Strain–Microstructure–Optoelectronic Inter-Relationship toward Engineering Mechano-Optoelectronic Conjugated Polymer Thin Films
Abstract
1. Introduction
2. Design of Conjugated Polymer MO Systems
2.1. Regioregularity
2.2. Crystallinity
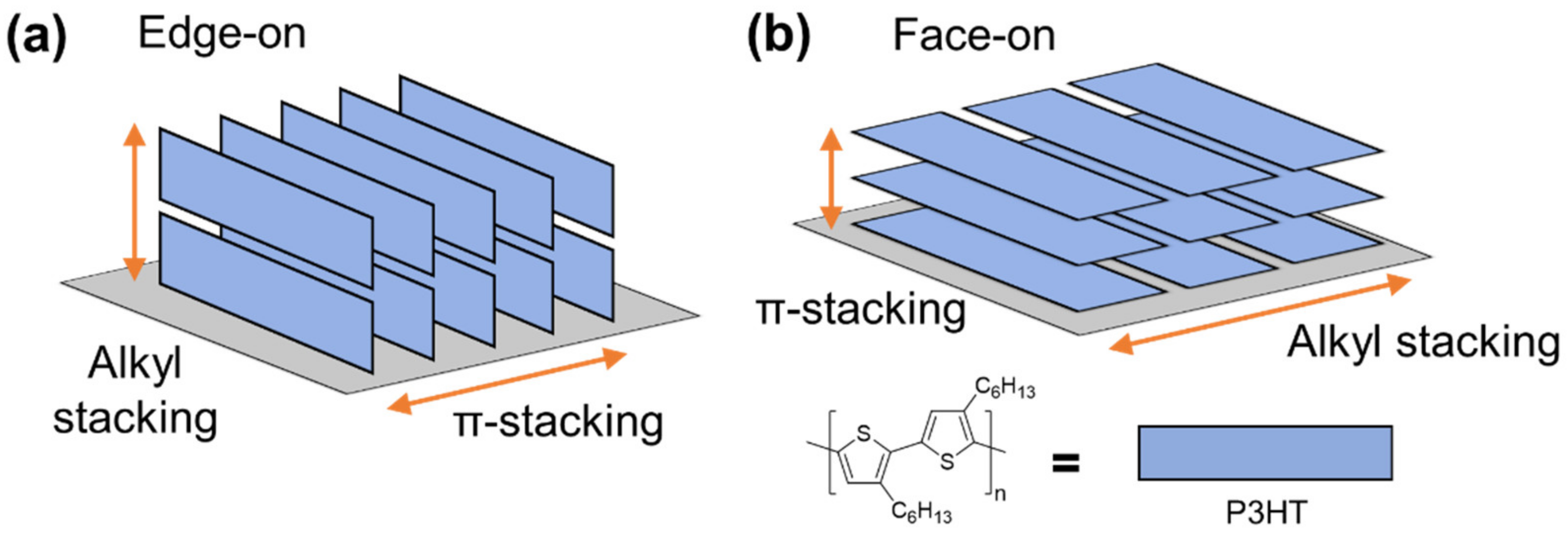
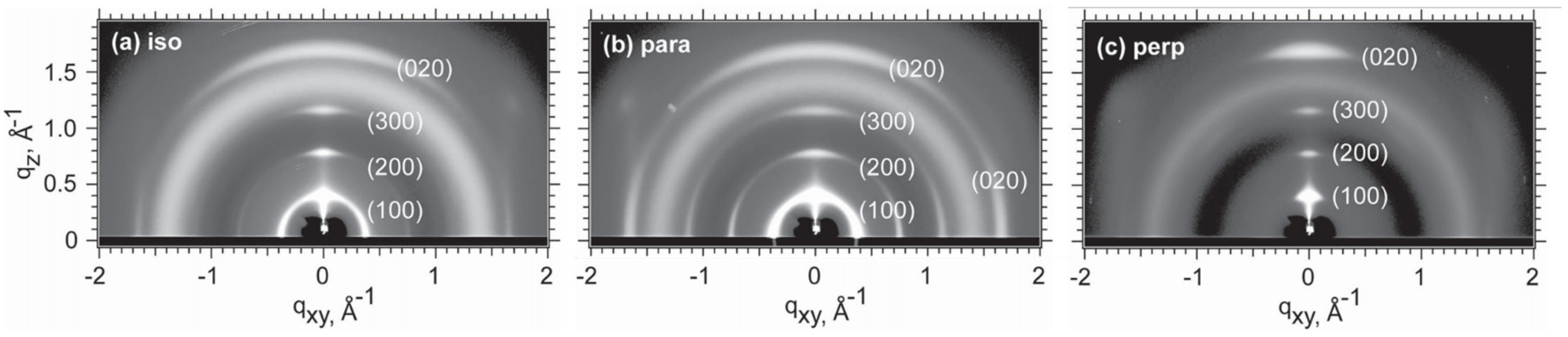
2.3. Orientation
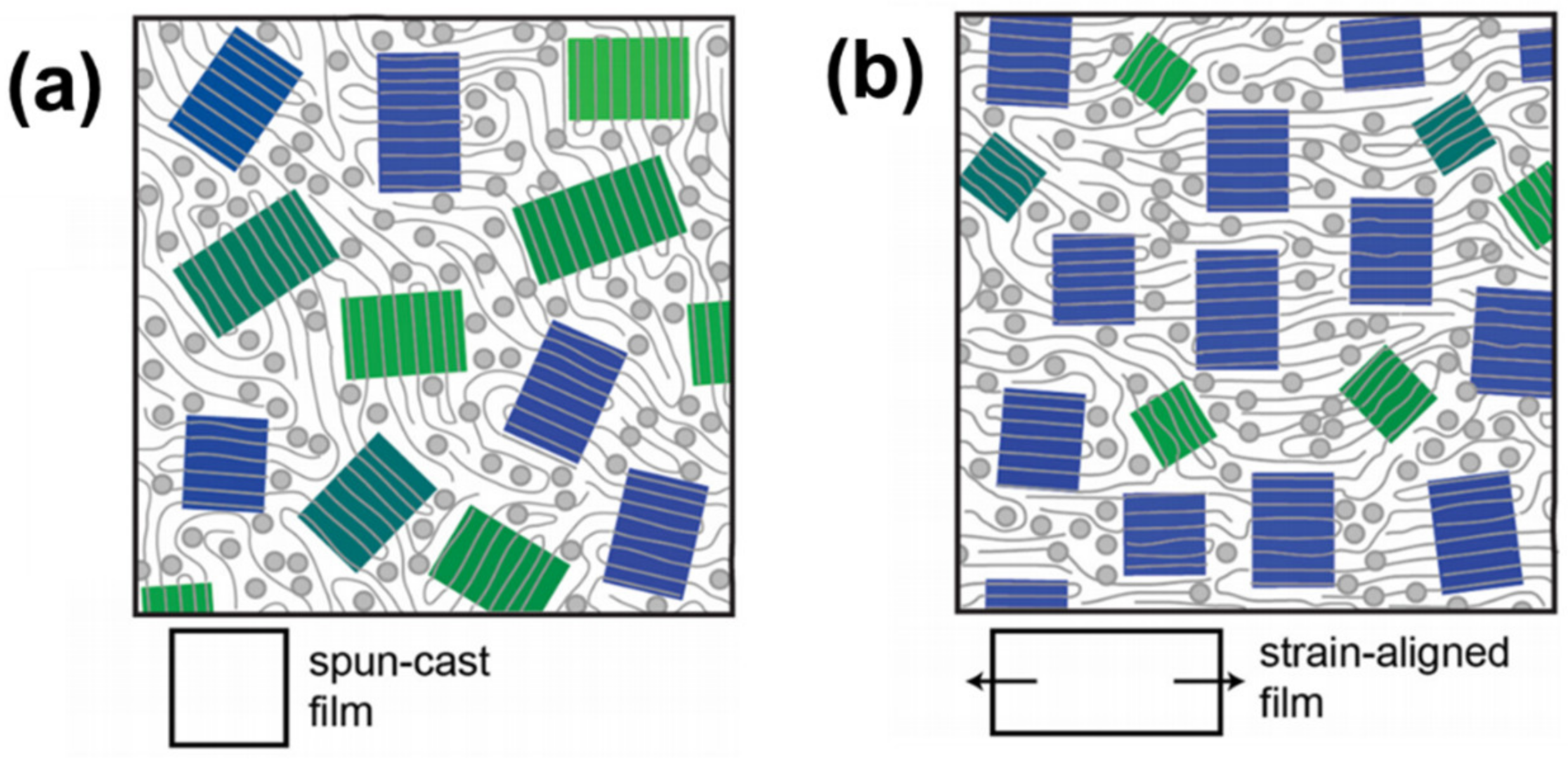
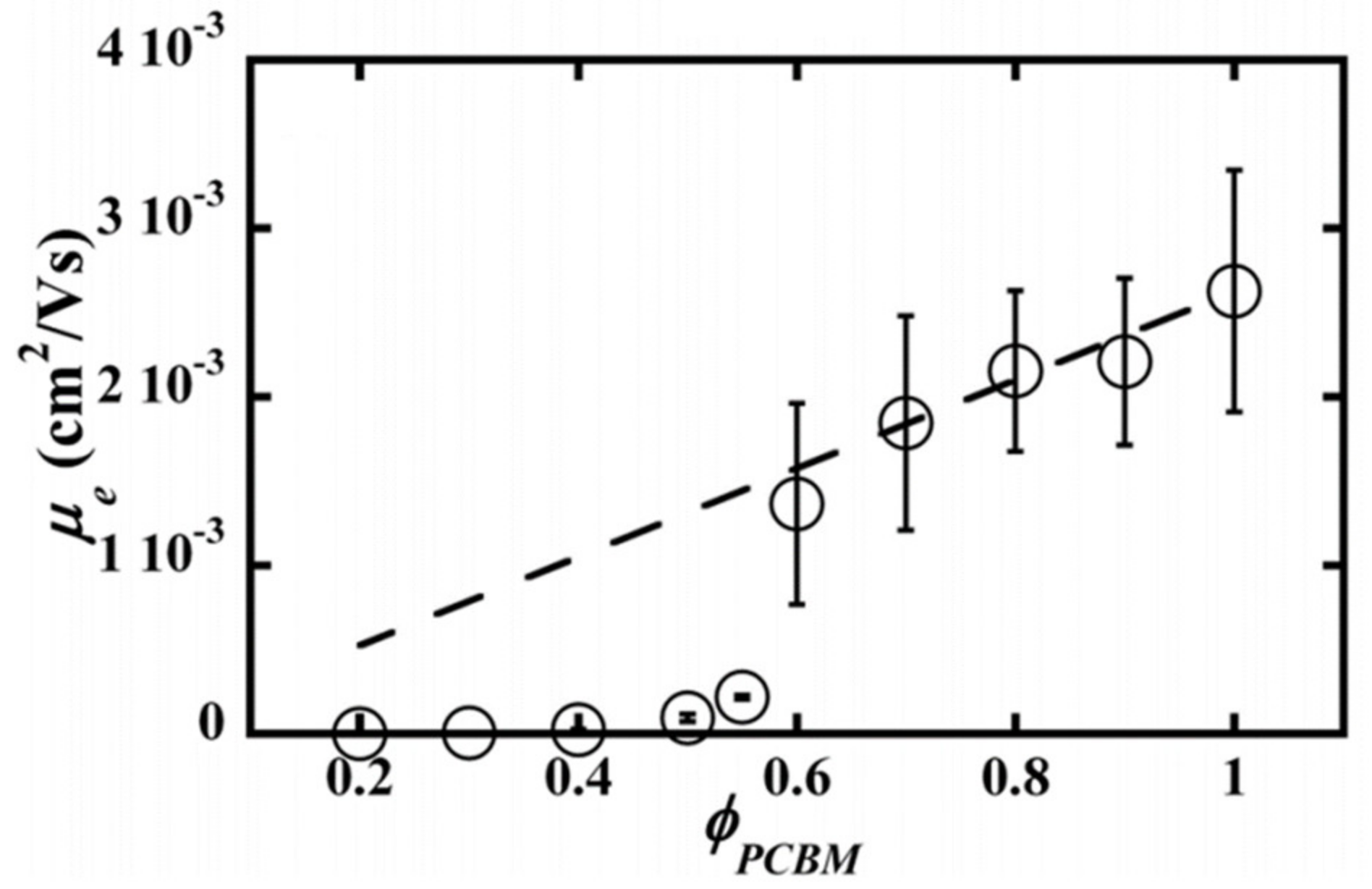
2.4. Blend Morphology
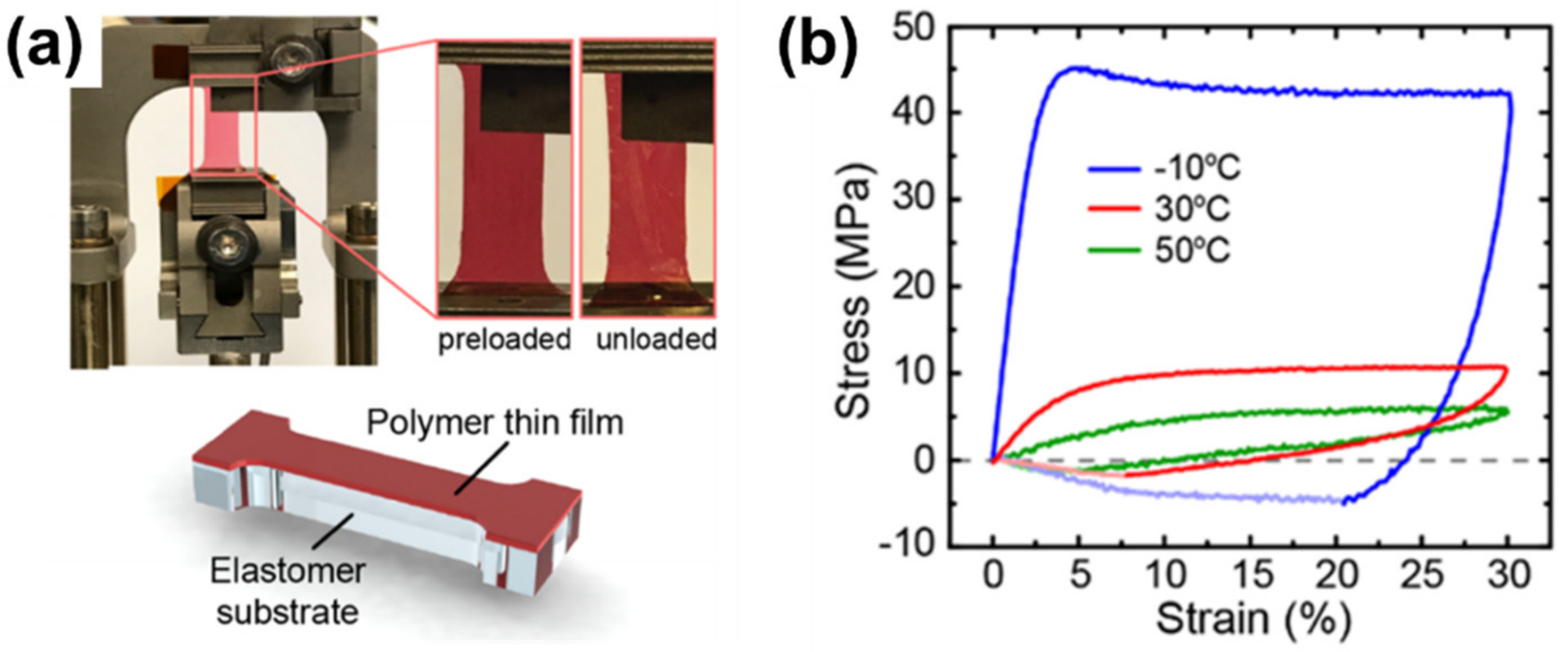
2.5. Mechanical Properties
3. Manufacturing of Conjugated Polymer MO Systems
4. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Le, T.-H.; Kim, Y.; Yoon, H. Electrical and Electrochemical Properties of Conducting Polymers. Polymers 2017, 9, 150. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-W.; Lee, J.-H.; Lin, B.-Y.; Chen, S.; Wu, S.-T. Liquid crystal display and organic light-emitting diode display: Present status and future perspectives. Light Sci. Appl. 2018, 7, 17168. [Google Scholar] [CrossRef] [PubMed]
- McNeill, C.R.; Greenham, N.C. Conjugated-Polymer Blends for Optoelectronics. Adv. Mater. 2009, 21, 3840–3850. [Google Scholar] [CrossRef]
- Pandey, M.; Kumari, N.; Nagamatsu, S.; Pandey, S.S. Recent advances in the orientation of conjugated polymers for organic field-effect transistors. J. Mater. Chem. C 2019, 7, 13323–13351. [Google Scholar] [CrossRef]
- Benten, H.; Mori, D.; Ohkita, H.; Ito, S. Recent research progress of polymer donor/polymer acceptor blend solar cells. J. Mater. Chem. A 2016, 4, 5340–5365. [Google Scholar] [CrossRef]
- Sirringhaus, H. 25th Anniversary Article: Organic Field-Effect Transistors: The Path Beyond Amorphous Silicon. Adv. Mater. 2014, 26, 1319–1335. [Google Scholar] [CrossRef]
- Berger, P.R.; Kim, M. Polymer solar cells: P3HT:PCBM and beyond. J. Renew. Sustain. Energy 2018, 10, 013508. [Google Scholar] [CrossRef]
- Gupta, A.S.P.; Kumar, A.; Shree, P.; Mishra, S.; Joseph, C.M. Preperation and Characterization of P3HT-PCBM Organic Solar Cells. Int. J. Electron. Elect. Eng. 2013, 2, 5. [Google Scholar]
- Dang, M.T.; Hirsch, L.; Wantz, G. P3HT:PCBM, Best Seller in Polymer Photovoltaic Research. Adv. Mater. 2011, 23, 3597–3602. [Google Scholar] [CrossRef] [PubMed]
- Jinno, H.; Fukuda, K.; Xu, X.; Park, S.; Suzuki, Y.; Koizumi, M.; Yokota, T.; Osaka, I.; Takimiya, K.; Someya, T. Stretchable and waterproof elastomer-coated organic photovoltaics for washable electronic textile applications. Nat. Energy 2017, 2, 780–785. [Google Scholar] [CrossRef]
- Xu, J.; Wang, S.; Wang, G.-J.N.; Zhu, C.; Luo, S.; Jin, L.; Gu, X.; Chen, S.; Feig, V.R.; To, J.W.F.; et al. Highly stretchable polymer semiconductor films through the nanoconfinement effect. Science 2017, 355, 59–64. [Google Scholar] [CrossRef]
- Ocheje, M.U.; Charron, B.P.; Nyayachavadi, A.; Rondeau-Gagné, S. Stretchable electronics: Recent progress in the preparation of stretchable and self-healing semiconducting conjugated polymers. Flex. Print. Electron. 2017, 2, 043002. [Google Scholar] [CrossRef]
- Lipomi, D.J.; Tee, B.C.-K.; Vosgueritchian, M.; Bao, Z. Stretchable Organic Solar Cells. Adv. Mater. 2011, 23, 1771–1775. [Google Scholar] [CrossRef]
- Kaur, N.; Singh, M.; Pathak, D.; Wagner, T.; Nunzi, J. Organic materials for photovoltaic applications: Review and mechanism. Synth. Met. 2014, 190, 20–26. [Google Scholar] [CrossRef]
- Leblebici, S.; Lee, J.; Weber-Bargioni, A.; Ma, B. Dielectric Screening to Reduce Charge Transfer State Binding Energy in Organic Bulk Heterojunction Photovoltaics. J. Phys. Chem. C 2017, 121, 3279–3285. [Google Scholar] [CrossRef]
- Brabec, C.J.; Gowrisanker, S.; Halls, J.J.M.; Laird, D.; Jia, S.; Williams, S.P. Polymer-Fullerene Bulk-Heterojunction Solar Cells. Adv. Mater. 2010, 22, 3839–3856. [Google Scholar] [CrossRef] [PubMed]
- Brabec, C.J.; Heeney, M.; McCulloch, I.; Nelson, J. Influence of blend microstructure on bulk heterojunction organic photovoltaic performance. Chem. Soc. Rev. 2010, 40, 1185–1199. [Google Scholar] [CrossRef]
- Finn, M.; Martens, C.J.; Zaretski, A.V.; Roth, B.; Søndergaard, R.R.; Krebs, F.C.; Lipomi, D.J. Mechanical stability of roll-to-roll printed solar cells under cyclic bending and torsion. Sol. Energy Mater. Sol. Cells 2018, 174, 7–15. [Google Scholar] [CrossRef]
