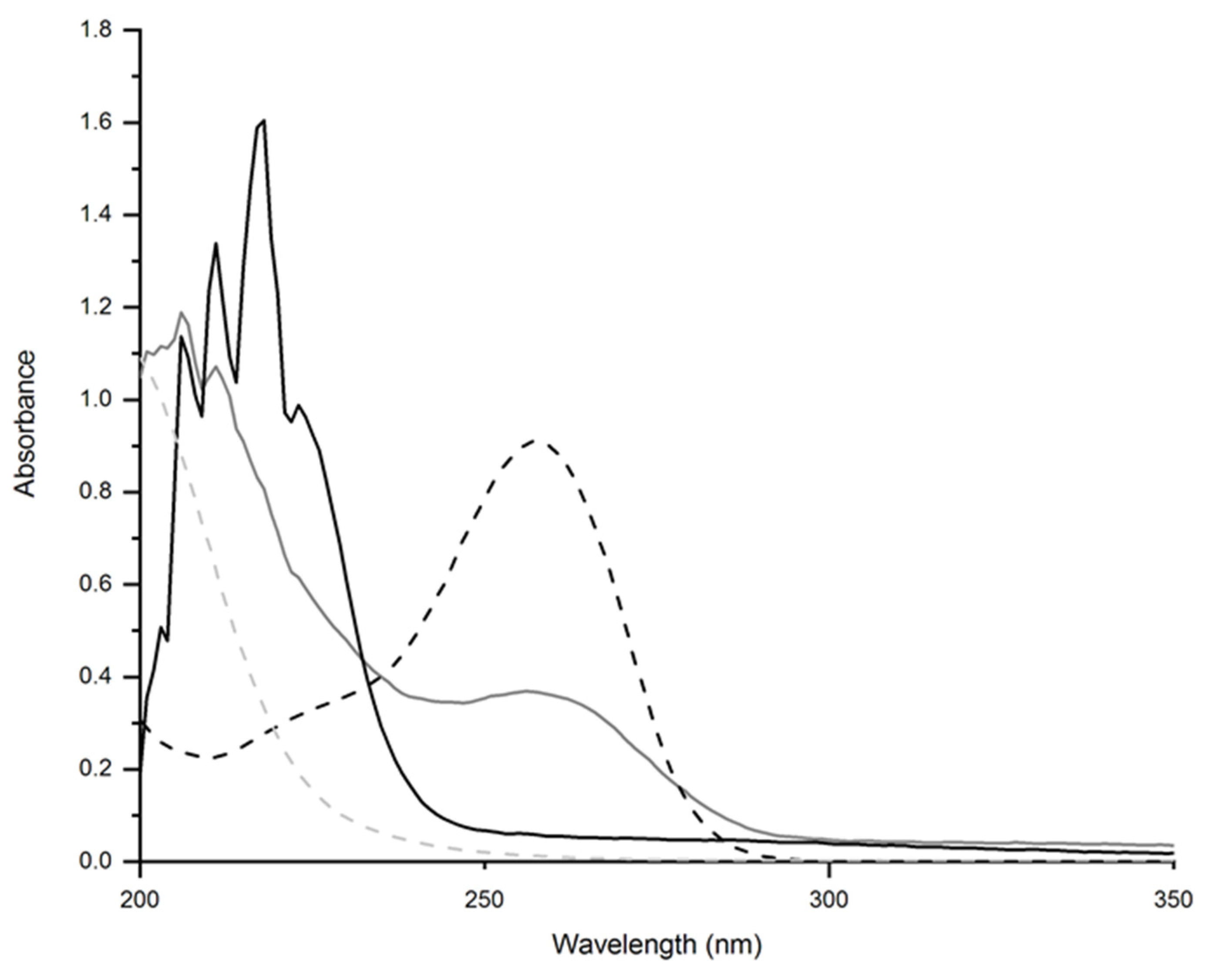
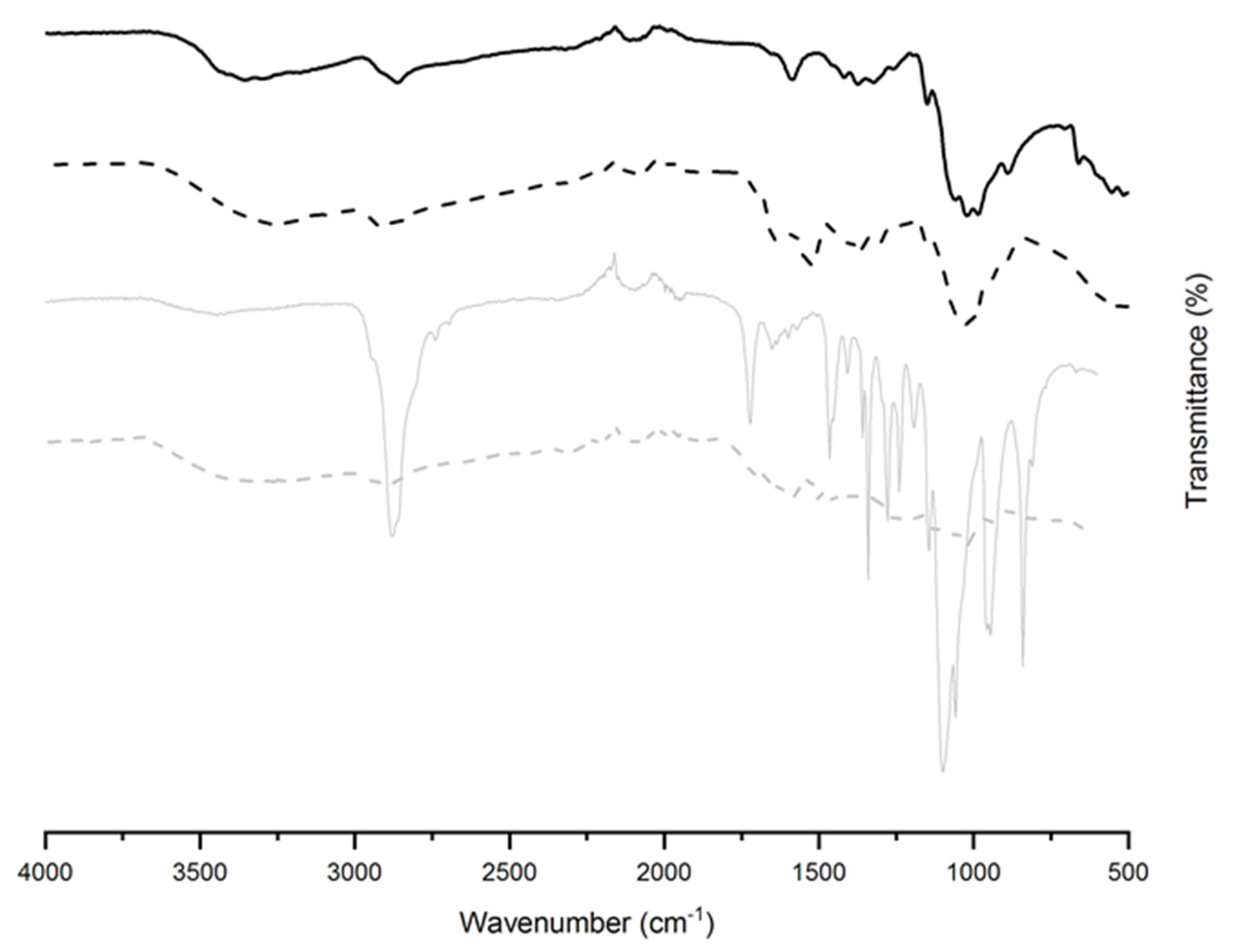
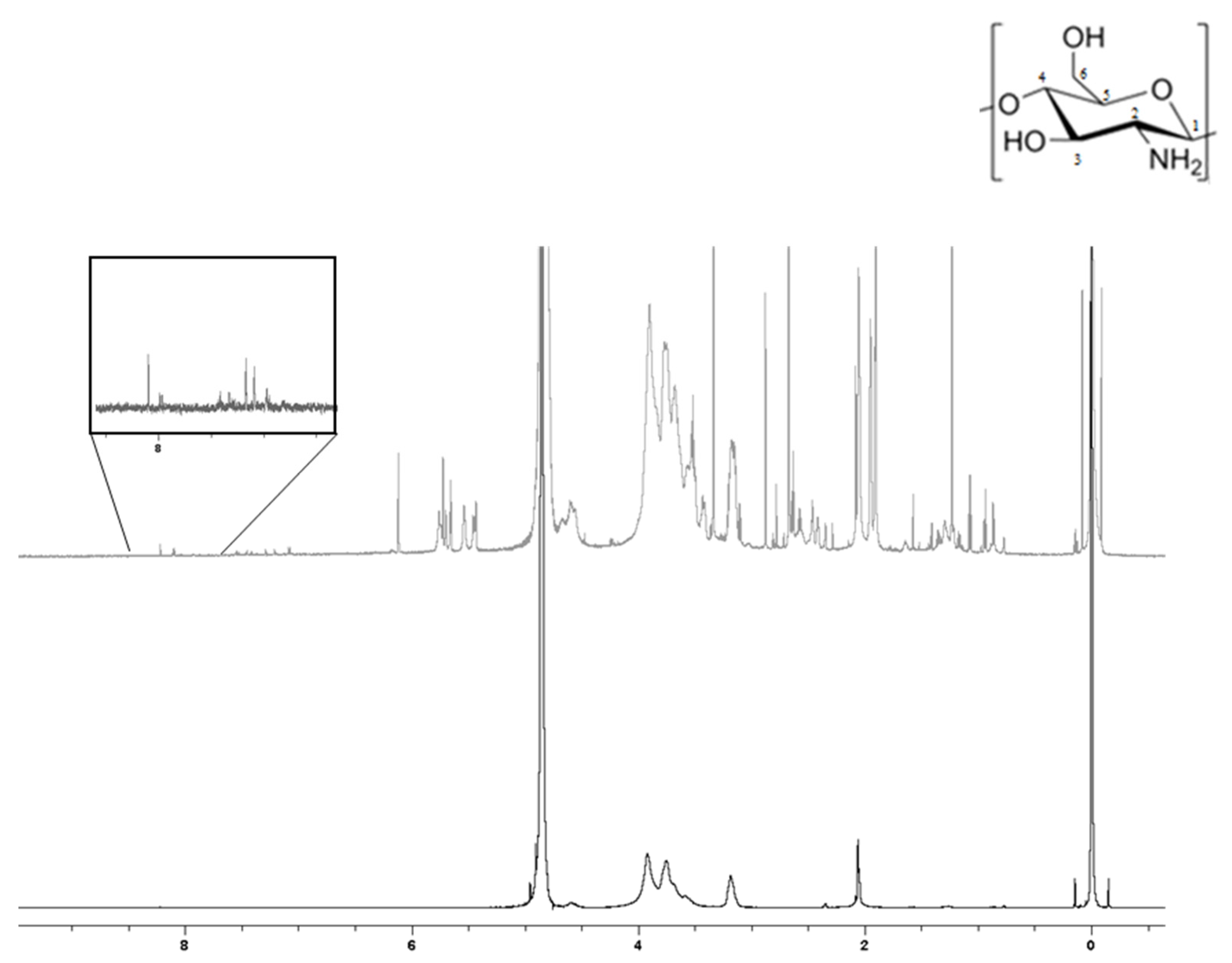

| Component | Non-Conductive Gels | Conductive Gels |
|---|---|---|
| Water | 1 mL | 1 mL |
| PEGDA | 50 mg | 50 mg |
| Modified CS derivative | 4.5 mg | 4.5 mg |
| Lignin | N/A | 100 mg |
| Pyrrole | N/A | 200 μL |
| Variable | Non-Conductive Gels (Wet) | Conductive Gels (Dry) | Conductive Gels (Wet) |
|---|---|---|---|
| G′ | 350 Pa | 650 Pa | 30,000 Pa |
| G″ | 22 Pa | 54 Pa | 19,000 Pa |
| Yield point | 15% | 7% | 0.8% |
