Effect of Polymer Dissolution Temperature and Conditioning Time on the Morphological and Physicochemical Characteristics of Poly(Vinylidene Fluoride) Membranes Prepared by Non-Solvent Induced Phase Separation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
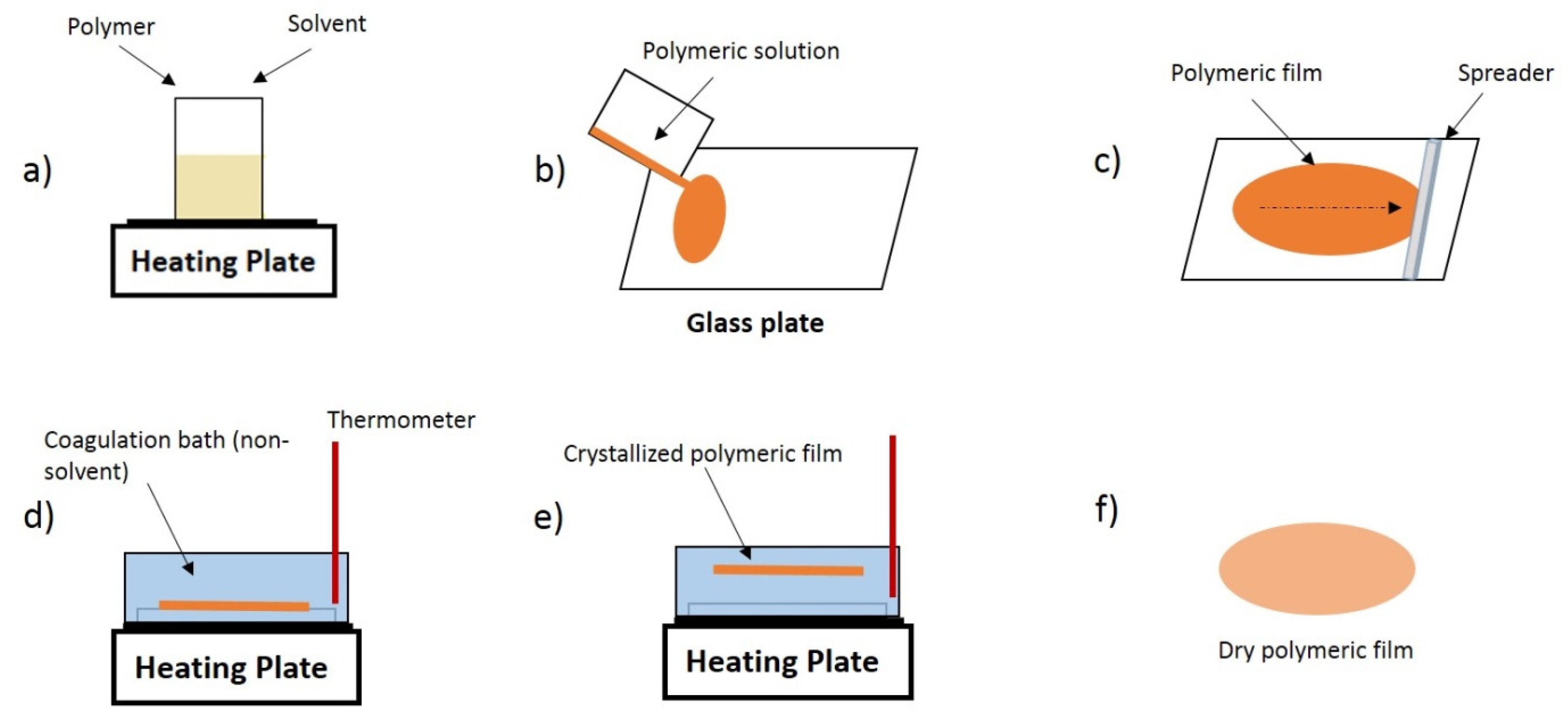
2.2. Membrane Preparation
2.3. Membrane Characterization
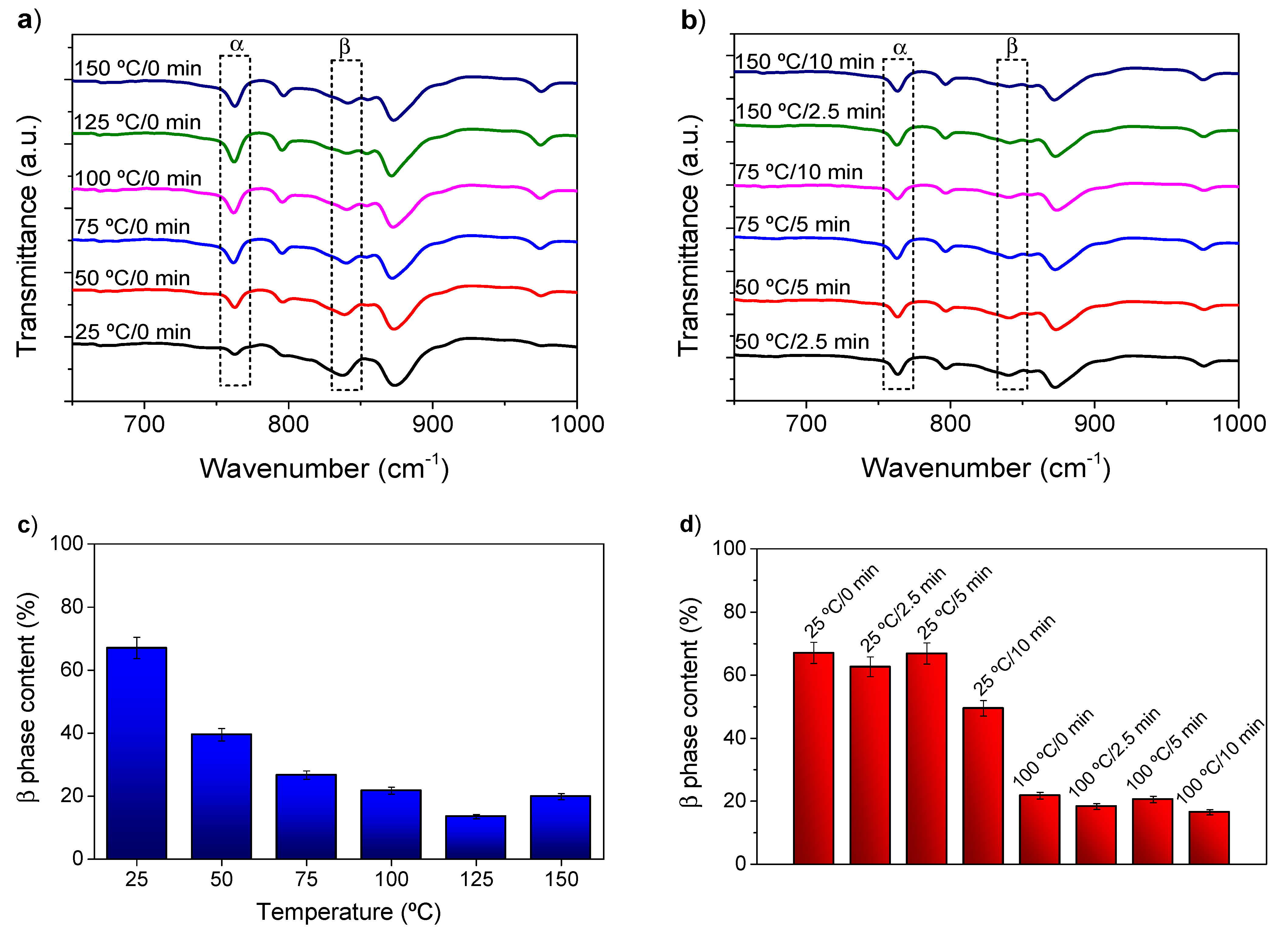
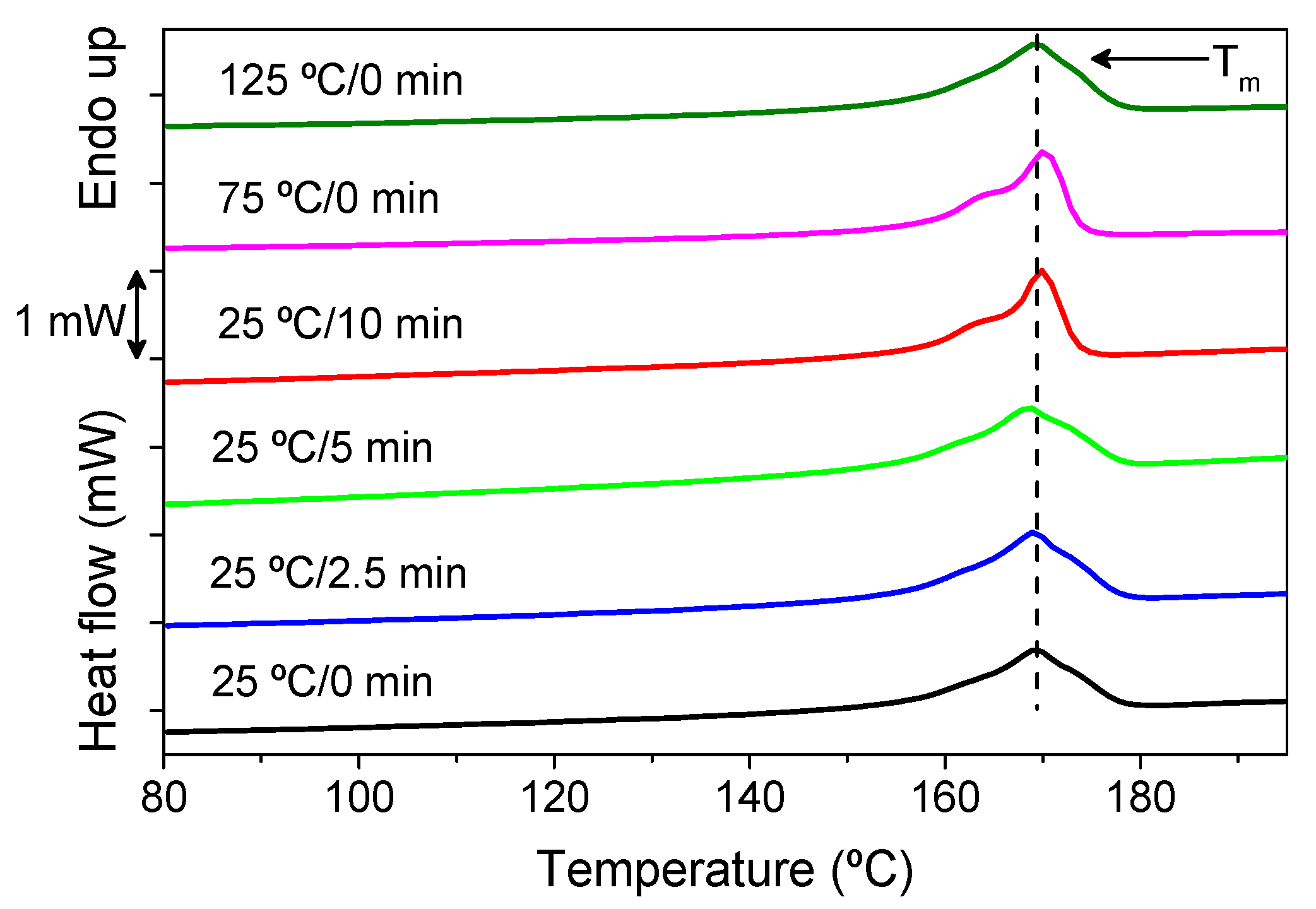
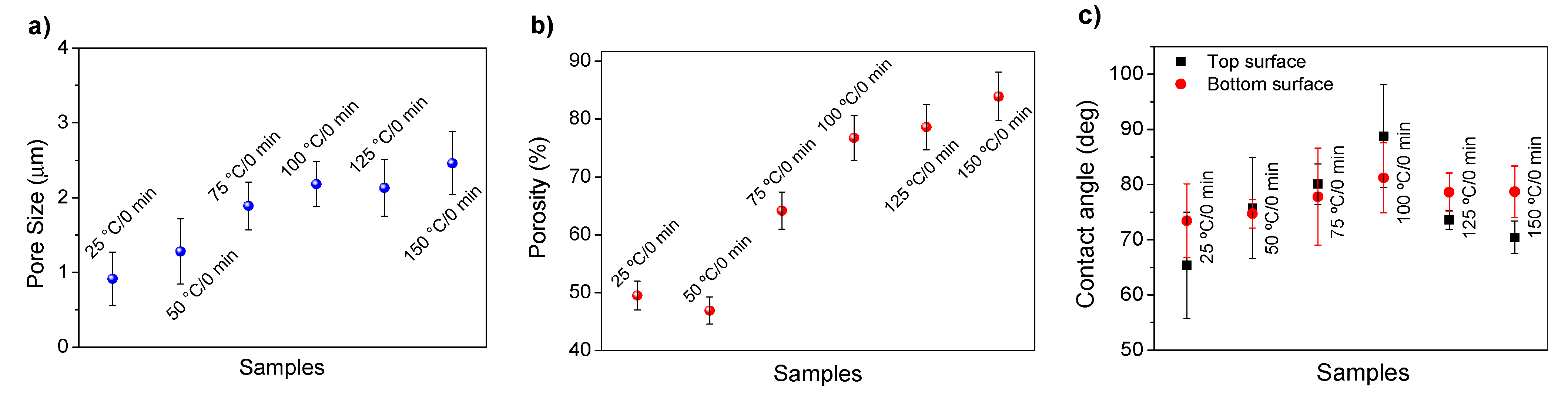
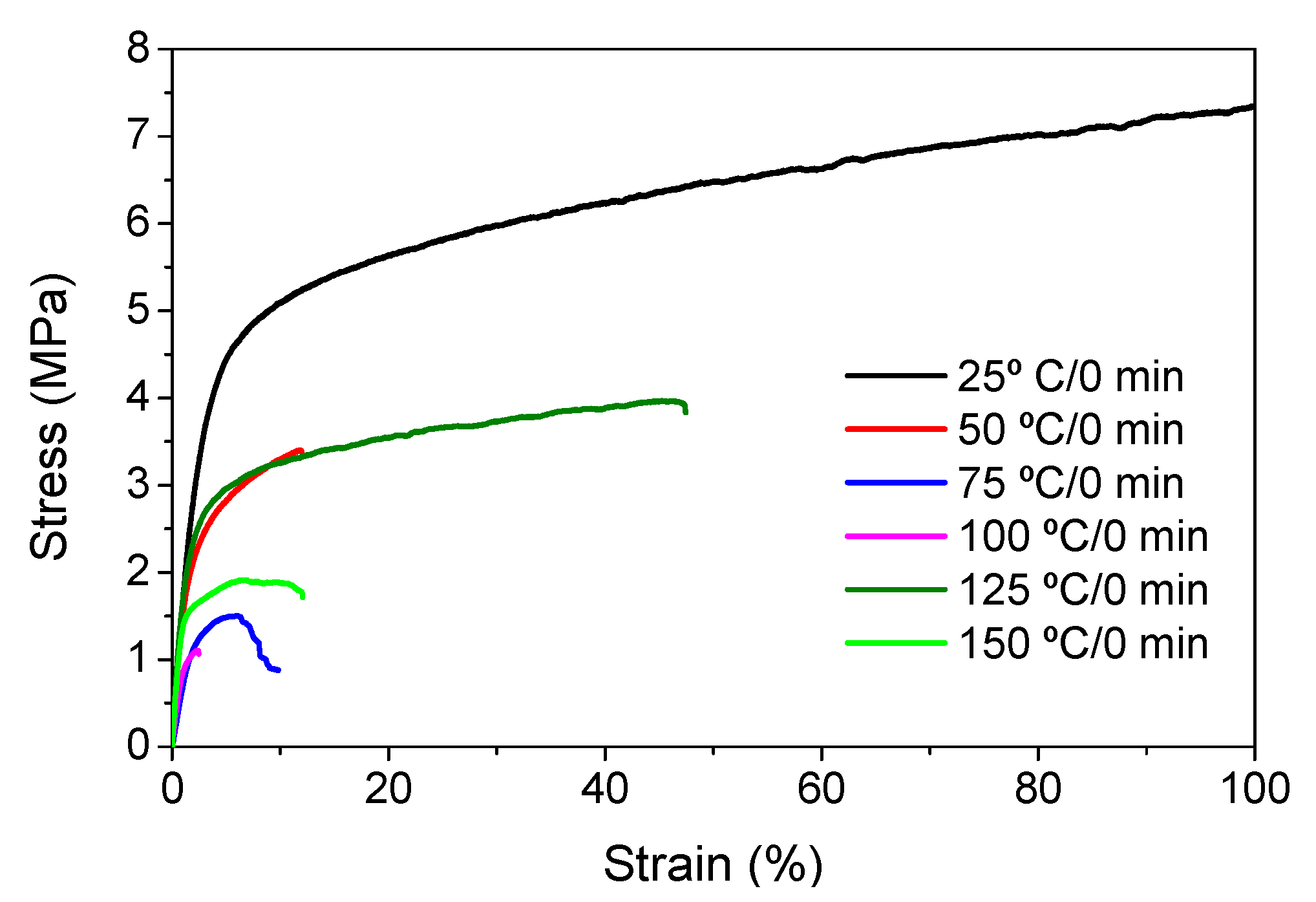
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baker, R.W. (Ed.) Overview of membrane science and technology. In Membrane Technology and Applications, 3rd ed.; John Wiley and Sons Ltd.: Chichester, UK, 2012; pp. 1–13. [Google Scholar]
- Liu, F.; Hashim, N.A.; Liu, Y.; Abed, M.R.M.; Li, K. Progress in the production and modification of PVDF membranes. J. Membr. Sci. 2011, 375, 1–27. [Google Scholar] [CrossRef]
- Gören, A.; Costa, C.M.; Tamaño Machiavello, M.N.; Cíntora-Juárez, D.; Nunes-Pereira, J.; Tirado, J.L.; Silva, M.M.; Gomez Ribelles, J.L.; Lanceros-Méndez, S. Effect of the degree of porosity on the performance of poly(vinylidene fluoride-trifluoroethylene)/poly(ethylene oxide) blend membranes for lithium-ion battery separators. Solid State Ion. 2015, 280, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Nunes-Pereira, J.; Costa, C.M.; Leones, R.; Silva, M.M.; Lanceros-Méndez, S. Li-ion battery separator membranes based on poly(vinylidene fluoride-trifluoroethylene)/carbon nanotube composites. Solid State Ion. 2013, 249–250, 63–71. [Google Scholar] [CrossRef]
- Correia, D.M.; Costa, C.M.; Nunes-Pereira, J.; Silva, M.M.; Botelho, G.; Ribelles, J.L.G.; Lanceros-Méndez, S. Physicochemical properties of poly(vinylidene fluoride-trifluoroethylene)/poly(ethylene oxide) blend membranes for lithium ion battery applications: Influence of poly(ethylene oxide) molecular weight. Solid State Ion. 2014, 268, 54–67. [Google Scholar] [CrossRef]
- Pasquini, L.; Zhakisheva, B.; Sgreccia, E.; Narducci, R.; Di Vona, M.L.; Knauth, P. Stability of proton exchange membranes in phosphate buffer for enzymatic fuel cell application: Hydration, conductivity and mechanical properties. Polymers 2021, 13, 475. [Google Scholar] [CrossRef]
- Pal, S.; Mondal, R.; Chatterjee, U. Sulfonated polyvinylidene fluoride and functional copolymer based blend proton exchange membrane for fuel cell application and studies on methanol crossover. Renew. Energy 2021, 170, 974–984. [Google Scholar] [CrossRef]
- Nawi, N.I.M.; Sait, N.R.; Bilad, M.R.; Shamsuddin, N.; Jaafar, J.; Nordin, N.A.H.; Narkkun, T.; Faungnawakij, K.; Mohshim, D.F. Polyvinylidene fluoride membrane via vapour induced phase separation for oil/water emulsion filtration. Polymers 2021, 13, 427. [Google Scholar] [CrossRef]
- Palanisamy, T.; Tabatabai, S.A.A.; Zhang, T.; Leiknes, T. Role of surfactants in cleaning of PVDF ultrafiltration membranes fouled by emulsified cutting oil. J. Water Process Eng. 2021, 40, 101923. [Google Scholar] [CrossRef]
