Impact of Traces of Hydrogen Sulfide on the Efficiency of Ziegler–Natta Catalyst on the Final Properties of Polypropylene
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Polymerization
2.3. Analysis
3. Results
3.1. Polymerization Stage
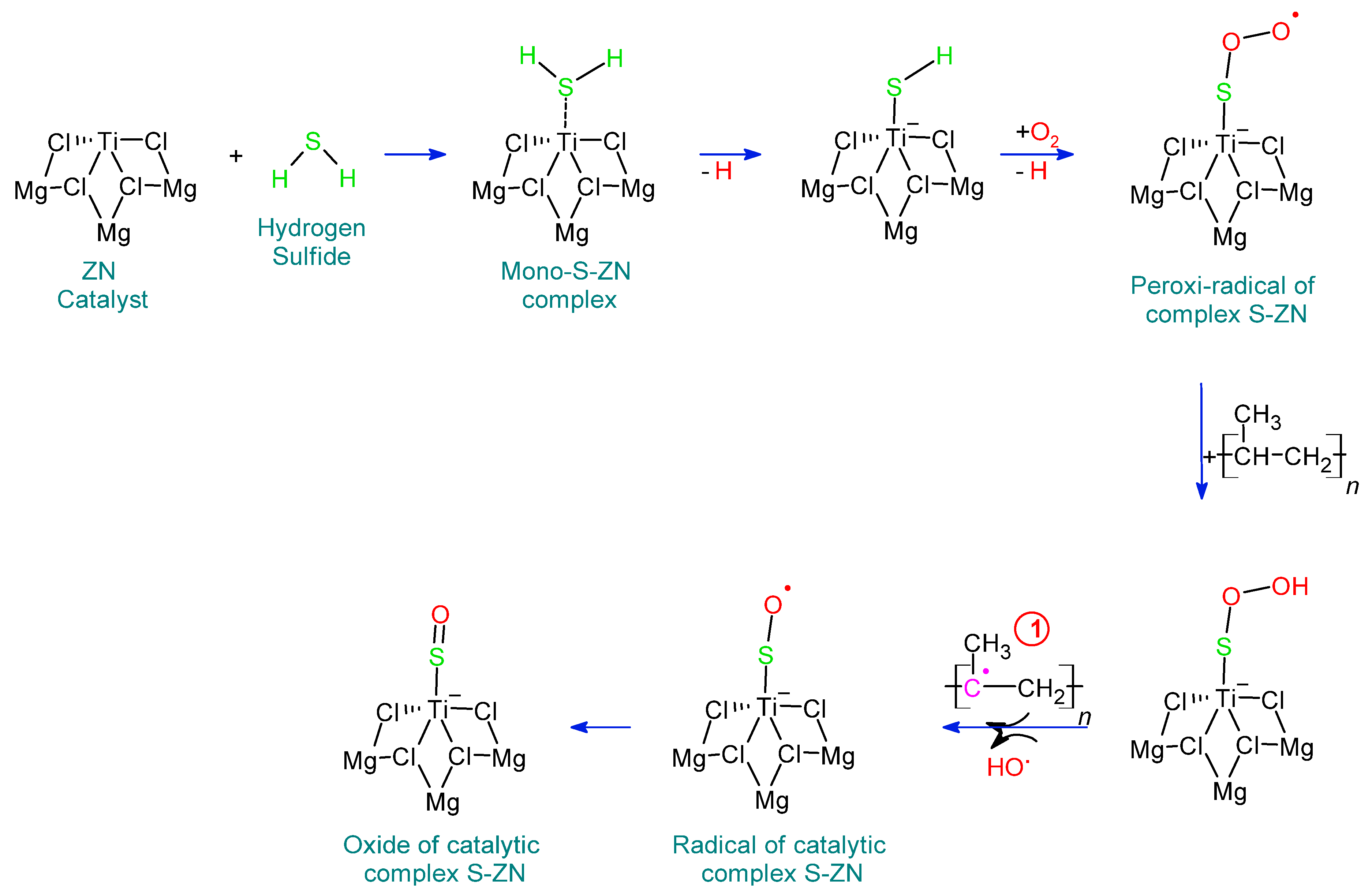
Identification of H2S in the Polymerization Stage and Proposed Reaction Mechanism Reaction of H2S with TiCl4/MgCl2
3.2. The Degassing Stage
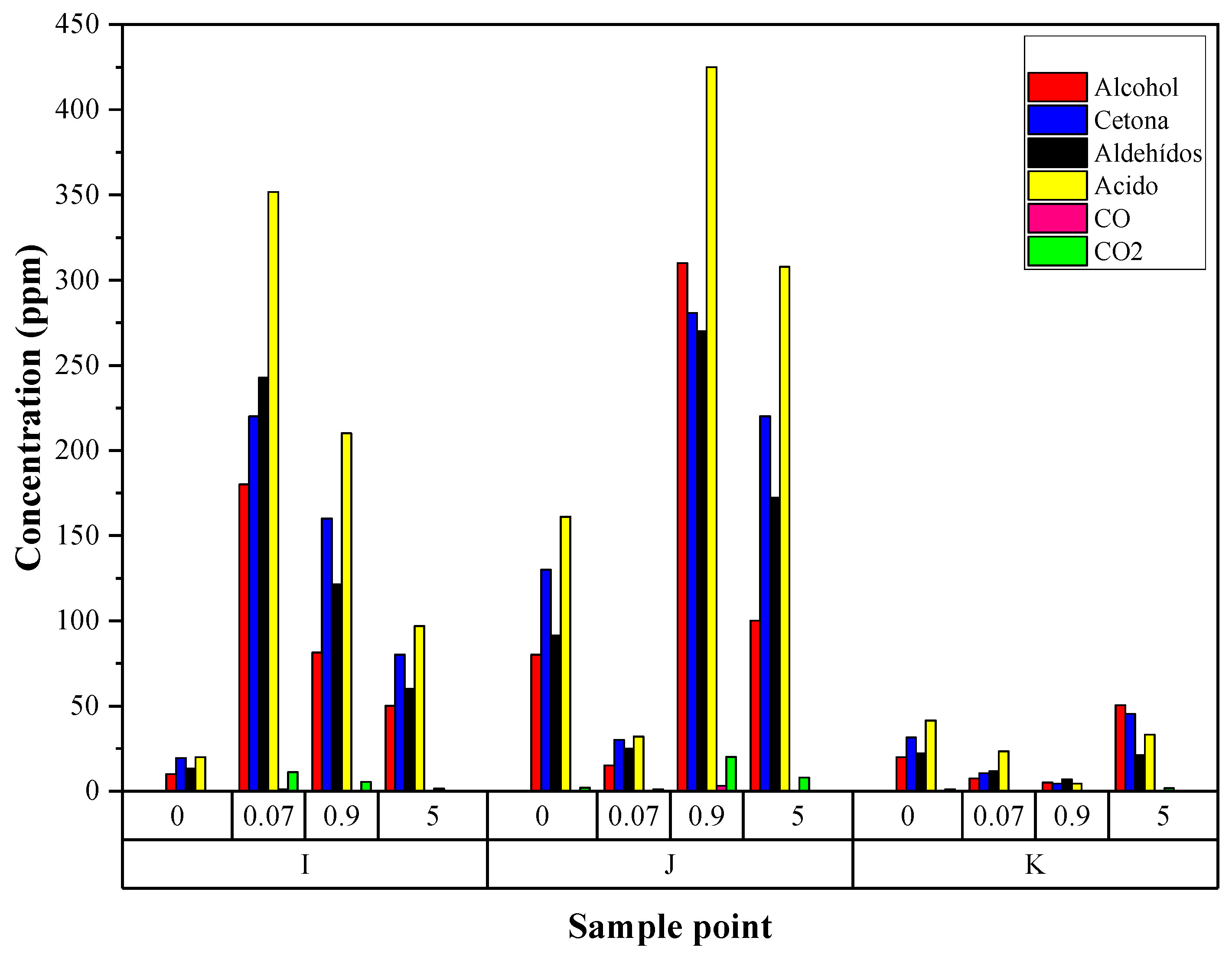
Detection and Quantification of H2S in PP Resin Degassing
3.3. Influence of H2S on the Properties of PP
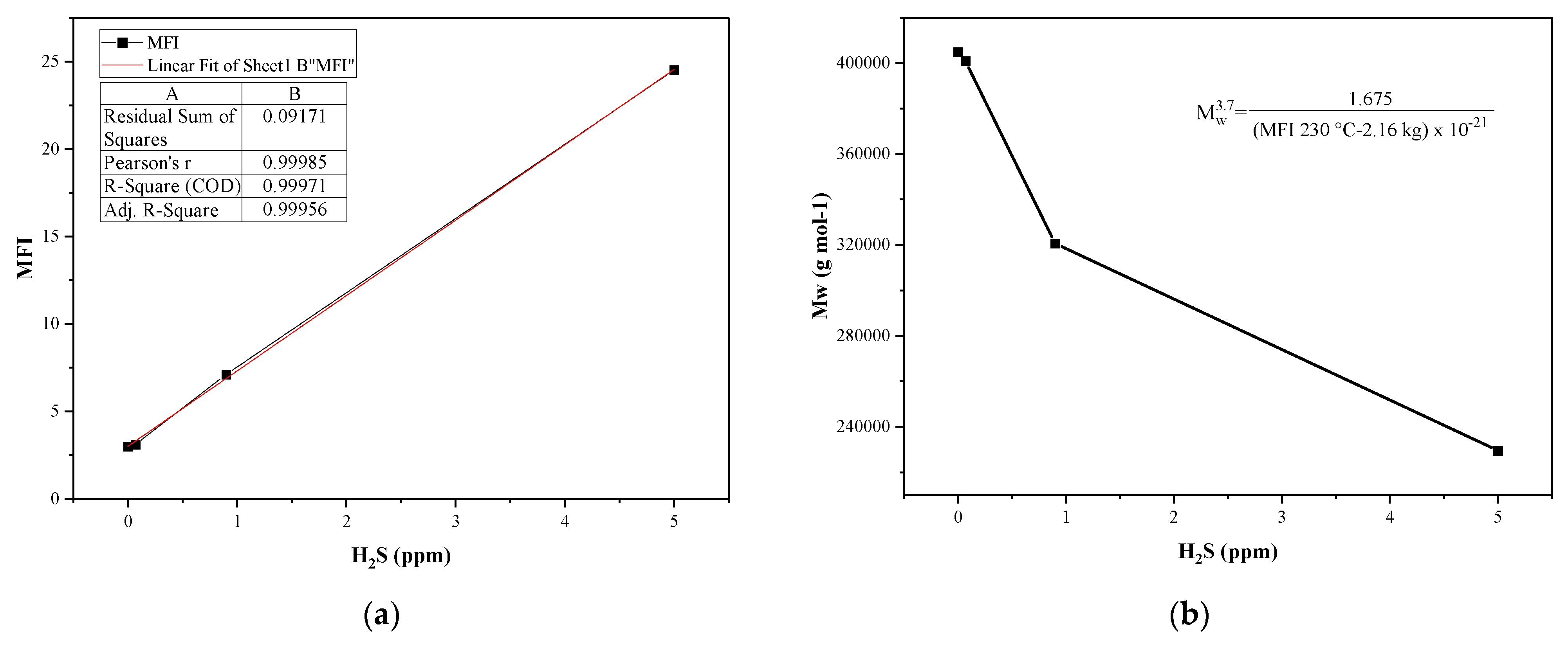
3.3.1. Effects on the Melt Flow Index (MFI) of PP
3.3.2. Thermal Degradation of the Material
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kurahashi, E.; Wada, T.; Nagai, T.; Chammingkwan, P.; Terano, M.; Taniike, T. Synthesis of Polypropylene Functionalized with a Trace Amount of Reactive Functional Groups and Its Utilization in Graft-Type Nanocomposites. Polymer 2018, 158, 46–52. [Google Scholar] [CrossRef]
- Karol, F.J.; Jacobson, F.I. Catalysis and the Unipol Process. In Studies in Surface Science and Catalysis; Elsevier: Amsterdam, The Netherlands, 1986; Volume 25. [Google Scholar]
- Mier, J.; Artiaga, R.; García Soto, L. Síntesis de Polímeros. Pesos Moleculares. Conformación y Configuración. In Elementos Estructurales con Materiales Polímeros: Ferrol; Universidade, Servicio de Publicacións: A Coruña, Spain, 1997; pp. 11–48. [Google Scholar]
- Bailar, J.C.; Emeléus, H.J.; Nyholm, R.; Trotman-Dickenson, A.F. Comprehensive Inorganic Chemistry; Elsevier: Amsterdam, The Netherlands, 1973; Volume 3, ISBN 9781483283135. [Google Scholar]
- Nikolaeva, M.; Mikenas, T.; Matsko, M.; Zakharov, V. Effect of AlEt3 and an External Donor on the Distribution of Active Sites According to Their Stereospecificity in Propylene Polymerization over TiCl4/MgCl2 Catalysts with Different Titanium Content. Macromol. Chem. Phys. 2016, 217, 1384–1395. [Google Scholar] [CrossRef]
