Epoxidation of Cardanol’s Terminal Double Bond
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
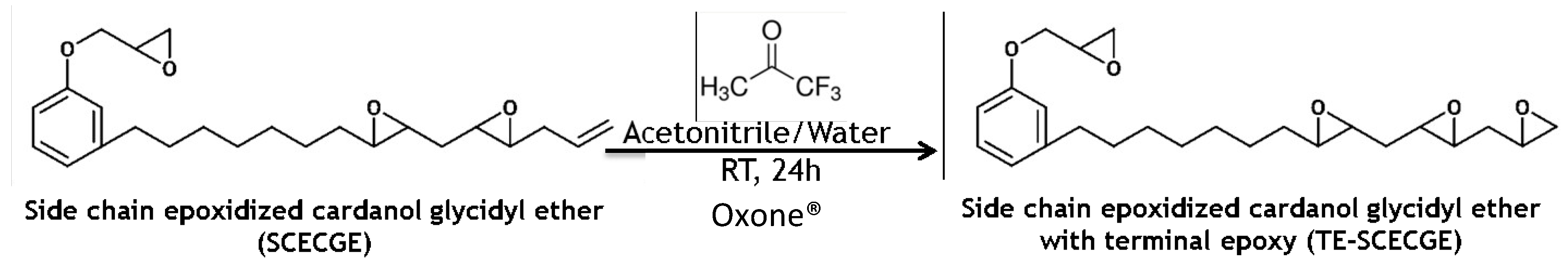
2.2. Epoxidation of the Terminal Double Bond via Oxone®
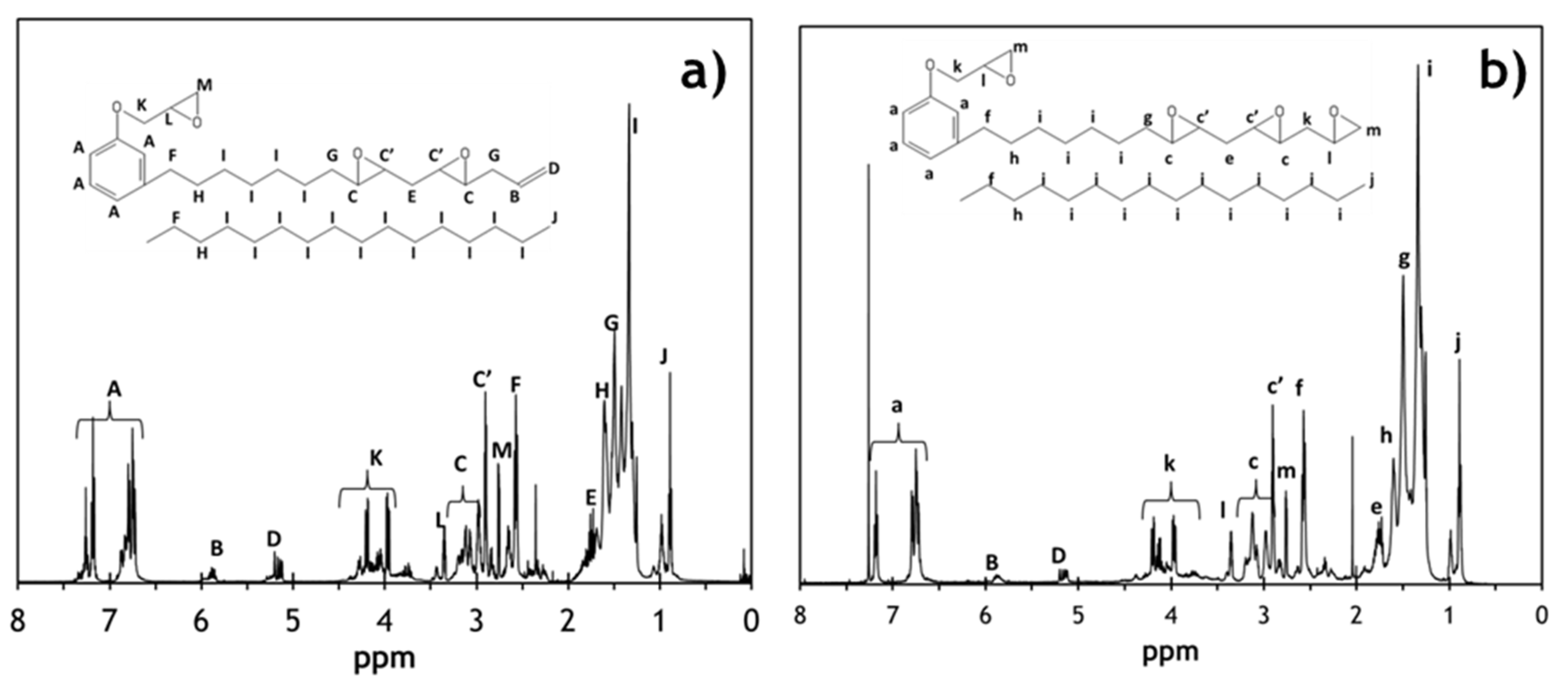
2.3. Characterization of the TE-SCECGE Resin
2.4. Preparation of Cured Samples of SCECGE and TE-SECCGE with PACM
2.5. Properties of the Cured Epoxy–Amine Polymer
3. Results and Discussion
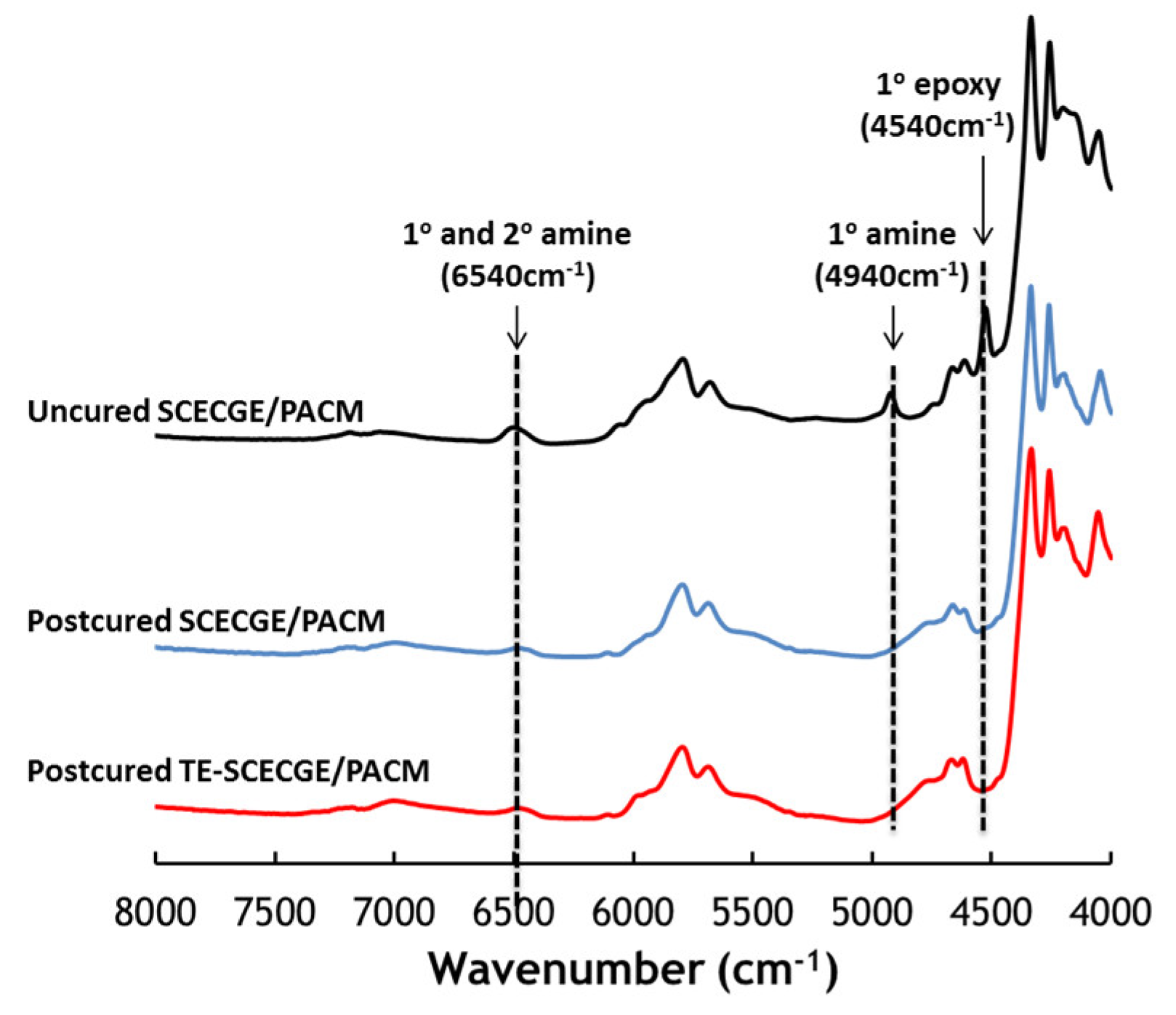
3.1. Characterization of the Epoxidized Cardanol
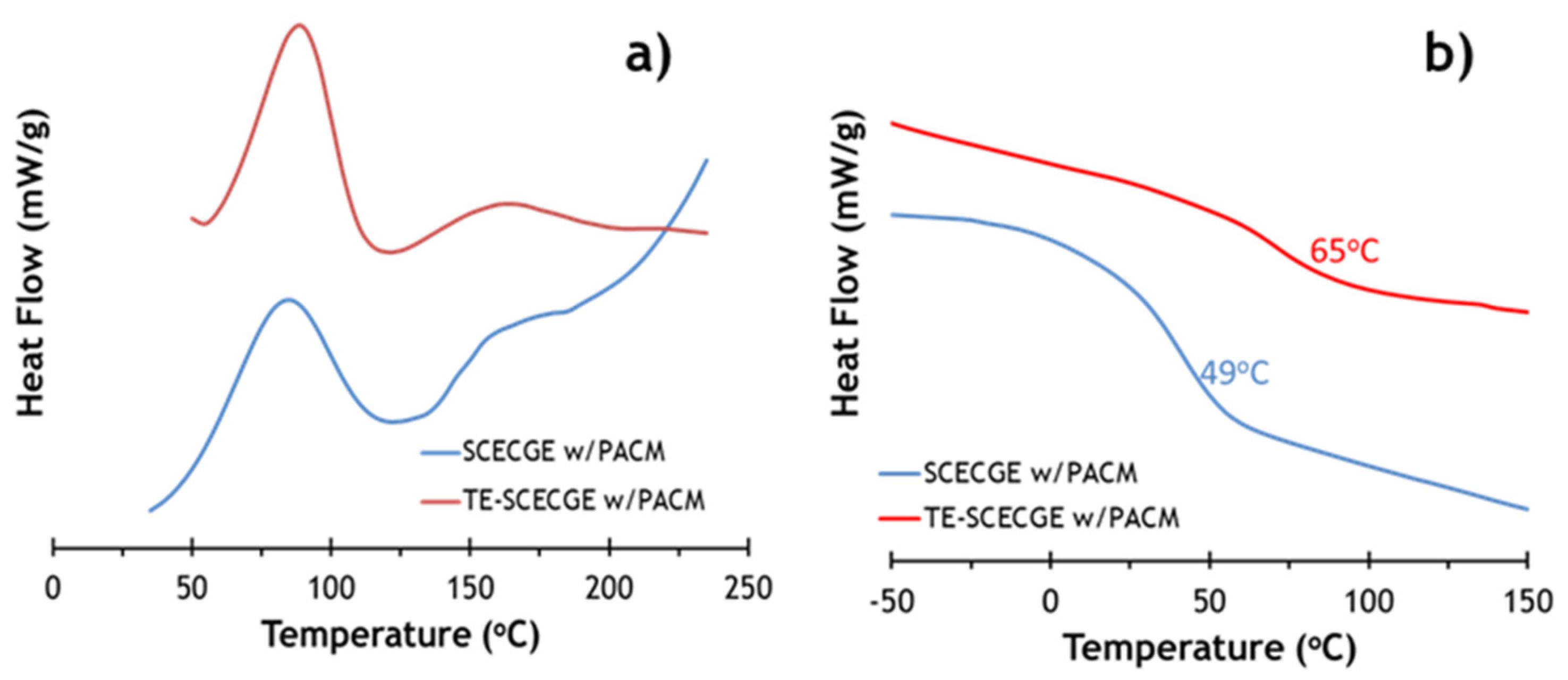
3.2. Curing of the Epoxy Resins with PACM
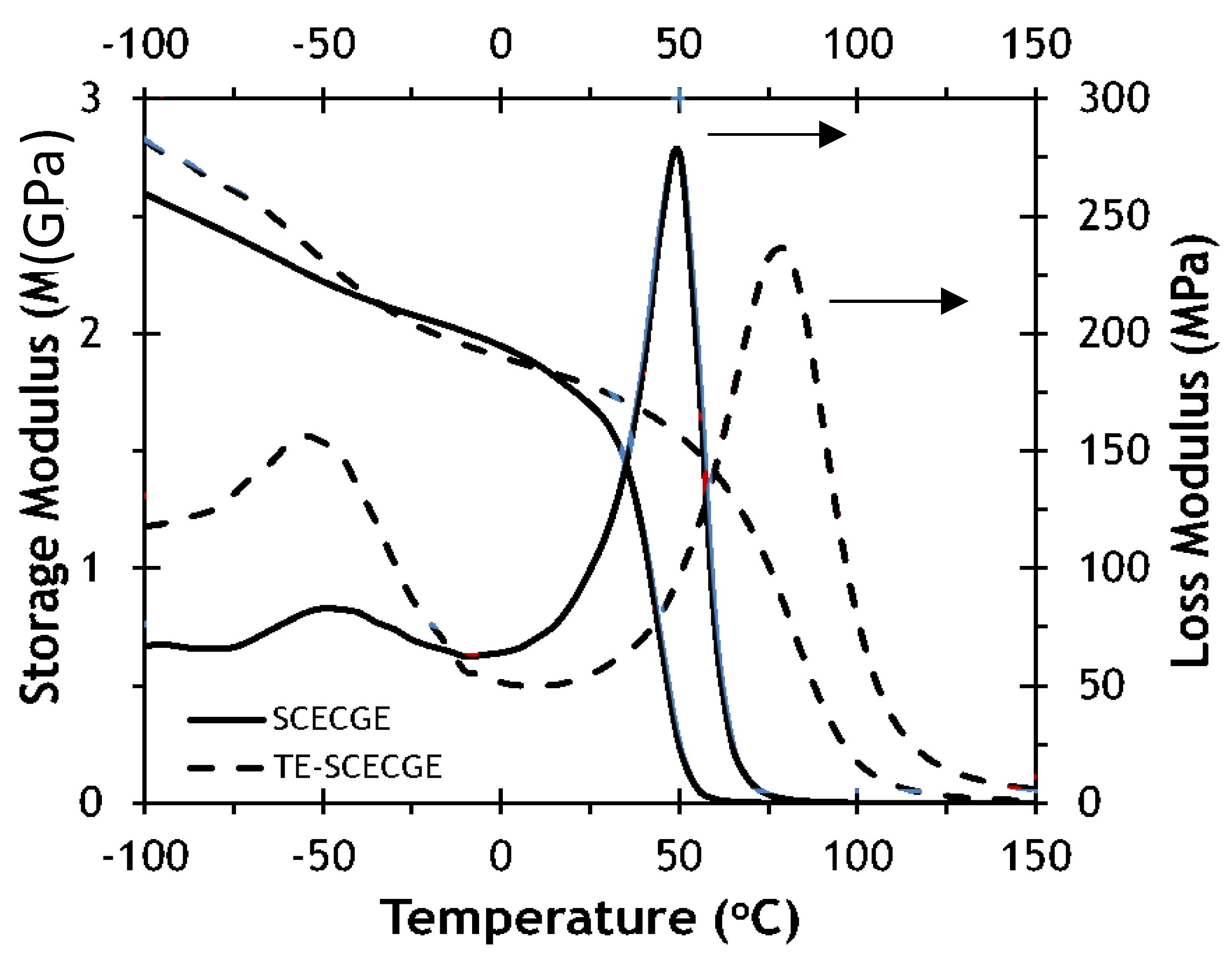
3.3. Comparison of the Thermomechanical and Mechanical Properties of SCECGE and TE-SCECGE
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Can, E.; Kınacı, E.; Palmese, G.R. Preparation and characterization of novel vinyl ester formulations derived from cardanol. Eur. Polym. J. 2015, 72, 129–147. [Google Scholar] [CrossRef]
- Voirin, C.; Caillol, S.; Sadavarte, N.V.; Tawade, B.V.; Boutevin, B.; Wadgaonkar, P.P. Functionalization of cardanol: Towards biobased polymers and additives. Polym. Chem. 2014, 5, 3142–3162. [Google Scholar] [CrossRef]
- Jaillet, F.; Nouailhas, H.; Auvergne, R.; Ratsimihety, A.; Boutevin, B.; Caillol, S. Synthesis and characterization of novel vinylester prepolymers from cardanol. Eur. J. Lipid Sci. Technol. 2014, 116, 928–939. [Google Scholar] [CrossRef]