- Feng, F.; Liang, C.-Z.; Wu, J.; Weber, M.; Maletzko, C.; Zhang, S.; Chung, T.-S. Polyphenylsulfone (PPSU)-based copolymeric membranes: Effects of chemical structure and content on gas permeation and separation. Polymers 2021, 13, 2745. [Google Scholar] [CrossRef]
- Wu, X.; Zhao, B.; Wang, L.; Zhang, Z.; Li, M. Preparation and characterization of superhydrophobic PVDF/HMSNs hybrid membrane for CO2 absorption. Polymer 2021, 214, 123242. [Google Scholar] [CrossRef]
- Alosaimi, A.M. Polysulfone membranes based hybrid nanocomposites for the adsorptive removal of Hg(II) ions. Polymers 2021, 13, 2792. [Google Scholar] [CrossRef]
- Salazar, H.; Nunes-Pereira, J.; Correia, D.M.; Cardoso, V.F.; Gonçalves, R.; Martins, P.M.; Ferdov, S.; Martins, M.D.; Botelho, G.; Lanceros-Méndez, S. Poly(vinylidene fluoride-hexafluoropropylene)/bayerite composite membranes for efficient arsenic removal from water. Mater. Chem. Phys. 2016, 183, 430–438. [Google Scholar] [CrossRef]
- Nunes-Pereira, J.; Lima, R.; Choudhary, G.; Sharma, P.R.; Ferdov, S.; Botelho, G.; Sharma, R.K.; Lanceros-Méndez, S. Highly efficient removal of fluoride from aqueous media through polymer composite membranes. Sep. Purif. Technol. 2018, 205, 1–10. [Google Scholar] [CrossRef]
- Xu, Y.; Yang, D.; Huo, S.; Ren, J.; Gao, N.; Chen, Z.; Liu, Y.; Xie, Z.; Zhou, S.; Qu, X. Carbon dots and ruthenium doped oxygen sensitive nanofibrous membranes for monitoring the respiration of agricultural products. Polym. Test. 2021, 93, 106957. [Google Scholar] [CrossRef]
- Krishnamoorthy, V.; Rajiv, S. Tailoring electrospun polymer blend carriers for nutrient delivery in seed coating for sustainable agriculture. J. Clean. Prod. 2018, 177, 69–78. [Google Scholar] [CrossRef]
- Chen, Y.-P.; Lo, T.-S.; Lin, Y.-T.; Chien, Y.-H.; Lu, C.-J.; Liu, S.-J. Fabrication of drug-eluting polycaprolactone/poly(lactic-co-glycolic acid) prolapse mats using solution-extrusion 3D printing and coaxial electrospinning techniques. Polymers 2021, 13, 2295. [Google Scholar] [CrossRef]
- Briddon, S.J.; Kilpatrick, L.E.; Hill, S.J. Studying GPCR pharmacology in membrane microdomains: Fluorescence correlation spectroscopy comes of age. Trends Pharmacol. Sci. 2018, 39, 158–174. [Google Scholar] [CrossRef]
- Pandey, A.; Yang, T.-S.; Yang, T.-I.; Belem, W.F.; Teng, N.-C.; Chen, I.-W.; Huang, C.-S.; Kareiva, A.; Yang, J.-C. An insight into nano silver fluoride-coated silk fibroin bioinspired membrane properties for guided tissue regeneration. Polymers 2021, 13, 2659. [Google Scholar] [CrossRef]
- Kang, Y.; Chen, P.; Shi, X.; Zhang, G.; Wang, C. Multilevel structural stereocomplex polylactic acid/collagen membranes by pattern electrospinning for tissue engineering. Polymer 2018, 156, 250–260. [Google Scholar] [CrossRef]
