Gel-Forming Soil Conditioners of Combined Action: Laboratory Tests for Functionality and Stability
Abstract
1. Introduction
- thermodynamic assessment and modeling of the water retention of soil substrates under the influence of the gel-forming conditioners;
- analysis of the saturated and unsaturated hydraulic conductivity of soil substrates with the synthetic gel structures;
- assessment of the dispersity, structural organization and aggregation strength under the influence of gel-forming conditioners;
- study of the kinetics of swelling and freezing point dynamics in the synthetic gel structures;
- assessment of the resistance of the gel-forming polymeric materials to biodegradation;
- analysis of the fungicidal properties of synthetic gel structures.
2. Materials and Methods
2.1. Gel-Forming Soil Conditioners and Their Composition
2.2. Mineral Soil Substrates
2.3. Preparation of Gel-Forming Materials
2.4. Methodological Guidelines for Laboratory Testing of Gel-Forming Polymer Cnditioners in Soils
2.4.1. Water-Retention Characteristic
2.4.2. WRC-Calculated Criteria of Soil Structure, Dispersity, Water Retention Energy and Capacity
2.4.3. Soil Hydraulic Properties
2.4.4. Evaporation Rate
2.4.5. Intensity and Degree of Swelling
2.4.6. Strength of Samples Treated with Gel-Forming Soil Conditioners
2.4.7. Biological Activity and Resistance to Biodegradation
2.4.8. Biological Activity against Pathogenic Organisms
2.4.9. Guidelines for the Main Quality Indicators
2.5. Other Laboratory Methods; Experimental Data Processing
3. Results
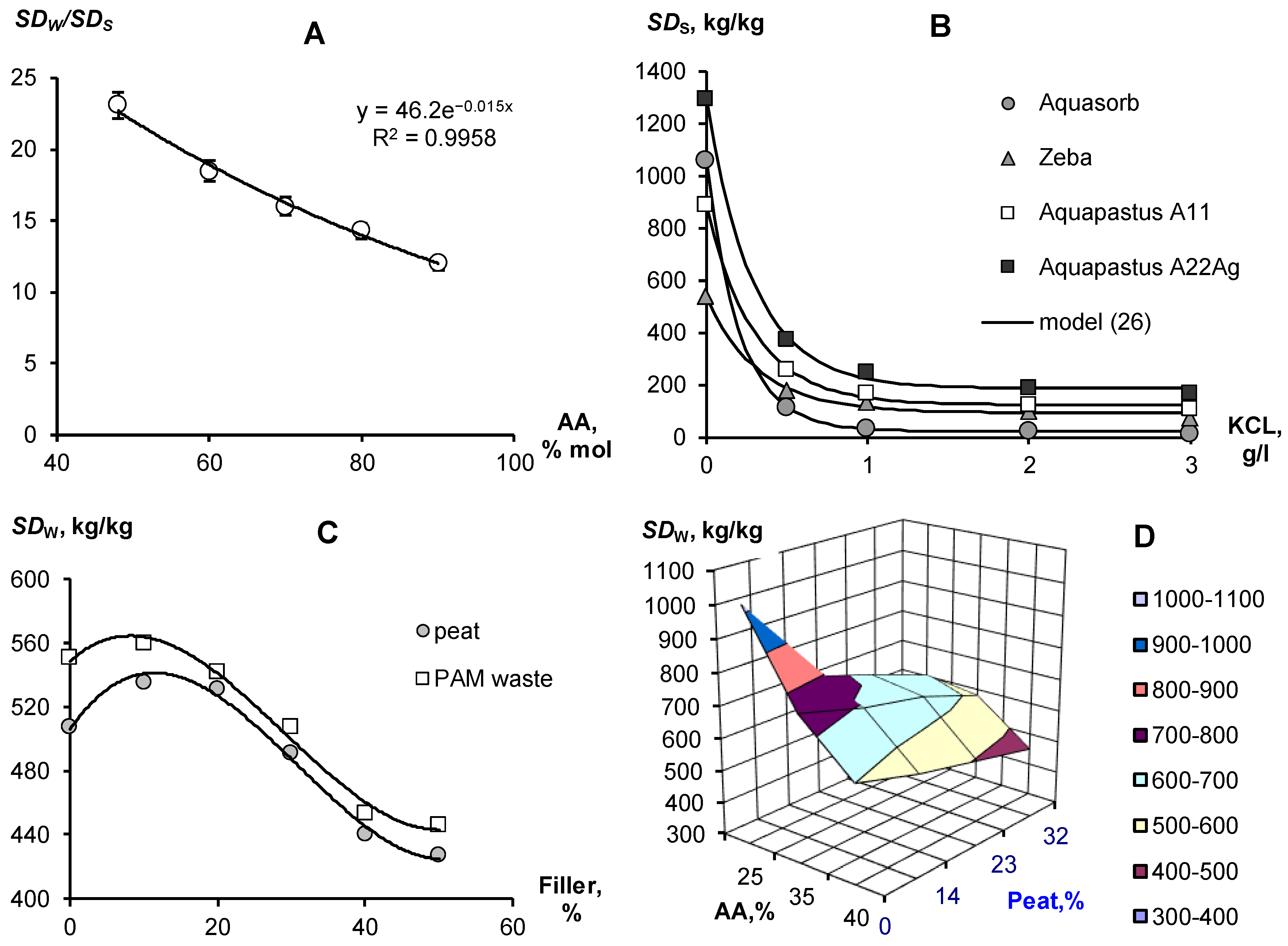
