Alkalization of Kraft Pulps from Pine and Eucalyptus and Its Effect on Enzymatic Saccharification and Viscosity Control of Cellulose
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Bleached Kraft Pulps
2.3. Cold Caustic Extraction (CCE)
2.4. Chemical Characterization
2.5. Fiber Biometry
2.6. Intrinsic Viscosity
2.7. Carboxyl Content
2.8. Specific Surface Area (SSA) from Methylene Blue (MB) Adsorption (SSAMB)
MB Adsorption and Langmuir Isotherm
2.9. X Ray Diffraction of Pulp Samples
2.10. Enzymatic Multicomponent Saccharification of Pulps
2.11. Endoglucanase Treatment of Pulps for Viscosity Reduction
2.12. Data Analysis
3. Results and Discussion
3.1. Chemical Composition and Fiber Biometry
3.2. Specific Surface Area by Methylene Blue (MB) Adsorption (SSAMB)
3.3. Cellulose Crystallinity
3.4. Intrinsic Viscosity
3.5. Carboxyl Content
3.6. Influence of CCE Treatment on Pulp Features
3.7. Enzymatic Multicomponent Saccharification of Pulps
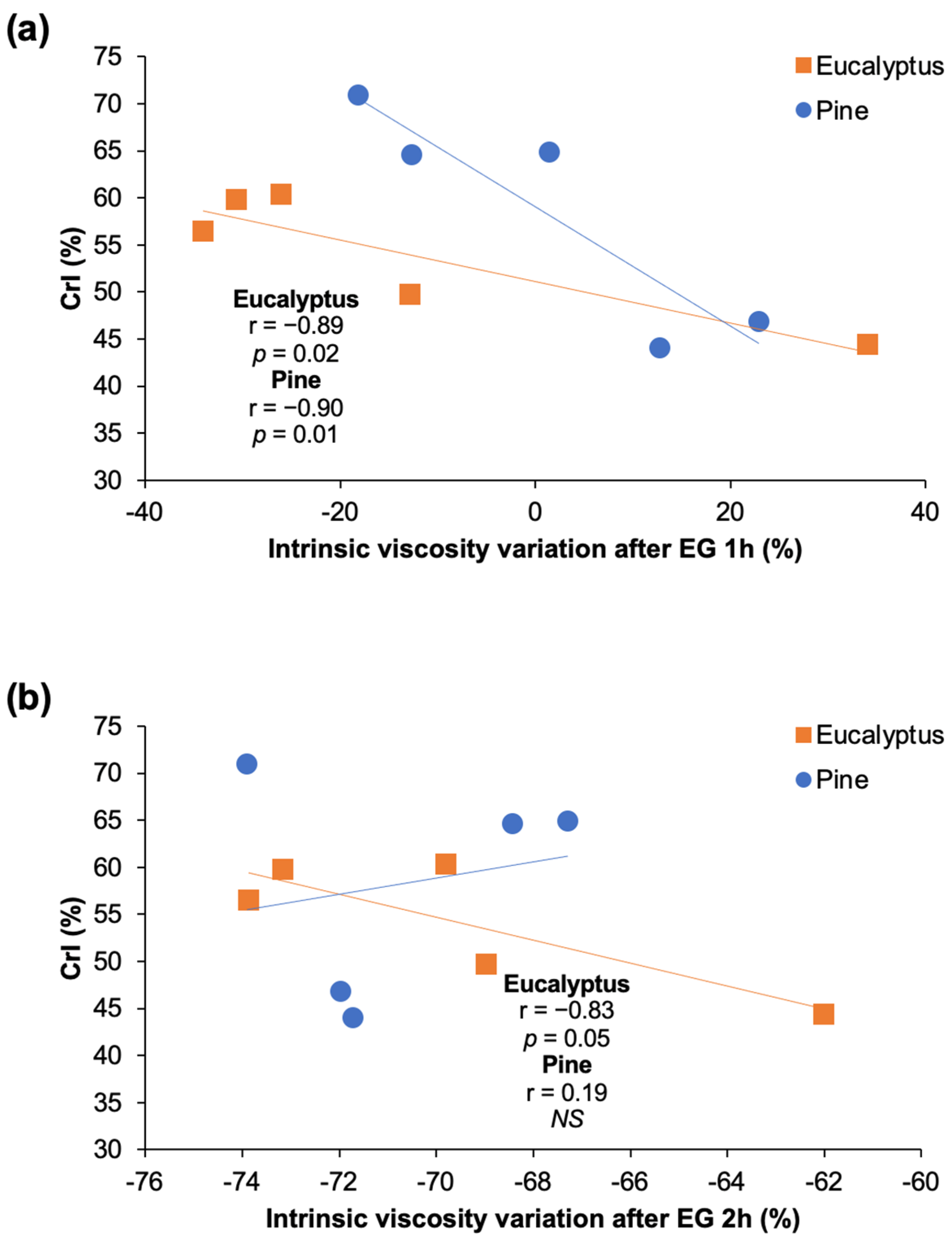
3.8. Endoglucanase (EG) Treatment of Pulps for Viscosity Control
4. Conclusions
- (1)
- CCE treatment was able to generate modifications in chemical composition and crystallinity of eucalyptus and pine bleached kraft pulps. NaOH concentrations above 17.5% cause a depolymerization of the cellulose chains, a decrease in fiber length and in CrI. Furthermore, eucalyptus started the conversion of cellulose I to cellulose II at NaOH 10%, while in pine it occurs at 17.5% NaOH.
- (2)
- Enzymatic saccharification yields higher than 90% where obtained with CCE-treated eucalyptus pulps while in bleached pulp it was 80%. In pine, only CCE-treated pulps with higher proportion of cellulose II showed saccharification yields higher than 90%.
- (3)
- CCE combined with EG treatment is an efficient method to control the intrinsic viscosity of pulps. The eucalyptus samples showed higher susceptibility to the EG treatment than the pine samples, which was mainly related to the low crystallinity of the pulps.
- (4)
- Overall, eucalyptus pulps are more accessible and reactive than pine pulps. The objective of increasing the reactivity, converting cellulose I to cellulose II hydrate, proved to be relevant only for the saccharification of pine pulps.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ciolacu, D.; Gorgieva, S.; Tampu, D.; Kokol, V. Enzymatic hydrolysis of different allomorphic forms of microcrystalline cellulose. Cellulose 2011, 18, 1527–1541. [Google Scholar] [CrossRef]