- Trombino, S.; Poerio, T.; Ferrarelli, T.; Mauro, M.V.; Giraldi, C.; Giorno, L.; Cassano, R. Multifunctional membranes based on natural polymers: Preparation, characterization and in vitro performance evaluation. Polym. Int. 2015, 64, 344–351. [Google Scholar] [CrossRef]
- Li, J.; Hu, Y.; He, T.; Huang, M.; Zhang, X.; Yuan, J.; Wei, Y.; Dong, X.; Liu, W.; Ko, F.; et al. Electrospun sandwich-structure composite membranes for wound dressing scaffolds with high antioxidant and antibacterial activity. Macromol. Mater. Eng. 2018, 303, 1700270. [Google Scholar] [CrossRef]
- Saleh, T.A.; Gupta, V.K. An overview of membrane science and technology. In Nanomaterial and Polymer Membranes; Saleh, T.A., Gupta, V.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–23. [Google Scholar]
- Sun, A.C.; Kosar, W.; Zhang, Y.; Feng, X. A study of thermodynamics and kinetics pertinent to formation of PVDF membranes by phase inversion. Desalination 2013, 309, 156–164. [Google Scholar] [CrossRef]
- Kuo, C.-Y.; Lin, H.-N.; Tsai, H.-A.; Wang, D.-M.; Lai, J.-Y. Fabrication of a high hydrophobic PVDF membrane via nonsolvent induced phase separation. Desalination 2008, 233, 40–47. [Google Scholar] [CrossRef]
- Baek, G.; Yang, S.-C. Effect of the two-dimensional magnetostrictive fillers of CoFe2O4-intercalated graphene oxide sheets in 3-2 type poly(vinylidene fluoride)-based magnetoelectric films. Polymers 2021, 13, 1782. [Google Scholar] [CrossRef]
- Nunes-Pereira, J.; Costa, P.; Fernandes, L.; Carvalho, E.O.; Fernandes, M.M.; Carabineiro, S.A.C.; Buijnsters, J.G.; Tubio, C.R.; Lanceros-Mendez, S. Antimicrobial and antibiofilm properties of fluorinated polymers with embedded functionalized nanodiamonds. ACS Appl. Polym. Mater. 2020, 2, 5014–5024. [Google Scholar] [CrossRef]
- Russo, F.; Marino, T.; Galiano, F.; Gzara, L.; Gordano, A.; Organji, H.; Figoli, A. Tamisolve® NxG as an alternative non-toxic solvent for the preparation of porous poly (vinylidene fluoride) membranes. Polymers 2021, 13, 2579. [Google Scholar] [CrossRef]
- Zioui, D.; Arous, O.; Mameri, N.; Kerdjoudj, H.; Sebastian, M.S.; Vilas, J.L.; Nunes-Pereira, J.; Lanceros-Méndez, S. Membranes based on polymer miscibility for selective transport and separation of metallic ions. J. Hazard. Mater. 2017, 336, 188–194. [Google Scholar] [CrossRef]
- Zeng, X.; Lin, J.; Cai, W.; Lu, Q.; Fu, S.; Li, J.; Yan, X.; Wen, X.; Zhou, C.; Zhang, M. Fabrication of superhydrophilic PVDF membranes by one-step modification with eco-friendly phytic acid and polyethyleneimine complex for oil-in-water emulsions separation. Chemosphere 2021, 264, 128395. [Google Scholar] [CrossRef]
- Kang, D.H.; Kim, N.K.; Kang, H.W. Electrostatic charge retention in PVDF nanofiber-nylon mesh multilayer structure for effective fine particulate matter filtration for face masks. Polymers 2021, 13, 3235. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.M.; Nunes-Pereira, J.; Sencadas, V.; Silva, M.M.; Lanceros-Méndez, S. Effect of fiber orientation in gelled poly(vinylidene fluoride) electrospun membranes for Li-ion battery applications. J. Mater. Sci. 2013, 48, 6833–6840. [Google Scholar] [CrossRef]
