Multifunctional Electrospun Nanofibers Based on Biopolymer Blends and Magnetic Tubular Halloysite for Medical Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Magnetic Modification of Halloysite
2.2.2. Preparation of PCL/Gel Solutions
2.2.3. Electrospinning Process
2.3. Characterization
2.3.1. Structure and Composition of Raw and Magnetic Halloysite
2.3.2. Magnetic Properties
2.3.3. Mechanical Properties
3. Results and Discussion
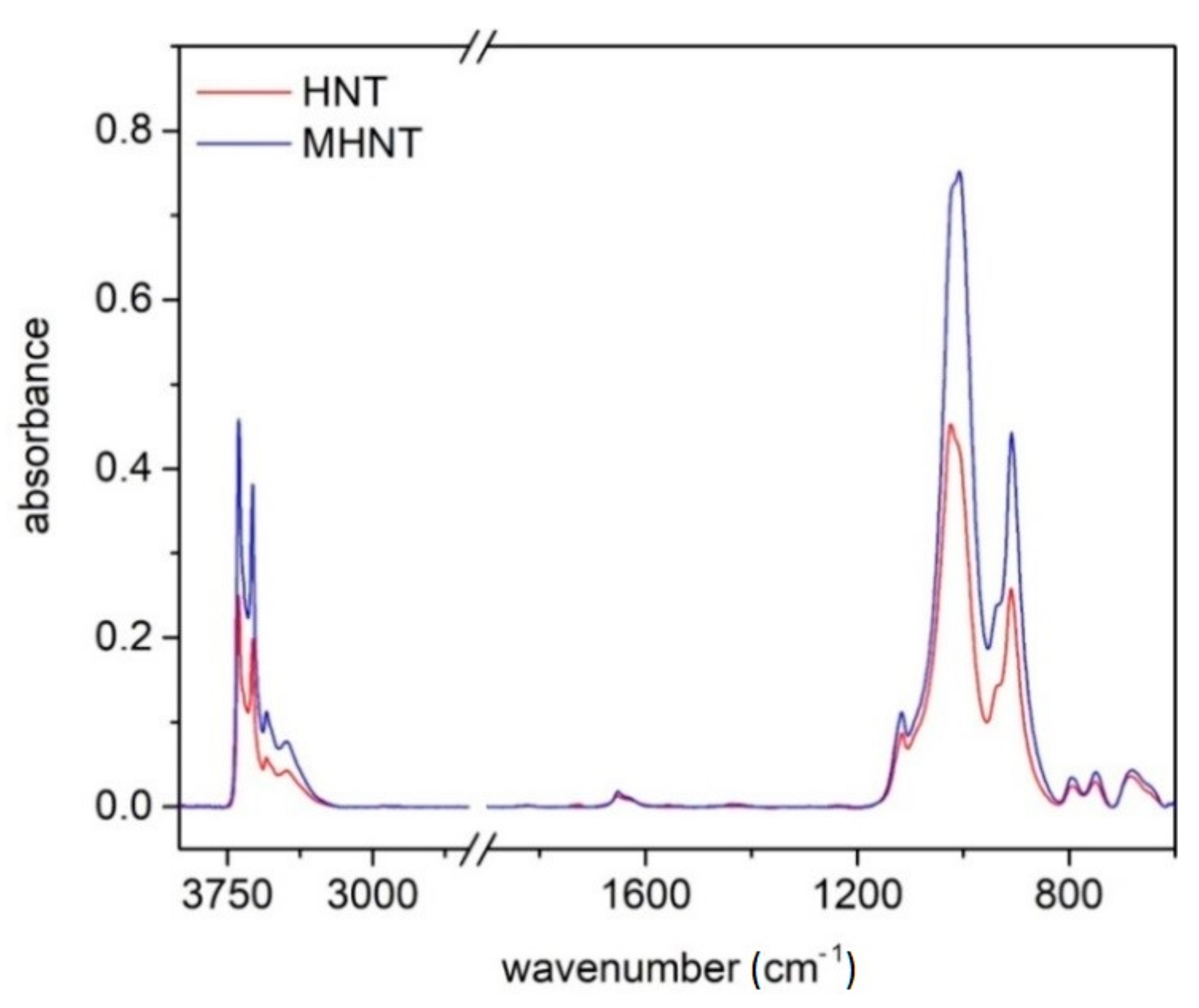
3.1. Structure and Composition of Magnetized Halloysite (MHNT)
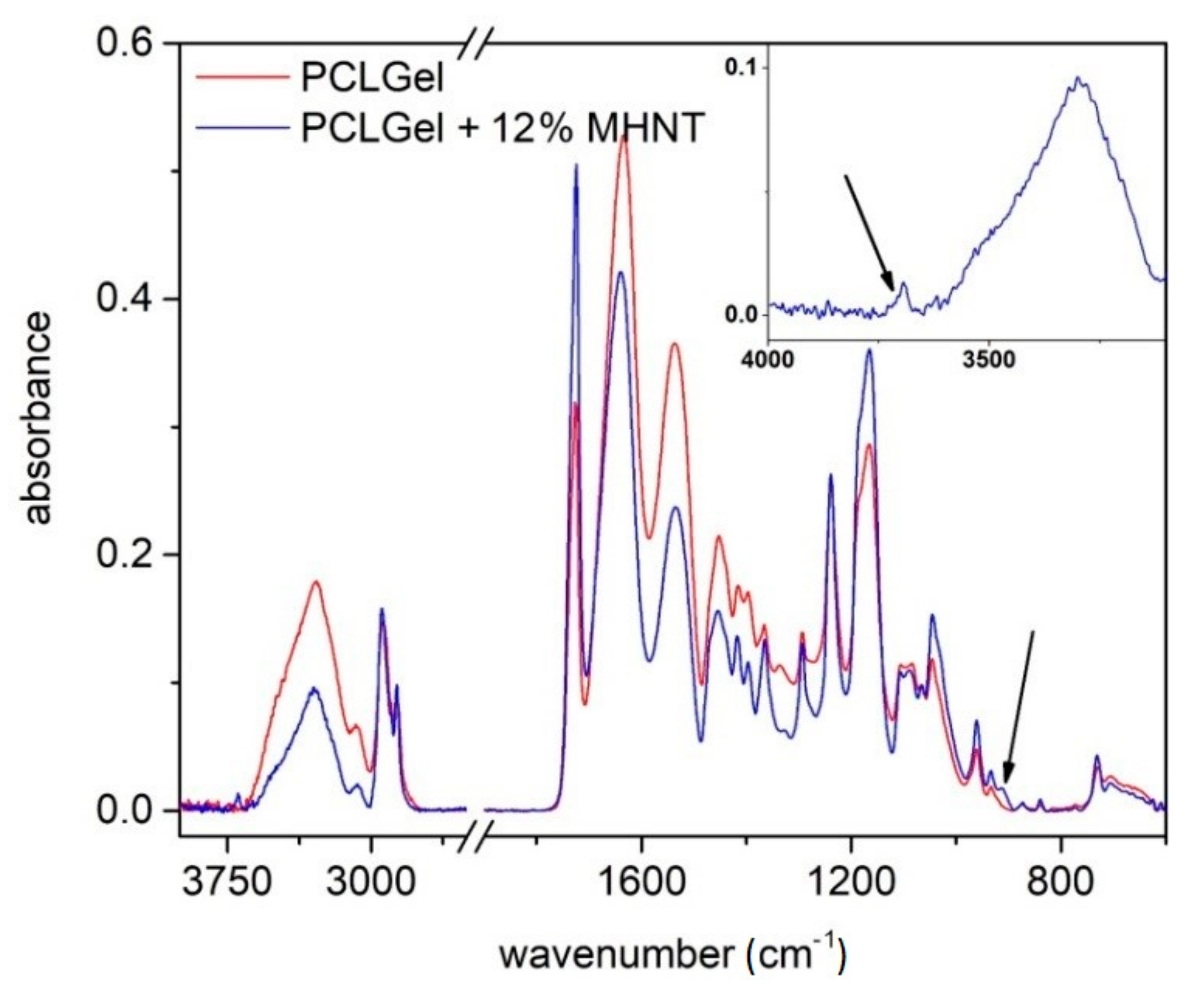
3.2. Structure and Composition of PCL/Gel/MHNT Nanofibers
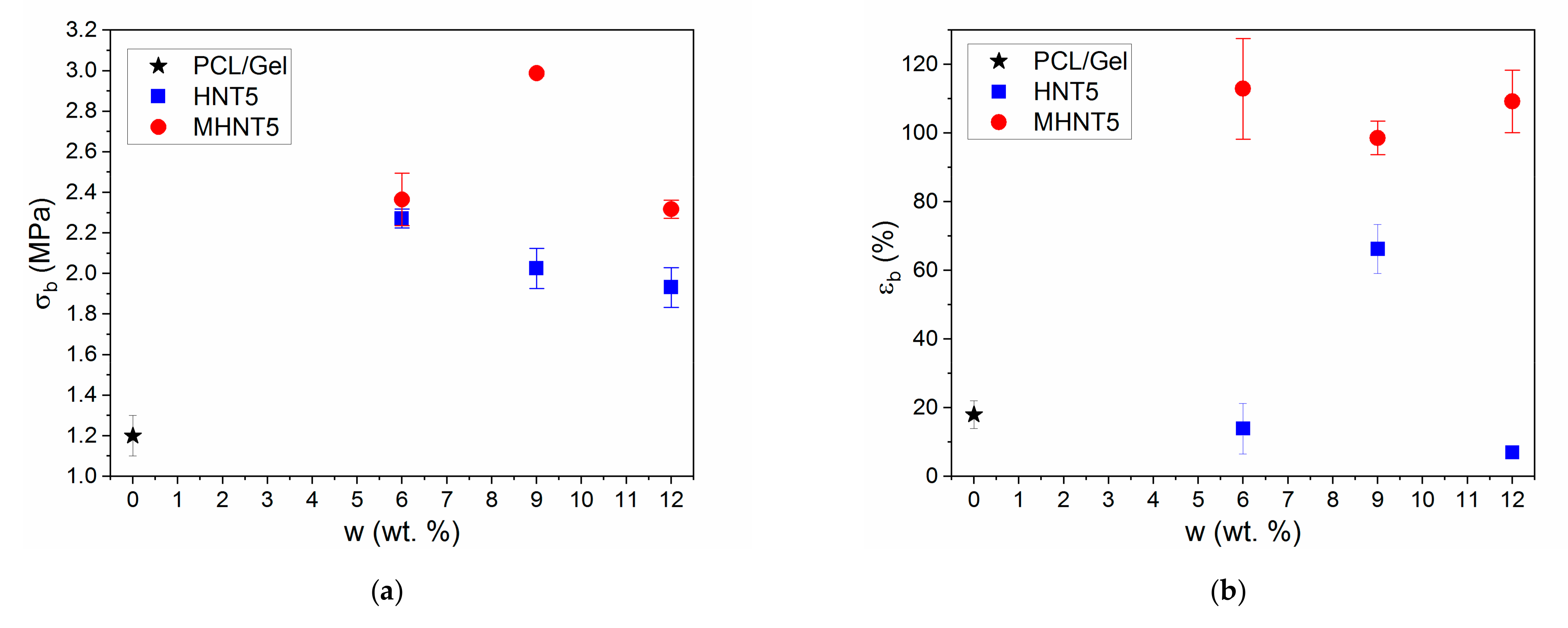
3.3. Mechanical Properties of PCL/Gel/MHNT Nanofibers
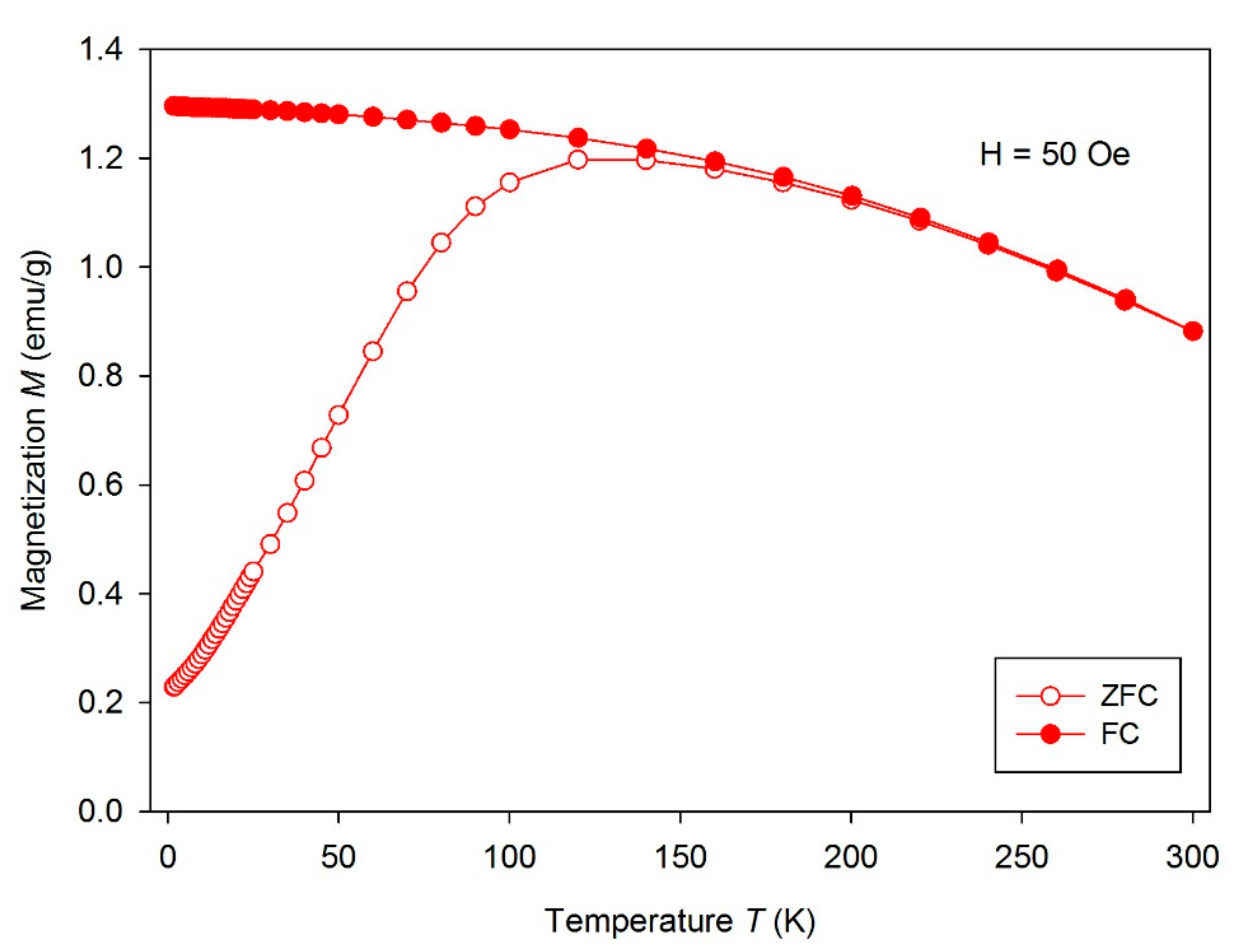
3.4. Magnetic Properties of PCL/Gel/MHNT Nanofibers
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Y.-W.; Zhan, Q.-F.; Li, R.-W. Fabrication, properties, and applications of flexible magnetic films. Chin. Phys. B 2013, 22, 127502. [Google Scholar] [CrossRef]
- Safarik, I.; Pospiskova, K.; Horska, K.; Safarikova, M. Potential of magnetically responsive (nano)biocomposites. Soft Matter 2012, 8, 5407–5413. [Google Scholar] [CrossRef]
- Nawaz, S.; Rashid, E.U.; Bagheri, A.R.; Aramesh, N.; Bhatt, P.; Ali, N.; Nguyen, T.A.; Bilal, M. Mitigation of environmentally hazardous pollutants by magnetically responsive composite materials. Chemosphere 2021, 276, 130241. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Zaman, H.; Bilal, M.; Shah, A.-u.-H.A.; Nazir, M.S.; Iqbal, H.M.N. Environmental perspectives of interfacially active and magnetically recoverable composite materials—A review. Sci. Total Environ. 2019, 670, 523–538. [Google Scholar] [CrossRef]
- Lalegül-Ülker, Ö.; Vurat, M.T.; Elçin, A.E.; Elçin, Y.M. Magnetic silk fibroin composite nanofibers for biomedical applications: Fabrication and evaluation of the chemical, thermal, mechanical, and in vitro biological properties. J. Appl. Polym. Sci. 2019, 136, 48040. [Google Scholar] [CrossRef]
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Melnikova, L.; Pospiskova, K.; Mitroova, Z.; Kopcansky, P.; Safarik, I. Peroxidase-like activity of magnetoferritin. Microchim. Acta 2014, 181, 295–301. [Google Scholar] [CrossRef]
- Safarik, I.; Prochazkova, J.; Schroer, M.A.; Garamus, V.M.; Kopcansky, P.; Timko, M.; Rajnak, M.; Karpets, M.; Ivankov, O.I.; Avdeev, M.V.; et al. Cotton textile/iron oxide manozyme composites with peroxidase-like activity: Preparation, characterization, and application. ACS Appl. Mater. Interfaces 2021, 13, 23627–23637. [Google Scholar] [CrossRef] [PubMed]
