Effect of Spherical Elements of Biosensors and Bioreactors on the Physicochemical Properties of a Peroxidase Protein
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Protein
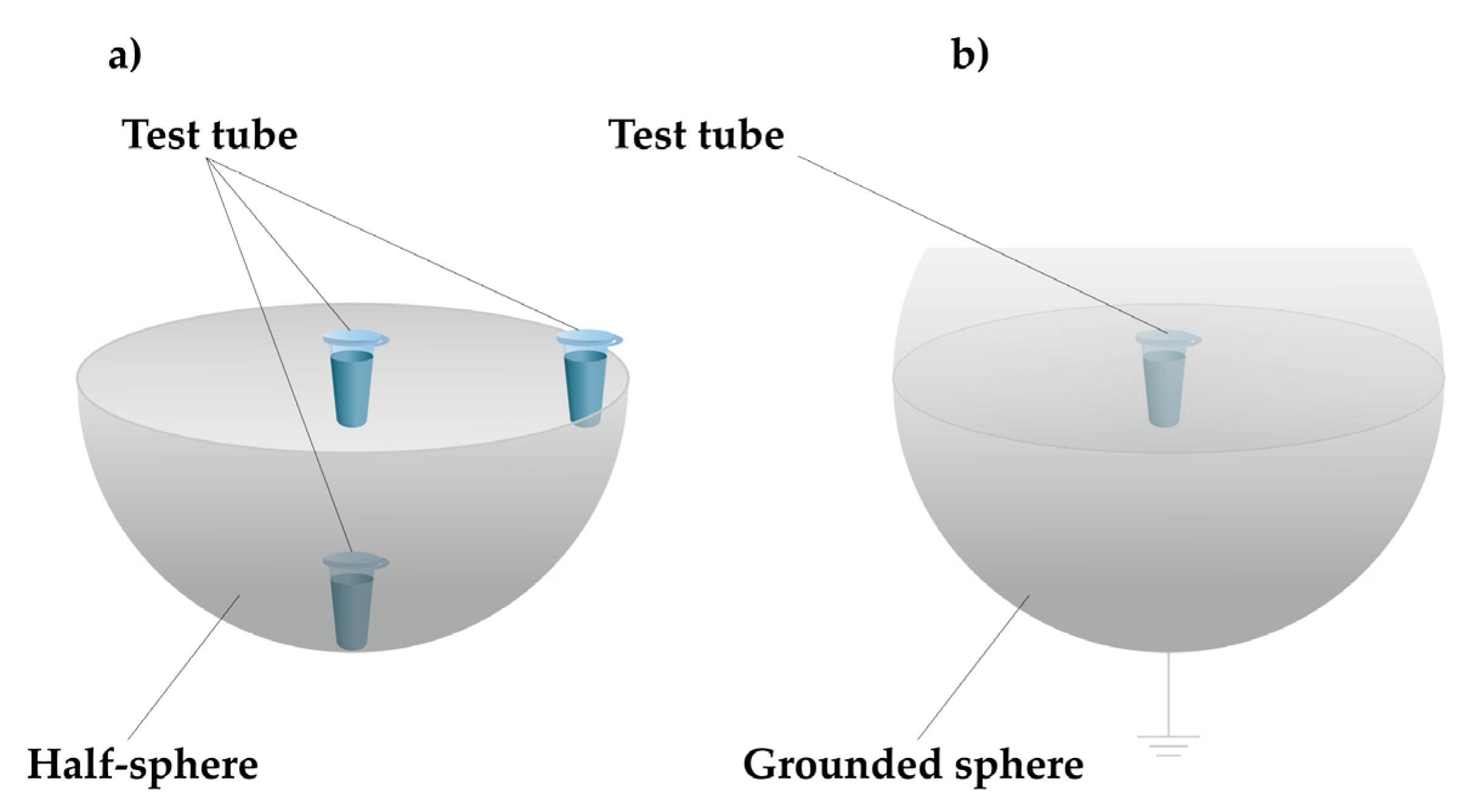
2.2. Experimental Setup
2.3. AFM Sample Preparation
2.4. AFM Measurements
2.5. ATR-FTIR Measurements
2.6. Spectrophotometry Measurements
3. Results
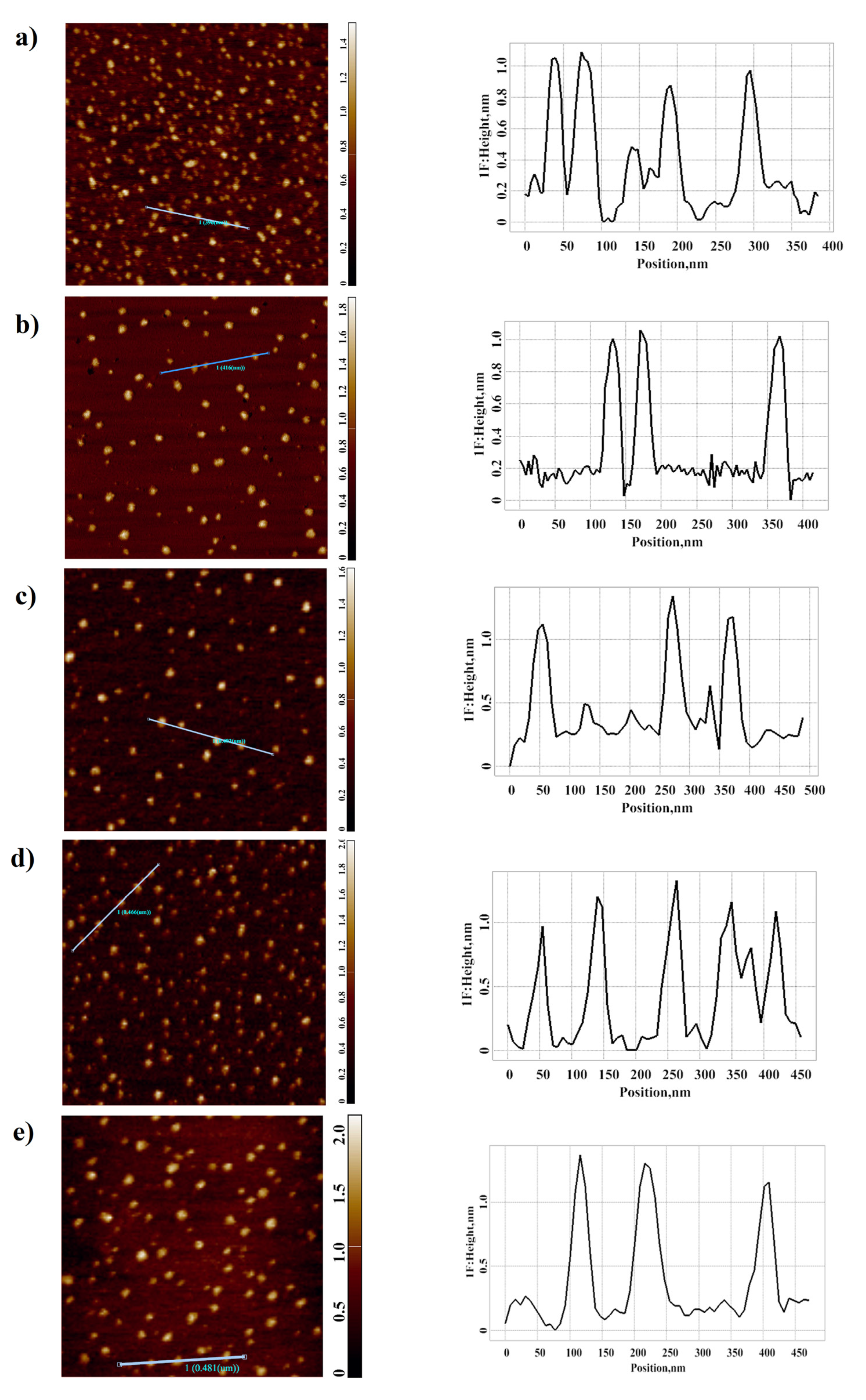
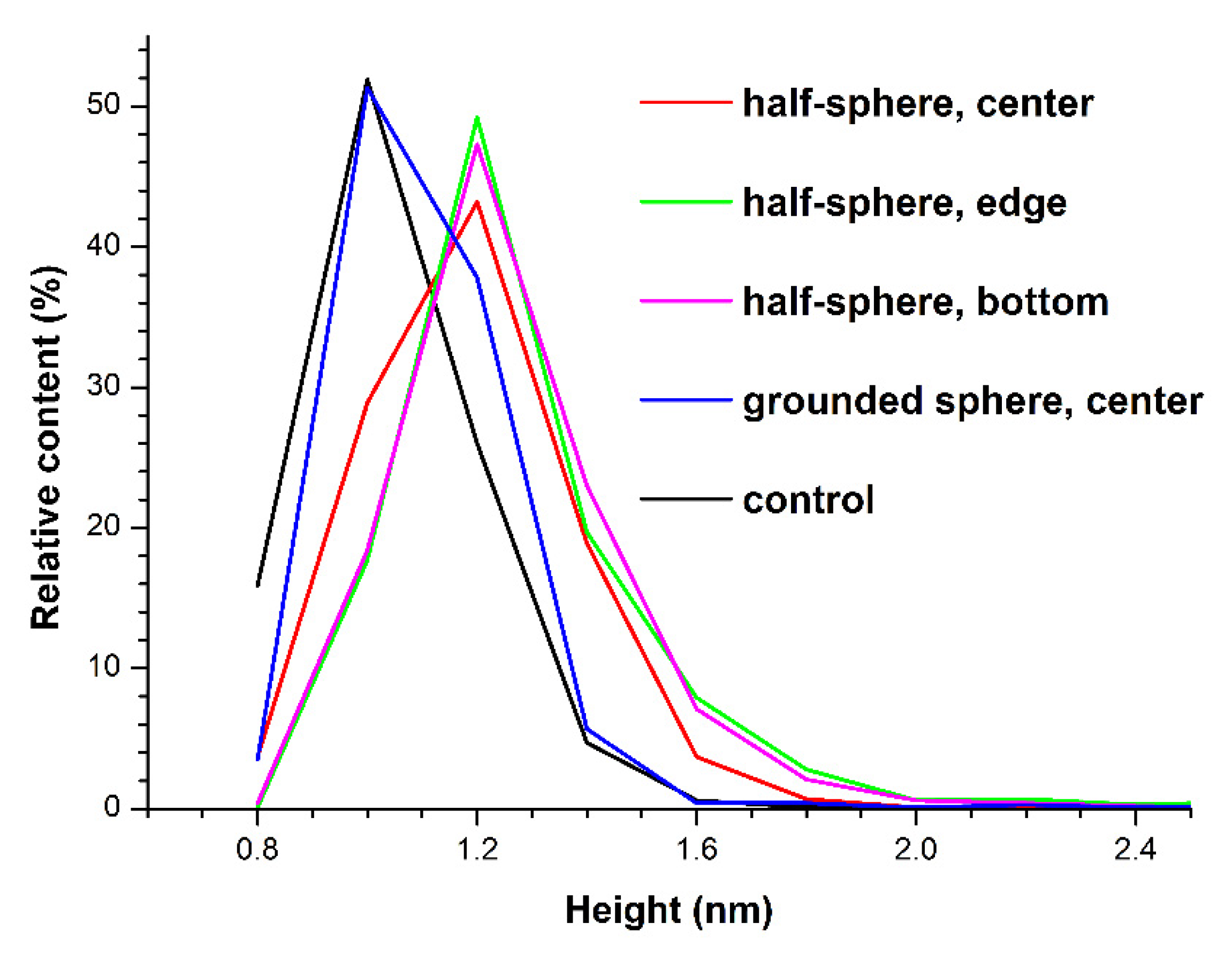
3.1. Atomic Force Microscopy
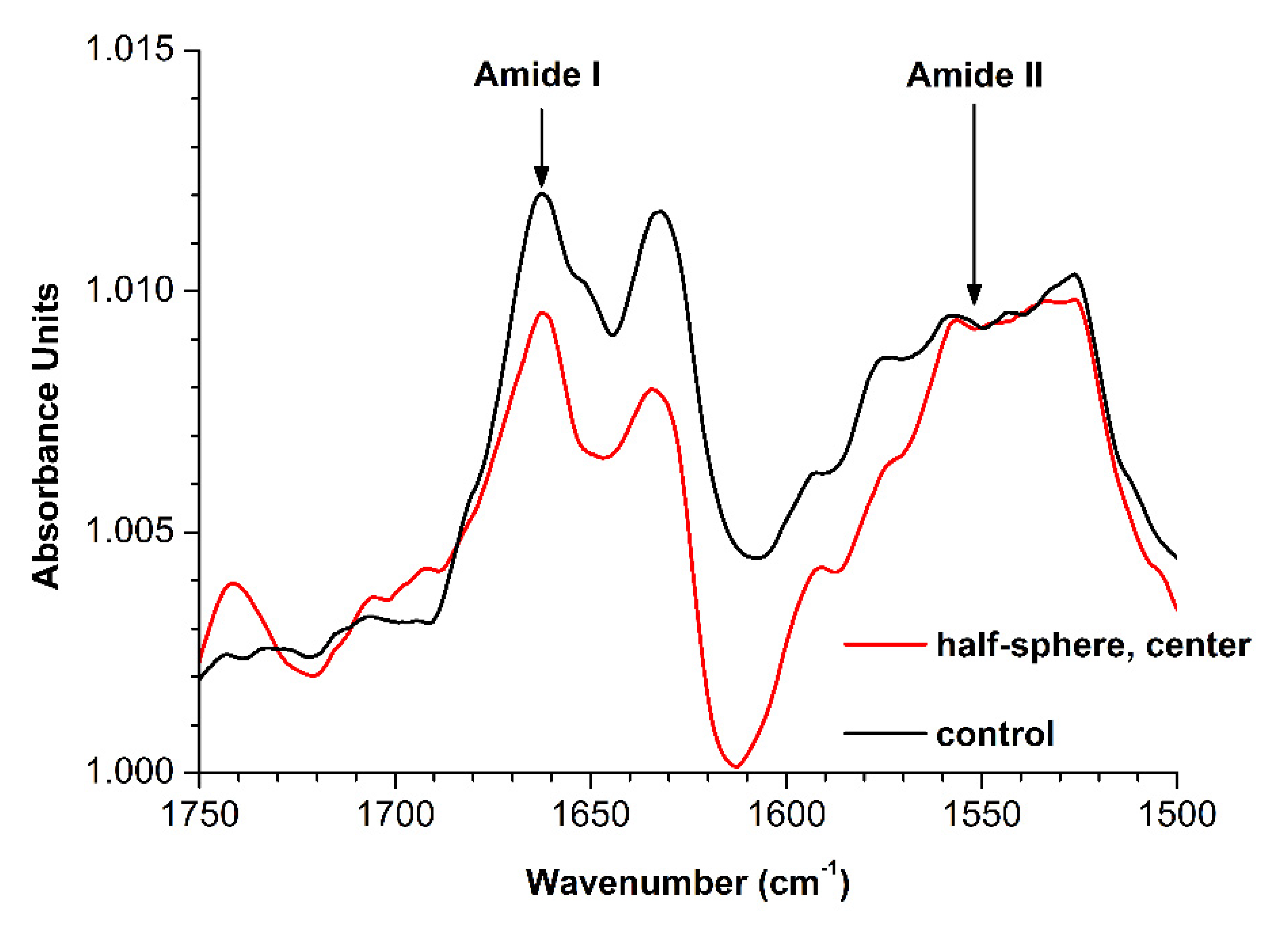
3.2. Attenuated Total Reflection Fourier Transform Infrared Spectroscopy
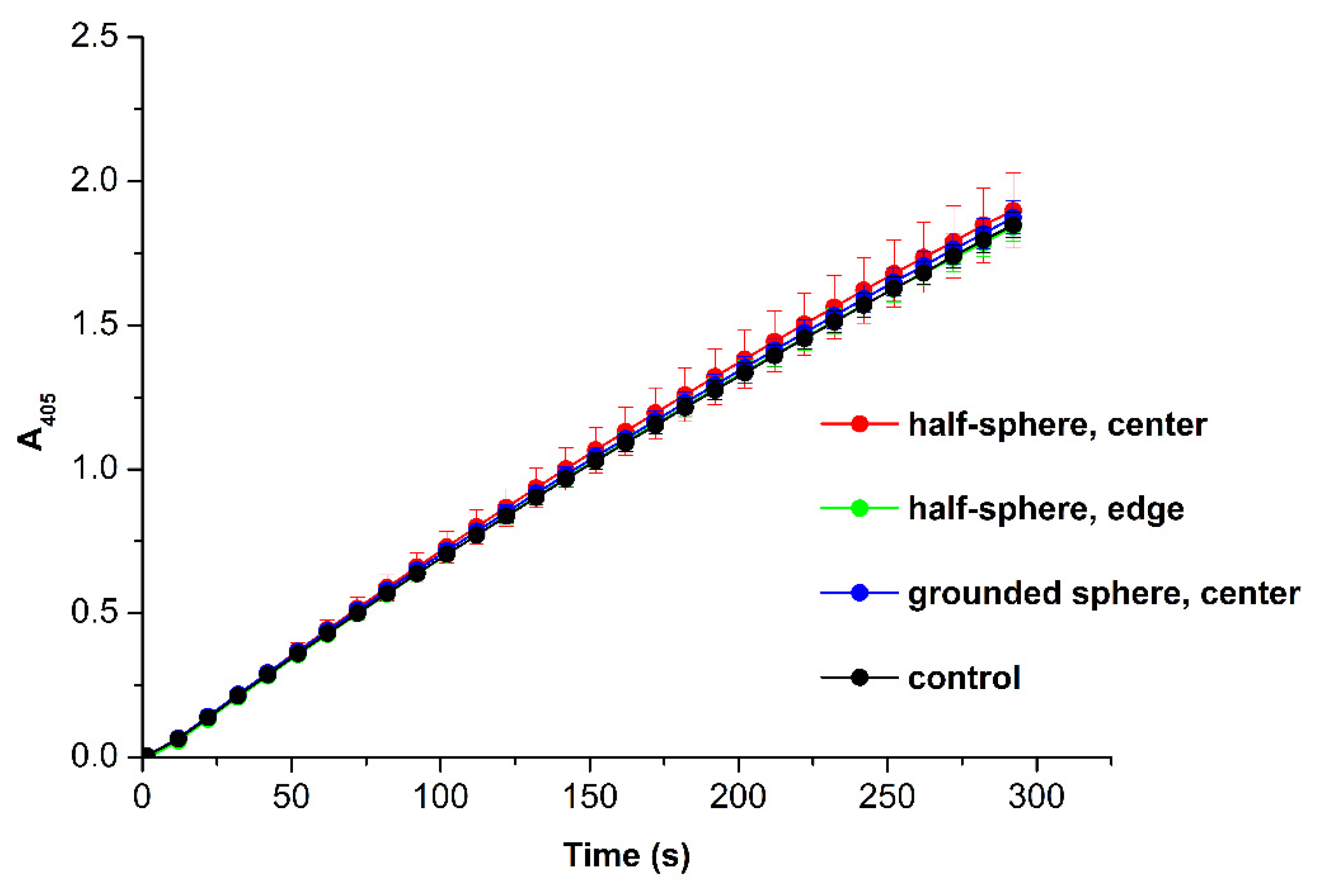