- Jin, E.; Zhang, Y.; Hu, F.; Yang, F.; Wu, S.; Jin, Y.; Song, J. To understand the superior hydrolytic activity after polymorphic conversion from cellulose I to II from the adsorption behaviors of enzymes. Cellulose 2017, 24, 1371–1381. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink, H.-P.; Bohn, A. Cellulose: Fascinating Biopolymer and Sustainable Raw Material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, I.; Mendonça, R.T.; Ago, M.; Rojas, O.J. Comparative study of cellulosic components isolated from different Eucalyptus species. Cellulose 2018, 25, 1011–1029. [Google Scholar] [CrossRef]
- Zugenmaier, P. Cellulose. Crystalline Cellulose and Cellulose Derivatives: Characterization and Structures; Springer: Berlin/Heidelberg, Germany, 2008; pp. 101–174. [Google Scholar]
- Ek, M.; Gellerstedt, G.; Henriksson, G. Wood Chemistry and Wood Biotechnology. In Pulp and Paper Chemistry and Technology; Ek, M., Gellerstedt, G., Henriksson, G., Eds.; Walter de Gruyter GmbH & Co. KG.: Berlin, Germany, 2009; Volume 1, pp. 1–301. [Google Scholar]
- Rojas, O.J. Cellulose Chemistry and Properties: Fibers, Nanocelluloses and Advanced Materials; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–329. [Google Scholar]
- Okano, T.; Sarko, A. Mercerization of cellulose. II. Alkali-cellulose intermediate and a possible mercerization mechanism. J. Appl. Polym. Sci. 1985, 30, 325–332. [Google Scholar] [CrossRef]
- Dinand, E.; Vignon, M.; Chanzy, H.; Heux, L. Mercerization of primary wall cellulose and its implication for the conversion of cellulose I→cellulose II. Cellulose 2002, 9, 7–18. [Google Scholar] [CrossRef]
- Porro, F.; Bedue, I.; Chanzy, H.; Heux, L. Solid-state 13C NMR study of Na-cellulose complexes. Biomacromolecules 2007, 8, 2586–2593. [Google Scholar] [CrossRef] [PubMed]
- Duchemin, B.J.C. Mercerisation of cellulose in aqueous NaOH at low concentrations. Green Chem. 2015, 17, 3941–3947. [Google Scholar] [CrossRef]
- Arnoul-Jarriault, B.; Lachenal, D.; Chirat, C.; Heux, L. Upgrading softwood bleached kraft pulp to dissolving pulp by cold caustic treatment and acid-hot caustic treatment. Ind. Crops. Prod. 2015, 65, 565–571. [Google Scholar] [CrossRef]
- Quintana, E.; Valls, C.; Vidal, T.; Roncero, M.B. Comparative evaluation of the action of two different endoglucanases. Part I: On a fully bleached, commercial acid sulfite dissolving pulp. Cellulose 2015, 22, 2067–2079. [Google Scholar] [CrossRef]
- Carrillo-Varela, I.; Retamal, R.; Pereira, M.; Mendonça, R.T. Structure and reactivity of cellulose from bleached kraft pulps of different Eucalyptus species upgraded to dissolving pulp. Cellulose 2019, 26, 5731–5744. [Google Scholar] [CrossRef]
- Sixta, H. Handbook of Pulp; Sixta, H., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Wenheim, Germany, 2008; Volume 1–2, ISBN 3527309993. [Google Scholar]