- Nikolaevna Panchenko, V.; Viktorovna Vorontsova, L.; Aleksandrovich Zakharov, V. Ziegler-Natta Catalysts for Propylene Polymerization—Interaction of an External Donor with the Catalyst. Polyolefins J. 2017, 4, 87–97. [Google Scholar] [CrossRef]
- Vizen, E.I.; Rishina, L.A.; Sosnovskaja, L.N.; Dyachkovsky, F.S.; Dubnikova, I.L.; Ladygina, T.A. Study of Hydrogen Effect in Propylene Polymerization on (with) the MgCl2-Supported Ziegler-Natta Catalyst-Part 2. Effect of CS2 on Polymerization Centres. Eur. Polym. J. 1994, 30, 1315–1318. [Google Scholar] [CrossRef]
- Kallio, K.; Wartmann, A.; Reichert, K.-H. Reactivation of a Poisoned Metallocene Catalyst by Irradiation with Visible Light; Wiley: Hoboken, NJ, USA, 2002; Volume 23. [Google Scholar]
- Bahri-Laleh, N. Interaction of Different Poisons with MgCl2/TiCl4 Based Ziegler-Natta Catalysts. Appl. Surf. Sci. 2016, 379, 395–401. [Google Scholar] [CrossRef]
- Asynkiewicz, S.P. Reactions of Organoaluminium Compounds with Electron Donors. Pure Appl. Chem. 1972, 30, 509–522. [Google Scholar] [CrossRef]
- Hernández-Fernández, J. Quantification of Oxygenates, Sulphides, Thiols and Permanent Gases in Propylene. A Multiple Linear Regression Model to Predict the Loss of Efficiency in Polypropylene Production on an Industrial Scale. J. Chromatogr. A 2020, 1628, 461478. [Google Scholar] [CrossRef]
- Li, Z.; Yin, Y.; Wang, X.; Tu, D.M.; Kao, K.C. Formation and Inhibition of Free Radicals in Electrically Stressed and Aged Insulating Polymers. J. Appl. Polym. Sci. 2003, 89, 3416–3425. [Google Scholar] [CrossRef]
- Biswal, H.S. Hydrogen Bonds Involving Sulfur: New Insights from Ab Initio Calculations and Gas Phase Laser Spectroscopy. In Challenges and Advances in Computational Chemistry and Physics; Springer: Berlin/Heidelberg, Germany, 2015; Volume 19, pp. 15–45. ISBN 9783319141633. [Google Scholar]