- Awartani, O.; Lemanski, B.I.; Ro, H.W.; Richter, L.J.; Delongchamp, D.M.; O’Connor, B.T. Correlating Stiffness, Ductility, and Morphology of Polymer:Fullerene Films for Solar Cell Applications. Adv. Energy Mater. 2012, 3, 399–406. [Google Scholar] [CrossRef]
- Kaltenbrunner, M.; White, M.S.; Glowacki, E.D.; Sekitani, T.; Someya, T.; Sariciftci, N.S.; Bauer, S. Ultrathin and lightweight organic solar cells with high flexibility. Nat. Commun. 2012, 3, 770. [Google Scholar] [CrossRef]
- Yan, T.; Song, W.; Huang, J.; Peng, R.; Huang, L.; Ge, Z. 16.67% Rigid and 14.06% Flexible Organic Solar Cells Enabled by Ternary Heterojunction Strategy. Adv. Mater. 2019, 31, e1902210. [Google Scholar] [CrossRef]
- Ryu, D.; Loh, K.J. Strain sensing using photocurrent generated by photoactive P3HT-based nanocomposites. Smart Mater. Struct. 2012, 21, 65016. [Google Scholar] [CrossRef]
- Ryu, D.; Loh, K.J. Multi-modal sensing using photoactive thin films. Smart Mater. Struct. 2014, 23, 85011. [Google Scholar] [CrossRef]
- Ryu, D.; Meyers, F.N.; Loh, K.J. Inkjet-printed, flexible, and photoactive thin film strain sensors. J. Intell. Mater. Syst. Struct. 2014, 26, 1699–1710. [Google Scholar] [CrossRef]
- Ryu, D.; Mongare, A. Corrugated Photoactive Thin Films for Flexible Strain Sensor. Materials 2018, 11, 1970. [Google Scholar] [CrossRef]
- Dang, M.T.; Wantz, G.; Bejbouji, H.; Urien, M.; Dautel, O.J.; Vignau, L.; Hirsch, L. Polymeric solar cells based on P3HT:PCBM: Role of the casting solvent. Sol. Energy Mater. Sol. Cells 2011, 95, 3408–3418. [Google Scholar] [CrossRef]
- Guo, B.; Li, W.; Guo, X.; Meng, X.; Maojie, Z.; Zhang, M.; Li, Y. High Efficiency Nonfullerene Polymer Solar Cells with Thick Active Layer and Large Area. Adv. Mater. 2017, 29, 1702291. [Google Scholar] [CrossRef]
- Hwang, Y.-J.; Courtright, B.A.E.; Ferreira, A.S.; Tolbert, S.H.; Jenekhe, S.A. 7.7% Efficient All-Polymer Solar Cells. Adv. Mater. 2015, 27, 4578–4584. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Kim, J.-H.; Kang, T.E.; Lee, C.; Kang, H.; Shin, M.; Wang, C.; Ma, B.; Jeong, U.; Kim, T.-S.; et al. Flexible, highly efficient all-polymer solar cells. Nat. Commun. 2015, 6, 8547. [Google Scholar] [CrossRef]
- Liu, J.; Loewe, R.S.; McCullough, R.D. Employing MALDI-MS on Poly(alkylthiophenes): Analysis of Molecular Weights, Molecular Weight Distributions, End-Group Structures, and End-Group Modifications. Macromolecules 1999, 32, 5777–5785. [Google Scholar] [CrossRef]
- Nieuwendaal, R.C.; Snyder, C.R.; Delongchamp, D.M. Measuring Order in Regioregular Poly(3-hexylthiophene) with Solid-State13C CPMAS NMR. ACS Macro Lett. 2014, 3, 130–135. [Google Scholar] [CrossRef]
- Mulherin, R.C.; Jung, S.; Huettner, S.; Johnson, K.; Kohn, P.; Sommer, M.; Allard, S.; Scherf, U.; Greenham, N.C. Ternary Photovoltaic Blends Incorporating an All-Conjugated Donor–Acceptor Diblock Copolymer. Nano Lett. 2011, 11, 4846–4851. [Google Scholar] [CrossRef]
- Kochemba, W.M.; Kilbey, S.M.; Pickel, D.L. End-group composition of poly(3-hexylthiophene)s prepared by in situ quenching of the grignard metathesis polymerization: Influence of additives and reaction conditions. J. Polym. Sci. Part A Polym. Chem. 2012, 50, 2762–2769. [Google Scholar] [CrossRef]
- Lee, Y.; Aplan, M.P.; Seibers, Z.D.; Kilbey, S.M.; Wang, Q.; Gomez, E.D. Tuning the synthesis of fully conjugated block copolymers to minimize architectural heterogeneity. J. Mater. Chem. A 2017, 5, 20412–20421. [Google Scholar] [CrossRef]
- Lee, C.S.; Dadmun, M.D. Important thermodynamic characteristics of poly(3-hexyl thiophene). Polymers 2014, 55, 4–7. [Google Scholar] [CrossRef]
- Snyder, C.R.; Henry, J.S.; Delongchamp, D.M. Effect of Regioregularity on the Semicrystalline Structure of Poly(3-hexylthiophene). Macromolecules 2011, 44, 7088–7091. [Google Scholar] [CrossRef]
- Xie, R.; Lee, Y.; Aplan, M.P.; Caggiano, N.J.; Müller, C.; Colby, R.H.; Gomez, E.D. Glass Transition Temperature of Conjugated Polymers by Oscillatory Shear Rheometry. Macromolecules 2017, 50, 5146–5154. [Google Scholar] [CrossRef]