- Arantes, V.; Dias, I.K.R.; Berto, G.; Pereira, B.; Marotti, B.S.; Nogueira, C.F.O. The current status of the enzyme- mediated isolation and functionalization of nanocelluloses: Production, properties, techno-economics, and opportunities. Cellulose 2020, 27, 10571–10630. [Google Scholar] [CrossRef]
- Chen, X.; Xiong, L.; Li, H.; Zhang, L.; Yuan, G.; Chen, X.; Wang, C.; Chen, X. The inhibitory effect of xylan on enzymatic hydrolysis of cellulose is dependent on cellulose ultrastructure. Cellulose 2020, 27, 4417–4428. [Google Scholar] [CrossRef]
- Wang, Z.; Winestrand, S.; Gillgren, T.; Jonsson, L.J. Chemical and structural factors influencing enzymatic saccharification of wood from aspen, birch and spruce. Biomass Bioenerg. 2018, 109, 125–134. [Google Scholar] [CrossRef]
- Duan, C.; Verma, S.K.; Li, J.; Ma, X.; Ni, Y. Combination of mechanical, alkaline and enzymatic treatments to upgrade paper-grade pulp to dissolving pulp with high reactivity. Bioresour. Technol. 2016, 200, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Legere, S.; He, Z.; Zhang, H.; Li, J.; Yang, B.; Zhang, S.; Zhang, L.; Zheng, L.; Ni, Y. Methods to increase the reactivity of dissolving pulp in the viscose rayon production process: A review. Cellulose 2018, 25, 3733–3753. [Google Scholar] [CrossRef]
- Nakano, T. Mechanism of microfibril contraction and anisotropic dimensional changes for cells in wood treated with aqueous NaOH solution. Cellulose 2010, 17, 711–719. [Google Scholar] [CrossRef][Green Version]
- Nakano, S.; Nakano, T. Morphological changes induced in wood samples by aqueous NaOH treatment and their effects on the conversion of cellulose I to cellulose II. Holzforschung 2015, 69, 483–491. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, H. X-ray diffraction study of bamboo fibers treated with NaOH. Fiber. Polym. 2008, 9, 735–739. [Google Scholar] [CrossRef]
- Carrillo-Varela, I.; Pereira, M.; Mendonça, R.T. Determination of polymorphic changes in cellulose from Eucalyptus spp. fibres after alkalization. Cellulose 2018, 25, 6831–6845. [Google Scholar] [CrossRef]
- Peretti, S.W.; Barton, R.; Mendonça, R.T. Lignin as Feedstock for Fibers and Chemicals in Commercializing Biobased Products: Opportunities, Challenges, Benefits, and Risks; Snyder, S.W., Ed.; RSC Green Chemistry: Cambridge, UK, 2015. [Google Scholar] [CrossRef]
- Li, J.; Liu, Y.; Duan, C.; Zhang, H.; Ni, Y. Mechanical pretreatment improving hemicelluloses removal from cellulosic fibers during cold caustic extraction. Bioresour. Technol. 2015, 136, 501–506. [Google Scholar] [CrossRef]