- Safarik, I.; Pospiskova, K.; Horska, K.; Maderova, Z.; Safarikova, M. Magnetically responsive (nano)biocomposites. In Intracellular Delivery; Prokop, A., Iwasaki, Y., Harada, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; Volume 2, pp. 17–34. [Google Scholar]
- Pospiskova, K.; Mohr, G.J.; Prochazkova, J.; Timko, M.; Rajnak, M.; Paulovicova, K.; Kopcansky, P.; Giovannini, G.; Boesel, L.F.; Safarik, I. Scalable production of magnetic fluorescent cellulose microparticles. Cellulose 2021, 28, 7675–7685. [Google Scholar] [CrossRef]
- Safarik, I.; Prochazkova, J.; Baldikova, E.; Timko, M.; Kopcansky, P.; Rajnak, M.; Torma, N.; Pospiskova, K. Modification of diamagnetic materials using magnetic fluids. Ukr. J. Phys. 2020, 65, 751. [Google Scholar] [CrossRef]
- Safarik, I.; Safarikova, M. One-step magnetic modification of non-magnetic solid materials. Int. J. Mater. Res. 2014, 105, 104–107. [Google Scholar] [CrossRef]
- Pospiskova, K.; Safarik, I. Low-temperature magnetic modification of sensitive biological materials. Mater. Lett. 2015, 142, 184–188. [Google Scholar] [CrossRef]
- Padmakumar, S.; Paul-Prasanth, B.; Pavithran, K.; Vijaykumar, D.K.; Rajanbabu, A.; Sivanarayanan, T.B.; Kadakia, E.; Amiji, M.M.; Nair, S.V.; Menon, D. Long-term drug delivery using implantable electrospun woven polymeric nanotextiles. Nanomed. Nanotechnol. Biol. Med. 2019, 15, 274–284. [Google Scholar] [CrossRef]
- Ignatova, M.; Rashkov, I.; Manolova, N. Drug-loaded electrospun materials in wound-dressing applications and in local cancer treatment. Expert Opin. Drug Deliv. 2013, 10, 469–483. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.G.; Yu, J.H.; Chen, L.; Williams, G.R.; Wang, X. Modified coaxial electrospinning for the preparation of high-quality ketoprofen-loaded cellulose acetate nanofibers. Carbohydr. Polym. 2012, 90, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Chen, S.; Li, W.; Lou, J.; Wang, J. Flurbiprofen axetil loaded coaxial electrospun poly(vinyl pyrrolidone)–nanopoly(lactic-co-glycolic acid) core–shell composite nanofibers: Preparation, characterization, and anti-adhesion activity. J. Appl. Polym. Sci. 2015, 132, 41982. [Google Scholar] [CrossRef]
