Functional Nanocellulose, Alginate and Chitosan Nanocomposites Designed as Active Film Packaging Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. CNF Production
2.1.2. BNC Production
2.1.3. CNC Production
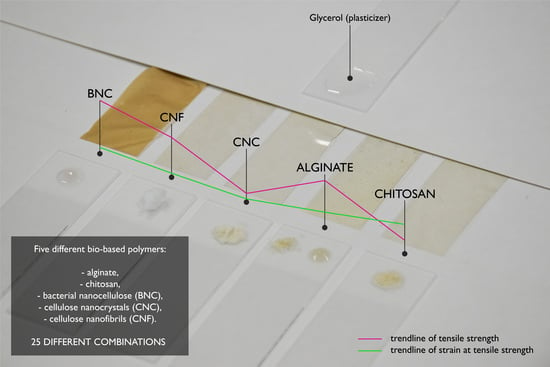
2.2. Films Preparation
2.3. Tensile Properties
2.4. Water Contact Angle
2.5. Film Density
2.6. Scanning Electron Microscope (SEM)
2.7. Water Vapour Transmission Rate (WVTR) and Water Vapor Permeability (WVP) Determination
2.8. Oxygen Permeability (OTR)
2.9. Moisture Absorption
2.10. Statistical Analysis
3. Results and Discussion
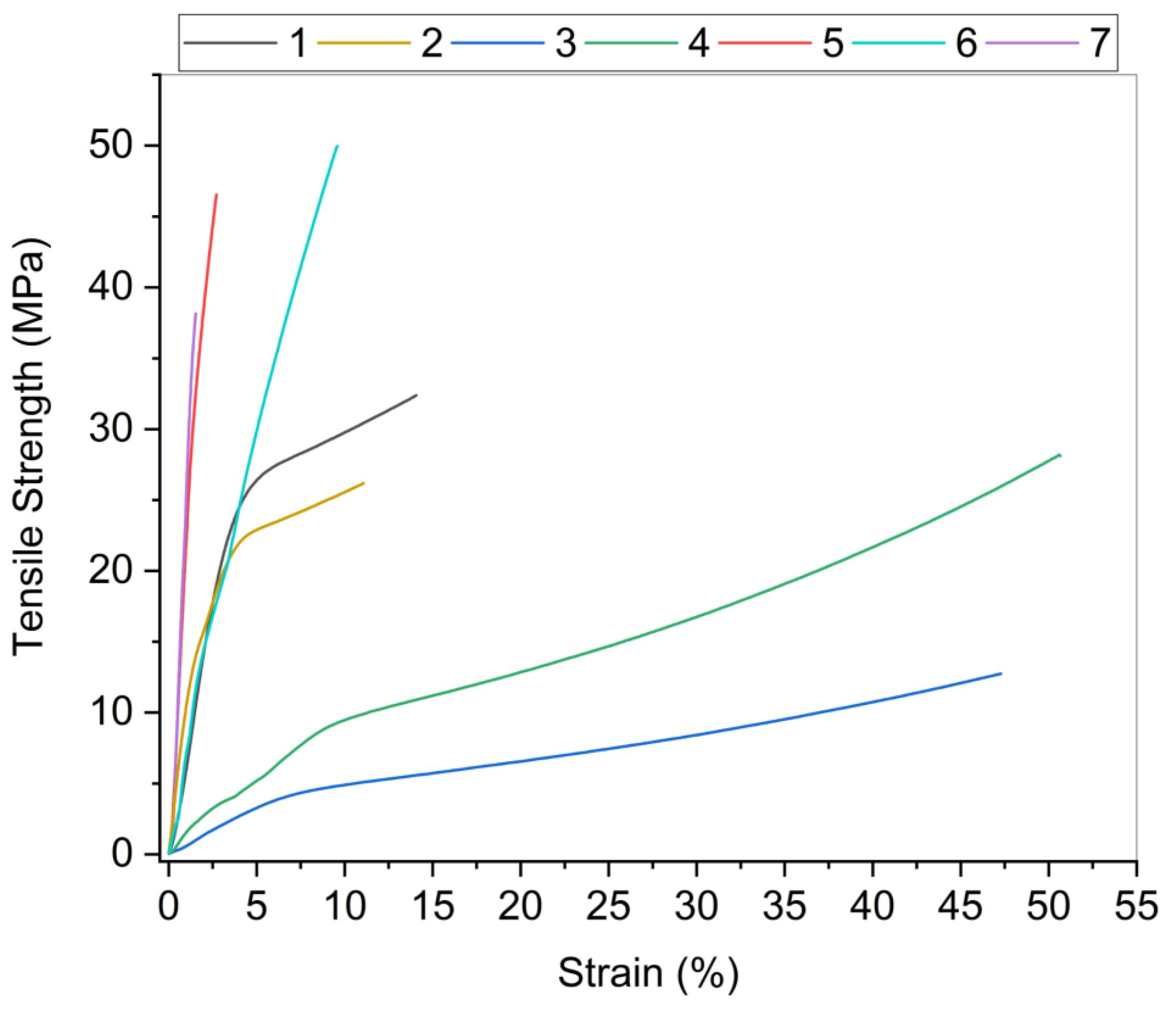
3.1. Tensile Properties
3.2. Water Contact Angle
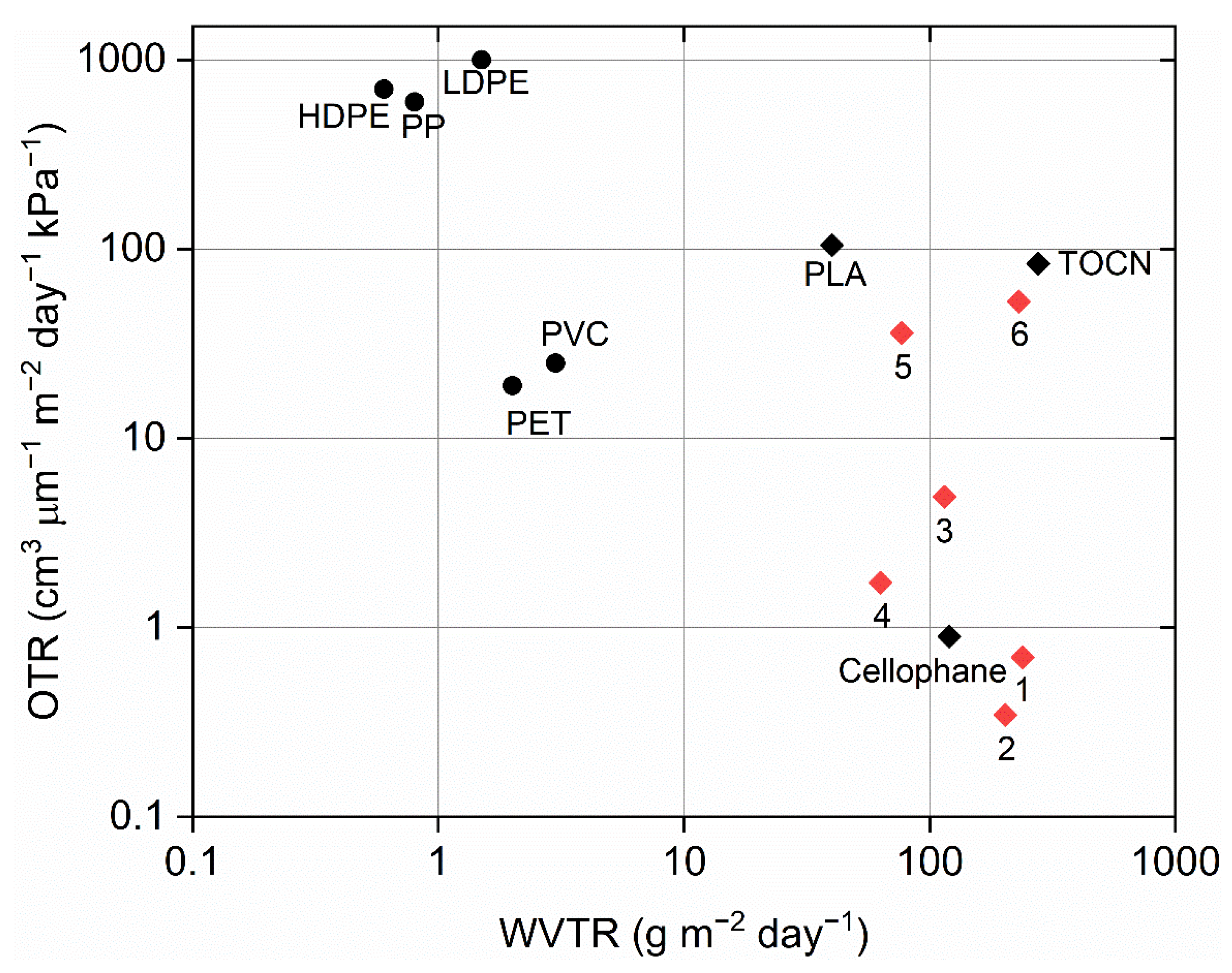
3.3. Barrier Properties of the Films
3.4. Visualization and Morphology of the Films
3.5. Film Density and Moisture Adsorption
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rochman, C.M. Microplastics Research—From Sink to Source. Science 2018, 360, 28–29. [Google Scholar] [CrossRef]
- Kawecki, D.; Nowack, B. Polymer-Specific Modeling of the Environmental Emissions of Seven Commodity Plastics as Macro- and Microplastics. Environ. Sci. Technol. 2019, 53, 9664–9676. [Google Scholar] [CrossRef] [PubMed]
- Kunwar, B.; Cheng, H.; Chandrashekaran, S.R.; Sharma, B.K. Plastics to fuel: A review. Renew. Sustain. Energy Rev. 2016, 54, 421–428. [Google Scholar] [CrossRef]
- Carus, M.; Dammer, L.; Raschka, A.; Skoczinski, P. Renewable Carbon: Key to a Sustainable and Future-oriented Chemical and Plastic Industry: Definition, Strategy, Measures and Potential. Greenh. Gases Sci. Technol. 2020, 10, 488–505. [Google Scholar] [CrossRef]
- SusChem. Sustainable Plastics Strategy. 2020. Available online: http://suschem.org/files/library/Publications/Suschem_Sustainable_Plastics_Brochure-FINAL_2101.pdf (accessed on 29 May 2021).
- Ghosh, K.; Jones, B.H. Roadmap to Biodegradable Plastics—Current State and Research Needs. ACS Sustain. Chem. Eng. 2021, 9, 6170–6187. [Google Scholar] [CrossRef]
