Abstract
Depositing minute light emitters into a regular array is a basic but essential technique in display technology. However, conventional lithographic methodologies involve multistep and energy-consuming processes. Here, we develop a facile method in which organic and polymeric fluorescent dyes spontaneously aggregate to form a patterned microarray. We find that a thin film of fluorescent π-conjugated polymer transforms into micrometer-sized aggregates when exposed to binary organic vapor at ambient temperature. The arrayed microaggregates can be formed over the whole substrate surface when using a quartz substrate that is prepatterned with regular hydrophilic boxes and hydrophobic grids. The resultant microarray is applicable to optical memories and displays when photoswitchable fluorophores are doped into the polymer matrix.
1. Introduction
Regular arrays of micro- and nanometer-sized devices integrated with light emitters enable the multipixeled displaying or collective assay of trace amounts of chemical or biological compounds [1,2,3]. The structural and electrical complexity of these arrays are usually realized by means of photo- or electron-beam lithography techniques. However, these top-down techniques are costly in terms of, for instance, the facilities required for the fabrication, the time and labor devoted for multistep processing, and the chemicals utilized for the protecting and rinsing steps. These drawbacks can be readily circumvented by using their counterpart bottom-up technology. The bottom-up method relies on the self-assembling behavior of small molecules and thus requires a low energy and simple procedure, which is especially useful for the fabrication of submicron-scale objects. In this field, a major focus is currently put on the arbitral molding and positioning of the nano-objects over a wide area [4,5,6]. Several successful examples have recently been reported, but they typically involve complex devices and enormous energy and time consumption during the fabrication process [7,8].
In this article, we report a facile method for the fabrication of regular device arrays by combining the advantages of the top-down and bottom-up strategies. We arbitrarily pattern a substrate with a hydrophobic surface modifier. Fluorescent polymers were spin-cast onto the prepatterned substrate to form a homogeneous thin film. Upon exposure to a binary vapor of good- and nonsolvents with an appropriate mixing ratio (cosolvent vapor annealing, coSVA), the polymer film spontaneously transforms into an array of microdisks. This technique does not involve any developing or molding processes, which drastically reduce the difficulty and complexity of the patterning procedure in comparison to the typical lithography process. Moreover, the fluorescence of the microdisks can be switched by doping photochromic fluorescent diarylethenes, which alter the fluorescence of the host polymer via a photoinduced energy transfer. The rich functionalities that are achievable with the coSVA method are highly valuable for constructing micrometer-scale optical memories, displays and sensing devices.
