Effect of In-Mold Annealing on the Properties of Asymmetric Poly(l-lactide)/Poly(d-lactide) Blends Incorporated with Nanohydroxyapatite
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Samples Preparation
2.3. Differential Scanning Calorimetry (DSC)
2.4. Mechanical Measurement
2.5. Impact Resistance
2.6. Thermo-Mechanical Analysis
2.7. Morphological Characteristic
3. Results and Discussion
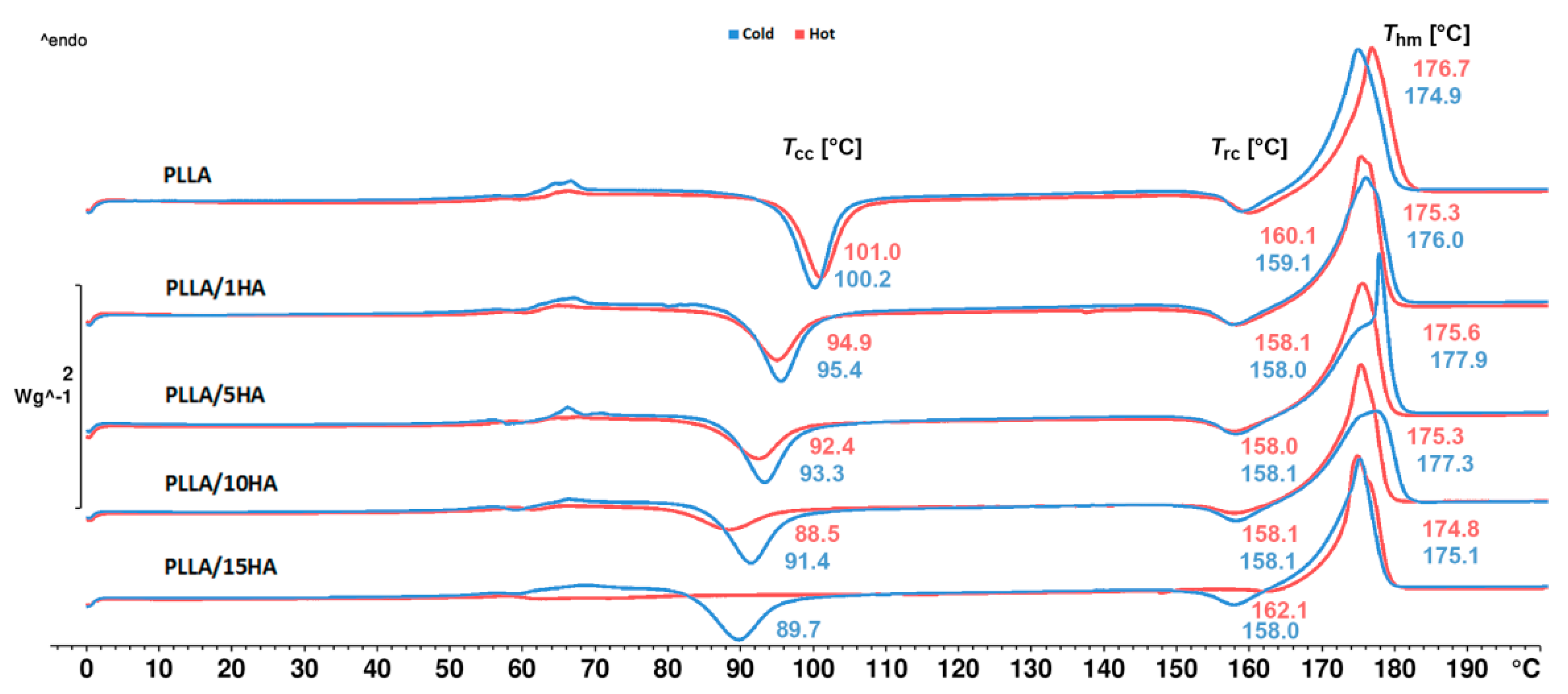
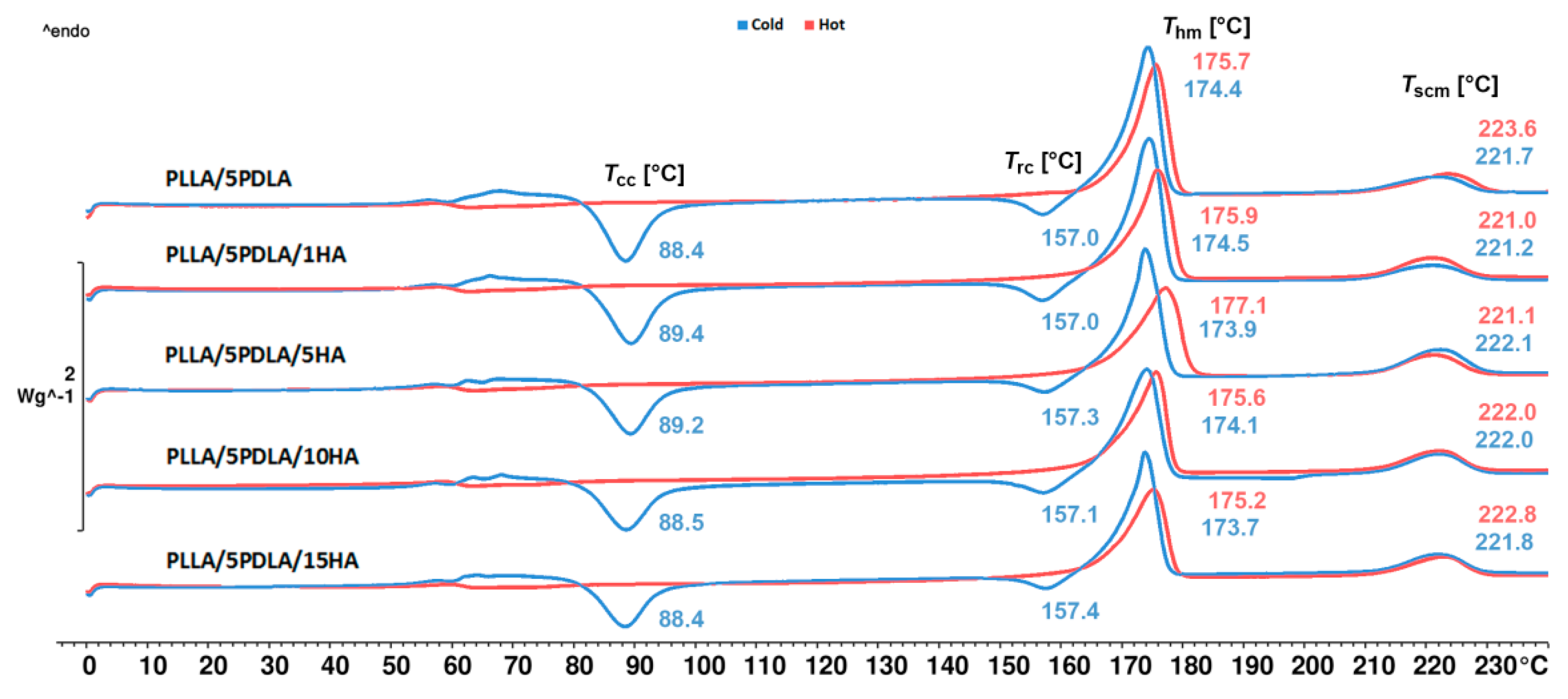
3.1. Non-Isothermal Crystallization (DSC Measurements)
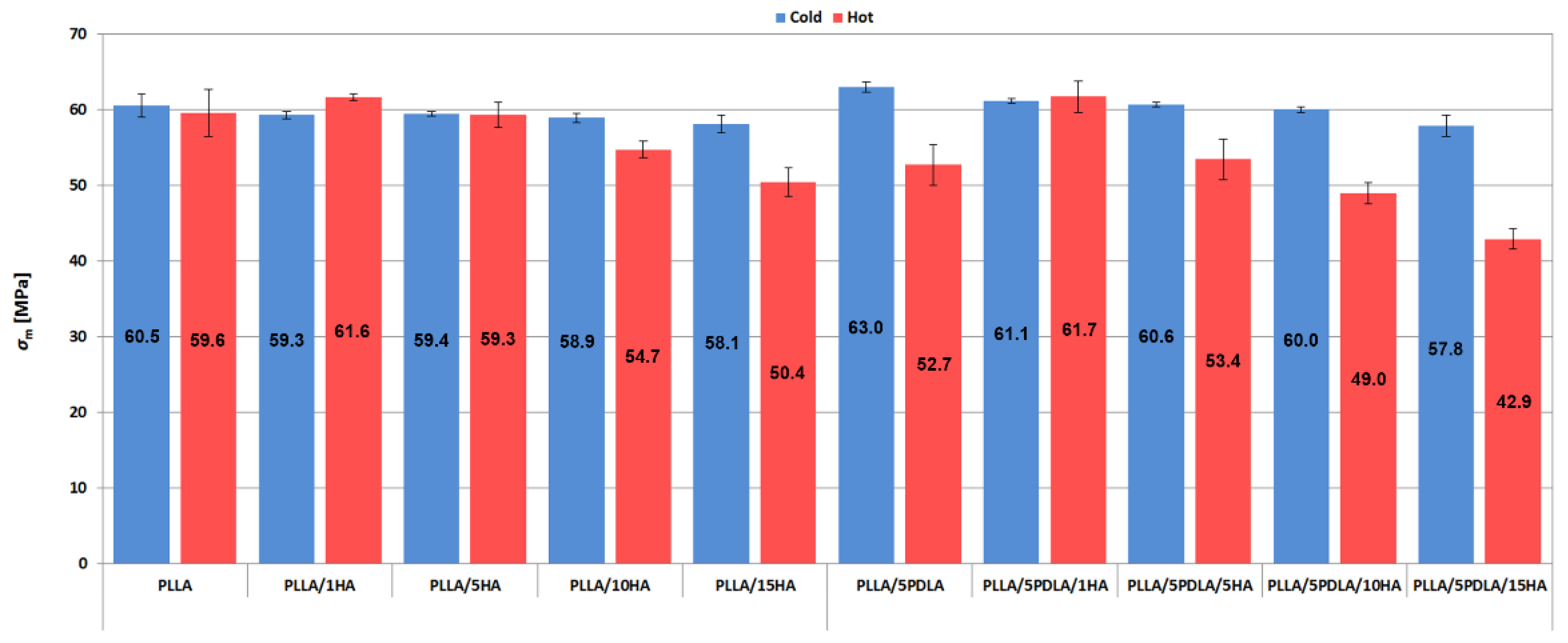
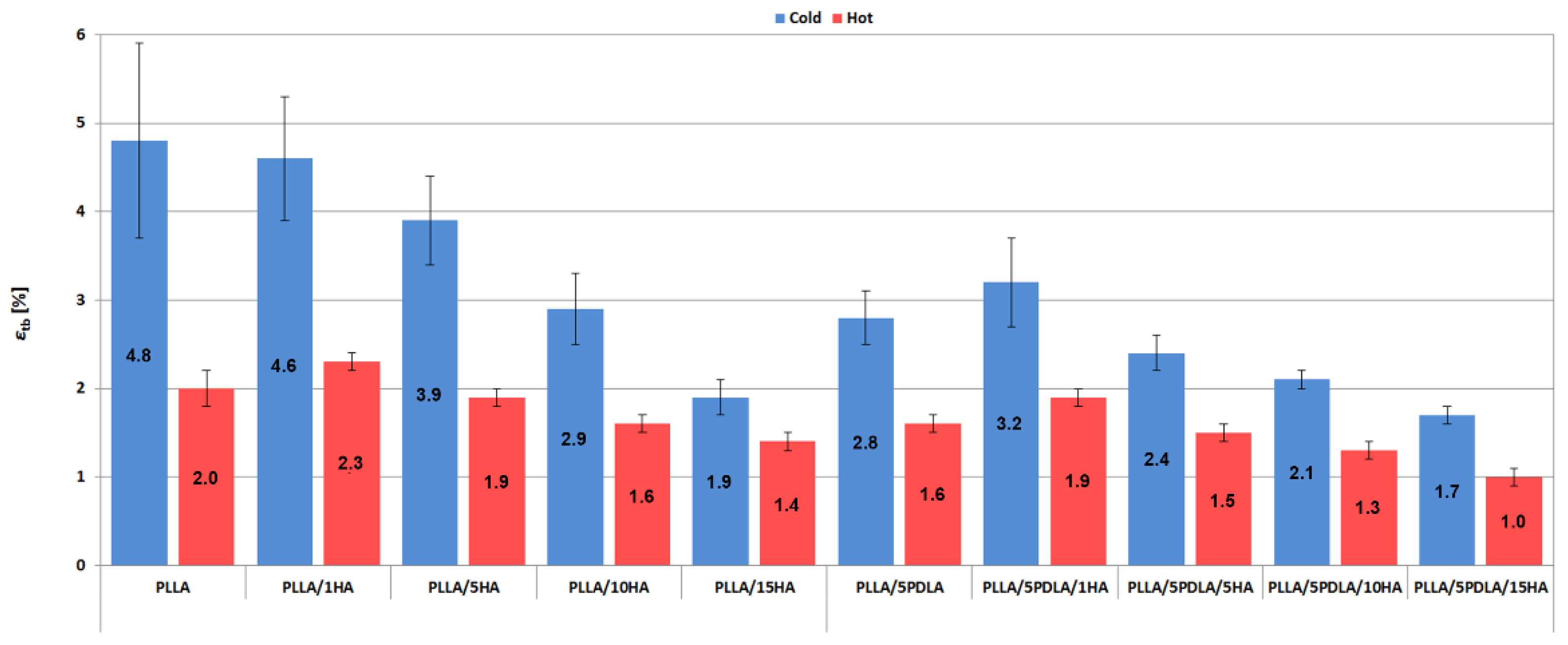
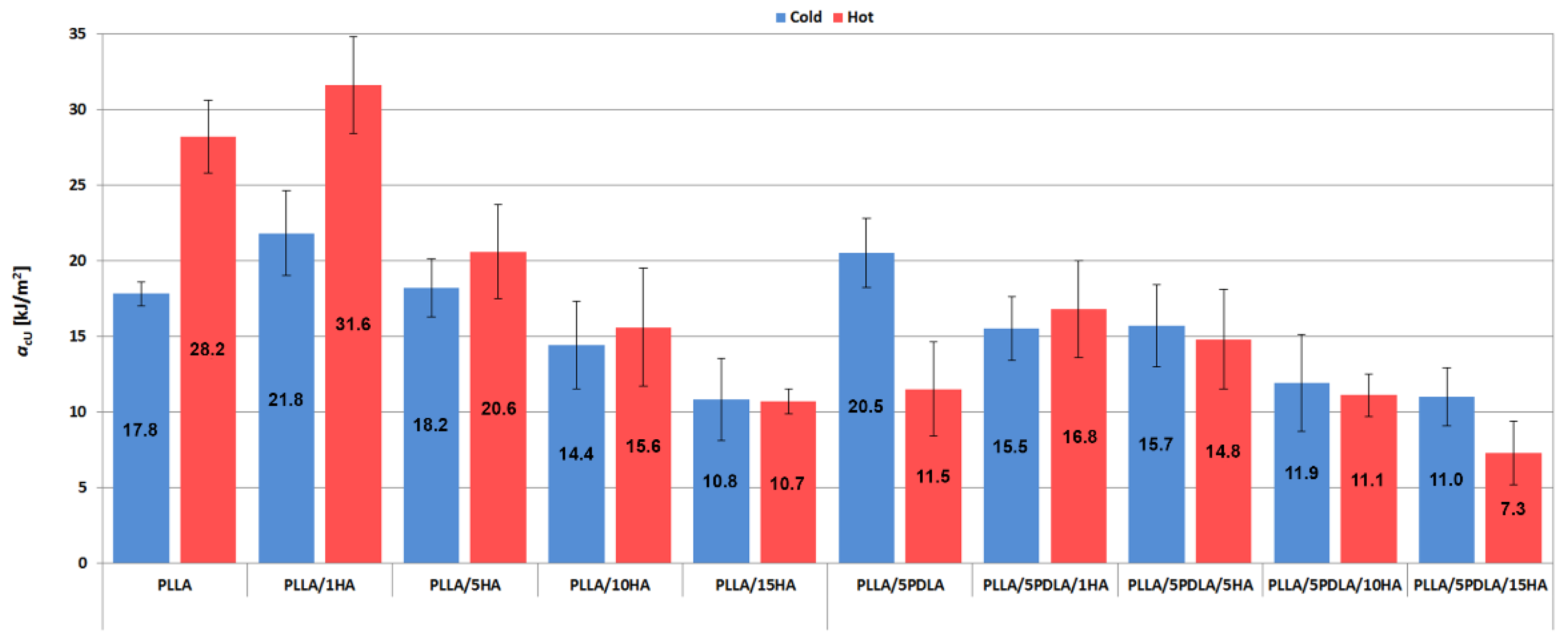
3.2. Mechanical Properties (Static Tensile and Charphy Impact Tests)
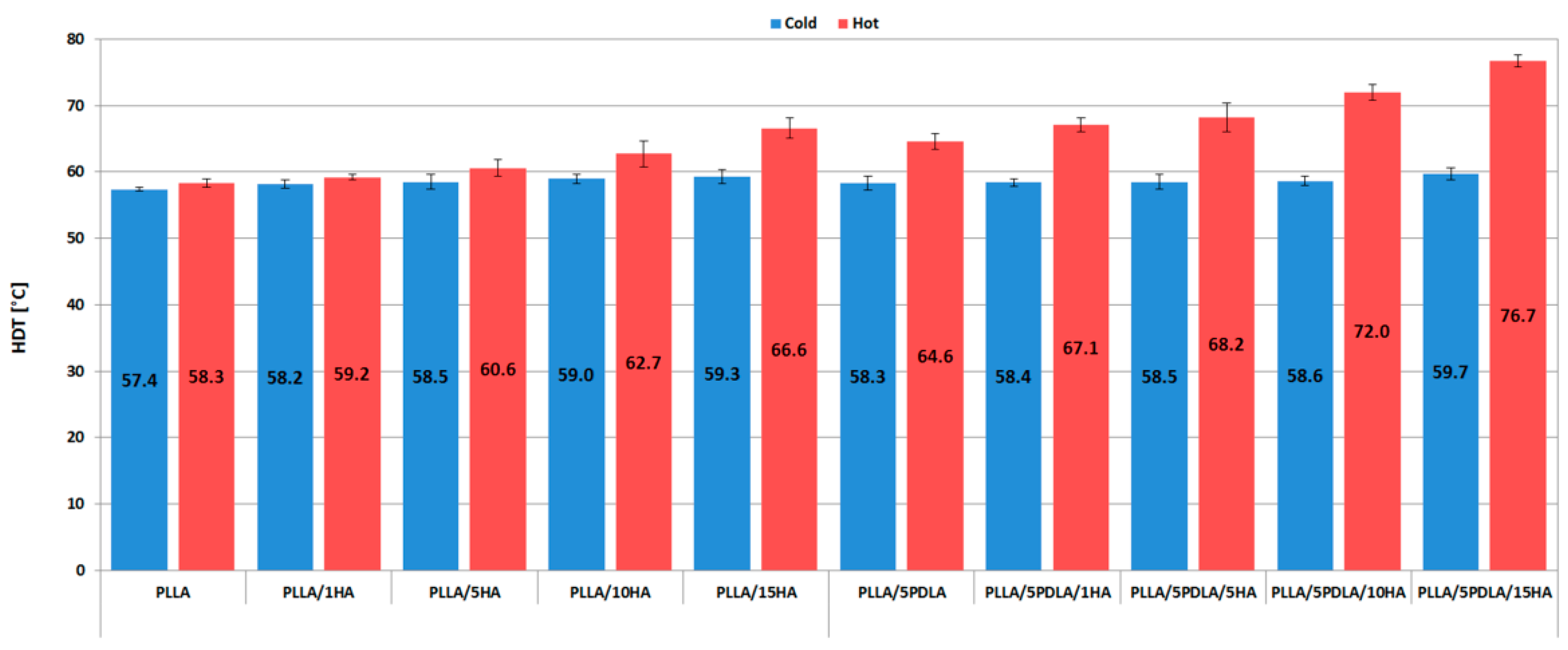
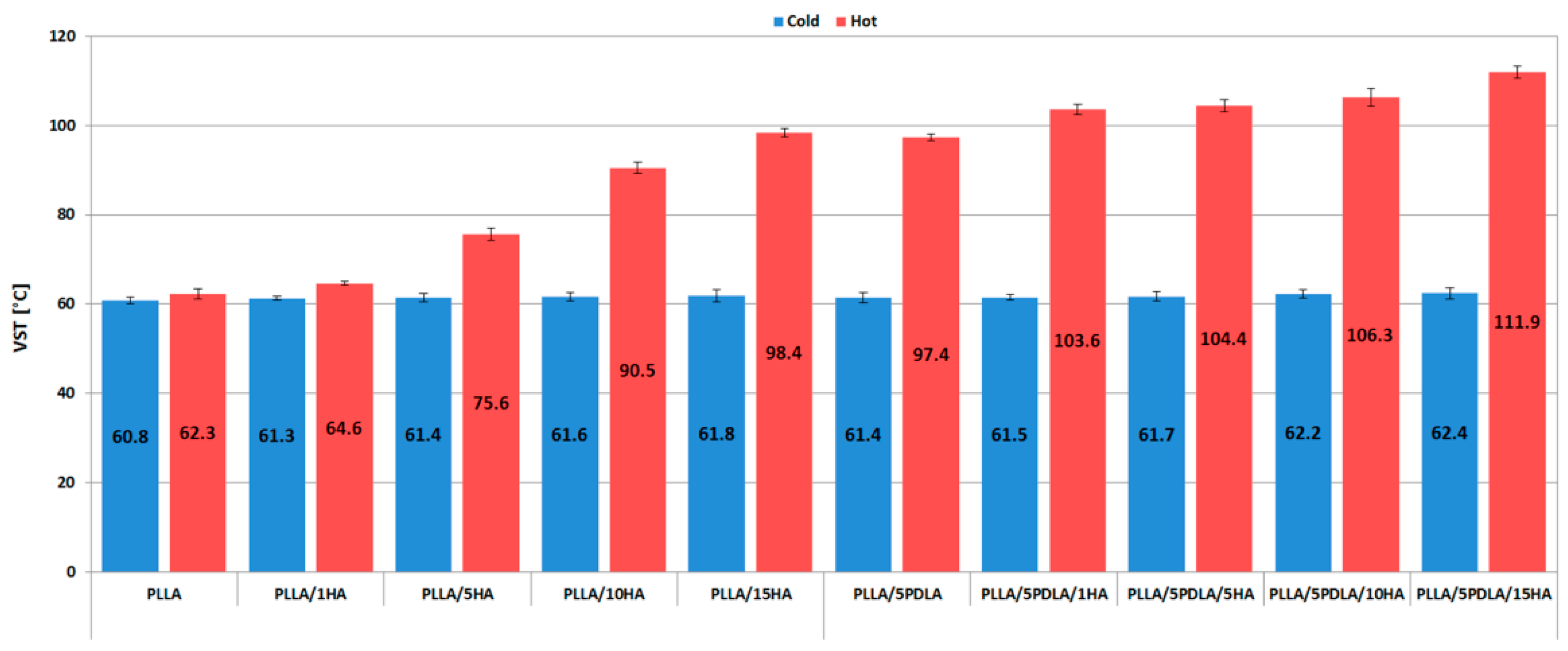
3.3. Thermo-Mechanical Properties (HDT and VST Measurements)
3.4. Fracture Surface Morphology (SEM Analysis)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nagalakshmaiah, M.; Afrin, S.; Malladi, R.P.; Elkoun, S.; Robert, M.; Ansari, M.A.; Svedberg, A.; Karim, Z. Biocomposites: Present trends and challenges for the future. In Green Composites for Automotive Applications; Woodhead Publishing: Sawston, UK, 2019; pp. 197–215. [Google Scholar]
- Ganapini, W. Bioplastics: A Case Study of Bioeconomy in Italy; Edizioni Ambiente: Milan, Italy, 2014; ISBN 88-6627-099-7. [Google Scholar]
- Watkins, E.; Schweitzer, J.-P. Moving towards a circular economy for plastics in the EU by 2030. In Think 2030. Brussels: Institute for European Environmental Policy (IEEP); IEEP: Brussels, Belgium, 2018. [Google Scholar]
- Masutani, K.; Kimura, Y. Present situation and future perspectives of poly (lactic acid). In Synthesis, Structure and Properties of Poly(Lactic Acid); Springer: Cham, Switzerland, 2017; pp. 1–25. [Google Scholar]
- Fiorentino, G.; Ripa, M.; Ulgiati, S. Chemicals from Biomass: Technological versus Environmental Feasibility. A Review. Biofuel. Bioprod. Biorefin. 2017, 11, 195–214. [Google Scholar] [CrossRef]