- Kim, J.-S.; Kim, J.-H.; Lee, W.; Yu, H.; Kim, H.J.; Song, I.; Shin, M.; Oh, J.H.; Jeong, U.; Kim, T.-S.; et al. Tuning Mechanical and Optoelectrical Properties of Poly(3-hexylthiophene) through Systematic Regioregularity Control. Macromolecules 2015, 48, 4339–4346. [Google Scholar] [CrossRef]
- Tsoi, W.C.; Spencer, S.J.; Yang, L.; Ballantyne, A.M.; Nicholson, P.G.; Turnbull, A.; Shard, A.G.; Murphy, C.E.; Bradley, D.D.C.; Nelson, J.; et al. Effect of Crystallization on the Electronic Energy Levels and Thin Film Morphology of P3HT:PCBM Blends. Macromolecules 2011, 44, 2944–2952. [Google Scholar] [CrossRef]
- Mauer, R.; Kastler, M.; Laquai, F. The Impact of Polymer Regioregularity on Charge Transport and Efficiency of P3HT:PCBM Photovoltaic Devices. Adv. Funct. Mater. 2010, 20, 2085–2092. [Google Scholar] [CrossRef]
- Müller, C. On the Glass Transition of Polymer Semiconductors and Its Impact on Polymer Solar Cell Stability. Chem. Mater. 2015, 27, 2740–2754. [Google Scholar] [CrossRef]
- Li, L.; Lu, G.; Yang, X.; Zhou, E. Progress in polymer solar cell. Chin. Sci. Bull. 2007, 52, 145–158. [Google Scholar] [CrossRef]
- Kim, J.Y.; Frisbie, C.D. Correlation of Phase Behavior and Charge Transport in Conjugated Polymer/Fullerene Blends. J. Phys. Chem. C 2008, 112, 17726–17736. [Google Scholar] [CrossRef]
- Hajduk, B.; Bednarski, H.; Trzebicka, B. Temperature-Dependent Spectroscopic Ellipsometry of Thin Polymer Films. J. Phys. Chem. B 2020, 124, 3229–3251. [Google Scholar] [CrossRef] [PubMed]
- Hajduk, B.; Bednarski, H.; Jarząbek, B.; Janeczek, H.; Nitschke, P. P3HT:PCBM blend films phase diagram on the base of variable-temperature spectroscopic ellipsometry. Beilstein J. Nanotechnol. 2018, 9, 1108–1115. [Google Scholar] [CrossRef]
- Yu, G.; Gao, J.; Hummelen, J.C.; Wudl, F.; Heeger, A.J. Polymer Photovoltaic Cells: Enhanced Efficiencies via a Network of Internal Donor-Acceptor Heterojunctions. Science 1995, 270, 1789–1791. [Google Scholar] [CrossRef]
- Hopkinson, P.E.; Staniec, P.A.; Pearson, A.J.; Dunbar, A.D.F.; Wang, T.; Ryan, A.J.; Jones, R.A.L.; Lidzey, D.G.; Donald, A.M. A Phase Diagram of the P3HT:PCBM Organic Photovoltaic System: Implications for Device Processing and Performance. Macromolecules 2011, 44, 2908–2917. [Google Scholar] [CrossRef]
- Ngo, T.T.; Nguyen, D.N.; Nguyen, V.T. Glass transition of PCBM, P3HT and their blends in quenched state. Adv. Nat. Sci. Nanosci. Nanotechnol. 2012, 3, 045001. [Google Scholar] [CrossRef]
- Brande, N.V.D.; Van Assche, G.; Van Mele, B. Thermal behaviour below and inside the glass transition region of a submicron P3HT layer studied by fast scanning chip calorimetry. Polymers 2016, 83, 59–66. [Google Scholar] [CrossRef]
- Beljonne, D.; Cornil, J.; Sirringhaus, H.; Brown, P.J.; Shkunov, M.; Friend, R.H.; Brédas, J.L. Optical Signature of Delocalized Polarons in Conjugated Polymers. Adv. Funct. Mater. 2001, 11, 229–234. [Google Scholar] [CrossRef]
- Shen, X.; Hu, W.; Russell, T.P. Measuring the Degree of Crystallinity in Semicrystalline Regioregular Poly(3-hexylthiophene). Macromolecules 2016, 49, 4501–4509. [Google Scholar] [CrossRef]
- Snyder, C.R.; Nieuwendaal, R.C.; Delongchamp, D.M.; Luscombe, C.K.; Sista, P.; Boyd, S.D. Quantifying Crystallinity in High Molar Mass Poly(3-hexylthiophene). Macromolecules 2014, 47, 3942–3950. [Google Scholar] [CrossRef]
- Balko, J.; Lohwasser, R.H.; Sommer, M.; Thelakkat, M.; Thurn-Albrecht, T. Determination of the Crystallinity of Semicrystalline Poly(3-hexylthiophene) by Means of Wide-Angle X-ray Scattering. Macromolecules 2013, 46, 9642–9651. [Google Scholar] [CrossRef]
- Kline, R.J.; McGehee, M.D.; Kadnikova, E.N.; Liu, J.; Fréchet, J.M.J.; Toney, M.F. Dependence of Regioregular Poly(3-hexylthiophene) Film Morphology and Field-Effect Mobility on Molecular Weight. Macromolecules 2005, 38, 3312–3319. [Google Scholar] [CrossRef]
- Zhang, R.; Li, B.; Iovu, M.C.; Jeffries-El, M.; Sauvé, G.; Cooper, J.; Jia, S.; Tristram-Nagle, S.; Smilgies, D.M.; Lambeth, D.N.; et al. Nanostructure Dependence of Field-Effect Mobility in Regioregular Poly(3-hexylthiophene) Thin Film Field Effect Transistors. J. Am. Chem. Soc. 2006, 128, 3480–3481. [Google Scholar] [CrossRef] [PubMed]
