Assessing the Influence of Dyes Physico-Chemical Properties on Incorporation and Release Kinetics in Silk Fibroin Matrices
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Silk Fibroin Aqueous Solution
2.3. Preparation of Silk Fibroin Microparticles
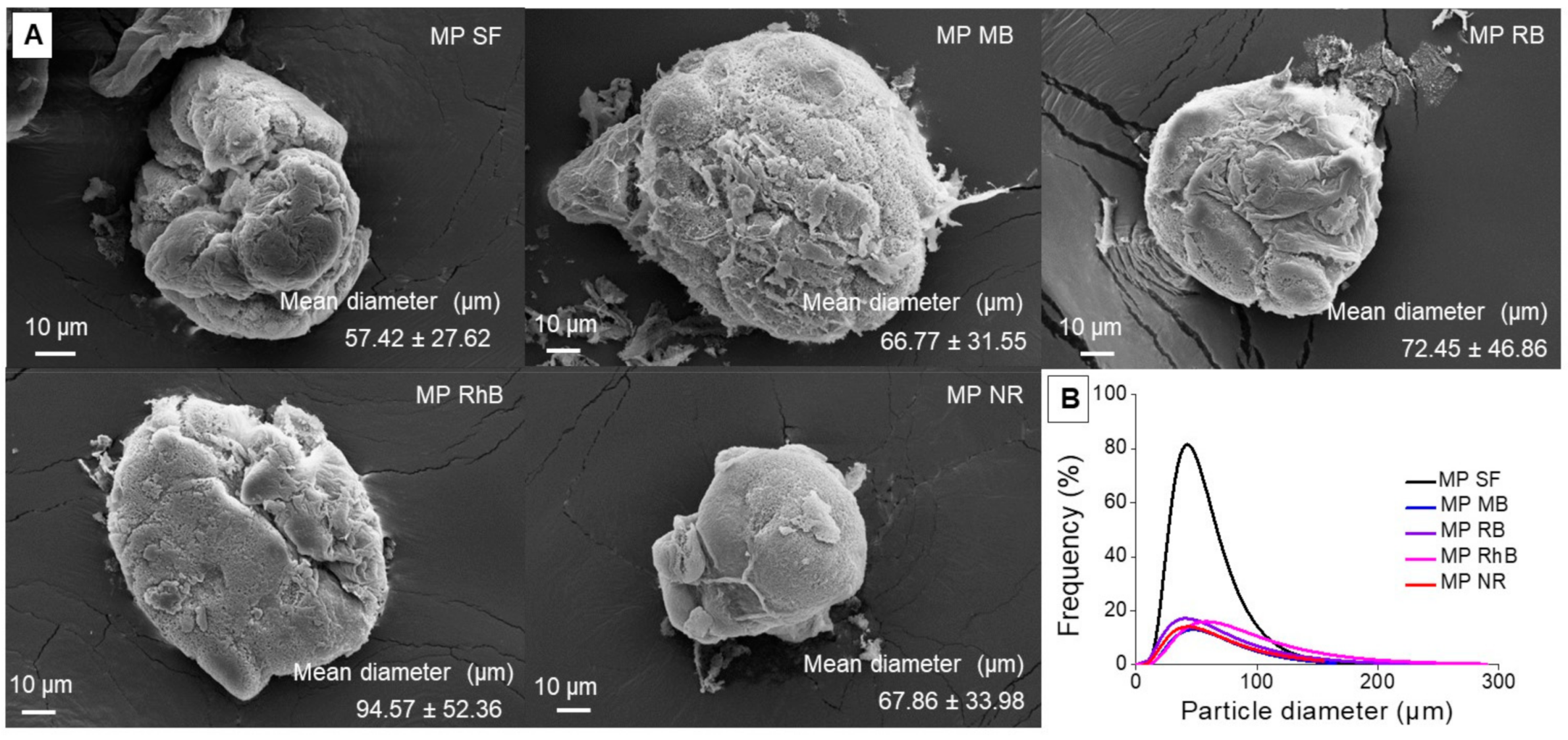
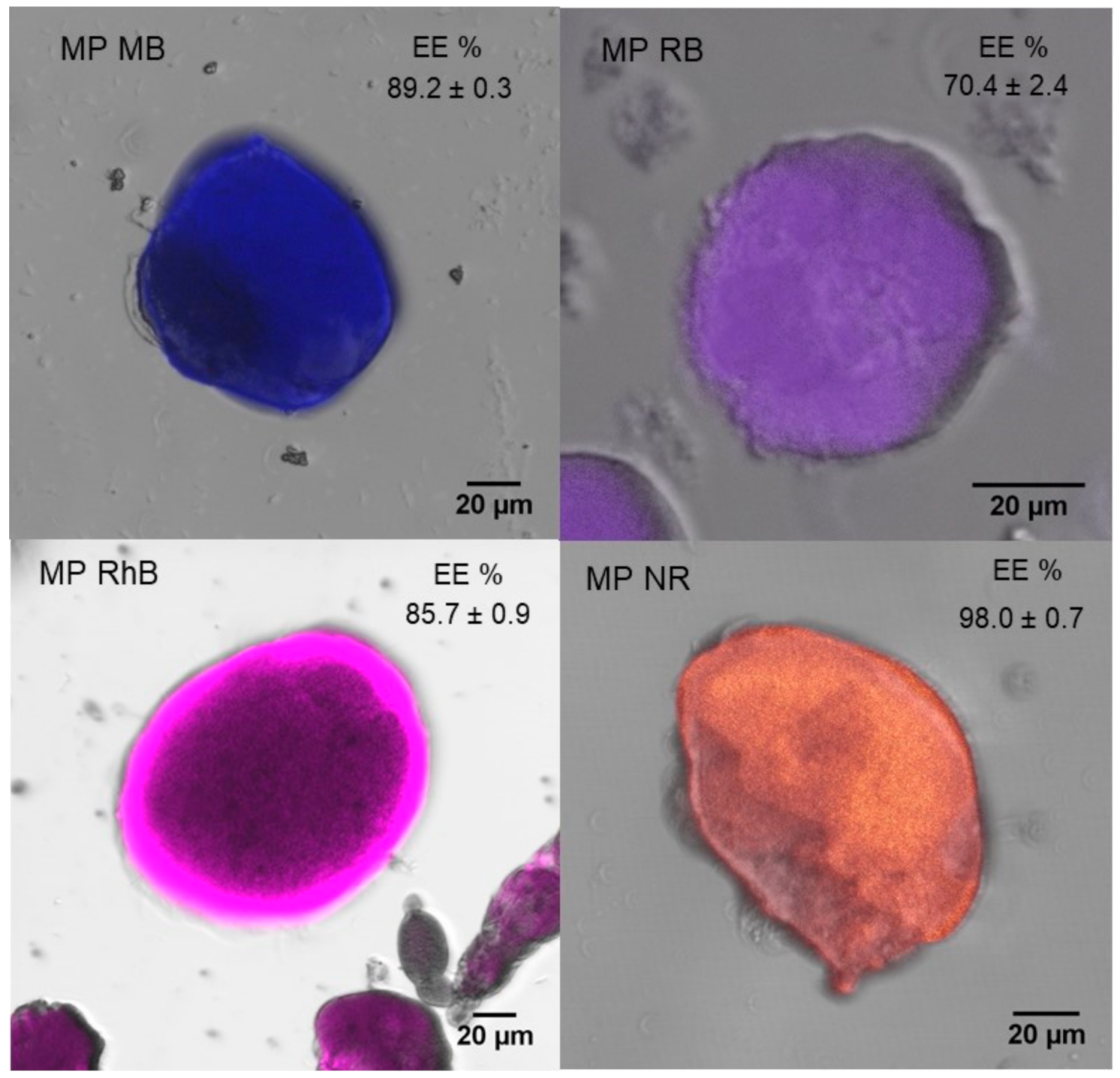
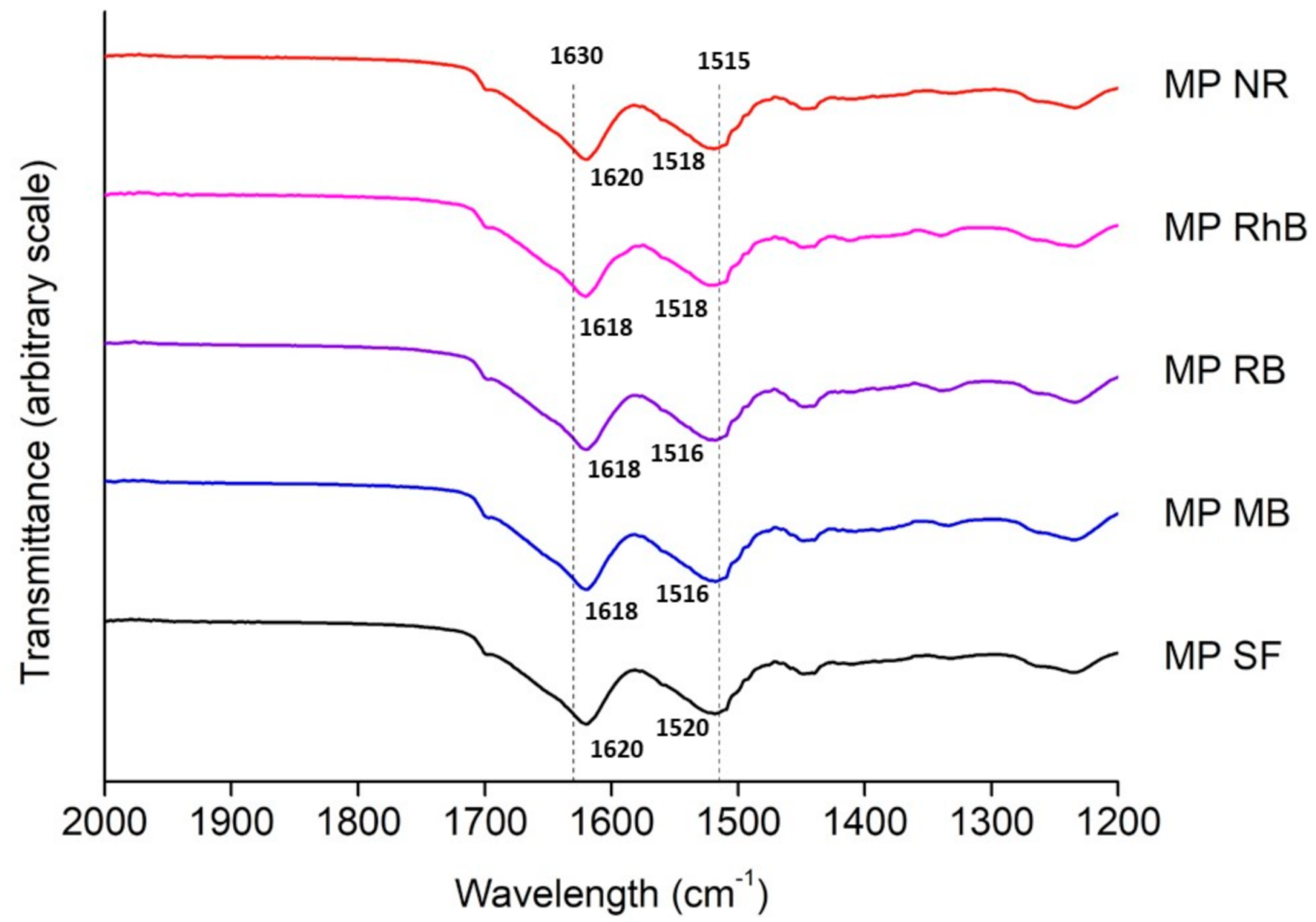
2.4. Microparticles Characterization
2.5. Preparation of Silk Fibroin Hydrogels
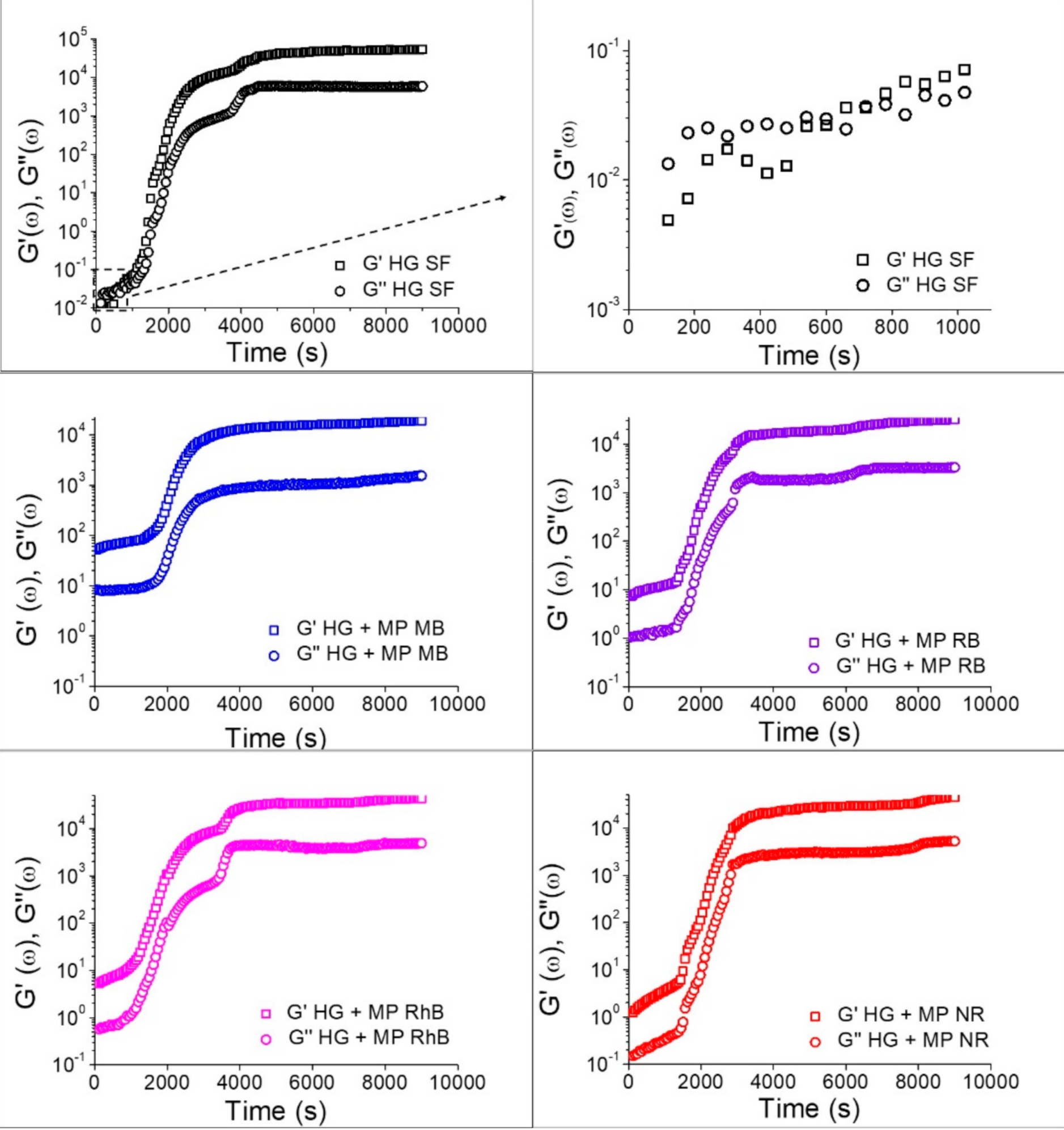
2.6. Hydrogel Characterization
2.7. Release Kinetics of the Dyes
3. Results and Discussion
3.1. Silk Fibroin Microparticles
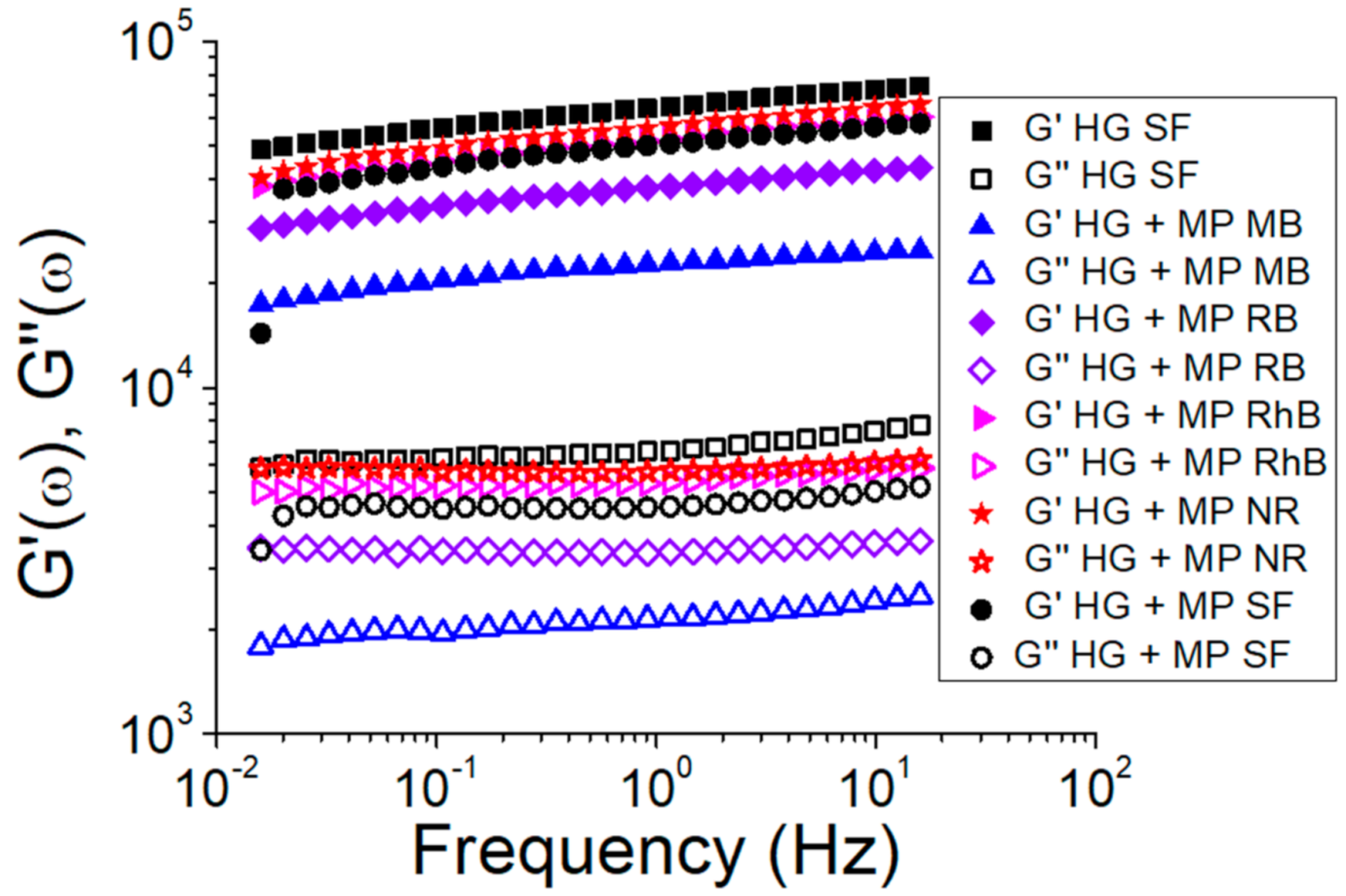
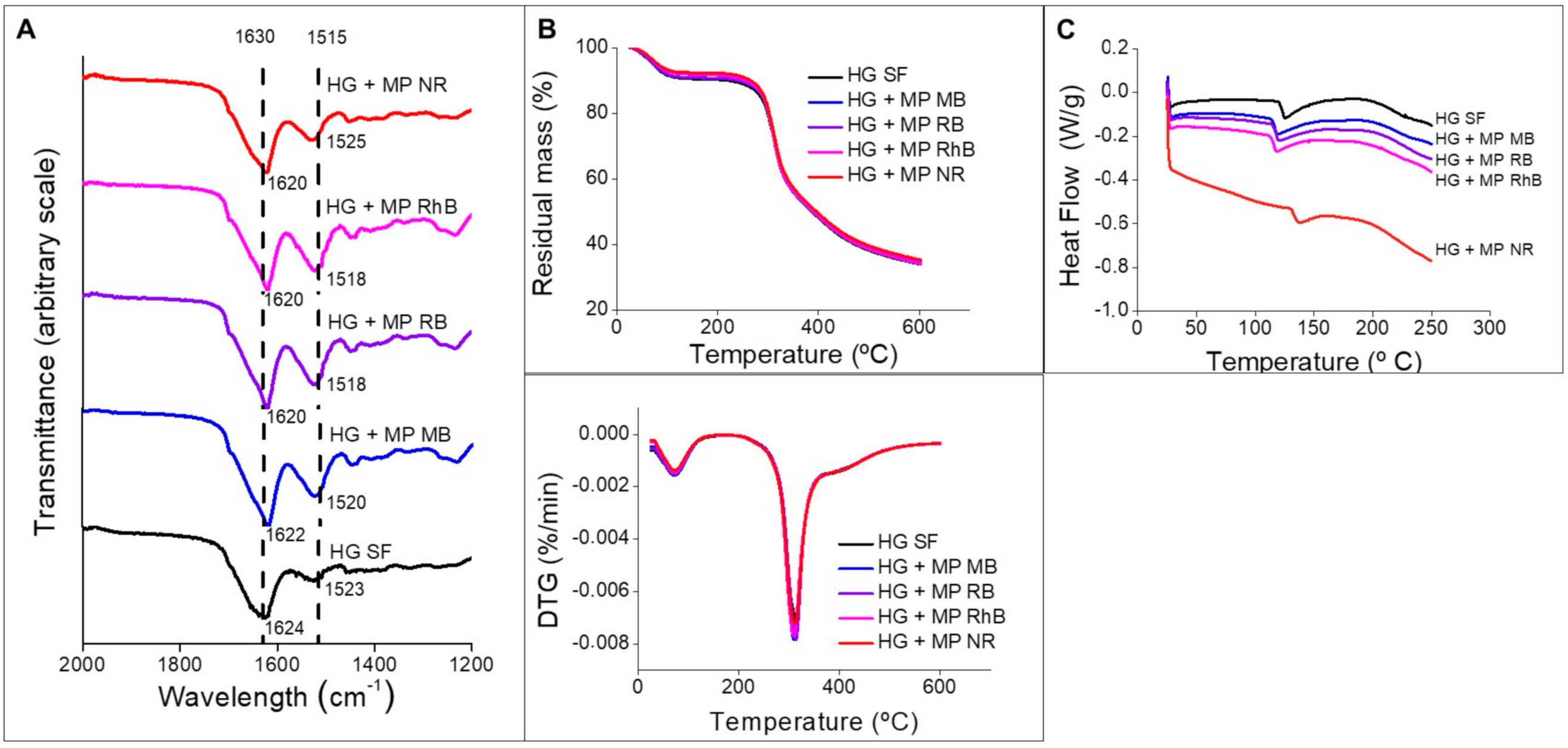
3.2. Silk Fibroin Hydrogels
3.3. Release Kinetics of the Dyes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24, 401–416. [Google Scholar] [CrossRef]
- Qi, Y.; Wang, H.; Wei, K.; Yang, Y.; Zheng, R.-Y.; Kim, I.S.; Zhang, K.-Q. A Review of Structure Construction of Silk Fibroin Biomaterials from Single Structures to Multi-Level Structures. Int. J. Mol. Sci. 2017, 18, 237. [Google Scholar] [CrossRef] [PubMed]
- Vepari, C.; Kaplan, D.L. Silk as a biomaterial. Prog. Polym. Sci. 2007, 32, 991–1007. [Google Scholar] [CrossRef]
- Koh, L.-D.; Cheng, Y.; Teng, C.-P.; Khin, Y.-W.; Loh, X.-J.; Tee, S.-Y.; Low, M.; Ye, E.; Yu, H.-D.; Zhang, Y.-W.; et al. Structures, mechanical properties and applications of silk fibroin materials. Prog. Polym. Sci. 2015, 46, 86–110. [Google Scholar] [CrossRef]