- Jaillet, F.; Nouailhas, H.; Boutevin, B.; Caillol, S. Synthesis of novel bio-based vinyl ester from dicyclopentadiene prepolymer, cashew nut shell liquid, and soybean oil. Eur. J. Lipid Sci. Technol. 2016, 118. [Google Scholar] [CrossRef]
- Suresh, K.I.; Kishanprasad, V.S. Synthesis, structure, and properties of novel polyols from cardanol and developed polyurethanes. Ind. Eng. Chem. Res. 2005, 44, 4504–4512. [Google Scholar] [CrossRef]
- Suresh, K.I. Rigid Polyurethane Foams from Cardanol: Synthesis, Structural Characterization, and Evaluation of Polyol and Foam Properties. ACS Sustain. Chem. Eng. 2013, 1, 232–242. [Google Scholar] [CrossRef]
- Kathalewar, M.; Sabnis, A.; D’Melo, D. Polyurethane coatings prepared from CNSL based polyols: Synthesis, characterization and properties. Prog. Org. Coat. 2014, 77, 616–626. [Google Scholar] [CrossRef]
- Kathalewar, M.; Sabnis, A.; D’Mello, D. Isocyanate free polyurethanes from new CNSL based bis-cyclic carbonate and its application in coatings. Eur. Polym. J. 2014, 57, 99–108. [Google Scholar] [CrossRef]
- Devi, A.; Srivastava, D. Studies on the blends of cardanol-based epoxidized novolac resin and CTPB. Eur. Polym. J. 2007, 43, 2422–2432. [Google Scholar] [CrossRef]
