Development of a New Eco-Friendly Copolymer Based on Chitosan for Enhanced Removal of Pb and Cd from Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
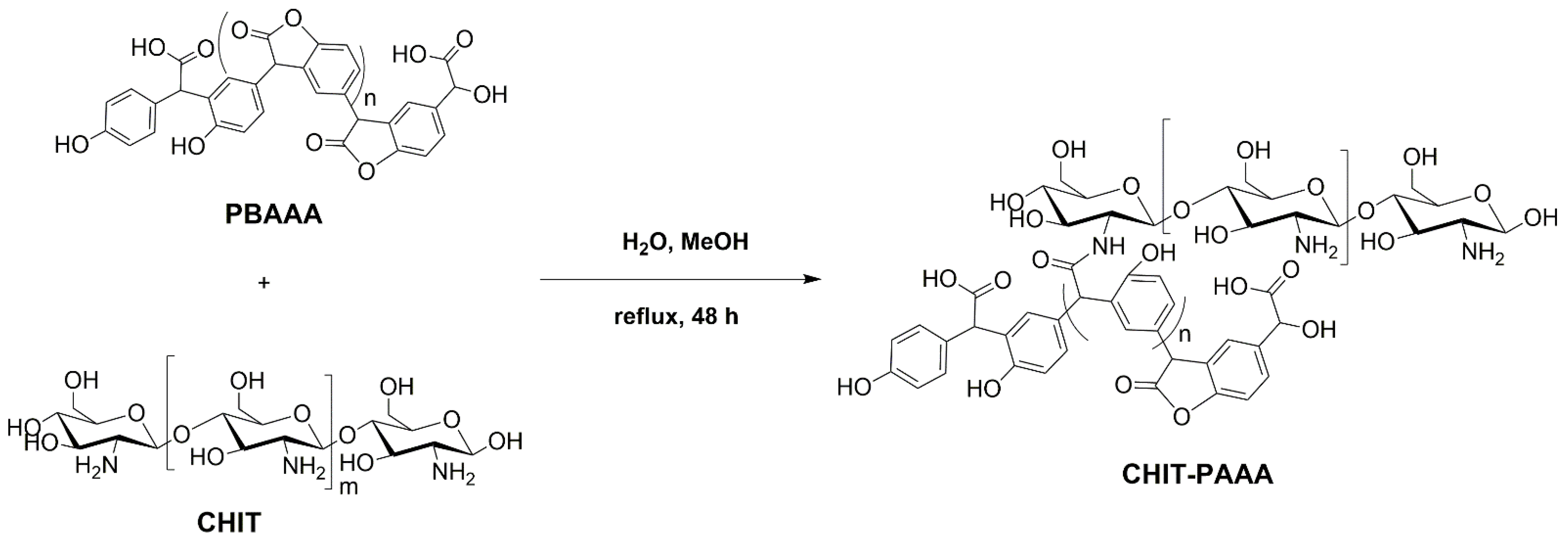
2.2. Synthesis of the Adsorbent Material
2.3. Characterization Methods
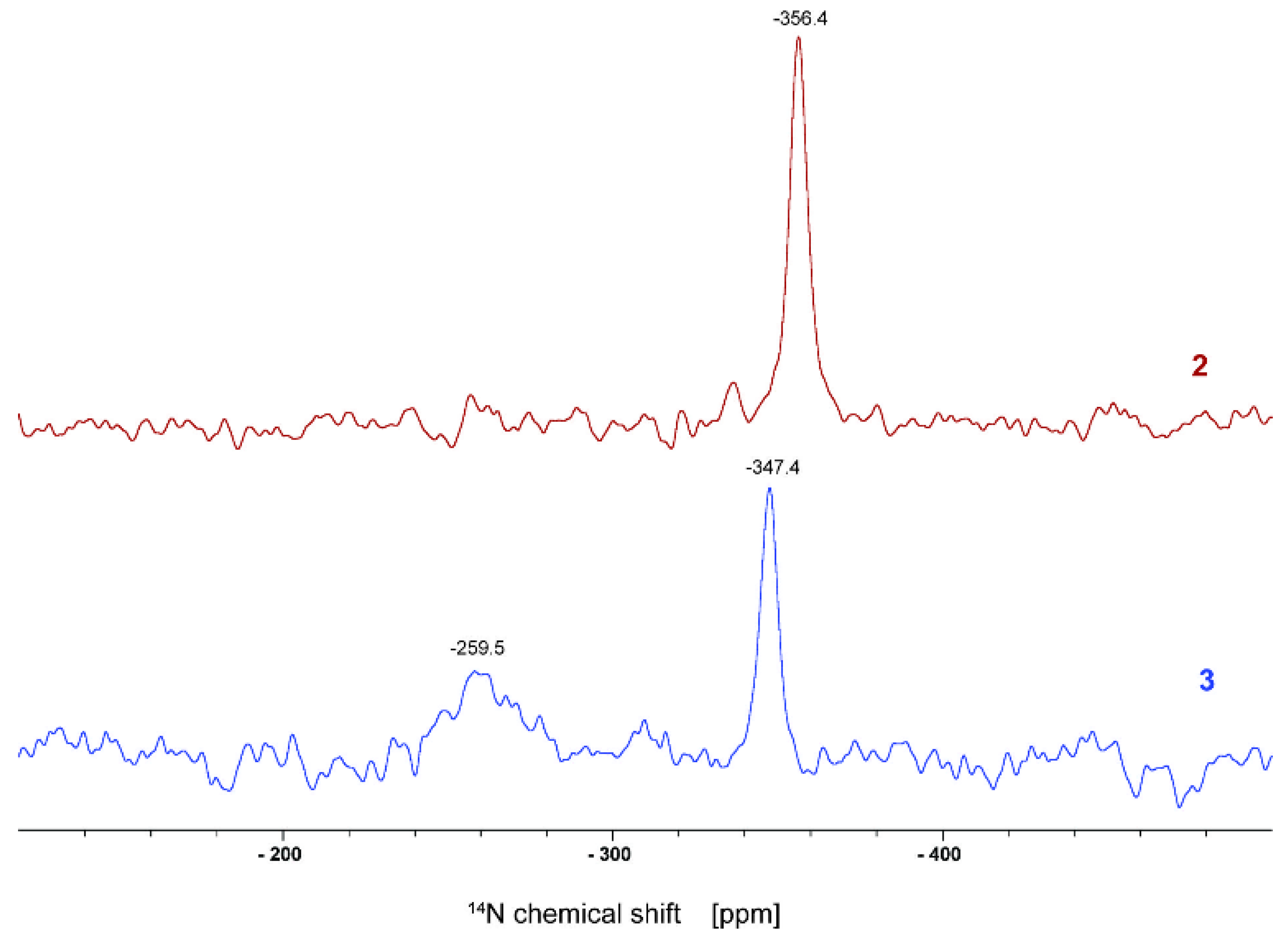
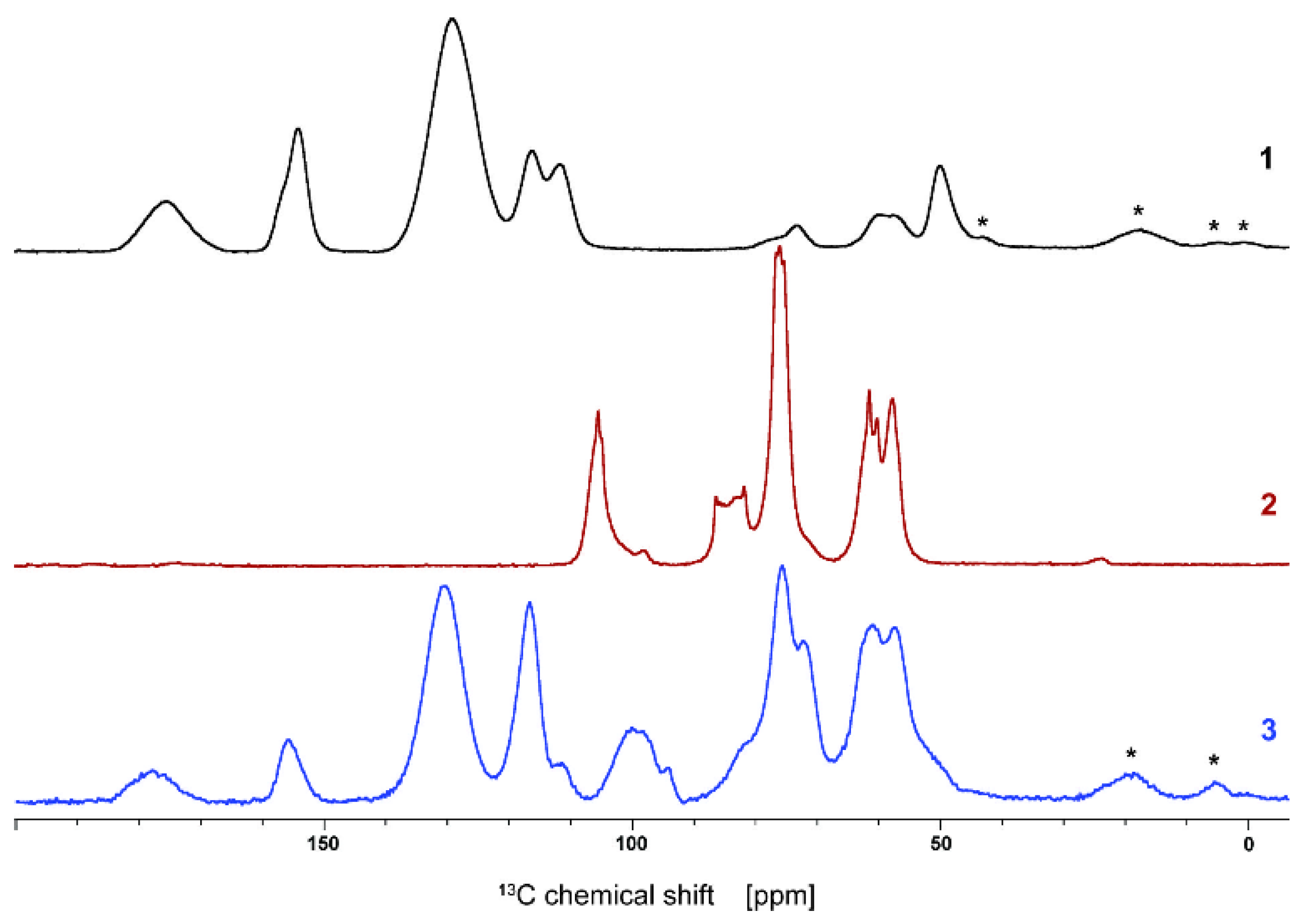
2.3.1. Solid-State Nuclear Magnetic Resonance
2.3.2. Fourier Transform Infrared Spectroscopy
2.3.3. Scanning Electron Microscopy
2.3.4. Thermo-Gravimetric Analysis
2.3.5. X-ray Photon Electron Spectroscopy
2.3.6. Brunauer-Emmett-Teller Surface Area Analysis
2.3.7. Flame Atomic Absorption Spectrometry
2.4. Batch Adsorption Experiments
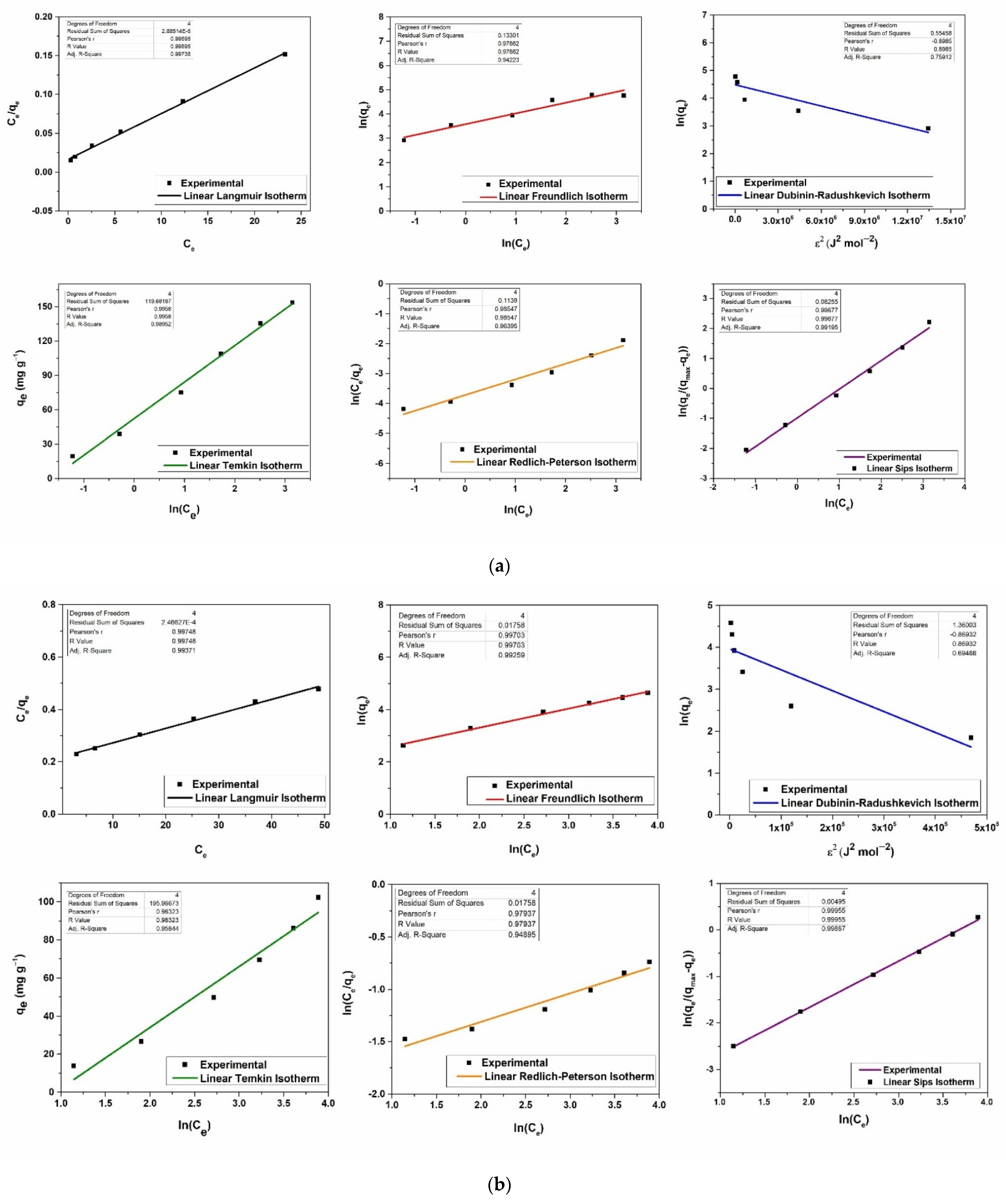
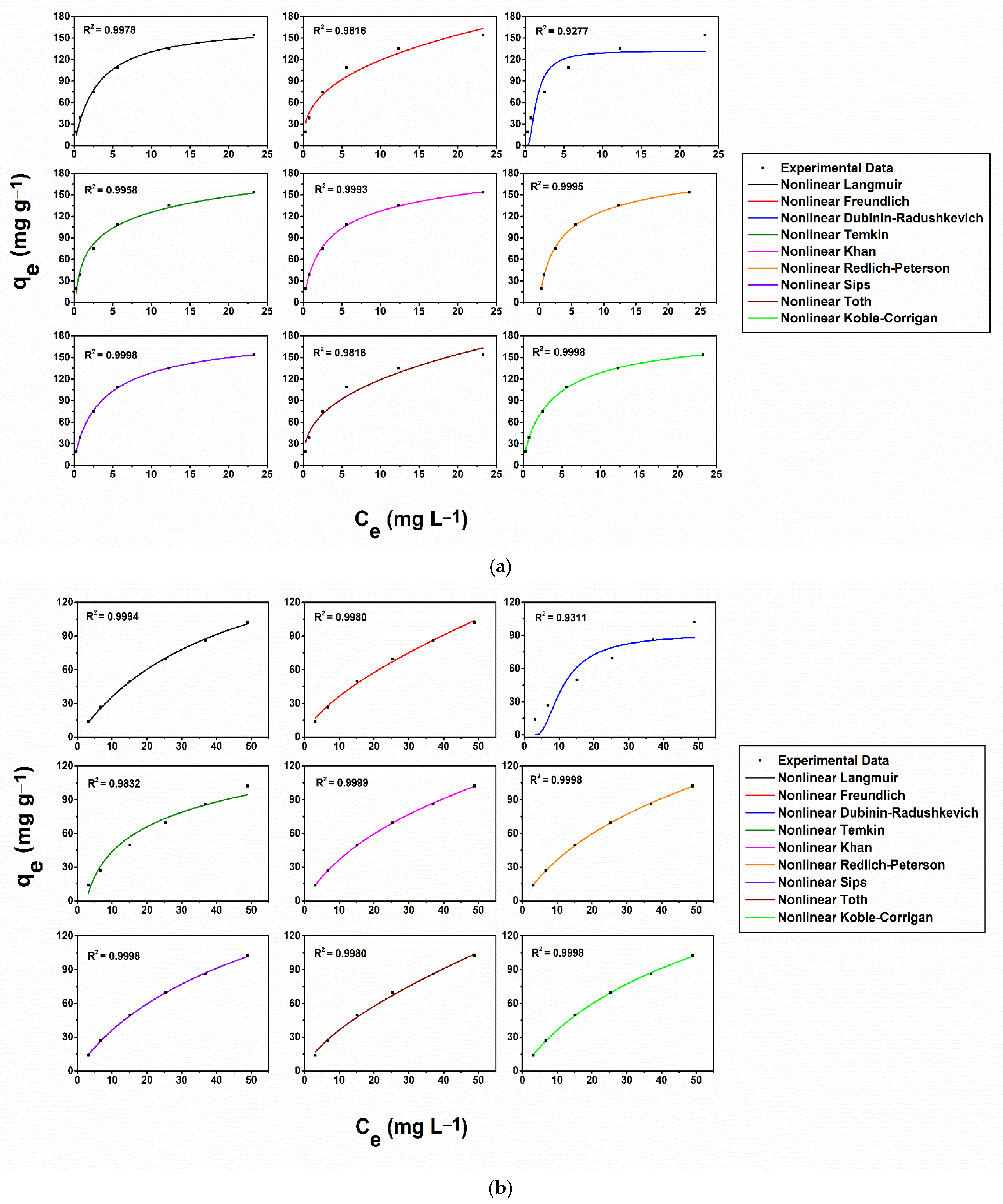
2.5. Equilibrium Adsorption Isotherms
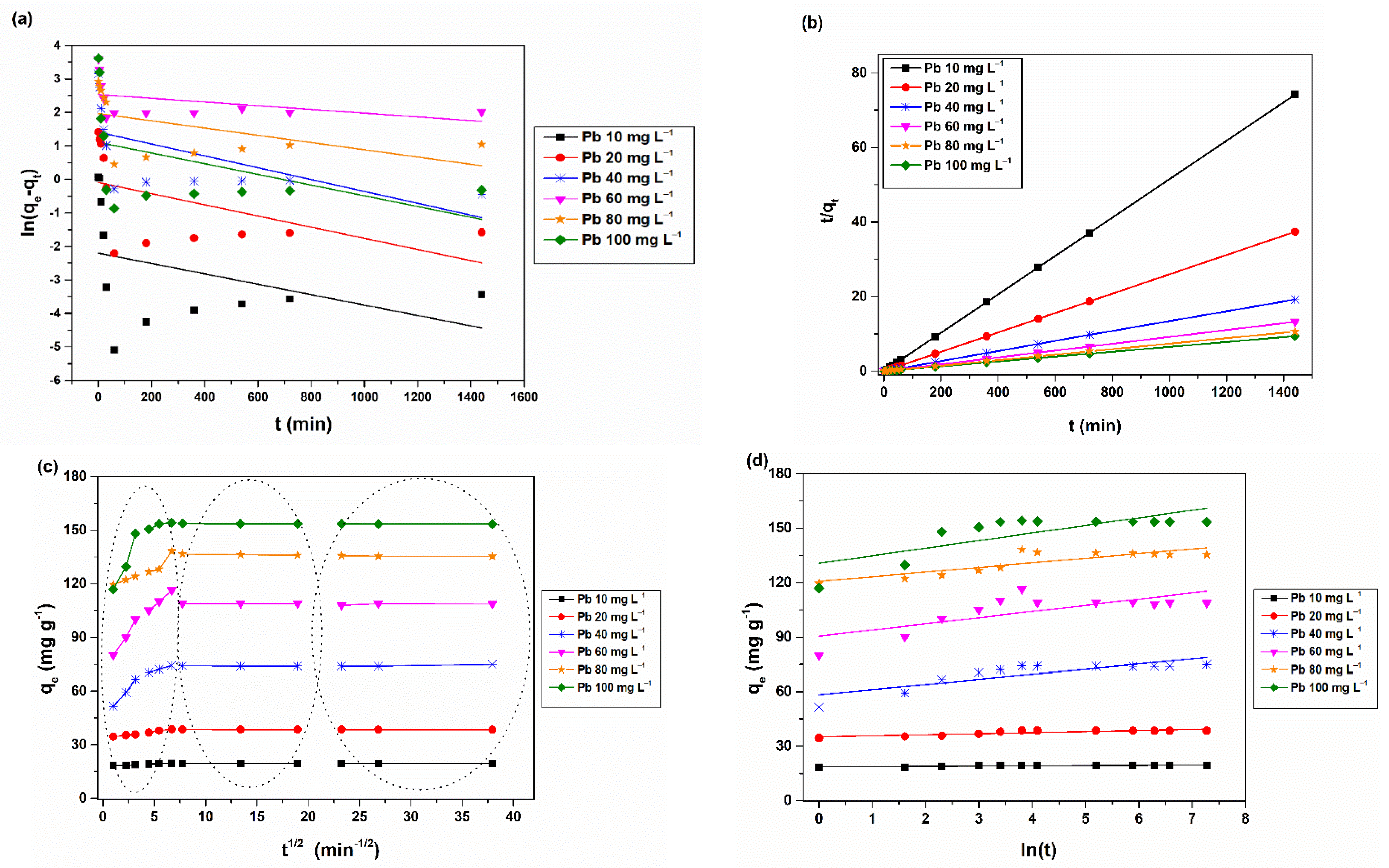
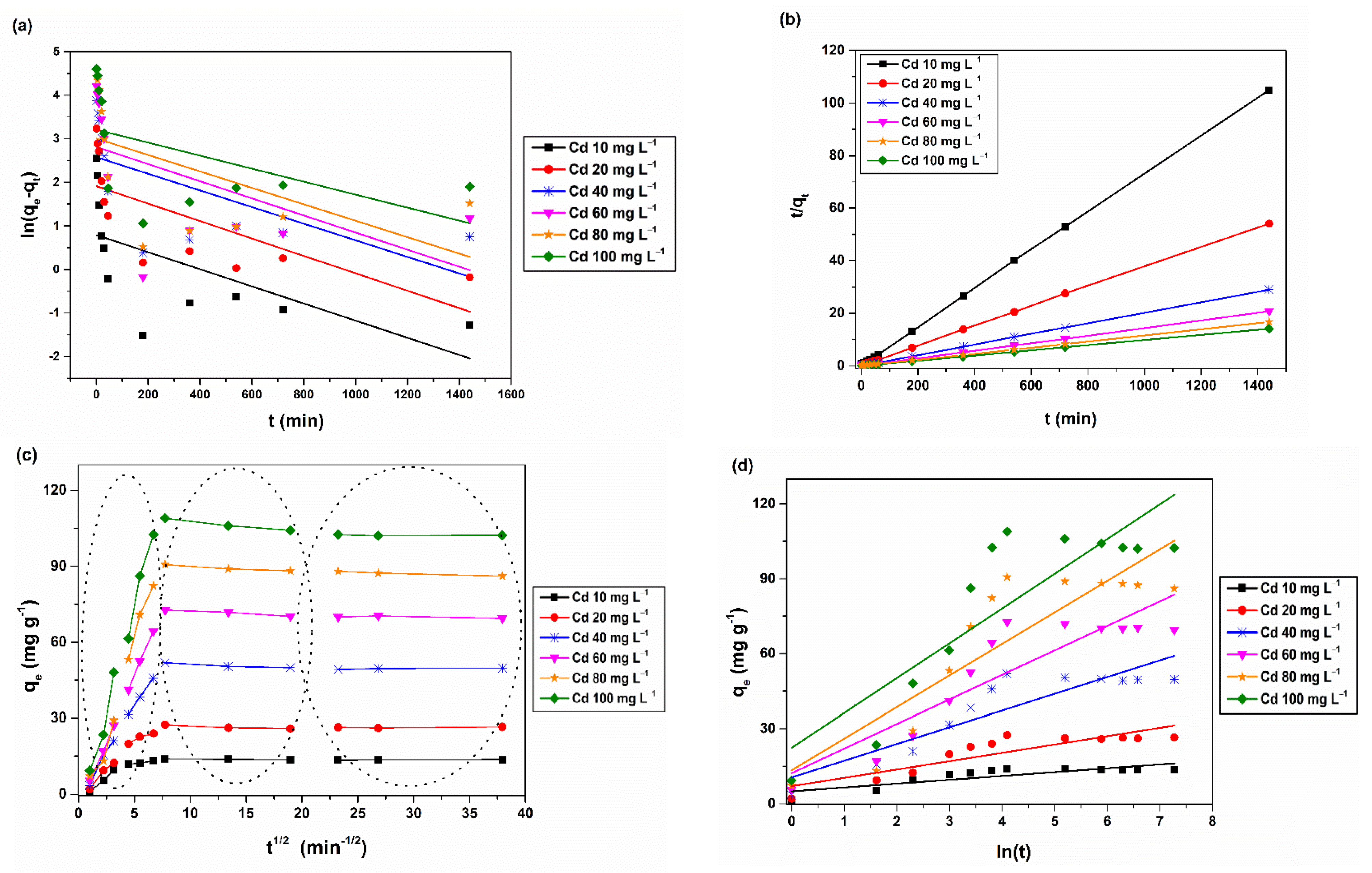
2.6. Kinetic Studies
2.7. Statistics
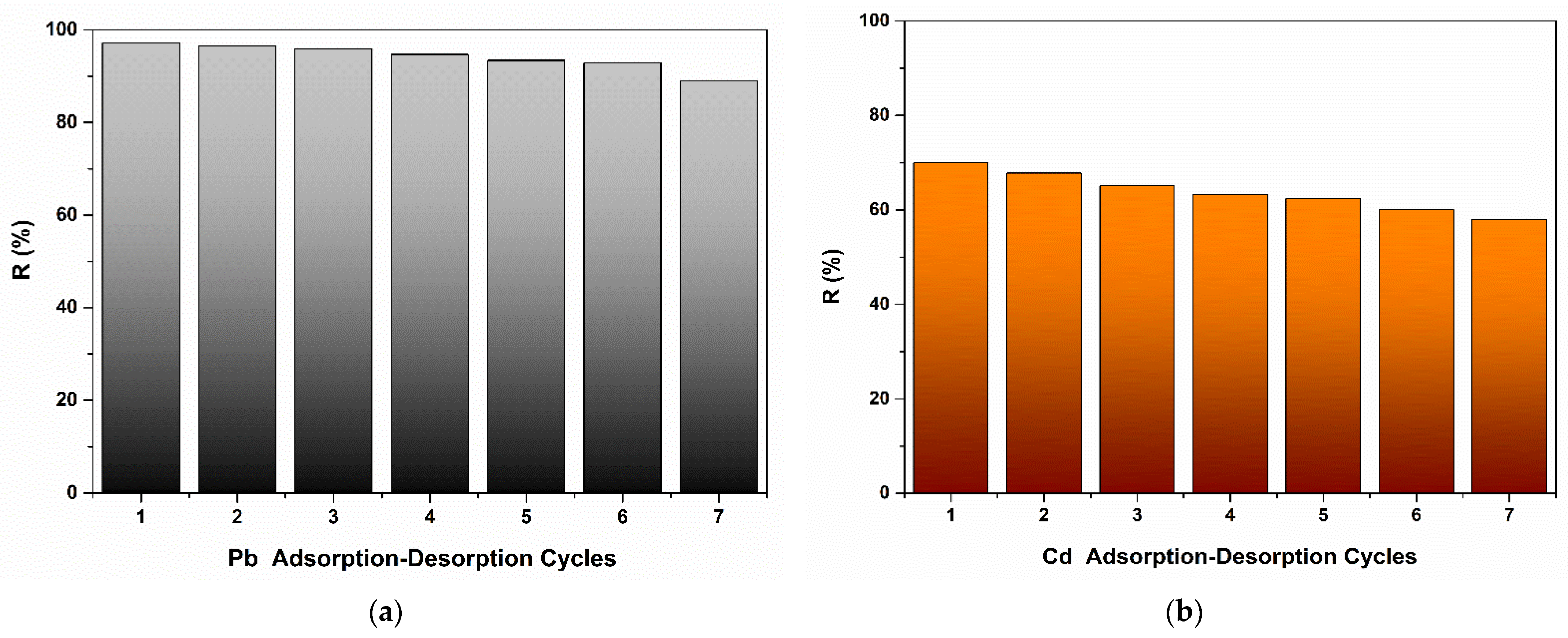
2.8. Recyclability Studies
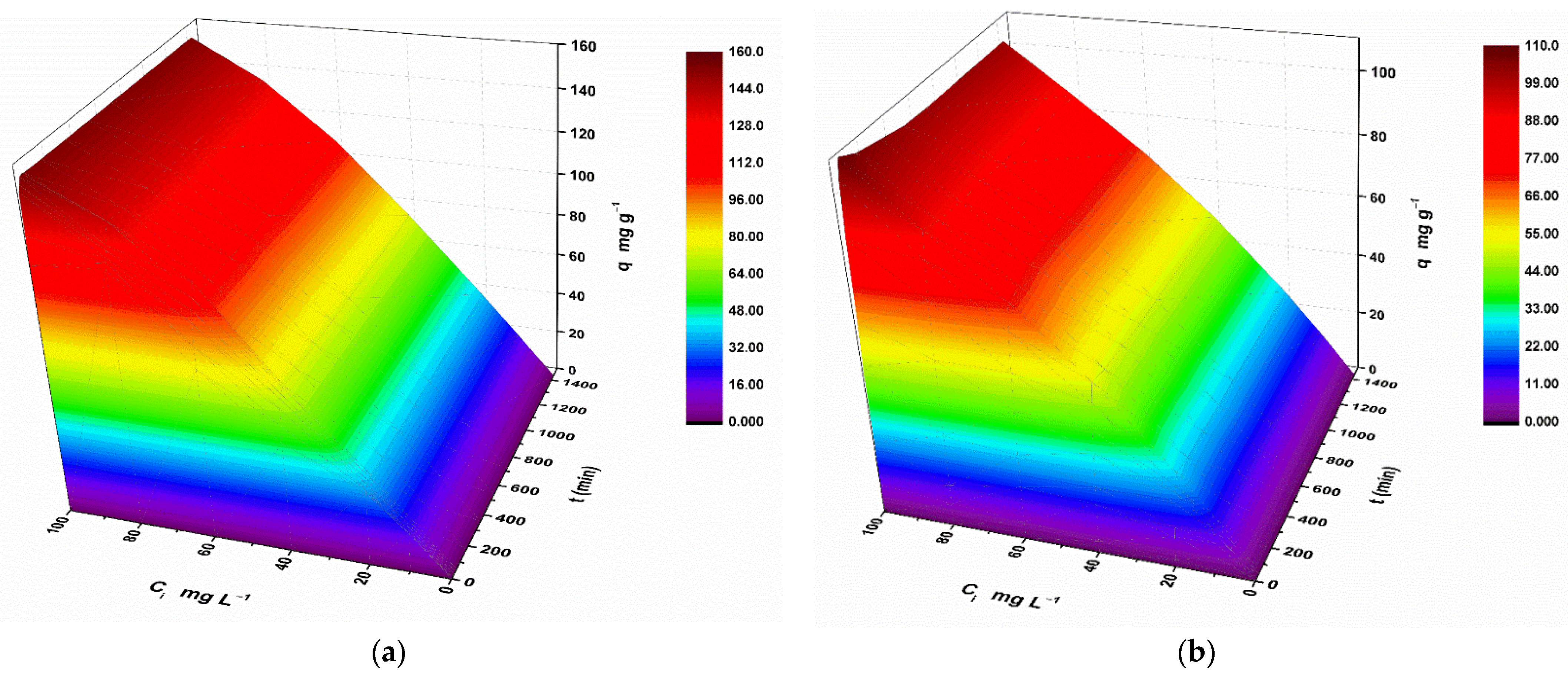
2.9. 3D Adsorption Rate Models
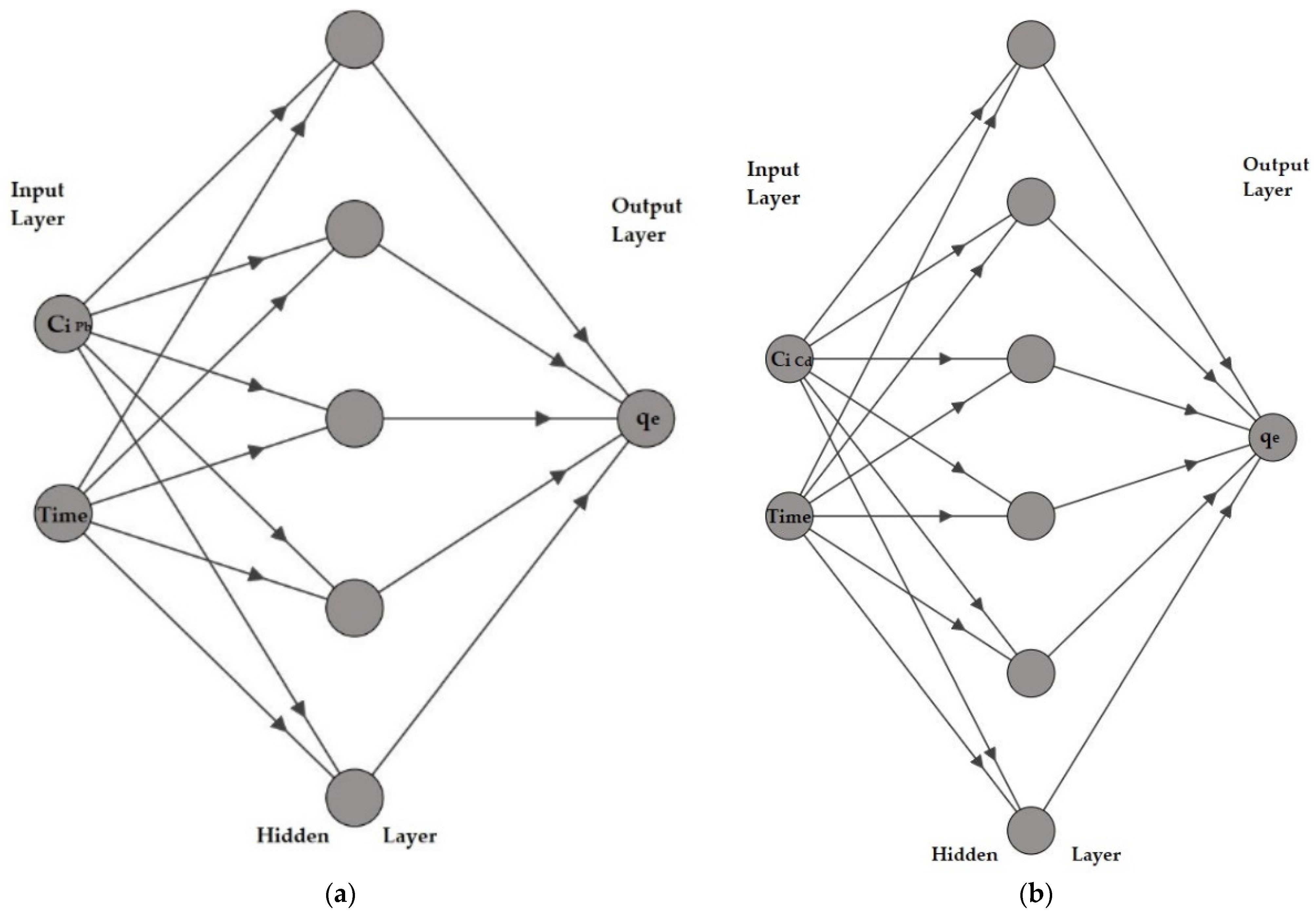
2.10. Artificial Neural Networks Models
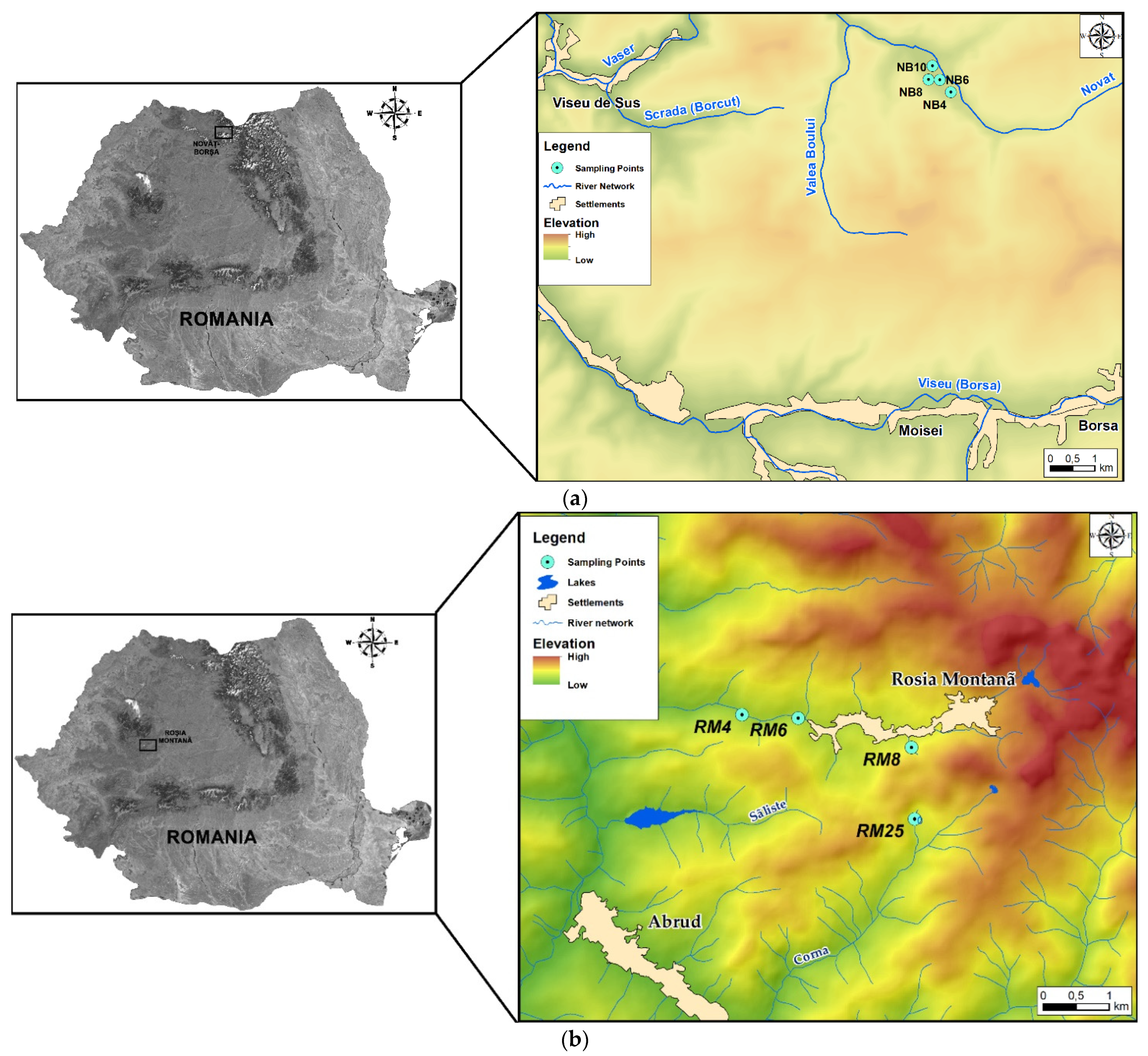
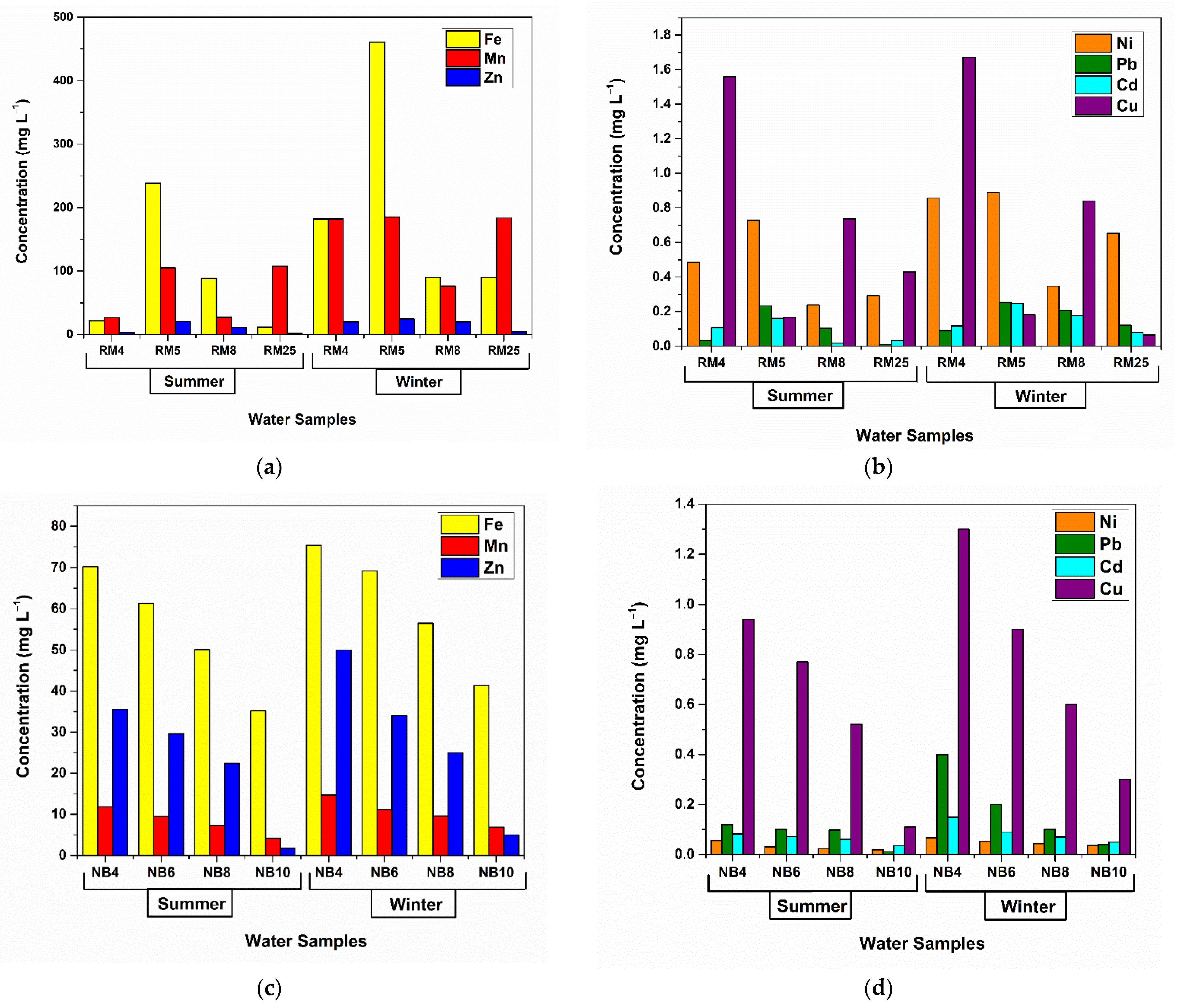
2.11. Adsorption Assays on Metal-Polluted Water Samples
3. Results and Discussion
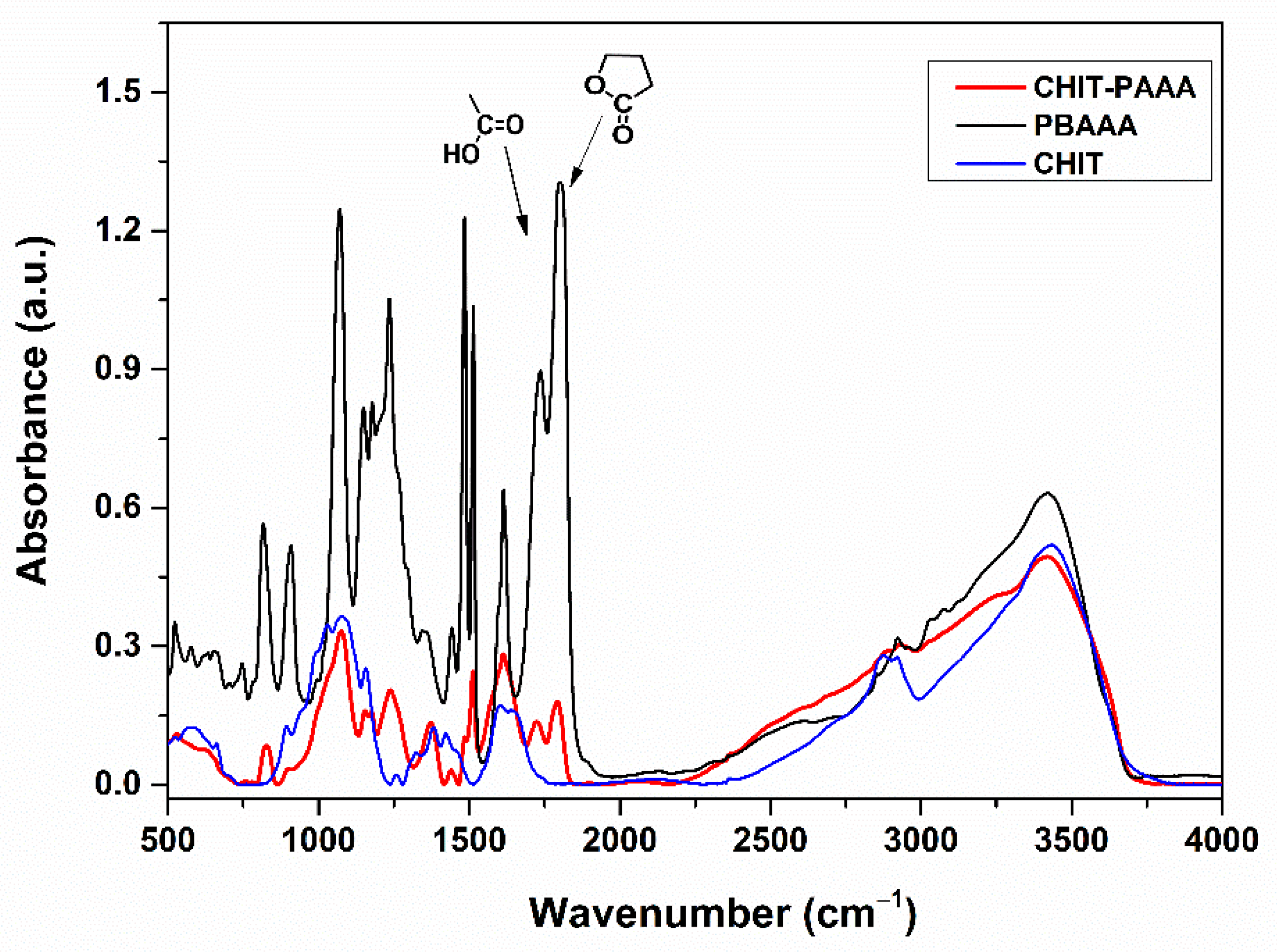
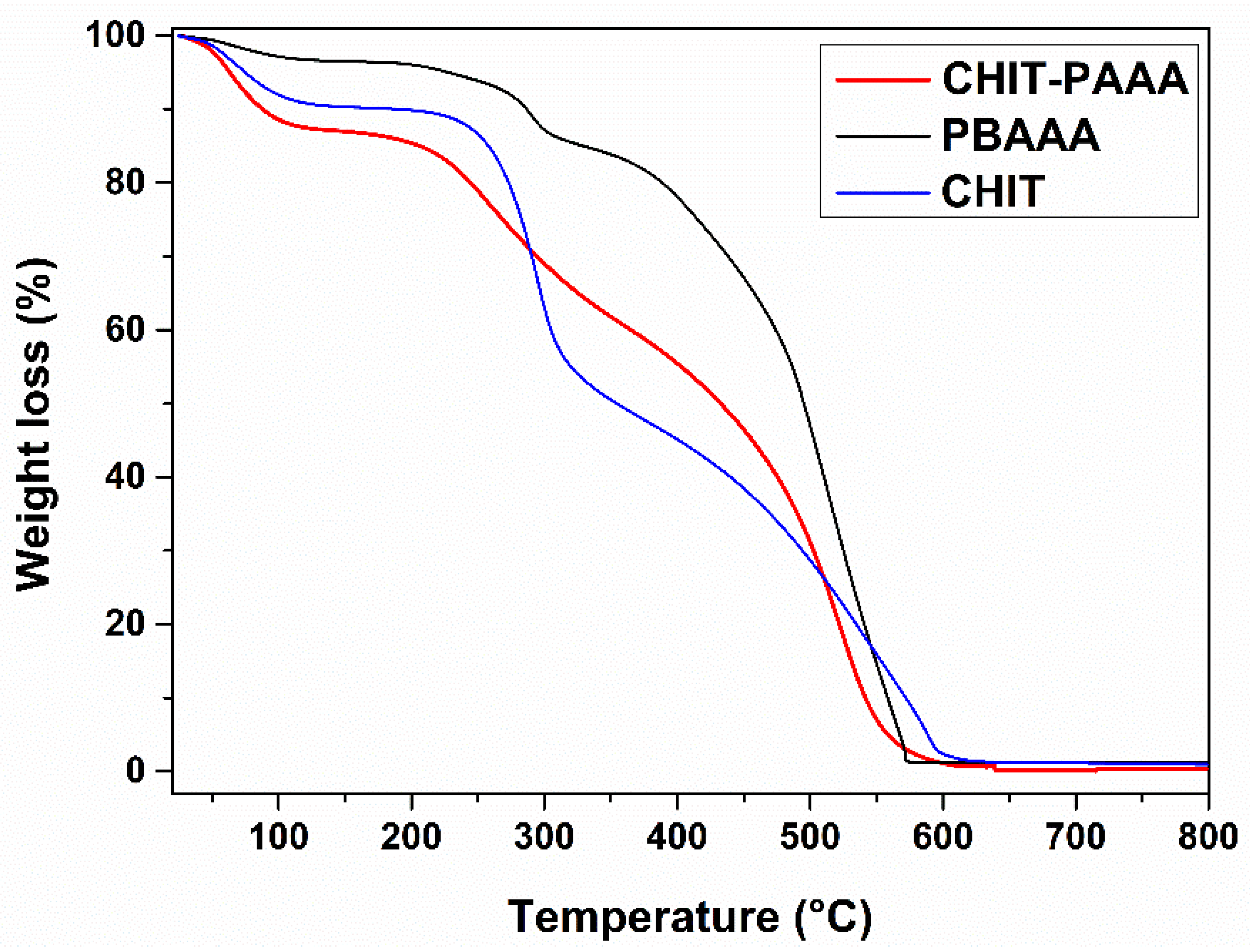
3.1. Synthesis and Characterization of the Adsorbent Material
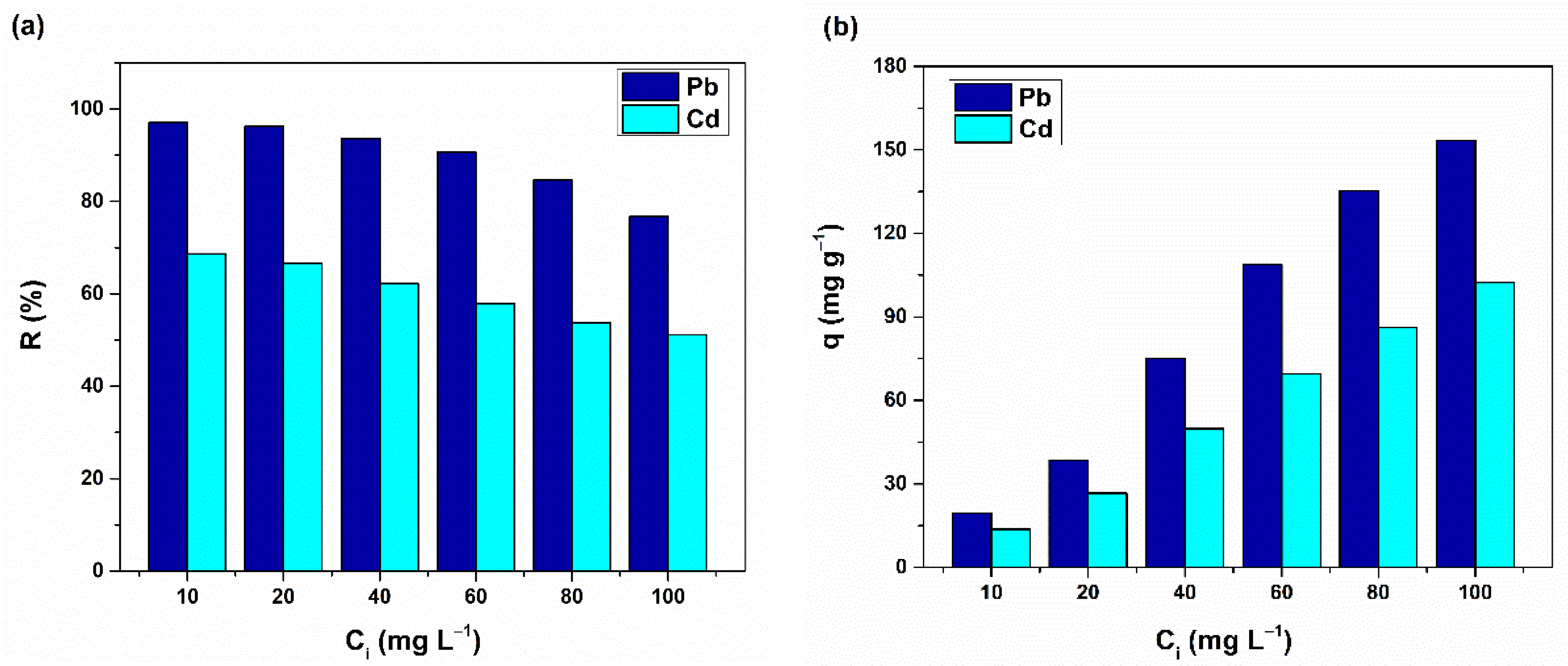
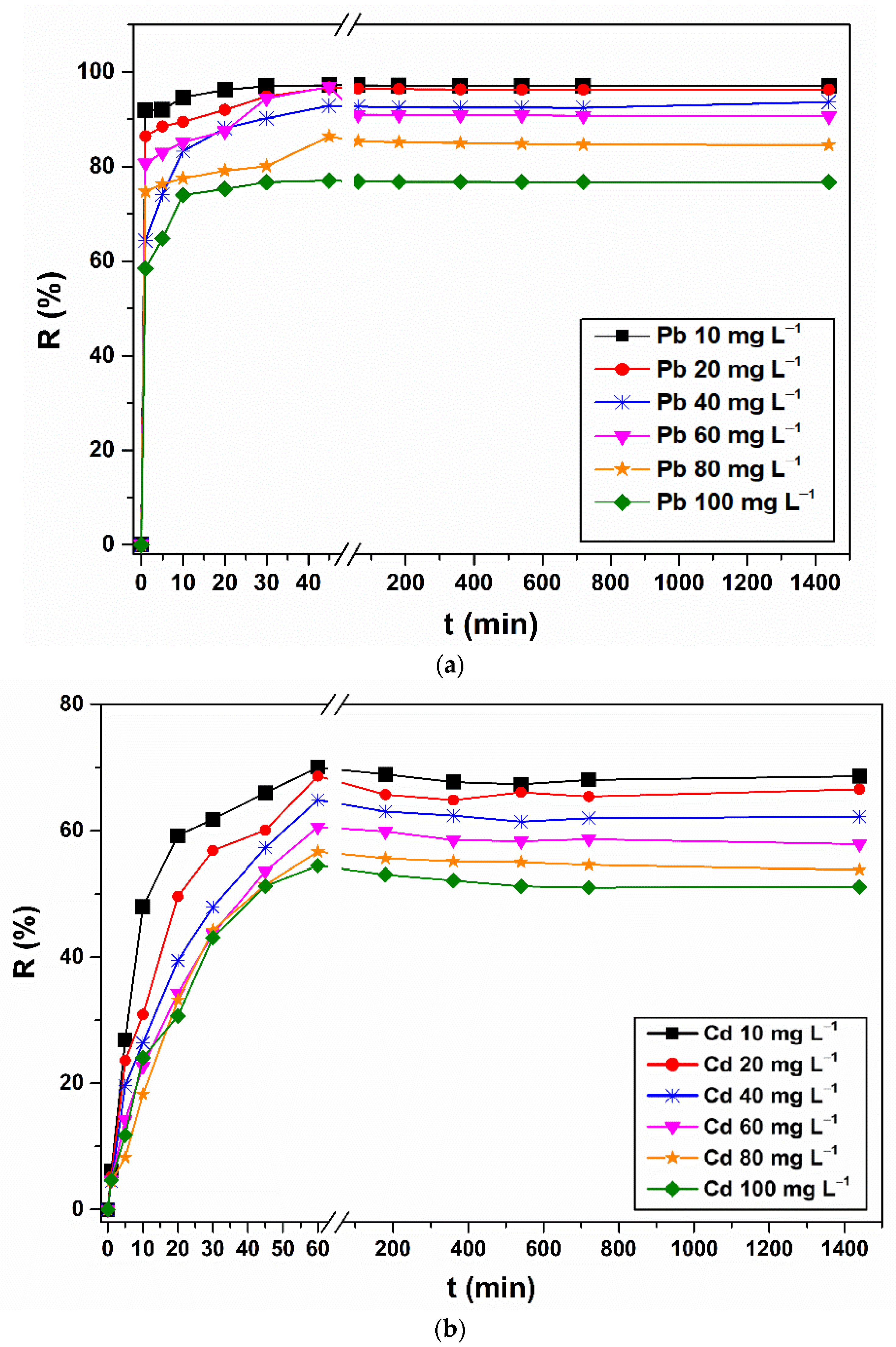
3.2. Metal Removal Assays on Stock Solutions
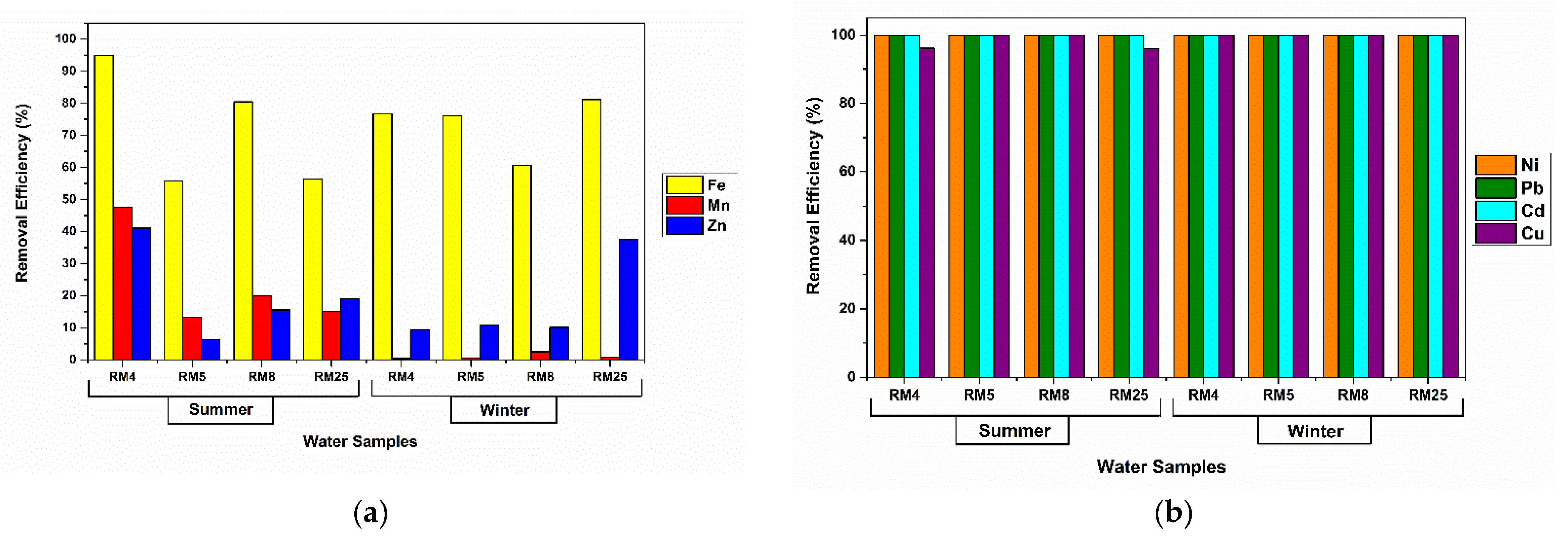
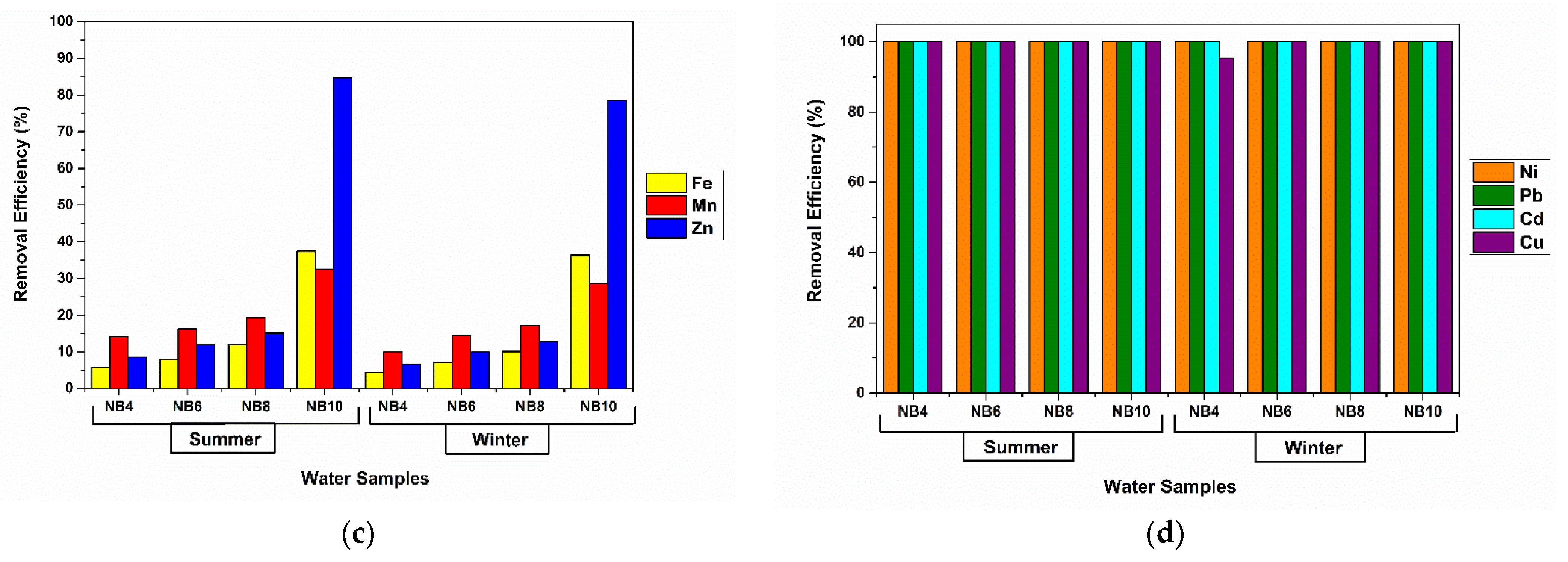
3.3. Assays on Metal-Polluted Water Samples
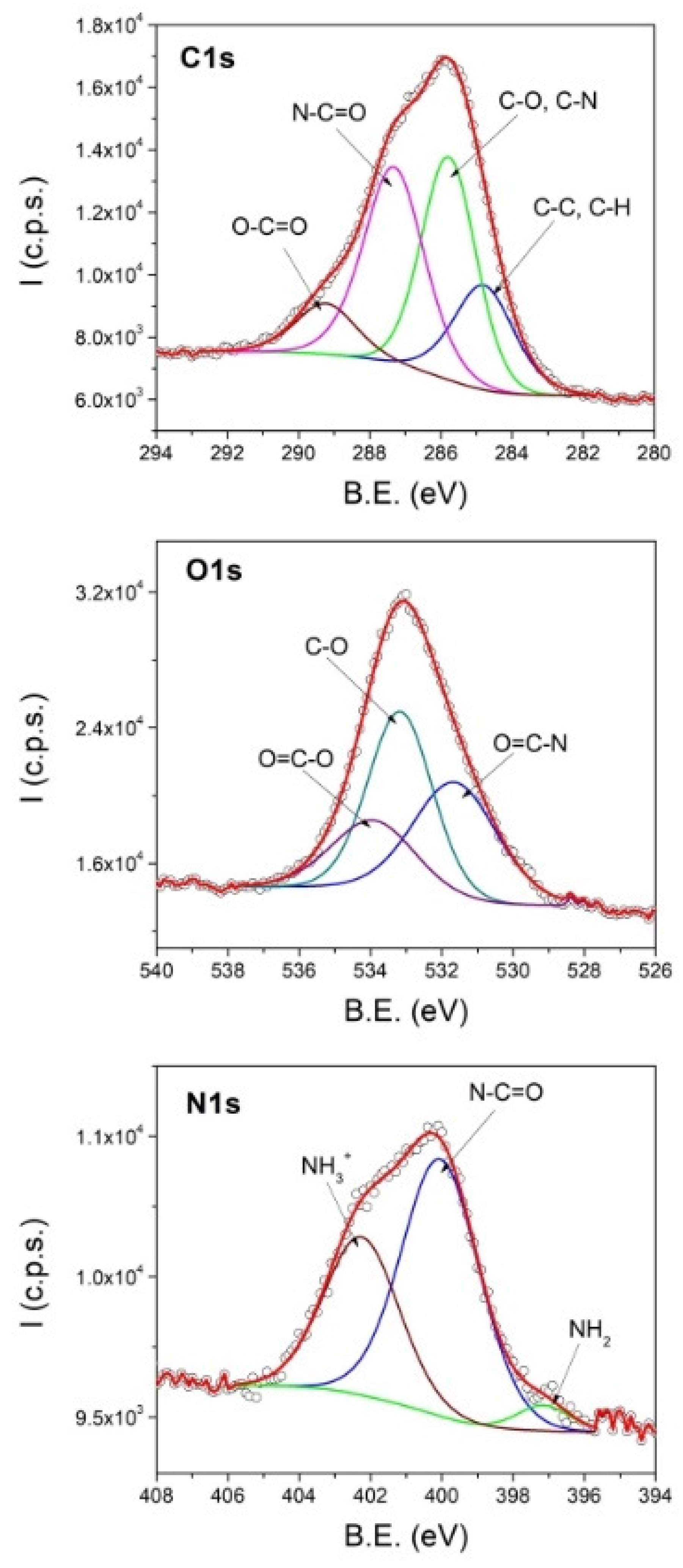
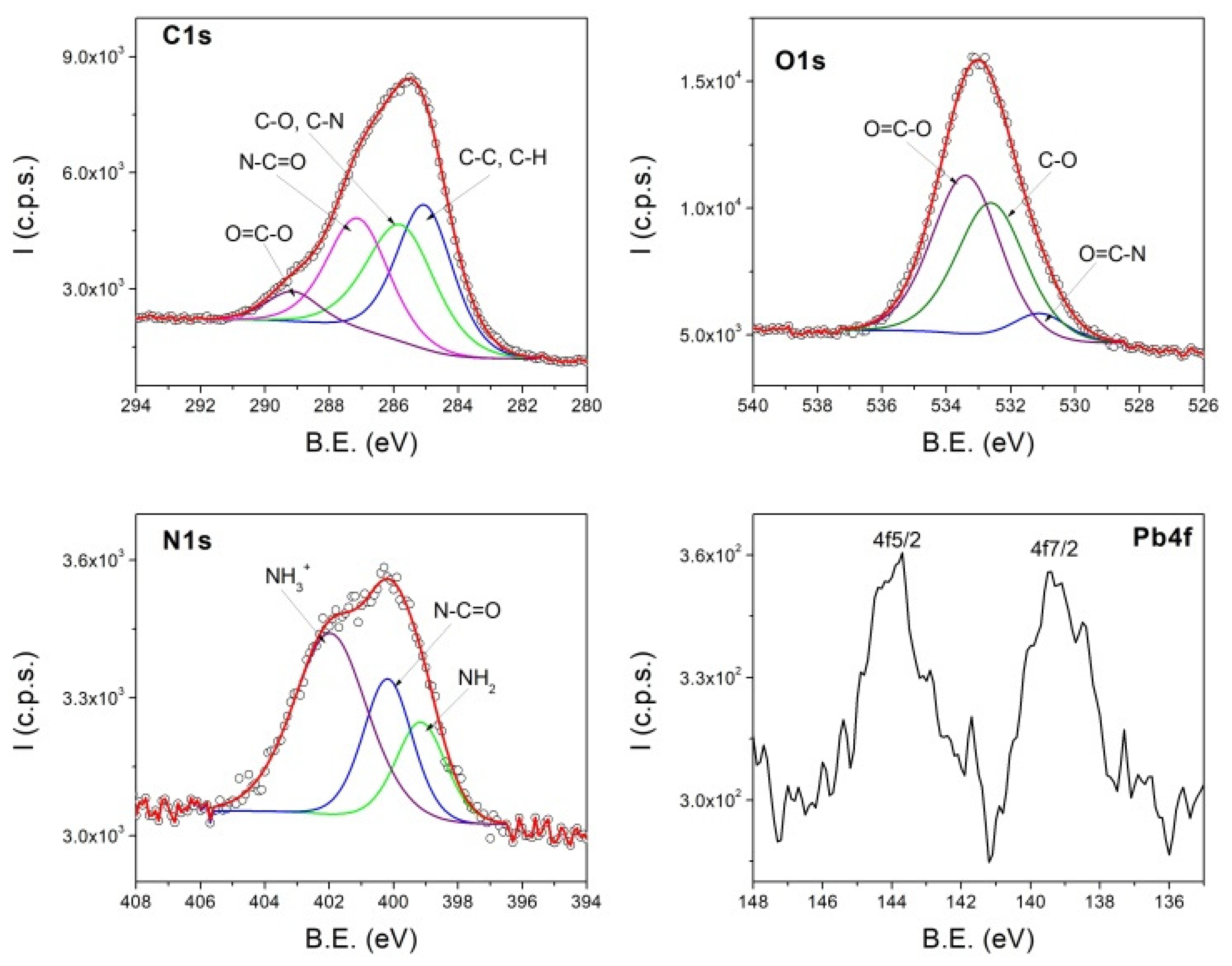
3.4. XPS Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schwarzenbach, R.P.; Escher, B.I.; Fenner, K.; Hofstetter, T.B.; Johnson, C.A.; Von Gunten, U.; Wehrli, B. The Challenge of Micropollutants in Aquatic Systems. Science 2006, 313, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Delpla, I.; Jung, A.V.; Baures, E.; Clement, M.; Thomas, O. Impacts of Climate Change on Surface Water Quality in Relation to Drinking Water Production. Environ. Int. 2009, 35, 1225–1233. [Google Scholar] [CrossRef]
- Kallenborn, R. Persistent Organic Pollutants (POPs) as Environmental Risk Factors in Remote High-Altitude Ecosystems. Ecotoxicol. Environ. Saf. 2006, 63, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.-I.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Ne Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater Biodiversity: Importance, Threats, Status and Conservation Challenges. Cambridge Philos. Soc. 2006, 81, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Schindler, D.E.; Hilborn, R. Prediction, Precaution, and Policy under Global Change. Emphasize Robustness, Monitoring, and Flexibility. Science 2015, 347, 953–954. [Google Scholar] [CrossRef]
- Malmqvist, B.; Rundle, S. Threats to the Running Water Ecosystems of the World. Environ. Conserv. 2002, 29, 134–153. [Google Scholar] [CrossRef]
- Narayan, S.S.; Dipak, P. Heavy Metal Tolerance and Accumulation by Bacterial Strains Isolated from Waste Water. J. Chem. Biol. Phys. Sci. 2014, 4, 812–817. [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Lead; U.S. Department of Health and Human Services: Washington, DC, USA, 2007.
