Polyvinylidene Fluoride Membrane Via Vapour Induced Phase Separation for Oil/Water Emulsion Filtration
Abstract
1. Introduction
2. Materials and Methods
2.1. Membrane Fabrication
2.2. Membrane Characterization
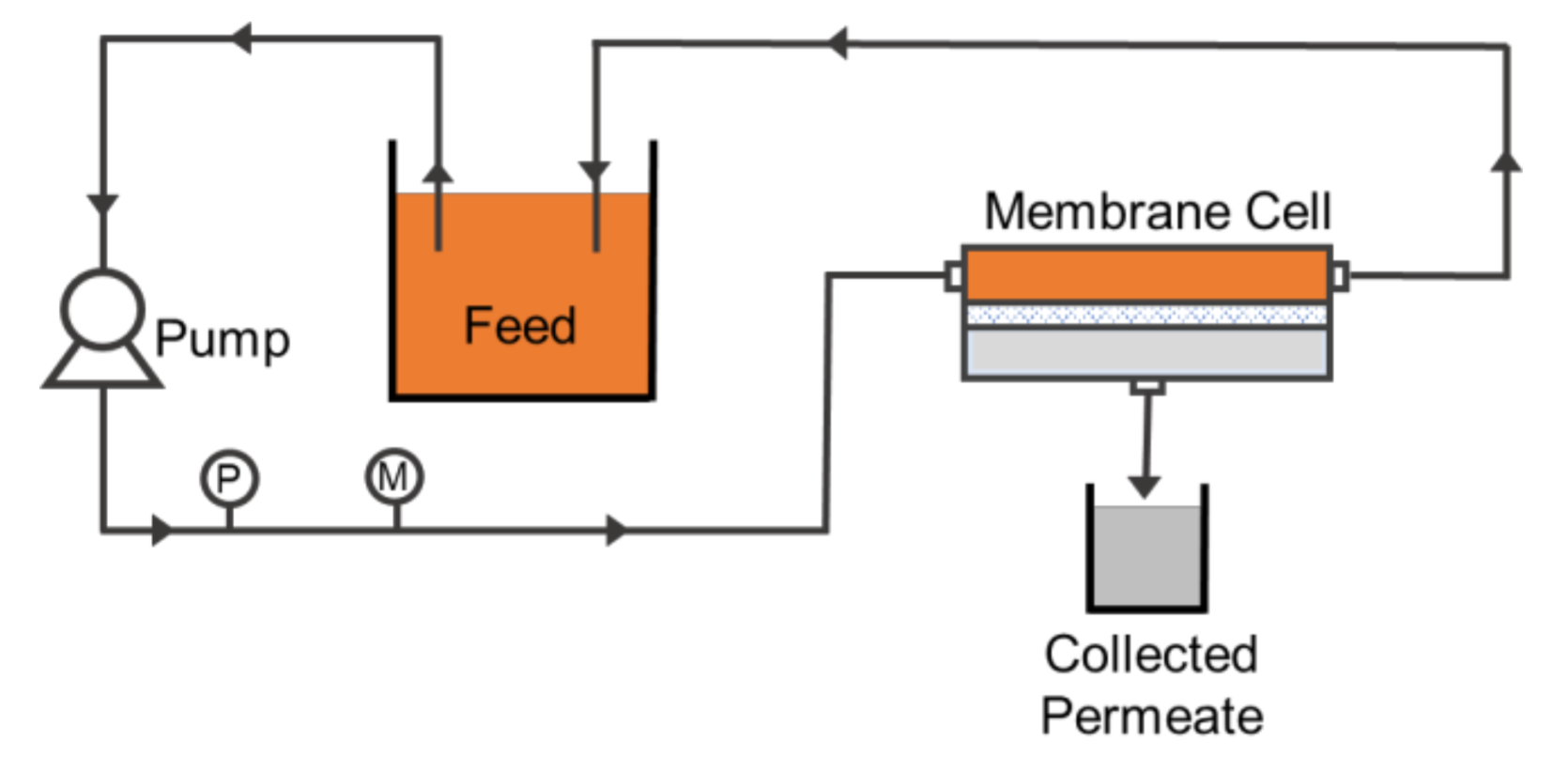
2.3. Filtration Configuration
2.4. Anti Fouling Analysis
3. Results and Discussion
3.1. Membrane Characterization
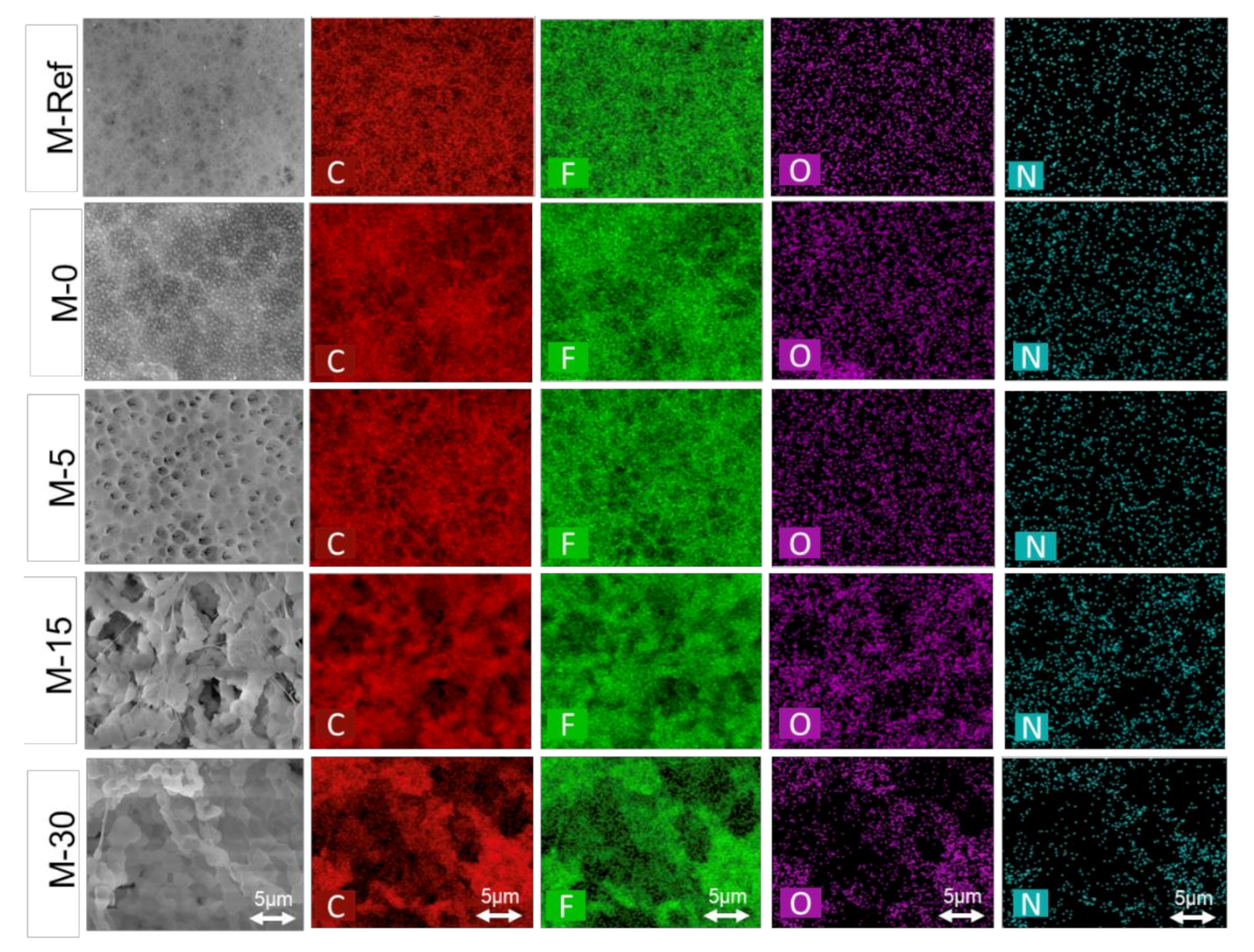
3.1.1. Membrane Morphology
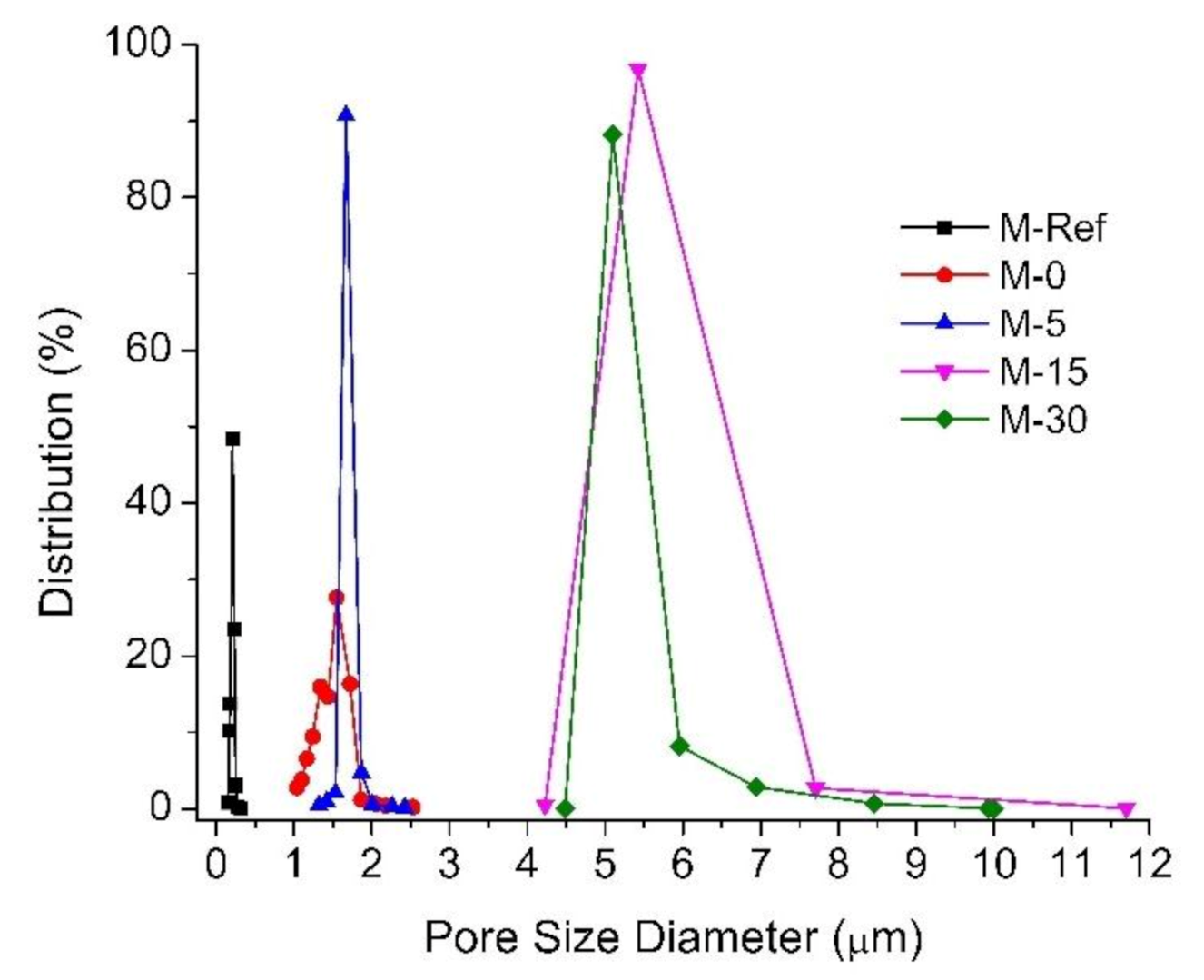
3.1.2. Membrane Pore Size and Distribution
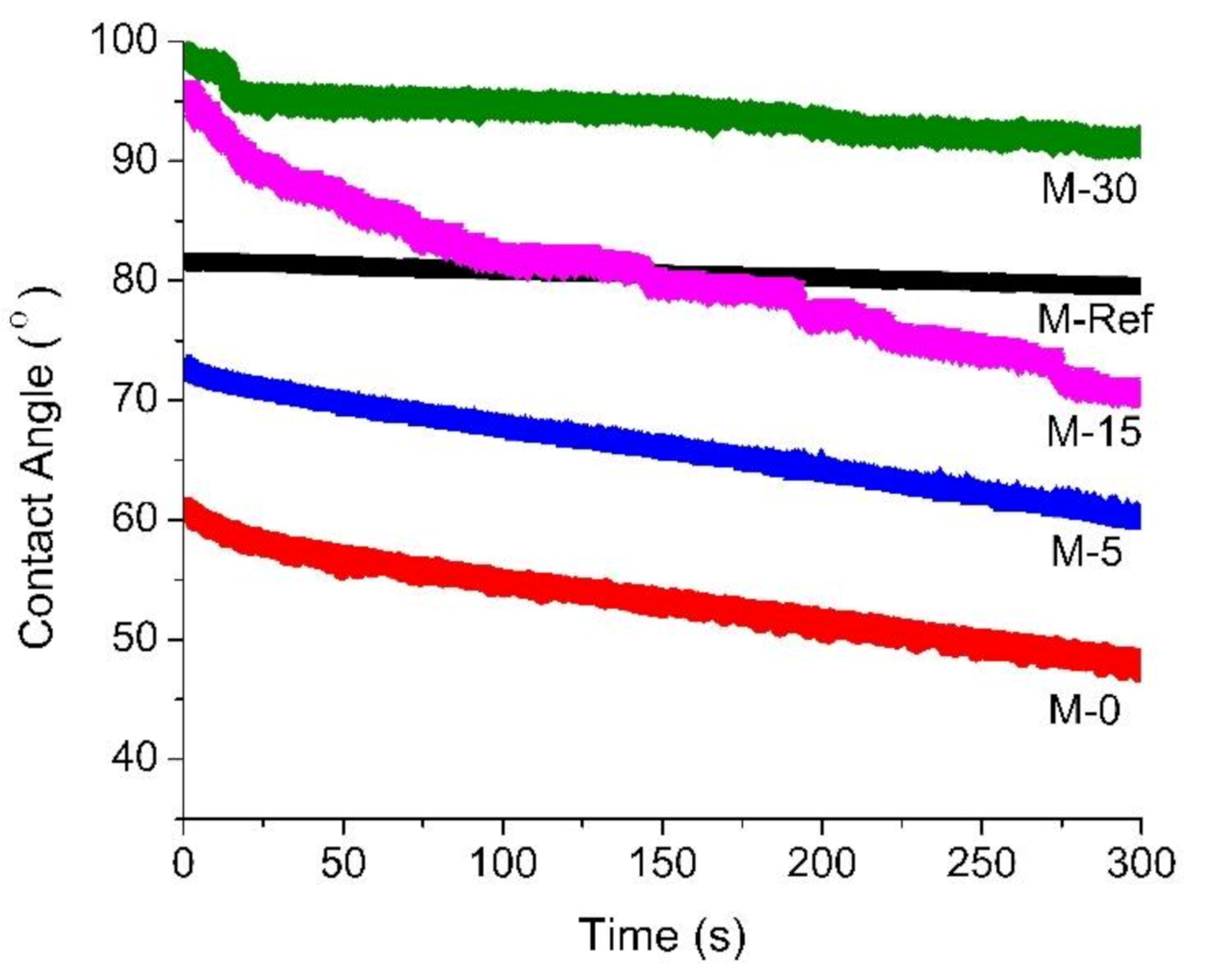
3.1.3. Surface Contact Angle
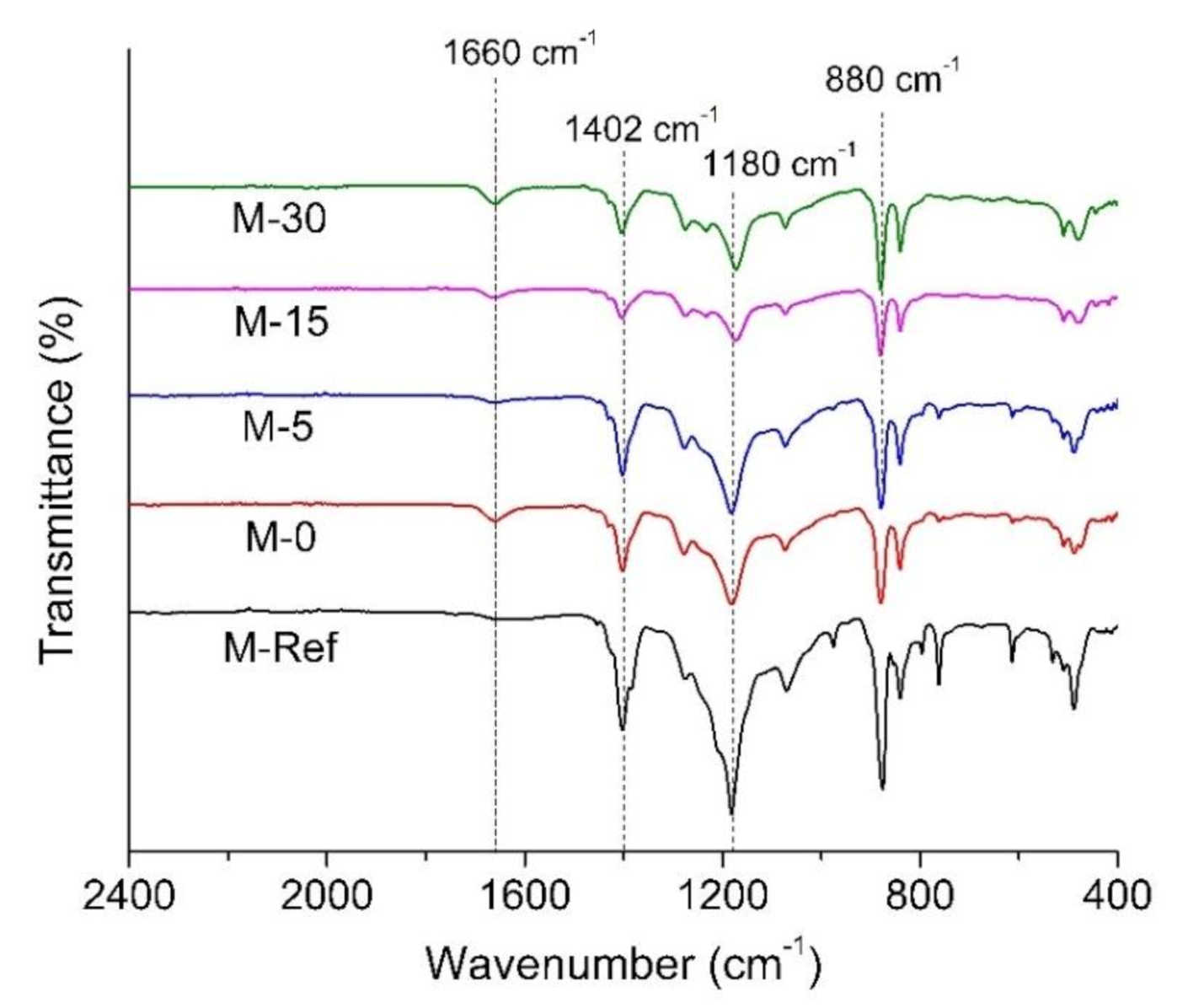
3.1.4. Fourier Transform Infrared (FTIR)
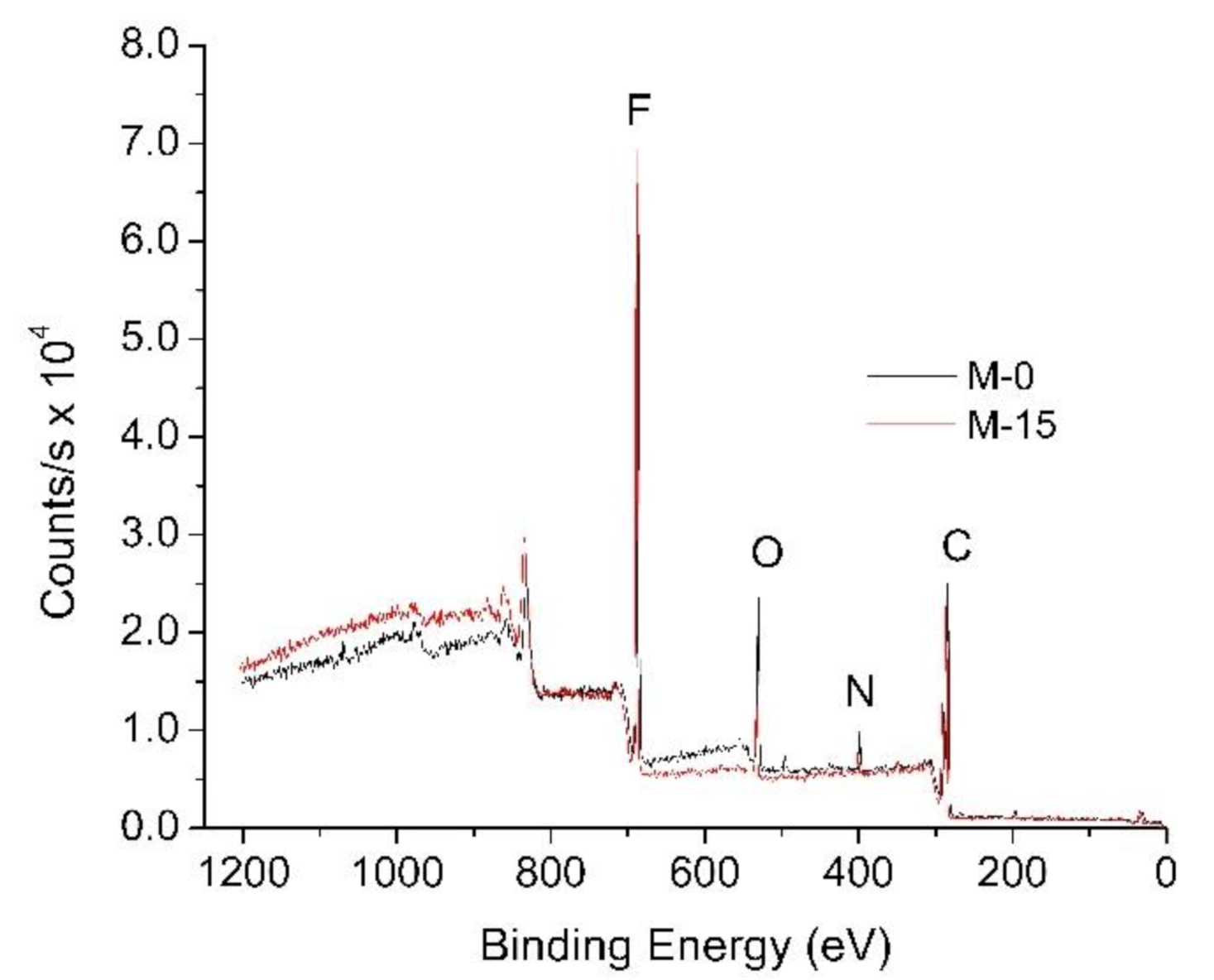
3.1.5. Surface Chemical Composition
3.1.6. Clean Water Permeability
3.2. Effect of Exposure Time on Membrane Hydraulic Performance
3.2.1. Permeance Recovery Analysis
3.2.2. Fouling Resistance Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Padaki, M.; Murali, R.S.; Abdullah, M.S.; Misdan, N.; Moslehyani, A.; Kassim, M.; Hilal, N.; Ismail, A. Membrane technology enhancement in oil–water separation. A review. Desalination 2015, 357, 197–207. [Google Scholar] [CrossRef]
- Kamyab, H.; Chelliapan, S.; Din, M.F.M.; Rezania, S.; Khademi, T.; Kumar, A. Palm oil mill effluent as an environmental pollutant. Palm Oil 2018, 13. [Google Scholar] [CrossRef]
- Aziz, M.M.A.; Kassim, K.A.; ElSergany, M.; Anuar, S.; Jorat, M.E.; Yaacob, H.; Ahsan, A.; Imteaz, M.A. Recent advances on palm oil mill effluent (POME) pretreatment and anaerobic reactor for sustainable biogas production. Renew. Sustain. Energy Rev. 2020, 119, 109603. [Google Scholar] [CrossRef]