- Huang, W.; Zou, T.; Li, S.; Jing, J.; Xia, X.; Liu, X. Drug-loaded zein nanofibers prepared using a modified coaxial electrospinning process. AAPS PharmSciTech 2013, 14, 675–681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhattarai, R.S.; Bachu, R.D.; Boddu, S.H.S.; Bhaduri, S. Biomedical applications of electrospun nanofibers: Drug and nanoparticle delivery. Pharmaceutics 2018, 11, 5. [Google Scholar] [CrossRef] [Green Version]
- Savva, I.; Odysseos, A.D.; Evaggelou, L.; Marinica, O.; Vasile, E.; Vekas, L.; Sarigiannis, Y.; Krasia-Christoforou, T. Fabrication, characterization, and evaluation in drug release properties of magnetoactive poly(ethylene oxide)–poly(l-lactide) electrospun membranes. Biomacromolecules 2013, 14, 4436–4446. [Google Scholar] [CrossRef] [PubMed]
- Molcan, M.; Safarik, I.; Pospiskova, K.; Paulovicova, K.; Timko, M.; Kopcansky, P.; Torma, N. Magnetically modified electrospun nanofibers for hyperthermia treatment. Ukr. J. Phys. 2020, 65, 655. [Google Scholar] [CrossRef]
- Faridi-Majidi, R.; Sharifi-Sanjani, N. In Situ synthesis of iron oxide nanoparticles on poly(ethylene oxide) nanofibers through an electrospinning process. J. Appl. Polym. Sci. 2007, 105, 1351–1355. [Google Scholar] [CrossRef]
- Prochazkova, J.; Pospiskova, K.; Safarik, I. Magnetically modified electrospun nanotextile exhibiting peroxidase-like activity. J. Magn. Magn. Mater. 2019, 473, 335–340. [Google Scholar] [CrossRef]
- Safarik, I.; Pospiskova, K.; Baldikova, E.; Savva, I.; Vekas, L.; Marinica, O.; Tanasa, E.; Krasia-Christoforou, T. Fabrication and bioapplications of magnetically modified chitosan-based electrospun nanofibers. Electrospinning 2018, 2, 29–39. [Google Scholar] [CrossRef]
- Jia, Y.; Yang, C.; Chen, X.; Xue, W.; Hutchins-Crawford, H.J.; Yu, Q.; Topham, P.D.; Wang, L. A review on electrospun magnetic nanomaterials: Methods, properties and applications. J. Mater. Chem. C 2021, 9, 9042–9082. [Google Scholar] [CrossRef]
- Ortolani, A.; Bianchi, M.; Mosca, M.; Caravelli, S.; Fuiano, M.; Marcacci, M.; Russo, A. The prospective opportunities offered by magnetic scaffolds for bone tissue engineering: A review. Joints 2017, 4, 228–235. [Google Scholar] [CrossRef] [Green Version]
- Nikolaou, M.; Avraam, K.; Kolokithas-Ntoukas, A.; Bakandritsos, A.; Lizal, F.; Misik, O.; Maly, M.; Jedelsky, J.; Savva, I.; Balanean, F.; et al. Superparamagnetic electrospun microrods for magnetically-guided pulmonary drug delivery with magnetic heating. Mater. Sci. Eng. C 2021, 126, 112117. [Google Scholar] [CrossRef]
