Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability
Abstract
1. Introduction
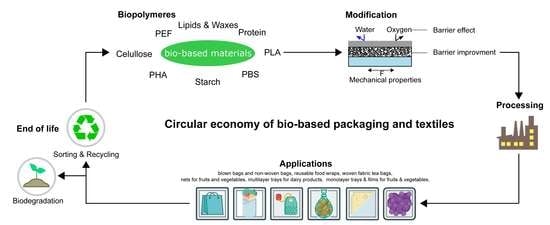
2. Bio-Based Materials for Packaging Applications
2.1. Synthetically Produced Bio-Polyesters
2.1.1. Polylactic Acid (PLA)
2.1.2. Polyethylene Furanoate (PEF)
2.1.3. Polybutylene Succinate (PBS)
2.2. Non-Synthetically Produced Polymers
2.2.1. Polyhydroxyalkanoate (PHA)
2.2.2. Cellulose and Derivatives
2.2.3. Starch
2.2.4. Proteins
2.2.5. Lipids and Waxes
3. Materials Modification, Treatment, and Processing
3.1. Biocomposites, Fillers, and Additives
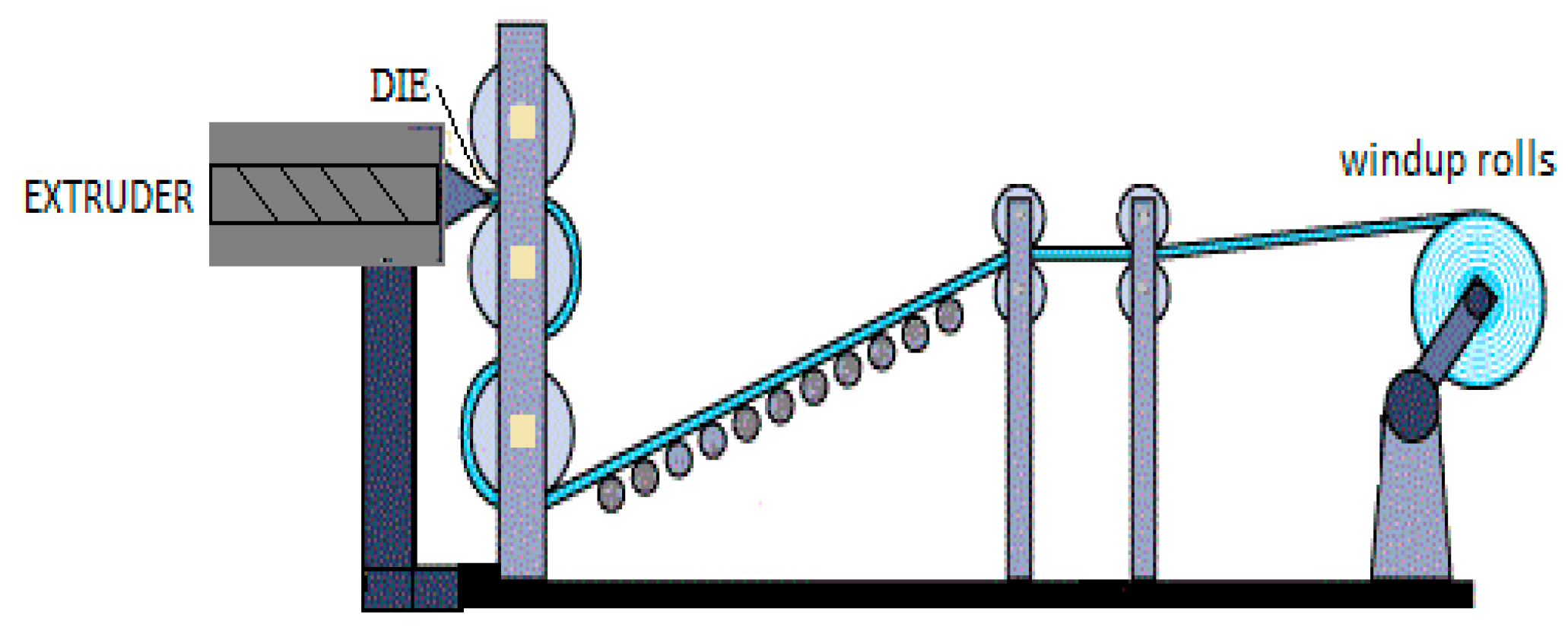
3.2. Processing of Bio-Based Materials for Packaging
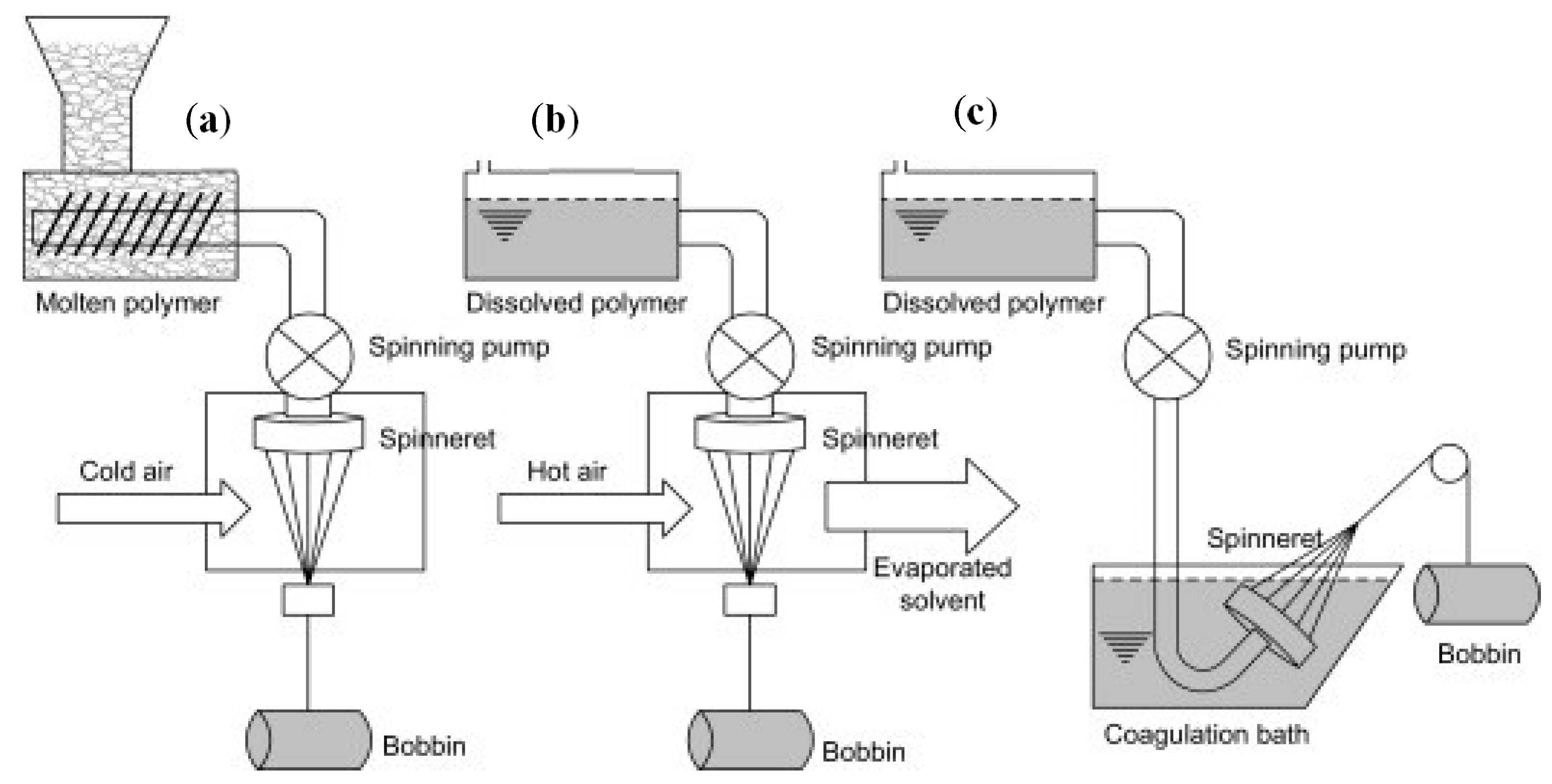
3.3. Textile Production Techniques and Processing
4. Industrial Applications
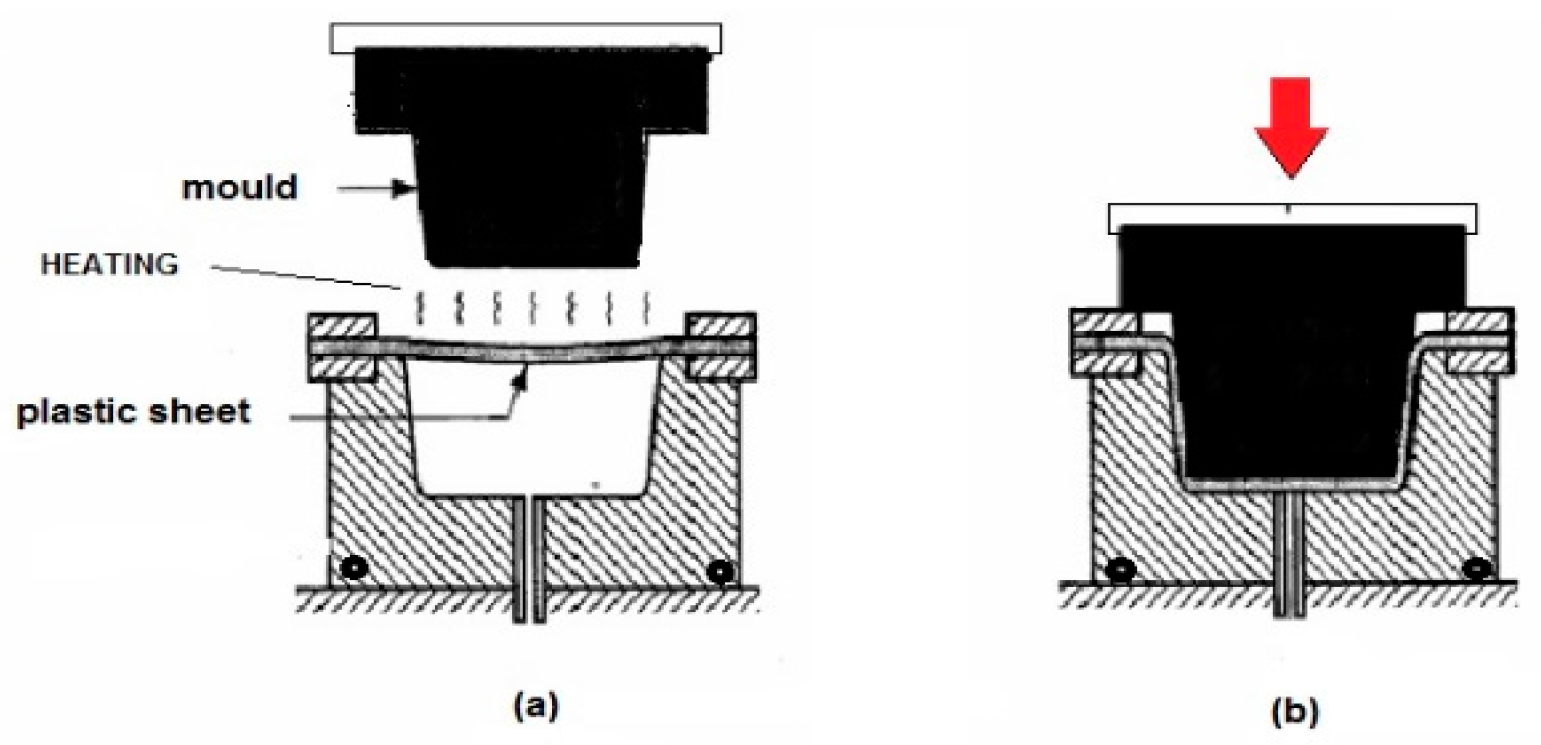
4.1. Bio-Based Films and Trays
4.2. Bio-Based Pouches and Bags
4.3. Bio-Based Textiles and Nets
5. Sustainability, Biodegradation, and Recycling
5.1. Sustainability
5.2. Biodegradable Materials
5.3. Recyclable Materials
6. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Coltelli, M.-B.; Gigante, V.; Cinelli, P.; Lazzeri, A. Chapter 15 Flexible Food Packaging Using Polymers from Biomass. In Bionanotechnology to Save the Environment; Plant and Fishery’s Biomass as Alternative to Petrol; Morganti, P., Ed.; MDPI: Basel, Switzerland, 2018; pp. 272–296. [Google Scholar]
- Álvarez-Chávez, C.R.; Edwards, S.; Moure-Eraso, R.; Geiser, K. Sustainability of bio-based plastics: General comparative analysis and recommendations for improvement. J. Clean. Prod. 2012, 23, 47–56. [Google Scholar] [CrossRef]
- Prashanth, K.H.; Tharanathan, R. Chitin/Chitosan: Modifications and their unlimited application potential—An overview. Trends Food Sci. Technol. 2007, 18, 117–131. [Google Scholar] [CrossRef]
- European Bioplastics. Bioplastics Market Data. Available online: https://www.european-bioplastics.org/ (accessed on 10 March 2020).
- Bajpai, P. Biobased Polymers. Properties and Applications in Packaging; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 0128184043. [Google Scholar]
- Amcor. 2018 Sustainability Review. Available online: https://www.amcor.com/sustainability/reports (accessed on 11 March 2020).
- Geueke, B.; Groh, K.; Muncke, J. Food packaging in the circular economy: Overview of chemical safety aspects for commonly used materials. J. Clean. Prod. 2018, 193, 491–505. [Google Scholar] [CrossRef]
- Kaiser, K.M.A.; Schmid, M.; Schlummer, M. Recycling of Polymer-Based Multilayer Packaging: A Review. Recycling 2017, 3, 1. [Google Scholar] [CrossRef]
- Thielen, M. Bioplastics: Basics, Applications, Markets; Polymedia Publisher: Mönchengladbach, Germany, 2012. [Google Scholar]
- Spierling, S.; Knüpffer, E.; Behnsen, H.; Mudersbach, M.; Krieg, H.; Springer, S.; Albrecht, S.; Herrmann, C.; Endres, H.-J. Bio-based plastics—A review of environmental, social and economic impact assessments. J. Clean. Prod. 2018, 185, 476–491. [Google Scholar] [CrossRef]
- Lee, R.; Lavoie, J.-M. From first- to third-generation biofuels: Challenges of producing a commodity from a biomass of increasing complexity. Anim. Front. 2013, 3, 6–11. [Google Scholar] [CrossRef]