- Vakhshouri, K.; Smith, B.H.; Chan, E.P.; Wang, C.; Salleo, A.; Wang, C.; Hexemer, A.; Gomez, E.D. Signatures of Intracrystallite and Intercrystallite Limitations of Charge Transport in Polythiophenes. Macromolecules 2016, 49, 7359–7369. [Google Scholar] [CrossRef]
- Agostinelli, T.; Lilliu, S.; Labram, J.G.; Campoy-Quiles, M.; Hampton, M.; Pires, E.; Rawle, J.; Bikondoa, O.; Bradley, D.D.C.; Anthopoulos, T.D.; et al. Real-Time Investigation of Crystallization and Phase-Segregation Dynamics in P3HT:PCBM Solar Cells During Thermal Annealing. Adv. Funct. Mater. 2011, 21, 1701–1708. [Google Scholar] [CrossRef]
- Verploegen, E.; Miller, C.E.; Schmidt, K.; Bao, Z.; Toney, M.F. Manipulating the Morphology of P3HT–PCBM Bulk Heterojunction Blends with Solvent Vapor Annealing. Chem. Mater. 2012, 24, 3923–3931. [Google Scholar] [CrossRef]
- Baek, W.-H.; Yang, H.; Yoon, T.-S.; Kang, C.; Lee, H.H.; Kim, Y.-S. Effect of P3HT:PCBM concentration in solvent on performances of organic solar cells. Sol. Energy Mater. Sol. Cells 2009, 93, 1263–1267. [Google Scholar] [CrossRef]
- Zhang, W.; Gomez, E.D.; Milner, S.T. Predicting Chain Dimensions of Semiflexible Polymers from Dihedral Potentials. Macromolecules 2014, 47, 6453–6461. [Google Scholar] [CrossRef]
- McCulloch, B.; Ho, V.; Hoarfrost, M.; Stanley, C.; Do, C.; Heller, W.T.; Segalman, R.A. Polymer Chain Shape of Poly(3-alkylthiophenes) in Solution Using Small-Angle Neutron Scattering. Macromolecules 2013, 46, 1899–1907. [Google Scholar] [CrossRef]
- Noriega, R.; Rivnay, J.; Vandewal, K.; Koch, F.P.V.; Stingelin, N.; Smith, P.; Toney, M.F.; Salleo, A. A general relationship between disorder, aggregation and charge transport in conjugated polymers. Nat. Mater. 2013, 12, 1038–1044. [Google Scholar] [CrossRef]
- Gu, K.; Loo, Y. The Polymer Physics of Multiscale Charge Transport in Conjugated Systems. J. Polym. Sci. Part B Polym. Phys. 2019, 57, 1559–1571. [Google Scholar] [CrossRef]
- Kline, R.J.; McGehee, M.D.; Toney, M.F. Highly oriented crystals at the buried interface in polythiophene thin-film transistors. Nat. Mater. 2006, 5, 222–228. [Google Scholar] [CrossRef]
- Kim, D.H.; Jang, Y.; Park, Y.D.; Cho, K. Controlled One-Dimensional Nanostructures in Poly(3-hexylthiophene) Thin Film for High-Performance Organic Field-Effect Transistors. J. Phys. Chem. B 2006, 110, 15763–15768. [Google Scholar] [CrossRef] [PubMed]
- Jimison, L.H.; Toney, M.F.; McCulloch, I.; Heeney, M.; Salleo, A. Charge-Transport Anisotropy Due to Grain Boundaries in Directionally Crystallized Thin Films of Regioregular Poly(3-hexylthiophene). Adv. Mater. 2009, 21, 1568–1572. [Google Scholar] [CrossRef]
- Oconnor, B.T.; Kline, R.J.; Conrad, B.R.; Richter, L.J.; Gundlach, D.J.; Toney, M.F.; Delongchamp, D.M. Anisotropic Structure and Charge Transport in Highly Strain-Aligned Regioregular Poly(3-hexylthiophene). Adv. Funct. Mater. 2011, 21, 3697–3705. [Google Scholar] [CrossRef]
- O’Connor, B.T.; Reid, O.G.; Zhang, X.; Kline, R.J.; Richter, L.J.; Gundlach, D.J.; Delongchamp, D.M.; Toney, M.F.; Kopidakis, N.; Rumbles, G. Morphological Origin of Charge Transport Anisotropy in Aligned Polythiophene Thin Films. Adv. Funct. Mater. 2014, 24, 3422–3431. [Google Scholar] [CrossRef]
- Awartani, O.; Kudenov, M.W.; O’Connor, B.T. Organic photovoltaic cells with controlled polarization sensitivity. Appl. Phys. Lett. 2014, 104, 93306. [Google Scholar] [CrossRef]
- Yang, H.; Lefevre, S.W.; Ryu, C.Y.; Bao, Z. Solubility-driven thin film structures of regioregular poly(3-hexyl thiophene) using volatile solvents. Appl. Phys. Lett. 2007, 90, 172116. [Google Scholar] [CrossRef]
- Vohra, V.; Anzai, T. Molecular Orientation of Conjugated Polymer Chains in Nanostructures and Thin Films: Review of Processes and Application to Optoelectronics. J. Nanomater. 2017, 2017, 1–18. [Google Scholar] [CrossRef]
- Kim, D.H.; Jang, Y.; Park, Y.D.; Cho, K. Surface-Induced Conformational Changes in Poly(3-hexylthiophene) Monolayer Films. Langmuir 2005, 21, 3203–3206. [Google Scholar] [CrossRef] [PubMed]