- Devi, A.; Srivastava, D. Studies on the blends of cardanol-based epoxidized novolac type phenolic resin and carboxyl-terminated polybutadiene (CTPB), I. Mater. Sci. Eng. A 2007, 458, 336–347. [Google Scholar] [CrossRef]
- Calò, E.; Maffezzoli, A.; Mele, G.; Martina, F.; Mazzetto, S.E.; Tarzia, A.; Stifani, C. Synthesis of a novel cardanol-based benzoxazine monomer and environmentally sustainable production of polymers and bio-composites. Green Chem. 2007, 9, 754–759. [Google Scholar] [CrossRef]
- Rao, B.S.; Palanisamy, A. Synthesis of bio based low temperature curable liquid epoxy, benzoxazine monomer system from cardanol: Thermal and viscoelastic properties. Eur. Polym. J. 2013, 49, 2365–2376. [Google Scholar] [CrossRef]
- Rao, B.; Palanisamy, A. Monofunctional benzoxazine from cardanol for bio-composite applications. React. Funct. Polym. 2011, 71, 148–154. [Google Scholar] [CrossRef]
- Jaillet, F.; Darroman, E.; Ratsimihety, A.; Auvergne, R.; Boutevin, B.; Caillol, S. New biobased epoxy materials from cardanol. Eur. J. Lipid Sci. Technol. 2014, 116, 63–73. [Google Scholar] [CrossRef]
- Kanehashi, S.; Yokoyama, K.; Masuda, R.; Kidesaki, T.; Nagai, K.; Miyakoshi, T. Preparation and characterization of cardanol-based epoxy resin for coating at room temperature curing. J. Appl. Polym. Sci. 2013, 130, 2468–2478. [Google Scholar] [CrossRef]