- Oberlintner, A.; Bajić, M.; Kalčíková, G.; Likozar, B.; Novak, U. Biodegradability study of active chitosan biopolymer films enriched with Quercus polyphenol extract in different soil types. Environ. Technol. Innov. 2021, 21, 101318. [Google Scholar] [CrossRef]
- Simona, J.; Dani, D.; Petr, S.; Marcela, N.; Jakub, T.; Bohuslava, T. Edible Films from Carrageenan/Orange Essential Oil/Trehalose—Structure, Optical Properties, and Antimicrobial Activity. Polymers 2021, 13, 332. [Google Scholar] [CrossRef]
- Novak, U.; Bajić, M.; Kõrge, K.; Oberlintner, A.; Murn, J.; Lokar, K.; Triler, K.V.; Likozar, B. From waste/residual marine biomass to active biopolymer-based packaging film materials for food industry applications—A review. Phys. Sci. Rev. 2019, 5, 5. [Google Scholar] [CrossRef]
- Khalil, H.A.; Davoudpour, Y.; Islam, N.; Mustapha, A.; Sudesh, K.; Dungani, R.; Jawaid, M. Production and modification of nanofibrillated cellulose using various mechanical processes: A review. Carbohydr. Polym. 2014, 99, 649–665. [Google Scholar] [CrossRef]
- Haghighi, H.; Gullo, M.; La China, S.; Pfeifer, F.; Siesler, H.W.; Licciardello, F.; Pulvirenti, A. Characterization of bio-nanocomposite films based on gelatin/polyvinyl alcohol blend reinforced with bacterial cellulose nanowhiskers for food packaging applications. Food Hydrocoll. 2021, 113, 106454. [Google Scholar] [CrossRef]
- Bai, L.; Huan, S.; Zhu, Y.; Chu, G.; McClements, D.J.; Rojas, O.J. Recent Advances in Food Emulsions and Engineering Foodstuffs Using Plant-Based Nanocelluloses. Annu. Rev. Food Sci. Technol. 2021, 12, 383–406. [Google Scholar] [CrossRef]
- Oberlintner, A.; Likozar, B.; Novak, U. Hydrophobic functionalization reactions of structured cellulose nanomaterials: Mechanisms, kinetics and in silico multi-scale models. Carbohydr. Polym. 2021, 259, 117742. [Google Scholar] [CrossRef]
- Zhao, D.; Yu, S.; Sun, B.; Gao, S.; Guo, S.; Zhao, K. Biomedical Applications of Chitosan and Its Derivative Nanoparticles. Polymers 2018, 10, 462. [Google Scholar] [CrossRef] [Green Version]
- Yan, N.; Chen, X. Sustainability: Don’t waste seafood waste. Nat. News 2015, 524, 155–157. [Google Scholar] [CrossRef] [PubMed]