- Rockwood, D.N.; Preda, R.C.; Yücel, T.; Wang, X.; Lovett, M.L.; Kaplan, D.L. Materials fabrication from Bombyx mori silk fibroin. Nat. Protoc. 2011, 6, 1612–1631. [Google Scholar] [CrossRef]
- Wenk, E.; Merkle, H.P.; Meinel, L. Silk fibroin as a vehicle for drug delivery applications. J. Control. Release 2011, 150, 128–141. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Minoura, N.; Tanioka, A. Transport of pharmaceuticals through silk fibroin membrane. Polymer 1994, 35, 2853–2856. [Google Scholar] [CrossRef]
- Sashina, E.S.; Bochek, A.M.; Novoselov, N.P.; Kirichenko, D.A. Structure and Solubility of Natural Silk Fibroin. Russ. J. Appl. Chem. 2006, 79, 869–876. [Google Scholar] [CrossRef]
- Mandal, B.B.; Mann, J.K.; Kundu, S.C. Silk fibroin/gelatin multilayered films as a model system for controlled drug release. Eur. J. Pharm. Sci. 2009, 37, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Ratner, B.D.; Hoffman, A.S. Synthetic Hydrogels for Biomedical Applications. In Hydrogels for Medical and Related Applications; American Chemical Society: Washington, DC, USA, 1976; pp. 1–36. [Google Scholar]
- Nogueira, G.M.; De Moraes, M.A.; Rodas, A.C.D.; Higa, O.Z.; Beppu, M.M. Hydrogels from silk fibroin metastable solution: Formation and characterization from a biomaterial perspective. Mater. Sci. Eng. C 2011, 31, 997–1001. [Google Scholar] [CrossRef]
- Kasoju, N.; Hawkins, N.; Pop-Georgievski, O.; Kubies, D.; Vollrath, F. Silk fibroin gelation via non-solvent induced phase separation. Biomater. Sci. 2016, 4, 460–473. [Google Scholar] [CrossRef]