- Jimison, L.H.; Himmelberger, S.; Duong, D.T.; Rivnay, J.; Toney, M.F.; Salleo, A. Vertical confinement and interface effects on the microstructure and charge transport of P3HT thin films. J. Polym. Sci. Part B Polym. Phys. 2013, 51, 611–620. [Google Scholar] [CrossRef]
- Brinkmann, M.; Wittmann, J.-C. Orientation of Regioregular Poly(3-hexylthiophene) by Directional Solidification: A Simple Method to Reveal the Semicrystalline Structure of a Conjugated Polymer. Adv. Mater. 2006, 18, 860–863. [Google Scholar] [CrossRef]
- Dörling, B.; Vohra, V.; Dao, T.T.; Garriga, M.; Murata, H.; Campoy-Quiles, M. Uniaxial macroscopic alignment of conjugated polymer systems by directional crystallization during blade coating. J. Mater. Chem. C 2014, 2, 3303–3310. [Google Scholar] [CrossRef]
- Brinkmann, M.; Hartmann, L.; Biniek, L.; Tremel, K.; Kayunkid, N. Orienting Semi-Conducting π-Conjugated Polymers. Macromol. Rapid Commun. 2014, 35, 9–26. [Google Scholar] [CrossRef]
- Kajiya, D.; Ozawa, S.; Koganezawa, T.; Saitow, K.-I. Enhancement of Out-of-plane Mobility in P3HT Film by Rubbing: Aggregation and Planarity Enhanced with Low Regioregularity. J. Phys. Chem. C 2015, 119, 7987–7995. [Google Scholar] [CrossRef]
- Aziz, M.Z.A.; Higashimine, K.; Shioya, N.; Shimoaka, T.; Hasegawa, T.; Sakai, H.; Vohra, V.; Murata, H. Controlling the concentration gradient in sequentially deposited bilayer organic solar cells via rubbing and annealing. RSC Adv. 2020, 10, 37529–37537. [Google Scholar] [CrossRef]
- Aryal, M.; Trivedi, K.; Hu, W. Nano-Confinement Induced Chain Alignment in Ordered P3HT Nanostructures Defined by Nanoimprint Lithography. ACS Nano 2009, 3, 3085–3090. [Google Scholar] [CrossRef]
- Zhou, M.; Aryal, M.; Mielczarek, K.; Zakhidov, A.; Hu, W. Hole mobility enhancement by chain alignment in nanoimprinted poly(3-hexylthiophene) nanogratings for organic electronics. J. Vac. Sci. Technol. B 2010, 28, C6M63–C6M67. [Google Scholar] [CrossRef]
- Soeda, J.; Matsui, H.; Okamoto, T.; Osaka, I.; Takimiya, K.; Takeya, J. Highly Oriented Polymer Semiconductor Films Compressed at the Surface of Ionic Liquids for High-Performance Polymeric Organic Field-Effect Transistors. Adv. Mater. 2014, 26, 6430–6435. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Pandey, M.; Nagamatsu, S.; Pandey, S.S.; Hayase, S.; Takashima, W. Casting Control of Floating-films into Ribbon-shape Structure by modified Dynamic FTM. J. Phys. Conf. Ser. 2017, 924, 012014. [Google Scholar] [CrossRef]
- Tripathi, A.S.; Gupta, R.K.; Sharma, S.; Nagamatsu, S.; Pandey, S.S. Molecular orientation and anisotropic charge transport in the large area thin films of regioregular Poly(3-alkylthiophenes) fabricated by ribbon-shaped FTM. Org. Electron. 2020, 81, 105687. [Google Scholar] [CrossRef]
- Awartani, O.; Kudenov, M.W.; Kline, R.J.; O’Connor, B.T. In-Plane Alignment in Organic Solar Cells to Probe the Morphological Dependence of Charge Recombination. Adv. Funct. Mater. 2015, 25, 1296–1303. [Google Scholar] [CrossRef]
- Gu, K.; Wang, Y.; Li, R.; Tsai, E.; Onorato, J.W.; Luscombe, C.K.; Priestley, R.D.; Loo, Y.-L. Role of Postdeposition Thermal Annealing on Intracrystallite and Intercrystallite Structuring and Charge Transport in Poly(3-hexylthiophene). ACS Appl. Mater. Interfaces 2021, 13, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Vakhshouri, K.; Kozub, D.R.; Wang, C.; Salleo, A.; Gomez, E.D. Effect of Miscibility and Percolation on Electron Transport in Amorphous Poly(3-Hexylthiophene)/Phenyl-C61-Butyric Acid Methyl Ester Blends. Phys. Rev. Lett. 2012, 108, 026601. [Google Scholar] [CrossRef] [PubMed]
- Kohn, P.; Rong, Z.; Scherer, K.H.; Sepe, A.; Sommer, M.; Müller-Buschbaum, P.; Friend, R.H.; Steiner, U.; Hüttner, S. Crystallization-Induced 10-nm Structure Formation in P3HT/PCBM Blends. Macromolecules 2013, 46, 4002–4013. [Google Scholar] [CrossRef]
- Sanyal, M.; Schmidt-Hansberg, B.; Klein, M.F.; Munuera, C.; Vorobiev, A.; Colsmann, A.; Scharfer, P.; Lemmer, U.; Schabel, W.; Dosch, H.; et al. Effect of Photovoltaic Polymer/Fullerene Blend Composition Ratio on Microstructure Evolution during Film Solidification Investigated in Real Time by X-ray Diffraction. Macromolecules 2011, 44, 3795–3800. [Google Scholar] [CrossRef]