- Groot, W.; Van Krieken, J.; Sliekersl, O.; De Vos, S. Production and Purification of Lactic Acid and Lactide. In POLY (LACTIC ACID): Synthesis, Structures, Properties, Processing, and Applications; Wiley: Hoboken, NJ, USA, 2010; Volume 1, pp. 1–18. [Google Scholar]
- Lim, L.-T.; Auras, R.; Rubino, M. Processing Technologies for Poly(Lactic Acid). Prog. Polym. Sci. 2008, 33, 820–852. [Google Scholar] [CrossRef]
- Reddy, M.M.; Vivekanandhan, S.; Misra, M.; Bhatia, S.K.; Mohanty, A.K. Biobased Plastics and Bionanocomposites: Current Status and Future Opportunities. Prog. Polym. Sci. 2013, 38, 1653–1689. [Google Scholar] [CrossRef]
- Tsuji, H. Poly(Lactic Acid) Stereocomplexes: A Decade of Progress. Adv. Drug Deliv. Rev. 2016, 107, 97–135. [Google Scholar] [CrossRef] [PubMed]
- Na, B.; Zhu, J.; Lv, R.; Ju, Y.; Tian, R.; Chen, B. Stereocomplex Formation in Enantiomeric Polylactides by Melting Recrystallization of Homocrystals: Crystallization Kinetics and Crystal Morphology. Macromolecules 2014, 47, 347–352. [Google Scholar] [CrossRef]
- Tan, B.H.; Muiruri, J.K.; Li, Z.; He, C. Recent Progress in Using Stereocomplexation for Enhancement of Thermal and Mechanical Property of Polylactide. ACS Sustain. Chem. Eng. 2016, 4, 5370–5391. [Google Scholar] [CrossRef]
- Ikada, Y.; Jamshidi, K.; Tsuji, H.; Hyon, S.H. Stereocomplex Formation between Enantiomeric Poly (Lactides). Macromolecules 1987, 20, 904–906. [Google Scholar] [CrossRef]
- Tsuji, H.; Ikada, Y. Stereocomplex Formation between Enantiomeric Poly(Lactic Acid) s. XI. Mechanical Properties and Morphology of Solution-Cast Films. Polymer 1999, 40, 6699–6708. [Google Scholar] [CrossRef]
- Tsuji, H. Poly (Lactide) Stereocomplexes: Formation, Structure, Properties, Degradation, and Applications. Macromol. Biosci. 2005, 5, 569–597. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.-Y.; Wei, X.-F.; Bao, R.-Y.; Dong, Q.-X.; Liu, Z.-Y.; Yang, W.; Xie, B.-H.; Yang, M.-B. Enantiomeric Poly(d-lactide) with a Higher Melting Point Served as a Significant Nucleating Agent for Poly (l-Lactide). CrystEngComm 2015, 17, 4334–4342. [Google Scholar] [CrossRef]