- Chen, S.; Boda, S.K.; Batra, S.K.; Li, X.; Xie, J. Emerging roles of electrospun nanofibers in cancer research. Adv. Healthc. Mater. 2018, 7, e1701024. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Cáceres, R.; Cabeza, L.; Perazzoli, G.; Díaz, A.; López-Romero, J.M.; Melguizo, C.; Prados, J. Electrospun nanofibers: Recent applications in drug delivery and cancer therapy. Nanomaterials 2019, 9, 656. [Google Scholar] [CrossRef] [Green Version]
- Matos, A.M.; Gonçalves, A.I.; Rodrigues, M.T.; Miranda, M.S.; Haj, A.J.E.; Reis, R.L.; Gomes, M.E. Remote triggering of TGF-β/Smad2/3 signaling in human adipose stem cells laden on magnetic scaffolds synergistically promotes tenogenic commitment. Acta Biomater. 2020, 113, 488–500. [Google Scholar] [CrossRef]
- Gonçalves, A.I.; Rodrigues, M.T.; Carvalho, P.P.; Bañobre-López, M.; Paz, E.; Freitas, P.; Gomes, M.E. Exploring the potential of starch/polycaprolactone aligned magnetic responsive ccaffolds for tendon regeneration. Adv. Healthc. Mater. 2016, 5, 213–222. [Google Scholar] [CrossRef]
- Hao, S.; Meng, J.; Zhang, Y.; Liu, J.; Nie, X.; Wu, F.; Yang, Y.; Wang, C.; Gu, N.; Xu, H. Macrophage phenotypic mechanomodulation of enhancing bone regeneration by superparamagnetic scaffold upon magnetization. Biomaterials 2017, 140, 16–25. [Google Scholar] [CrossRef]
- Shuai, C.; Yang, W.; He, C.; Peng, S.; Gao, C.; Yang, Y.; Qi, F.; Feng, P. A magnetic micro-environment in scaffolds for stimulating bone regeneration. Mater. Des. 2020, 185, 108275. [Google Scholar] [CrossRef]
- Sapir, Y.; Polyak, B.; Cohen, S. Cardiac tissue engineering in magnetically actuated scaffolds. Nanotechnology 2014, 25, 014009. [Google Scholar] [CrossRef] [PubMed]
- Sapir, Y.; Cohen, S.; Friedman, G.; Polyak, B. The promotion of in vitro vessel-like organization of endothelial cells in magnetically responsive alginate scaffolds. Biomaterials 2012, 33, 4100–4109. [Google Scholar] [CrossRef] [Green Version]
- Caizer, C. Nanoparticle size effect on some magnetic properties. In Handbook of Nanoparticles; Aliofkhazraei, M., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 475–519. [Google Scholar]
- Sagare, R.D. Halloysite nanotubes: Design, characterization and applications. A review. Farmacia 2021, 69, 208–214. [Google Scholar] [CrossRef]
- Feldman, D. Polymer nanocomposites in medicine. J. Macromol. Sci. Part A 2016, 53, 55–62. [Google Scholar] [CrossRef]
- Kalia, S.; Kango, S.; Kumar, A.; Haldorai, Y.; Kumari, B.; Kumar, R. Magnetic polymer nanocomposites for environmental and biomedical applications. Colloid Polym. Sci. 2014, 292, 2025–2052. [Google Scholar] [CrossRef]