- Iskandar, M.J.; Baharum, A.; Anuar, F.H.; Othaman, R. Palm oil industry in South East Asia and the effluent treatment technology—A review. Environ. Technol. Innov. 2018, 9, 169–185. [Google Scholar] [CrossRef]
- Yu, L.; Han, M.; He, F. A review of treating oily wastewater. Arab. J. Chem. 2017, 10, S1913–S1922. [Google Scholar] [CrossRef]
- Annuar, A.M.; Nawi, N.I.M.; Bilad, M.R.; Jaafar, J.; Marbelia, L.; Nandianto, A.B.D. Improved bubbling for membrane fouling control in filtration of palm oil mill effluent anaerobic digester sludge. J. Water Process Eng. 2020, 36, 101350. [Google Scholar] [CrossRef]
- Hafyan, R.H.; Bhullar, L.K.; Mahadzir, S.; Bilad, M.R.; Nordin, N.A.H.; Wirzal, M.D.H.; Putra, Z.A.; Rangaiah, G.P.; Abdullah, B. Integrated biorefinery of empty fruit bunch from palm oil industries to produce valuable biochemicals. Processes 2020, 8, 868. [Google Scholar] [CrossRef]
- Ahmad, T.; Guria, C.; Mandal, A. A review of oily wastewater treatment using ultrafiltration membrane: A parametric study to enhance the membrane performance. J. Water Process Eng. 2020, 36, 101289. [Google Scholar] [CrossRef]
- Tanudjaja, H.J.; Hejase, C.A.; Tarabara, V.V.; Fane, A.G.; Chew, J.W. Membrane-based separation for oily wastewater: A practical perspective. Water Res. 2019, 156, 347–365. [Google Scholar] [CrossRef]
- Ismail, N.; Salleh, W.; Ismail, A.; Hasbullah, H.; Yusof, N.; Aziz, F.; Jaafar, J. Hydrophilic polymer-based membrane for oily wastewater treatment: A review. Sep. Purif. Technol. 2020, 233, 116007. [Google Scholar] [CrossRef]