- Galiano, F.; Song, X.; Marino, T.; Boerrigter, M.; Saoncella, O.; Simone, S.; Faccini, M.; Chaumette, C.; Drioli, E.; Figoli, A. Novel photocatalytic PVDF/nano-TiO2 hollow fibers for environmental remediation. Polymers 2018, 10, 1134. [Google Scholar] [CrossRef] [Green Version]
- Kong, X.; Lu, X.; Liu, J.; Wu, C.; Zhang, S. Improved desalination performance of polyvinylidene fluoride hollow fiber membranes by the intermediate role of surfactants. Macromol. Mater. Eng. 2021, 306, 2000538. [Google Scholar] [CrossRef]
- Wang, X.; Xiao, C.; Liu, H.; Chen, M.; Xu, H.; Luo, W.; Zhang, F. Robust functionalization of underwater superoleophobic PVDF-HFP tubular nanofiber membranes and applications for continuous dye degradation and oil/water separation. J. Membr. Sci. 2020, 596, 117583. [Google Scholar] [CrossRef]
- Akbari, A.; Hamadanian, M.; Bojaran, M.; Lehi, A.Y.; Jabbari, V. The role of solution and coagulation temperatures in crystalline structure, morphology, roughness, pore diameter distribution, and separation properties of nanoporous membranes fabricated via phase inversion. Sep. Sci. Technol. 2012, 47, 1866–1873. [Google Scholar] [CrossRef]
- Feng, C.; Wang, R.; Wu, Y.; Li, G. Preliminary analysis of a linear pore pattern formed on poly(vinylidene fluoride-co-hexafluoro propylene) porous membrane surfaces. J. Membr. Sci. 2010, 352, 255–261. [Google Scholar] [CrossRef]
- Ribeiro, C.; Costa, C.M.; Correia, D.M.; Nunes-Pereira, J.; Oliveira, J.; Martins, P.; Goncalves, R.; Cardoso, V.F.; Lanceros-Mendez, S. Electroactive poly(vinylidene fluoride)-based structures for advanced applications. Nat. Protoc. 2018, 13, 681–704. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Pan, J.; Wang, Z.; Frappa, M.; Drioli, E.; Macedonio, F. Hyflon/PVDF membranes prepared by NIPS and TIPS: Comparison in MD performance. Sep. Purif. Technol. 2020, 247, 116992. [Google Scholar] [CrossRef]
- Jung, J.T.; Kim, J.F.; Wang, H.H.; di Nicolo, E.; Drioli, E.; Lee, Y.M. Understanding the non-solvent induced phase separation (NIPS) effect during the fabrication of microporous PVDF membranes via thermally induced phase separation (TIPS). J. Membr. Sci. 2016, 514, 250–263. [Google Scholar] [CrossRef]
- Bottino, A.; Capannelli, G.; Munari, S.; Turturro, A. Solubility parameters of poly(vinylidene fluoride). J. Polym. Sci. Part B Polym. Phys. 1988, 26, 785–794. [Google Scholar] [CrossRef]
- Bottino, A.; Camera-Roda, G.; Capannelli, G.; Munari, S. The formation of microporous polyvinylidene difluoride membranes by phase separation. J. Membr. Sci. 1991, 57, 1–20. [Google Scholar] [CrossRef]
- Guillen, G.R.; Pan, Y.; Li, M.; Hoek, E.M.V. Preparation and Characterization of Membranes Formed by Nonsolvent Induced Phase Separation: A Review. Ind. Eng. Chem. Res. 2011, 50, 3798–3817. [Google Scholar] [CrossRef]