- Carina, D.; Sharma, S.; Jaiswal, A.K.; Jaiswal, S. Seaweeds polysaccharides in active food packaging: A review of recent progress. Trends Food Sci. Technol. 2021, 110, 559–572. [Google Scholar] [CrossRef]
- Khan, A.; Khan, R.A.; Salmieri, S.; Le Tien, C.; Riedl, B.; Bouchard, J.; Chauve, G.; Tan, V.; Kamal, M.R.; Lacroix, M. Mechanical and barrier properties of nanocrystalline cellulose reinforced chitosan based nanocomposite films. Carbohydr. Polym. 2012, 90, 1601–1608. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, Y.; Cao, J.; Jiang, W. Improving the performance of edible food packaging films by using nanocellulose as an additive. Int. J. Biol. Macromol. 2021, 166, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Lengowski, E.C.; Júnior, E.A.B.; Simon, L.; De Muñiz, G.I.B.; De Andrade, A.S.; Nisgoski, S.; Klock, U. Different degree of fibrillation: Strategy to reduce permeability in nanocellulose-starch films. Cellulose 2020, 27, 10855–10872. [Google Scholar] [CrossRef]
- Fang, Z.; Zhu, H.; Preston, C.; Hu, L. Development, application and commercialization of transparent paper. Transl. Mater. Res. 2014, 1, 015004. [Google Scholar] [CrossRef]
- Shanmugam, K.; Doosthosseini, H.; Varanasi, S.; Garnier, G.; Batchelor, W. Nanocellulose films as air and water vapour barriers: A recyclable and biodegradable alternative to polyolefin packaging. Sustain. Mater. Technol. 2019, 22, e00115. [Google Scholar] [CrossRef]
- Yu, Z.; Alsammarraie, F.K.; Nayigiziki, F.X.; Wang, W.; Vardhanabhuti, B.; Mustapha, A.; Lin, M. Effect and mechanism of cellulose nanofibrils on the active functions of biopolymer-based nanocomposite films. Food Res. Int. 2017, 99, 166–172. [Google Scholar] [CrossRef]
- Atef, M.; Rezaei, M.; Behrooz, R. Characterization of physical, mechanical, and antibacterial properties of agar-cellulose bionanocomposite films incorporated with savory essential oil. Food Hydrocoll. 2015, 45, 150–157. [Google Scholar] [CrossRef]
- Kõrge, K.; Šeme, H.; Bajić, M.; Likozar, B.; Novak, U. Reduction in Spoilage Microbiota and Cyclopiazonic Acid Mycotoxin with Chestnut Extract Enriched Chitosan Packaging: Stability of Inoculated Gouda Cheese. Foods 2020, 9, 1645. [Google Scholar] [CrossRef]
