Chitosan-Based Drug Delivery System: Applications in Fish Biotechnology
Abstract
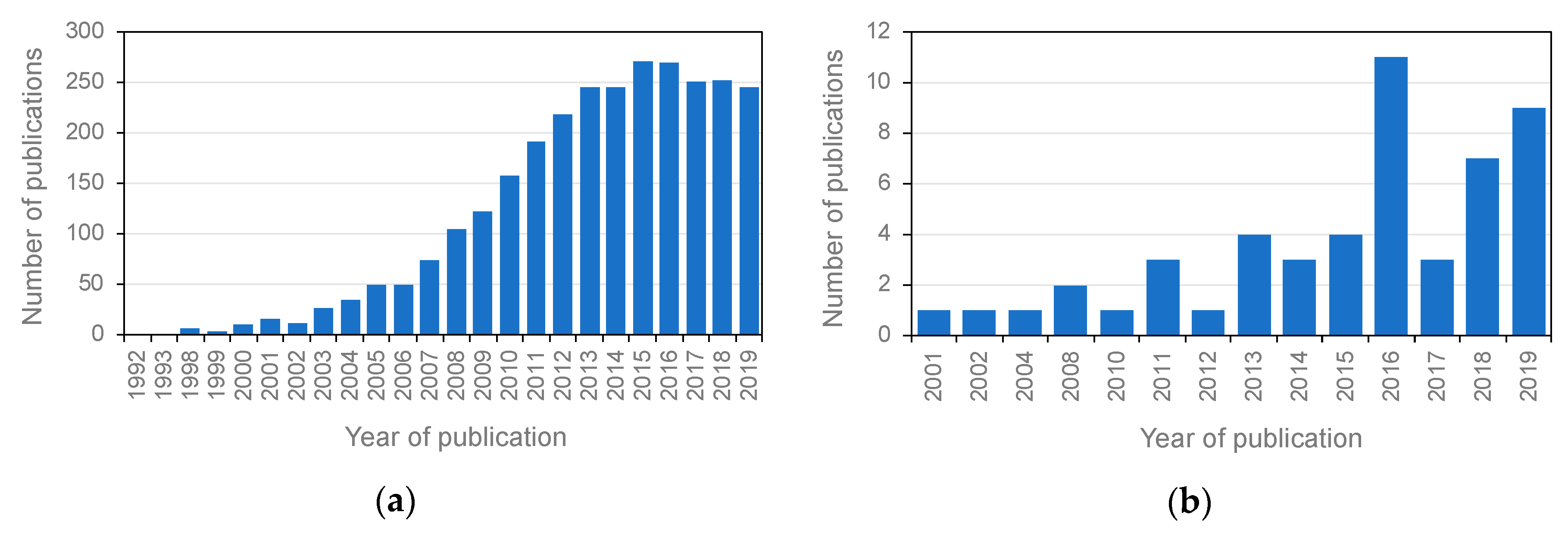
1. Introduction
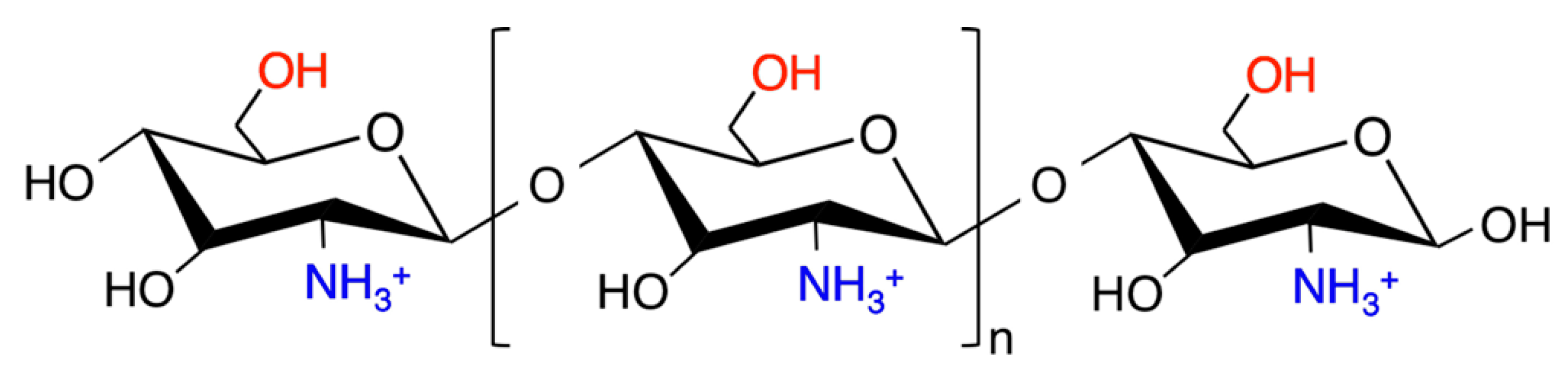
2. Chitosan as a Nucleic Acid Delivery Vector
2.1. Chitosan Derivatization
2.2. Chitosan Solubility
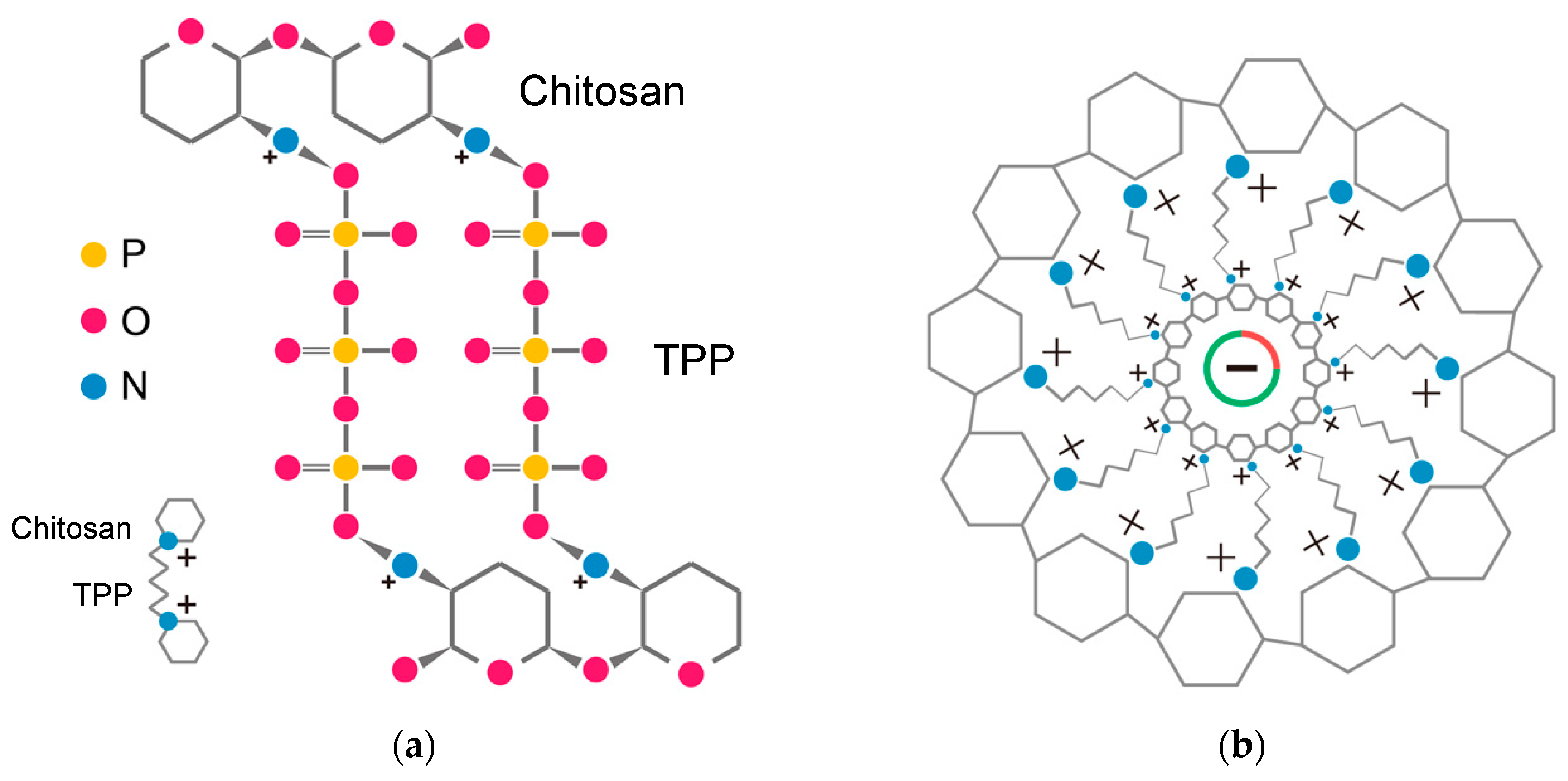
2.3. Stability of Chitosan Polyplexes
2.4. Targeting Drug Delivery, Cellular Uptake and Intracellular Trafficking
2.4.1. Targeting Drug Delivery with Chitosan Derivatives
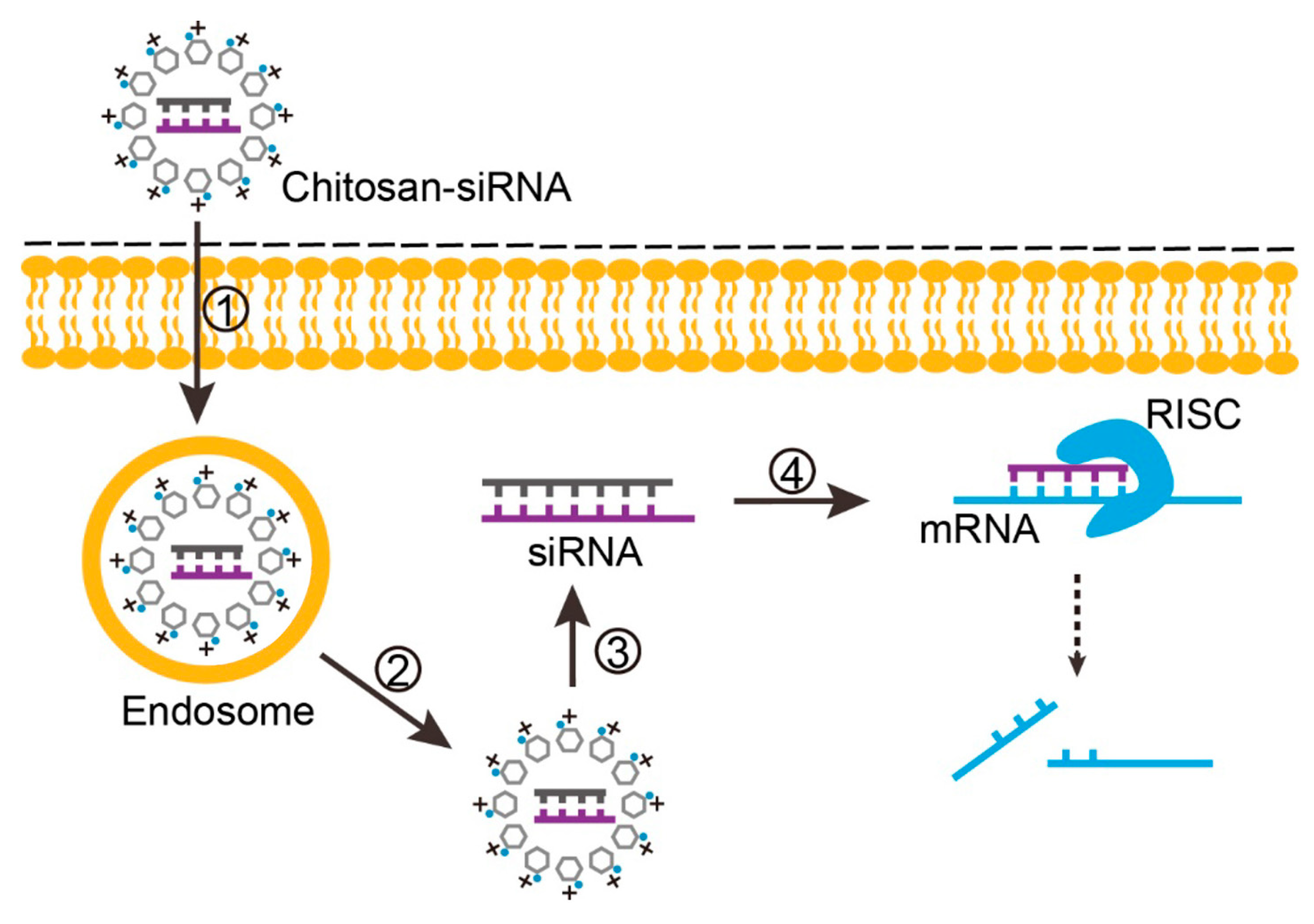
2.4.2. Endosomal Escape, Unpacking and Nuclear Import of DNA
3. Use of Chitosan in Fish Biotechnology
3.1. Chitosan and Its Derivatives as Dietary Additives
3.1.1. Dietary Supplementation with Chitosan
3.1.2. Dietary Supplementation with Chitosan Nanoparticles
3.1.3. Dietary Supplementation with Chitin and Chitooligosaccharide
3.2. Chitosan as a Carrier for Drug Delivery in Fish
3.2.1. Chitosan Loading Chemical Compounds
3.2.2. Chitosan Loading Metal Ions
3.2.3. Chitosan Loading Inactivated Pathogens
3.2.4. Chitosan Loading Proteins
3.2.5. Chitosan Loading Nucleic Acids
3.3. Chitosan-Based Applications in Fish Biotechnology and Gene Therapy
3.3.1. Fish Vaccination
3.3.2. Control of Gonadal Development
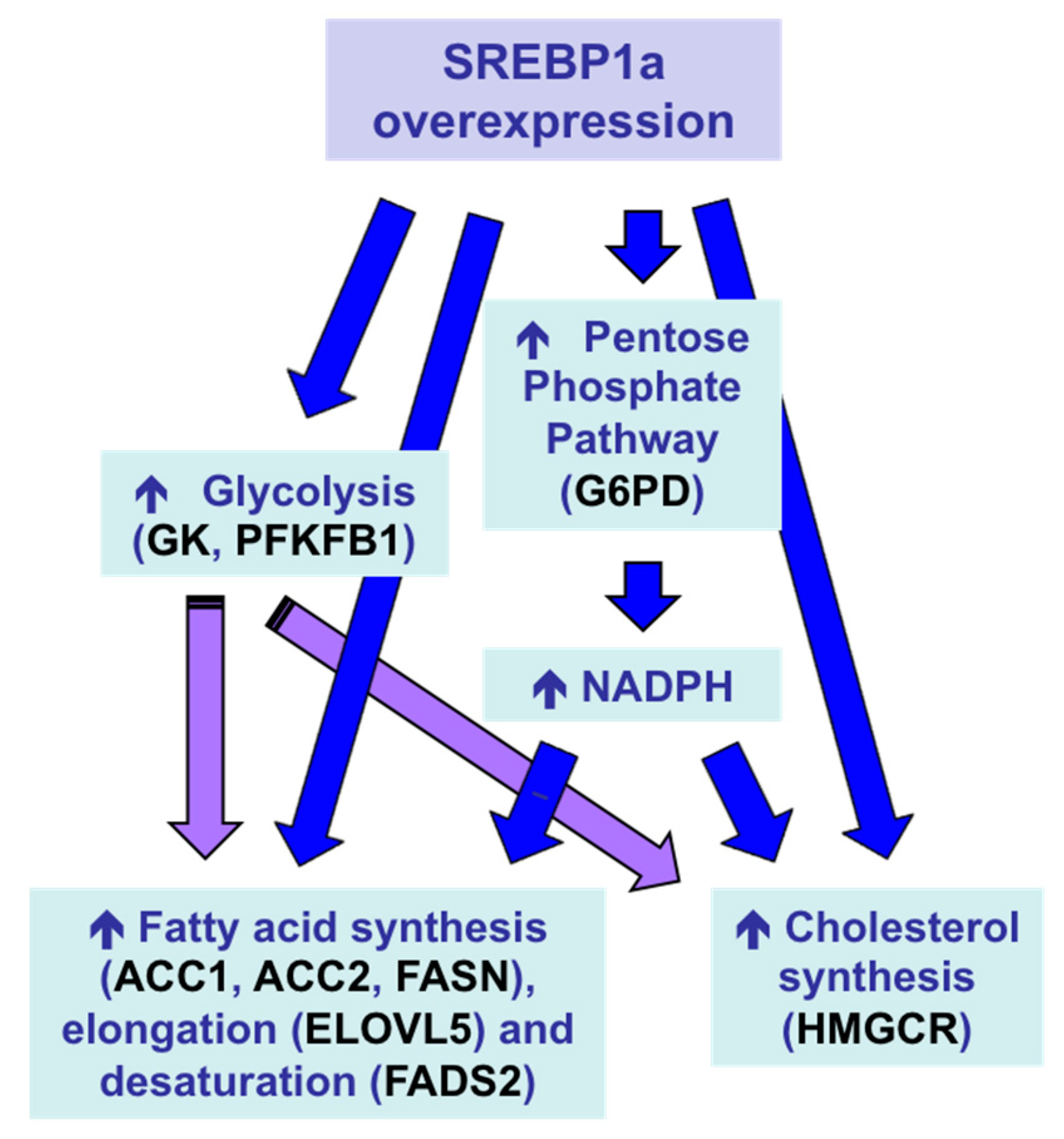
3.3.3. Control of Fish Metabolism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mahdy Samar, M.; El-Kalyoubi, M.H.; Khalaf, M.M.; Abd El-Razik, M.M. Physicochemical, functional, antioxidant and antibacterial properties of chitosan extracted from shrimp wastes by microwave technique. Ann. Agric. Sci. 2013, 58, 33–41. [Google Scholar] [CrossRef]
- Sun, M.; Wang, T.; Pang, J.; Chen, X.; Liu, Y. Hydroxybutyl chitosan centered biocomposites for potential curative applications: A critical review. Biomacromolecules 2020, 21, 1351–1367. [Google Scholar] [CrossRef] [PubMed]