- De Moraes, M.A.; Mahl, C.R.A.; Silva, M.F.; Beppu, M.M. Formation of silk fibroin hydrogel and evaluation of its drug release profile. J. Appl. Polym. Sci. 2015, 132, 1–6. [Google Scholar] [CrossRef]
- Numata, K.; Yamazaki, S.; Naga, N. Biocompatible and Biodegradable Dual-Drug Release System Based on Silk Hydrogel Containing Silk Nanoparticles. Biomacromolecules 2012, 13, 1383–1389. [Google Scholar] [CrossRef]
- Suri, S.; Ruan, G.; Winter, J.; Schmidt, C.E. Microparticles and Nanoparticles. In Biomaterials Science; Elsevier: Amsterdam, The Netherlands, 2013; pp. 360–388. [Google Scholar]
- Wenk, E.; Wandrey, A.J.; Merkle, H.P.; Meinel, L. Silk fibroin spheres as a platform for controlled drug delivery. J. Control. Release 2008, 132, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Mwangi, T.K.; Bowles, R.D.; Tainter, D.M.; Bell, R.D.; Kaplan, D.L.; Setton, L.A. Synthesis and characterization of silk fibroin microparticles for intra-articular drug delivery. Int. J. Pharm. 2015, 485, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Numata, K.; Cebe, P.; Kaplan, D.L. Mechanism of enzymatic degradation of beta-sheet crystals. Biomaterials 2010, 31, 2926–2933. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhu, J.; Chen, S.; Jia, L.; Ma, Y. Fabrication of aqueous-based dual drug loaded silk fibroin electrospun nanofibers embedded with curcumin-loaded RSF nanospheres for drugs controlled release. RSC Adv. 2017, 7, 56550–56558. [Google Scholar] [CrossRef]
- Wu, P.; Liu, Q.; Wang, Q.; Qian, H.; Yu, L.; Liu, B.; Li, R. Novel silk fibroin nanoparticles incorporated silk fibroin hydrogel for inhibition of cancer stem cells and tumor growth. Int. J. Nanomed. 2018, 13, 5405–5418. [Google Scholar] [CrossRef]