- Abdullayev, E.; Lvov, Y. Chapter 22—Halloysite for controllable loading and release. In Developments in Clay Science; Yuan, P., Thill, A., Bergaya, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; Volume 7, pp. 554–605. [Google Scholar]
- Steele, L.; Margolis, G.; Cohen, S.; Polyak, B. Chapter 11: Applications of magnetic-responsive materials for cardiovascular tissue engineering. In Smart Materials for Tissue Engineering: Applications; The Royal Society of Chemistry: London, UK, 2017; pp. 290–328. [Google Scholar]
- Riela, G.S.; Fakhrullin, R.F. Clay-based drug-delivery systems: What does the future hold? Ther. Deliv. 2017, 8, 633–646. [Google Scholar] [CrossRef]
- Yendluri, R.; Lvov, Y.; de Villiers, M.M.; Vinokurov, V.; Naumenko, E.; Tarasova, E.; Fakhrullin, R. Paclitaxel encapsulated in halloysite clay nanotubes for intestinal and intracellular delivery. J. Pharm. Sci. 2017, 106, 3131–3139. [Google Scholar] [CrossRef] [PubMed]
- Satish, S.; Tharmavaram, M.; Rawtani, D. Halloysite nanotubes as a nature’s boon for biomedical applications. Nanobiomedicine 2019, 6, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prinz Setter, O.; Segal, E. Halloysite nanotubes—The nano-bio interface. Nanoscale 2020, 12, 23444–23460. [Google Scholar] [CrossRef]
- Kushwaha, S.K.; Kushwaha, N.; Pandey, P.; Fatma, B. Halloysite nanotubes for nanomedicine: Prospects, challenges and applications. BioNanoScience 2021, 11, 200–208. [Google Scholar] [CrossRef]
- Kausar, A. Review on polymer/halloysite nanotube nanocomposite. Polym.-Plast. Technol. Eng. 2018, 57, 548–564. [Google Scholar] [CrossRef]
- Safarik, I.; Baldikova, E.; Prochazkova, J.; Safarikova, M.; Pospiskova, K. Magnetically modified agricultural and food waste: Preparation and application. J. Agric. Food Chem. 2018, 66, 2538–2552. [Google Scholar] [CrossRef]
- Janacek, D.; Kvitek, L.; Karlikova, M.; Pospiskova, K.; Safarik, I. Removal of silver nanoparticles with native and magnetically modified halloysite. Appl. Clay Sci. 2018, 162, 10–14. [Google Scholar] [CrossRef]
- Khunová, V.; Šafařík, I.; Škrátek, M.; Kelnar, I.; Tomanová, K. Biodegradable polymer nanocomposites based on natural nanotubes: Effect of magnetically modified halloysite on the behaviour of polycaprolactone. Clay Miner. 2018, 51, 435–444. [Google Scholar] [CrossRef]
- Khunová, V.; Pavliňáková, V.; Škrátek, M.; Šafařík, I.; Pavliňák, D. Magnetic halloysite reinforced biodegradable nanofibres: New challenge for medical applications. AIP Conf. Proc. 2018, 1981, 020074. [Google Scholar] [CrossRef]