3.1. Composition and Technological Properties of Polymer Hydrogels
3.1.1. Polymeric Matrix Composition and Swelling
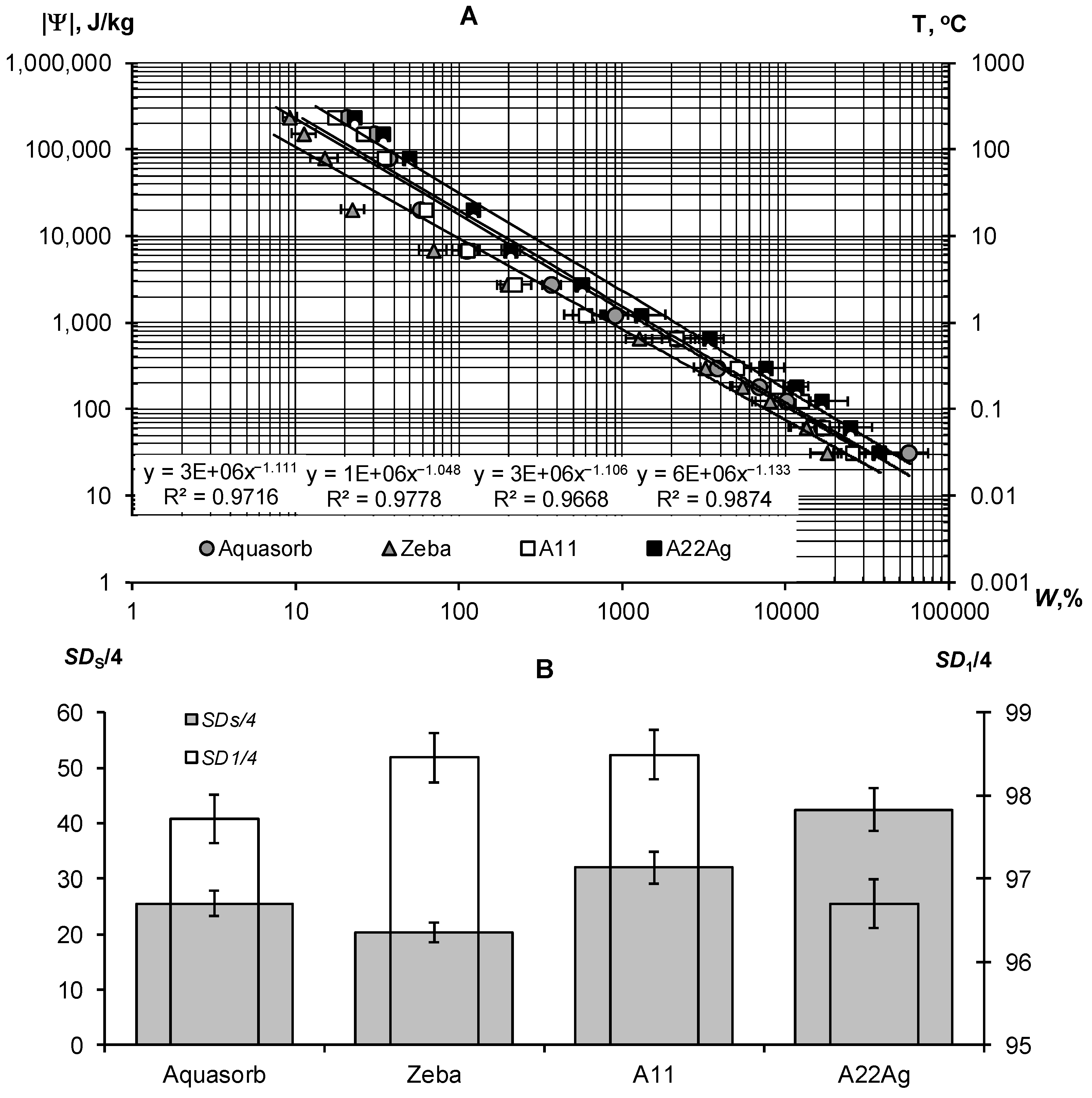
3.1.2. Water Retention, Dispersity and Stability of Gels with Regards to Physical and Chemical Factors
3.1.3. Biodegradation of Hydrogels and Its Inhibition
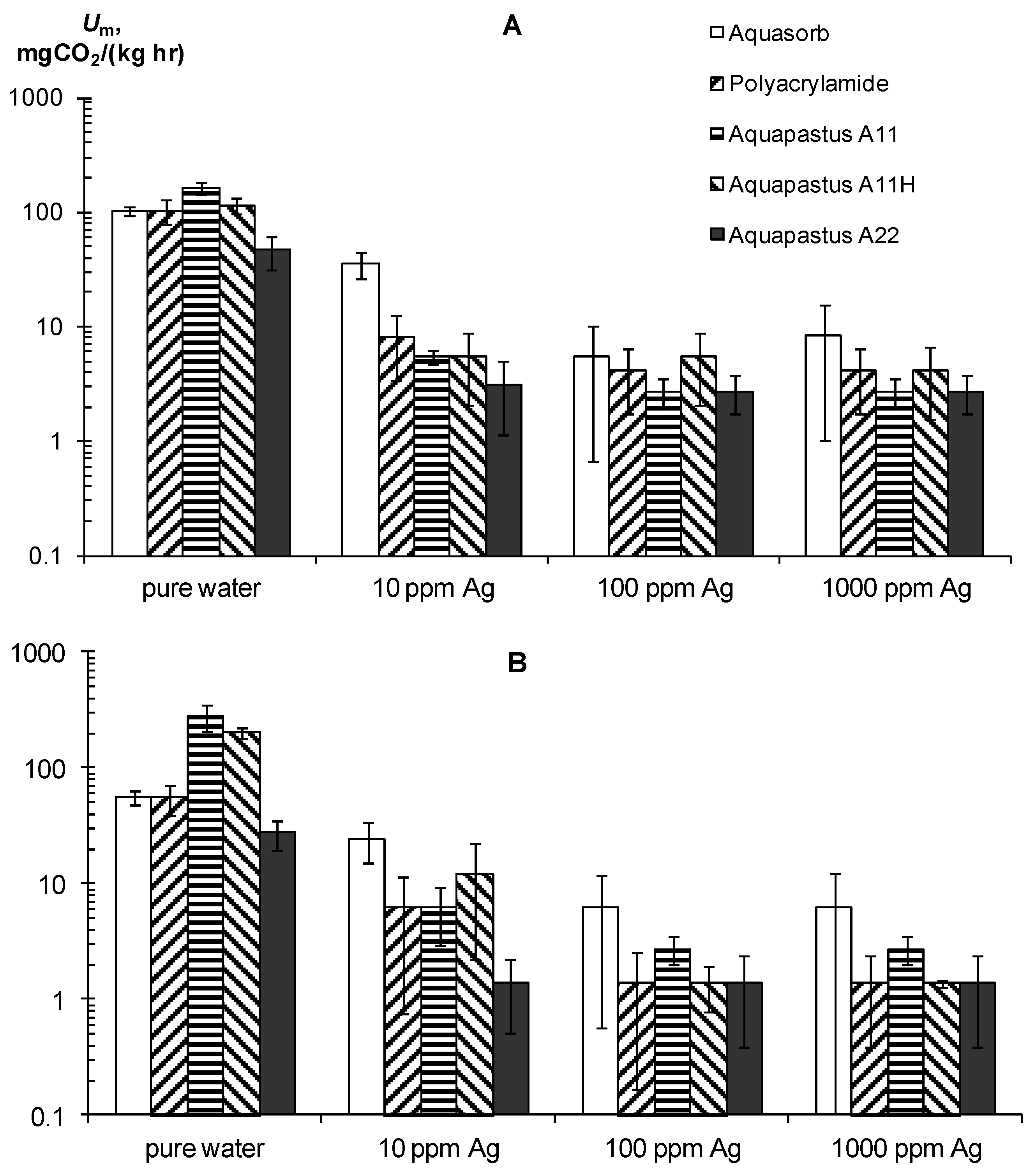
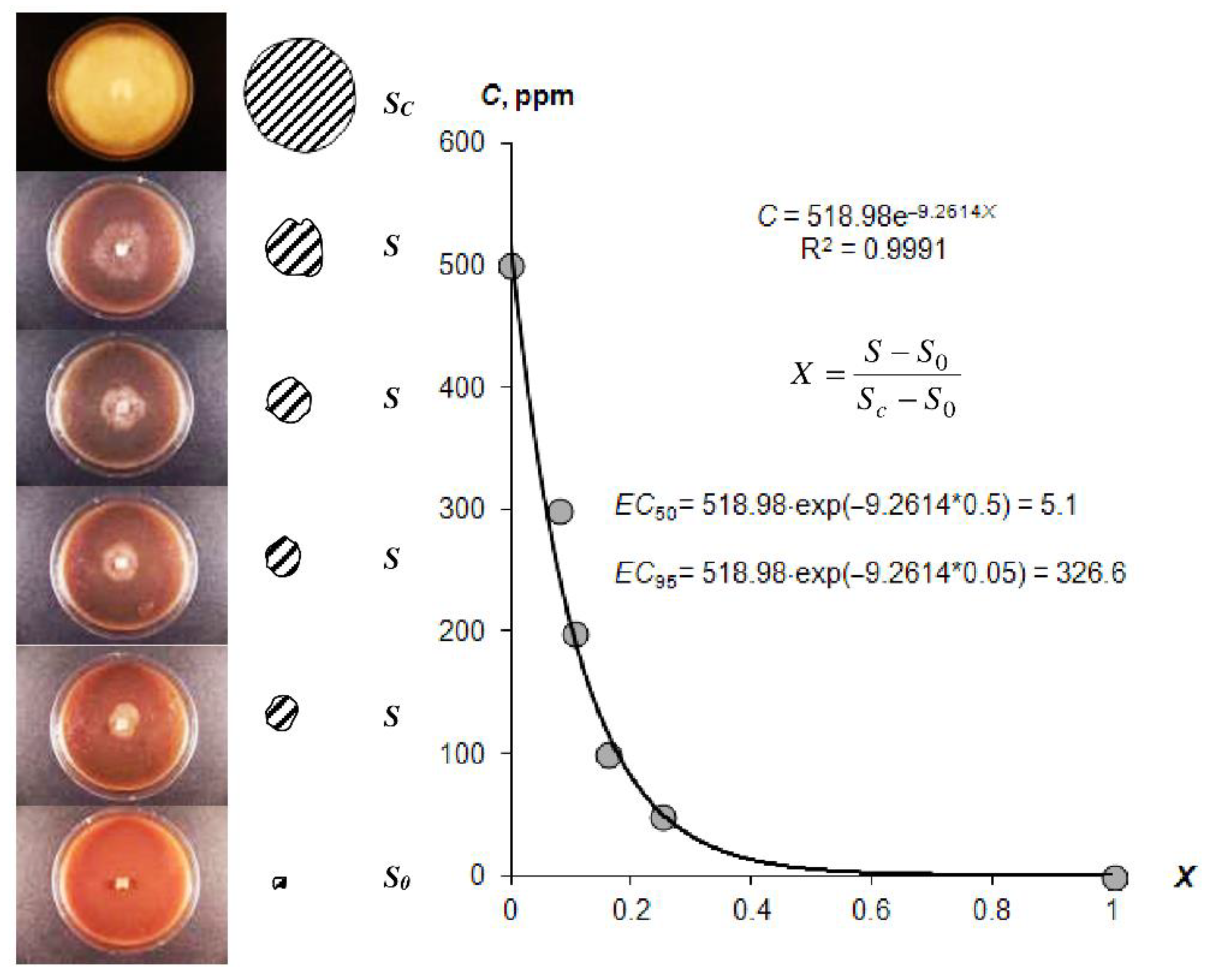
3.1.4. Laboratory Analysis of SGS Protective Antimicrobial Properties
3.2. Laboratory Testing of Soil–Gel Compositions
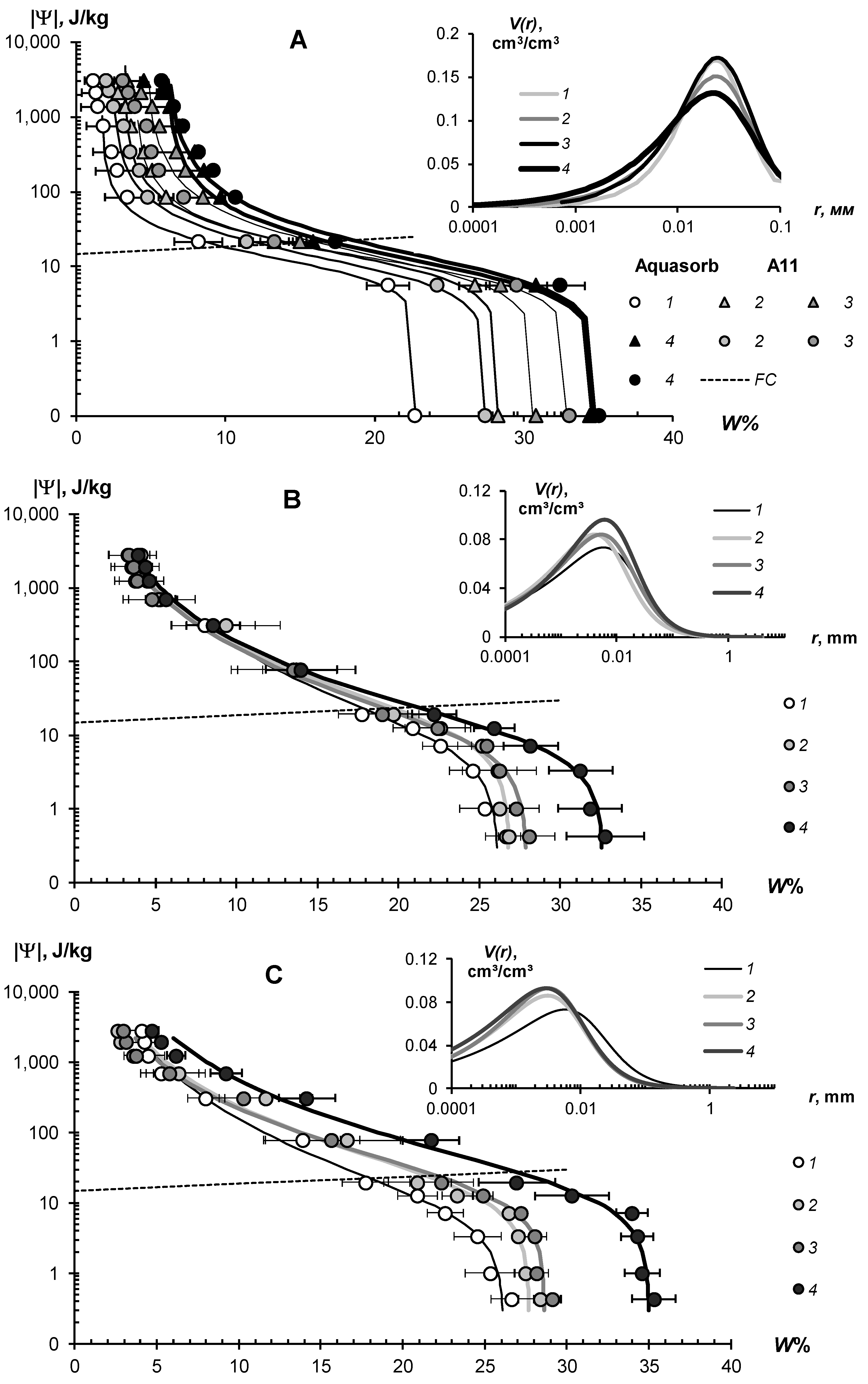
3.2.1. Thermodynamic Assessment of Water Retention and Dispersity
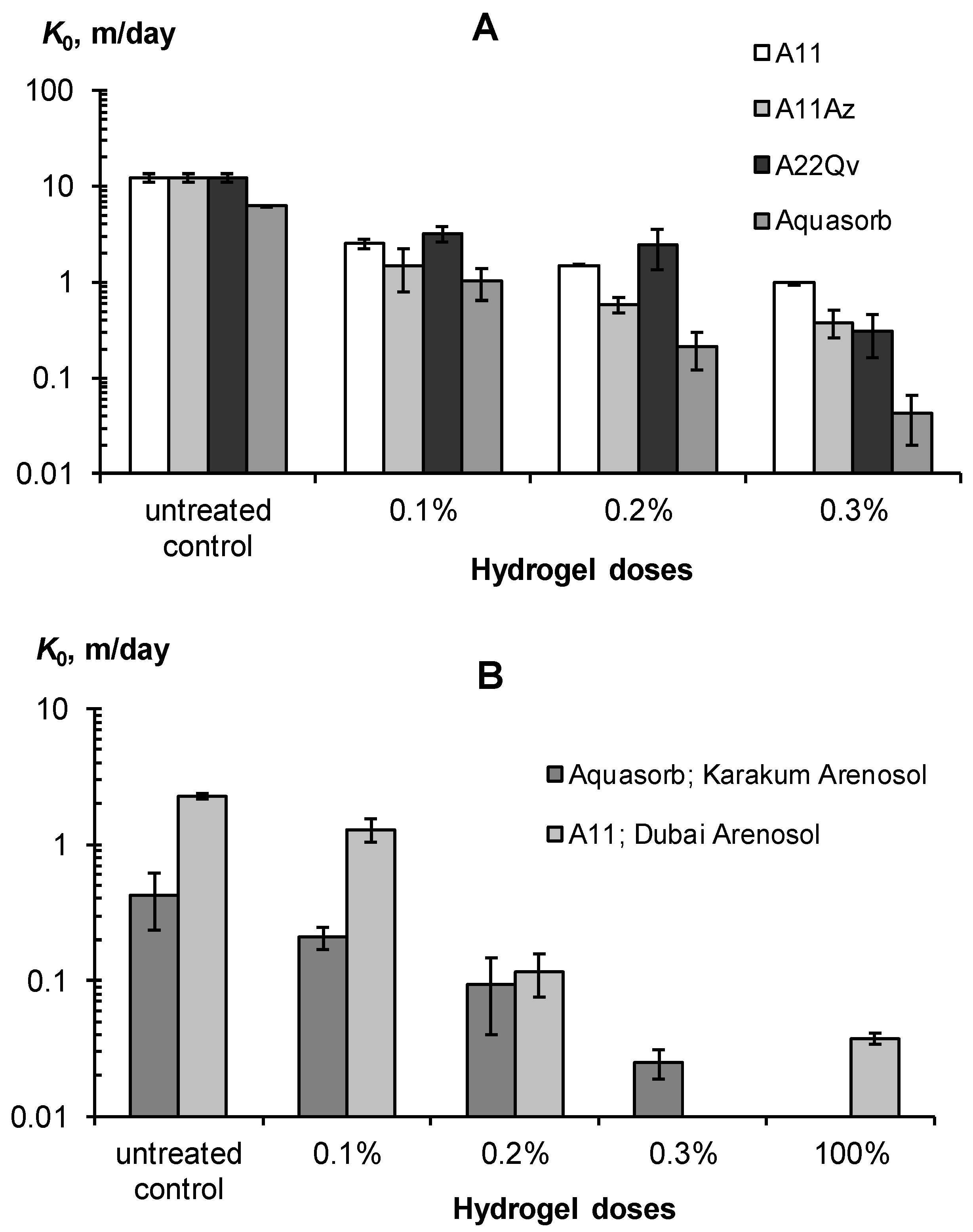
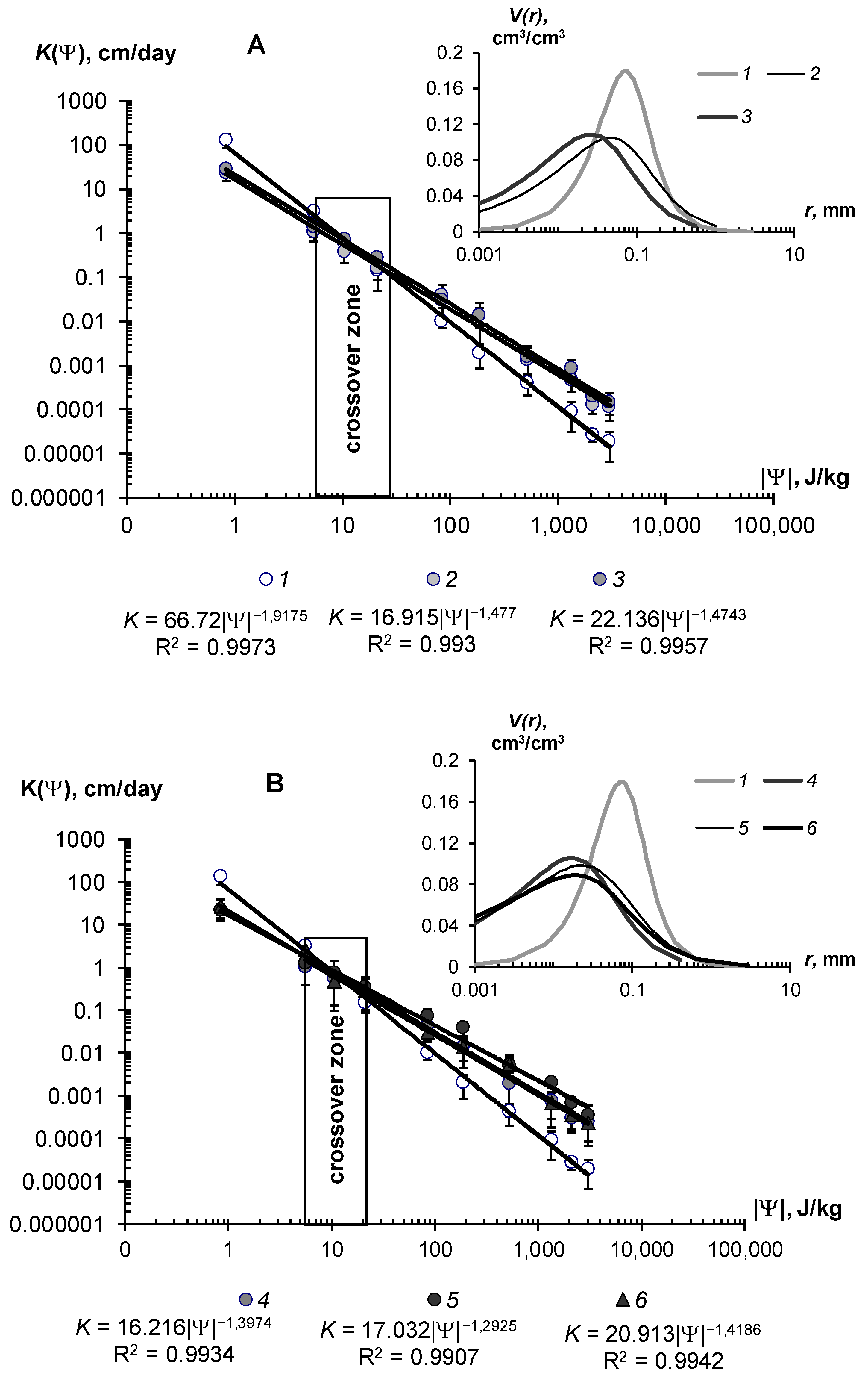
3.2.2. Saturated and Unsaturated Hydraulic Conductivity
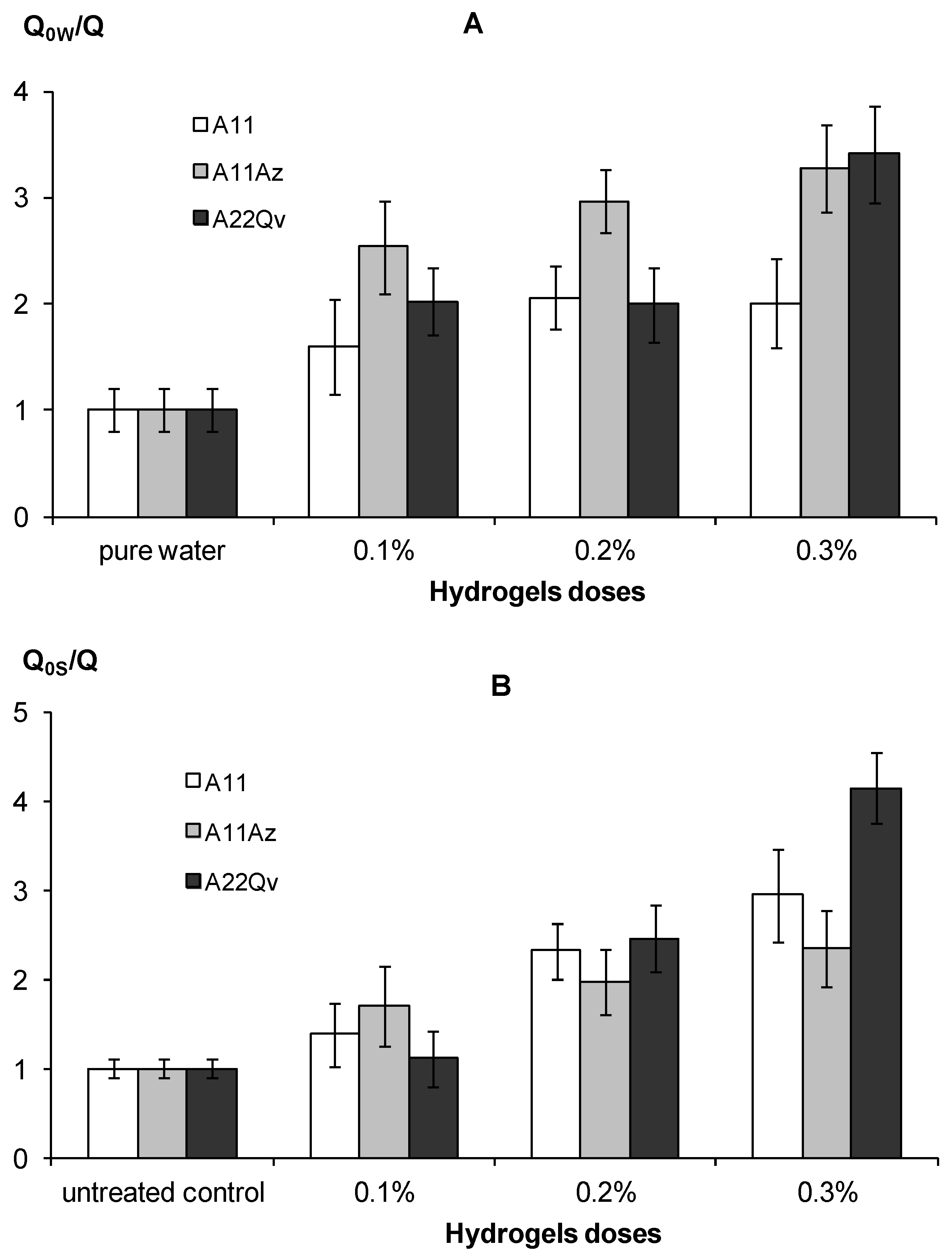
3.2.3. Evaporation of Water
3.2.4. Strength of Soil Aggregation
3.2.5. Basal Respiration and Resistance to Biodegradation
4. Discussion
4.1. Technological Properties of Gel-Forming Materials
4.1.1. Swelling Degree and Kinetics
4.1.2. Water Retention and Associated Quality Indicators
4.1.3. Biodegradation of Acrylic Composite Materials
4.1.4. Antimicrobial Properties of Composite Gel-Forming Materials
4.2. Technological Properties of Soil-Gel Compositions
4.2.1. Water Retention and Other Hydrophysical Properties
4.2.2. Aggregation Strength and Biodegradation of Gel Structures in Soils
5. Conclusions
6. Patents
- patent RU №2726561 (https://findpatent.ru/patent/272/2726561.html; accessed on 2 July 2022)
- patent RU 2639789(http://www.findpatent.ru/patent/263/2639789.html; accessed on 2 July 2022).