3.3. Spectrophotometry-Based Estimation of HRP Enzymatic Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Metzler, D.E. Biochemistry: The Chemical Reactions of Living Cells; Academic Press: Oxford, UK, 1970. [Google Scholar]
- Emamdadi, N.; Gholizadeh, M.; Housaindokht, M.R. Investigation of static magnetic field effect on horseradish peroxidase enzyme activity and stability in enzymatic oxidation process. Int. J. Biol. Macromol. 2021, 170, 189–195. [Google Scholar] [CrossRef]
- Wasak, A.; Drozd, R.; Jankowiak, D.; Rakoczy, R. The influence of rotating magnetic field on bio-catalytic dye degradation using the horseradish peroxidase. Biochem. Eng. J. 2019, 147, 81–88. [Google Scholar] [CrossRef]
- Ivanov, Y.D.; Pleshakova, T.O.; Shumov, I.D.; Kozlov, A.F.; Ivanova, I.A.; Valueva, A.A.; Tatur, V.Y.; Smelov, M.V.; Ivanova, N.D.; Ziborov, V.S. AFM Imaging of Protein Aggregation in Studying the Impact of Knotted Electromagnetic Field on A Peroxidase. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Ivanov, Y.D.; Pleshakova, T.O.; Shumov, I.D.; Kozlov, A.F.; Romanova, T.S.; Valueva, A.A.; Tatur, V.Y.; Stepanov, I.N.; Ziborov, V.S. Investigation of the Influence of Liquid Motion in a Flow-Based System on an Enzyme Aggregation State with an Atomic Force Microscopy Sensor: The Effect of Water Flow. Appl. Sci. 2020, 10, 4560. [Google Scholar] [CrossRef]
- Ivanov, Y.D.; Pleshakova, T.O.; Shumov, I.D.; Kozlov, A.F.; Ivanova, I.A.; Ershova, M.O.; Tatur, V.Y.; Ziborov, V.S. AFM Study of the Influence of Glycerol Flow on