- Huang, K.; Zhang, Y.; Li, M.; Lian, J.; Yang, X.; Xia, J. Preparation of a light color cardanol-based curing agent and epoxy resin composite: Cure-induced phase separation and its effect on properties. Prog. Org. Coat. 2012, 74, 240–247. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, S.; Guo, W.-W.; Wang, P.-L.; Xing, W.; Song, L.; Hu, Y. Renewable Cardanol-Based Phosphate as a Flame Retardant Toughening Agent for Epoxy Resins. ACS Sustain. Chem. Eng. 2017, 5. [Google Scholar] [CrossRef]
- Chen, J.; Nie, X.; Liu, Z.; Mi, Z.; Zhou, Y. Synthesis and application of polyepoxide cardanol glycidyl ether as biobased polyepoxide reactive diluent for epoxy resin. ACS Sustain. Chem. Eng. 2015, 3, 1164–1171. [Google Scholar] [CrossRef]
- Kim, Y.H.; An, E.S.; Park, S.Y.; Song, B.K. Enzymatic epoxidation and polymerization of cardanol obtained from a renewable resource and curing of epoxide-containing polycardanol. J. Mol. Catal. B Enzym. 2007, 45, 39–44. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, Y.H.; Won, K.; Song, B.K. Enzymatic synthesis and curing of polycardol from renewable resources. J. Mol. Catal. B Enzym. 2009, 57, 312–316. [Google Scholar] [CrossRef]
- Kinaci, E.; Can, E.; La Scala, J.J.; Palmese, G.R. Influence of epoxidized cardanol functionality and reactivity on network formation and properties. Polymers 2020, 12, 1956. [Google Scholar] [CrossRef]