- Thakur, V.K.; Voicu, S.I. Recent advances in cellulose and chitosan based membranes for water purification: A concise review. Carbohydr. Polym. 2016, 146, 148–165. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Gupta, R.K. Recent progress on ferroelectric polymer-based nanocomposites for high energy density capacitors: Synthesis, dielectric properties, and future aspects. Chem. Rev. 2016, 116, 4260–4317. [Google Scholar]
- Otitoju, T.; Ahmad, A.; Ooi, B. Polyvinylidene fluoride (PVDF) membrane for oil rejection from oily wastewater: A performance review. J. Water Process Eng. 2016, 14, 41–59. [Google Scholar] [CrossRef]
- Huang, S.; Ras, R.H.; Tian, X. Antifouling membranes for oily wastewater treatment: Interplay between wetting and membrane fouling. Curr. Opin. Colloid Interface Sci. 2018, 36, 90–109. [Google Scholar] [CrossRef]
- Ates, B.; Koytepe, S.; Ulu, A.; Gurses, C.; Thakur, V.K. Chemistry, structures, and advanced applications of nanocomposites from biorenewable resources. Chem. Rev. 2020, 120, 9304–9362. [Google Scholar] [CrossRef]
- Fahrina, A.; Arahman, N.; Mulyati, S.; Aprilia, S.; Mat Nawi, N.I.; Aqsha, A.; Bilad, M.R.; Takagi, R.; Matsuyama, H. Development of polyvinylidene fluoride membrane by incorporating bio-based ginger extract as additive. Polymers 2020, 12, 2003. [Google Scholar] [CrossRef]