- Lu, P.; Yang, Y.; Liu, R.; Liu, X.; Ma, J.; Wu, M.; Wang, S. Preparation of sugarcane bagasse nanocellulose hydrogel as a colourimetric freshness indicator for intelligent food packaging. Carbohydr. Polym. 2020, 249, 116831. [Google Scholar] [CrossRef]
- Skočaj, M. Bacterial nanocellulose in papermaking. Cellulose 2019, 26, 6477–6488. [Google Scholar] [CrossRef]
- Lavrič, G.; Medvešček, D.; Skočaj, M. Papermaking properties of bacterial nanocellulose produced from mother of vinegar, a waste product after classical vinegar production. Tappi J. 2020, 19, 197–203. [Google Scholar] [CrossRef]
- Kunaver, M.; Anžlovar, A.; Žagar, E. The fast and effective isolation of nanocellulose from selected cellulosic feedstocks. Carbohydr. Polym. 2016, 148, 251–258. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, W.; Teng, A.; Zhang, K.; Ma, Y.; Duan, S.; Li, S.; Guo, Y. Using cellulose nanofibers to reinforce polysaccharide films: Blending vs. layer-by-layer casting. Carbohydr. Polym. 2020, 227, 115264. [Google Scholar] [CrossRef]
- Soni, B.; Hassan, E.B.; Schilling, M.W.; Mahmoud, B. Transparent bionanocomposite films based on chitosan and TEMPO-oxidized cellulose nanofibers with enhanced mechanical and barrier properties. Carbohydr. Polym. 2016, 151, 779–789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nan, F.; Nagarajan, S.; Chen, Y.; Liu, P.; Duan, Y.; Men, Y.; Zhang, J. Enhanced Toughness and Thermal Stability of Cellulose Nanocrystal Iridescent Films by Alkali Treatment. ACS Sustain. Chem. Eng. 2017, 5, 8951–8958. [Google Scholar] [CrossRef]
- Wang, J.; Gardner, D.J.; Stark, N.M.; Bousfield, D.W.; Tajvidi, M.; Cai, Z. Moisture and Oxygen Barrier Properties of Cellulose Nanomaterial-Based Films. ACS Sustain. Chem. Eng. 2018, 6, 49–70. [Google Scholar] [CrossRef]