- Armstrong, A.; Hayter, B.R. α-Functionalised ketones as promoters of alkene epoxidation by Oxone®. Tetrahedron 1999, 55, 11119–11126. [Google Scholar] [CrossRef]
- Bez, G.; Zhao, C.-G. First highly enantioselective epoxidation of alkenes with aldehyde/Oxone®. Tetrahedron Lett. 2003, 44, 7403–7406. [Google Scholar] [CrossRef]
- Ho, C.-Y.; Chen, Y.-C.; Wong, M.-K.; Yang, D. Fluorinated chiral secondary amines as catalysts for epoxidation of olefins with oxone. J. Org. Chem. 2005, 70, 898–906. [Google Scholar] [CrossRef]
- Denmark, S.E.; Wu, Z.; Crudden, C.M.; Matsuhashi, H. Catalytic epoxidation of alkenes with oxone. 2. Fluoro ketones. J. Org. Chem. 1997, 62, 8288–8289. [Google Scholar] [CrossRef] [PubMed]
- Aouf, C.; Nouailhas, H.; Fache, M.; Caillol, S.; Boutevin, B.; Fulcrand, H. Multi-functionalization of gallic acid. Synthesis of a novel bio-based epoxy resin. Eur. Polym. J. 2013, 49, 1185–1195. [Google Scholar] [CrossRef]
- McAninch, I.M.; Palmese, G.R.; Lenhart, J.L.; La Scala, J.J. DMA testing of epoxy resins: The importance of dimensions. Polym. Eng. Sci. 2015, 55, 2761–2774. [Google Scholar] [CrossRef]
- Hu, F.; Yadav, S.K.; La Scala, J.J.; Sadler, J.M.; Palmese, G.R. Preparation and Characterization of Fully Furan-Based Renewable Thermosetting Epoxy-Amine Systems. Macromol. Chem. Phys. 2015, 216, 1441–1446. [Google Scholar] [CrossRef]