- Kaushik, R.; Ghosh, A.; Amilan Jose, D. Recent Progress in Hydrogen Sulphide (H2S) Sensors by Metal Displacement Approach. Coord. Chem. Rev. 2017, 347, 141–157. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X. Hydrogen Bonding in the Complexes Formed by Arsine and H-X Molecules: A Theoretical Study. Chem. Phys. Lett. 2019, 735, 136767. [Google Scholar] [CrossRef]
- Barnabas, F.A. Solution Reactions of HX Molecules (X = SH, CI, Br) With Dinuclear Palladium(I) Complexes Containing Bis(Diphenylphosphino)Methane; University of British Columbia: Vancouver, BC, Canada, 1989. [Google Scholar]
- Pluth, M.D.; Tonzetich, Z.J. Hydrosulfide Complexes of the Transition Elements: Diverse Roles in Bioinorganic, Cluster, Coordination, and Organometallic Chemistry. Chem. Soc. Rev. 2020, 49, 4070–4134. [Google Scholar] [CrossRef]
- Livingstone, S.E. Metal Complexes of Ligands Containing Sulphur, Selenium, or Tellurium as Donor Atoms. Q. Rev. Chem. Soc. 1965, 19, 386. [Google Scholar] [CrossRef]
- Lindoy, L.F. Reactions Involving Metal Complexes of Sulphur Ligands. Coord. Chem. Rev. 1969, 4, 41–71. [Google Scholar] [CrossRef]
- Enríquez Rodríguez, M. Funcionalización de Ligandos Coordinados; Universidad de Coruña: A Coruña, Spain, 2017. [Google Scholar]
- Hernández-Fernández, J. Quantification of Arsine and Phosphine in Industrial Atmospheric Emissions in Spain and Colombia. Implementation of Modified Zeolites to Reduce the Environmental Impact of Emissions. Atmos. Pollut. Res. 2021, 12, 167–176. [Google Scholar] [CrossRef]
- Mensforth, E.J.; Hill, M.R.; Batten, S.R. Coordination Polymers of Sulphur-Donor Ligands. Inorganica. Chim. Acta 2013, 403, 9–24. [Google Scholar] [CrossRef]
- Jafarinejad, S. Control and Treatment of Sulfur Compounds Specially Sulfur Oxides (SOx) Emissions from the Petroleum Industry: A Review. Chem. Int. 2016, 2, 242–253. [Google Scholar]