- Gad, H.M.H.; El-Sayed, A.A. Activated carbon from agricultural by-products for the removal of Rhodamine-B from aqueous solution. J. Hazard. Mater. 2009, 168, 1070–1081. [Google Scholar] [CrossRef]
- Viganó, J.; Meirelles, A.A.D.; Náthia-Neves, G.; Baseggio, A.M.; Cunha, R.L.; Maróstica Junior, M.R.; Meireles, M.A.A.; Gurikov, P.; Smirnova, I.; Martínez, J. Impregnation of passion fruit bagasse extract in alginate aerogel microparticles. Int. J. Biol. Macromol. 2019, 155, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, T. Mechanism of Sustained-Action Medication. Theoretical Analysis of Rate of Release of Solid Drugs Dispersed in Solid Matrices. J. Pharm. Sci. 1963, 52, 1145–1149. [Google Scholar] [CrossRef]
- Ritger, P.L.; Peppas, N.A. A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J. Control. Release 1987, 5, 37–42. [Google Scholar] [CrossRef]
- Peppas, N.A.; Sahlin, J.J. A simple equation for the description of solute release. III. Coupling of diffusion and relaxation. Int. J. Pharm. 1989, 57, 169–172. [Google Scholar] [CrossRef]
- Kim, H.; Fassihi, R. Application of Binary Polymer System in Drug Release Rate Modulation. 2. Influence of Formulation Variables and Hydrodynamic Conditions on Release Kinetics. J. Pharm. Sci. 1997, 86, 323–328. [Google Scholar] [CrossRef]
- Hopfenberg, H.B. Controlled Release from Erodible Slabs, Cylinders, and Spheres. Control. Release Polym. Formul. 1976, 33, 26–32. [Google Scholar] [CrossRef]