Horseradish Peroxidase near the in/out Linear Sections of a Coil. Appl. Sci. 2021, 11, 1723. [Google Scholar] [CrossRef]
- Ivanov, Y.D.; Pleshakova, T.O.; Shumov, I.D.; Kozlov, A.F.; Valueva, A.A.; Ivanova, I.A.; Ershova, M.O.; Larionov, D.I.; Repnikov, V.V.; Ivanova, N.D.; et al. AFM and FTIR Investigation of the Effect of Water Flow on Horseradish Peroxidase. Molecules 2021, 26, 306. [Google Scholar] [CrossRef]
- Hamedi, N.; Saviz, M.; Banaei, A.; Shabani, S.; Qafary, M.; Moosavi-Movahedi, A.A.; Faraji-Dana, R. Effects of circularly polarized electromagnetic fields on solvated hemoglobin structure. J. Mol. Liq. 2020, 312, 113283. [Google Scholar] [CrossRef]
- Lopes, L.C.; Barreto, M.T.; Gonçalves, K.M.; Alvarez, H.M.; Heredia, M.F.; De Souza, R.O.M.; Cordeiro, Y.; Dariva, C.; Fricks, A.T. Stability and structural changes of horseradish peroxidase: Microwave versus conventional heating treatment. Enzym. Microb. Technol. 2015, 69, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, Y.D.; Pleshakova, T.O.; Shumov, I.D.; Kozlov, A.F.; Ivanova, I.A.; Valueva, A.A.; Ershova, M.O.; Tatur, V.Y.; Stepanov, I.N.; Repnikov, V.V.; et al. AFM study of changes in properties of horseradish peroxidase after incubation of its solution near a pyramidal structure. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- CEM. Discover SP—Specifications. Available online: https://cem.com/en/discover-sp-specifications (accessed on 6 May 2021).