- Zuo, J.-H.; Cheng, P.; Chen, X.-F.; Yan, X.; Guo, Y.-J.; Lang, W.-Z. Ultrahigh flux of polydopamine-coated PVDF membranes quenched in air via thermally induced phase separation for oil/water emulsion separation. Sep. Purif. Technol. 2018, 192, 348–359. [Google Scholar] [CrossRef]
- Chiao, Y.-H.; Chen, S.-T.; Yap Ang, M.B.M.; Patra, T.; Castilla-Casadiego, D.A.; Fan, R.; Almodovar, J.; Hung, W.-S.; Wickramasinghe, S.R. High-performance polyacrylic acid-grafted PVDF nanofiltration membrane with good antifouling property for the textile industry. Polymers 2020, 12, 2443. [Google Scholar] [CrossRef]
- Wan, L.S.; Xu, Z.K.; Wang, Z.G. Leaching of PVP from polyacrylonitrile/PVP blending membranes: A comparative study of asymmetric and dense membranes. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 1490–1498. [Google Scholar] [CrossRef]
- Ismail, N.; Venault, A.; Mikkola, J.-P.; Bouyer, D.; Drioli, E.; Kiadeh, N.T.H. Investigating the potential of membranes formed by the vapor induced phase separation process. J. Membr. Sci. 2020, 597, 117601. [Google Scholar] [CrossRef]
- Park, H.C.; Kim, Y.P.; Kim, H.Y.; Kang, Y.S. Membrane formation by water vapor induced phase inversion. J. Membr. Sci. 1999, 156, 169–178. [Google Scholar] [CrossRef]
- Menut, P.; Su, Y.; Chinpa, W.; Pochat-Bohatier, C.; Deratani, A.; Wang, D.; Huguet, P.; Kuo, C.; Lai, J.; Dupuy, C. A top surface liquid layer during membrane formation using vapor-induced phase separation (VIPS)—Evidence and mechanism of formation. J. Membr. Sci. 2008, 310, 278–288. [Google Scholar] [CrossRef]