- Shi, Q.; Wu, J. Review on Sulfur Compounds in Petroleum and Its Products: State-of-the-Art and Perspectives. Energy Fuels 2021, 35, 14445–14461. [Google Scholar] [CrossRef]
- Zhang, L.L.; Wang, C.L.; Zhao, Y.S.; Yang, G.H.; Su, M.; Yang, C.H. Speciation and Quantification of Sulfur Compounds in Petroleum Asphaltenes by Derivative XANES Spectra. J. Fuel Chem. Technol. 2013, 41, 1328–1335. [Google Scholar] [CrossRef]
- Han, Y.; Zhang, Y.; Xu, C.; Hsu, C.S. Molecular Characterization of Sulfur-Containing Compounds in Petroleum. Fuel 2018, 221, 144–158. [Google Scholar] [CrossRef]
- Joaquin, H.-F.; Juan, L. Quantification of Poisons for Ziegler Natta Catalysts and Effects on the Production of Polypropylene by Gas Chromatographic with Simultaneous Detection: Pulsed Discharge Helium Ionization, Mass Spectrometry and Flame Ionization. J. Chromatogr. A 2020, 1614, 460736. [Google Scholar] [CrossRef]
- van Krevelen, D.W.; te Nijenhuis, K. Typology of Properties. In Properties of Polymers; Elsevier: Amsterdam, The Netherlands, 2009; pp. 49–67. [Google Scholar] [CrossRef]
- Halasa, A.F.; Massie, J.M.; Ceresa, R.J. The Chemical Modification of Polymers. In The Science and Technology of Rubber; Academic Press: Cambridge, MA, USA, 2013; pp. 517–546. [Google Scholar] [CrossRef]
- Soroush, M.; Grady, M.C. Polymers, Polymerization Reactions, and Computational Quantum Chemistry. In Computational Quantum Chemistry; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–16. [Google Scholar] [CrossRef]
- Chong, B.Y.K.; Krstina, J.; Le, T.P.T.; Moad, G.; Postma, A.; Rizzardo, E.; Thang, S.H. Thiocarbonylthio Compounds [S=C(Ph)S-R) in Free Radical Polymerization with Reversible Addition-Fragmentation Chain Transfer (RAFT Polymerization). Role of the Free-Radical Leaving Group (R). Macromolecules 2003, 36, 2256–2272. [Google Scholar] [CrossRef]
- Shen, X.R.; Fu, Z.S.; Hu, J.; Wang, Q.; Fan, Z.Q. Mechanism of Propylene Polymerization with MgCl2-Supported Ziegler-Natta Catalysts Based on Counting of Active Centers: The Role of External Electron Donor. J. Phys. Chem. C 2013, 117, 15174–15182. [Google Scholar] [CrossRef]