- Luo, F.; Fortenberry, A.; Ren, J.; Qiang, Z. Recent Progress in Enhancing Poly(Lactic Acid) Stereocomplex Formation for Material Property Improvement. Front. Chem. 2020, 8, 688. [Google Scholar] [CrossRef]
- Yamane, H.; Sasai, K.; Takano, M.; Takahashi, M. Poly (d-Lactic Acid) as a Rheological Modifier of Poly (l-Lactic Acid): Shear and Biaxial Extensional Flow Behavior. J. Rheol. 2004, 48, 599–609. [Google Scholar] [CrossRef]
- Inkinen, S.; Stolt, M.; Södergård, A. Effect of Blending Ratio and Oligomer Structure on the Thermal Transitions of Stereocomplexes Consisting of Ad-lactic Acid Oligomer and Poly (l-lactide). Polym. Adv. Technol. 2011, 22, 1658–1664. [Google Scholar] [CrossRef]
- Shi, X.; Jing, Z.; Zhang, G. Influence of PLA Stereocomplex Crystals and Thermal Treatment Temperature on the Rheology and Crystallization Behavior of Asymmetric Poly(l-lactide)/Poly(d-lactide) Blends. J. Polym. Res. 2018, 25, 71. [Google Scholar] [CrossRef]
- Wei, X.-F.; Bao, R.-Y.; Cao, Z.-Q.; Yang, W.; Xie, B.-H.; Yang, M.-B. Stereocomplex Crystallite Network in Asymmetric PLLA/PDLA Blends: Formation, Structure, and Confining Effect on the Crystallization Rate of Homocrystallites. Macromolecules 2014, 47, 1439–1448. [Google Scholar] [CrossRef]
- Wang, J.; Lv, R.; Wang, B.; Na, B.; Liu, H. Direct Observation of a Stereocomplex Crystallite Network in the Asymmetric Polylactide Enantiomeric Blends. Polymer 2018, 143, 52–57. [Google Scholar] [CrossRef]
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly(Lactic Acid) Modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Yang, S.; Leong, K.-F.; Du, Z.; Chua, C.-K. The Design of Scaffolds for Use in Tissue Engineering. Part I. Traditional Factors. Tissue Eng. 2001, 7, 679–689. [Google Scholar] [CrossRef] [Green Version]
- Nazhat, S.; Kellomäki, M.; Törmälä, P.; Tanner, K.; Bonfield, W. Dynamic Mechanical Characterization of Biodegradable Composites of Hydroxyapatite and Polylactides. J. Biomed. Mater. Res. 2001, 58, 335–343. [Google Scholar] [CrossRef]