- Bugnicourt, E.; Schmid, M.; Nerney, O.M.; Wildner, J.; Smykala, L.; Lazzeri, C.; Cinelli, P. Processing and Validation of Whey-Protein-Coated Films and Laminates at Semi-Industrial Scale as Novel Recyclable Food Packaging Materials with Excellent Barrier Properties. Adv. Mater. Sci. Eng. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Cinelli, P.; Schmid, M.; Bugnicourt, E.; Wildner, J.; Bazzichi, A.; Anguillesi, I.; Lazzeri, C. Whey protein layer applied on biodegradable packaging film to improve barrier properties while maintaining biodegradability. Polym. Degrad. Stab. 2014, 108, 151–157. [Google Scholar] [CrossRef]
- Schmid, M.; Benz, A.; Stinga, C.; Samain, D.; Zeyer, K.P. Fundamental Investigations Regarding Barrier Properties of Grafted PVOH Layers. Int. J. Polym. Sci. 2012, 2012, 1–6. [Google Scholar] [CrossRef]
- Schmid, M.; Dallmann, K.; Bugnicourt, E.; Cordoni, D.; Wild, F.; Lazzeri, C.; Noller, K. Properties of Whey-Protein-Coated Films and Laminates as Novel Recyclable Food Packaging Materials with Excellent Barrier Properties. Int. J. Polym. Sci. 2012, 1–7. [Google Scholar] [CrossRef]
- Zink, J.; Wyrobnik, T.; Prinz, T.; Schmid, M. Physical, Chemical and Biochemical Modifications of Protein-Based Films and Coatings: An Extensive Review. Int. J. Mol. Sci. 2016, 17, 1376. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.; Müller, K. Chapter 11—Whey Protein-Based Packaging Films and Coatings; Deeth, H.C., Bansal, N., Eds.; Whey Proteins, Academic Press: New York, NY, USA, 2019; pp. 407–437. [Google Scholar]
- Hong, S.-I.; Krochta, J. Oxygen barrier performance of whey-protein-coated plastic films as affected by temperature, relative humidity, base film, and protein type. J. Food Eng. 2006, 77, 739–745. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, S.; Gunasekaran, S. Preparation and Characterization of Whey Protein Film Incorporated with TiO2 Nanoparticles. J. Food Sci. 2009, 74, N50–N56. [Google Scholar] [CrossRef] [PubMed]
- Ustunol, Z.; Mert, B. Water Solubility, Mechanical, Barrier, and Thermal Properties of Cross-linked Whey Protein Isolate-based Films. J. Food Sci. 2006, 69, FEP129–FEP133. [Google Scholar] [CrossRef]
- Samain, D. Method for Treating a Solid Material to Make It Hydrophobic, Material Obtained and Uses. CA2301073A1; US6342268B1, 29 January 2002. [Google Scholar]
- Chang, Y.; Joo, E.; Song, H.-G.; Choi, I.; Yoon, C.S.; Choi, Y.J.; Han, J. Development of Protein-Based High-Oxygen Barrier Films Using an Industrial Manufacturing Facility. J. Food Sci. 2019, 84, 303–310. [Google Scholar] [CrossRef]
- Hu, B. Biopolymer-Based Lightweight Materials for Packaging Applications. ACS Symp. Series 2014, 1175, 239–255. [Google Scholar] [CrossRef]
- Terzopoulou, Z.; Tsanaktsis, V.; Bikiaris, D.N.; Exarhopoulos, S.; Papageorgiou, D. Biobased poly(ethylene furanoate-co-ethylene succinate) copolyesters: Solid state structure, melting point depression and biodegradability. RSC Adv. 2016, 6, 84003–84015. [Google Scholar] [CrossRef]
- Camebridge Consultants. PHA: Plastic the Way Nature Intendend? Available online: https://www.cambridgeconsultants.com/insights/bioplastics-pha-whitepaper (accessed on 10 March 2020).