- Horie, K.; Hiura, H.; Sawada, M.; Mita, I.; Kambe, H. Calorimetric investigation of polymerization reactions. III. Curing reaction of epoxides with amines. J. Polym. Sci. Part A-1 Polym. Chem. 1970, 8, 1357–1372. [Google Scholar] [CrossRef]
- Riccardi, C.; Adabbo, H.; Williams, R. Curing reaction of epoxy resins with diamines. J. Appl. Polym. Sci. 1984, 29, 2481–2492. [Google Scholar] [CrossRef]
- Hill, L.W. Calculation of crosslink density in short chain networks. Prog. Org. Coat. 1997, 31, 235–243. [Google Scholar] [CrossRef]
- Soles, C.L.; Yee, A.F. A discussion of the molecular mechanisms of moisture transport in epoxy resins. J. Polym. Sci. Part B Polym. Phys. 2000, 38, 792–802. [Google Scholar] [CrossRef]
- La Scala, J.; Wool, R.P. Fundamental thermo-mechanical property modeling of triglyceride-based thermosetting resins. J. Appl. Polym. Sci. 2013, 127, 1812–1826. [Google Scholar] [CrossRef]
- Choi, S.C. Introductory Applied Statistics in Science; Prentice-Hall: Upper Saddle River, NJ, USA, 1978. [Google Scholar]
| Epoxy Resin | Terminal Double Bond Functionality (per Molecule) | Primary Epoxy Functionality (per Molecule) | Secondary Epoxy Functionality (per Molecule) | Total Epoxy Functionality (per Molecule) | EEW (g/Equivalent) |
|---|---|---|---|---|---|
| SCECGE [21] | 0.32 | 1.05 | 1.40 | 2.45 | 177 |
| TE-SCECGE | 0.10 | 1.25 | 1.40 | 2.65 | 165 |
| Epoxy Resin | ΔHRXN (J/mol) | Tg (DSC) (°C) | Cross-Link Density (mol/m3) | Tg (DMA) (°C) |