- Otsu, T.; Matsumoto, A. Controlled Synthesis of Polymers Using the Iniferter Technique: Developments in Living Radical Polymerization. In Microencapsulation Microgels Iniferters; Springer: Berlin/Heidelberg, Germany, 1998; pp. 75–137. [Google Scholar]
- Hernández-Fernandez, J.; Rodríguez, E. Determination of Phenolic Antioxidants Additives in Industrial Wastewater from Polypropylene Production Using Solid Phase Extraction with High-Performance Liquid Chromatography. J. Chromatogr. A 2019, 1607, 460442. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; Lopez-Martinez, J.; Barceló, D. Quantification and Elimination of Substituted Synthetic Phenols and Volatile Organic Compounds in the Wastewater Treatment Plant during the Production of Industrial Scale Polypropylene. Chemosphere 2021, 263, 128027. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; López-Martínez, J. Experimental Study of the Auto-Catalytic Effect of Triethylaluminum and TiCl4 Residuals at the Onset of Non-Additive Polypropylene Degradation and Their Impact on Thermo-Oxidative Degradation and Pyrolysis. J. Anal. Appl. Pyrolysis. 2021, 155, 105052. [Google Scholar] [CrossRef]
- Zhang, S.; Li, B.; Lin, M.; Li, Q.; Gao, S.; Yi, W. Effect of a Novel Phosphorus-Containing Compound on the Flame Retardancy and Thermal Degradation of Intumescent Flame Retardant Polypropylene. J. Appl. Polym. Sci. 2011, 122, 3430–3439. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; Rayón, E.; López, J.; Arrieta, M.P. Enhancing the Thermal Stability of Polypropylene by Blending with Low Amounts of Natural Antioxidants. Macromol. Mater. Eng. 2019, 304, 1900379. [Google Scholar] [CrossRef]
- Standard Test Method for Melt Flow Rates of Thermoplastics by Extrusion Plastometer. Available online: https://www.astm.org/d1238-10.html (accessed on 28 August 2022).