- Pearson, A.J.; Wang, T.; Jones, R.A.L.; Lidzey, D.G.; Staniec, P.A.; Hopkinson, P.E.; Donald, A.M. Rationalizing Phase Transitions with Thermal Annealing Temperatures for P3HT:PCBM Organic Photovoltaic Devices. Macromolecules 2012, 45, 1499–1508. [Google Scholar] [CrossRef]
- Reisdorffer, F.; Haas, O.; Le Rendu, P.; Nguyen, T. Co-solvent effects on the morphology of P3HT:PCBM thin films. Synth. Met. 2012, 161, 2544–2548. [Google Scholar] [CrossRef]
- Chang, L.; Lademann, H.W.A.; Bonekamp, J.-B.; Meerholz, K.; Moulé, A.J. Effect of Trace Solvent on the Morphology of P3HT:PCBM Bulk Heterojunction Solar Cells. Adv. Funct. Mater. 2011, 21, 1779–1787. [Google Scholar] [CrossRef]
- Wang, W.; Song, L.; Magerl, D.; González, D.M.; Körstgens, V.; Philipp, M.; Moulin, J.-F.; Müller-Buschbaum, P. Influence of Solvent Additive 1,8-Octanedithiol on P3HT:PCBM Solar Cells. Adv. Funct. Mater. 2018, 28, 1800209. [Google Scholar] [CrossRef]
- Arca, F.; Loch, M.; Lugli, P. Enhancing Efficiency of Organic Bulkheterojunction Solar Cells by Using 1,8-Diiodooctane as Processing Additive. IEEE J. Photovolt. 2014, 4, 1560–1565. [Google Scholar] [CrossRef]
- Park, M.S.; Meresa, A.A.; Kwon, C.-M.; Kim, F.S. Selective Wet-Etching of Polymer/Fullerene Blend Films for Surface- and Nanoscale Morphology-Controlled Organic Transistors and Sensitivity-Enhanced Gas Sensors. Polymers 2019, 11, 1682. [Google Scholar] [CrossRef] [PubMed]
- Verploegen, E.; Mondal, R.; Bettinger, C.J.; Sok, S.; Toney, M.F.; Bao, Z. Effects of Thermal Annealing Upon the Morphology of Polymer-Fullerene Blends. Adv. Funct. Mater. 2010, 20, 3519–3529. [Google Scholar] [CrossRef]
- Li, G.; Shrotriya, V.; Huang, J.; Yao, Y.; Moriarty, T.; Emery, K.; Yang, Y. High-efficiency solution processable polymer photovoltaic cells by self-organization of polymer blends. Nat. Mater. 2005, 4, 864–868. [Google Scholar] [CrossRef]
- Ma, W.; Yang, C.; Gong, X.; Lee, K.; Heeger, A.J. Thermally Stable, Efficient Polymer Solar Cells with Nanoscale Control of the Interpenetrating Network Morphology. Adv. Funct. Mater. 2005, 15, 1617–1622. [Google Scholar] [CrossRef]
- Huq, A.F.; Karim, A. Comparative solvent quality dependent crystallization in solvent vapor annealing of P3HT:PCBM thin films by in-situ GIWAXS. Polymers 2019, 165, 101–111. [Google Scholar] [CrossRef]
- Hu, S.; Dyck, O.; Chen, H.; Hsiao, Y.-C.; Hu, B.; Duscher, G.; Dadmun, M.; Khomami, B. The impact of selective solvents on the evolution of structure and function in solvent annealed organic photovoltaics. RSC Adv. 2014, 4, 27931–27938. [Google Scholar] [CrossRef]
- Janssen, G.; Aguirre, A.; Goovaerts, E.; Vanlaeke, P.; Poortmans, J.; Manca, J. Optimization of morphology of P3HT/PCBM films for organic solar cells: Effects of thermal treatments and spin coating solvents. Eur. Phys. J. Appl. Phys. 2007, 37, 287–290. [Google Scholar] [CrossRef]
- Tournebize, A.; Rivaton, A.; Peisert, H.; Chassé, T. The Crucial Role of Confined Residual Additives on the Photostability of P3HT:PCBM Active Layers. J. Phys. Chem. C 2015, 119, 9142–9148. [Google Scholar] [CrossRef]
- O’Connor, T.F.; Zaretski, A.V.; Shiravi, B.A.; Savagatrup, S.; Printz, A.D.; Diaz, M.I.; Lipomi, D.J. Stretching and conformal bonding of organic solar cells to hemispherical surfaces. Energy Environ. Sci. 2014, 7, 370–378. [Google Scholar] [CrossRef]
- Khang, D.-Y.; Rogers, J.A.; Lee, H.H. Mechanical Buckling: Mechanics, Metrology, and Stretchable Electronics. Adv. Funct. Mater. 2009, 19, 1526–1536. [Google Scholar] [CrossRef]
- Zhang, S.; Ocheje, M.U.; Luo, S.; Ehlenberg, D.; Appleby, B.; Weller, D.; Zhou, D.; Rondeau-Gagné, S.; Gu, X. Probing the Viscoelastic Property of Pseudo Free-Standing Conjugated Polymeric Thin Films. Macromol. Rapid Commun. 2018, 39, e1800092. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Schrickx, H.; Balar, N.; Siddika, S.; Sheikh, N.; O’Connor, B.T. Unveiling the Stress–Strain Behavior of Conjugated Polymer Thin Films for Stretchable Device Applications. Macromolecules 2020, 53, 1988–1997. [Google Scholar] [CrossRef]