- Huq, T.; Salmieri, S.; Khan, A.; Khan, R.A.; Le Tien, C.; Riedl, B.; Fraschini, C.; Bouchard, J.; Uribe-Calderon, J.; Kamal, M.R.; et al. Nanocrystalline cellulose (NCC) reinforced alginate based biodegradable nanocomposite film. Carbohydr. Polym. 2012, 90, 1757–1763. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.; Wei, C.; Gong, Y.; Wang, S.; Ding, W. Mechanical and Water-Resistant Properties of Eco-Friendly Chitosan Membrane Reinforced with Cellulose Nanocrystals. Polymers 2019, 11, 166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cazón, P.; Velazquez, G.; Vázquez, M. UV-protecting films based on bacterial cellulose, glycerol and polyvinyl alcohol: Effect of water activity on barrier, mechanical and optical properties. Cellulose 2020, 27, 8199–8213. [Google Scholar] [CrossRef]
- Faradilla, R.F.; Lee, G.; Roberts, J.; Martens, P.; Stenzel, M.; Arcot, J. Effect of glycerol, nanoclay and graphene oxide on physicochemical properties of biodegradable nanocellulose plastic sourced from banana pseudo-stem. Cellulose 2017, 25, 399–416. [Google Scholar] [CrossRef]
- Filipova, I.; Serra, F.; Tarrés, Q.; Mutjé, P.; Delgado-Aguilar, M. Oxidative treatments for cellulose nanofibers production: A comparative study between TEMPO-mediated and ammonium persulfate oxidation. Cellulose 2020, 27, 10671–10688. [Google Scholar] [CrossRef]
- Zhao, Y.; Moser, C.; Lindström, M.E.; Henriksson, G.; Li, J. Cellulose Nanofibers from Softwood, Hardwood, and Tunicate: Preparation–Structure–Film Performance Interrelation. ACS Appl. Mater. Interfaces 2017, 9, 13508–13519. [Google Scholar] [CrossRef]
- Azeredo, H.M.; Mattoso, L.H.C.; Avena-Bustillos, R.J.; Filho, G.C.; Munford, M.L.; Wood, D.; McHugh, T.H. Nanocellulose Reinforced Chitosan Composite Films as Affected by Nanofiller Loading and Plasticizer Content. J. Food Sci. 2010, 75, N1–N7. [Google Scholar] [CrossRef]
- Qing, Y.; Sabo, R.; Wu, Y.; Cai, Z. High-Performance Cellulose Nanofibril Composite Films. BioResources 2012, 7, 3064–3075. [Google Scholar]
- Carneiro-Da-Cunha, M.G.; Cerqueira, M.; Souza, B.W.; Carvalho, S.; Quintas, M.A.; Teixeira, J.; Vicente, A.A. Physical and thermal properties of a chitosan/alginate nanolayered PET film. Carbohydr. Polym. 2010, 82, 153–159. [Google Scholar] [CrossRef]