- Ma, W.; Cao, Y.; Gong, F.; Liu, C.; Tao, G.; Wang, X. Poly(vinylidene fluoride) membranes prepared via nonsolvent induced phase separation combined with the gelation. Colloids Surf. A Physicochem. Eng. Asp. 2015, 479, 25–34. [Google Scholar] [CrossRef]
- Dong, X.; Lu, D.; Harris, T.A.L.; Escobar, I.C. Polymers and Solvents Used in Membrane Fabrication: A Review Focusing on Sustainable Membrane Development. Membranes 2021, 11, 309. [Google Scholar] [CrossRef]
- Wang, H.H.; Jung, J.T.; Kim, J.F.; Kim, S.; Drioli, E.; Lee, Y.M. A novel green solvent alternative for polymeric membrane preparation via nonsolvent-induced phase separation (NIPS). J. Membr. Sci. 2019, 574, 44–54. [Google Scholar] [CrossRef]
- Russo, F.; Ursino, C.; Sayinli, B.; Koyuncu, I.; Galiano, F.; Figoli, A. Advancements in sustainable PVDF copolymer membrane preparation using Rhodiasolv® PolarClean as an alternative eco-friendly solvent. Clean Technol. 2021, 3, 761–786. [Google Scholar] [CrossRef]
- Marino, T.; Russo, F.; Figoli, A. The Formation of Polyvinylidene Fluoride Membranes with Tailored Properties via Vapour/Non-Solvent Induced Phase Separation. Membranes 2018, 8, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Zhang, L.; Sun, D.; An, Q.; Chen, H. Effect of coagulation bath temperature on formation mechanism of poly(vinylidene fluoride) membrane. J. Appl. Polym. Sci. 2008, 110, 1656–1663. [Google Scholar] [CrossRef]
- Cheng, L.-P. Effect of Temperature on the Formation of Microporous PVDF Membranes by Precipitation from 1-Octanol/DMF/PVDF and Water/DMF/PVDF Systems. Macromolecules 1999, 32, 6668–6674. [Google Scholar] [CrossRef]
- Haponska, M.; Trojanowska, A.; Nogalska, A.; Jastrzab, R.; Gumi, T.; Tylkowski, B. PVDF membrane morphology—Influence of polymer molecular weight and preparation temperature. Polymers 2017, 9, 718. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Lu, X.; Liu, Z.; Ma, Z.; Wu, S.; Li, Z.; Kong, X.; Liu, J.; Wu, C. The unidirectional regulatory role of coagulation bath temperature on cross-section radius of the PVDF hollow-fiber membrane. J. Membr. Sci. 2018, 550, 9–17. [Google Scholar] [CrossRef]
- Young, T.-H.; Cheng, L.-P.; Lin, D.-J.; Fane, L.; Chuang, W.-Y. Mechanisms of PVDF membrane formation by immersion-precipitation in soft (1-octanol) and harsh (water) nonsolvents. Polymer 1999, 40, 5315–5323. [Google Scholar] [CrossRef]
- Cheng, L.-P.; Young, T.-H.; Fang, L.; Gau, J.-J. Formation of particulate microporous poly(vinylidene fluoride) membranes by isothermal immersion precipitation from the 1-octanol/dimethylformamide/poly(vinylidene fluoride) system. Polymer 1999, 40, 2395–2403. [Google Scholar] [CrossRef]
- Thürmer, M.B.; Poletto, P.; Marcolin, M.; Duarte, J.; Zeni, M. Effect of non-solvents used in the coagulation bath on morphology of PVDF membranes. Mater. Res. 2012, 15, 884–890. [Google Scholar] [CrossRef]