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Cadmium; U.S. Department of Health and Human Services: Washington, DC, USA, 2012.
- Wan Ngah, W.S.; Teong, L.C.; Hanafiah, M.A.K.M. Adsorption of Dyes and Heavy Metal Ions by Chitosan Composites: A Review. Carbohydr. Polym. 2011, 83, 1446–1456. [Google Scholar] [CrossRef]
- Reddy, D.H.K.; Lee, S.M. Application of Magnetic Chitosan Composites for the Removal of Toxic Metal and Dyes from Aqueous Solutions. Adv. Colloid Interface Sci. 2013, 201–202, 68–93. [Google Scholar] [CrossRef] [PubMed]
- Varma, A.J.; Deshpande, S.V.; Kennedy, J.F. Metal Complexation by Chitosan and Its Derivatives: A Review. Carbohydr. Polym. 2004, 55, 77–93. [Google Scholar] [CrossRef]
- Guibal, E. Interactions of Metal Ions with Chitosan-Based Sorbents: A Review. Sep. Purif. Technol. 2004, 38, 43–74. [Google Scholar] [CrossRef]
- Fang, C.; Xiong, Z.; Qin, H.; Huang, G.; Liu, J.; Ye, M.; Feng, S.; Zou, H. One-Pot Synthesis of Magnetic Colloidal Nanocrystal Clusters Coated with Chitosan for Selective Enrichment of Glycopeptides. Anal. Chim. Acta 2014, 841, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Bai, R. Highly Enhanced Adsorption of Lead Ions on Chitosan Granules Functionalized with Poly(Acrylic Acid). Ind. Eng. Chem. Res. 2006, 45, 7897–7904. [Google Scholar] [CrossRef]
- Charpentier, T.V.J.; Neville, A.; Lanigan, J.L.; Barker, R.; Smith, M.J.; Richardson, T. Preparation of Magnetic Carboxymethylchitosan Nanoparticles for Adsorption of Heavy Metal Ions. ACS Omega 2016, 1, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Mahaweero, T. Extraction of Heavy Metals from Aqueous Solutions Using Chitosan/Montmorillonite Hybrid Hydrogels. Master’s Thesis, Case Western Reserve University, Cleveland, OH, USA, 2013. [Google Scholar]
- Zhang, L.; Zeng, Y.; Cheng, Z. Removal of Heavy Metal Ions Using Chitosan and Modified Chitosan: A Review. J. Mol. Liq. 2016, 214, 175–191. [Google Scholar] [CrossRef]
- Gokila, S.; Gomathi, T.; Sudha, P.N.; Anil, S. Removal of the Heavy Metal Ion Chromiuim(VI) Using Chitosan and Alginate Nanocomposites. Int. J. Biol. Macromol. 2017, 104, 1459–1468. [Google Scholar] [CrossRef] [PubMed]
- Medeiros Borsagli, F.G.L.; Mansur, A.A.P.; Chagas, P.; Oliveira, L.C.A.; Mansur, H.S. O-Carboxymethyl Functionalization of Chitosan: Complexation and Adsorption of Cd (II) and Cr (VI) as Heavy Metal Pollutant Ions. React. Funct. Polym. 2015, 97, 37–47. [Google Scholar] [CrossRef]
- Sun, X.; Li, Q.; Yang, L.; Liu, H. Chemically Modified Magnetic Chitosan Microspheres for Cr(VI) Removal from Acidic Aqueous Solution. Particuology 2016, 26, 79–86. [Google Scholar] [CrossRef]
- Kulkarni, P.S.; Deshmukh, P.G.; Jakhade, A.P.; Kulkarni, S.D.; Chikate, R.C. 1,5 Diphenyl Carbazide Immobilized Cross-Linked Chitosan Films: An Integrated Approach towards Enhanced Removal of Cr(VI). J. Mol. Liq. 2017, 247, 254–261. [Google Scholar] [CrossRef]