- Bremner, T.; Rudin, A.; Cook, D.G. Melt Flow Index Values and Molecular Weight Distributions of Commercial Thermoplastics. J. Appl. Polym. Sci. 1990, 41, 1617–1627. [Google Scholar] [CrossRef]
- Ivin, K.J.; Rooney, J.J.; Stewart, C.D.; Green, M.L.H.; Mahtab, R. Mechanism for the Stereospecific Polymerization of Olefins by Ziegler–Natta Catalysts. J. Chem. Soc. Chem. Commun. 1978, 14, 604–606. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; Guerra, Y.; Puello-Polo, E.; Marquez, E. Effects of Different Concentrations of Arsine on the Synthesis and Final Properties of Polypropylene. Polymers 2022, 14, 3123. [Google Scholar] [CrossRef] [PubMed]
- Padilla Paz, R.M. Síntesis y Estudio de Cpmplejos Organometálicos de Iridio Con N-Aril-4,5-Dimetilen-1,3-Oxazolidin-2-Onas y Complejos de Cobre Con Furoiltioureas; Universidad Autónoma del estado de Hidalgo: Hidalgo, Mexico, 2006. [Google Scholar]
- Eguren, L.; Korswagen, R. Catalizadores Ziegler-Natta Utilizados Para Polimerizar Propileno y Etileno. Revista de Química 1987, 1, 5–13. [Google Scholar]
- NATTA, G.; PASQUON, I.; GIACHETTI, E. Kinetics of the Stereospecific Polymerization of Polypropylene to Isotactic Polymers. In Stereoregular Polymers and Stereospecific Polymerizations; Elsevier: Amsterdam, The Netherlands, 1967. [Google Scholar]
- Zakharov, I.I.; Zakharov, V.A.; Zhidomirov, G.M. Quantum Chemical Studies of Propene, Ethylene, Acetylene and Dihydrogen Reactivity in the Insertion Reaction into the Titanium-Alkyl Bond; Wiley: Hoboken, NJ, USA, 1996; Volume 5. [Google Scholar]
- Cheremisinoff, N.P. Handbook of Polymer Science and Technology: Synthesis and Properties; Dekker, M., Ed.; Routledge: London, UK, 1989. [Google Scholar]
- Chien, J.C.W.; Bres, P. Magnesium Chloride Supported High Mileage Catalysts for Olefin Polymerization. XIII. Effect of External Lewis Base on Ethylene Polymerization. J. Polym. Sci. A Polym. Chem. 1986, 24, 1967–1988. [Google Scholar] [CrossRef]
- Bhaduri, S.; Mukhopadhyay, S.; Kulkarni, S.A. Role of Titanium Oxidation States in Polymerization Activity of Ziegler-Natta Catalyst: A Density Functional Study. J. Organomet. Chem. 2006, 691, 2810–2820. [Google Scholar] [CrossRef]
- Clough, R.L. Isotopic Exchange in Gamma-irradiated Mixtures of C24H50 and C24D50: Evidence of Free Radical Migration in the Solid State. J. Chem. Phys. 1987, 87, 1588. [Google Scholar] [CrossRef]
- Bahlouli, N.; Pessey, D.; Raveyre, C.; Guillet, J.; Ahzi, S.; Dahoun, A.; Hiver, J.M. Recycling Effects on the Rheological and Thermomechanical Properties of Polypropylene-Based Composites. Mater. Des. 2012, 33, 451–458. [Google Scholar] [CrossRef]