- Stloukal, P.; Pekařová, S.; Kalendova, A.; Mattausch, H.; Laske, S.; Holzer, C.; Chitu, L.; Bodner, S.; Maier, G.; Slouf, M.; et al. Kinetics and mechanism of the biodegradation of PLA/clay nanocomposites during thermophilic phase of composting process. Waste Manag. 2015, 42, 31–40. [Google Scholar] [CrossRef]
- Padamati, R.B.; O’Connor, K.E.; Ramakrishna, S. Current progress on bio-based polymers and their future trends. Prog. Biomater. 2013, 2, 8. [Google Scholar] [CrossRef]
- Blackburn, R.S. Sustainable Textiles. Life Cycle and Environmental Impact; Woodhead: Cambridge, UK, 2009; ISBN 9781845694. [Google Scholar]
- IfBB Institute for Bioplastics and Biocomposites. Biopolymers Facts and Statistics: Production Capacities, Processing Routes, Feedstock, Land and Water Use (ISSN (Online) 2510-3431); IfBB – Institute for Bioplastics and Biocomposites Hochschule Hannover: Hannover, Germany, 2019. [Google Scholar]
- Singhvi, M.; Gokhale, D. Biomass to biodegradable polymer (PLA). RSC Adv. 2013, 3, 13558. [Google Scholar] [CrossRef]
- Hanstveit, A.O. Biodegradability of petroleum waxes and beeswax in an adapted CO2 evolution test. Chemosphere 1992, 25, 605–620. [Google Scholar] [CrossRef]
- Poly(lactic acid). Synthesis, Structures, Properties, Processing, and Applications; Auras, R., Lim, L.-T., Selke, S.E.M., Tsuji, H., Eds.; Wiley: Hoboken, NJ, USA, 2010; ISBN 978-0-470-29366-9. [Google Scholar]
- Garlotta, D. A Literature Review of Poly(Lactic Acid). J. Polym. Environ. 2001, 9, 63–84. [Google Scholar] [CrossRef]
- Perinelli, D.R.; Cespi, M.; Bonacucina, G.; Palmieri, G.F. PEGylated polylactide (PLA) and poly(lactic-co-glycolic acid) (PLGA) copolymers for the design of drug delivery systems. J. Pharm. Investig. 2019, 49, 443–458. [Google Scholar] [CrossRef]
- Eerhart, A.J.J.E.; Faaij, A.P.C.; Patel, M.K. Replacing fossil-based PET with biobased PEF; Process analysis, energy and GHG balance. Energy Environ. Sci. 2012, 5, 6407. [Google Scholar] [CrossRef]
- Avantium. Internal Results YXY Technologies. Available online: https://www.avantium.com/technologies/yxy/ (accessed on 2 July 2020).
- Xu, J.; Guo, B.-H. Microbial Succinic Acid, Its Polymer Poly(butylene succinate), and Applications. In Prokaryotic Symbionts in Plants; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2009; Volume 14, pp. 347–388. [Google Scholar]
- Guettler, M.V.; Jain, M.K.; Rumler, D. Method for Making Succinic Acid, Bacterial Variants for Use in Process and Methods for Obtaining Variants. U.S. Patent 5,573,931A, 1995. [Google Scholar]
- Jompang, L.; Thumsorn, S.; On, J.W.; Surin, P.; Apawet, C.; Chaichalermwong, T.; Kaabbuathong, N.; O-Charoen, N.; Srisawat, N. Poly(Lactic Acid) and Poly(Butylene Succinate) Blend Fibers Prepared by Melt Spinning Technique. Energy Proc. 2013, 34, 493–499. [Google Scholar] [CrossRef]
- Seggiani, M.; Gigante, V.; Cinelli, P.; Coltelli, M.-B.; Sandroni, M.; Anguillesi, I.; Lazzeri, A. Processing and mechanical performances of Poly(Butylene Succinate–co–Adipate) (PBSA) and raw hydrolyzed collagen (HC) thermoplastic blends. Polym. Test. 2019, 77, 105900. [Google Scholar] [CrossRef]
- Rameshkumar, S.; Shaiju, P.; O’Connor, K.E.; Padamati, R.B. Bio-based and biodegradable polymers—State-of-the-art, challenges, and emerging trends. Curr. Opin. Green Sustain. Chem. 2020, 21, 75–81. [Google Scholar] [CrossRef]
- Sabapathy, P.C.; Devaraj, S.; Meixner, K.; Anburajan, P.; Kathirvel, P.; Ravikumar, Y.; Zabed, H.M.; Qi, X. Recent developments in Polyhydroxyalkanoates (PHAs) production—A review. Bioresour. Technol. 2020, 306, 123132. [Google Scholar] [CrossRef] [PubMed]
- Bugnicourt, E. Polyhydroxyalkanoate (PHA): Review of synthesis, characteristics, processing, and potential applications in packaging. Express Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Dilkes-Hoffman, L.S.; Lant, P.A.; Laycock, B.; Pratt, S. The rate of biodegradation of PHA bioplastics in the marine environment: A meta-study. Mar. Pollut. Bull. 2019, 142, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.K.; Ha, S.K.; Verma, K.; Tiwari, S.K.; Kumar, R. Recent progress in selected bio-nanomaterials and their engineering applications: An overview. J. Sci. Adv. Mater. Devices 2018, 3, 263–288. [Google Scholar] [CrossRef]
- Pandey, J.K.; Kumar, A.P.; Misra, M.; Mohanty, A.K.; Drzal, L.T.; Singh, R.P. Recent advances in biodegradable nanocomposites. J. Nanosci. Nanotechnol. 2005, 5, 497–526. [Google Scholar] [CrossRef] [PubMed]
- Endres, H.-J.; Siebert-Raths, A. Engineering Biopolymers. Markets, Manufacturing, Properties, and Applications; Hanser Publishers: Munich, Germany, 2011; ISBN 9781569904619. [Google Scholar]
- Johansson, C.; Bras, J.; Mondragon, I.; Nechita, P.; Plackett, D.; Simon, P.; Svetec, D.G.; Virtanen, S.; Baschetti, M.G.; Breen, C.; et al. Renewable fibers and bio-based materials for packaging applications-a review of recent developments. Bioresources 2012, 7, 2506–2552. [Google Scholar] [CrossRef]
- Lawton, J.; Fanta, G. Glycerol-plasticized films prepared from starch—poly(vinyl alcohol) mixtures: Effect of poly(ethylene-co-acrylic acid). Carbohydr. Polym. 1994, 23, 275–280. [Google Scholar] [CrossRef]
- Imam, S.H.; Gordon, S.H.; Shogren, R.L.; Tosteson, T.R.; Govind, N.S.; Greene, R.V. Degradation of Starch–Poly(β-Hydroxybutyrate-Co-β-Hydroxyvalerate) Bioplastic in Tropical Coastal Waters. Appl. Environ. Microbiol. 1999, 65, 431–437. [Google Scholar] [CrossRef]
- Bertolini, A. Starches. Characterization, Properties, and Applications; CRC: Boca Raton, FL, USA, 2010; ISBN 9781420080230. [Google Scholar]
- Callegarin, F.; Gallo, J.-A.Q.; Debeaufort, F.; Voilley, A. Lipids and biopackaging. J. Am. Oil Chem. Soc. 1997, 74, 1183–1192. [Google Scholar] [CrossRef]
- Dhall, R.K. Advances in Edible Coatings for Fresh Fruits and Vegetables: A Review. Crit. Rev. Food Sci. Nutr. 2013, 53, 435–450. [Google Scholar] [CrossRef] [PubMed]
- Castro-Aguirre, E.; Iñiguez-Franco, F.; Samsudin, H.; Fang, X.; Auras, R. Poly(lactic acid)—Mass production, processing, industrial applications, and end of life. Adv. Drug Deliv. Rev. 2016, 107, 333–366. [Google Scholar] [CrossRef]
- Mehta, R.; Kumar, V.; Bhunia, H.; Upadhyay, S.N. Synthesis of Poly(Lactic Acid): A Review. J. Macromol. Sci. Part C 2005, 45, 325–349. [Google Scholar] [CrossRef]
- Ren, J. Biodegradable Poly(Lactic Acid): Synthesis, Modification, Processing and Applications; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Li, B.; Chen, S.-C.; Qiu, Z.-C.; Yang, K.-K.; Tang, S.; Yu, W.-J.; Wang, Y. Synthesis of poly(lactic acid-b-p-dioxanone) block copolymers from ring opening polymerization of p-dioxanone by poly(L-lactic acid) macroinitiators. Polym. Bull. 2008, 61, 139–146. [Google Scholar] [CrossRef]
- Ovitt, T.M.; Coates, G.W. Stereoselective ring-opening polymerization of rac-lactide with a single-site, racemic aluminum alkoxide catalyst: Synthesis of stereoblock poly(lactic acid). J. Polym. Sci. Part A Polym. Chem. 2000, 38, 4686–4692. [Google Scholar] [CrossRef]
- Radano, C.P.; Baker, G.L.; Smith, M.R. Stereoselective Polymerization of a Racemic Monomer with a Racemic Catalyst: Direct Preparation of the Polylactic Acid Stereocomplex from Racemic Lactide. J. Am. Chem. Soc. 2000, 122, 1552–1553. [Google Scholar] [CrossRef]
- NatureWorks. Where Ingeo Comes From. Available online: https://www.natureworksllc.com/ (accessed on 20 December 2019).