- Zhao, N.; Xie, Q.; Weng, L.; Wang, S.; Zhang, X.; Xu, J. Superhydrophobic surface from vapor-induced phase separation of copolymer micellar solution. Macromolecules 2005, 38, 8996–8999. [Google Scholar] [CrossRef]
- Mat Nawi, N.I.; Chean, H.M.; Shamsuddin, N.; Bilad, M.R.; Narkkun, T.; Faungnawakij, K.; Khan, A.L. Development of hydrophilic PVDF membrane using vapour induced phase separation method for produced water treatment. Membranes 2020, 10, 121. [Google Scholar] [CrossRef]
- Peng, Y.; Dong, Y.; Fan, H.; Chen, P.; Li, Z.; Jiang, Q. Preparation of polysulfone membranes via vapor-induced phase separation and simulation of direct-contact membrane distillation by measuring hydrophobic layer thickness. Desalination 2013, 316, 53–66. [Google Scholar] [CrossRef]
- Tsai, H.; Kuo, C.; Lin, J.; Wang, D.; Deratani, A.; Pochat-Bohatier, C.; Lee, K.; Lai, J. Morphology control of polysulfone hollow fiber membranes via water vapor induced phase separation. J. Membr. Sci. 2006, 278, 390–400. [Google Scholar] [CrossRef]
- Dehban, A.; Kargari, A.; Ashtiani, F.Z. Preparation and characterization of an antifouling poly(phenyl sulfone) ultrafiltration membrane by vapor-induced phase separation technique. Sep. Purif. Technol. 2019, 212, 986–1000. [Google Scholar] [CrossRef]
- He, T. Finger-Like Structure; Springer: Berlin/Heidelberg, Germany, 2015; pp. 1–2. [Google Scholar]
- Tan, X.; Rodrigue, D. A review on porous polymeric membrane preparation. Part I: Production techniques with polysulfone and poly (vinylidene fluoride). Polymers 2019, 11, 1160. [Google Scholar] [CrossRef]