- Mendonça, R.T.; Jara, J.; González, V.; Elissetche, J.; Freer, J. Evaluation of the white-rot fungi Ganoderma austral and Ceriposipsis subvermispora in biotechnological applications. J. Ind. Microbiol. Biotechnol. 2008, 35, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Aguayo, M.G.; Ferraz, A.; Elissetche, J.P.; Masarin, F.; Mendonça, R.T. Lignin chemistry and topochemistry during kraft delignification of Eucalyptus globulus genotypes with contrasting pulpwood characteristics. Holzforschung 2014, 68, 623–629. [Google Scholar] [CrossRef]
- Carrillo, I.; Valenzuela, S.; Elissetche, J.P. Comparative evaluation of Eucalyptus globulus and E. nitens wood and fibre quality. IAWA J. 2017, 38, 105–116. [Google Scholar] [CrossRef]
- Saito, T.; Isogai, A. TEMPO-mediated oxidation of native cellulose. The effect of oxidation conditions on chemical and crystal structure of the water-insoluble fractions. Biomacromolecules 2004, 5, 1983–1989. [Google Scholar] [CrossRef] [PubMed]
- Kashcheyeva, E.I.; Gladysheva, E.K.; Skiba, E.A.; Budaeva, V.V. A study of properties and enzymatic hydrolysis of bacterial cellulose. Cellulose 2019, 26, 2255–2265. [Google Scholar] [CrossRef]
- Skripkina, T.; Podgorbunskikh, E.; Bychkov, A.; Lomovsky, O. Sorption of methylene blue for studying the specific surface properties of biomass carbohydrates. Coatings 2020, 10, 1115. [Google Scholar] [CrossRef]
- Kaewprasit, C.; Hequet, E.; Abidi, N.; Gourlot, J.P. Application of methylene blue adsorption to cotton fiber specific surface area measurement: Part I Methodology. J. Cotton Sci. 1998, 2, 164–173. [Google Scholar]
- Vilar, V.J.P.; Motelho, C.M.S.; Boaventura, R.A.E. Methylene blue adsorption by algal biomass based materials: Biosor-bents characterization and process behaviour. J. Hazard. Mater. 2007, 147, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Aung, E.I.; Fujii, S.; Hosomi, S.; Kimizu, M.; Ninomiya, K.; Takahashia, K. Investigation of accessibility and reactivity of cellulose pretreated by ionic liquid at high loading. Carbohydr. Polym. 2017, 176, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Nam, S.; French, A.D.; Condon, B.D.; Concha, M. Segal crystallinity index revisited by the simulation of X-ray diffraction patterns of cotton cellulose Ib and cellulose II. Carbohydr. Polym. 2016, 135, 1–9. [Google Scholar] [CrossRef]
- Carrillo-Varela, I.; Mendonça, R.T.; Pereira, M.; Reyes-Contreras, P.; Contreras, D. Methylene blue adsorption onto hydrogels made from different Eucalyptus dissolving pulps. Cellulose 2022, 29, 445–468. [Google Scholar] [CrossRef]