- Basso, A.; Serban, S. Industrial applications of immobilized enzymes: A review. Mol. Catal. 2019, 479, 110607. [Google Scholar] [CrossRef]
- Bilal, M.; Barceló, D.; Iqbal, H.M. Nanostructured materials for harnessing the power of horseradish peroxidase for tailored environmental applications. Sci. Total Environ. 2020, 749, 142360. [Google Scholar] [CrossRef]
- Delafosse, A.; Loubière, C.; Calvo, S.; Toye, D.; Olmos, E. Solid-liquid suspension of microcarriers in stirred tank bioreactor: Experimental and numerical analysis. Chem. Eng. Sci. 2018, 180, 52–63. [Google Scholar] [CrossRef]
- Sarkar, J.; Shekhawat, L.K.; Loomba, V.; Rathore, A.S.A.S. CFD of mixing of multi-phase flow in a bioreactor using population balance model. Biotechnol. Prog. 2016, 32, 613–628. [Google Scholar] [CrossRef]
- Balezin, M.; Baryshnikova, K.V.; Kapitanova, P.; Evlyukhin, A.B. Electromagnetic properties of the Great Pyramid: First multipole resonances and energy concentration. J. Appl. Phys. 2018, 124, 034903. [Google Scholar] [CrossRef]
- El-kaliuoby, M.I.; El-khatib, A.; Mohamed, M.; Roston, G.; Khalil, A.M.; Ahmed, M.A.M.; AbdulSalam, H. Monitoring of Rat’s Blood Rheology after Exposure to 50-Hz Electric Fields (Occupational Study). OSR J. Environ. Sci. Toxicol. Food Technol. (IOSR-JESTFT) 2016, 10, 4–8. [Google Scholar] [CrossRef]
- Pershin, S.M. Conversion of ortho-para H2O isomers in water and a jump in erythrocyte fluidity through a microcapillary at a temperature of 36.6 ± 0.3 °C. Phys. Wave Phenom. 2009, 17, 241–250. [Google Scholar] [CrossRef]
- Davies, P.; Rennke, H.; Cotran, R. Influence of molecular charge upon the endocytosis and intracellular fate of peroxidase activity in cultured arterial endothelium. J. Cell Sci. 1981, 49, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Welinder, K.G. Amino Acid Sequence Studies of Horseradish Peroxidase. Amino and Carboxyl Termini, Cyanogen Bromide and Tryptic Fragments, the Complete Sequence, and Some Structural Characteristics of Horseradish Peroxidase C. JBIC J. Biol. Inorg. Chem. 1979, 96, 483–502. [Google Scholar] [CrossRef]
- Veitch, N.C. Horseradish peroxidase: A modern view of a classic enzyme. Phytochemistry 2004, 65, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Tams, J.; Welinder, K.G. Mild Chemical Deglycosylation of Horseradish Peroxidase Yields a Fully Active, Homogeneous Enzyme. Anal. Biochem. 1995, 228, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Ignatenko, O.V.; Sjölander, A.; Hushpulian, D.M.; Kazakov, S.V.; Ouporov, I.V.; Chubar, T.A.; Poloznikov, A.A.; Ruzgas, T.; Tishkov, V.I.; Gorton, L.; et al. Electrochemistry of chemically trapped dimeric and monomeric recombinant horseradish peroxidase. Adv. Biosens. Bioelectron. 2013, 2, 25–34. [Google Scholar]
- Ivanov, Y.D.; Danichev, V.V.; Pleshakova, T.O.; Shumov, I.D.; Ziborov, V.S.; Krokhin, N.V.; Zagumenniy, M.N.; Ustinov, V.S.; Smirnov, L.P.; Shironin, A.V.; et al. Irreversible chemical AFM-based fishing for detection of low-copied proteins. Biochem. (Moscow) Suppl. Ser. B Biomed. Chem. 2013, 7, 46–61. [Google Scholar] [CrossRef]
- Kumaki, J.; Nishikawa, Y.; Hashimoto, T. Visualization of Single-Chain Conformations of a Synthetic Polymer with Atomic Force Microscopy. J. Am. Chem. Soc. 1996, 118, 3321–3322. [Google Scholar] [CrossRef]