- Saljoughi, E.; Amirilargani, M.; Mohammadi, T. Effect of PEG additive and coagulation bath temperature on the morphology, permeability and thermal/chemical stability of asymmetric CA membranes. Desalination 2010, 262, 72–78. [Google Scholar] [CrossRef]
- Boom, R.; Wienk, I.; Van den Boomgaard, T.; Smolders, C. Microstructures in phase inversion membranes. Part 2. The role of a polymeric additive. J. Membr. Sci. 1992, 73, 277–292. [Google Scholar] [CrossRef]
- Marino, T.; Russo, F.; Figoli, A. The formation of polyvinylidene fluoride membranes with tailored properties via vapour/non-solvent induced phase separation. Membranes 2018, 8, 71. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Xie, R.; Luo, F.; Faraj, Y.; Liu, Z.; Ju, X.-J.; Wang, W.; Chu, L.-Y. Preparation of high strength poly (vinylidene fluoride) porous membranes with cellular structure via vapor-induced phase separation. J. Membr. Sci. 2018, 549, 151–164. [Google Scholar] [CrossRef]
- Masuelli, M.; Grasselli, M.; Marchese, J.; Ochoa, N. Preparation, structural and functional characterization of modified porous PVDF membranes by γ-irradiation. J. Membr. Sci. 2012, 389, 91–98. [Google Scholar] [CrossRef]
- Han, G.-H.; Lee, S.-H.; Seo, M.-g.; Lee, K.-Y. Effect of polyvinylpyrrolidone (PVP) on palladium catalysts for direct synthesis of hydrogen peroxide from hydrogen and oxygen. RSC Adv. 2020, 10, 19952–19960. [Google Scholar] [CrossRef]
- Mavukkandy, M.O.; Bilad, M.R.; Giwa, A.; Hasan, S.W.; Arafat, H.A. Leaching of PVP from PVDF/PVP blend membranes: Impacts on membrane structure and fouling in membrane bioreactors. J. Mater. Sci. 2016, 51, 4328–4341. [Google Scholar] [CrossRef]