- Gopal Reddi, M.R.; Gomathi, T.; Saranya, M.; Sudha, P.N. Adsorption and Kinetic Studies on the Removal of Chromium and Copper onto Chitosan-g-Maliec Anhydride-g-Ethylene Dimethacrylate. Int. J. Biol. Macromol. 2017, 104, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Anitha, T.; Senthil Kumar, P.; Sathish Kumar, K.; Ramkumar, B.; Ramalingam, S. Adsorptive Removal of Pb(II) Ions from Polluted Water by Newly Synthesized Chitosan–Polyacrylonitrile Blend: Equilibrium, Kinetic, Mechanism and Thermodynamic Approach. Process Saf. Environ. Prot. 2015, 98, 187–197. [Google Scholar] [CrossRef]
- Sobahi, T.R.A.; Abdelaal, M.Y.; Makki, M.S.I. Chemical Modification of Chitosan for Metal Ion Removal. Arab. J. Chem. 2014, 7, 741–746. [Google Scholar] [CrossRef]
- Dubey, R.; Bajpai, J.; Bajpai, A.K. Chitosan-Alginate Nanoparticles (CANPs) as Potential Nanosorbent for Removal of Hg (II) Ions. Environ. Nanotechnol. Monit. Manag. 2016, 6, 32–44. [Google Scholar] [CrossRef]
- Gupta, V.K.; Gupta, D.; Agarwal, S.; Kothiyal, N.C.; Asif, M.; Sood, S.; Pathania, D. Fabrication of Chitosan-g-Poly(Acrylamide)/Cu Nanocomposite for the Removal of Pb(II) from Aqueous Solutions. J. Mol. Liq. 2016, 224, 1319–1325. [Google Scholar] [CrossRef]
- Miretzky, P.; Cirelli, A.F. Hg(II) Removal from Water by Chitosan and Chitosan Derivatives: A Review. J. Hazard. Mater. 2009, 167, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Mousa, N.E.; Simonescu, C.M.; Pătescu, R.E.; Onose, C.; Tardei, C.; Culiţă, D.C.; Oprea, O.; Patroi, D.; Lavric, V. Pb2 + Removal from Aqueous Synthetic Solutions by Calcium Alginate and Chitosan Coated Calcium Alginate. React. Funct. Polym. 2016, 109, 137–150. [Google Scholar] [CrossRef]
- Ji, G.; Bao, W.; Gao, G.; An, B.; Zou, H.; Gan, S. Removal of Cu (II) from Aqueous Solution Using a Novel Crosslinked Alumina-Chitosan Hybrid Adsorbent. Chin. J. Chem. Eng. 2012, 20, 641–648. [Google Scholar] [CrossRef]
- Lalhmunsiama; Lalchhingpuii;; Nautiyal, B.P.; Tiwari, D.; Choi, S.I.; Kong, S.H.; Lee, S.M. Silane Grafted Chitosan for the Efficient Remediation of Aquatic Environment Contaminated with Arsenic(V). J. Colloid Interface Sci. 2016, 467, 203–212. [Google Scholar] [CrossRef]
- Pal, P.; Pal, A. Surfactant-Modified Chitosan Beads for Cadmium Ion Adsorption. Int. J. Biol. Macromol. 2017, 104, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Kyzas, G.Z.; Kostoglou, M. Swelling–Adsorption Interactions during Mercury and Nickel Ions Removal by Chitosan Derivatives. Sep. Purif. Technol. 2015, 149, 92–102. [Google Scholar] [CrossRef]
- Hu, C.; Zhu, P.; Cai, M.; Hu, H.; Fu, Q. Comparative Adsorption of Pb(II), Cu(II) and Cd(II) on Chitosan Saturated Montmorillonite: Kinetic, Thermodynamic and Equilibrium Studies. Appl. Clay Sci. 2017, 143, 320–326. [Google Scholar] [CrossRef]
- Shankar, P.; Gomathi, T.; Vijayalakshmi, K.; Sudha, P.N. Comparative Studies on the Removal of Heavy Metals Ions onto Cross Linked Chitosan-g-Acrylonitrile Copolymer. Int. J. Biol. Macromol. 2014, 67, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Sutirman, Z.A.; Sanagi, M.M.; Abd Karim, K.J.; Wan Ibrahim, W.A. Preparation of Methacrylamide-Functionalized Crosslinked Chitosan by Free Radical Polymerization for the Removal of Lead Ions. Carbohydr. Polym. 2016, 151, 1091–1099. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, B.; Zhang, L.; Huang, R. Adsorptive Removal of Cr(VI) from Aqueous Solutions by Cross-Linked Chitosan/Bentonite Composite. Korean J. Chem. Eng. 2015, 32, 1314–1322. [Google Scholar] [CrossRef]