- Sacco, P.; Cok, M.; Scognamiglio, F.; Pizzolitto, C.; Vecchies, F.; Marfoglia, A.; Marsich, E.; Donati, I. Glycosylated-chitosan derivatives: A systematic review. Molecules 2020, 25, 1534. [Google Scholar] [CrossRef] [PubMed]
- Chuan, D.; Jin, T.; Fan, R.; Zhou, L.; Guo, G. Chitosan for gene delivery: Methods for improvement and applications. Adv. Colloid Interface Sci. 2019, 268, 25–38. [Google Scholar] [CrossRef]
- Ivanova, D.G.; Yaneva, Z.L. Antioxidant properties and redox-modulating activity of chitosan and its derivatives: Biomaterials with application in cancer therapy. Biores. Open Access 2020, 9, 64–72. [Google Scholar] [CrossRef]
- Wang, W.; Meng, Q.; Li, Q.; Liu, J.; Zhou, M.; Jin, Z.; Zhao, K. Chitosan derivatives and their application in biomedicine. Int. J. Mol. Sci. 2020, 21, 487. [Google Scholar] [CrossRef]
- Raftery, R.; O’Brien, F.J.; Cryan, S.-A. Chitosan for gene delivery and orthopedic tissue engineering applications. Molecules 2013, 18, 5611–5647. [Google Scholar] [CrossRef]
- Lostalé-Seijo, I.; Montenegro, J. Synthetic materials at the forefront of gene delivery. Nat. Rev. Chem. 2018, 2, 258–277. [Google Scholar] [CrossRef]
- Cao, Y.; Tan, Y.F.; Wong, Y.S.; Liew, M.W.J.; Venkatraman, S. Recent Advances in Chitosan-Based Carriers for Gene Delivery. Mar. Drugs 2019, 17, 381. [Google Scholar] [CrossRef]
- Santos-Carballal, B.; Fernández Fernández, E.; Goycoolea, F. Chitosan in non-viral gene delivery: Role of structure, characterization methods, and insights in cancer and rare diseases therapies. Polymers 2018, 10, 444. [Google Scholar] [CrossRef]
- Ginn, S.L.; Amaya, A.K.; Alexander, I.E.; Edelstein, M.; Abedi, M.R. Gene therapy clinical trials worldwide to 2017: An update. J. Gene Med. 2018, 20, e3015. [Google Scholar] [CrossRef] [PubMed]