- Raquez, J.-M.; Ramy-Ratiarison, R.; Murariu, M.; Dubois, P. CHAPTER 4. Reactive Extrusion of PLA-based Materials: From Synthesis to Reactive Melt-blending. Polymer Chem. Series 2014, 99–123. [Google Scholar] [CrossRef]
- Dugan, J.S. Novel Properties of PLA Fibers. Int. Nonwovens J. 2001, 29–33. [Google Scholar] [CrossRef]
- Fazita, M.N.; Jayaraman, K.; Bhattacharyya, D.; Haafiz, M.M.; Saurabh, C.; Hussin, M.H.; Khalil, H.A. Green Composites Made of Bamboo Fabric and Poly(Lactic) Acid for Packaging Applications—A Review. Materials 2016, 9, 435. [Google Scholar] [CrossRef] [PubMed]
- Maharana, T.; Mohanty, B.; Negi, Y.S. Melt–solid polycondensation of lactic acid and its biodegradability. Prog. Polym. Sci. 2009, 34, 99–124. [Google Scholar] [CrossRef]
- Lyu, S.; Schley, J.; Loy, B.; Lind, D.; Hobot, C.; Sparer, R.; Untereker, D. Kinetics and Time−Temperature Equivalence of Polymer Degradation. Biomacromolecules 2007, 8, 2301–2310. [Google Scholar] [CrossRef] [PubMed]
- El Sawy, M.; Kim, K.-H.; Park, J.-W.; Deep, A. Hydrolytic degradation of polylactic acid (PLA) and its composites. Renew. Sustain. Energy Rev. 2017, 79, 1346–1352. [Google Scholar] [CrossRef]
- Husarova, L.; Pekařová, S.; Stloukal, P.; Kucharzcyk, P.; Verney, V.; Commereuc, S.; Ramoné, A.; Koutný, M. Identification of important abiotic and biotic factors in the biodegradation of poly(l-lactic acid). Int. J. Boil. Macromol. 2014, 71, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Mueller, R.-J. Biological degradation of synthetic polyesters—Enzymes as potential catalysts for polyester recycling. Process. Biochem. 2006, 41, 2124–2128. [Google Scholar] [CrossRef]
- Kijchavengkul, T.; Auras, R.; Rubino, M.; Ngouajio, M.; Fernandez, R.T. Assessment of aliphatic–aromatic copolyester biodegradable mulch films. Part II: Laboratory simulated conditions. Chemosphere 2008, 71, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Gorrasi, G.; Pantani, R. Effect of PLA grades and morphologies on hydrolytic degradation at composting temperature: Assessment of structural modification and kinetic parameters. Polym. Degrad. Stab. 2013, 98, 1006–1014. [Google Scholar] [CrossRef]
- Ghorpade, V.M.; Gennadios, A.; Hanna, M.A. Laboratory composting of extruded poly(lactic acid) sheets. Bioresour. Technol. 2001, 76, 57–61. [Google Scholar] [CrossRef]
- Marty, A.; Duquesne, S.; Guicherd, M.; Gueroult, M.; Andre, I. Novel Proteases and Uses Therof. WO2019122308, 27 June 2019. [Google Scholar]
- Tokiwa, Y.; Jarerat, A. Biodegradation of poly(L-lactide). Biotechnol. Lett. 2004, 26, 771–777. [Google Scholar] [CrossRef]
- Blasi, P. Poly(lactic acid)/poly(lactic-co-glycolic acid)-based microparticles: An overview. J. Pharm. Investig. 2019, 49, 337–346. [Google Scholar] [CrossRef]
- Sharma, D.; Lipp, L.; Arora, S.; Singh, J. Diblock and triblock copolymers of polylactide and polyglycolide. Mater. Biomed.Eng. 2019, 449–477. [Google Scholar] [CrossRef]
- Koosomsuan, W.; Yamaguchi, M.; Phinyocheep, P.; Sirisinha, K. High-Strain Shape Memory Behavior of PLA-PEG Multiblock Copolymers and Its Microstructural Origin. J. Polym. Sci. Part B Polym. Phys. 2018, 57, 241–256. [Google Scholar] [CrossRef]
- Liu, X.; Corciulo, C.; Arabagian, S.; Ulman, A.; Cronstein, B. Adenosine-Functionalized Biodegradable PLA-b-PEG Nanoparticles Ameliorate Osteoarthritis in Rats. Sci. Rep. 2019, 9, 7430. [Google Scholar] [CrossRef]
- Lee, G.S.; Moon, B.R.; Jeong, H.; Shin, J.; Kim, J.G. Mechanochemical synthesis of poly(lactic acid) block copolymers: Overcoming the miscibility of the macroinitiator, monomer and catalyst under solvent-free conditions. Polym. Chem. 2019, 10, 539–545. [Google Scholar] [CrossRef]
- Werpy, T.; Petersen, G. Top Value-Added Chemicals from Biomass: Volume I—Results of Screening for Potential Candidates from Sugars and Synthesis Gas (technical report); Office of Scientific and Technical Information (OSTI):National Renewable Energy Lab.: Golden, CO, USA, 2004.
- Thessrimuang, N.; Prachayawarakorn, J. Characterization and Properties of High Amylose Mung Bean Starch Biodegradable Films Cross-linked with Malic Acid or Succinic Acid. J. Polym. Environ. 2018, 27, 234–244. [Google Scholar] [CrossRef]
- Cheng, C.; Zhou, Y.; Lin, M.; Wei, P.; Yang, S.-T. Polymalic acid fermentation by Aureobasidium pullulans for malic acid production from soybean hull and soy molasses: Fermentation kinetics and economic analysis. Bioresour. Technol. 2017, 223, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Iyyappan, J.; Bharathiraja, B.; Baskar, G.; Kamalanaban, E. Process optimization and kinetic analysis of malic acid production from crude glycerol using Aspergillus niger. Bioresour. Technol. 2019, 281, 18–25. [Google Scholar] [CrossRef]
- Gazzotti, S.; Todisco, S.A.; Picozzi, C.; Ortenzi, M.A.; Farina, H.; Lesma, G.; Silvani, A. Eugenol-grafted aliphatic polyesters: Towards inherently antimicrobial PLA-based materials exploiting OCA’s chemistry. Eur. Polym. J. 2019, 114, 369–379. [Google Scholar] [CrossRef]
- Zhang, Y.; Ni, C.; Shi, G.; Wang, J.; Zhang, M.; Li, W. The polyion complex nano-prodrug of doxorubicin (DOX) with poly(lactic acid-co-malic acid)-block-polyethylene glycol: Preparation and drug-controlled release. Med. Chem. Res. 2014, 24, 1189–1195. [Google Scholar] [CrossRef]
- Oyama, H.T.; Tanishima, D.; Maekawa, S. Poly(malic acid-co-L-lactide) as a superb degradation accelerator for Poly(l-lactic acid) at physiological conditions. Polym. Degrad. Stab. 2016, 134, 265–271. [Google Scholar] [CrossRef]
- Ferone, M.; Raganati, F.; Olivieri, G.; Marzocchella, A. Bioreactors for succinic acid production processes. Crit. Rev. Biotechnol. 2019, 39, 571–586. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Kopitzky, R.; Tolga, S.; Kabasci, S. Polylactide (PLA) and Its Blends with Poly(butylene succinate) (PBS): A Brief Review. Polymer 2019, 11, 1193. [Google Scholar] [CrossRef]
- Cadar, O.; Paul, M.; Roman, C.; Miclean, M.; Majdik, C. Biodegradation behaviour of poly(lactic acid) and (lactic acid-ethylene glycol-malonic or succinic acid) copolymers under controlled composting conditions in a laboratory test system. Polym. Degrad. Stab. 2012, 97, 354–357. [Google Scholar] [CrossRef]
- Robert, T.; Friebel, S. Itaconic acid—A versatile building block for renewable polyesters with enhanced functionality. Green Chem. 2016, 18, 2922–2934. [Google Scholar] [CrossRef]
- Sood, S.; Gupta, V.K.; Agarwal, S.; Dev, K.; Pathania, D. Controlled release of antibiotic amoxicillin drug using carboxymethyl cellulose-cl-poly(lactic acid-co-itaconic acid) hydrogel. Int. J. Boil. Macromol. 2017, 101, 612–620. [Google Scholar] [CrossRef]
- Gupta, V.K.; Sood, S.; Agarwal, S.; Saini, R.V.; Pathania, D. Antioxidant activity and controlled drug delivery potential of tragacanth gum-cl- poly(lactic acid-co-itaconic acid) hydrogel. Int. J. Boil. Macromol. 2018, 107, 2534–2543. [Google Scholar] [CrossRef] [PubMed]
- Aliotta, L.; Cinelli, P.; Coltelli, M.B.; Righetti, M.C.; Gazzano, M.; Lazzeri, C. Effect of nucleating agents on crystallinity and properties of poly(lactic acid) (PLA). Eur. Polym. J. 2017, 93, 822–832. [Google Scholar] [CrossRef]
- Castiello, S.; Coltelli, M.-B.; Bronco, S.; Conzatti, L. Comparative study about preparation of poly(lactide)/Organophilic montmorillonites nanocomposites through melt blending or ring opening polymerization methods. J. Appl. Polym. Sci. 2012, 125, 413. [Google Scholar] [CrossRef]
- Scatto, M.; Salmini, E.; Castiello, S.; Coltelli, M.-B.; Conzatti, L.; Stagnaro, P.; Andreotti, L.; Bronco, S. Plasticized and nanofilled poly(lactic acid)-based cast films: Effect of plasticizer and organoclay on processability and final properties. J. Appl. Polym. Sci. 2012, 127, 4947–4956. [Google Scholar] [CrossRef]
- Shogren, R. Water vapor permeability of biodegradable polymers. J. Polym. Environ. 1997, 5, 91–95. [Google Scholar] [CrossRef]
- Singh, A.A.; Sharma, S.; Srivastava, M.; Majumdar, A. Modulating the properties of polylactic acid for packaging applications using biobased plasticizers and naturally obtained fillers. Int. J. Boil. Macromol. 2020, 153, 1165–1175. [Google Scholar] [CrossRef]
- Jem, K.J.; Tan, B. The development and challenges of poly(lactic acid) and poly(glycolic acid). Adv. Ind. Eng. Polym. Res. 2020, 3, 60–70. [Google Scholar] [CrossRef]
- Burgess, S.K.; Karvan, O.; Johnson, J.; Kriegel, R.M.; Koros, W.J. Oxygen sorption and transport in amorphous poly(ethylene furanoate). Polymer 2014, 55, 4748–4756. [Google Scholar] [CrossRef]
- Poulopoulou, N.; Kasmi, N.; Siampani, M.; Terzopoulou, Z.N.; Bikiaris, D.N.; Achilias, D.S.; Papageorgiou, D.G.; Papageorgiou, G.Z. Exploring Next-Generation Engineering Bioplastics: Poly(alkylene furanoate)/Poly(alkylene terephthalate) (PAF/PAT) Blends. Polymers 2019, 11, 556. [Google Scholar] [CrossRef]
- Materials, E.E.P.O.F.C. Scientific Opinion on the safety assessment of the substance, furan 2, 5? Dicarboxylic acid, CAS No 3238?40? 2, for use in food contact materials. EFSA J. 2014, 12. [Google Scholar] [CrossRef]
- Chen, G.-Q. A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry. Chem. Soc. Rev. 2009, 38, 2434. [Google Scholar] [CrossRef]
- Nagarajan, V.; Andrzejewski, J.; Misra, M. Perspective on Polylactic Acid (PLA) based Sustainable Materials for Durable Applications: Focus on Toughness and Heat Resistance. ACS Sustain. Chem. Eng. 2016, 4, 2899–2916. [Google Scholar] [CrossRef]
- van den Oever, M.; Molenveld, K.; van der Zee, M.; Bos, H. Bio-Based and Biodegradable Plastics—Facts and Figures; Wageningen Food & Biobased Research number 1722: Wageningen, The Netherlands, 2017. [Google Scholar]
- Danimer Scientific. PHA Impact. Available online: https://danimerscientific.com/pha-the-future-of-biopolymers/ (accessed on 2 January 2020).