- Pagliero, M.; Bottino, A.; Comite, A.; Costa, C. Novel hydrophobic PVDF membranes prepared by nonsolvent induced phase separation for membrane distillation. J. Membr. Sci. 2020, 596, 117575. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Z.; Liu, M.; Zhao, F.; Wu, Z. In-situ modification of PVDF membrane during phase-inversion process using carbon nanosphere sol as coagulation bath for enhancing anti-fouling ability. J. Membr. Sci. 2017, 526, 272–280. [Google Scholar] [CrossRef]
- Ashtiani, S.; Khoshnamvand, M.; Číhal, P.; Dendisová, M.; Randová, A.; Bouša, D.; Shaliutina-Kolešová, A.; Sofer, Z.; Friess, K. Fabrication of a PVDF membrane with tailored morphology and properties via exploring and computing its ternary phase diagram for wastewater treatment and gas separation applications. RSC Adv. 2020, 10, 40373–40383. [Google Scholar] [CrossRef]
- Sousa, R.E.; Ferreira, J.C.C.; Costa, C.M.; Machado, A.V.; Silva, M.M.; Lanceros-Mendez, S. Tailoring poly(vinylidene fluoride-co-chlorotrifluoroethylene) microstructure and physicochemical properties by exploring its binary phase diagram with dimethylformamide. J. Polym. Sci. Part B Polym. Phys. 2015, 53, 761–773. [Google Scholar] [CrossRef] [Green Version]
- Tabani, Z.; Maghsoudi, H.; Fathollahi Zonouz, A. High electrochemical stability of polyvinylidene fluoride (PVDF) porous membranes using phase inversion methods for lithium-ion batteries. J. Solid State Electrochem. 2021, 25, 651–657. [Google Scholar] [CrossRef]
- Ajkidkarn, P.; Manuspiya, H. Novel bacterial cellulose nanocrystals/polyether block amide microporous membranes as separators for lithium-ion batteries. Int. J. Biol. Macromol. 2020, 164, 3580–3588. [Google Scholar] [CrossRef]
- Shi, M.; Dai, Q.; Li, F.; Li, T.; Hou, G.; Zhang, H.; Li, X. Membranes with well-defined selective layer regulated by controlled solvent diffusion for high power density flow battery. Adv. Energy Mater. 2020, 10, 2001382. [Google Scholar] [CrossRef]
- Guan, X.; Zhang, Z.; Zhang, S.; Wang, Y.; Yang, H.; Wang, J.; Li, M.; Lu, H.; Li, Y.; Huang, Q.; et al. NIPS derived three-dimensional porous copper membrane for high-energy-density lithium-ion batteries. Electrochim. Acta 2019, 312, 424–431. [Google Scholar] [CrossRef]
- Ferreira, C.G.; Cardoso, V.F.; Lopes, A.C.; Botelho, G.; Lanceros-Méndez, S. Tailoring microstructure and physical properties of poly(vinylidene fluoride–hexafluoropropylene) porous films. J. Mater. Sci. 2015, 50, 5047–5058. [Google Scholar] [CrossRef]
- Martins, P.; Lopes, A.C.; Lanceros-Mendez, S. Electroactive phases of poly(vinylidene fluoride): Determination, processing and applications. Prog. Polym. Sci. 2014, 39, 683–706. [Google Scholar] [CrossRef]
- Benz, M.; Euler, W.B. Determination of the crystalline phases of poly(vinylidene fluoride) under different preparation conditions using differential scanning calorimetry and infrared spectroscopy. J. Appl. Polym. Sci. 2003, 89, 1093–1100. [Google Scholar] [CrossRef]