- Picanço-Castro, V.; Pereira, C.G.; Covas, D.T.; Porto, G.S.; Athanassiadou, A.; Figueiredo, M.L. Emerging patent landscape for non-viral vectors used for gene therapy. Nat. Biotechnol. 2020, 38, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Simões, S.; Filipe, A.; Faneca, H.; Mano, M.; Penacho, N.; Düzgünes, N.; de Lima, M.P. Cationic liposomes for gene delivery. Expert Opin. Drug Deliv. 2005, 2, 237–254. [Google Scholar] [CrossRef] [PubMed]
- Saffari, M.; Moghimi, H.; Dass, C. Barriers to liposomal gene delivery: From application site to the target. Iran. J. Pharm. Res. 2016, 15, 3–17. [Google Scholar]
- Ramamoorth, M.; Narvekar, A. Non viral vectors in gene therapy- an overview. J. Clin. Diagn. Res. 2015, 9, GE01–GE06. [Google Scholar] [CrossRef]
- Patil, S.; Gao, Y.G.; Lin, X.; Li, Y.; Dang, K.; Tian, Y.; Zhang, W.J.; Jiang, S.F.; Qadir, A.; Qian, A.R. The development of functional non-viral vectors for gene delivery. Int. J. Mol. Sci. 2019, 20, 5491. [Google Scholar] [CrossRef]
- Mumper, R.; Wang, J.; Claspell, J.; Rolland, A. Novel polymeric condensing carriers for gene delivery. Proc. Int. Symp. Control. Release Bioact. Mater. 1995, 22, 178–179. [Google Scholar]
- MacLaughlin, F.C.; Mumper, R.J.; Wang, J.; Tagliaferri, J.M.; Gill, I.; Hinchcliffe, M.; Rolland, A.P. Chitosan and depolymerized chitosan oligomers as condensing carriers for in vivo plasmid delivery. J. Control. Release 1998, 56, 259–272. [Google Scholar] [CrossRef]
- Katas, H.; Alpar, H.O. Development and characterisation of chitosan nanoparticles for siRNA delivery. J. Control. Release 2006, 115, 216–225. [Google Scholar] [CrossRef]
- Richardson, S.C.; Kolbe, H.V.; Duncan, R. Potential of low molecular mass chitosan as a DNA delivery system: Biocompatibility, body distribution and ability to complex and protect DNA. Int. J. Pharm. 1999, 178, 231–243. [Google Scholar] [CrossRef]