- Coltelli, M.-B.; Danti, S.; Trombi, L.; Morganti, P.; Donnarumma, G.; Baroni, A.; Fusco, A.; Lazzeri, C. Preparation of Innovative Skin Compatible Films to Release Polysaccharides for Biobased Beauty Masks. Cosmetics 2018, 5, 70. [Google Scholar] [CrossRef]
- Jost, V. Packaging related properties of commercially available biopolymers—An overview of the status quo. Express Polym. Lett. 2018, 12, 429–435. [Google Scholar] [CrossRef]
- Yang, H.-S.; Gardner, D.J.; Nader, J.W. Dispersion evaluation of microcrystalline cellulose/cellulose nanofibril-filled polypropylene composites using thermogravimetric analysis. J. Therm. Anal. Calorim. 2010, 103, 1007–1015. [Google Scholar] [CrossRef]
- Chen, J. Chapter 4—Synthetic Textile Fibers: Regenerated Cellulose Fibers. In Textiles and Fashion: Woodhead Publishing Series in Textiles; Sinclair, R., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 79–95. ISBN 978-1-84569-931-4. [Google Scholar]
- Weller, I.; Lange, H. Nachhaltigkeit. Soziologische Rev. 2007, 30, 393–402. [Google Scholar] [CrossRef]
- Calva-Estrada, S.J.; Jiménez-Fernández, M.; Lugo-Cervantes, E. Protein-Based Films: Advances in the Development of Biomaterials Applicable to Food Packaging. Food Eng. Rev. 2019, 11, 78–92. [Google Scholar] [CrossRef]
- Wheylayer 2. Available online: http://www.wheylayer.eu/de/ (accessed on 10 November 2019).
- ThermoWhey. Available online: http://thermowhey.eu/ (accessed on 10 November 2019).
- Stäbler, A.; Schmid, M. Thermoformbarer Mehrschichtverbund Sowie Proteinbasierte Formulierung zum Erhalt Einer Thermoformbaren Schicht mit Sauerstoffbarriere im Verbund. Patent application 102016108214.7; Germany, 3 May 2016. [Google Scholar]
- Bugnicourt, E.; Noller, K.; Schmid, M.; Wild, F. Whey Protein Coated Films; 2011. WO002013014493A1, 31 January 2013. [Google Scholar]
- Corso, J.F. Bone-Conduction Thresholds for Sonic and Ultrasonic Frequencies. J. Acoust. Soc. Am. 1963, 35, 1738. [Google Scholar] [CrossRef]
- Kadam, D.M.; Thunga, M.; Wang, S.; Kessler, M.; Grewell, D.; Lamsal, B.P.; Yu, C. Preparation and characterization of whey protein isolate films reinforced with porous silica coated titania nanoparticles. J. Food Eng. 2013, 117, 133–140. [Google Scholar] [CrossRef]
- Fachin, L.; Viotto, W.H. Effect of pH and heat treatment of cheese whey on solubility and emulsifying properties of whey protein concentrate produced by ultrafiltration. Int. Dairy J. 2005, 15, 325–332. [Google Scholar] [CrossRef]
- Nicorescu, I.; Loisel, C.; Vial, C.; Riaublanc, A.; Djelveh, G.; Cuvelier, G.; Legrand, J. Combined effect of dynamic heat treatment and ionic strength on the properties of whey protein foams—Part II. Food Res. Int. 2008, 41, 980–988. [Google Scholar] [CrossRef]
- Nicolai, T.; Britten, M.; Schmitt, C. β-Lactoglobulin and WPI aggregates: Formation, structure, and applications. Food Hydrocoll. 2011, 25, 1945–1962. [Google Scholar] [CrossRef]
- Jambrak, A.R.; Lelas, V.; Mason, T.J.; Krešić, G.; Badanjak, M. Physical properties of ultrasound treated soy proteins. J. Food Eng. 2009, 93, 386–393. [Google Scholar] [CrossRef]
- Weng, W.; Zheng, H. Effect of transglutaminase on properties of tilapia scale gelatin films incorporated with soy protein isolate. Food Chem. 2015, 169, 255–260. [Google Scholar] [CrossRef] [PubMed]
- di Pierro, P.; Chico, B.; Villalonga, R.; Mariniello, L.; Damiao, A.E.; Masi, P.; Porta, R. Chitosan−Whey Protein Edible Films Produced in the Absence or Presence of Transglutaminase: Analysis of Their Mechanical and Barrier Properties. Biomacromolecules 2006, 7, 744–749. [Google Scholar] [CrossRef]
- Hernàndez-Balada, E.; Taylor, M.M.; Phillips, J.G.; Marmer, W.N.; Brown, E.M. Properties of biopolymers produced by transglutaminase treatment of whey protein isolate and gelatin. Bioresour. Technol. 2009, 100, 3638–3643. [Google Scholar] [CrossRef]
- Banerjee, R.; Chen, H. Functional Properties of Edible Films Using Whey Protein Concentrate. J. Dairy Sci. 1995, 78, 1673–1683. [Google Scholar] [CrossRef]
- Dangaran, K.; Krochta, J. Whey Protein Films and Coatings (chapter 6); Whey Processing, Functionality and Health Benefits, 1st ed.; Onwulata, C., Huth, P.J., Eds.; Wiley-Blackwell: Ames, IA, USA, 2009; pp. 133–167. [Google Scholar] [CrossRef]
- Fornes, T.; Paul, D.R. Modeling properties of nylon 6/clay nanocomposites using composite theories. Polymer 2003, 44, 4993–5013. [Google Scholar] [CrossRef]
- Zeng, Q.; Yu, A.; Lu, G.; Paul, D.R. Clay-Based Polymer Nanocomposites: Research and Commercial Development. J. Nanosci. Nanotechnol. 2005, 5, 1574–1592. [Google Scholar] [CrossRef]
- Kumar, P.; Sandeep, K.; Alavi, S.; Truong, V.; Gorga, R. Effect of Type and Content of Modified Montmorillonite on the Structure and Properties of Bio-Nanocomposite Films Based on Soy Protein Isolate and Montmorillonite. J. Food Sci. 2010, 75, N46–N56. [Google Scholar] [CrossRef]
- Kumar, P.; Sandeep, K.; Alavi, S.; Truong, V.-D.; Gorga, R. Preparation and characterization of bio-nanocomposite films based on soy protein isolate and montmorillonite using melt extrusion. J. Food Eng. 2010, 100, 480–489. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, Y.; Liu, F.; Ren, F.; Zhao, G.; Leng, X. Fabrication and characterization of TiO2/whey protein isolate nanocomposite film. Food Hydrocoll. 2011, 25, 1098–1104. [Google Scholar] [CrossRef]
- Pérez-Gago, M.B.; Krochta, J.M. Lipid particle size effect on water vapor permeability and mechanical properties of whey protein/beeswax emulsion films. J. Agric. Food Chem. 2001, 49, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Simelane, S.; Ustunol, Z. Mechanical Properties of Heat? Cured Whey Protein? Based Edible Films Compared with Collagen Casings under Sausage Manufacturing Conditions. J. Food Sci. 2005, 70, 131. [Google Scholar] [CrossRef]
- Rhim, J.W.; Gennadios, A.; Fu, D.; Weller, C.L.; Hanna, M.A. Properties of Ultraviolet Irradiated Protein Films. LWT 1999, 32, 129–133. [Google Scholar] [CrossRef]
- Gennadios, A.; Rhim, J.; Handa, A.; Weller, C.; Hanna, M. Ultraviolet Radiation Affects Physical and Molecular Properties of Soy Protein Films. J. Food Sci. 2008, 63, 225–228. [Google Scholar] [CrossRef]
- Morillon, V.; Debeaufort, F.; Blond, G.; Capelle, M.; Voilley, A. Factors Affecting the Moisture Permeability of Lipid-Based Edible Films: A Review. Crit. Rev. Food Sci. Nutr. 2002, 42, 67–89. [Google Scholar] [CrossRef]
- Ashok, A.; Rejeesh, C.R.; Renjith, R. Biodegradable Polymers for Sustainable Packaging Applications: A Review. Int. J. Bionics Biomater. 2016, 1–11. [Google Scholar]
- Bravin, B.; Peressini, D.; Sensidoni, A. Development and application of polysaccharide–lipid edible coating to extend shelf-life of dry bakery products. J. Food Eng. 2006, 76, 280–290. [Google Scholar] [CrossRef]
- Shellhammer, T.; Krochta, J. Whey Protein Emulsion Film Performance as Affected by Lipid Type and Amount. J. Food Sci. 1997, 62, 390–394. [Google Scholar] [CrossRef]
- European Bioplastics. What Are Bioplasctics? Available online: https://www.european-bioplastics.org/bioplastics/ (accessed on 7 May 2020).