- Park, S.; Baker, J.O.; Himmel, M.E.; Parilla, P.A.; Johnson, D.K. Cellulose crystallinity index: Measurement techniques and their impact on interpreting cellulose performance. Biotechnol. Biofuels 2010, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.Y.; Yoo, D.I.; Shin, Y.; Kim, H.C.; Kim, H.Y.; Chung, Y.S.; Park, W.H.; Youk, J.H. Crystalline structure analysis of cellulose treated with sodium hydroxide and carbon dioxide by means of X-ray diffraction and FTIR spectroscopy. Carbohydr. Res. 2005, 340, 2376–2391. [Google Scholar] [CrossRef] [PubMed]
- Xing, L.; Gu, J.; Zhang, W.; Tu, D.; Hu, C. Cellulose I and II nanocrystals produced by sulfuric acid hydrolysis of tetra pak cellulose I. Carbohydr. Polym. 2018, 192, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Scherrer, P. Bestimmung der Grosse und der Inneren Struktur von Kolloidteilchen Mittels Rontgenstrahlen; Nachrichten von der Gesellschaft der Wissenschaften: Gottingen, Germany, 1918. [Google Scholar]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Choi, K.H.; Kim, A.R.; Cho, B.U. Effects of alkali swelling and beating treatments on properties of kraft pulp fibers. BioRes 2016, 11, 3769–3782. [Google Scholar] [CrossRef]
- French, A.D. Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 2014, 21, 885–896. [Google Scholar] [CrossRef]
- Wada, M.; Okano, T.; Sugiyama, J. Allomorphs of native crystalline cellulose I evaluated by two equatorial d-spacings. J. Wood Sci. 2001, 47, 124–128. [Google Scholar] [CrossRef]
- Isogai, A.; Atalla, R.H. Dissolution of cellulose in aqueous NaOH solutions. Cellulose 1998, 5, 309–319. [Google Scholar] [CrossRef]
- Duchemin, B.; Thuault, A.; Vicente, A.; Rigaud, B.; Fernandez, C.; Eve, S. Ultrastructure of cellulose crystallites in flax textile fibres. Cellulose 2012, 19, 1837–1854. [Google Scholar] [CrossRef]
- Hakansson, H.; Ahlgren, P. Acid hydrolysis of some industrial pulps: Effect of hydrolysis conditions and raw material. Cellulose 2005, 12, 177–183. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Maltha, C.R.A.; Demuner, A.J.; Cazal, C.M.; Reis, E.L.; Colodette, J.L. A rapid method for quantification of carboxyl groups in cellulose pulp. BioRes 2013, 8, 1043–1054. [Google Scholar] [CrossRef]
- Johansson, M.H.; Samuelson, O. The formation of end groups in cellulose during alkali cooking. Carbohydr. Res. 1973, 34, 33–43. [Google Scholar] [CrossRef]
- Sjostrom, E. The origino f charge on cellulosic fibers. Nord. Pulp Pap. Res. J. 1989, 4, 90–93. [Google Scholar] [CrossRef]
- Zemljic, L.F.; Persin, Z.; Stenius, P.; Kleinschek, K.S. Carboxyl groups in pre-treated regenerated cellulose fibers. Cellulose 2008, 15, 681–690. [Google Scholar] [CrossRef]
- Pere, J.; Paakkonen, E.; Ji, Y.; Retulainen, E. Influence of the hemicellulose content on the fiber properties, strength, and formability of handsheets. BioResources 2019, 14, 251–263. [Google Scholar] [CrossRef]