- Xu, W.; Shen, Y.; Jiang, Z.; Wang, Y.; Chu, Y.; Xiong, S. Intranasal delivery of chitosan-DNA vaccine generates mucosal SIgA and anti-CVB3 protection. Vaccine 2004, 22, 3603–3612. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-J.; Pan, S.-P.; Xie, Q.-X.; Xiao, L.-J. Preparation of chitosan-plasmid DNA nanoparticles encoding zona pellucida glycoprotein-3alpha and its expression in mouse. Mol. Reprod. Dev. 2004, 68, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Revuri, V.; Lee, S.-J.; Cho, S.; Park, I.-K.; Cho, K.J.; Bae, W.K.; Lee, Y. Oral siRNA delivery to treat colorectal liver metastases. ACS Nano 2017, 11, 10417–10429. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Jia, H.-R.; Wu, F.-G. Glycol chitosan: A water-soluble polymer for cell imaging and drug delivery. Molecules 2019, 24, 4371. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Fong, C.-W.; Khor, E.; Lim, L.-Y. Transfection efficiency of chitosan vectors: Effect of polymer molecular weight and degree of deacetylation. J. Control. Release 2005, 106, 391–406. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Khor, E.; Lim, L.-Y. Uptake and cytotoxicity of chitosan molecules and nanoparticles: Effects of molecular weight and degree of deacetylation. Pharm. Res. 2004, 21, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Howard, K.A.; Dong, M.; Andersen, M.Ø.; Rahbek, U.L.; Johnsen, M.G.; Hansen, O.C.; Besenbacher, F.; Kjems, J. The influence of polymeric properties on chitosan/siRNA nanoparticle formulation and gene silencing. Biomaterials 2007, 28, 1280–1288. [Google Scholar] [CrossRef]
- Razmi, F.A.; Ngadi, N.; Wong, S.; Inuwa, I.M.; Opotu, L.A. Kinetics, thermodynamics, isotherm and regeneration analysis of chitosan modified pandan adsorbent. J. Clean. Prod. 2019, 231, 98–109. [Google Scholar] [CrossRef]