- Savagatrup, S.; Printz, A.D.; Wu, H.; Rajan, K.M.; Sawyer, E.J.; Zaretski, A.V.; Bettinger, C.J.; Lipomi, D.J. Viability of stretchable poly(3-heptylthiophene) (P3HpT) for organic solar cells and field-effect transistors. Synth. Met. 2015, 203, 208–214. [Google Scholar] [CrossRef]
- Savagatrup, S.; Printz, A.D.; Rodriquez, D.; Lipomi, D.J. Best of Both Worlds: Conjugated Polymers Exhibiting Good Photovoltaic Behavior and High Tensile Elasticity. Macromolecules 2014, 47, 1981–1992. [Google Scholar] [CrossRef]
- Rodriquez, D.; Kohl, J.G.; Morel, P.; Burrows, K.; Favaro, G.; Root, S.E.; Ramírez, J.; Alkhadra, M.A.; Carpenter, C.W.; Fei, Z.; et al. Measurement of Cohesion and Adhesion of Semiconducting Polymers by Scratch Testing: Effect of Side-Chain Length and Degree of Polymerization. ACS Macro Lett. 2018, 7, 1003–1009. [Google Scholar] [CrossRef]
- Mok, J.W.; Hu, Z.; Sun, C.; Barth, I.; Muñoz, R.; Jackson, J.; Terlier, T.; Yager, K.G.; Verduzco, R. Network-Stabilized Bulk Heterojunction Organic Photovoltaics. Chem. Mater. 2018, 30, 8314–8321. [Google Scholar] [CrossRef]
- Zhao, R.; Wang, N.; Yu, Y.; Liu, J. Organoboron Polymer for 10% Efficiency All-Polymer Solar Cells. Chem. Mater. 2020, 32, 1308–1314. [Google Scholar] [CrossRef]
- Genene, Z.; Mammo, W.; Wang, E.; Andersson, M.R. Recent Advances in n-Type Polymers for All-Polymer Solar Cells. Adv. Mater. 2019, 31, e1807275. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Melkonyan, F.S.; Facchetti, A.; Marks, T.J. All-Polymer Solar Cells: Recent Progress, Challenges, and Prospects. Angew. Chem. Int. Ed. 2019, 58, 4129–4142. [Google Scholar] [CrossRef]
- Kim, W.; Choi, J.; Kim, J.-H.; Kim, T.; Lee, C.; Lee, S.; Kim, M.; Kim, B.J.; Kim, T.-S. Comparative Study of the Mechanical Properties of All-Polymer and Fullerene–Polymer Solar Cells: The Importance of Polymer Acceptors for High Fracture Resistance. Chem. Mater. 2018, 30, 2102–2111. [Google Scholar] [CrossRef]
- Krebs, F.C.; Gevorgyan, S.A.; Alstrup, J. A roll-to-roll process to flexible polymer solar cells: Model studies, manufacture and operational stability studies. J. Mater. Chem. 2009, 19, 5442–5451. [Google Scholar] [CrossRef]
- Hoth, C.N.; Schilinsky, P.; Choulis, S.A.; Brabec, C.J. Printing Highly Efficient Organic Solar Cells. Nano Lett. 2008, 8, 2806–2813. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.H.; Park, H.; Mujawar, S.; Yoon, S.C.; Kim, S.-S.; Na, S.-I.; Kang, S.-J.; Khim, D.; Kim, D.-Y.; Lee, S.-H. High efficiency polymer solar cells via sequential inkjet-printing of PEDOT:PSS and P3HT:PCBM inks with additives. Org. Electron. 2010, 11, 1516–1522. [Google Scholar] [CrossRef]
- Lange, A.; Wegener, M.; Boeffel, C.; Fischer, B.; Wedel, A.; Neher, D. A new approach to the solvent system for inkjet-printed P3HT:PCBM solar cells and its use in devices with printed passive and active layers. Sol. Energy Mater. Sol. Cells 2010, 94, 1816–1821. [Google Scholar] [CrossRef]
- Aernouts, T.; Aleksandrov, T.; Girotto, C.; Genoe, J.; Poortmans, J. Polymer based organic solar cells using ink-jet printed active layers. Appl. Phys. Lett. 2008, 92, 033306. [Google Scholar] [CrossRef]
- Lange, A.; Wegener, M.; Fischer, B.; Janietz, S.; Wedel, A. Solar Cells with Inkjet Printed Polymer Layers. Energy Procedia 2012, 31, 150–158. [Google Scholar] [CrossRef]
- Hoth, C.N.; Choulis, S.A.; Schilinsky, P.; Brabec, C.J. On the effect of poly(3-hexylthiophene) regioregularity on inkjet printed organic solar cells. J. Mater. Chem. 2009, 19, 5398–5404. [Google Scholar] [CrossRef]
- Na, S.-I.; Yu, B.-K.; Kim, S.-S.; Vak, D.; Kim, T.-S.; Yeo, J.-S.; Kim, D.-Y. Fully spray-coated ITO-free organic solar cells for low-cost power generation. Sol. Energy Mater. Sol. Cells 2010, 94, 1333–1337. [Google Scholar] [CrossRef]
- Espinosa, N.; García-Valverde, R.; Urbina, A.; Krebs, F.C. A life cycle analysis of polymer solar cell modules prepared using roll-to-roll methods under ambient conditions. Sol. Energy Mater. Sol. Cells 2011, 95, 1293–1302. [Google Scholar] [CrossRef]