- Fearon, O.; Nykanen, V.; Kuitunen, S.; Ruuttunen, K.; Alen, R.; Alopaeus, V.; Vuorinen, T. Detailed modeling of the kraft pulping chemistry: Carbohydrate reactions. AIChE J. 2020, 66, e16252. [Google Scholar] [CrossRef]
- Zhai, R.; Hu, J.; Saddler, J.N. The inhibition of hemicellulosic sugars on cellulose hydrolysis are highly dependant on the cellulase productive binding, processivity, and substrate surface charges. Bioresour. Technol. 2018, 258, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Wada, M.; Ike, M.; Tokuyasu, K. Enzymatic hydrolysis of cellulose I is greatly accelerated via its conversion to the cellulose II hydrate form. Polym. Degrad. Stab. 2010, 95, 543–548. [Google Scholar] [CrossRef]
- Linder, M.; Teeri, T.T. The Roles and Function of Cellulose—Binding Domains. J. Biotechnol. 1997, 57, 15–28. [Google Scholar] [CrossRef]
- Liu, Y.S.; Baker, J.O.; Zeng, Y.; Himmel, M.E.; Haas, T.; Ding, S.Y. Cellobiohydrolase Hydrolyzes Crystalline Cellulose on Hydrophobic Faces. J. Biol. Chem. 2011, 286, 11195–11201. [Google Scholar] [CrossRef] [PubMed]
- Ciolacu, D.; Chiriac, A.I.; Pastor, F.I.J.; Kokol, V. The influence of supramolecular structure of cellulose allomorphs on the interactions with cellulose-binding domain, CBD3b from Paenibacillus barcinonensis. Bioresour. Technol. 2014, 157, 14–21. [Google Scholar] [CrossRef]
- Ling, Z.; Zhang, Z.; Yang, G.; Takabe, K.; Xu, F. Nanocrystals of cellulose allomorphs have different adsorption of cellulase and subsequent degradation. Ind. Crops. Prod. 2018, 112, 541–549. [Google Scholar] [CrossRef]
- Gehmayr, V.; Sixta, H. Pulp properties and their influence on enzymatic degradability. Biomacromolecules 2012, 13, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Verma, S.K.; Li, J.; Ma, X.; Ni, Y. Viscosity control and reactivity improvements of cellulose fibers by cellulase treatment. Cellulose 2016, 23, 269–276. [Google Scholar] [CrossRef]
- Karkkainen, E. Industrial Production of Different Prehydrolysis Kraft Dissolving Pulp Grades. Master’s Thesis, Aalto University, Espoo, Finland, 8 March 2021. Master of Science in Technology. [Google Scholar]
- Wennerstrom, M.; Bylund, S. Method for Controlling Viscosity in Dissolving Pulps; Valmet, A.B., Sundsvall, S.E., Eds.; WO 2017/105322 A1; World Intellectual Property Organization: Geneva, Switzerland, 22 June 2017. [Google Scholar]
- Loureiro, P.E.G.; Cadete, S.M.S.; Tokin, R.; Evtuguin, D.V.; Lund, H.; Johansen, K.S. Enzymatic fibre modification during production of dissolving wood pulp for regenerated cellulosic materials. Front. Plant. Sci. 2021, 12, 717776. [Google Scholar] [CrossRef] [PubMed]