Author Contributions
Funding
Conflicts of Interest
References
- Campos, E.V.R.; de Oliveira, J.L.; Fraceto, L.F. Applications of Controlled Release Systems for Fungicides, Herbicides, Acaricides, Nutrients, and Plant Growth Hormones: A Review. Adv. Sci. Eng. Med. 2014, 6, 373–387. [Google Scholar] [CrossRef]
- Campos, E.V.R.; de Oliveira, J.L.; Fraceto, L.F.; Singh, B. Polysaccharides as Safer Release Systems for Agrochemicals. Agron. Sustain. Dev. 2015, 35, 47–66. [Google Scholar] [CrossRef]
- Smagin, A.; Sadovnikova, N.; Smagina, M. Synthetic Gel Structures in Soils for Sustainable Potato Farming. Sci. Rep. 2019, 9, 18588. [Google Scholar] [CrossRef] [PubMed]
- Behera, S.; Mahanwar, P.A. Superabsorbent Polymers in Agriculture and Other Applications: A review. Polym. Plast. Technol. Mat. 2020, 59, 341–356. [Google Scholar] [CrossRef]
- Rizwan, M.; Gilani, S.R.; Durani, A.I.; Naseem, S. Materials diversity of hydrogel: Synthesis, polymerization process and soil conditioning properties in agricultural field. J. Adv. Res. 2021, 33, 15–40. [Google Scholar] [CrossRef] [PubMed]
- Certini, G.; Scalenghe, R. Soils: Basic Concepts and Future Challenges; Cambridge University Press: Cambridge, UK, 2006; 310p. [Google Scholar] [CrossRef]
- Narjary, B.; Aggarwal, P.; Singh, A.; Chakraborty, D.; Singh, R. Water Availability in Different Soils in Relation to Hydrogel Application. Geoderma 2012, 187, 94–101. [Google Scholar] [CrossRef]
- Abdallah, A.M. Influence of Hydrogel Type and Concentration, and Water Application Rate on some Hydraulic Properties of a Sandy Soil. Alexandria Sci. Exch. J. 2019, 40, 347–360. [Google Scholar] [CrossRef]
- Chen, J.; Zhao, Y. Relationship between Water Absorbency and Reaction Conditions in Aqueous Solution Polymerization of Polyacrylate Superabsorbents. J. Appl. Polym. Sci. 2000, 75, 808–814. [Google Scholar] [CrossRef]
- Tayel, M.Y.; El-Hady, O.A. Super Gel as a Soil Conditioner. Egypt. J. Soil. Sci. 1981, 19, 103–106. [Google Scholar]
- Johnson, M.S. The effects of Gel-forming Polyacrylamides on Moisture Storage in Sandy Soils. J. Sci. Food Agric. 1984, 35, 1196–1200. [Google Scholar] [CrossRef]
- Azzam, R. Tailoring Polymeric Gels for Soil Reclamation and Hydroponics. Commun. Soil Sci. Plant Anal. 1985, 16, 1123–1138. [Google Scholar] [CrossRef]
- Smagin, A.V.; Sadovnikova, N.B. Impact of Strongly Swelling Hydrohels on Water-Holding Capacity of Light-Textured Soils. Eurasian Soil Sci. 1995, 27, 50–55. [Google Scholar]
- Al-Darby, A.M. The Hydraulic Properties of a Sandy Soil Treated with Gel-forming Soil Conditioner. Soil Technol. 1996, 9, 15–28. [Google Scholar] [CrossRef]
- Al-Darby, A.M.; Al-Asfoor, S.I.; El-Shafei, Y.Z. Effect of Soil Gel-Conditioner on the Hydrophysical Properties of Sandy Soil. J. Saudi Soc. Agric. Sci. 2002, 1, 14–40. [Google Scholar]
- El-Rehim, H.A.A.; Hegazy, E.S.A.; El-Mohdy, H.L.A. Radiation Synthesis of Hydrogels to Enhance Sandy Soils Water Retention and Increase Plant Performance. J. Appl. Polym. Sci. 2004, 93, 1360–1371. [Google Scholar] [CrossRef]
- Abedi-Koupai, J.; Sohrab, F.; Swarbrick, G. Evaluation of Hydrogel Application on Soil Water Retention Characteristics. J. Plant Nutr. 2008, 31, 317–331. [Google Scholar] [CrossRef]
- Shahid, S.A.; Qidwai, A.A.; Anwar, F.; Ullah, I.; Rashid, U. Improvement in the Water Retention Characteristics of Sandy Loam Soil Using a Newly Synthesized Poly(acrylamide-co-acrylic Acid)/AlZnFe2O4 Superabsorbent Hydrogel Nanocomposite Material. Molecules 2012, 17, 9397–9412. [Google Scholar] [CrossRef]
- Yang, L.; Yang, Y.; Chen, Z. Influence of Super Absorbent Polymer on Soil Water Retention, Seed Germination and Plant Survivals for Rocky Slopes Eco-engineering. Ecol. Eng. 2014, 62, 27–32. [Google Scholar] [CrossRef]
- Banedjschafie, S.; Durner, W. Water Retention Properties of a Sandy Soil with Superabsorbent Polymers as Affected by Aging and Water Quality. J. Plant Nutr. Soil Sci. 2015, 178, 798–806. [Google Scholar] [CrossRef]
- Novoskoltseva, O.A.; Panova, I.G.; Loiko, N.G.; Nikolaev, Y.A.; Litmanovich, E.A.; Yaroslavov, A.A. Polyelectrolytes and Polycomplexes for Stabilizing Sandy Grounds. Polym. Sci. Ser. B 2021, 63, 488–495. [Google Scholar] [CrossRef]
- Wu, L.; Liu, M.; Rui-Liang, R.L. Preparation and properties of a double-coated slow-release NPK compound fertilizer with superabsorbent and water-retention. Biores. Technol. 2008, 99, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Singh, S.K.; Bajpai, J.; Bajpai, A.K. Controlled pesticide release from biodegradable polymers. Cent. Eur. J. Chem. 2014, 12, 453–469. [Google Scholar] [CrossRef]
- Vashist, A.; Ahmad, S. Hydrogels: Smart Materials for Drug Delivery. Orient. J. Chem. 2013, 29, 861–870. [Google Scholar] [CrossRef]
- Ullah, F.; Othman, M.B.H.; Javed, F. Classification, Processing and Application of Hydrogels: A Review. Mater. Sci. Eng. 2015, 57, 414–433. [Google Scholar] [CrossRef]
- Johnson, M.S. Effect of Soluble Salts on Water Absorption by Gel-forming Soil Conditioners. J. Sci. Food Agric. 1984, 35, 1063–1066. [Google Scholar] [CrossRef]
- Seybold, C.A. Polyacrylamide review: Soil Conditioning and Environmental Fate. Comm. Soil Sci. Plant Anal. 1994, 25, 2171–2185. [Google Scholar] [CrossRef]
- Sojka, R.E.; Bjorneberg, D.L.; Entry, J.A. Polyacrylamide in Agriculture and Environmental Land Management. Adv. Agron. 2007, 92, 75–162. [Google Scholar]
- Lentz, R.D.; Andrawes, F.F.; Barvenik, F.W.; Koehn, A.C. Acrylamide Monomer Leaching from Polyacrylamide-treated Irrigation Furrows. J. Environ. Qual. 2008, 37, 2293–2298. [Google Scholar] [CrossRef]
- Smagin, A.V.; Sadovnikova, N.B.; Vasenev, V.I.; Smagina, M.V. Biodegradation of Some Organic Materials in Soils and Soil Constructions: Experiments, Modeling and Prevention. Materials 2018, 11, 1889. [Google Scholar] [CrossRef]
- Smagin, A.V. Column-Centrifugation Method for Determining Water Retention Curves of Soils and Disperse Sediments. Eurasian Soil Sci. 2012, 45, 416–422. [Google Scholar] [CrossRef]
- Smagin, A.V. Thermogravimetric Determination of Specific Surface Area for Soil Colloids. Colloid J. 2016, 78, 391–396. [Google Scholar] [CrossRef]
- Van Genuchten, M.T. A closed form equation for predicting the hydraulic conductivity of unsaturated soils. Soil Sci. Soc. Am. J. 1980, 44, 892–898. [Google Scholar] [CrossRef]
- Smagin, A.V. Thermodynamic Concept of Water Retention and Physical Quality of the Soil. Agronomy 2021, 11, 1686. [Google Scholar] [CrossRef]
- Smagin, A.V. About Thermodynamic Theory of Water Retention Capacity and Dispersity of Soils. Eurasian Soil Sci. 2018, 51, 782–796. [Google Scholar] [CrossRef]
- Dexter, A.R. Soil physical quality: Part I. Theory, effects of soil texture, density, and organic matter, and effects on root growth. Geoderma 2004, 120, 201–214. [Google Scholar] [CrossRef]
- Smagin, A.V.; Kol’tsov, I.N.; Pepelov, I.L.; Kirichenko, A.V.; Sadovnikova, N.B. The physical state of finely dispersed soil-like systems with drilling sludge as an example. Eurasian Soil Sci. 2011, 44, 163–172. [Google Scholar] [CrossRef]
- Richards, L.A.; Weaver, L.R. Fifteem-Atmosphere-Percentage as Related to the Permanent Wilting Percentage. Soil Sci. 1943, 56, 331–339. [Google Scholar] [CrossRef]
- Voronin, A.D. Energy Concept of the Physical State of Soils. Eurasian Soil Sci. 1990, 23, 7–19. [Google Scholar]
- Campbell, G.S. Soil Physics with BASIC; Elsevier: Amsterdam, The Netherlands, 1985; p. 268. [Google Scholar]
- Simunek, J.; van Genuchten, M.T.; Sejna, M. Development and Application of the HYDRUS and STANMOD Software Packages and Related Codes. Vadose Zone J. 2008, 7, 587–600. [Google Scholar] [CrossRef]
- ISO 16072; Soil Quality—Laboratory Methods for Determination of Microbial Soil Respiration. International Organization for Standardization: Geneva, Switzerland, 2002.