- Ierardi, V.; Ferrera, F.; Millo, E.; Damonte, G.; Filaci, G.; Valbusa, U. Bioactive surfaces for antibody-antigen complex detection by Atomic Force Microscopy. J. Phys. Conf. Ser. 2013, 439, 012001. [Google Scholar] [CrossRef]
- Ivanov, Y.D.; Bukharina, N.S.; Frantsuzov, P.A.; Pleshakova, T.O.; Kanashenko, S.L.; Medvedeva, N.V.; Argentova, V.V.; Zgoda, V.G.; Munro, A.W.; Archakov, A.I. AFM study of cytochrome CYP102A1 oligomeric state. Soft Matter 2012, 8, 4602–4608. [Google Scholar] [CrossRef]
- Kaysheva, A.L.; Frantsuzov, P.A.; Kopylov, A.T.; Pleshakova, T.O.; Stepanov, A.A.; Malsagova, K.A.; Archakov, A.I.; Ivanov, Y.D. Mass Spectrometric Identification of Proteins Enhanced by the Atomic Force Microscopy Immobilization Surface. Int. J. Mol. Sci. 2021, 22, 431. [Google Scholar] [CrossRef]
- Pleshakova, T.O.; Bukharina, N.S.; Archakov, A.I.; Ivanov, Y.D. Atomic Force Microscopy for Protein Detection and Their Physicochemical Characterization. Int. J. Mol. Sci. 2018, 19, 1142. [Google Scholar] [CrossRef]
- Crampton, N.; Bonass, W.A.; Kirkham, J.; Thomson, N.H. Formation of Aminosilane-Functionalized Mica for Atomic Force Microscopy Imaging of DNA. Langmuir 2005, 21, 7884–7891. [Google Scholar] [CrossRef]
- Limanskaya, L.A.; Limanskii, A.P. Compaction of single supercoiled DNA molecules adsorbed onto amino mica. Russ. J. Bioorg. Chem. 2006, 32, 444–459. [Google Scholar] [CrossRef]
- Lyubchenko, Y.L.; Gall, A.A.; Shlyakhtenko, L.S.; Harrington, R.E.; Jacobs, B.L.; Oden, P.I.; Lindsay, S.M. Atomic Force Microscopy Imaging of Double Stranded DNA and RNA. J. Biomol. Struct. Dyn. 1992, 10, 589–606. [Google Scholar] [CrossRef]
- Lyubchenko, Y.; Shlyakhtenko, L.; Harrington, R.; Oden, P.; Lindsay, S. Atomic force microscopy of long DNA: Imaging in air and under water. Proc. Natl. Acad. Sci. USA 1993, 90, 2137–2140. [Google Scholar] [CrossRef] [PubMed]
- Shlyakhtenko, L.S.; Gall, A.A.; Weimer, J.J.; Hawn, D.D.; Lyubchenko, Y.L. Atomic Force Microscopy Imaging of DNA Covalently Immobilized on a Functionalized Mica Substrate. Biophys. J. 1999, 77, 568–576. [Google Scholar] [CrossRef]
- Braga, P.C.; Ricci, D. Atomic Force Microscopy. Biology: Biomedical Methods and Applications. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2003; Volume 242, p. 382. [Google Scholar]
- Archakov, A.I.; Ivanov, Y.D. Analytical nanobiotechnology for medicine diagnostics. Mol. BioSyst. 2007, 3, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, D.; Strzelecki, J.; Pawlak, K.; Dahm, H.; Balter, A. Effect of ampicillin on adhesive properties of bacteria examined by atomic force microscopy. Micron 2018, 112, 84–90. [Google Scholar] [CrossRef]
- Kiselyova, O.I.; Yaminsky, I.V.; Ivanov, Y.D.; Kanaeva, I.P.; Kuznetsov, V.Y.; Archakov, A.I. AFM Study of Membrane Proteins, Cytochrome P450 2B4, and NADPH–Cytochrome P450 Reductase and Their Complex Formation. Arch. Biochem. Biophys. 1999, 371, 1–7. [Google Scholar] [CrossRef]
- Pleshakova, T.O.; Kaysheva, A.L.; Shumov, I.D.; Ziborov, V.S.; Bayzyanova, J.M.; Konev, V.A.; Uchaikin, V.F.; Archakov, A.I.; Ivanov, Y.D. Detection of Hepatitis C Virus Core Protein in Serum Using Aptamer-Functionalized AFM Chips. Micromachines 2019, 10, 129. [Google Scholar] [CrossRef]