- Seyedmohammadi, J.; Motavassel, M.; Maddahi, M.H.; Nikmanesh, S. Application of Nanochitosan and Chitosan Particles for Adsorption of Zn(II) Ions Pollutant from Aqueous Solution to Protect Environment. Model. Earth Syst. Environ. 2016, 2, 165. [Google Scholar] [CrossRef]
- Ganea, I.-V.; Nan, A.; Neamt, I.; Baciu, C.; Serrano, A.R. Neoteric Material Based on Renewable Resources for Metal-Contaminated Waters. Environ. Sci. Proc. 2021, 9, 3. [Google Scholar] [CrossRef]
- Nan, A.; Bunge, A.; Cîrcu, M.; Petran, A.; Hǎdade, N.D.; Filip, X. Poly(Benzofuran-Co-Arylacetic Acid)—A New Type of Highly Functionalized Polymers. Polym. Chem. 2017, 8, 3504–3514. [Google Scholar] [CrossRef]
- Ortega, A.; Sánchez, A.; Burillo, G. Binary Graft of Poly(N-Vinylcaprolactam) and Poly(Acrylic Acid) onto Chitosan Hydrogels Using Ionizing Radiation for the Retention and Controlled Release of Therapeutic Compounds. Polymers 2021, 13, 2641. [Google Scholar] [CrossRef]
- Langmuir, I. The Constitution and Fundamental Properties of Solids and Liquids. Part II.-Liquids. J. Franklin Inst. 1917, 184, 721. [Google Scholar] [CrossRef]
- Freundlich, H. Über Die Absorption in Lösungen. Z. Phys. Chem. Stöch. Verwand. 1907, 57, 385–470. [Google Scholar] [CrossRef]
- Dubinin, M.M.; Radushkevich, L.V. The Equation of the Characteristic Curve of Activated Charcoal. Proc. Acad. Sci. USSR Phys. Chem. Sect. 1947, 55, 331–337. [Google Scholar]
- Temkin, M.J.; Pyzhev, V. Kinetics of Ammonia Synthesis on Promoted Iron Catalysts. Acta Physicochim. URSS 1940, 12, 217–222. [Google Scholar]
- Khan, A.R.; Ataullah, R.; Al-Haddad, A. Equilibrium Adsorption Studies of Some Aromatic Pollutants from Dilute Aqueous Solutions on Activated Carbon at Different Temperatures. J. Colloid Interface Sci. 1994, 194, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Redlich, O.; Peterson, D.L. A Useful Adsorption Isotherm. J. Phys. Chem. 1959, 63, 1024. [Google Scholar] [CrossRef]
- Sips, R. Combined Form of Langmuir and Freundlich Equations. J. Phys. Chem. 1948, 16, 490–495. [Google Scholar] [CrossRef]
- Toth, J. State Equation of the Solid Gas Interface Layer. Acta Chim. 1971, 69, 311–317. [Google Scholar]
- Koble, R.A.; Corrigan, T.E. Adsorption Isotherms for Pure Hydrocarbons. Ind. Eng. Chem. 1952, 44, 383–387. [Google Scholar] [CrossRef]
- Hamzaoui, M.; Bestani, B.; Benderdouche, N. The Use of Linear and Nonlinear Methods for Adsorption Isotherm Optimization of Basic Green 4-Dye onto Sawdust-Based Activated Carbon. J. Mater. Environ. Sci. 2018, 9, 1110–1118. [Google Scholar]
- Kocadagistan, B.; Kocadagistan, E. The Effects of Sunflower Seed Shell Modifying Process on Textile Dye Adsorption: Kinetic, Thermodynamic and Equilibrium Study. Desalination Water Treat. 2014, 57, 3168–3178. [Google Scholar] [CrossRef]
- Lagergren, S.; Sven, K. Zur Theorie Der Sogennanten Adsorptiongeloster Stoffe. K. Sevenska Vetenskapsakademiens. Handl. 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; McKay, G. The Kinetics of Sorption of Divalent Metal Ions onto Sphagnum Moss Peat. Water Res. 2000, 34, 735–742. [Google Scholar] [CrossRef]
- Weber, W.J.; Morris, J.C. Kinetic of Adsorption on Carbon from Solution. Am. Soc. Civ. Eng. 1963, 89, 31–59. [Google Scholar] [CrossRef]