- Galagan, Y.; De Vries, I.G.; Langen, A.P.; Andriessen, R.; Verhees, W.J.; Veenstra, S.C.; Kroon, J.M. Technology development for roll-to-roll production of organic photovoltaics. Chem. Eng. Process. Process. Intensif. 2011, 50, 454–461. [Google Scholar] [CrossRef]
- Andersen, T.R.; Dam, H.F.; Hösel, M.; Helgesen, M.; Carlé, J.E.; Larsen-Olsen, T.T.; Gevorgyan, S.A.; Andreasen, J.W.; Adams, J.; Li, N.; et al. Scalable, ambient atmosphere roll-to-roll manufacture of encapsulated large area, flexible organic tandem solar cell modules. Energy Environ. Sci. 2014, 7, 2925–2933. [Google Scholar] [CrossRef]
- Carlé, J.E.; Helgesen, M.; Hagemann, O.; Hösel, M.; Heckler, I.M.; Bundgaard, E.; Gevorgyan, S.A.; Søndergaard, R.R.; Jørgensen, M.; García-Valverde, R.; et al. Overcoming the Scaling Lag for Polymer Solar Cells. Joule 2017, 1, 274–289. [Google Scholar] [CrossRef]
- Reinspach, J.A.; Diao, Y.; Giri, G.; Sachse, T.; England, K.; Zhou, Y.; Tassone, C.; Worfolk, B.J.; Presselt, M.; Toney, M.F.; et al. Tuning the Morphology of Solution-Sheared P3HT:PCBM Films. ACS Appl. Mater. Interfaces 2016, 8, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Yan, H.; Kurosawa, T.; Schroeder, B.C.; Gu, K.L.; Zhou, Y.; To, J.W.F.; Oosterhout, S.D.; Savikhin, V.; Molina-Lopez, F.; et al. Comparison of the Morphology Development of Polymer-Fullerene and Polymer-Polymer Solar Cells during Solution-Shearing Blade Coating. Adv. Energy Mater. 2016, 6, 1601225. [Google Scholar] [CrossRef]
- Gu, X.; Shaw, L.; Gu, K.; Toney, M.F.; Bao, Z. The meniscus-guided deposition of semiconducting polymers. Nat. Commun. 2018, 9, 1–16. [Google Scholar] [CrossRef]
- Zhang, S.; Alesadi, A.; Selivanova, M.; Cao, Z.; Qian, Z.; Luo, S.; Galuska, L.; Teh, C.; Ocheje, M.U.; Mason, G.T.; et al. Toward the Prediction and Control of Glass Transition Temperature for Donor–Acceptor Polymers. Adv. Funct. Mater. 2020, 30, 2002221. [Google Scholar] [CrossRef]
- Xie, R.; Weisen, A.R.; Lee, Y.; Aplan, M.A.; Fenton, A.M.; Masucci, A.E.; Kempe, F.; Sommer, M.; Pester, C.W.; Colby, R.H.; et al. Glass transition temperature from the chemical structure of conjugated polymers. Nat. Commun. 2020, 11, 1–8. [Google Scholar] [CrossRef]
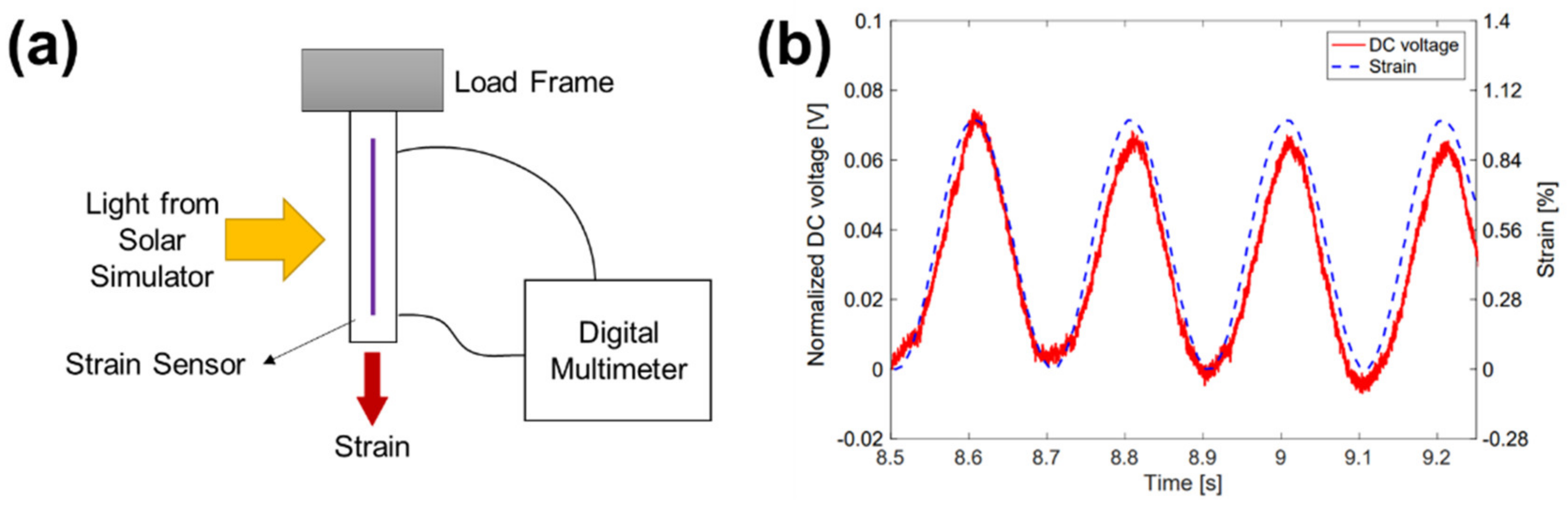
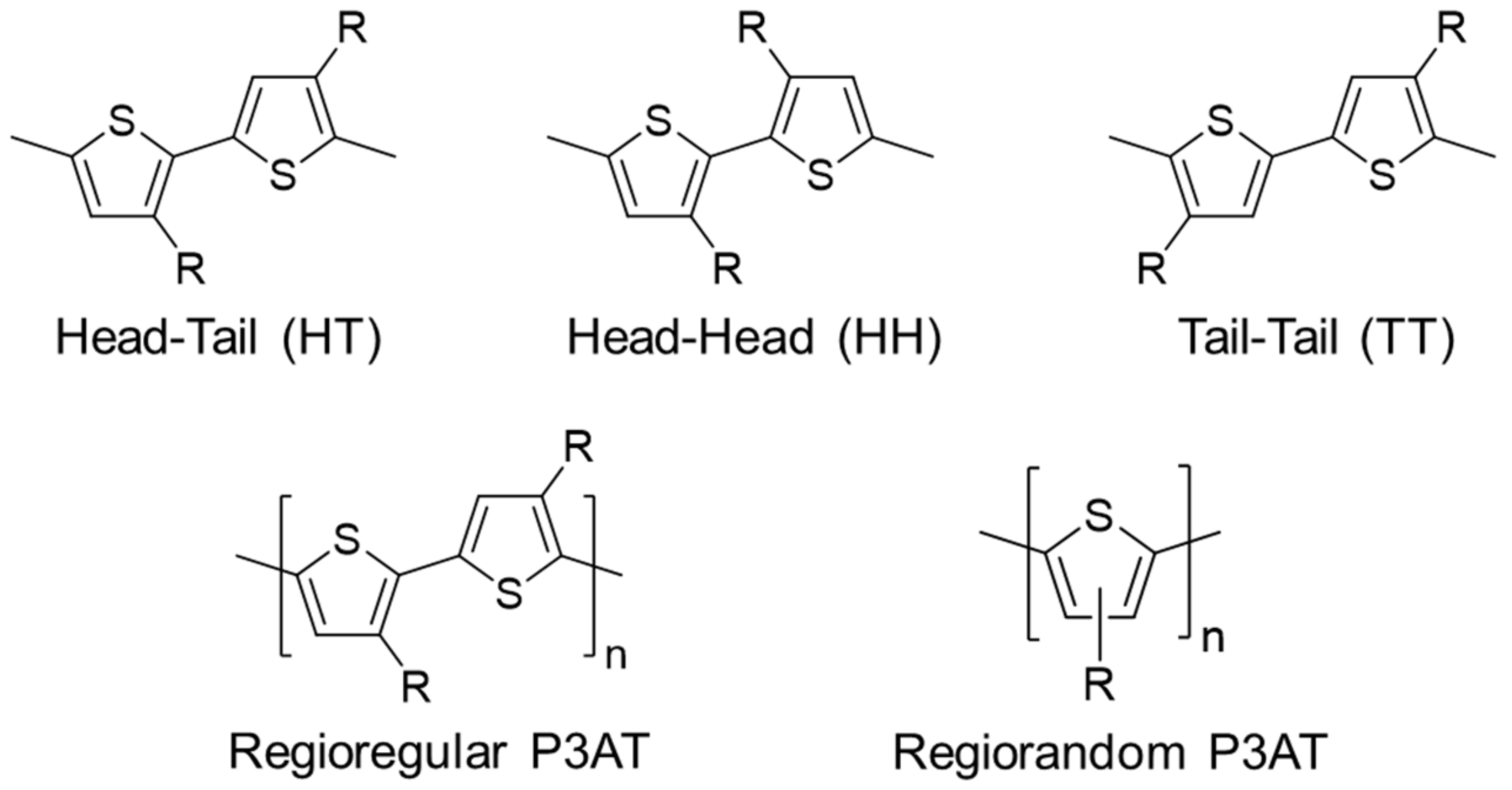
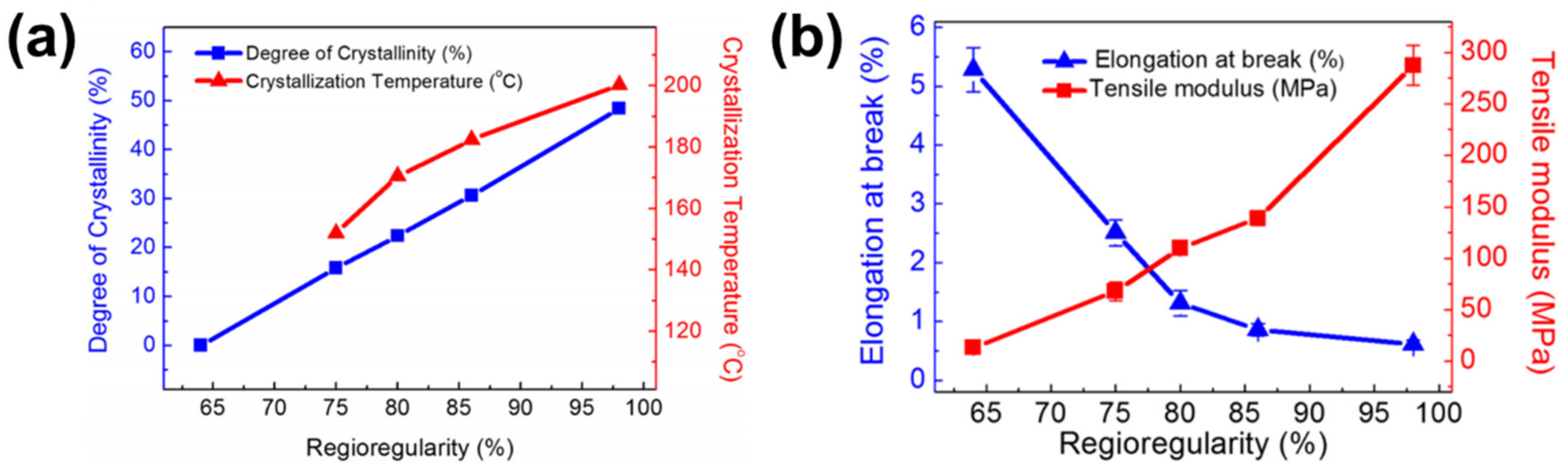
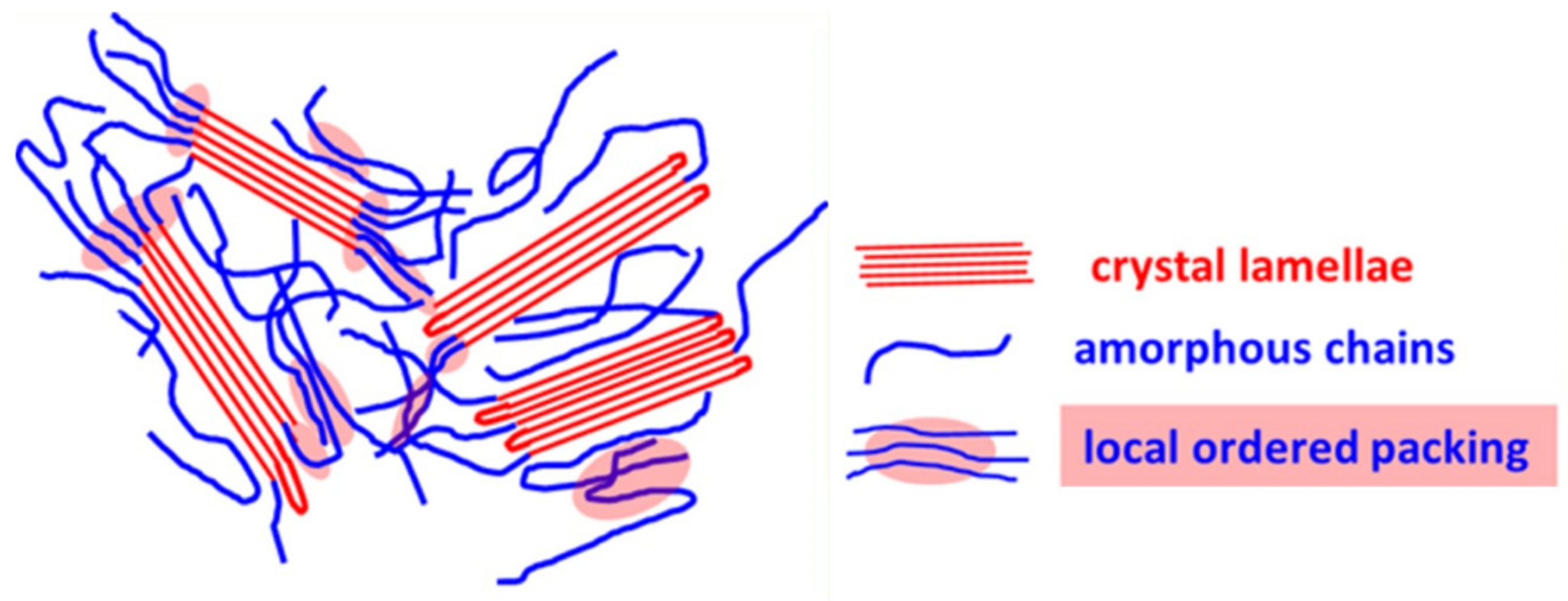

| JSC (mA/cm2) | VOC (V) | FF | PCE (%) | Ref. | |
|---|---|---|---|---|---|
| Normal architecture by roll-to-roll printing | 9.30 | 0.56 | 0.52 | 2.70 | [114] |
| Inverted architecture by roll-to-roll printing | 8.85 | 0.54 | 0.56 | 2.67 | [114] |
| Inkjet printing on ITO substrate | 10.05 | 0.54 | 0.64 | 3.47 | [115] |
| Spin coating as reference | 9.69 | 0.60 | 0.45 | 2.64 | [117] |
| P3HT/PCBM by inkjet printing | 9.34 | 0.57 | 0.45 | 2.40 | [117] |
| P3HT/PCBM and PEDOT:PSS by inkjet printing | 8.94 | 0.51 | 0.34 | 1.54 | [117] |
| Spray-coated P3HT/PCBM on ITO electrode | 8.06 | 0.59 | 0.60 | 2.86 | [121] |
| Fully spray-coated device with P3HT/PCBM on PEDOT:PSS electrode | 6.62 | 0.61 | 0.54 | 2.17 | [121] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.; Mongare, A.; Plant, A.; Ryu, D. Strain–Microstructure–Optoelectronic Inter-Relationship toward Engineering Mechano-Optoelectronic Conjugated Polymer Thin Films. Polymers 2021, 13, 935. https://doi.org/10.3390/polym13060935
Lee Y, Mongare A, Plant A, Ryu D. Strain–Microstructure–Optoelectronic Inter-Relationship toward Engineering Mechano-Optoelectronic Conjugated Polymer Thin Films. Polymers. 2021; 13(6):935. https://doi.org/10.3390/polym13060935
Chicago/Turabian StyleLee, Youngmin, Alfred Mongare, Aaron Plant, and Donghyeon Ryu. 2021. "Strain–Microstructure–Optoelectronic Inter-Relationship toward Engineering Mechano-Optoelectronic Conjugated Polymer Thin Films" Polymers 13, no. 6: 935. https://doi.org/10.3390/polym13060935
APA StyleLee, Y., Mongare, A., Plant, A., & Ryu, D. (2021). Strain–Microstructure–Optoelectronic Inter-Relationship toward Engineering Mechano-Optoelectronic Conjugated Polymer Thin Films. Polymers, 13(6), 935. https://doi.org/10.3390/polym13060935