- Alameh, M.; Lavertu, M.; Tran-Khanh, N.; Chang, C.-Y.; Lesage, F.; Bail, M.; Darras, V.; Chevrier, A.; Buschmann, M.D. siRNA delivery with chitosan: Influence of chitosan molecular weight, degree of deacetylation, and amine to phosphate ratio on in vitro silencing efficiency, hemocompatibility, biodistribution, and in vivo efficacy. Biomacromolecules 2018, 19, 112–131. [Google Scholar] [CrossRef]
- Al-Remawi, M. Application of N-hexoyl chitosan derivatives with high degree of substitution in the preparation of super-disintegrating pharmaceutical matrices. J. Drug Deliv. Sci. Technol. 2015, 29, 31–41. [Google Scholar] [CrossRef]
- Chung, Y.-C.; Kuo, C.-L.; Chen, C.-C. Preparation and important functional properties of water-soluble chitosan produced through Maillard reaction. Bioresour. Technol. 2005, 96, 1473–1482. [Google Scholar] [CrossRef] [PubMed]
- Gullón, B.; Montenegro, M.I.; Ruiz-Matute, A.I.; Cardelle-Cobas, A.; Corzo, N.; Pintado, M.E. Synthesis, optimization and structural characterization of a chitosan-glucose derivative obtained by the Maillard reaction. Carbohydr. Polym. 2016, 137, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Uccello-Barretta, G.; Balzano, F.; Aiello, F.; Senatore, A.; Fabiano, A.; Zambito, Y. Mucoadhesivity and release properties of quaternary ammonium-chitosan conjugates and their nanoparticulate supramolecular aggregates: An NMR investigation. Int. J. Pharm. 2014, 461, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, Z.; Bao, X.; Xu, G.; Yao, P. Fatty acid and quaternary ammonium modified chitosan nanoparticles for insulin delivery. Colloids Surf. B. Biointerfaces 2018, 170, 136–143. [Google Scholar] [CrossRef]
- Jiang, X.; Dai, H.; Leong, K.W.; Goh, S.-H.; Mao, H.-Q.; Yang, Y.-Y. Chitosan-g-PEG/DNA complexes deliver gene to the rat liver via intrabiliary and intraportal infusions. J. Gene Med. 2006, 8, 477–487. [Google Scholar] [CrossRef]
- Ping, Y.; Liu, C.; Zhang, Z.; Liu, K.L.; Chen, J.; Li, J. Chitosan-graft-(PEI-β-cyclodextrin) copolymers and their supramolecular PEGylation for DNA and siRNA delivery. Biomaterials 2011, 32, 8328–8341. [Google Scholar] [CrossRef]