- Woods End Research Laboratory, Inc. Woods End Research Guide to Solvita Testing and Managing Your Soil; Woods End Research Laboratory, Inc.: Mt. Vernon, ME, USA, 1997. [Google Scholar]
- Loizeau, J.L.; Arbouille, D.; Santiago, S.; Vernet, J.P. Evaluation of Wide Range Laser Diffraction Grain Size Analyzer for Use With Sediments. Sedimentology 1994, 41, 353–361. [Google Scholar] [CrossRef]
- Richards, L.A. (Ed.) Diagnosis and Improvement of Saline and Alkali Soils; U.S. Department Agriculture Handbook 60; U.S. Gov. Printing Office: Washington, DC, USA, 1954. [Google Scholar]
- Hadas, A.; Kautsky, L.; Goek, M.; Kara, E.E. Rates of Decomposition of Plant Residues and Available Nitrogen in Soil, Related to Residue Composition through Simulation of Carbon and Nitrogen Turnover. Soil Biol. Biochem. 2004, 36, 255–266. [Google Scholar] [CrossRef]
- Smagin, A.V.; Sadovnikova, N.B.; Belyaeva, E.A.; Kirichenko, A.V.; Krivtsova, V.N. Capillary Effects in Polydisperse Systems and Their Use in Soil Engineering. Eurasian Soil Sci. 2021, 54, 1433–1446. [Google Scholar] [CrossRef]
- Vorobieva, E.V. Swelling of polyacrylamide-based hydrogel in aqueous solutions of low-molecular salts. Dokl. Nat. Acad. Sci. Belarus 2020, 64, 293–299. (In Russian) [Google Scholar] [CrossRef]
- Shirinov, S.D.; Jalilov, A.T. Investigation of the swelling kinetics of synthesized hydrogels based on hydrolyzed polyacrylonitrile. Univers. Chem. Biol. Electron. Sci. J. 2018, 1–3. Available online: https://7universum.com/ru/nature/archive/item/5601 (accessed on 21 August 2022).
- Lagutina, M.A.; Dubrovskii, S.A. The swelling pressure of weakly ionic acrylamide gels. Polym. Sci. Ser. A 1996, 38, 1059–1064. [Google Scholar]
- Dubrovskii, S.A.; Lagutina, M.A.; Kazanskii, K.S. Method of measuring the swelling pressure of superabsorbent gels. Polym. Gels Net. 1994, 2, 49–58. [Google Scholar] [CrossRef]
- Wacka, H.; Ulbricht, M. Effect of synthesis composition on the swelling pressure of polymeric hydrogels. Polymer 2009, 50, 2075–2080. [Google Scholar] [CrossRef]
- Arthur, E.; Tuller, M.; Moldrup, P.; de Jonge, L.W. Evaluation of theoretical and empirical water vapor sorption isotherm models for soils. Water Resour. Res. 2016, 52, 190–205. [Google Scholar] [CrossRef]
- Andrade, R.D.; Lemus, R.; Perez, C.E. Models of sorption isotherms for food: Uses and limitations. Vitae 2011, 18, 325–334. [Google Scholar]
- Xu, C.Y.; Zhou, T.T.; Wang, C.L.; Liu, H.Y.; Zhang, C.T.; Hu, F.N.; Zhao, S.W.; Geng, Z.C. Aggregation of polydisperse soil colloidal particles: Dependence of Hamaker constant on particle size. Geoderma 2019, 21, 113999. [Google Scholar] [CrossRef]
- Lande, S.S.; Bosch, S.J.; Howard, P.H. Degradation and Leaching of Acrylamide in soil. J. Environ. Qual. 1979, 8, 133–137. [Google Scholar] [CrossRef]
- Abdelmagid, H.M.; Tabatabai, M.A. Decomposition of Acrylamide in Soils. J. Environ. Qual. 1982, 11, 701–704. [Google Scholar] [CrossRef]
- Shanker, R.; Ramakrishna, C.; Seth, P.K. Microbial Degradation of Acrylamide Monomer. Arch. Microb. 1990, 154, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Kay-Shoemake, J.L.; Watwood, M.E.; Sojka, R.E.; Lentz, R.D. Polyacrylamide as a Substrate for Microbial Amidase in Culture and in Soil. Soil Biol. Biochem. 1998, 30, 1647–1654. [Google Scholar] [CrossRef]
- Sojka, R.E.; Entry, J.A. Influence of Polyacrylamide Application to Soil on Movement of Microorganisms in Runoff Water. Environ. Pollut. 2000, 108, 405–412. [Google Scholar] [CrossRef]
- Kim, S.W.; Jung, J.H.; Lamsal, K.; Kim, Y.S.; Min, J.S.; Lee, Y.S. Antifungal effect of silver nanoparticles (AgNPs) against various plant pathogenic fungi. Mycobiology 2012, 40, 53–58. [Google Scholar] [CrossRef]
- Pulit, J.; Banach, M.; Szczygłowska, R.; Bryk, M. Nanosilver against Fungi. Silver Nanoparticles as an Effective. Acta Biochim. Pol. 2013, 60, 795-3. [Google Scholar] [CrossRef]
- Ouda, S.M. Antifungal Activity of Silver and Copper Nanoparticles on Two Plant Pathogens, Alternaria alternata and Botrytis cinerea. Res. J. Microbiol. 2014, 9, 34–42. [Google Scholar] [CrossRef]
- De Lucca, J.; Boue, S.; Sien, T.; Cleveland, T.E.; Walsh, T.J. Silver Enhances the in Vitro Antifungal Activity of the Saponin, CAY-1. Mycoses 2011, 54, e1–e8. [Google Scholar] [CrossRef]
- Ali, M.; Kim, B.; Belfield, K.D.; Norman, D.; Brennan, M.; Ali, G.S. Inhibition of Phytophthora parasitica and P. capsici by Silver Nanoparticles Synthesized Using Aqueous Extract of Artemisia absinthium. Phytopathology 2015, 105, 1183–1190. [Google Scholar] [CrossRef]
- Rai, M.K.; Deshmukh, S.D.; Ingle, A.P.; Gade, A.K. Silver Nanoparticles: The Powerful Nanoweapon against Multidrug-Resistant Bacteria. J. Appl. Microbiol. 2012, 112, 841–852. [Google Scholar] [CrossRef]
- Ratte, H.T. Bioaccumulation and Toxicity of Silver Compounds: A Review. Environ. Toxicol. Chem. 1999, 18, 89–108. [Google Scholar] [CrossRef]
- Jiang, H.S.; Li, M.; Chang, F.Y.; Li, W.; Yin, L.Y. Physiological Analysis of Silver Nanoparticles and AgNO3 Toxicity to Spirodela polyrhiza. Environ. Toxicol. Chem. 2012, 31, 1880–1886. [Google Scholar] [CrossRef]
- Langdon, K.A.; McLaughlin, M.J.; Kirby, J.K.; Merrington, G. Influence of Soil Properties and Soil Leaching on the Toxicity of Ionic Silver to Plants. Environ. Toxicol. Chem. 2015, 34, 2503–2512. [Google Scholar] [CrossRef]
- Schlich, K.; Klawonn, T.; Terytze, K.; Hund-Rinke, K. Effects of Silver Nanoparticles and Silver Nitrate in the Earthworm Reproduction Test. Environ. Toxicol. Chem. 2013, 32, 181–187. [Google Scholar] [CrossRef]
- Moiseev, K.G.; Romanov, I.A. Soil aggregate strength as affected by long-term plowing. Eurasian Soil Sci. 2004, 37, 604–607. [Google Scholar]




| Granulometric Fractions: | Soil Substrates: | ||
|---|---|---|---|
| № 1 – Monomineral Fine-Grained Quartz Sand | № 2 – Polymineral Loamy-Sandy Arenosol from the Karakum Desert | № 3– Carbonate Loamy-Sandy Arenosol from the Emirate of Dubai | |
| Clay (<2 µm) | 0 | 2.1 | 4.2 |