- Marbelia, L.; Bilad, M.R.; Vankelecom, I.F. Gradual PVP leaching from PVDF/PVP blend membranes and its effects on membrane fouling in membrane bioreactors. Sep. Purif. Technol. 2019, 213, 276–282. [Google Scholar] [CrossRef]
- Liao, Y.; Bokhary, A.; Maleki, E.; Liao, B. A review of membrane fouling and its control in algal-related membrane processes. Bioresour. Technol. 2018, 264, 343–358. [Google Scholar] [CrossRef]
- Chollom, M.N.; Rathilal, S. Fouling and cleaning in osmotically driven membranes. In Osmotically Driven Membrane Processes: Approach, Development and Current Status; IntechOpen: London, UK, 2018; pp. 179–205. [Google Scholar]
- Huang, X.; Wang, W.; Liu, Y.; Wang, H.; Zhang, Z.; Fan, W.; Li, L. Treatment of oily waste water by PVP grafted PVDF ultrafiltration membranes. Chem. Eng. J. 2015, 273, 421–429. [Google Scholar] [CrossRef]
- Tsuyuhara, T.; Hanamoto, Y.; Miyoshi, T.; Kimura, K.; Watanabe, Y. Influence of membrane properties on physically reversible and irreversible fouling in membrane bioreactors. Water Sci. Technol. 2010, 61, 2235–2240. [Google Scholar] [CrossRef]





| Type of Membranes | Composition (wt.%) | Exposure Time before Immersion (minutes) | |||
|---|---|---|---|---|---|
| PVDF * | PVP * | LiCl * | DMAC * | ||
| M-Ref | 15 | 0 | 0 | 85 | 0 |
| M-0 | 13 | 3 | 0.1 | 83.9 | 0 |
| M-5 | 13 | 3 | 0.1 | 83.9 | 5 |
| M-15 | 13 | 3 | 0.1 | 83.9 | 15 |
| M-30 | 13 | 3 | 0.1 | 83.9 | 30 |
| Membrane | Composition (%) | |||
|---|---|---|---|---|