- Fu, X.Y.; Sotani, T.; Matsuyama, H. Effect of membrane preparation method on the outer surface roughness of cellulose acetate butyrate hollow fiber membrane. Desalination 2008, 233, 10–18. [Google Scholar] [CrossRef]
- Purkait, M.K.; Sinha, M.K.; Mondal, P.; Singh, R. Chapter 1—Introduction to Membranes. In Stimuli Responsive Polymeric Membranes: Smart Polymeric Membranes; Purkait, M.K., Sinha, M.K., Mondal, P., Singh, R., Eds.; Elsevier: London, UK, 2018; Volume 25, pp. 1–37. [Google Scholar]
- Ferreira, J.C.C.; Monteiro, T.S.; Lopes, A.C.; Costa, C.M.; Silva, M.M.; Machado, A.V.; Lanceros-Mendez, S. Variation of the physicochemical and morphological characteristics of solvent casted poly(vinylidene fluoride) along its binary phase diagram with dimethylformamide. J. Non Cryst. Solids 2015, 412, 16–23. [Google Scholar] [CrossRef]
- Lanceros-Méndez, S.; Mano, J.F.; Costa, A.M.; Schmidt, V.H. FTIR and Dsc studies of mechanically deformed β-Pvdf films. J. Macromol. Sci. Part B 2007, 40, 517–527. [Google Scholar] [CrossRef] [Green Version]
- Gregorio, R., Jr. Determination of the α, β, and γ crystalline phases of poly(vinylidene fluoride) films prepared at different conditions. J. Appl. Polym. Sci. 2006, 100, 3272–3279. [Google Scholar] [CrossRef]
- Tashiro, K. Crystal structure and phase transition of PVDF and related copolymers. In Ferroelectric Polymers: Chemistry, Physics and Aplications; Nalwa, H.S., Ed.; Marcel Dekker: New York, NY, USA, 1995; pp. 63–182. [Google Scholar]
- Lovinger, A.J.; Wang, T.T. Investigation of the properties of directionally solidified poly(vinylidene fluoride). Polymer 1979, 20, 725–732. [Google Scholar] [CrossRef]
- Gregorio, R.; Capitão, R.C. Morphology and phase transition of high melt temperature crystallized poly(vinylidene fluoride). J. Mater. Sci. 2000, 35, 299–306. [Google Scholar] [CrossRef]
- Uragami, T.; Fujimoto, M.; Sugihara, M. Studies on syntheses and permeabilities of special polymer membranes. 27. Concentration of poly(styrene sulphonic acid) in various aqueous solutions using poly(vinylidene fluoride) membranes. Polymer 1981, 22, 240–244. [Google Scholar] [CrossRef]
- Rezaei, H.; Ashtiani, F.Z.; Fouladitajar, A. Effects of operating parameters on fouling mechanism and membrane flux in cross-flow microfiltration of whey. Desalination 2011, 274, 262–271. [Google Scholar] [CrossRef]
- Lee, A.; Elam, J.W.; Darling, S.B. Membrane materials for water purification: Design, development, and application. Environ. Sci. Water Res. Technol. 2016, 2, 17–42. [Google Scholar] [CrossRef]
- Zhang, P.-Y.; Yang, H.; Xu, Z.-L.; Wei, Y.-M.; Guo, J.-L.; Chen, D.-G. Characterization and preparation of poly(vinylidene fluoride) (PVDF) microporous membranes with interconnected bicontinuous structures via non-solvent induced phase separation (NIPS). J. Polym. Res. 2013, 20, 66. [Google Scholar] [CrossRef]