- Perrechil, F.A.; Sato, A.C.K.; Cunha, R.L. κ-Carrageenan-sodium caseinate microgel production by atomization: Critical analysis of the experimental procedure. J. Food Eng. 2011, 104, 123–133. [Google Scholar] [CrossRef]
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, Microspheres, and Microcapsules for Advanced Drug Delivery. Sci. Pharm. 2019, 87, 20. [Google Scholar] [CrossRef]
- Baimark, Y.; Srihanam, P.; Srisuwan, Y.; Phinyocheep, P. Preparation of Porous Silk Fibroin Microparticles by a Water-in-Oil Emulsification-Diffusion Method. J. Appl. Polym. Sci. 2010, 118, 1127–1133. [Google Scholar] [CrossRef]
- Dyakonov, T.; Yang, C.H.; Bush, D.; Gosangari, S.; Majuru, S.; Fatmi, A. Design and Characterization of a Silk-Fibroin-Based Drug Delivery Platform Using Naproxen as a Model Drug. J. Drug Deliv. 2012, 2012, 490514. [Google Scholar] [CrossRef]
- Lammel, A.S.; Hu, X.; Park, S.H.; Kaplan, D.L.; Scheibel, T.R. Controlling silk fibroin particle features for drug delivery. Biomaterials 2010, 31, 4583–4591. [Google Scholar] [CrossRef] [PubMed]
- Rusa, C.C.; Bridges, C.; Ha, S.; Tonelli, A.E. Conformational Changes Induced in Bombyx mori Silk Fibroin by Cyclodextrin Inclusion Complexation. Macromolecules 2005, 38, 5640–5646. [Google Scholar] [CrossRef]
- Silva, M.F.; De Moraes, M.A.; Nogueira, G.M.; Rodas, A.C.D.; Higa, O.Z.; Beppu, M.M. Glycerin and Ethanol as Additives on Silk Fibroin Films: Insoluble and Malleable Films. J. Appl. Pol. 2012, 128, 115–122. [Google Scholar] [CrossRef]