| Carbon (C) | Fluoride (F) | Oxygen (O) | Nitrogen (N) | |
| M-Ref | 55.55 | 42.84 | 1.61 | 0.00 |
| M-0 | 56.04 | 41.11 | 2.85 | 0.00 |
| M-5 | 52.93 | 45.36 | 1.71 | 0.00 |
| M-15 | 56.11 | 38.81 | 4.29 | 0.79 |
| M-30 | 60.66 | 33.61 | 4.73 | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nawi, N.I.M.; Sait, N.R.; Bilad, M.R.; Shamsuddin, N.; Jaafar, J.; Nordin, N.A.H.; Narkkun, T.; Faungnawakij, K.; Mohshim, D.F. Polyvinylidene Fluoride Membrane Via Vapour Induced Phase Separation for Oil/Water Emulsion Filtration. Polymers 2021, 13, 427. https://doi.org/10.3390/polym13030427
Nawi NIM, Sait NR, Bilad MR, Shamsuddin N, Jaafar J, Nordin NAH, Narkkun T, Faungnawakij K, Mohshim DF. Polyvinylidene Fluoride Membrane Via Vapour Induced Phase Separation for Oil/Water Emulsion Filtration. Polymers. 2021; 13(3):427. https://doi.org/10.3390/polym13030427
Chicago/Turabian StyleNawi, Normi Izati Mat, Nur Rifqah Sait, Muhammad Roil Bilad, Norazanita Shamsuddin, Juhana Jaafar, Nik Abdul Hadi Nordin, Thanitporn Narkkun, Kajornsak Faungnawakij, and Dzeti Farhah Mohshim. 2021. "Polyvinylidene Fluoride Membrane Via Vapour Induced Phase Separation for Oil/Water Emulsion Filtration" Polymers 13, no. 3: 427. https://doi.org/10.3390/polym13030427
APA StyleNawi, N. I. M., Sait, N. R., Bilad, M. R., Shamsuddin, N., Jaafar, J., Nordin, N. A. H., Narkkun, T., Faungnawakij, K., & Mohshim, D. F. (2021). Polyvinylidene Fluoride Membrane Via Vapour Induced Phase Separation for Oil/Water Emulsion Filtration. Polymers, 13(3), 427. https://doi.org/10.3390/polym13030427