- Zeldowitsch, J. Über Den Mechanismus Der Katalytischen Oxidation Von CO a MnO2. URSS Acta Physiochim. 1934, 1, 364–449. [Google Scholar]
- Marczewski, A.W. Application of Mixed Order Rate Equations to Adsorption of Methylene Blue on Mesoporous Carbons. Appl. Surf. Sci. 2010, 256, 5145–5152. [Google Scholar] [CrossRef]
- Derylo-Marczewska, A.; Marczewski, A.W.; Winter, S.; Sternik, D. Studies of Adsorption Equilibria and Kinetics in the Systems: Aqueous Solution of Dyes–Mesoporous Carbons. Appl. Surf. Sci. 2010, 256, 5164–5170. [Google Scholar] [CrossRef]
- Tvrdík, J.; Křivý, I.; Mišík, L. Adaptive Population-Based Search: Application to Estimation of Nonlinear Regression Parameters. Comput. Stat. Data Anal. 2007, 52, 713–724. [Google Scholar] [CrossRef]
- Roman, T.; Asavei, R.L.; Karkalos, N.E.; Roman, C.; Virlan, C.; Cimpoesu, N.; Istrate, B.; Zaharia, M.; Markopoulos, A.P.; Kordatos, K.; et al. Synthesis and Adsorption Properties of Nanocrystalline Ferrites for Kinetic Modeling Development. Int. J. Appl. Ceram. Technol. 2019, 16, 693–705. [Google Scholar] [CrossRef]
- Ghaedi, M.; Ansari, A.; Nejad, P.A.; Ghaedi, A.; Vafaei, A.; Habibi, M.H. Artificial Neural Network and Bees Algorithm for Removal of Eosin B Using Cobalt Oxide Nanoparticle-Activated Carbon: Isotherm and Kinetics Study. Environ. Prog. Sustain. Energy 2015, 34, 155–168. [Google Scholar] [CrossRef]
- Ghaedi, M.; Hosaininia, R.; Ghaedi, A.M.; Vafaei, A.; Taghizadeh, F. Adaptive Neuro-Fuzzy Inference System Model for Adsorption of 1,3,4-Thiadiazole-2,5-Dithiol onto Gold Nanoparticales-Activated Carbon. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 131, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Ghaedi, M.; Ghaedi, A.M.; Negintaji, E.; Ansari, A.; Vafaei, A.; Rajabi, M. Random Forest Model for Removal of Bromophenol Blue Using Activated Carbon Obtained from Astragalus Bisulcatus Tree. J. Ind. Eng. Chem. 2014, 20, 1793–1803. [Google Scholar] [CrossRef]
- Elemen, S.; Akçakoca Kumbasar, E.P.; Yapar, S. Modeling the Adsorption of Textile Dye on Organoclay Using an Artificial Neural Network. Dye. Pigment. 2012, 95, 102–111. [Google Scholar] [CrossRef]
- Despagne, F.; Massart, D.L. Neural Networks in Multivariate Calibration. Analyst 1998, 123, 157R–178R. [Google Scholar] [CrossRef]
- Khan, T.; Mustafa, M.R.U.; Isa, M.H.; Manan, T.S.B.A.; Ho, Y.C.; Lim, J.W.; Yusof, N.Z. Artificial Neural Network (ANN) for Modelling Adsorption of Lead (Pb (II)) from Aqueous Solution. Water. Air. Soil Pollut. 2017, 228, 426. [Google Scholar] [CrossRef]
- Narayana, P.L.; Maurya, A.K.; Wang, X.S.; Harsha, M.R.; Srikanth, O.; Alnuaim, A.A.; Hatamleh, W.A.; Hatamleh, A.A.; Cho, K.K.; Paturi, U.M.R.; et al. Artificial Neural Networks Modeling for Lead Removal from Aqueous Solutions Using Iron Oxide Nanocomposites from Bio-Waste Mass. Environ. Res. 2021, 199, 111370. [Google Scholar] [CrossRef]
- Olanrewaju, R.F.; Mariam, R.; Ahmed, A.A. Modeling of ANN to Determine Optimum Adsorption Capacity for Removal of Pollutants in Wastewater. In Proceedings of the 2017 IEEE 4th International Conference on Smart Instrumentation, Measurement and Application (ICSIMA), Putrajaya, Malaysia, 28–30 November 2017; pp. 1–5. [Google Scholar] [CrossRef]