- Matsumoto, A.; Chen, J.; Collette, A.L.; Kim, U.J.; Altman, G.H.; Cebe, P.; Kaplan, D.L. Mechanisms of silk fibroin sol-gel transitions. J. Phys. Chem. B 2006, 110, 21630–21638. [Google Scholar] [CrossRef]
- Kim, U.J.; Park, J.; Li, C.; Jin, H.J.; Valluzzi, R.; Kaplan, D.L. Structure and properties of silk hydrogels. Biomacromolecules 2004, 5, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Yucel, T.; Cebe, P.; Kaplan, D.L. Vortex-Induced Injectable Silk Fibroin Hydrogels. Biophys. J. 2009, 97, 2044–2050. [Google Scholar] [CrossRef]
- Steffe, J.F. Rheological Methods in Food Process Engineering, 2nd ed.; Freeman Press: Walled Lake, MI, USA, 1996; ISBN 0963203614. [Google Scholar]
- Nejadnik, M.R.; Yang, X.; Bongio, M.; Alghamdi, H.S.; Van den Beucken, J.J.J.P.; Huysmans, M.C.; Jansen, J.A.; Hilborn, J.; Ossipov, D.; Leeuwenburgh, S.C.G. Self-healing hybrid nanocomposites consisting of bisphosphonated hyaluronan and calcium phosphate nanoparticles. Biomaterials 2014, 35, 6918–6929. [Google Scholar] [CrossRef] [PubMed]
- Braga, A.L.M.; Cunha, R.L. The effects of xanthan conformation and sucrose concentration on the rheological properties of acidified sodium caseinate-xanthan gels. Food Hydrocoll. 2004, 18, 977–986. [Google Scholar] [CrossRef]
- Ribeiro, M.; de Moraes, M.A.; Beppu, M.M.; Monteiro, F.J.; Ferraz, M.P. The role of dialysis and freezing on structural conformation, thermal properties and morphology of silk fibroin hydrogels. Biomatter 2014, 4, e28536. [Google Scholar] [CrossRef] [PubMed]
- Magoshi, J.; Nakamura, S. Studies on Physical Properties and Structure of Silk. Glass Transition and Crystallization of Silk Fibroin. J. Appl. Polym. Sci. 1975, 19, 1013–1015. [Google Scholar] [CrossRef]