- European Commission. Sustainable Development. Available online: https://ec.europa.eu/trade/policy/policy-making/sustainable-development/ (accessed on 6 May 2020).
- Wayan, S.I.; Suriadi, I.G.A.K.; Arnis, K. Mechanical Properties of Rice Husks Fiber Reinforced Polyester Composites. Int. J. Mater. Mech. Manuf. 2014, 2, 165–168. [Google Scholar] [CrossRef]
- Schmid, M.; Herbst, C.; Müller, K.; Stäbler, A.; Schlemmer, D.; Coltelli, M.-B.; Lazzeri, C. Effect of Potato Pulp Filler on the Mechanical Properties and Water Vapor Transmission Rate of Thermoplastic WPI/PBS Blends. Polym. Technol. Eng. 2015, 55, 510–517. [Google Scholar] [CrossRef]
- Baştürka, S.B.; Kanbur, K.; Polatoğluc, I.; Yüreklid, Y. Mechanical Properties of Acorn and Pinecone Filled Polymer Composites. Am. Sci. Res. J. Eng. Technol. Sci. 2015, 144–153. [Google Scholar]
- Bledzki, A.; Gassan, J. Composites reinforced with cellulose based fibres. Prog. Polym. Sci. 1999, 24, 221–274. [Google Scholar] [CrossRef]
- Faruk, O.; Bledzki, A.K.; Fink, H.-P.; Sain, M. Biocomposites reinforced with natural fibers: 2000–2010. Prog. Polym. Sci. 2012, 37, 1552–1596. [Google Scholar] [CrossRef]
- Pandey, J.K.; Ahn, S.H.; Lee, C.S.; Mohanty, A.K.; Misra, M. Recent Advances in the Application of Natural Fiber Based Composites. Macromol. Mater. Eng. 2010, 295, 975–989. [Google Scholar] [CrossRef]
- Poulose, A.M.; Elnour, A.Y.; Anis, A.; Shaikh, H.; Al-Zahrani, S.; George, J.; Al-Wabel, M.I.; Usman, A.R.; Ok, Y.S.; Tsang, D.C.; et al. Date palm biochar-polymer composites: An investigation of electrical, mechanical, thermal and rheological characteristics. Sci. Total. Environ. 2017, 619, 311–318. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, D.; Xu, H.; Lu, W.; Ren, X.; Cai, H.; Lei, H.; Huo, E.; Zhao, Y.; Qian, M.; et al. Biochar filled high-density polyethylene composites with excellent properties: Towards maximizing the utilization of agricultural wastes. Ind. Crop. Prod. 2020, 146, 112185. [Google Scholar] [CrossRef]
- Aliotta, L.; Gigante, V.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, C.; Seggiani, M. Thermo-Mechanical Properties of PLA/Short Flax Fiber Biocomposites. Appl. Sci. 2019, 9, 3797. [Google Scholar] [CrossRef]
- Righetti, M.C.; Cinelli, P.; Mallegni, N.; Stäbler, A.; Lazzeri, C. Thermal and Mechanical Properties of Biocomposites Made of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and Potato Pulp Powder. Polymer 2019, 11, 308. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Cinelli, P.; Gigante, V.; Aliotta, L.; Morganti, P.; Panariello, L.; Lazzeri, C. Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties. Int. J. Mol. Sci. 2019, 20, 504. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Aliotta, L.; Vannozzi, A.; Morganti, P.; Panariello, L.; Danti, S.; Neri, S.; Fernandez-Avila, C.; Fusco, A.; Donnarumma, G.; et al. Properties and Skin Compatibility of Films Based on Poly(Lactic Acid) (PLA) Bionanocomposites Incorporating Chitin Nanofibrils (CN). J. Funct. Biomater. 2020, 11, 21. [Google Scholar] [CrossRef]
- Aliotta, L.; Gigante, V.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, C. Evaluation of Mechanical and Interfacial Properties of Bio-Composites Based on Poly(Lactic Acid) with Natural Cellulose Fibers. Int. J. Mol. Sci. 2019, 20, 960. [Google Scholar] [CrossRef] [PubMed]
- Aliotta, L.; Lazzeri, C. A proposal to modify the Kelly-Tyson equation to calculate the interfacial shear strength (IFSS) of composites with low aspect ratio fibers. Compos. Sci. Technol. 2020, 186, 107920. [Google Scholar] [CrossRef]
- Gigante, V.; Aliotta, L.; Phuong, V.T.; Coltelli, M.B.; Cinelli, P.; Lazzeri, C. Effects of waviness on fiber-length distribution and interfacial shear strength of natural fibers reinforced composites. Compos. Sci. Technol. 2017, 152, 129–138. [Google Scholar] [CrossRef]
- Seggiani, M.; Cinelli, P.; Verstichel, S.; Puccini, M.; Vitolo, S.; Anguillesi, I.; Lazzeri, A. Development of fibres-reinforced biodegradable composites. Chem. Eng. Trans. 2015, 43, 1813–1815. [Google Scholar]
- Cinelli, P.; Seggiani, M.; Mallegni, N.; Gigante, V.; Lazzeri, C. Processability and Degradability of PHA-Based Composites in Terrestrial Environments. Int. J. Mol. Sci. 2019, 20, 284. [Google Scholar] [CrossRef]
- Greene, J. PLA and PHA Biodegradation in the Marine Enviornment; Department of Resources Recycling and Recovery: Sacramento, CA, USA, 2012.
- Seggiani, M.; Cinelli, P.; Balestri, E.; Mallegni, N.; Stefanelli, E.; Rossi, A.; Lardicci, C.; Lazzeri, C. Novel Sustainable Composites Based on Poly(hydroxybutyrate-co-hydroxyvalerate) and Seagrass Beach-CAST Fibers: Performance and Degradability in Marine Environments. Materials 2018, 11, 772. [Google Scholar] [CrossRef]
- Chiellini, E.; Cinelli, P.; Imam, S.H.; Mao, L. Composite films based on biorelated agro-industrial waste and poly(vinyl alcohol). Preparation and mechanical properties characterization. Biomacromolecules 2001, 2, 1029–1037. [Google Scholar] [CrossRef]
- Chiellini, E.; Cinelli, P.; Chiellini, F.; Imam, S.H. Environmentally Degradable Bio-Based Polymeric Blends and Composites. Macromol. Biosci. 2004, 4, 218–231. [Google Scholar] [CrossRef]
- Righetti, M.C.; Cinelli, P.; Mallegni, N.; Massa, C.A.; Aliotta, L.; Lazzeri, C. Thermal, Mechanical, Viscoelastic and Morphological Properties of Poly(lactic acid) based Biocomposites with Potato Pulp Powder Treated with Waxes. Materials 2019, 12, 990. [Google Scholar] [CrossRef]
- Sakai, T. Screw extrusion technology—Past, present, and future. Polimery 2013, 58, 847–857. [Google Scholar] [CrossRef]
- Gigante, V. Processing and Characterization of PLA/PBAT Blends for Toughened Rigid and Flexible Items. Ph.D. Thesis, University of Pisa, Pisa, Italy, Date of Completion. 5 July 2020. [Google Scholar]
- Dhanasekharan, K.M.; Kokini, J. Design and scaling of wheat dough extrusion by numerical simulation of flow and heat transfer. J. Food Eng. 2003, 60, 421–430. [Google Scholar] [CrossRef]
- Shah, A.; Gupta, M. Proceedings of the Comparision of the Flow in Co-Rotating and Counter-Rotating Twin-Screw Extruders, Annual Technical Conference—ANTEC; Chicago’s Navy Pier: Chicago, IL, USA, 2004; pp. 443–447. [Google Scholar]
- Lazzeri, A.; Phuong, T.V.; Cinelli, P. Copolymers Based on Reactive Polyesters and Plasticisers for the Manufacture of Transparent, Biodegradable Packaging Film; 2013. EP2844685A1, 22 June 2016. [Google Scholar]
- Mallegni, N.; Phuong, T.V.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, C. Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion. Materilas 2018, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Cinelli, P.; Coltelli, M.-B.; Lazzeri, A. Naturally Made Hard Containers for Food Packaging: Actual and Future Perspectives. In Bionanotechnology to Save the Environment; Plant and Fishery’s Biomass as Alternative to Petrol; Morganti, P., Ed.; MDPI: Basel, Switzerland, 2018; pp. 297–318. [Google Scholar]
- Ruys, L.; van Olmen, R. New Developments in Agrotextiles Based on the Results of the FP7 Project “Bioagrotex”; Grant agreement ID: 213501; EU research project; 2012. [Google Scholar]
- Drumright, R.E.; Gruber, P.R.; Henton, D.E. Polylactic Acid Technology. Adv. Mater. 2000, 12, 1841–1846. [Google Scholar] [CrossRef]
- Bonaldi, R. Functional finishes for high-performance apparel. High-Perform. Apparel 2018, 129–156. [Google Scholar] [CrossRef]
- Centexbel—VKC. Extrusie (Textiel). Available online: https://www.centexbel.be/en (accessed on 20 December 2019).