2. Materials and Methods
2.1. Materials and Settings
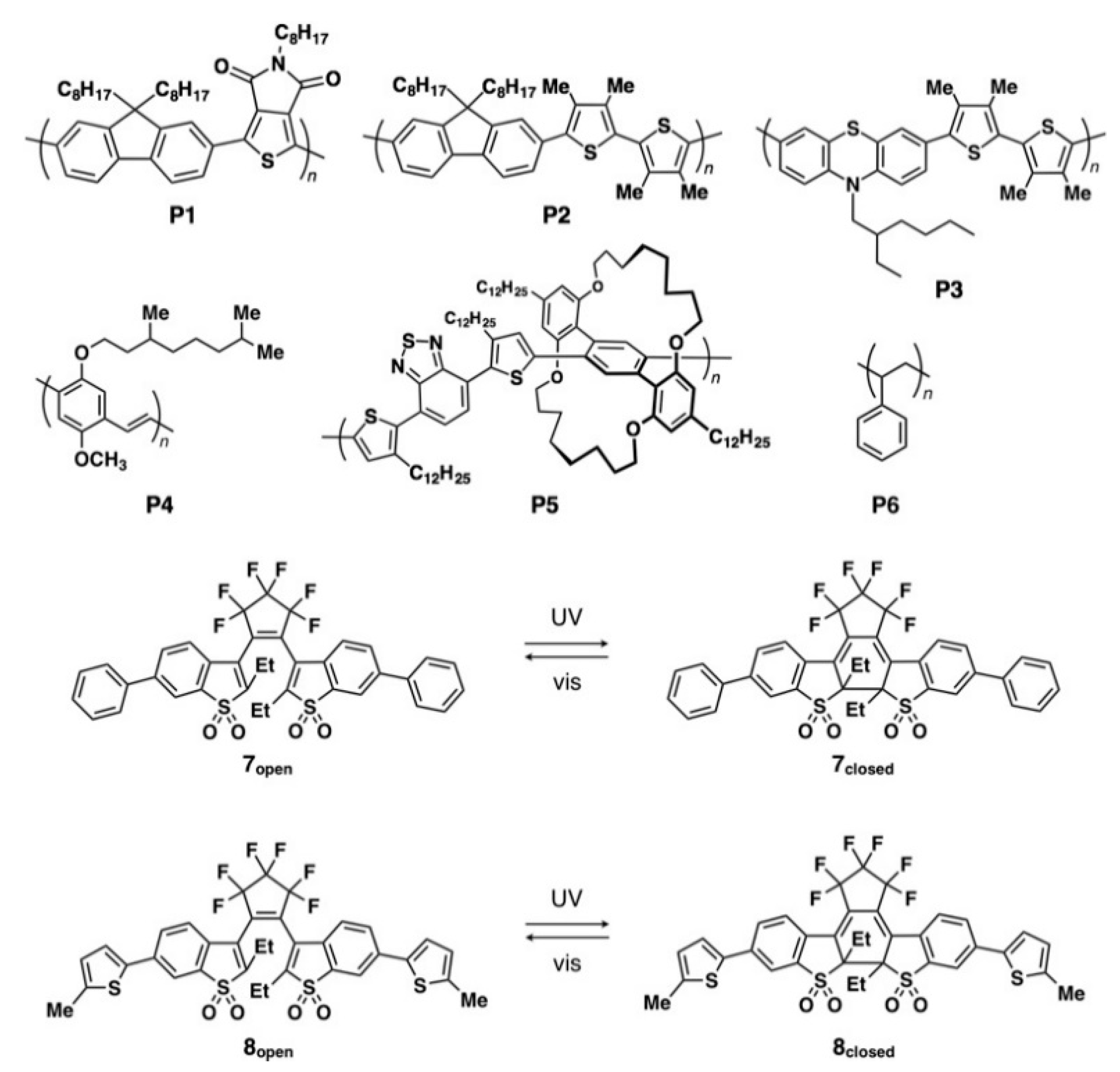
π-Conjugated polymers P1 (poly[(9,9-dioctylfluorene-2,7-diyl)-alt-(5-octylthieno[3,4-c]pyrrole-4,6-dione-1,3-diyl)], number-averaged molecular weight (Mn) = 43,000 g mol−1, polydispersity index (PDI) = 2.36), P2 (poly[(9,9-dioctylfuorenyl-2,7-diyl)-alt-(3,3′,4,4′-tetramethylbithiophene-2,5′-diyl)], Mn = 31,800 g mol−1, PDI = 2.46), P3 (poly[(N-(2-ethylhexyl)phenothiazine-3,7-diyl)-alt-(3,3′,4,4′-tetramethylbithiophene-2,5′-diyl)], Mn = 21,000 g mol−1, PDI = 2.82), P5 (an alternating copolymer containing a phenylene moiety covered with 1,10,17,26-tetraoxa[10.10]metacyclophane as one part and 3-n-dodecylthiophene-alt-benzothiadiazole-3′-n-dodecylthiophene as the counterpart, Mn = 8600 g mol−1, Mw/Mn = 1.2), and photochromic diarylethenes 7 and 8 were synthesized according to the reported procedures (Figure 1) [9,10,11,12,13,14,15,16,17]. π-Conjugated polymer P4 (poly[2-methoxy-5-(3′,7′-dimethyloctyloxy)-1,4-phenylenevinylene]) and nonconjugated polystyrene P6 were purchased from Aldrich Co. Ltd. (Figure 1). Unless otherwise noted, all solvents and reagents were used as purchased. Steady-state photoluminescence (PL) spectra were measured on a JASCO FP-6200 spectrofluorometer. Optical and fluorescent microscopic observations were carried out using an Olympus model BX53 Upright Microscope. Quartz and SiO2 (200 nm)-covered silicon (Si) were used as a substrate. Atomic force microscopy (AFM) measurements were conducted on SII-Nanotechnology model S-image scanning probe microscopy.
Figure 1.
Molecular structures of π-conjugated polymers P1–P5, nonconjugated polystyrene P6 and photochromic diarylethenes 7 and 8.
2.2. Cosolvent Vapor Annealing (coSVA)
Typically, 1 mg of conjugated polymer was dissolved in CHCl3 (1 mL), and the solution (20 µL) was spin-cast on a quartz or Si substrate (2000 rpm, 40 s). The thickness of the polymer film was 3–10 nm. A 50-mL vial containing 0.6 mL of binary solution of CHCl3 and MeOH was sealed with a cap, where a polymer-coated substrate was adhered at the backside of the cap. Then, the vial stood at 30 °C for 1 to 18 h to carry out the cosolvent vapor annealing (coSVA, Figure 2a).
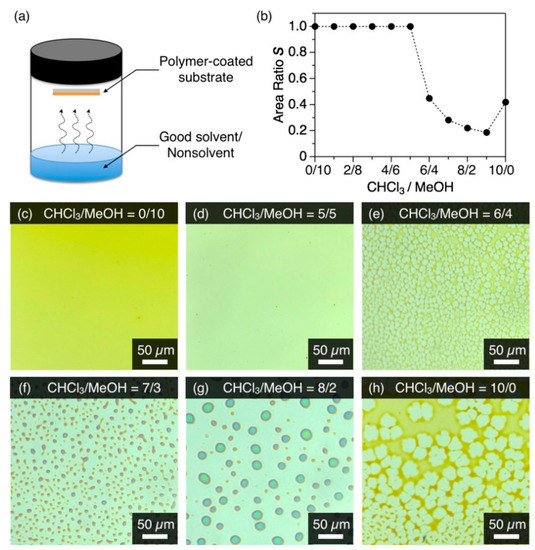
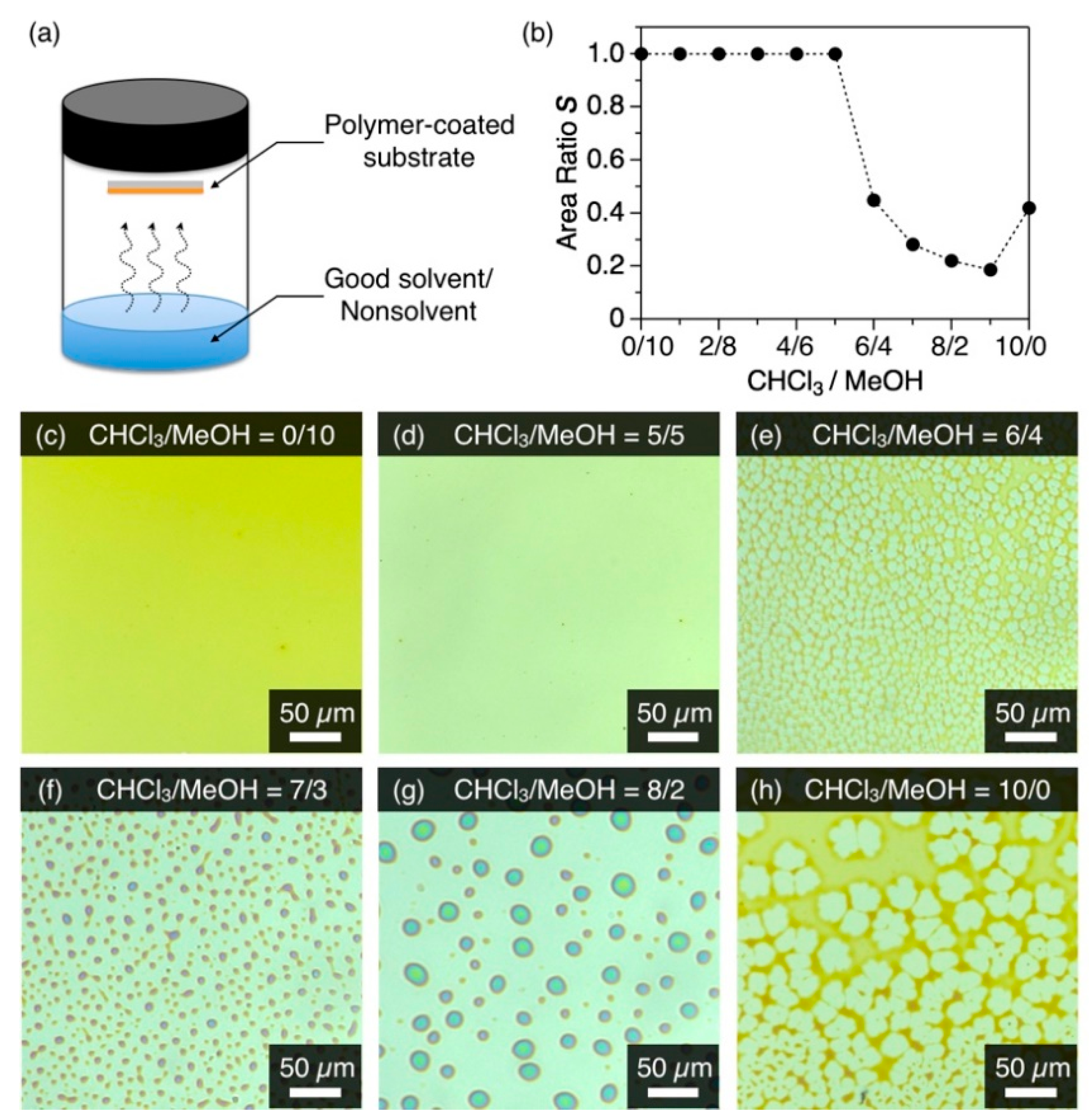
Figure 2.