- Teo, W.; Liao, S.; Chan, C.; Ramakrishna, S. Fabrication and Characterization of Hierarchically Organized Nanoparticle-Reinforced Nanofibrous Composite Scaffolds. Acta Biomater. 2011, 7, 193–202. [Google Scholar] [CrossRef]
- Hanemann, T.; Szabó, D.V. Polymer-Nanoparticle Composites: From Synthesis to Modern Applications. Materials 2010, 3, 3468–3517. [Google Scholar] [CrossRef]
- Hasnain, M.S.; Ahmad, S.A.; Chaudhary, N.; Hoda, M.N.; Nayak, A.K. 1-Biodegradable polymer matrix nanocomposites for bone tissue engineering. In Applications of Nanocomposite Materials in Orthopedics; Inamuddin, Asiri, A.M., Mohammad, A., Eds.; Woodhead Publishing Series in Biomaterials; Woodhead Publishing: Sawston, UK, 2019; pp. 1–37. ISBN 978-0-12-813740-6. [Google Scholar]
- Bharadwaz, A.; Jayasuriya, A.C. Recent Trends in the Application of Widely Used Natural and Synthetic Polymer Nanocomposites in Bone Tissue Regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 110, 110698. [Google Scholar] [CrossRef]
- Seunarine, K.; Gadegaard, N.; Tormen, M.; Meredith, D.; Riehle, M.; Wilkinson, C. 3D Polymer Scaffolds for Tissue Engineering. Nanomedicine 2006, 1, 281–296. [Google Scholar] [CrossRef]
- Ishii, S.; Tamura, J.; Furukawa, T.; Nakamura, T.; Matsusue, Y.; Shikinami, Y.; Okuno, M. Long-Term Study of High-Strength Hydroxyapatite/Poly(l-lactide) Composite Rods for the Internal Fixation of Bone Fractures: A 2–4-Year Follow-up Study in Rabbits. J. Biomed. Mater. Res. B App. Biomater. 2003, 66B, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.-L.; Fang, H.-W.; Tseng, T.; Lee, W.-H. Effects of Hydroxyapatite Dosage on Mechanical and Biological Behaviors of Polylactic Acid Composite Materials. ASC Mater. Lett. 2007, 61, 3009–3013. [Google Scholar] [CrossRef]
- Salerno, A.; Fernández-Gutiérrez, M.; del Barrio, J.S.R.; Pascual, C.D. Macroporous and Nanometre Scale Fibrous PLA and PLA–HA Composite Scaffolds Fabricated by a Bio Safe Strategy. RSC Adv. 2014, 4, 61491–61502. [Google Scholar] [CrossRef] [Green Version]