- EU Project EcobioNet. Available online: https://ec.europa.eu/environment/eco-innovation/projects/en/projects/ecobionet (accessed on 12 March 2020).
- European Commission. A European Strategy for Plastics in a Circular Economy. Available online: https://ec.europa.eu/environment/circular-economy/pdf/plastics-strategy-brochure.pdf (accessed on 2 July 2020).
- Gesellschaft für Verpackungsmarktforschung. Flexible Plastic Packaging Market in Germany and in Europe; GVM Gesellschaft für Verpackungsmarktforschung mbH: Mainz, Germany, 2018. [Google Scholar]
- Peelman, N.; Ragaert, P.; de Meulenaer, B.; Adons, D.; Peeters, R.; Cardon, L.; van Impe, F.; Devlieghere, F. Application of bioplastics for food packaging. Trends Food Sci. Technol. 2013, 32, 128–141. [Google Scholar] [CrossRef]
- Jiménez-Rosado, M.; Zarate-Ramírez, L.; Romero, A.; Bengoechea, C.; Partal, P.; Guerrero, A. Bioplastics based on wheat gluten processed by extrusion. J. Clean. Prod. 2019, 239, 117994. [Google Scholar] [CrossRef]
- Global biodegradable plastics market report 2018: Forecast to 2023 featuring NatureWorks, BASF, Total Corbion PLA, Mitsubishi Chemical, and Biome Bioplastics. Focus Catal. 2019, 2019, 2. [CrossRef]
- Novamont GmbH. Available online: https://germany.novamont.com/page.php?id_page=2&id_first=2 (accessed on 11 March 2020).
- Piscopo, A.; Zappia, A.; de Bruno, A.; Pozzo, S.; Limbo, S.; Piergiovanni, L.; Poiana, M. Use of biodegradable materials as alternative packaging of typical Calabrian Provola cheese. Food Packag. Shelf Life 2019, 21, 100351. [Google Scholar] [CrossRef]
- Koide, S.; Shi, J. Microbial and quality evaluation of green peppers stored in biodegradable film packaging. Food Control. 2007, 18, 1121–1125. [Google Scholar] [CrossRef]
- Khakalo, A.; Filpponen, I.; Rojas, O.J. Protein-mediated interfacial adhesion in composites of cellulose nanofibrils and polylactide: Enhanced toughness towards material development. Compos. Sci. Technol. 2018, 160, 145–151. [Google Scholar] [CrossRef]
- Reis, M.O.; Olivato, J.B.; Bilck, A.P.; Zanela, J.; Grossmann, M.V.E.; Yamashita, F. Biodegradable trays of thermoplastic starch/poly(lactic acid) coated with beeswax. Ind. Crop. Prod. 2018, 112, 481–487. [Google Scholar] [CrossRef]
- Soares, F.C.; Yamashita, F.; Müller, C.; Pires, A.T. Thermoplastic starch/poly(lactic acid) sheets coated with cross-linked chitosan. Polym. Test. 2013, 32, 94–98. [Google Scholar] [CrossRef]
- Bio-Based News. Available online: http://news.bio-based.eu/globally-2014-a-good-year-for-renewables/ (accessed on 12 March 2020).
- GualapackGroup. Available online: https://gualapackgroup.com (accessed on 11 March 2020).
- Nawab, A.; Alam, F.; Haq, M.A.; Haider, M.S.; Lutfi, Z.; Kamaluddin, S.; Hasnain, A. Innovative edible packaging from mango kernel starch for the shelf life extension of red chili powder. Int. J. Boil. Macromol. 2018, 114, 626–631. [Google Scholar] [CrossRef]
- Nielsen, T.D.; Holmberg, K.; Stripple, J. Need a bag? A review of public policies on plastic carrier bags—Where, how and to what effect? Waste Manag. 2019, 87, 428–440. [Google Scholar] [CrossRef] [PubMed]
- Martinho, G.; Balaia, N.; Pires, A. The Portuguese plastic carrier bag tax: The effects on consumer’s behavior. Waste Manag. 2017, 61, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Lunt, J.; Bone, J. Properties and dyeability of fibers and fabrics produced from polylactide (PLA) polymers. Aatcc Rev. 2001, 1, 20–23. [Google Scholar]
- Palade, L.-I.; Lehermeier, H.J.; Dorgan, J.R. Melt Rheology of Highl-Content Poly(lactic acid). Macromolecules 2001, 34, 1384–1390. [Google Scholar] [CrossRef]
- Oever, M.V.D.; Molenveld, K.; van der Zee, M.; Bos, H. FBR Sustainable Chemistry & Technology; Bio-based and Biodegradable Plastics: Facts and Figures: Focus on Food Packaging in the Netherlands; Wageningen Food & Biobased Research: Wageningen, The Netherlands, 2017; Volume 1722. [Google Scholar] [CrossRef]
- Toray. ecodear® Plant-based Synthetic Fibe. Available online: https://www.toray.com/products/fibers/fib_0131.html (accessed on 12 March 2020).
- Polisilk. PLA Filament Yarns. Available online: http://www.yarnsource.co.uk/films-fibres-fabrics/ (accessed on 12 March 2020).
- Swicofil. Polyester High Tenacity. Available online: https://www.swicofil.com/commerce/products/polyester/165/hightenacity (accessed on 12 March 2020).
- Coats Group. Available online: https://www.coats.com (accessed on 12 March 2020).
- Bio4Pack. Available online: https://www.bio4pack.com/de/aktuell/10-jahre-bio4pack-10-jahre-innovation-in-nachhaltiger-verpackung/ (accessed on 12 March 2020).
- Niaounakis, M. Biopolymers. Processing and Products; William Andrew: Oxford, UK, 2015; ISBN 9780323266987. [Google Scholar]
- FKuR. Available online: https://fkur.com/en/ (accessed on 12 March 2020).
- Bio-Netting®—Netco BV. Available online: https://www.netco-bv.nl/en/bio-netting.html (accessed on 6 May 2020).
- Hottle, T.A.; Bilec, M.M.; Landis, A.E. Sustainability assessments of bio-based polymers. Polym. Degrad. Stab. 2013, 98, 1898–1907. [Google Scholar] [CrossRef]
- Khalil, H.A.; Davoudpour, Y.; Saurabh, C.; Hossain, S.; Adnan, A.S.; Dungani, R.; Paridah, M.T.; Sarker, Z.I.; Fazita, M.N.; Syakir, M.; et al. A review on nanocellulosic fibres as new material for sustainable packaging: Process and applications. Renew. Sustain. Energy Rev. 2016, 64, 823–836. [Google Scholar] [CrossRef]
- Leceta, I.; Etxabide, A.; Cabezudo, S.; de la Caba, K.; Guerrero, P. Bio-based films prepared with by-products and wastes: Environmental assessment. J. Clean. Prod. 2014, 64, 218–227. [Google Scholar] [CrossRef]
- Detzel, A.; Kauertz, B.; Derreza-Greeven, C. Study of the Environmental Impacts of Packagings Made of Biodegradable Plastics. 2012. Available online: https://www.umweltbundesamt.de/sites/default/files/medien/461/publikationen/4446.pdf (accessed on 2 July 2020).
- Vink, E.T.; Glassner, D.; Kolstad, J.J.; Wooley, R.J.; O’Connor, R.P. Original Research: The eco-profiles for current and near-future NatureWorks® polylactide (PLA) production. Ind. Biotechnol. 2007, 3, 58–81. [Google Scholar] [CrossRef]
- Vink, E.T.; Davies, S. Life Cycle Inventory, and Impact Assessment Data for 2014 Ingeo™ Polylactide Production. Ind. Biotechnol. 2015, 11, 167–180. [Google Scholar] [CrossRef]
- Hann, S.; Scholes, R.; Molteno, S.; Hilton, M.; Favoino, E.; Jakobsen, L.G. Relevance of Biodegradable and Compostable Consumer Plastic Products and Packaging in a Circular Economy; Project conducted under Framework Contract No ENV.B.3/FRA/2017/005; Final Report for the European Commission DG Environment: Bristol, UK, 2020. [Google Scholar]
- European Commission. Environmental Impact Assessments of Innovative Bio-Based Products—Summary of methodology and conclusions. Task 1 of “Study on Support to R&I Policy in the Area of Bio-based Products and Services”; European Commission: Brussels, Belgium, 2018. [Google Scholar]
- Pilz, H.; Brandt, B.; Fehringer, R. The Impact of Plastics on Life Cycle Energy Consumption and Greenhouse Gas Emissions in Europe. Summary report. June 2010. Available online: https://www.plasticseurope.org/application/files/9015/1310/4686/september-2010-the-impact-of-plastic.pdf (accessed on 15 May 2020).
- European Bioplastics. Environmental Benefits of Bioplastics. Available online: https://www.european-bioplastics.org/ (accessed on 10 March 2020).
- European Bioplastics. Environmental Benefits of Bioplastics; European Bioplastics. Available online: https://www.european-bioplastics.org/bioplastics/environment/ (accessed on 2 July 2020).