(a) Schematic representation of cosolvent vapor annealing (coSVA) method. (b) Plot of the area ratio S of P1 versus CHCl3/MeOH (v/v) utilized for coSVA. (c–h) Optical microscope images of spin-cast films of P1 after coSVA for 4 h with CHCl3/MeOH of (c) 0/10, (d) 5/5, (e) 6/4, (f) 7/3, (g) 8/2, and (h) 10/0.
2.3. Hydrophobic/Hydrophilic Micropatterning on a Substrate
A quartz or Si substrate was immersed into a CHCl3 solution (5 mL) containing hexamethyldisilazane (HMDS, 10 µL). After 12 h, the substrate was fully covered with a monolayer of methyl group that enhanced the hydrophobicity of the surface. The hydrophobic/hydrophilic cross micropattern was fabricated by irradiating parallel vacuum ultraviolet (PVUV) light (λ = 150–200 nm, 15 Hz, 130 s, pulse duration: 10 ns) through a photomask with chromium cross-pattern (width of the boxes and lines: 3 and 2 µm, respectively). The intense PVUV light selectively removes the methyl group on the substrate to form a hydrophilic box array pattern on the surface (Figure S1) [18].
3. Results and Discussions
3.1. Fabrication of Microstructures by coSVA Method
We previously reported that conjugated polymers P1–P5 (Figure 1) self-assemble into microspherical particles upon sluggish precipitation from solution, which is driven by the diffusion of nonsolvent into the polymer solution [19,20,21,22]. However, it is difficult to arrange the resultant microspheres into a regular array on a substrate. To overcome this problem, we attempt the direct transformation of the morphology of the polymers from a thin film to minute disks on a substrate surface by the solvent vapor annealing (SVA) method [23].
A spin-cast film of P1 on a bare quartz substrate was exposed to a binary mixture of CHCl3 and MeOH vapor for the coSVA method. When the composition of the vapor, CHCl3/MeOH (v/v), was less than 6/4, the thin film hardly displayed a morphological change even after a 4 h-exposure to the vapor (Figure 2c,d). In contrast, the thin film transformed into a reticular or dotted pattern when CHCl3/MeOH was larger than 6/4 (Figure 2e–h). The morphological transformation of the film upon coSVA was quantitatively assessed in terms of the area ratio S, which was given by the coverage area of the film P1 divided by the whole area observed. The plot of S against the CHCl3/MeOH ratio showed a clear threshold at CHCl3/MeOH = 6/4 and immediately hit a plateau thereafter with a coverage ratio of 0.2 (Figure 2b and Figure S2). When only CHCl3 vapor was exposed, the S value increased slightly to 0.4. Considering that CHCl3 is a good solvent for P1, the transformation of the film at a higher CHCl3 content is triggered by the partial dewetting of the film with CHCl3 and subsequent fluidization. MeOH, a nonsolvent for P1, does not fluidize the film but plausibly accelerates the transformation of the film into a separated microdisk morphology.