- Lee, H.; Jeong, J.H.; Park, T.G. PEG grafted polylysine with fusogenic peptide for gene delivery: High transfection efficiency with low cytotoxicity. J. Control. Release 2002, 79, 283–291. [Google Scholar] [CrossRef]
- Strand, S.P.; Issa, M.M.; Christensen, B.E.; Vårum, K.M.; Artursson, P. Tailoring of chitosans for gene delivery: Novel self-branched glycosylated chitosan oligomers with improved functional properties. Biomacromolecules 2008, 9, 3268–3276. [Google Scholar] [CrossRef]
- Thanou, M.; Florea, B.I.; Geldof, M.; Junginger, H.E.; Borchard, G. Quaternized chitosan oligomers as novel gene delivery vectors in epithelial cell lines. Biomaterials 2002, 23, 153–159. [Google Scholar] [CrossRef]
- Kean, T.; Roth, S.; Thanou, M. Trimethylated chitosans as non-viral gene delivery vectors: Cytotoxicity and transfection efficiency. J. Control. Release 2005, 103, 643–653. [Google Scholar] [CrossRef]
- Ren, Y.; Zhao, X.; Liang, X.; Ma, P.X.; Guo, B. Injectable hydrogel based on quaternized chitosan, gelatin and dopamine as localized drug delivery system to treat Parkinson’s disease. Int. J. Biol. Macromol. 2017, 105, 1079–1087. [Google Scholar] [CrossRef]
- Raik, S.V.; Andranovitš, S.; Petrova, V.A.; Xu, Y.; Lam, J.K.-W.; Morris, G.A.; Brodskaia, A.V.; Casettari, L.; Kritchenkov, A.S.; Skorik, Y.A. Comparative study of diethylaminoethyl-chitosan and methylglycol-chitosan as potential non-viral vectors for gene therapy. Polymers 2018, 10, 442. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Remuñán-López, C.; Vila-Jato, J.L.; Alonso, M.J. Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. J. Appl. Polym. Sci. 1997, 63, 125–132. [Google Scholar] [CrossRef]