- Plastics Europe. The European Association of Plastics Recycling Recovery Organisations. Plastics—The Facts 2017: An analysis of European plastics production, demand, and waste data. Available online: https://www.plasticseurope.org/application/files/5715/1717/4180/Plastics_the_facts_2017_FINAL_for_website_one_page.pdf (accessed on 2 July 2020).
- Beckham, G.T.; Johnson, C.W.; Donohoe, B.S.; Rorrer, N.; Mcgeehan, J.E.; Austin, H.P.; Allen, M.D. Enzymes for Polymer Degradation. WO2019168811, 6 September 2019. [Google Scholar]
- Narancic, T.; Verstichel, S.; Chaganti, S.R.; Morales-Gamez, L.; Kenny, S.T.; de Wilde, B.; Padamati, R.B.; O’Connor, K.E. Biodegradable Plastic Blends Create New Possibilities for End-of-Life Management of Plastics but They Are Not a Panacea for Plastic Pollution. Environ. Sci. Technol. 2018, 52, 10441–10452. [Google Scholar] [CrossRef]
- EnviroSim—Wastewater Modeling Software. Available online: https://envirosim.com/ (accessed on 10 June 2020).
- Punch, W.F. A System for Predicting the Biodegradability of New Compounds. Research Project Database, Grantee Research Project. EPA Grant Number: R826114. Available online: https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/949 (accessed on 2 July 2020).
- Abi-Akl, R.; Ledieu, E.; Enke, T.N.; Cordero, O.X.; Cohen, T. Physics-based prediction of biopolymer degradation. Soft Matter 2019, 15, 4098–4108. [Google Scholar] [CrossRef] [PubMed]
- Babu, G.L.S.; Reddy, K.R.; Chouskey, S.K.; Kulkarni, H.S. Prediction of Long-Term Municipal Solid Waste Landfill Settlement Using Constitutive Model. Pr. Period. Hazardous Toxic Radioact. Waste Manag. 2010, 14, 139–150. [Google Scholar] [CrossRef]
- EagleVizion—Plastics sorting. Available online: http://www.eaglevizion.com/plastics-sorting (accessed on 30 March 2020).
- NatureWorks LLC. Using Near-Infrared Sorting of Recycle PLA Bottles. Available online: https://www.natureworksllc.com/~/media/The_Ingeo_Journey/EndofLife_Options/mech_recycling/20090708_NatureWorks_UsingNIRSortingtoRecyclePLABottles_pdf.pdf (accessed on 20 December 2019).
- de Andrade, M.F.C.; Souza, P.M.S.; Cavalett, O.; Morales, A.R. Life Cycle Assessment of Poly(Lactic Acid) (PLA): Comparison Between Chemical Recycling, Mechanical Recycling and Composting. J. Polym. Environ. 2016, 24, 372–384. [Google Scholar] [CrossRef]
- Badia, J.; Ribes-Greus, A. Mechanical recycling of polylactide, upgrading trends and combination of valorization techniques. Eur. Polym. J. 2016, 84, 22–39. [Google Scholar] [CrossRef]
- Najafi, N.; Heuzey, M.-C.; Carreau, P.; Wood-Adams, P.M. Control of thermal degradation of polylactide (PLA)-clay nanocomposites using chain extenders. Polym. Degrad. Stab. 2012, 97, 554–565. [Google Scholar] [CrossRef]
- Phuong, V.T.; Gigante, V.; Aliotta, L.; Coltelli, M.B.; Cinelli, P.; Lazzeri, C. Reactively extruded ecocomposites based on poly(lactic acid)/bisphenol A polycarbonate blends reinforced with regenerated cellulose microfibers. Compos. Sci. Technol. 2017, 139, 127–137. [Google Scholar] [CrossRef]
- Takatani, M.; Ikeda, K.; Sakamoto, K.; Okamoto, T. Cellulose esters as compatibilizers in wood/poly(lactic acid) composite. J. Wood Sci. 2008, 54, 54–61. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Bronco, S.; Chinea, C. The effect of free radical reactions on structure and properties of poly(lactic acid) (PLA) based blends. Polym. Degrad. Stab. 2010, 95, 332–341. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Toncelli, C.; Ciardelli, F.; Bronco, S. Compatible blends of biorelated polyesters through catalytic transesterification in the melt. Polym. Degrad. Stab. 2011, 96, 982–990. [Google Scholar] [CrossRef]
- Henton, D.; Gruber, P.; Lunt, J.; Randall, J. Polylactic Acid Technology. Natur. Fibers Biopolym. Biocom. 2005. [Google Scholar] [CrossRef]
- Piemonte, V.; Sabatini, S.; Gironi, F. Chemical Recycling of PLA: A Great Opportunity Towards the Sustainable Development? J. Polym. Environ. 2013, 21, 640–647. [Google Scholar] [CrossRef]


| Bio-Based Material | Synthetic/Non-Synthetic Produced Polymer | Classification | Common Feedstock or Source | Monomer/Sub-Unit | Common Production Technique | Main Intrinsic Variables | Expected Production Capacity by 2023 (Million Tonnes) [29] | End of Life (Common) | Recent Investigations in the Field of Packaging | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Polylactic Acid (PLA) | Synthetic | Aliphatic Polyester | Corn, Corn Stover, Sugarcane Bagasse, Sugar Beet, Rice Hulls | Lactic Acid (L- and D-Isomers) | Ring Opening Polymerisation Technique; Polycondensation | Ratio of Isomers; Molecular Weight | 0.83 | Industrial Composting, Mechanical and Chemical recy Cling | CopolymerizAtion with Dicarboxylic Acids; Degradation Accelerating Modifications or Additives; Blending PLA | [30,31,32,33,34] |
| Polyethylene Furanoate (PEF) | Synthetic | Aromatic Polyester | Corn, Wheat | 2,5-FurandicarboXylic Acid, Monoethylene Glycol | Polycondensation | Molecular Weight | No Data | Target Recycling when Market Grows | Alternative Feedstocks: Techniques to Lower the price | [35,36] |
| Polybutylene Succinate (PBS) | Synthetic | Aliphatic Polyester | Sugar Cane, Sugar Beet, Corn, Potato, Wheat | Succinic Acid and 1,4-Butanediol, | Polyconcensation | Molecular Weight | 0.54 (Includes PBS, PBAT and PCL) | Chemical/Catalytic Recycling, Enzymatic Depolymerisation | PBS in Composites to Enhance Processability | [29,37,38,39,40,41] |
| PolyhydroxyalkaNoate (PHA) | Non-Synthetic | Aliphatic Polyester (Family) | Sugars and Emerging Trials with Waste Biomass | Depending on the Sub Type | Microbial Fermentation | Molecular Weight; Monomer Types; Crystallinity | 0.17 | Industrial and Home Composting; Biogas Installation | Reduction of Production Costs (Mixed Microbial Cultures; Low Cost Feedstocks; Fillers or Blends) | [9,25,41,42,43,44] |
| Cellulose | Non-Synthetic | Polysaccharide | Plant material | β-D-Glucose | Naturally Occurring | Regenerated Cellulose | 0.03 | Home composting, Industrial Composting, Anaerobic Digestion; | Cellulose Nanocrystal and Cellulose Nanofiber Films and Composites | [41,45,46,47] |
| Starch | Non-Synthetic | Polysaccharide | Potatoes, Corn, Wheat, Rice, Tapioca | D-Glucose | Naturally Occurring | Thermoplastic Starch | 0.22 | Totally Biodegradabl; Industrial or Home Compost | Starch in Composite Films | [41,48,49,50,51] |
| Protein | Non-Synthetic | Proteins | Plants and Animals | Amino Acids | Naturally Occurring | Amino Acid Composition, Source; Degree of Denaturation; Purity | No Data | Totally Biodegradable | Physical, Chemical, and Enzymatic Modifications of Protein Films and Coatings | [12,13,15,16,20] |
| Lipids and Waxes | Non-Synthetic | Lipids | Plants and Animals | Fatty Acids and Other Hydrocarbons | Naturally Occurring | Carbon Length of Fatty Acid; Source; Accompanying Substances | No Data | Totally Biodegradable | Edible Coatings; Multilayer | [31,52,53] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reichert, C.L.; Bugnicourt, E.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A.; Canesi, I.; Braca, F.; Martínez, B.M.; Alonso, R.; Agostinis, L.; et al. Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers 2020, 12, 1558. https://doi.org/10.3390/polym12071558
Reichert CL, Bugnicourt E, Coltelli M-B, Cinelli P, Lazzeri A, Canesi I, Braca F, Martínez BM, Alonso R, Agostinis L, et al. Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers. 2020; 12(7):1558. https://doi.org/10.3390/polym12071558
Chicago/Turabian StyleReichert, Corina L., Elodie Bugnicourt, Maria-Beatrice Coltelli, Patrizia Cinelli, Andrea Lazzeri, Ilaria Canesi, Francesca Braca, Belén Monje Martínez, Rafael Alonso, Lodovico Agostinis, and et al. 2020. "Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability" Polymers 12, no. 7: 1558. https://doi.org/10.3390/polym12071558
APA StyleReichert, C. L., Bugnicourt, E., Coltelli, M.-B., Cinelli, P., Lazzeri, A., Canesi, I., Braca, F., Martínez, B. M., Alonso, R., Agostinis, L., Verstichel, S., Six, L., Mets, S. D., Gómez, E. C., Ißbrücker, C., Geerinck, R., Nettleton, D. F., Campos, I., Sauter, E., ... Schmid, M. (2020). Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers, 12(7), 1558. https://doi.org/10.3390/polym12071558