- Akindoyo, J.O.; Beg, M.D.; Ghazali, S.; Alam, A.; Heim, H.P.; Feldmann, M. Synergized Poly(Lactic Acid)–Hydroxyapatite Composites: Biocompatibility Study. J. Appl. Polym. Sci. 2019, 136, 47400. [Google Scholar] [CrossRef]
- Šupová, M. Problem of Hydroxyapatite Dispersion in Polymer Matrices: A Review. J. Mater. Sci. Mater. Med. 2009, 20, 1201–1213. [Google Scholar] [CrossRef] [PubMed]
- Akindoyo, J.O.; Beg, M.D.; Ghazali, S.; Heim, H.P.; Feldmann, M. Effects of Surface Modification on Dispersion, Mechanical, Thermal and Dynamic Mechanical Properties of Injection Molded PLA-Hydroxyapatite Composites. Compos. Part A Appl. Sci. Manuf. 2017, 103, 96–105. [Google Scholar] [CrossRef]
- Akindoyo, J.O.; Beg, M.D.; Ghazali, S.; Heim, H.P.; Feldmann, M. Impact Modified PLA-Hydroxyapatite Composites–Thermo-Mechanical Properties. Compos. Part A Appl. Sci. Manuf. 2018, 107, 326–333. [Google Scholar] [CrossRef]
- Hong, Z.; Qiu, X.; Sun, J.; Deng, M.; Chen, X.; Jing, X. Grafting Polymerization of l-Lactide on the Surface of Hydroxyapatite Nano-Crystals. Polymer 2004, 45, 6699–6706. [Google Scholar] [CrossRef]
- Hong, Z.; Zhang, P.; He, C.; Qiu, X.; Liu, A.; Chen, L.; Chen, X.; Jing, X. Nano-Composite of Poly(l-lactide) and Surface Grafted Hydroxyapatite: Mechanical Properties and Biocompatibility. Biomaterials 2005, 26, 6296–6304. [Google Scholar] [CrossRef]
- Ko, H.-S.; Lee, S.; Lee, D.; Jho, J.Y. Mechanical Properties and Bioactivity of Poly(Lactic Acid) Composites Containing Poly(Glycolic Acid) Fiber and Hydroxyapatite Particles. Nanomaterials 2021, 11, 249. [Google Scholar] [CrossRef] [PubMed]
- Shuai, C.; Yu, L.; Yang, W.; Peng, S.; Zhong, Y.; Feng, P. Phosphonic Acid Coupling Agent Modification of HAP Nanoparticles: Interfacial Effects in PLLA/HAP Bone Scaffold. Polymers 2020, 12, 199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferri, J.; Jordá, J.; Montanes, N.; Fenollar, O.; Balart, R. Manufacturing and Characterization of Poly(Lactic Acid) Composites with Hydroxyapatite. J. Thermoplast. Compos. Mater. 2018, 31, 865–881. [Google Scholar] [CrossRef]