- Türkeş, E.; Sağ Açıkel, Y. Synthesis and characterization of magnetic halloysite–chitosan nanocomposites: Use in the removal of methylene blue in wastewaters. Int. J. Environ. Sci. Technol. 2020, 17, 1281–1294. [Google Scholar] [CrossRef]
- Maleki, A.; Hajizadeh, Z.; Firouzi-Haji, R. Eco-friendly functionalization of magnetic halloysite nanotube with SO3H for synthesis of dihydropyrimidinones. Microporous Mesoporous Mater. 2018, 259, 46–53. [Google Scholar] [CrossRef]
- Hamza, H.; Ferretti, A.M.; Innocenti, C.; Fidecka, K.; Licandro, E.; Sangregorio, C.; Maggioni, D. An approach for magnetic halloysite nanocomposite with selective loading of superparamagnetic magnetite nanoparticles in the lumen. Inorg. Chem. 2020, 59, 12086–12096. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Qian, D.; Wu, D.; Ma, X. Magnetic halloysite nanotubes/iron oxide composites for the adsorption of dyes. Chem. Eng. J. 2011, 168, 959–963. [Google Scholar] [CrossRef]
- Rihova, M.; Ince, A.E.; Cicmancova, V.; Hromadko, L.; Castkova, K.; Pavlinak, D.; Vojtova, L.; Macak, J.M. Water-born 3D nanofiber mats using cost-effective centrifugal spinning: Comparison with electrospinning process: A complex study. J. Appl. Polym. Sci. 2021, 138, 49975. [Google Scholar] [CrossRef]
- Calisir, M.D.; Kilic, A. A comparative study on SiO2 nanofiber production via two novel non-electrospinning methods: Centrifugal spinning vs solution blowing. Mater. Lett. 2020, 258, 126751. [Google Scholar] [CrossRef]
- Heseltine, P.L.; Ahmed, J.; Edirisinghe, M. Developments in pressurized gyration for the mass production of polymeric fibers. Macromol. Mater. Eng. 2018, 303, 1800218. [Google Scholar] [CrossRef] [Green Version]
- Hassan, M.A.; Yeom, B.Y.; Wilkie, A.; Pourdeyhimi, B.; Khan, S.A. Fabrication of nanofiber meltblown membranes and their filtration properties. J. Membr. Sci. 2013, 427, 336–344. [Google Scholar] [CrossRef]
- Mahalingam, S.; Matharu, R.; Homer-Vanniasinkam, S.; Edirisinghe, M. Current methodologies and approaches for the formation of core–sheath polymer fibers for biomedical applications. Appl. Phys. Rev. 2020, 7, 041302. [Google Scholar] [CrossRef]
- Yu, M.; Dong, R.-H.; Yan, X.; Yu, G.-F.; You, M.-H.; Ning, X.; Long, Y.-Z. Recent advances in needleless electrospinning of ultrathin fibers: From academia to industrial production. Macromol. Mater. Eng. 2017, 302, 1700002. [Google Scholar] [CrossRef]