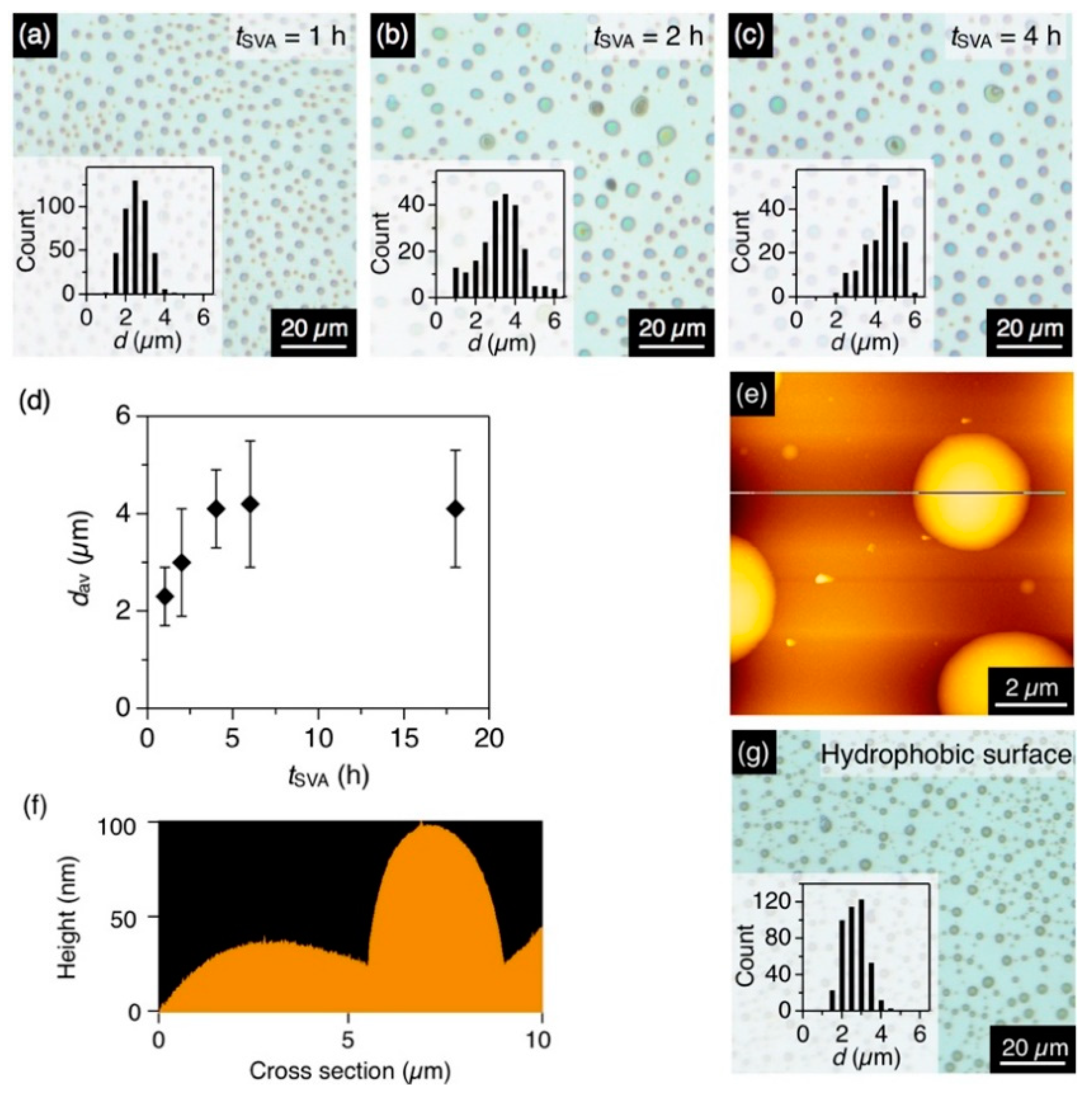
The time-dependent observation of the structural transformation was conducted with a binary vapor (CHCl3/MeOH = 1/0.5, v/v). After 1 h of coSVA, microdisks formed with an average diameter (dav) of 2.3 µm and a standard deviation (σ) of 0.6 µm (Figure 3a). Upon prolonging the annealing time (tSVA), dav increased and hit a plateau of 4.1 µm (σ = 0.8 µm) in 4 h (Figure 3b–d). The average height of the microdisks was ~100 nm according to the height profiles of the AFM images (Figure 3e,f).
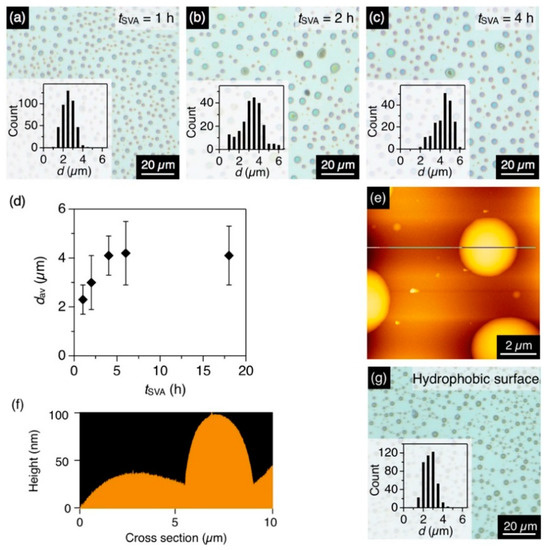
Figure 3.
(a–c) Optical micrographs of the spin-cast films of P1 with a tSVA of (a) 1 h, (b) 2 h and (c) 4 h. The CHCl3/MeOH ratio is 1/0.5. Insets show histograms of d of the resultant microdisks. (d) Plot of dav of the microdisk of P1 versus tSVA. (e,f): (e) AFM image and (f) its cross-section profile of the microdisk of P1. (g) Optical micrographs of the spin-cast films of P1 on an HMDS-treated hydrophobic quartz substrate with a tSVA of 4 h. The CHCl3/MeOH ratio is 1/0.5. The inset shows histograms of d of the resultant microdisks.
A hydrophobic quartz substrate, treated with HMDS, also afforded microdisks upon coSVA (CHCl3/MeOH = 1/0.5 v/v), while the dav of the resultant microdisks was 2.4 µm, which was smaller than that observed on a bare quartz substrate (4.0 µm, Figure 3g). This is because of the lesser affinity of the hydrophobic surface with P1. P1 tend to aggregate with each other tightly to form smaller microdisks on the hydrophobic surface.
3.2. Fabrication of Microstructures by coSVA Method
Patterned surfaces of quartz and Si substrates were prepared by methylation of the surface and subsequent selected-area demethylation by PVUV irradiation through a patterned photomask. The hydrophobically modified substrate was fabricated as described in Section 2.3. PVUV light was irradiated to the substrate through a cross-patterned photomask. The methyl group on the substrate was photochemically removed upon exposure to PVUV light to form hydrophilic box patterns (Figure 4a and Figure S1). The patterned substrate was then coated with polymer P1, for instance, and subjected to coSVA to form a regular array (Figure 4b).