- McCulloch, W.S.; Pitts, W. A Logical Calculus of the Ideas Immanent in Nervous Activity. Bull. Math. Biophys. 1943, 5, 115–133. [Google Scholar] [CrossRef]
- Mohanraj, M.; Jayaraj, S.; Muraleedharan, C. Applications of Artificial Neural Networks for Thermal Analysis of Heat Exchangers—A Review. Int. J. Therm. Sci. 2015, 90, 150–172. [Google Scholar] [CrossRef]
- Sha, W.; Edwards, K.L. The Use of Artificial Neural Networks in Materials Science Based Research. Mater. Des. 2007, 28, 1747–1752. [Google Scholar] [CrossRef]
- Mjalli, F.S.; Al-Asheh, S.; Alfadala, H.E. Use of Artificial Neural Network Black-Box Modeling for the Prediction of Wastewater Treatment Plants Performance. J. Environ. Manag. 2007, 83, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Cavas, L.; Karabay, Z.; Alyuruk, H.; Doĝan, H.; Demir, G.K. Thomas and Artificial Neural Network Models for the Fixed-Bed Adsorption of Methylene Blue by a Beach Waste Posidonia oceanica (L.) Dead Leaves. Chem. Eng. J. 2011, 171, 557–562. [Google Scholar] [CrossRef]
- Chowdhury, S.; Saha, P. Das Artificial Neural Network (ANN) Modeling of Adsorption of Methylene Blue by NaOH-Modified Rice Husk in a Fixed-Bed Column System. Environ. Sci. Pollut. Res. Int. 2013, 20, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Ghaedi, A.M.; Vafaei, A. Applications of Artificial Neural Networks for Adsorption Removal of Dyes from Aqueous Solution: A Review. Adv. Colloid Interface Sci. 2017, 245, 20–39. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, S. Artificial Neural Network (ANN) Approach for Modeling Zn(II) Adsorption in Batch Process. Korean J. Chem. Eng. 2017, 34, 2423–2434. [Google Scholar] [CrossRef]
- Luu, T.T.; Dinh, V.P.; Nguyen, Q.H.; Tran, N.Q.; Nguyen, D.K.; Ho, T.H.; Nguyen, V.D.; Tran, D.X.; Kiet, H.A.T. Pb(II) Adsorption Mechanism and Capability from Aqueous Solution Using Red Mud Modified by Chitosan. Chemosphere 2021, 287, 132279. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.H.; Liu, S.C.; Chen, C.Y.; Chen, C.Y. Comparative Adsorption of Cu(II), Zn(II), and Pb(II) Ions in Aqueous Solution on the Crosslinked Chitosan with Epichlorohydrin. J. Hazard. Mater. 2008, 154, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Ngah, W.S.W.; Fatinathan, S. Pb(II) Biosorption Using Chitosan and Chitosan Derivatives Beads: Equilibrium, Ion Exchange and Mechanism Studies. J. Environ. Sci. 2010, 22, 338–346. [Google Scholar] [CrossRef]
- Tran, H.V.; Tran, L.D.; Nguyen, T.N. Preparation of Chitosan/Magnetite Composite Beads and Their Application for Removal of Pb(II) and Ni(II) from Aqueous Solution. Mater. Sci. Eng. C 2010, 30, 304–310. [Google Scholar] [CrossRef]
- Rasoulzadeh, H.; Dehghani, M.H.; Mohammadi, A.S.; Karri, R.R.; Nabizadeh, R.; Nazmara, S.; Kim, K.H.; Sahu, J.N. Parametric Modelling of Pb(II) Adsorption onto Chitosan-Coated Fe3O4 Particles through RSM and DE Hybrid Evolutionary Optimization Framework. J. Mol. Liq. 2020, 297, 111893. [Google Scholar] [CrossRef]
- ALSamman, M.T.; Sánchez, J. Recent Advances on Hydrogels Based on Chitosan and Alginate for the Adsorption of Dyes and Metal Ions from Water. Arab. J. Chem. 2021, 14, 103455. [Google Scholar] [CrossRef]