- Henton, D.E.; Gruber, P.; Lunt, J.; Randall, J. Polylactic Acid Technology. In Natural Fibers, Biopolymers, and Biocomposites, 1st ed.; CRC Press: Boca Raton, FL, USA, 2005; Volume 16, pp. 527–577. [Google Scholar]
- Sarasua, J.-R.; Prud’Homme, R.E.; Wisniewski, M.; Le Borgne, A.; Spassky, N. Crystallization and Melting Behavior of Polylactides. Macromolecules 1998, 31, 3895–3905. [Google Scholar] [CrossRef]
- Sarasua, J.; Arraiza, A.L.; Balerdi, P.; Maiza, I. Crystallization and Thermal Behaviour of Optically Pure Polylactides and Their Blends. J. Mater. Sci. 2005, 40, 1855–1862. [Google Scholar] [CrossRef]
- Zhou, S.; Zheng, X.; Yu, X.; Wang, J.; Weng, J.; Li, X.; Feng, B.; Yin, M. Hydrogen Bonding Interaction of Poly(d,l-lactide)/Hydroxyapatite Nanocomposites. Chem. Mater. 2007, 19, 247–253. [Google Scholar] [CrossRef]
- Lv, T.; Li, J.; Huang, S.; Wen, H.; Li, H.; Chen, J.; Jiang, S. Synergistic Effects of Chain Dynamics and Enantiomeric Interaction on the Crystallization in PDLA/PLLA Mixtures. Polymer 2021, 222, 123648. [Google Scholar] [CrossRef]
- Vadori, R.; Mohanty, A.K.; Misra, M. The Effect of Mold Temperature on the Performance of Injection Molded Poly(Lactic Acid)-Based Bioplastic: The Effect of Mold Temperature on the Performance of Injection Molded Poly(Lactic Acid)-Based Bioplastic. Macromol. Mater. Eng. 2013, 298, 981–990. [Google Scholar] [CrossRef]
- Kawamoto, N.; Sakai, A.; Horikoshi, T.; Urushihara, T.; Tobita, E. Physical and Mechanical Properties of Poly (l-lactic Acid) Nucleated by Dibenzoylhydrazide Compound. J. Appl. Polym. Sci. 2007, 103, 244–250. [Google Scholar] [CrossRef]
- Tang, Z.; Zhang, C.; Liu, X.; Zhu, J. The Crystallization Behavior and Mechanical Properties of Polylactic Acid in the Presence of a Crystal Nucleating Agent. J. Appl. Polym. Sci. 2012, 125, 1108–1115. [Google Scholar] [CrossRef]
- Kramer, E.J.; Berger, L.L. Fundamental processes of craze growth and fracture. In Crazing in Polymers Vol. 2; Springer: Cham, Switzerland, 1990; pp. 1–68. [Google Scholar]