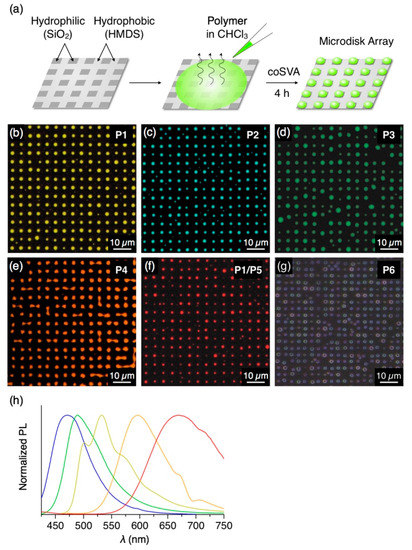
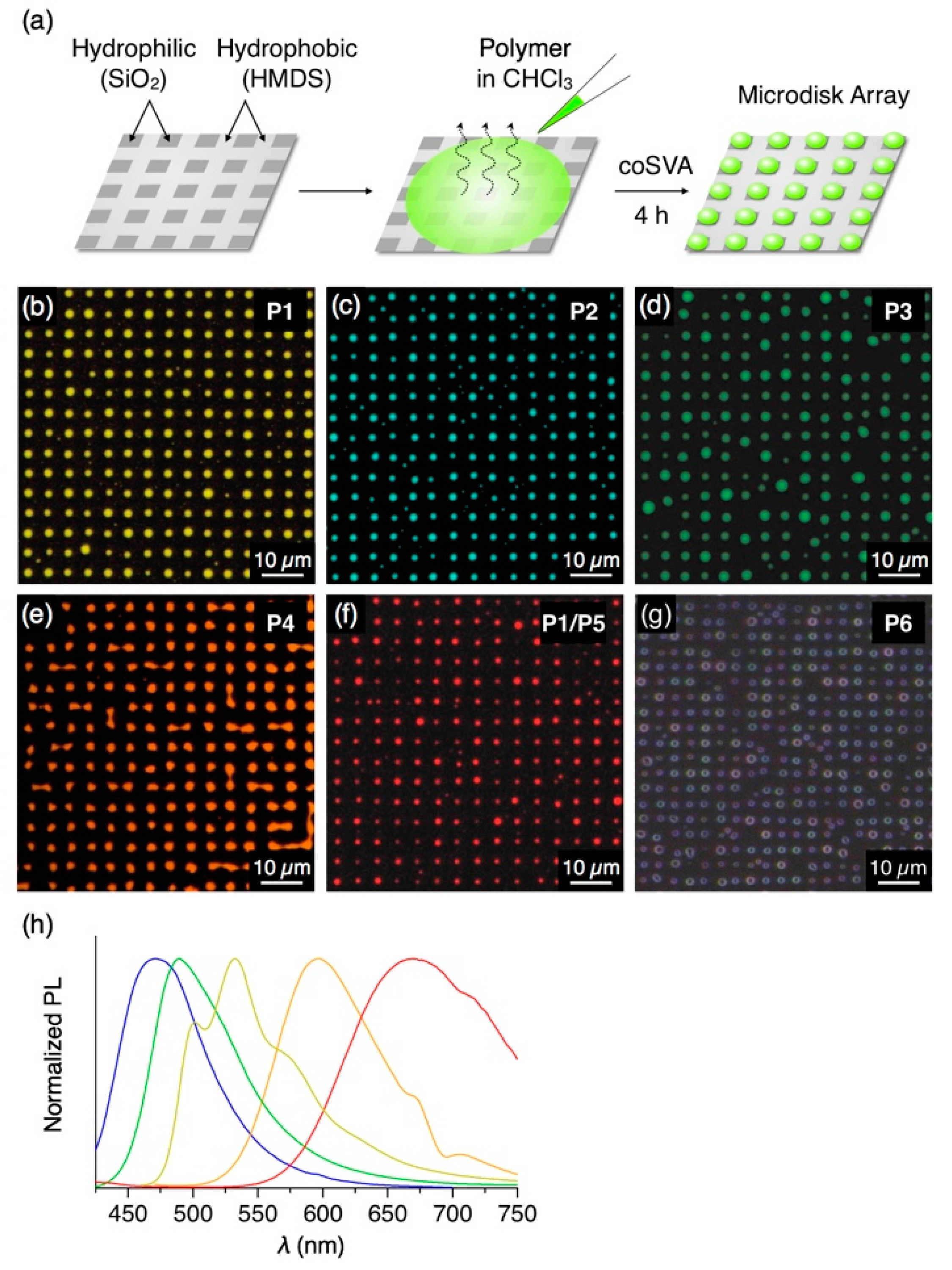
Figure 4.
(a) Schematic representation of the preparation procedure of the microdisk array of P1–P6 by coSVA on a hydrophobic/hydrophilic patterned substrate. (b–f) Fluorescent micrographs (λex = 400–440 nm) of microdisk arrays of (b) P1, (c) P2, (d) P3, (e) P4, (f) P1/P5 (7/3 w/w) and (g) P6 prepared by coSVA of the corresponding spin-cast films with tSVA = 4 h. (h) PL spectra (λex = 400 nm) of thin films of P1 (yellow), P2 (blue), P3 (green), P4 (orange) and P1/P5 (red) prepared by drop-cast from CHCl3 solutions of polymers on a quartz substrate.