- Aurrekoetxea, J.; Sarrionandia, M.A.; Urrutibeascoa, I.; Maspoch, M.L. Effects of Recycling on the Microstructure and the Mechanical Properties of Isotactic Polypropylene. J. Mater. Sci. 2001, 36, 2607–2613. [Google Scholar] [CrossRef]
- Alvarado Chacon, F.; Brouwer, M.T.; Thoden van Velzen, E.U.; Smeding, I.W. A First Assessment of the Impact of Impurities in PP and PE Recycled Plastics; Wageningen Food & Biobased Research: Wageningen, The Netherlands, 2020. [Google Scholar]
- Sheng, B.-R.; Li, B.; Xie, B.-H.; Yang, W.; Feng, J.-M.; Yang, M.-B. Influences of Molecular Weight and Crystalline Structure on Fracture Behavior of Controlled-Rheology-Polypropylene Prepared by Reactive Extrusion. Polym. Degrad. Stab. 2008, 93, 225–232. [Google Scholar] [CrossRef]
- Dusseault, J.J.A.; Hsu, C.C. MgCI2-Supported Ziegler-Natta Catalysts for Olefin Polymerization: Basic Structure, Mechanism, and Kinetic Behavior. J. Macromol. Sci. Part C 2006, 33, 103–145. [Google Scholar] [CrossRef]
- Kissin, Y.V.; Marin, V.P.; Nelson, P.J. Propylene Polymerization Reactions with Supported Ziegler–Natta Catalysts: Observing Polymer Material Produced by a Single Active Center. J. Polym. Sci. A Polym. Chem. 2017, 55, 3832–3841. [Google Scholar] [CrossRef]
- Noristi, L.; Marchetti, E.; Baruzzi, G.; Sgarzi, P. Investigation on the Particle Growth Mechanism in Propylene Polymerization with MgCl2-Supported Ziegler–Natta Catalysts. J. Polym. Sci. A Polym. Chem. 1994, 32, 3047–3059. [Google Scholar] [CrossRef]
- Ashfaq, A.; Clochard, M.C.; Coqueret, X.; Dispenza, C.; Driscoll, M.S.; Ulański, P.; Al-Sheikhly, M. Polymerization Reactions and Modifications of Polymers by Ionizing Radiation. Polymers 2020, 12, 2877. [Google Scholar] [CrossRef]
- Bhanu, V.A.; Kishore, K. Role of Oxygen in Polymerization Reactions. Chem. Rev. 1991, 91, 99–117. [Google Scholar] [CrossRef]
- Newby, T.E. Study of Spontaneous Polymerisation Inhibition. Ph.D. Thesis, University of York, York, UK, 2014. [Google Scholar]
- George, A.; Harper, P. Melt Flow Index Determination in Polymer Process Control. GB9803894.6, 25 February 1998. [Google Scholar]
- Richaud, E.; Fayolle, B.; Davies, P. Tensile Properties of Polypropylene Fibers. In Handbook of Properties of Textile and Technical Fibres; Elsevier: Amsterdam, The Netherlands, 2018; pp. 515–543. [Google Scholar] [CrossRef]
- Litvinov, V.M.; Ries, M.E.; Baughman, T.W.; Henke, A.; Matloka, P.P. Chain Entanglements in Polyethylene Melts. Why Is It Studied Again? Macromolecules 2013, 46, 541–547. [Google Scholar] [CrossRef]