- Li, H.; Ji, H.; Cui, X.; Che, X.; Zhang, Q.; Zhong, J.; Jin, R.; Wang, L.; Luo, Y. Kinetics, Thermodynamics, and Equilibrium of As(III), Cd(II), Cu(II) and Pb(II) Adsorption Using Porous Chitosan Bead-Supported MnFe2O4 Nanoparticles. Int. J. Min. Sci. Technol. 2021, 31, 1107–1115. [Google Scholar] [CrossRef]
- Karthik, R.; Meenakshi, S. Removal of Pb(II) and Cd(II) Ions from Aqueous Solution Using Polyaniline Grafted Chitosan. Chem. Eng. J. 2015, 263, 168–177. [Google Scholar] [CrossRef]
- Zhang, G.; Qu, R.; Sun, C.; Ji, C.; Chen, H.; Wang, C.; Niu, Y. Adsorption for Metal Ions of Chitosan Coated Cotton Fiber. J. Appl. Polym. Sci. 2008, 110, 2321–2327. [Google Scholar] [CrossRef]
- Paulino, A.T.; Belfiore, L.A.; Kubota, L.T.; Muniz, E.C.; Almeida, V.C.; Tambourgi, E.B. Effect of Magnetite on the Adsorption Behavior of Pb(II), Cd(II), and Cu(II) in Chitosan-Based Hydrogels. Desalination 2011, 275, 187–196. [Google Scholar] [CrossRef]
- Ge, H.; Fan, X. Adsorption of Pb2+ and Cd2+ onto a Novel Activated Carbon-Chitosan Complex. Chem. Eng. Technol. 2011, 34, 1745–1752. [Google Scholar] [CrossRef]
- Babakhani, A.; Sartaj, M. Competitive Adsorption of Nickel(II) and Cadmium(II) Ions by Chitosan Cross-Linked with Sodium Tripolyphosphate. Chem. Eng. Commun. 2021, 209, 1348–1366. [Google Scholar] [CrossRef]
- Bassi, R.; Prasher, S.O.; Simpson, B.K. Removal of Selected Metal Ions from Aqueous Solutions Using Chitosan Flakes. Sep. Sci. Technol. 2000, 35, 547–560. [Google Scholar] [CrossRef]
- Zielińska, K.; Chostenko, A.; Truszkowski, S. Adsorption of Cadmium Ions on Chitosan Membranes: Kinetics and Equilibrium Studies. Prog. Chem. Appl. Chitin Deriv. 2010, 15, 73–78. [Google Scholar]
- Sobhanardakani, S.; Zandipak, R.; Parvizimosaed, H.; Khoei, A.J.; Moslemi, M.; Tahergorabi, M.; Hosseini, S.M.; Delfieh, P. Efficiency of Chitosan for the Removal of Pb (II), Fe (II) and Cu (II) Ions from Aqueous Solutions. Iran. J. Toxicol. 2014, 8, 1145–1151. [Google Scholar]
- Chen, B.; Zhao, H.; Chen, S.; Long, F.; Huang, B.; Yang, B.; Pan, X. A Magnetically Recyclable Chitosan Composite Adsorbent Functionalized with EDTA for Simultaneous Capture of Anionic Dye and Heavy Metals in Complex Wastewater. Chem. Eng. J. 2019, 356, 69–80. [Google Scholar] [CrossRef]
- Xu, X.; Ouyang, X.-K.; Yang, L.-Y. Adsorption of Pb(II) from Aqueous Solutions Using Crosslinked Carboxylated Chitosan/Carboxylated Nanocellulose Hydrogel Beads. J. Mol. Liq. 2021, 322, 114523. [Google Scholar] [CrossRef]
- Guo, D.-M.; An, Q.-D.; Xiao, Z.-Y.; Zhai, S.-R.; Yang, D.-J. Efficient Removal of Pb(II), Cr(VI) and Organic Dyes by Polydopamine Modified Chitosan Aerogels. Carbohydr. Polym. 2018, 202, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Sharififard, H.; Shahraki, Z.H.; Rezvanpanah, E.; Rad, S.H. A Novel Natural Chitosan/Activated Carbon/Iron Bio-Nanocomposite: Sonochemical Synthesis, Characterization, and Application for Cadmium Removal in Batch and Continuous Adsorption Process. Bioresour. Technol. 2018, 270, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Wang, X.; Wang, G.; Hao, C.; Li, X.; Li, T. Adsorption Performance of a Polysaccharide Composite Hydrogel Based on Crosslinked Glucan/Chitosan for Heavy Metal Ions. Compos. Part B Eng. 2019, 169, 45–54. [Google Scholar] [CrossRef]