In an analogous manner to P1, π-conjugated polymers P2–P4, featuring blue, green and orange fluorescence, respectively (Figure 4h), were treated with the coSVA method, successfully resulting in the formation of microdisk arrays (CHCl3/MeOH = 1/0.5, annealing time: 4 h, Figure 4c–e). In contrast, P5 with red PL color hardly yields well-ordered microdisk arrays, despite the fact that P5 forms microspheres by a vapor diffusion self-assembly process in solution [21]. Meanwhile, P5 has a high miscibility with other π-conjugated polymers [12] and features an absorption band in 400–550 nm overlapping with the PL band of P1 (Figure S3), which is advantageous for using P5 as a dopant of P1 to give red PL through energy transfer from P1 to P5. With this concept in mind, we conducted coSVA with a spin-cast film of a mixture of P1 and P5 (7/3 w/w) and found the formation of the analogous microdisk arrays (Figure 4f). By doping P5 with a weight ratio of P5 of more than 20% (Figure S4), the fluorescent color of the microdisks turns to red via photoinduced energy transfer from P1 to P5. Nonconjugated polystyrene P6 also forms microdisk arrays via the coSVA method (Figure 4g).
3.3. Fluorescence Switching and Color Change
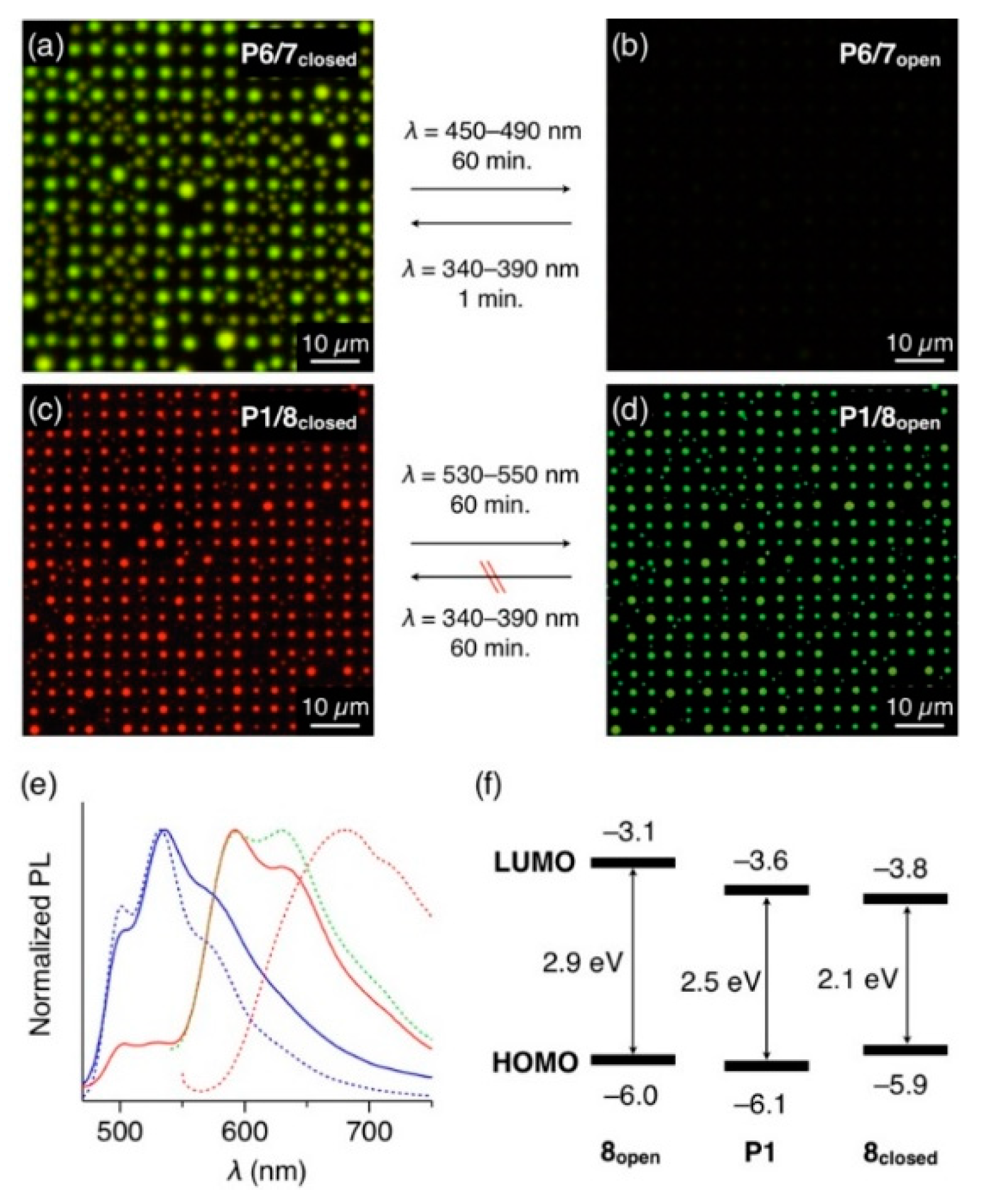
The turn-on fluorescence property and its color switching are of practical importance, especially for the development of optical displays and memories. To this end, we incorporated photoswitchable diarylethenes 7 and 8 as fluorescent dopants into the polymer media. Closed-form isomers of 7 and 8 (7closed and 8closed) are emissive with yellow and red colors, respectively, while their open-form isomers (7open and 8open) are nonemissive under excitation with 400–440-nm light [13,14,15,16,17].
Luminescence on/off switching of the polymer microdisk arrays was achieved from arrays of P6 doped with 7. A mixture of P6 and diarylethene 7closed (10 wt.%) dissolved in CHCl3 was spin-cast on a patterned substrate. The resultant film was then subjected to coSVA (CHCl3/MeOH = 1/0.5) to form a microdisk array with yellowish-green PL (Figure 5a). Upon photoirradiation (λ = 450–490 nm) for 60 min, 7closed photo-isomerized into 7open, and the microdisks turned nonemissive (Figure 5b). Conversely, upon irradiation of UV light (λ = 340–390 nm), fluorescence was recovered within 1 min by the backward photo-isomerization.