- Persano, L.; Camposeo, A.; Tekmen, C.; Pisignano, D. Industrial upscaling of electrospinning and applications of polymer nanofibers: A review. Macromol. Mater. Eng. 2013, 298, 504–520. [Google Scholar] [CrossRef]
- Pavliňáková, V.; Fohlerová, Z.; Pavliňák, D.; Khunová, V.; Vojtová, L. Effect of halloysite nanotube structure on physical, chemical, structural and biological properties of elastic polycaprolactone/gelatin nanofibers for wound healing applications. Mater. Sci. Eng. C 2018, 91, 94–102. [Google Scholar] [CrossRef]
- Safarik, I.; Lunackova, P.; Mosiniewicz-Szablewska, E.; Weyda, F.; Safarikova, M. Adsorption of water-soluble organic dyes on ferrofluid-modified sawdust. Holzforschung 2007, 61, 247–253. [Google Scholar] [CrossRef]
- Massart, R. Preparation of aqueous magnetic liquids in alkaline and acidic media. IEEE Trans. Magn. 1981, 17, 1247–1248. [Google Scholar] [CrossRef]
- Kiwada, H.; Sato, J.; Yamada, S.; Kato, Y. Feasibility of magnetic liposomes as a targeting device for drugs. Chem. Pharm. Bull. 1986, 34, 4253–4258. [Google Scholar] [CrossRef] [Green Version]
- Luo, P.; Zhao, Y.; Zhang, B.; Liu, J.; Yang, Y.; Liu, J. Study on the adsorption of Neutral Red from aqueous solution onto halloysite nanotubes. Water Res. 2010, 44, 1489–1497. [Google Scholar] [CrossRef]
- Jancar, J.; Ondreas, F.; Lepcio, P.; Zboncak, M.; Zarybnicka, K. Mechanical properties of glassy polymers with controlled NP spatial organization. Polym. Test. 2020, 90, 106640. [Google Scholar] [CrossRef]
- Dorigato, A.; D’Amato, M.; Pegoretti, A. Thermo-mechanical properties of high density polyethylene—Fumed silica nanocomposites: Effect of filler surface area and treatment. J. Polym. Res. 2012, 19, 9889. [Google Scholar] [CrossRef]
- Fu, S.; Sun, Z.; Huang, P.; Li, Y.; Hu, N. Some basic aspects of polymer nanocomposites: A critical review. Nano Mater. Sci. 2019, 1, 2–30. [Google Scholar] [CrossRef]
- Ondreas, F.; Lepcio, P.; Zboncak, M.; Zarybnicka, K.; Govaert, L.E.; Jancar, J. Effect of nanoparticle organization on molecular mobility and mechanical properties of polymer nanocomposites. Macromolecules 2019, 52, 6250–6259. [Google Scholar] [CrossRef]
- Zarybnicka, K.; Ondreas, F.; Lepcio, P.; Kalina, M.; Zboncak, M.; Jancar, J. Thermodynamic parameters controlling nanoparticle spatial packing in polymer solutions. Macromolecules 2020, 53, 8704–8713. [Google Scholar] [CrossRef]
- Lepcio, P.; Ondreas, F.; Zarybnicka, K.; Zboncak, M.; Caha, O.; Jancar, J. Bulk polymer nanocomposites with preparation protocol governed nanostructure: The origin and properties of aggregates and polymer bound clusters. Soft Matter 2018, 14, 2094–2103. [Google Scholar] [CrossRef]