- Ibarra, D.; Kopcke, V.; Ek, M. Behavior of different monocomponent endoglucanases on the accessibility and reactivity of dissolving-grade pulps for viscose process. Enzyme Microb. Technol. 2010, 47, 355–362. [Google Scholar] [CrossRef]
- Duan, C.; Long, Y.; Li, J.; Ma, X.; Ni, Y. Changes of cellulose accessibility to cellulase due to fiber hornification and its impact on enzymatic viscosity control of dissolving pulp. Cellulose 2015, 22, 2729–2736. [Google Scholar] [CrossRef]
- Henrissat, B.; Driguez, H.; Viet, C.; Schuelin, M. Synergism of cellulases from Trichoderma reesei in the degradation of cellulose. Nat. Biotechnol. 1985, 3, 722–726. [Google Scholar] [CrossRef]
- Gronqvist, S.; Hakala, T.K.; Kamppuri, T.; Vehvilainen, M.; Hanninen, T.; Liitia, T.; Maloney, T.; Suurnakki, A. Fibre porosity development of dissolving pulp during mechanical and enzymatic processing. Cellulose 2014, 21, 3667–3676. [Google Scholar] [CrossRef]










| Sample | Treatment | Glucans (%) | Xylans 1 or Mannans 2 (%) | Fiber Length (mm) | Fiber Width (um) | Fines Content (%) | Mean Kink Index |
|---|---|---|---|---|---|---|---|
| Eucalyptus 1 | Bleached | 90.2 ± 1 | 9.8 ± 0.2 | 0.69 ± 0.01 | 18.1 ± 0.1 | 4.4 ± 0.1 | 3 ± 0 |
| CCE5 | 93.6 ± 1 | 6.5 ± 0.1 | 0.647 ± 0.004 | 18.1 ± 0.1 | 4.5 ± 0.2 | 3.8 ± 0.3 | |
| CCE10 | >99 | <1 | 0.575 ± 0.001 | 18 ± 0 | 5.8 ± 0.3 | 3.79 ± 0.01 | |
| CCE17.5 | >99 | <1 | 0.536 ± 0.004 | 17 ± 0 | 7 ± 1 | 4 ± 0.03 | |
| CCE35 | >99 | <1 | 0.59 ± 0.08 | 17.3 ± 0.4 | 5 ± 1 | 3.83 ± 0.03 | |
| Pine 2 | Bleached | 92 ± 1 | 7.5 ± 0.4 | 2.224 ± 0.004 | 32.5 ± 0.2 | 3.1 ± 0.1 | 3 ± 0.1 |
| CCE5 | 93.2 ± 0.2 | 6.5 ± 0.2 | 2.19 ± 0.02 | 32.5 ± 0.1 | 2.3 ± 0.1 | 3.3 ± 0.1 | |
| CCE10 | 94 ± 3 | 5.5 ± 1.1 | 2.031 ± 0.001 | 33.1 ± 0.1 | 2.5 ± 0.1 | 3.99 ± 0.04 | |
| CCE17.5 | >99 | <1 | 1.79 ± 0.03 | 31.6 ± 0.4 | 3 ± 1 | 4.1 ± 0.4 | |
| CCE35 | >99 | <1 | 1.74 ± 0.02 | 31.5 ± 0.5 | 2.4 ± 0.4 | 4.2 ± 0.1 |
| Carboxyl Content | Intrinsic Viscosity | CrI | SSA | FL | FW | Fines | Kink Index | Xylans Content | Glucans Content | |
|---|---|---|---|---|---|---|---|---|---|---|
| Carboxyl content | 1 | 0.11 | 0.32 | 0.85 * | 0.80 | 0.47 | −0.65 | −0.93 * | 0.83 * | −0.83 * |
| Intrinsic viscosity | 1 | 0.91 * | 0.37 | 0.38 | 0.79 | −0.15 | −0.38 | 0.48 | −0.48 | |
| CrI | 1 | 0.63 | 0.69 | 0.96 * | −0.52 | −0.55 | 0.74 | −0.74 | ||
| SSA | 1 | 0.88 * | 0.70 | −0.72 | −0.81 | 0.97 * | −0.97 * | |||
| FL | 1 | 0.84 * | −0.94 * | −0.87 * | 0.95 * | −0.95 * | ||||
| FW | 1 | −0.73 | −0.69 | 0.83 * | −0.83 * | |||||
| Fines | 1 | 0.69 | −0.81 | 0.80 | ||||||
| Kink index | 1 | −0.85 * | 0.86 | |||||||