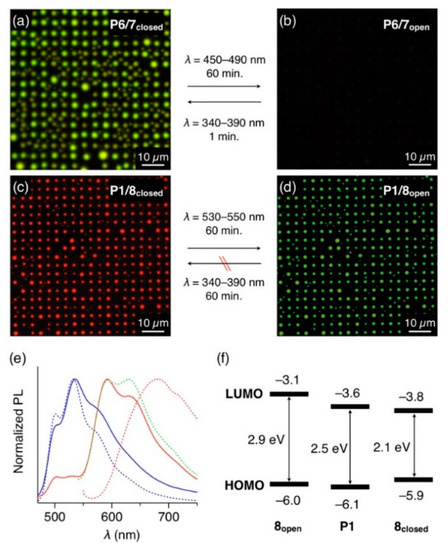
Figure 5.
(a,b) Fluorescent micrographs (λex = 400–440 nm) of microdisk arrays of 7closed-doped (10 wt.%) P6 (a) before and (b) after photoirradiation at 450–490 nm. (c,d) Fluorescent micrographs (λex = 400–440 nm) of microdisk arrays of 8closed-doped (10 wt.%) P1 (c) before and (d) after photoirradiation at 530–550 nm. (e) PL spectra (λex = 400 nm) of thin films of P1 doped with 10 wt.% of 8open (blue, solid line) and 8closed (red, solid line), prepared by drop-cast of CHCl3 solutions of P1 and 8 on a quartz substrate. The dashed lines show PL spectra of thin films of P1 (blue) and 8closed (red). The green dashed line shows the PL spectrum of a CHCl3 solution of 8closed. (f) HOMO and LUMO energy levels of P1, 8open, 8closed, obtained from a cyclic voltammogram.
Since 8closed acts as an energy acceptor for P1, the PL color of P1/8 mixed film is switchable by the photoisomerization of 8. In fact, the 8closed-doped P1 microdisk array displays red PL upon photoexcitation of P1 at λex = 400–440 nm (Figure 5c). The PL spectrum of the microdisks matches well with those of the solution of 8closed, not with the aggregate state of 8closed, indicating that 8closed is well-dispersed in the P1 medium and emits luminescence via energy transfer from P1 (Figure 5e). Upon photoirradiation at 530–550 nm, 8closed isomerized into nonemissive 8open. Accordingly, the PL color of the array changed from red to yellowish green, which was attributed to the PL of P1 (Figure 5d). However, the reverse photoisomerization reaction (from 8open to 8closed) barely occurred in the P1 matrix, even with a 60-min exposure to UV light. This is possibly because energy transfer from 8open to P1 occurs prior to the photoisomerization of 8open to 8closed, considering the HOMO and LUMO energy levels of P1 and 8open (Figure 5f), determined by their cyclic voltammetry (Figure S5) and photoabsorption spectra (Figure S6).
4. Conclusions
The newly developed cosolvent vapor annealing (coSVA) method is effective for the fabrication of regularly deposited microdisks. The morphology of the resultant microaggregates is tunable by changing the mixing ratio of good- and nonsolvents. The arrangement of the aggregates is arbitrarily controlled by the hydrophobic/hydrophilic micropatterned substrate over a whole substrate surface as large as a square centimeter scale. The turn-on/off of PL of the microdisks is switchable by doping photochromic molecules. Furthermore, the fluorescence color of the π-conjugated polymer microarray can be switched upon photoisomerization. The self-assembled fluorescent microdisk arrays with several micrometer periodic patterns will be applicable to future optical memories and displays, as well as tools for chemical and biological sensing.
5. Patents
Japanese patent applications 2019-021022 and 2018-016723, and Japanese patent B6420092.
Supplementary Materials
The following is available online at https://www.mdpi.com/2073-4360/13/2/269/s1, Figure S1: fabrication of patterned substrates, Figure S2: microscopic images, Figure S3: electronic absorption and PL spectra, Figure S4: fluorescent microscopy images, Figure S5: cyclic voltammograms, Figure S6: electronic absorption spectra.
Author Contributions
H.Y. and Y.Y. designed the research. H.Y., T.M., Y.K., Y.A. and L.T. performed the experiments. J.K., T.K., M.M. and M.I. performed the organic synthesis. H.Y. and Y.Y. prepared the manuscript with feedback from the other authors. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by JST CREST (JPMJCR20T4) and ACT-X (JPMJAX201J) from Japan Science and Technology Agency (JST), Grant-in-Aid for Scientific Research (A) (JP16H02081), Innovative Areas “π-System Figuration” (JP17H05141, JP17H05142), Young Scientists (JP19K15334) from Japan Society for the Promotion of Science (JSPS), Kato Foundation for Promotion of Science, The Kao Foundation for Arts and Sciences, and Ogasawara Foundation.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
The authors acknowledge Xuying Liu and Takeo Minari at National Institute for Materials Science (NIMS) for the use of photoexposure equipment, Masayuki Takeuchi and Kazunori Sugiyasu at NIMS for the synthesis of P5.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Coskun, A.F.; Topkaya, S.N.; Yetisen, A.K.; Cetin, A.E. Portable multiplex optical assays. Adv. Opt. Mater. 2019, 7, 1801109. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, S.; Zentgraf, T. Metasurface holography: From fundamentals to applications. Nanophotonics 2018, 7, 1169–1190. [Google Scholar] [CrossRef]
- Zhao, J.; Yan, Y.; Gao, Z.; Du, Y.; Dong, H.; Yao, J.; Zhao, Y.S. Full-color laser displays based on organic printed microlaser arrays. Nat. Commun. 2019, 10, 870. [Google Scholar] [CrossRef]