| Silt (2–50 µm) | 2.3 | 23.6 | 23.3 |
| Very fine sand (50–100 µm) | 5.3 | 23.2 | 22.4 |
| Fine sand (100–250 µm) | 63.8 | 47.5 | 39.2 |
| Medium sand (250–500 µm) | 28.6 | 3.6 | 9.6 |
| Coarse sand (500–1000 µm) | 0 | 0 | 1.3 |
| Very coarse sand (1000–2000 µm) | 0 | 0 | 0 |
| Indicators | Gradations of Quality; Comments | ||
|---|---|---|---|
| «Lack» | «Norm» | «Excess» | |
| SDW [kg/kg] | <300 0.1% gel dose does not bind all the water in the soil pores. | 300–600 0.1% dose of gel binds all the water in the pores of the soil. | >600 risk of viscous gel flow and its leaching from the soil. |
| SDS/4 [%] | <20 poor resistance to pressure in the soil. | 20–50 normal resistance to pressure in the soil. | >50 excessive stability; risk of poor water recovery. |
| ΔSD1/4 [%] | >98 loosely stitched polymer mesh, risk of gel leaching. | 90–98 normal strength of polymer mesh for soil conditioners. | <90 excessively strong polymer mesh, risk of loss of swelling. |
| Sst [m2/g] | <300 low dispersity, risks of poor water, agrochemicals and pesticides retention in the soil. | 300–800 normal dispersity, effective water, agrochemicals and pesticides retention in the soil. | >800 high dispersity, excessively strong water, agrochemicals and pesticides retention in the soil. |
| Et [J/kg] | <6000 low water retention, sorption capacity and structuring effect in the soil. | 60,000–160,000 normal surface energy of hydrogels providing optimal technological properties in the soil. | >160,000 excessively high surface energy; risks of poor aeration and clumpiness in the soil. |
| CcrWcr [mmol/kg] (if z = 1) | <20 low coagulation threshold; risk of loss of aggregate stability during drying and freezing of gels | 20–40 moderate coagulation threshold; normal aggregate stability and recovery during drying and freezing of gels | >40 high coagulation threshold; high aggregative stability and resistance to drying and freezing of gels |
| T0.5 [yr] | <2 low biodegradable stability; complex use of gels in soils is not cost-effective | 2–10 stability of gels acceptable for their effective use in the soil | >10 increased biodegradable stability, polymers cannot be used in control release systems |
| EC50 [ppm] | >200 Insufficient suppression of pathogens in the rhizosphere | 20–200 normal effectiveness of pathogen suppression in the rhizosphere | <20 excessive dose of biocides, risk of damage to plants and pedofauna of the rhizosphere |
| Indicators | Gradations of Quality; Comments | ||
|---|---|---|---|
| «Lack» | «Norm» | «Excess» | |
| γ1 = Sst/Sst0 | <2 weak increase in dispersity; poor quality or too small dose of soil conditioner. | 2–6 normal increase in dispersity; good structuring and water-retaining effect. | >6 strong increase in dispersity; high quality gels, doses can be reduced |
| γ2 = Et/Et0 | <1.5 weak increase in surface energy; poor quality or too small dose of soil conditioner. | 1.5–3 normal increase in surface energy; good technological properties of soil conditioners | >3 strong increase in surface energy; high quality gels, doses can be reduced |
| γ3 = (AG0/AG)2 | <2 weak aggregative stability, high risk of gel collapse under salinity | 2–4 normal aggregative stability; gels can be used in slightly saline soils | >4 high aggregative stability; gels are suitable for moderately saline soils and water |
| FC [%] | <10 low water capacity at the level of sandy substrates; poor quality or too small dose of soil conditioner | 10–20 normal water capacity at the level of sandy loams and loams; good quality gels | >20 high water capacity at the level silty and clayey loams; very good quality gels, doses can be reduced |
| AWR = FC − WP [%] | <8 lack of available water, poor effect of soil conditioners | 8–16 normal range for fertile soil, good effect from soil conditioners | >16 wide range of available water, high quality gels, doses can be reduced |
| K0 [m/day] | <0.3 low water conductivity, stagnant water, risk of poor soil aeration | 0.3–1 normal water conductivity, acceptable infiltration losses, good aeration and gas exchange with the atmosphere | >1 high water conductivity, large infiltration water losses |
| K0S/K0 | <5 rather weak decrease in water conductivity; poor quality or too small dose of soil conditioner | 5–40 effective reduction of infiltration; good quality air conditioner | >40 excessive reduction in infiltration; risks of stagnant water and low soil aeration |
| Q0W/Q or Q0S/Q within 5 days | <1.5 insufficient evaporation reduction. | 1.5–3 normal evaporation reduction efficiency. | >3 high evaporation reduction efficiency |
| PA [Pa] | <50 low particle consolidation, weak soil resistance to erosion | 50–100 normal particle consolidation and soil resistance to erosion | >100 high particle consolidation and soil resistance to erosion |
| Hydrogels/Treatment | EC50 | EC95 |
|---|---|---|
| Ionic silver | ||
| Aquasorb | 42.0 ± * 4.4 | 448 ± 31 |
| A11 | 63.0 ± 19.6 | 467 ± 29 |
| A22 | 17.0 ± 5.4 | 406 ± 44 |
| LSD0.05 * | 24.0 | 70.6 |
| LSD0.01 * | 36.4 | 106.9 |
| Silver nanoparticles | ||
| Aquasorb | 1.4 ± 0.2 | 264 ± 13 |
| A11 | 0.9 ± 0.1 | 272 ± 80 |
| A22 | 5.8 ± 2.4 | 241 ± 90 |
| LSD0.05 | 2.8 | 139.9 |
| LSD0.01 | 4.2 | 211.8 |
| Azoxystrobin in sythetic fungicide Quadris | ||
| Aquasorb | 14.1 ± 3.3 | 237 ± 18 |
| A11 | 4.9 ± 3.1 | 222 ± 10 |
| A22 | 17.3 ± 6.5 | 187 ± 13 |
| LSD0.05 | 9.1 | 28.1 |
| LSD0.01 | 13.9 | 42.6 |
| Hydrogels, Doses % | FC, % | WP, % | AWR,% | Ws, % | Wr, % | α, kPa−1 | n | Et, J/kg | Sst, m2/g | AG, 10−19 J |
|---|---|---|---|---|---|---|---|---|---|---|
| Monomineral quartz sands: | ||||||||||
| 0% | 4.6 | 1.3 | 3.3 | 27.5 | 1.3 | 0.310 | 2.26 | 2033 | 7.4 | 3.96 |
| 0% | 3.3 | 1.7 | 1.6 | 29.3 | 1.6 | 1.655 | 1.87 | 1973 | 7.2 | 3.95 |
| 0% | 3.0 | 0.6 | 2.4 | 26.4 | 0.6 | 0.523 | 2.11 | 727 | 3.1 | 3.22 |
| Aquasorb | ||||||||||
| 0.1% | 19.2 | 4.2 | 15.0 | 38.7 | 2.8 | 0.146 | 1.61 | 3320 | 26.6 | 1.79 |