| Flow point | 95% | 140% | 4% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Au-Yong, S.; Firlak, M.; Draper, E.R.; Municoy, S.; Ashton, M.D.; Akien, G.R.; Halcovitch, N.R.; Baldock, S.J.; Martin-Hirsch, P.; Desimone, M.F.; et al. Electrochemically Enhanced Delivery of Pemetrexed from Electroactive Hydrogels. Polymers 2022, 14, 4953. https://doi.org/10.3390/polym14224953
Au-Yong S, Firlak M, Draper ER, Municoy S, Ashton MD, Akien GR, Halcovitch NR, Baldock SJ, Martin-Hirsch P, Desimone MF, et al. Electrochemically Enhanced Delivery of Pemetrexed from Electroactive Hydrogels. Polymers. 2022; 14(22):4953. https://doi.org/10.3390/polym14224953
Chicago/Turabian StyleAu-Yong, Sophie, Melike Firlak, Emily R. Draper, Sofia Municoy, Mark D. Ashton, Geoffrey R. Akien, Nathan R. Halcovitch, Sara J. Baldock, Pierre Martin-Hirsch, Martin F. Desimone, and et al. 2022. "Electrochemically Enhanced Delivery of Pemetrexed from Electroactive Hydrogels" Polymers 14, no. 22: 4953. https://doi.org/10.3390/polym14224953
APA StyleAu-Yong, S., Firlak, M., Draper, E. R., Municoy, S., Ashton, M. D., Akien, G. R., Halcovitch, N. R., Baldock, S. J., Martin-Hirsch, P., Desimone, M. F., & Hardy, J. G. (2022). Electrochemically Enhanced Delivery of Pemetrexed from Electroactive Hydrogels. Polymers, 14(22), 4953. https://doi.org/10.3390/polym14224953