- Zboncak, M.; Ondreas, F.; Uhlir, V.; Lepcio, P.; Michalicka, J.; Jancar, J. Translation of segment scale stiffening into macroscale reinforcement in polymer nanocomposites. Polym. Eng. Sci. 2020, 60, 587–596. [Google Scholar] [CrossRef]
- Jancar, J.; Douglas, J.F.; Starr, F.W.; Kumar, S.K.; Cassagnau, P.; Lesser, A.J.; Sternstein, S.S.; Buehler, M.J. Current issues in research on structure–property relationships in polymer nanocomposites. Polymer 2010, 51, 3321–3343. [Google Scholar] [CrossRef]
- Lepcio, P.; Ondreáš, F.; Zárybnická, K.; Zbončák, M.; Svatík, J.; Jančář, J. Phase diagram of bare particles in polymer nanocomposites: Uniting solution and melt blending. Polymer 2021, 230, 124033. [Google Scholar] [CrossRef]




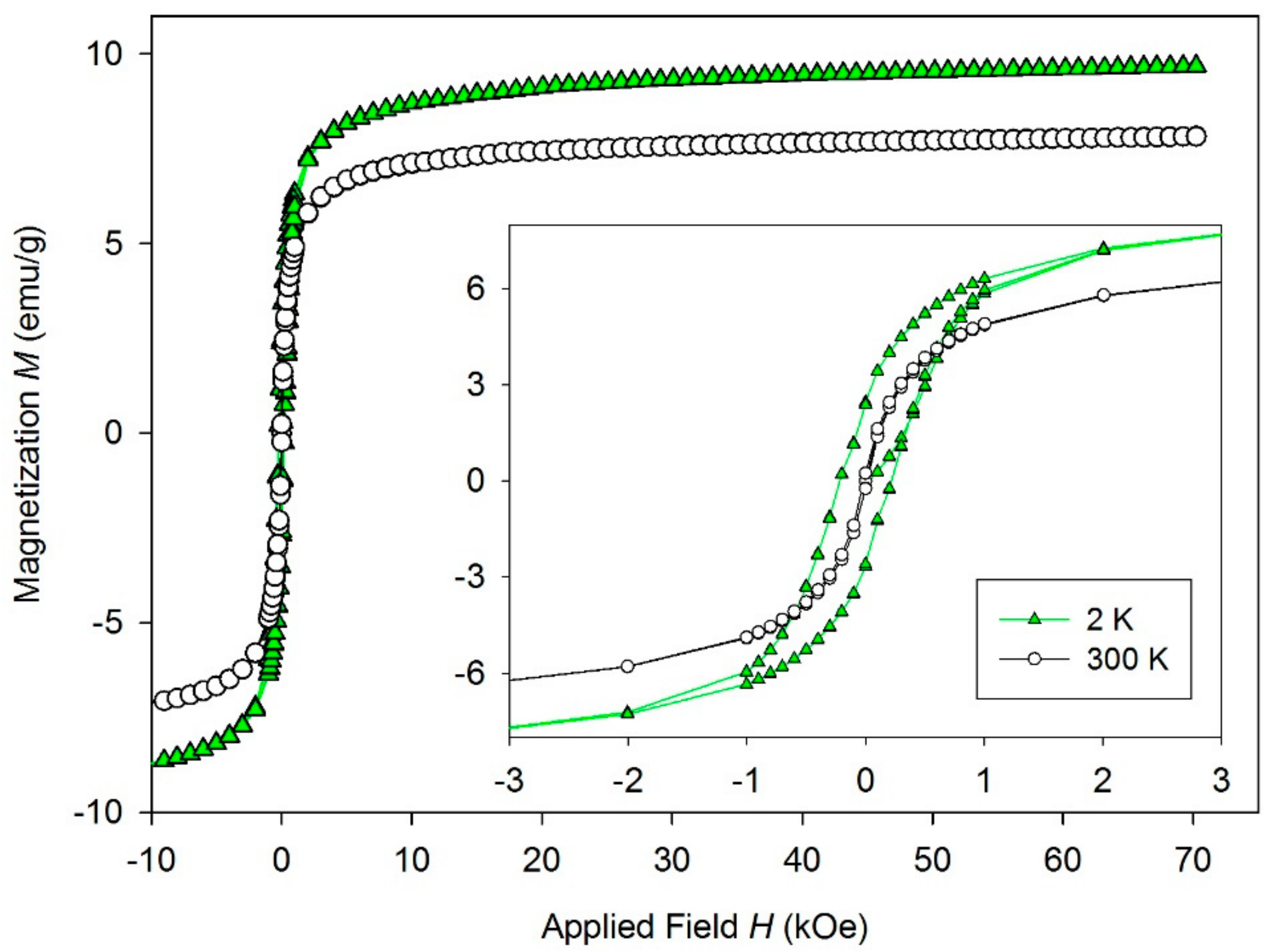
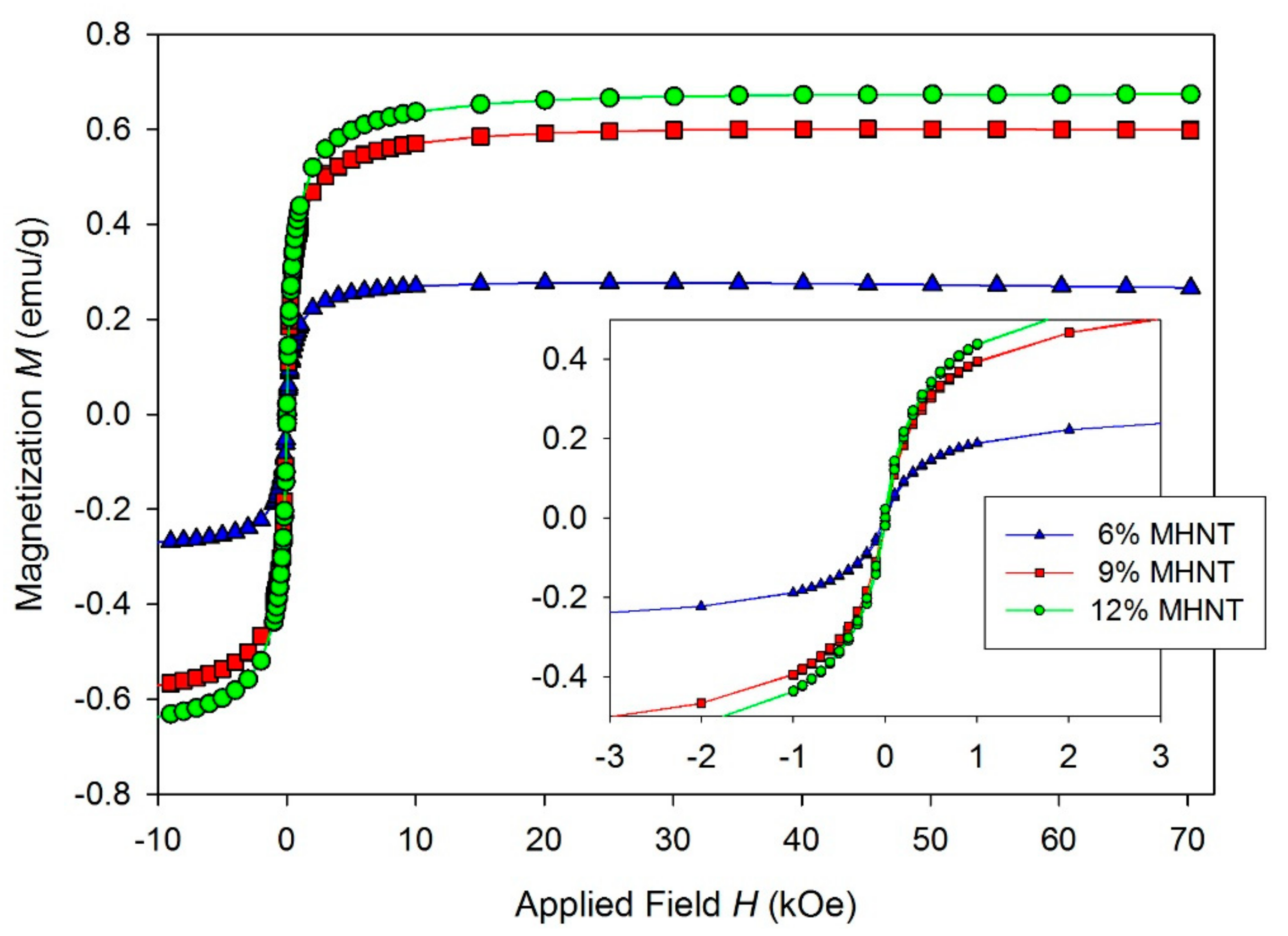
| wt.% | C | N | O | Al | Si | Fe |
|---|---|---|---|---|---|---|
| HNT | 0.0 | 0.0 | 64.9 | 17.6 | 15.5 | <0.3 |
| MHNT | 0.0 | 0.0 | 64.0 | 14.9 | 13.2 | 8.2 |
| PCL/Gel/MHNT (6–12 wt.%) | 56.3 | 16.8 | 24.8 | 0.6 | 0.5 | 0.3–0.5 |
| Sample | MS (emu/g) | MR (emu/g) | HC (Oe) | TB (K) |
|---|---|---|---|---|
| MHNT | 7.8 (9.6) | 0.22 (2.5) | 16 (216) | 120 K |
| 6 wt.% | 0.27 | 0.009 | 14 | - |
| 9 wt.% | 0.6 | 0.019 | 15 | - |
| 12 wt.% | 0.67 | 0.021 | 15 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khunová, V.; Pavliňák, D.; Šafařík, I.; Škrátek, M.; Ondreáš, F. Multifunctional Electrospun Nanofibers Based on Biopolymer Blends and Magnetic Tubular Halloysite for Medical Applications. Polymers 2021, 13, 3870. https://doi.org/10.3390/polym13223870
Khunová V, Pavliňák D, Šafařík I, Škrátek M, Ondreáš F. Multifunctional Electrospun Nanofibers Based on Biopolymer Blends and Magnetic Tubular Halloysite for Medical Applications. Polymers. 2021; 13(22):3870. https://doi.org/10.3390/polym13223870
Chicago/Turabian StyleKhunová, Viera, David Pavliňák, Ivo Šafařík, Martin Škrátek, and František Ondreáš. 2021. "Multifunctional Electrospun Nanofibers Based on Biopolymer Blends and Magnetic Tubular Halloysite for Medical Applications" Polymers 13, no. 22: 3870. https://doi.org/10.3390/polym13223870
APA StyleKhunová, V., Pavliňák, D., Šafařík, I., Škrátek, M., & Ondreáš, F. (2021). Multifunctional Electrospun Nanofibers Based on Biopolymer Blends and Magnetic Tubular Halloysite for Medical Applications. Polymers, 13(22), 3870. https://doi.org/10.3390/polym13223870