- Siepmann, J.; Siepmann, F. Mathematical modeling of drug delivery. Int. J. Pharm. 2008, 364, 328–343. [Google Scholar] [CrossRef] [PubMed]


| Dye | Hydrophilicity | Solubility in Water (mg/mL) * | Molar Mass (g/mol) * | pKa ** |
|---|---|---|---|---|
| MB | Hydrophilic | 50 | 319.85 | 3.14 |
| RB | Hydrophilic | 100 | 973.67 | 4.7 |
| RhB | Hydrophobic | 7.8 [22] | 479.02 | 3.7 |
| NR | Hydrophobic | 10 | 288.78 | 6.8 |
| Model | Equation | Parameters | MP MB | MP RB | MP RhB | MP NR |
|---|---|---|---|---|---|---|
| Peppas | K (1/sn) | 0.0238 | 0.0054 | 0.0128 | 0.0050 | |
| n | 0.4500 | 0.4504 | 0.4500 | 0.4500 | ||
| R² | 0.9009 | 0.9250 | 0.9799 | 0.9656 | ||
| Peppas–Sahlin | K1 (1/sn) | 0.0224 | 0.0050 | 0.0129 | 0.0050 | |
| K2 (1/s2n) | 0.0000 | 1.48 × 10−5 | 0.0000 | 0.0000 | ||
| n | 0.4500 | 0.4500 | 0.4500 | 0.4500 | ||
| R² | 0.9067 | 0.9083 | 0.9688 | 0.9531 | ||
| Higuchi | Kh (1/s0.5) | 0.0157 | 0.0037 | 0.0091 | 0.0035 | |
| R² | 0.9154 | 0.9346 | 0.9739 | 0.9593 | ||
| Burst release | K (1/sn) | 0.0206 | 0.0043 | 0.0124 | 0.0048 | |
| n | 0.4500 | 0.4748 | 0.4500 | 0.4500 | ||
| B | 0.0422 | 0.0055 | 0.0071 | 0.0035 | ||
| R² | 0.9235 | 0.9076 | 0.9718 | 0.9564 |
| Model | Equation | Parameters | HG + MP MB | HG + MP RB | HG + MP RhB | HG + MP NR |
|---|---|---|---|---|---|---|
| Peppas | K (1/sn) | 0.002 | 0.002 | 0.012 | 0.001 | |
| n | 0.813 | 0.890 | 0.483 | 0.890 | ||
| R² | 0.960 | 0.928 | 0.914 | 0.844 | ||
| Peppas–Sahlin | K1 (1/sn) | 0.002 | - | - | 0.000 | |