|---|---|---|---|---|
| SCECGE [21] | 45 | 49 | 375 | 52 |
| TE-SCECGE | 52 | 65 | 625 | 69 |
| Epoxy Resin | Young’s Modulus (E) (GPa) | Tensile Strength (σ) (MPa) | Failure Strain (ε) (%) |
|---|---|---|---|
| SCECGE [21] | 0.88 ± 0.1 | 13 ± 2 | 3.1 ± 0.4 |
| TE-SCECGE | 1.24 ± 0.3 | 24 ± 7 | 4.0 ± 1.2 |
| Functionality | SCECGE Epoxies | TE-SCECGE Epoxies | SCECGE Cured Epoxies | TE-SCECGE Cured Epoxies |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 8.4 | 8.3 | 33.0 | 29.8 |
| 2 | 44.5 | 41.7 | 47.9 | 42.8 |
| 3 | 42.5 | 30.4 | 17.5 | 20.4 |
| 4 | 4.5 | 19.6 | 1.6 | 7.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinaci, E.; Can, E.; Scala, J.J.L.; Palmese, G.R. Epoxidation of Cardanol’s Terminal Double Bond. Polymers 2020, 12, 2104. https://doi.org/10.3390/polym12092104
Kinaci E, Can E, Scala JJL, Palmese GR. Epoxidation of Cardanol’s Terminal Double Bond. Polymers. 2020; 12(9):2104. https://doi.org/10.3390/polym12092104
Chicago/Turabian StyleKinaci, Emre, Erde Can, John J. La Scala, and Giuseppe R. Palmese. 2020. "Epoxidation of Cardanol’s Terminal Double Bond" Polymers 12, no. 9: 2104. https://doi.org/10.3390/polym12092104
APA StyleKinaci, E., Can, E., Scala, J. J. L., & Palmese, G. R. (2020). Epoxidation of Cardanol’s Terminal Double Bond. Polymers, 12(9), 2104. https://doi.org/10.3390/polym12092104