| Xylans content | 1 | 0.99 * | ||||||||
| Glucans content | 1 |
| Carboxyl Content | Intrinsic Viscosity | CrI | SSA | FL | FW | Fines | Kink Index | Xylans Content | Glucans Content | |
|---|---|---|---|---|---|---|---|---|---|---|
| Carboxyl content | 1 | 0.45 | 0.56 | 0.52 | 0.75 | 0.13 | 0.37 | −0.93 * | 0.65 | −0.63 |
| Intrinsic viscosity | 1 | 0.89 * | 0.78 | 0.80 | 0.77 | −0.25 | −0.59 | 0.77 | −0.76 | |
| CrI | 1 | 0.98 * | 0.94 * | 0.85 * | 0.01 | −0.75 | 0.95 * | −0.95 * | ||
| SSA | 1 | 0.91 * | 0.84 * | 0.08 | −0.73 | 0.95 * | −0.95 * | |||
| FL | 1 | 0.76 | 0.31 | −0.92 * | 0.99 * | −0.98 * | ||||
| FW | 1 | 0.07 | −0.45 | 0.83 | −0.84 * | |||||
| Fines | 1 | −0.48 | 0.32 | −0.33 | ||||||
| Kink index | 1 | −0.86 * | 0.85 * | |||||||
| Xylans content | 1 | 0.98 * | ||||||||
| Glucans content | 1 |
| Specie | Treatment | Lateral Crystallite Size (nm) | D-Spacing (Å) | ||
|---|---|---|---|---|---|
| CI 1 (200) | CII 2 (110) | CI 1 (200) | CII 2 (110) | ||
| Eucalyptus | Bleached | 4.49 | - | 3.95 | - |
| CCE5 | 4.51 | - | 3.94 | - | |
| CCE10 | 5.01 | 6.79 | 3.94 | 4.40 | |
| CCE17.5 | 5.13 | 5.77 | 3.93 | 4.44 | |
| CCE35 | - | 6.29 | - | 4.42 | |
| Pine | Bleached | 4.97 | - | 3.95 | - |
| CCE5 | 5.04 | - | 3.98 | - | |
| CCE10 | 5.26 | - | 3.96 | - | |
| CCE17.5 | 5.00 | 5.82 | 3.91 | 4.47 | |
| CCE35 | - | 5.70 | - | 4.57 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carrillo-Varela, I.; Vidal, C.; Vidaurre, S.; Parra, C.; Machuca, Á.; Briones, R.; Mendonça, R.T. Alkalization of Kraft Pulps from Pine and Eucalyptus and Its Effect on Enzymatic Saccharification and Viscosity Control of Cellulose. Polymers 2022, 14, 3127. https://doi.org/10.3390/polym14153127
Carrillo-Varela I, Vidal C, Vidaurre S, Parra C, Machuca Á, Briones R, Mendonça RT. Alkalization of Kraft Pulps from Pine and Eucalyptus and Its Effect on Enzymatic Saccharification and Viscosity Control of Cellulose. Polymers. 2022; 14(15):3127. https://doi.org/10.3390/polym14153127
Chicago/Turabian StyleCarrillo-Varela, Isabel, Claudia Vidal, Sebastián Vidaurre, Carolina Parra, Ángela Machuca, Rodrigo Briones, and Regis Teixeira Mendonça. 2022. "Alkalization of Kraft Pulps from Pine and Eucalyptus and Its Effect on Enzymatic Saccharification and Viscosity Control of Cellulose" Polymers 14, no. 15: 3127. https://doi.org/10.3390/polym14153127
APA StyleCarrillo-Varela, I., Vidal, C., Vidaurre, S., Parra, C., Machuca, Á., Briones, R., & Mendonça, R. T. (2022). Alkalization of Kraft Pulps from Pine and Eucalyptus and Its Effect on Enzymatic Saccharification and Viscosity Control of Cellulose. Polymers, 14(15), 3127. https://doi.org/10.3390/polym14153127