- Montaudo, G.; Puglisi, C. Thermal Degradation Mechanisms in Condensation Polymers. In Developments in Polymer Degradation—7; Springer: Dordrecht, The Netherlands, 1987; pp. 35–80. [Google Scholar]
- Commereuc, S.; Vaillant, D.; Philippart, J.L.; Lacoste, J.; Lemaire, J.; Carlsson, D.J. Photo and Thermal Decomposition of IPP Hydroperoxides. Polym. Degrad. Stab. 1997, 57, 175–182. [Google Scholar] [CrossRef]
- Joaquin, H.-F.; Juan, L.-M. Autocatalytic Influence of Different Levels of Arsine on the Thermal Stability and Pyrolysis of Polypropylene. J. Anal. Appl. Pyrolysis 2022, 161, 105385. [Google Scholar] [CrossRef]
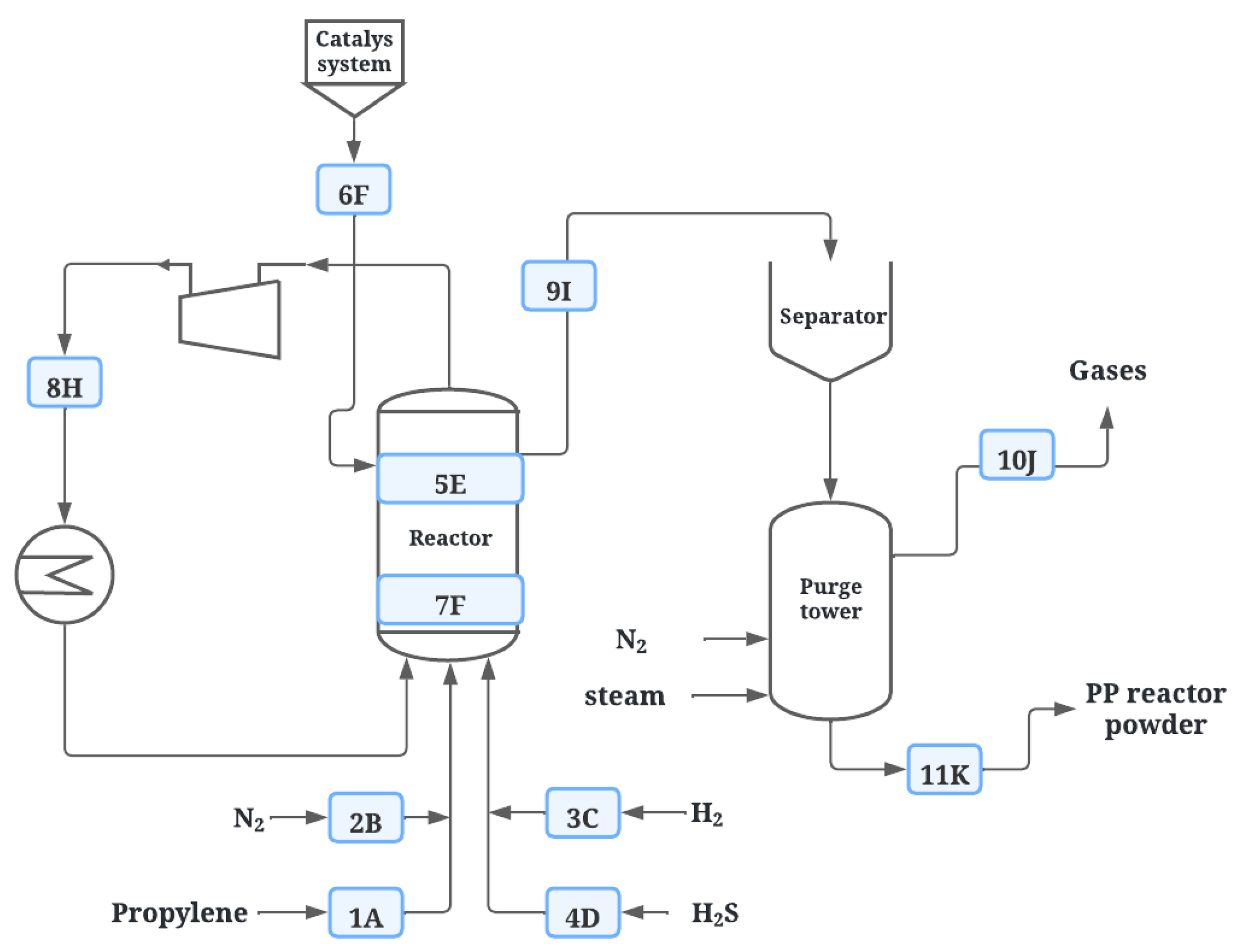

| Feeding | Reaction | Degassing | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Point | 1A | 2B | 3C | 4D | 5E | 7F | 6F | 8H | 9I | 10J | 11K |
| State of origin | LPG | Gas | Gas | LPG | Inside the reactor | Catalytic system | Recovered | Exits of reactor | Retired in the purge | Dust | |
| Substance | Propylene | N2 | H2 | H2S | Gases | ZN | ZN | Propylene | PP resin | Gases | PP resin |
| ID-0 | 1A-0 | 2B-0 | 3C-0 | 4D-0 | 5E-0 | 7F-0 | 6F-0 | 8H-0 | 9I-0 | 10J-0 | 11K-0 |
| ID-0.07 | 1A-0.07 | 2B-0.07 | 3C-0.07 | 4D-0.07 | 5E-0.07 | 7F-0.07 | 6F-0.07 | 8H-0.07 | 9I-0.07 | 10J-0.07 | 11K-0.07 |
| ID-0.9 | 1A-0.9 | 2B-0.9 | 3C-0.9 | 4D-0.9 | 5E-0.9 | 7F-0.9 | 6F-0.9 | 8H-0.9 | 9I-0.9 | 10J-0.9 | 11K-0.9 |
| ID-5 | 1A-5 | 2B-5 | 3C-5 | 4D-5 | 5E-5 | 7F-5 | 6F-5 | 8H-5 | 9I-5 | 10J-5 | 11K-5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Fernández, J.; Cano, H.; Aldas, M. Impact of Traces of Hydrogen Sulfide on the Efficiency of Ziegler–Natta Catalyst on the Final Properties of Polypropylene. Polymers 2022, 14, 3910. https://doi.org/10.3390/polym14183910
Hernández-Fernández J, Cano H, Aldas M. Impact of Traces of Hydrogen Sulfide on the Efficiency of Ziegler–Natta Catalyst on the Final Properties of Polypropylene. Polymers. 2022; 14(18):3910. https://doi.org/10.3390/polym14183910
Chicago/Turabian StyleHernández-Fernández, Joaquín, Heidi Cano, and Miguel Aldas. 2022. "Impact of Traces of Hydrogen Sulfide on the Efficiency of Ziegler–Natta Catalyst on the Final Properties of Polypropylene" Polymers 14, no. 18: 3910. https://doi.org/10.3390/polym14183910
APA StyleHernández-Fernández, J., Cano, H., & Aldas, M. (2022). Impact of Traces of Hydrogen Sulfide on the Efficiency of Ziegler–Natta Catalyst on the Final Properties of Polypropylene. Polymers, 14(18), 3910. https://doi.org/10.3390/polym14183910