| Effect of Varying Dissolution Temperature | ||||
|---|---|---|---|---|
| Top Surface | Bottom Surface | Cross Section | ||
| 25 °C/0 min |  |  |  |  |
| 50 °C/0 min |  |  |  |  |
| 75 °C/0 min | 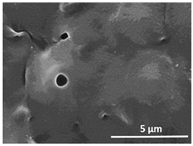 |  | 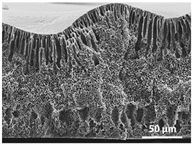 |  |
| 100 °C/0 min |  |  |  |  |
| 125 °C/0 min |  |  |  |  |
| 150 °C/0 min |  |  | 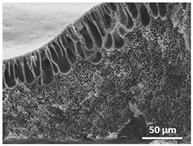 |  |
| Effect of Varying Drying Time | ||||
|---|---|---|---|---|
| Top Surface | Bottom Surface | Cross Section | ||
| 25 °C/0 min |  |  |  |  |
| 25 °C/2.5 min |  |  |  |  |
| 25 °C/5 min |  |  |  |  |
| 25 °C/10 min |  |  |  |  |
| Sample | 25 °C/0 min | 25 °C/2.5 min | 25 °C/5 min | 25 °C/10 min | 75 °C/0 min | 125 °C/0 min |
|---|---|---|---|---|---|---|
| ΔHf (J/g) | 57.2 ± 2.9 | 50 ± 2.5 | 58.7 ± 2.9 | 54.5 ± 2.7 | 50.1 ± 2.5 | 52.2 ± 2.6 |
| χc (%) | 57.5 ± 2.9 | 50.6 ± 2.5 | 59.2 ± 3.0 | 55.7 ± 2.8 | 52.2 ± 2.6 | 55.1 ± 2.8 |
| Sample | Young’s Modulus (MPa) | Yield Strain (%) | Strain at Break (%) |
|---|---|---|---|
| 25 °C/0 min | 300 ± 15 | 1.6 ± 0.1 | 81.2 ± 4.1 |
| 50 °C/0 min | 200 ± 10 | 1.4 ± 0.1 | 12.0 ± 0.8 |
| 75 °C/0 min | 70 ± 4 | 1.8 ± 0.1 | 6.4 ± 0.3 |
| 100 °C/0 min | 100 ± 5 | 1.0 ± 0.2 | 2.5 ± 0.3 |
| 125 °C/0 min | 200 ± 13 | 1.9 ± 0.1 | 47.4 ± 3.1 |
| 150 °C/0 min | 100 ± 6 | 1.1 ± 0.3 | 12.0 ± 2.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira, J.; Cardoso, V.F.; Botelho, G.; Morão, A.M.; Nunes-Pereira, J.; Lanceros-Mendez, S. Effect of Polymer Dissolution Temperature and Conditioning Time on the Morphological and Physicochemical Characteristics of Poly(Vinylidene Fluoride) Membranes Prepared by Non-Solvent Induced Phase Separation. Polymers 2021, 13, 4062. https://doi.org/10.3390/polym13234062
Teixeira J, Cardoso VF, Botelho G, Morão AM, Nunes-Pereira J, Lanceros-Mendez S. Effect of Polymer Dissolution Temperature and Conditioning Time on the Morphological and Physicochemical Characteristics of Poly(Vinylidene Fluoride) Membranes Prepared by Non-Solvent Induced Phase Separation. Polymers. 2021; 13(23):4062. https://doi.org/10.3390/polym13234062
Chicago/Turabian StyleTeixeira, João, Vanessa Fernandes Cardoso, Gabriela Botelho, António Miguel Morão, João Nunes-Pereira, and Senentxu Lanceros-Mendez. 2021. "Effect of Polymer Dissolution Temperature and Conditioning Time on the Morphological and Physicochemical Characteristics of Poly(Vinylidene Fluoride) Membranes Prepared by Non-Solvent Induced Phase Separation" Polymers 13, no. 23: 4062. https://doi.org/10.3390/polym13234062
APA StyleTeixeira, J., Cardoso, V. F., Botelho, G., Morão, A. M., Nunes-Pereira, J., & Lanceros-Mendez, S. (2021). Effect of Polymer Dissolution Temperature and Conditioning Time on the Morphological and Physicochemical Characteristics of Poly(Vinylidene Fluoride) Membranes Prepared by Non-Solvent Induced Phase Separation. Polymers, 13(23), 4062. https://doi.org/10.3390/polym13234062