- Renouf-Glauser, A.C.; Rose, J.; Farrar, D.F.; Cameron, R.E. The Effect of Crystallinity on the Deformation Mechanism and Bulk Mechanical Properties of PLLA. Biomaterials 2005, 26, 5771–5782. [Google Scholar] [CrossRef] [PubMed]
- Park, S.D.; Todo, M.; Arakawa, K.; Koganemaru, M. Effect of Crystallinity and Loading-Rate on Mode I Fracture Behavior of Poly(Lactic Acid). Polymer 2006, 47, 1357–1363. [Google Scholar] [CrossRef]
- Way, J.; Atkinson, J.; Nutting, J. The Effect of Spherulite Size on the Fracture Morphology of Polypropylene. J. Mater. Sci. 1974, 9, 293–299. [Google Scholar] [CrossRef]
- Andrews, E. Microfibrillar Textures in Polymer Fibers. J. Polym. Sci. Part A-2 Polym. Phys. 1966, 4, 668–672. [Google Scholar] [CrossRef]
- Graham, I.; Marshall, G.; Williams, J. The Fracture Mechanics of Crazes; Springer: Cham, Switzerland, 1973; pp. 261–272. [Google Scholar]







| Sample Code | PLLA (wt.%) | PDLA (wt.%) | HA (wt.%) |
|---|---|---|---|
| PLLA | 100 | - | - |
| PLLA/1HA | 99 | - | 1 |
| PLLA/5HA | 95 | - | 5 |
| PLLA/10HA | 90 | - | 10 |
| PLLA/15HA | 85 | - | 15 |
| PLLA/5PDLA | 95 | 5 | - |
| PLLA/5PDLA/1HA | 94 | 5 | 1 |
| PLLA/5PDLA/5HA | 90 | 5 | 5 |
| PLLA/5PDLA/10HA | 85 | 5 | 10 |
| PLLA/5PDLA/15HA | 80 | 5 | 15 |
| Samples | Tcc (°C) | ΔHcc (J∙g−1) | Trc (°C) | ΔHrc (J∙g−1) | Thm (°C) | ΔHhm (J∙g−1) | Tscm (°C) | ΔHscm (J∙g−1) | χc (%) |
|---|---|---|---|---|---|---|---|---|---|
| PLLA | 100.2 | 33.7 | 159.1 | 5.7 | 174.9 | 52.2 | - | - | 12.0 |
| PLLA/1HA | 95.4 | 29.5 | 158.0 | 7.0 | 176.0 | 49.3 | - | - | 13.7 |
| PLLA/5HA | 93.3 | 25.8 | 158.1 | 6.0 | 177.9 | 47.5 | - | - | 15.6 |
| PLLA/10HA | 91.4 | 24.9 | 158.1 | 5.6 | 177.3 | 46.1 | - | - | 16.4 |
| PLLA/15HA | 89.7 | 22.4 | 158.0 | 4.5 | 175.1 | 43.1 | - | - | 17.9 |
| PLLA/5PDLA | 88.4 | 23.1 | 157.0 | 4.4 | 174.4 | 44.5 | 221.7 | 8.6 | 16.1 |
| PLLA/5PDLA/1HA | 89.4 | 23.8 | 157.0 | 4.6 | 174.5 | 43.9 | 221.2 | 7.6 | 14.8 |
| PLLA/5PDLA/5HA | 89.2 | 20.1 | 157.3 | 3.4 | 173.9 | 38.0 | 222.1 | 11.5 | 14.5 |
| PLLA/5PDLA/10HA | 88.5 | 20.6 | 157.1 | 3.2 | 174.1 | 38.2 | 222.0 | 12.8 | 15.1 |