- Spoljaric, S.; Salminen, A.; Luong, N.D.; Seppälä, J. Ductile Nanocellulose-Based Films with High Stretchability and Tear Resistance. Eur. Polym. J. 2015, 69, 328–340. [Google Scholar] [CrossRef]
- Enescu, D.; Gardrat, C.; Cramail, H.; Le Coz, C.; Sèbe, G.; Coma, V. Bio-Inspired Films Based on Chitosan, Nanoclays and Cellulose Nanocrystals: Structuring and Properties Improvement by Using Water-Evaporation-Induced Self-Assembly. Cellulose 2019, 26, 2389–2401. [Google Scholar] [CrossRef]
- Wang, L.; Chen, C.; Wang, J.; Gardner, D.J.; Tajvidi, M. Cellulose nanofibrils versus cellulose nanocrystals: Comparison of performance in flexible multilayer films for packaging applications. Food Packag. Shelf Life 2020, 23, 100464. [Google Scholar] [CrossRef]
- Ahankari, S.S.; Subhedar, A.R.; Bhadauria, S.S.; Dufresne, A. Nanocellulose in food packaging: A review. Carbohydr. Polym. 2021, 255, 117479. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Thakur, M.; Bhattacharya, M.; Mandal, T.; Goswami, S. Commercial application of cellulose nano-composites—A review. Biotechnol. Rep. 2019, 21, e00316. [Google Scholar] [CrossRef]
- Mascheroni, E.; Rampazzo, R.; Ortenzi, M.A.; Piva, G.; Bonetti, S.; Piergiovanni, L. Comparison of cellulose nanocrystals obtained by sulfuric acid hydrolysis and ammonium persulfate, to be used as coating on flexible food-packaging materials. Cellulose 2016, 23, 779–793. [Google Scholar] [CrossRef] [Green Version]
- Gregory, D.A.; Tripathi, L.; Fricker, A.T.; Asare, E.; Orlando, I.; Raghavendran, V.; Roy, I. Bacterial cellulose: A smart biomaterial with diverse applications. Mater. Sci. Eng. R Rep. 2021, 145, 100623. [Google Scholar] [CrossRef]
- Ali, A.; Shaker, K.; Nawab, Y.; Jabbar, M.; Hussain, T.; Militky, J.; Baheti, V. Hydrophobic treatment of natural fibers and their composites—A review. J. Ind. Text. 2018, 47, 2153–2183. [Google Scholar] [CrossRef]


| Sample Name | Film Composition | Thickness (µm) | Tensile Strength (MPa) | Strain at Break (%) | Water Contact Angle (°) |
|---|---|---|---|---|---|
| Film 1 | Alginate | 55 ± 3 | 40 ± 10 | 22 ± 6 | 39 ± 2 |
| Film 2 | Alginate + 5% CNC | 50 ± 2 | 43 ± 4 | 28 ± 4 | 58 ± 3 |