- Faramarzi, V.; Niess, F.; Moulin, E.; Maaloum, M.; Dayen, J.F.; Beaufrand, J.B.; Zanettini, S.; Doudin, B.; Giuseppone, N. Light-triggered self-construction of supramolecular organic nanowires as metallic interconnects. Nat. Chem. 2012, 4, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Noorduin, W.L.; Grinthal, A.; Mahadevan, L.; Aizenberg, J. Rationally designed complex, hierarchical microarchitectures. Science 2013, 340, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Hosono, N.; Kajitani, T.; Fukushima, T.; Ito, K.; Sasaki, S.; Takata, M.; Aida, T. Large-area three-dimensional molecular ordering of a polymer brush by one-step processing. Science 2010, 330, 808–811. [Google Scholar] [CrossRef] [PubMed]
- Green, J.E.; Choi, J.W.; Boukai, A.; Bunimovich, Y.; Johnston-Halperin, E.; DeIonno, E.; Luo, Y.; Sheriff, B.A.; Xu, K.; Shin, Y.S.; et al. A 160-kilobit molecular electronic memory patterned at 10(11) bits per square centimetre. Nature 2007, 445, 414–417. [Google Scholar] [CrossRef]
- Gu, L.; Poddar, S.; Lin, Y.; Long, Z.; Zhang, D.; Zhang, Q.; Shu, L.; Qiu, X.; Kam, M.; Javey, A.; et al. A biomimetic eye with a hemispherical perovskite nanowire array retina. Nature 2020, 581, 278–282. [Google Scholar] [CrossRef]
- Saito, H.; Kuwabara, J.; Kanbara, T. Facile synthesis of fluorene-based π-conjugated polymers via sequential bromination/direct arylation polycondensation. J. Polym. Sci. Part A Polym. Chem. 2015, 53, 2198–2201. [Google Scholar] [CrossRef]
- Fujinami, Y.; Kuwabara, J.; Lu, W.; Hayashi, H.; Kanbara, T. Synthesis of Thiophene- and Bithiophene-Based alternating copolymers via Pd-catalyzed direct C–H arylation. ACS Macro Lett. 2012, 1, 67–70. [Google Scholar] [CrossRef]
- Kuwabara, J.; Nohara, Y.; Choi, S.J.; Fujinami, Y.; Lu, W.; Yoshimura, K.; Oguma, J.; Suenobu, K.; Kanbara, T. Direct arylation polycondensation for the synthesis of bithiophene-based alternating copolymers. Polym. Chem. 2013, 4, 947–953. [Google Scholar] [CrossRef]
- Pan, C.; Sugiyasu, K.; Wakayama, Y.; Sato, A.; Takeuchi, M. Thermoplastic fluorescent conjugated polymers: Benefits of preventing π-π stacking. Angew. Chem. Int. Ed. 2013, 52, 10775–10779. [Google Scholar] [CrossRef]
- Uno, K.; Niikura, H.; Morimoto, M.; Ishibashi, Y.; Miyasaka, H.; Irie, M. In situ preparation of highly fluorescent dyes upon photoirradiation. J. Am. Chem. Soc. 2011, 133, 13558–13564. [Google Scholar] [CrossRef]
- Takagi, Y.; Kunishi, T.; Katayama, T.; Ishibashi, Y.; Miyasaka, H.; Morimoto, M.; Irie, M. Photoswitchable fluorescent diarylethene derivatives with short alkyl chain substituents. Photochem. Photobiol. Sci. 2012, 11, 1661–1665. [Google Scholar] [CrossRef]
- Irie, M.; Fukaminato, T.; Matsuda, K.; Kobatake, S. Photochromism of diarylethene molecules and crystals: Memories, switches, and actuators. Chem. Rev. 2014, 114, 12174–12277. [Google Scholar] [CrossRef]
- Kashihara, R.; Morimoto, M.; Ito, S.; Miyasaka, H.; Irie, M. Fluorescence photoswitching of a diarylethene by irradiation with single-wavelength visible light. J. Am. Chem. Soc. 2017, 139, 16498–16501. [Google Scholar] [CrossRef]
- Irie, M.; Morimoto, M. Photoswitchable turn-on mode fluorescent diarylethenes: Strategies for controlling the switching response. Bull. Chem. Soc. Jpn. 2018, 91, 237–250. [Google Scholar] [CrossRef]
- Liu, X.; Kanehara, M.; Liu, C.; Sakamoto, K.; Yasuda, T.; Takeya, J.; Minari, T. Spontaneous patterning of high-resolution electronics via parallel vacuum ultraviolet. Adv. Mater. 2016, 28, 6568–6573. [Google Scholar] [CrossRef]
- Adachi, T.; Tong, L.; Kuwabara, J.; Kanbara, T.; Saeki, A.; Seki, S.; Yamamoto, Y. Spherical assemblies from π-conjugated alternating copolymers: Toward optoelectronic colloidal crystals. J. Am. Chem. Soc. 2013, 135, 870–876. [Google Scholar] [CrossRef]
- Tong, L.; Kushida, S.; Kuwabara, J.; Kanbara, T.; Ishii, N.; Saeki, A.; Seki, S.; Furumi, S.; Yamamoto, Y. Tetramethylbithiophene in π-conjugated alternating copolymers as an effective structural component for the formation of spherical assemblies. Polym. Chem. 2014, 5, 3583–3587. [Google Scholar] [CrossRef]
- Kushida, S.; Braam, D.; Pan, C.; Dao, T.D.; Tabata, K.; Sugiyasu, K.; Takeuchi, M.; Ishii, S.; Nagao, T.; Lorke, A.; et al. Whispering gallery resonance from self-assembled microspheres of highly fluorescent isolated conjugated polymers. Macromolecules 2015, 48, 3928–3933. [Google Scholar] [CrossRef]
- Kushida, S.; Braam, D.; Dao, T.D.; Saito, H.; Shibasaki, K.; Ishii, S.; Nagao, T.; Saeki, A.; Kuwabara, J.; Kanbara, T.; et al. Conjugated polymer blend microspheres for efficient, long-range light energy transfer. ACS Nano 2016, 10, 5543–5549. [Google Scholar] [CrossRef] [PubMed]
- Okada, D.; Lin, Z.-H.; Huang, J.-S.; Oki, O.; Morimoto, M.; Liu, X.; Minari, T.; Ishii, S.; Nagao, T.; Irie, M.; et al. Optical microresonator arrays of fluorescence-switchable diarylethenes with unreplicable spectral fingerprints. Mater. Horiz. 2020, 7, 1801–1808. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).