| PLLA/5PDLA/15HA | 88.4 | 19.1 | 157.4 | 2.5 | 173.7 | 35.9 | 221.8 | 9.5 | 15.8 |
| Samples | Tcc (°C) | ΔHcc (J∙g−1) | Trc (°C) | ΔHrc (J∙g−1) | Thm (°C) | ΔHhm (J∙g−1) | Tscm (°C) | ΔHscm (J∙g−1) | χc (%) |
|---|---|---|---|---|---|---|---|---|---|
| PLLA | 101.0 | 26.5 | 160.1 | 6.0 | 176.7 | 49.7 | - | - | 16.2 |
| PLLA/1HA | 94.9 | 20.4 | 158.1 | 5.7 | 175.3 | 49.1 | - | - | 22.0 |
| PLLA/5HA | 92.4 | 17.1 | 158.0 | 4.7 | 175.6 | 47.4 | - | - | 25.4 |
| PLLA/10HA | 88.5 | 11.0 | 158.1 | 2.9 | 175.3 | 45.2 | - | - | 32.8 |
| PLLA/15HA | - | - | 162.1 | 0.5 | 174.8 | 40.5 | - | - | 44.4 |
| PLLA/5PDLA | - | - | - | - | 175.7 | 42.5 | 223.6 | 9.8 | 40.1 |
| PLLA/5PDLA/1HA | - | - | - | - | 175.9 | 39.9 | 221.0 | 9.6 | 38.1 |
| PLLA/5PDLA/5HA | - | - | - | - | 177.1 | 38.6 | 221.1 | 9.8 | 38.3 |
| PLLA/5PDLA/10HA | - | - | - | - | 175.6 | 37.1 | 222.0 | 9.6 | 38.9 |
| PLLA/5PDLA/15HA | - | - | - | - | 175.2 | 35.5 | 222.8 | 9.3 | 39.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boruvka, M.; Cermak, C.; Behalek, L.; Brdlik, P. Effect of In-Mold Annealing on the Properties of Asymmetric Poly(l-lactide)/Poly(d-lactide) Blends Incorporated with Nanohydroxyapatite. Polymers 2021, 13, 2835. https://doi.org/10.3390/polym13162835
Boruvka M, Cermak C, Behalek L, Brdlik P. Effect of In-Mold Annealing on the Properties of Asymmetric Poly(l-lactide)/Poly(d-lactide) Blends Incorporated with Nanohydroxyapatite. Polymers. 2021; 13(16):2835. https://doi.org/10.3390/polym13162835
Chicago/Turabian StyleBoruvka, Martin, Cenek Cermak, Lubos Behalek, and Pavel Brdlik. 2021. "Effect of In-Mold Annealing on the Properties of Asymmetric Poly(l-lactide)/Poly(d-lactide) Blends Incorporated with Nanohydroxyapatite" Polymers 13, no. 16: 2835. https://doi.org/10.3390/polym13162835
APA StyleBoruvka, M., Cermak, C., Behalek, L., & Brdlik, P. (2021). Effect of In-Mold Annealing on the Properties of Asymmetric Poly(l-lactide)/Poly(d-lactide) Blends Incorporated with Nanohydroxyapatite. Polymers, 13(16), 2835. https://doi.org/10.3390/polym13162835