- Sanders, S.A.; Bray, R.C.; Smith, A.T. pH-Dependent properties of a mutant horseradish peroxidase isoenzyme C in which Arg38 has been replaced with lysine. Eur. J. Biochem. 1994, 224, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Kondyurin, A.V.; Naseri, P.; Tilley, J.M.R.; Nosworthy, N.J.; Bilek, M.M.M.; McKenzie, D.R. Mechanisms for Covalent Immobilization of Horseradish Peroxidase on Ion-Beam-Treated Polyethylene. Science 2012, 2012, 1–28. [Google Scholar] [CrossRef]
- Ryzhkina, I.S.; Kiseleva, Y.V.; Murtazina, L.I.; Mishina, O.A.; Sherman, E.D.; Konovalov, A.I. Comparative study of self-organization and physicochemical properties of highly diluted aqueous solutions of phenol bioantioxidants. Dokl. Phys. Chem. 2012, 447, 203–206. [Google Scholar] [CrossRef]
- Pershin, S.M. Konovalov effect in aqueous solutions of low concentrations: The role of spin ortho-paraisomers of H2O. Rep. Russ. Acad. Sci. 2014, 455, 44–47. [Google Scholar]
- Pershin, S.M. A New of the Action of EMF on Water/Aqueous Solutions, Taking into Account the Quantum Differences of the Ortho/Para of Spin Isomers of H2O. Online Biophysical Blog. Available online: http://www.biophys.ru/archive/sarov2013/proc-p17.pdf (accessed on 6 May 2021).
- Shannon, L.M.; Kay, E.; Lew, J.Y. Peroxidase Isozymes from Horseradish Roots. J. Biol. Chem. 1966, 241, 2166–2172. [Google Scholar] [CrossRef]
- Kaushal, S.; Nanda, S.S.; Samal, S.; Yi, D.K. Strategies for the Development of Metallic-Nanoparticle-Based Label-Free Biosensors and Their Biomedical Applications. ChemBioChem 2020, 21, 576–600. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivanov, Y.D.; Tatur, V.Y.; Pleshakova, T.O.; Shumov, I.D.; Kozlov, A.F.; Valueva, A.A.; Ivanova, I.A.; Ershova, M.O.; Ivanova, N.D.; Repnikov, V.V.; et al. Effect of Spherical Elements of Biosensors and Bioreactors on the Physicochemical Properties of a Peroxidase Protein. Polymers 2021, 13, 1601. https://doi.org/10.3390/polym13101601
Ivanov YD, Tatur VY, Pleshakova TO, Shumov ID, Kozlov AF, Valueva AA, Ivanova IA, Ershova MO, Ivanova ND, Repnikov VV, et al. Effect of Spherical Elements of Biosensors and Bioreactors on the Physicochemical Properties of a Peroxidase Protein. Polymers. 2021; 13(10):1601. https://doi.org/10.3390/polym13101601
Chicago/Turabian StyleIvanov, Yuri D., Vadim Yu. Tatur, Tatyana O. Pleshakova, Ivan D. Shumov, Andrey F. Kozlov, Anastasia A. Valueva, Irina A. Ivanova, Maria O. Ershova, Nina D. Ivanova, Victor V. Repnikov, and et al. 2021. "Effect of Spherical Elements of Biosensors and Bioreactors on the Physicochemical Properties of a Peroxidase Protein" Polymers 13, no. 10: 1601. https://doi.org/10.3390/polym13101601
APA StyleIvanov, Y. D., Tatur, V. Y., Pleshakova, T. O., Shumov, I. D., Kozlov, A. F., Valueva, A. A., Ivanova, I. A., Ershova, M. O., Ivanova, N. D., Repnikov, V. V., Stepanov, I. N., & Ziborov, V. S. (2021). Effect of Spherical Elements of Biosensors and Bioreactors on the Physicochemical Properties of a Peroxidase Protein. Polymers, 13(10), 1601. https://doi.org/10.3390/polym13101601