| Film 3 | Chitosan | 123 ± 3 | 14 ± 2 | 51 ± 4 | 75 ± 3 |
| Film 4 | Chitosan + 3% CNC | 70 ± 1 | 31 ± 2 | 55 ± 6 | 108 ± 2 |
| Film 5 | BNC | 66.4 ± 0.8 | 60 ± 11 | 4.2 ± 1 | 46 ± 3 |
| Film 6 | BNC + 30% glycerol | 56 ± 2 | 53 ± 5 | 10 ± 0.4 | 65 ± 4 |
| Film 7 | CNF + 30% glycerol | 31 ± 3 | 47 ± 3 | 2.4 ± 0.6 | 23 ± 1 |
| Sample Name | Film Composition | Film Density (g∙cm−3) | Moisture Absorption (%) | WVTR (g∙cm−1∙day−1) | WVP (g/(m∙s∙Pa)) |
|---|---|---|---|---|---|
| Film 1 | Alginate | 1.87 ± 0.2 | 57.4 ± 1.6 | 239 ± 8 | 9.36 |
| Film 2 | Alginate + 5% CNC | 1.34 ± 0.16 | 51.2 ± 1.9 | 203 ± 5 | 7.23 |
| Film 3 | Chitosan | 2.00 ± 0.35 | 49.2 ± 1.5 | 115 ± 9 | 10.07 |
| Film 4 | Chitosan + 3% CNC | 1.05 ± 0.10 | 36.5 ± 1.8 | 63 ± 2 | 1.39 |
| Film 5 | BNC | 0.74 ± 0.05 | 9.7 ± 0.8 | 77 ± 4 | 3.62 |
| Film 6 | BNC + 30% glycerol | 0.79 ± 0.10 | 21.3 ± 1.0 | 230 ± 11 | 9.17 |
| Film 7 | CNF + 30% glycerol | 1.29 ± 0.20 | 20.4 ± 0.6 | Not applicable | Not applicable |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lavrič, G.; Oberlintner, A.; Filipova, I.; Novak, U.; Likozar, B.; Vrabič-Brodnjak, U. Functional Nanocellulose, Alginate and Chitosan Nanocomposites Designed as Active Film Packaging Materials. Polymers 2021, 13, 2523. https://doi.org/10.3390/polym13152523
Lavrič G, Oberlintner A, Filipova I, Novak U, Likozar B, Vrabič-Brodnjak U. Functional Nanocellulose, Alginate and Chitosan Nanocomposites Designed as Active Film Packaging Materials. Polymers. 2021; 13(15):2523. https://doi.org/10.3390/polym13152523
Chicago/Turabian StyleLavrič, Gregor, Ana Oberlintner, Inese Filipova, Uroš Novak, Blaž Likozar, and Urška Vrabič-Brodnjak. 2021. "Functional Nanocellulose, Alginate and Chitosan Nanocomposites Designed as Active Film Packaging Materials" Polymers 13, no. 15: 2523. https://doi.org/10.3390/polym13152523
APA StyleLavrič, G., Oberlintner, A., Filipova, I., Novak, U., Likozar, B., & Vrabič-Brodnjak, U. (2021). Functional Nanocellulose, Alginate and Chitosan Nanocomposites Designed as Active Film Packaging Materials. Polymers, 13(15), 2523. https://doi.org/10.3390/polym13152523