- Gan, Q.; Wang, T.; Cochrane, C.; McCarron, P. Modulation of surface charge, particle size and morphological properties of chitosan–TPP nanoparticles intended for gene delivery. Colloids Surf. B Biointerfaces 2005, 44, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Raja, M.A.G.; Katas, H.; Jing Wen, T. Stability, intracellular delivery, and release of siRNA from chitosan nanoparticles using different cross-linkers. PLoS ONE 2015, 10, e0128963. [Google Scholar]
- Vimal, S.; Abdul Majeed, S.; Taju, G.; Nambi, K.S.N.; Sundar Raj, N.; Madan, N.; Farook, M.A.; Rajkumar, T.; Gopinath, D.; Sahul Hameed, A.S. Chitosan tripolyphosphate (CS/TPP) nanoparticles: Preparation, characterization and application for gene delivery in shrimp. Acta Trop. 2013, 128, 486–493. [Google Scholar] [CrossRef]
- Fàbregas, A.; Miñarro, M.; García-Montoya, E.; Pérez-Lozano, P.; Carrillo, C.; Sarrate, R.; Sánchez, N.; Ticó, J.R.; Suñé-Negre, J.M. Impact of physical parameters on particle size and reaction yield when using the ionic gelation method to obtain cationic polymeric chitosan-tripolyphosphate nanoparticles. Int. J. Pharm. 2013, 446, 199–204. [Google Scholar] [CrossRef]
- Serrano-Sevilla, I.; Artiga, Á.; Mitchell, S.G.; De Matteis, L.; de la Fuente, J.M. Natural polysaccharides for siRNA delivery: Nanocarriers based on chitosan, hyaluronic acid, and their derivatives. Molecules 2019, 24, 2570. [Google Scholar] [CrossRef]
- Köping-Höggård, M.; Tubulekas, I.; Guan, H.; Edwards, K.; Nilsson, M.; Vårum, K.; Artursson, P. Chitosan as a nonviral gene delivery system. Structure–property relationships and characteristics compared with polyethylenimine in vitro and after lung administration in vivo. Gene Ther. 2001, 8, 1108–1121. [Google Scholar] [CrossRef]
- Mao, H.Q.; Roy, K.; Troung-Le, V.L.; Janes, K.A.; Lin, K.Y.; Wang, Y.; August, J.T.; Leong, K.W. Chitosan-DNA nanoparticles as gene carriers: Synthesis, characterization and transfection efficiency. J. Control. Release 2001, 70, 399–421. [Google Scholar] [CrossRef]