- Li, X.; Zhou, H.; Wu, W.; Wei, S.; Xu, Y.; Kuang, Y. Studies of Heavy Metal Ion Adsorption on Chitosan/Sulfydryl-Functionalized Graphene Oxide Composites. J. Colloid Interface Sci. 2015, 448, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Kumara, N.T.R.N.; Hamdan, N.; Petra, M.I.; Tennakoon, K.U.; Ekanayake, P. Equilibrium Isotherm Studies of Adsorption of Pigments Extracted from Kuduk-Kuduk (Melastoma malabathricum L.) Pulp onto TiO2 Nanoparticles. J. Chem. 2014, 2014, 468975. [Google Scholar] [CrossRef]
- Giles, C.H.; Smith, D.; Huitson, A. A General Treatment and Classification of the Solute Adsorption Isotherm. J. Colloid Interface Sci. 1974, 47, 755–765. [Google Scholar] [CrossRef]
- Essington, M.E. Soil and Water Chemistry: An Integrative Approach; CRC Press: Boca Raton, FL, USA, 2004; ISBN 9772081415. [Google Scholar]

| Heavy Metal | qmax (mg g−1) | Adsorbent Material | Reference |
|---|---|---|---|
| Cd | 1.06 | Chitosan | [88] |
| Cd | 9.9 | [89] | |
| Cd | 94 | [90] | |
| Pb | 7.64 | [77] | |
| Pb | 34.98 | [79] | |
| Pb | 55.5 | [91] | |
| Pb | 34.13 | Epichlorohydrin crosslinked chitosan | [78] |
| Pb | 63.33 | Chitosan–magnetite | [80] |
| Pb | 112.98 | Magnetic chitosan nanocomposites | [81] |
| Pb | 142.67 | Geopolymer–alginate– chitosan | [82] |
| Pb | 189.60 | Magnetic chitosan functionalized with EDTA | [92] |
| Pb | 334.90 | Crosslinked carboxylated chitosan–carboxylated nanocellulose hydrogel beads | [93] |
| Pb | 441.20 | Polydopamine-modified chitosan | [94] |
| Cd | 344.00 | Chitosan–activated- carbon–iron bio-nanocomposite | [95] |
| Pb | 11.98 | Chitosan bead-supported MnFe2O4 nanoparticles | [83] |
| Cd | 9.73 | ||
| Pb | 13.23 | Polyaniline-grafted chitosan | [84] |
| Cd | 12.87 | ||
| Pb | 86.09 | Chitosan-coated cotton fibers | [85] |
| Cd | 14.14 | ||
| Pb | 96.62 | Chitosan-g-methylenebisacrylamide/poly(acrylic acid) | [86] |
| Cd | 80.57 | ||
| Pb | 125.40 | Activated carbon–chitosan complex | [87] |
| Cd | 69.40 | ||
| Pb | 395.00 | Crosslinked glucan–chitosan | [96] |
| Cd | 269.00 | ||
| Pb | 447.00 | Chitosan–sulfhydryl-functionalized graphene oxide composites | [97] |
| Cd | 177.00 | ||
| Pb | 170.07 | CHIT-PAAA | Current Study |
| Cd | 180.51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganea, I.-V.; Nan, A.; Roba, C.; Neamțiu, I.; Gurzău, E.; Turcu, R.; Filip, X.; Baciu, C. Development of a New Eco-Friendly Copolymer Based on Chitosan for Enhanced Removal of Pb and Cd from Water. Polymers 2022, 14, 3735. https://doi.org/10.3390/polym14183735
Ganea I-V, Nan A, Roba C, Neamțiu I, Gurzău E, Turcu R, Filip X, Baciu C. Development of a New Eco-Friendly Copolymer Based on Chitosan for Enhanced Removal of Pb and Cd from Water. Polymers. 2022; 14(18):3735. https://doi.org/10.3390/polym14183735
Chicago/Turabian StyleGanea, Iolanda-Veronica, Alexandrina Nan, Carmen Roba, Iulia Neamțiu, Eugen Gurzău, Rodica Turcu, Xenia Filip, and Călin Baciu. 2022. "Development of a New Eco-Friendly Copolymer Based on Chitosan for Enhanced Removal of Pb and Cd from Water" Polymers 14, no. 18: 3735. https://doi.org/10.3390/polym14183735
APA StyleGanea, I.-V., Nan, A., Roba, C., Neamțiu, I., Gurzău, E., Turcu, R., Filip, X., & Baciu, C. (2022). Development of a New Eco-Friendly Copolymer Based on Chitosan for Enhanced Removal of Pb and Cd from Water. Polymers, 14(18), 3735. https://doi.org/10.3390/polym14183735