| K2 (1/s2n) | 0.001 | - | - | 3.81 × 10−4 | ||
| n | 0.450 | - | - | 0.527 | ||
| R² | 0.949 | - | - | 0.863 | ||
| Hopfenberg | k0 (cm²/mol.s) | 0.000 | 0.000 | - | - | |
| R² | 0.953 | 0.943 | - | - | ||
| Higuchi | Kh (1/s0.5) | 0.013 | 0.016 | 0.011 | 0.011 | |
| R² | 0.845 | 0.768 | 0.920 | 0.690 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomoda, B.T.; Pacheco, M.S.; Abranches, Y.B.; Viganó, J.; Perrechil, F.; De Moraes, M.A. Assessing the Influence of Dyes Physico-Chemical Properties on Incorporation and Release Kinetics in Silk Fibroin Matrices. Polymers 2021, 13, 798. https://doi.org/10.3390/polym13050798
Tomoda BT, Pacheco MS, Abranches YB, Viganó J, Perrechil F, De Moraes MA. Assessing the Influence of Dyes Physico-Chemical Properties on Incorporation and Release Kinetics in Silk Fibroin Matrices. Polymers. 2021; 13(5):798. https://doi.org/10.3390/polym13050798
Chicago/Turabian StyleTomoda, Bruno Thorihara, Murilo Santos Pacheco, Yasmin Broso Abranches, Juliane Viganó, Fabiana Perrechil, and Mariana Agostini De Moraes. 2021. "Assessing the Influence of Dyes Physico-Chemical Properties on Incorporation and Release Kinetics in Silk Fibroin Matrices" Polymers 13, no. 5: 798. https://doi.org/10.3390/polym13050798
APA StyleTomoda, B. T., Pacheco, M. S., Abranches, Y. B., Viganó, J., Perrechil, F., & De Moraes, M. A. (2021). Assessing the Influence of Dyes Physico-Chemical Properties on Incorporation and Release Kinetics in Silk Fibroin Matrices. Polymers, 13(5), 798. https://doi.org/10.3390/polym13050798