| 0.2% | 26.0 | 8.3 | 17.6 | 49.2 | 3.8 | 0.209 | 1.40 | 6928 | 46.8 | 2.12 |
| 0.3% | 28.5 | 9.8 | 18.7 | 48.6 | 1.4 | 0.185 | 1.30 | 11410 | 63.5 | 2.58 |
| A22 (23% Peat) | ||||||||||
| 0.1% | 18.6 | 4.0 | 14.5 | 37.0 | 1.8 | 0.183 | 1.50 | 4212 | 26.5 | 2.28 |
| 0.1% + Ag 0.1% | 17.0 | 4.1 | 12.9 | 35.0 | 2.9 | 0.169 | 1.61 | 3573 | 28.2 | 1.82 |
| 0.1% + Ag 1% | 19.5 | 4.1 | 15.3 | 38.6 | 1.2 | 0.199 | 1.45 | 3050 | 26.0 | 1.68 |
| 0.2% | 23.7 | 5.8 | 17.9 | 40.7 | 2.0 | 0.130 | 1.44 | 5073 | 36.1 | 2.02 |
| 0.2% + Ag 0.1% | 22.7 | 6.6 | 16.1 | 39.6 | 1.7 | 0.188 | 1.36 | 5836 | 42.5 | 1.97 |
| 0.2% + Ag 1% | 21.6 | 6.9 | 14.6 | 38.6 | 5.6 | 0.118 | 1.63 | 7685 | 44.4 | 2.49 |
| 0.3% | 28.6 | 9.5 | 19.1 | 51.9 | 2.7 | 0.213 | 1.34 | 9726 | 56.8 | 2.46 |
| 0.3% + Ag 0.1% | 27.4 | 9.9 | 17.5 | 48.2 | 2.8 | 0.232 | 1.32 | 1154 | 53.6 | 2.97 |
| 0.3% + Ag 1% | 29.0 | 9.6 | 19.4 | 48.9 | 0.1 | 0.203 | 1.29 | 8206 | 61.1 | 1.93 |
| A11 (28% biocatalytic waste) | ||||||||||
| 0.1% | 10.2 | 3.6 | 6.6 | 37.6 | 2.4 | 1.913 | 1.42 | 3753 | 22.6 | 2.38 |
| 0.2% | 20.7 | 9.6 | 11.1 | 53.0 | 0.2 | 6.459 | 1.19 | 5630 | 62.7 | 1.29 |
| 0.3% | 28.4 | 11.4 | 17.0 | 50.0 | 1.9 | 0.323 | 1.26 | 7415 | 72.9 | 1.46 |
| A11H (12% Humates) | ||||||||||
| 0.1% | 8.6 | 4.5 | 4.1 | 50.0 | 3.9 | 7.257 | 1.47 | 3822 | 24.8 | 2.22 |
| 0.2% | 16.7 | 7.2 | 9.5 | 48.6 | 0.1 | 9.817 | 1.20 | 5652 | 45.7 | 1.74 |
| 0.3% | 28.1 | 9.7 | 18.3 | 47.5 | 0.0 | 0.230 | 1.27 | 6929 | 62.2 | 1.60 |
| A11HMZ (12% Humates, Zn, Mg 0.4%) | ||||||||||
| 0.1% | 9.4 | 4.6 | 4.8 | 57.0 | 3.9 | 7.337 | 1.46 | 4561 | 25.6 | 2.56 |
| 0.2% | 15.2 | 6.2 | 9.0 | 53.0 | 1.9 | 7.389 | 1.27 | 4587 | 39.1 | 1.68 |
| 0.3% | 29.7 | 9.5 | 20.2 | 53.3 | 0.2 | 0.231 | 1.29 | 6666 | 56.6 | 1.69 |
| A11Ag (ionic silver 1%) | ||||||||||
| 0.1% | 10.9 | 2.8 | 8.2 | 29.8 | 0.0 | 1.234 | 1.32 | 4025 | 21.5 | 2.69 |
| 0.2% | 17.2 | 5.6 | 11.6 | 42.6 | 0.0 | 1.360 | 1.27 | 7251 | 38.2 | 2.72 |
| 0.3% | 27.0 | 9.7 | 17.3 | 57.8 | 0.0 | 0.688 | 1.26 | 12547 | 68.4 | 3.26 |
| A11Az (10% Filterperlite, Azoxystorobin 1%) | ||||||||||
| 0.1% | 9.5 | 1.8 | 7.7 | 33.4 | 0.3 | 1.206 | 1.41 | 3303 | 16.6 | 2.86 |
| 0.2% | 16.1 | 4.7 | 11.4 | 41.8 | 0.0 | 1.203 | 1.29 | 6987 | 34.2 | 2.94 |
| 0.3% | 23.8 | 8.6 | 15.3 | 51.6 | 0.0 | 0.823 | 1.25 | 12553 | 56.8 | 3.17 |
| A22Qv (23% Peat, Quadris 1%) | ||||||||||
| 0.1% | 10.3 | 2.1 | 8.2 | 34.6 | 0.3 | 1.227 | 1.39 | 3951 | 17.6 | 3.23 |
| 0.2% | 19.1 | 7.1 | 11.9 | 48.1 | 0.0 | 2.195 | 1.24 | 10326 | 44.4 | 3.32 |
| 0.3% | 25.4 | 9.4 | 16.0 | 52.6 | 0.0 | 0.705 | 1.25 | 14737 | 64.2 | 3.29 |
| Hydrogels, doses % | FC, % | WP, % | AWR,% | Ws, % | Wr, % | α, kPa−1 | n | R2 | s, % | p-Value |
|---|---|---|---|---|---|---|---|---|---|---|
| Loamy-sandy Arenosol from the Karakum Desert, Aquasorb | ||||||||||
| 0% | 9.8 | 1.7 | 8.0 | 22.9 | 1.7 | 0.093 | 2.11 | 0.997 | 0.6 | 0.0005 |
| 0.1% | 12.5 | 2.6 | 9.9 | 27.4 | 2.5 | 0.111 | 2.06 | 0.998 | 0.6 | 0.0001 |
| 0.2% | 14.4 | 4.1 | 10.3 | 33.0 | 4.0 | 0.110 | 2.16 | 0.996 | 0.8 | 0.0002 |
| 0.3% | 18.4 | 6.5 | 12.0 | 35.1 | 6.1 | 0.112 | 1.83 | 0.995 | 1.0 | 0.0015 |
| Loamy-sandy Arenosol from the Karakum Desert, A11 | ||||||||||
| 0% | 9.8 | 1.7 | 8.0 | 22.9 | 1.7 | 0.093 | 2.11 | 0.997 | 0.6 | 0.0005 |
| 0.1% | 14.3 | 3.3 | 11.0 | 28.3 | 3.2 | 0.089 | 2.14 | 0.996 | 0.8 | 0.0007 |
| 0.2% | 16.0 | 5.0 | 11.0 | 30.8 | 4.8 | 0.105 | 1.92 | 0.993 | 1.0 | 0.0030 |
| 0.3% | 16.8 | 6.3 | 10.5 | 34.8 | 6.0 | 0.123 | 1.93 | 0.993 | 1.1 | 0.0023 |
| Loamy-sandy Arenosol from Dubai, Aquasorb | ||||||||||
| 0% | 18.0 | 4.7 | 13.3 | 26.1 | 0.0 | 0.111 | 1.34 | 0.995 | 0.7 | 0.0050 * |
| 0.1% | 19.7 | 4.2 | 15.5 | 26.8 | 0.0 | 0.069 | 1.40 | 0.992 | 1.6 | 0.0240 * |
| 0.2% | 19.2 | 4.1 | 15.1 | 27.9 | 0.0 | 0.091 | 1.39 | 0.995 | 0.9 | 0.0008 * |
| 0.3% | 21.2 | 4.6 | 16.6 | 32.6 | 0.9 | 0.097 | 1.43 | 0.992 | 1.4 | 0.0051 * |
| Loamy-sandy Arenosol from Dubai, A22 | ||||||||||
| 0% | 18.0 | 4.7 | 13.3 | 26.1 | 0.0 | 0.111 | 1.34 | 0.995 | 0.7 | 0.0050 * |
| 0.1% | 21.5 | 4.8 | 16.7 | 27.6 | 0.0 | 0.053 | 1.41 | 0.997 | 0.8 | 0.0005 * |
| 0.2% | 22.1 | 4.3 | 17.7 | 28.6 | 0.0 | 0.051 | 1.44 | 0.996 | 0.8 | 0.0019 * |
| 0.3% | 26.7 | 6.9 | 20.0 | 35.0 | 0.0 | 0.052 | 1.37 | 0.999 | 0.4 | 0.0001 * |
| Indicators: | Corg, % | Temperature: | ||
|---|---|---|---|---|
| 4 °C | 20 °C | 30 °C | ||
| A11 in quartz sand | ||||
| k0, yr−1 | 0.071 | 0.24 | 1.09 | 1.88 |
| T0.5, yr | 2.8 | 0.6 | 0.4 | |
| A22Ag in quartz sand | ||||
| k0, yr−1 | 0.070 | 0.01 | 0.08 | 0.14 |
| T0.5, yr | 76.4 | 8.5 | 5.1 | |
| A11in loamy-sandy Arenosol (UAE, Dubai) | ||||
| k0, yr−1 | 0.098 | 0.30 | 1.19 | 2.35 |
| T0.5, yr | 2.3 | 0.6 | 0.3 | |
| A22Ag in loamy-sandy Arenosol (UAE, Dubai) | ||||
| k0, yr−1 | 0.088 | 0.02 | 0.06 | 0.13 |
| T0.5, yr | 37.3 | 10.7 | 5.5 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smagin, A.V.; Budnikov, V.I.; Sadovnikova, N.B.; Kirichenko, A.V.; Belyaeva, E.A.; Krivtsova, V.N. Gel-Forming Soil Conditioners of Combined Action: Laboratory Tests for Functionality and Stability. Polymers 2022, 14, 4665. https://doi.org/10.3390/polym14214665
Smagin AV, Budnikov VI, Sadovnikova NB, Kirichenko AV, Belyaeva EA, Krivtsova VN. Gel-Forming Soil Conditioners of Combined Action: Laboratory Tests for Functionality and Stability. Polymers. 2022; 14(21):4665. https://doi.org/10.3390/polym14214665
Chicago/Turabian StyleSmagin, Andrey V., Viktor I. Budnikov, Nadezhda B. Sadovnikova, Anatoly V. Kirichenko, Elena A. Belyaeva, and Victoria N. Krivtsova. 2022. "Gel-Forming Soil Conditioners of Combined Action: Laboratory Tests for Functionality and Stability" Polymers 14, no. 21: 4665. https://doi.org/10.3390/polym14214665
APA StyleSmagin, A. V., Budnikov, V. I., Sadovnikova, N. B., Kirichenko, A. V., Belyaeva, E. A., & Krivtsova, V. N. (2022). Gel-Forming Soil Conditioners of Combined Action: Laboratory Tests for Functionality and Stability. Polymers, 14(21), 4665. https://doi.org/10.3390/polym14214665