- Chan, P.; Kurisawa, M.; Chung, J.E.; Yang, Y.-Y. Synthesis and characterization of chitosan-g-poly(ethylene glycol)-folate as a non-viral carrier for tumor-targeted gene delivery. Biomaterials 2007, 28, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Jhaveri, A.; Deshpande, P.; Pattni, B.; Torchilin, V. Transferrin-targeted, resveratrol-loaded liposomes for the treatment of glioblastoma. J. Control. Release 2018, 277, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Chen, J.; Xu, X.; Ding, Z.; Yang, Y.-H.; Hua, Z.; Zhang, J. Galactosylated low molecular weight chitosan as DNA carrier for hepatocyte-targeting. Int. J. Pharm. 2003, 255, 57–68. [Google Scholar] [CrossRef]
- Park, I.-K.; Yang, J.; Jeong, H.-J.; Bom, H.-S.; Harada, I.; Akaike, T.; Kim, S.-I.; Cho, C.-S. Galactosylated chitosan as a synthetic extracellular matrix for hepatocytes attachment. Biomaterials 2003, 24, 2331–2337. [Google Scholar] [CrossRef]
- Kim, T.H.; Nah, J.W.; Cho, M.-H.; Park, T.G.; Cho, C.S. Receptor-mediated gene delivery into antigen presenting cells using mannosylated chitosan/DNA nanoparticles. J. Nanosci. Nanotechnol. 2006, 6, 2796–2803. [Google Scholar] [CrossRef] [PubMed]
- Negm, N.A.; Hefni, H.H.H.; Abd-Elaal, A.A.A.; Badr, E.A.; Abou Kana, M.T.H. Advancement on modification of chitosan biopolymer and its potential applications. Int. J. Biol. Macromol. 2020, 152, 681–702. [Google Scholar] [CrossRef] [PubMed]
- Shastri, D.H. Thiolated chitosan: A boon to ocular delivery of therapeutics. MOJ Bioequivalence Bioavailab. 2017, 3, 34–37. [Google Scholar] [CrossRef]
- Mahmood, A.; Lanthaler, M.; Laffleur, F.; Huck, C.W.; Bernkop-Schnürch, A. Thiolated chitosan micelles: Highly mucoadhesive drug carriers. Carbohydr. Polym. 2017, 167, 250–258. [Google Scholar] [CrossRef]
- Boateng, J.S.; Ayensu, I. Preparation and characterization of laminated thiolated chitosan-based freeze-dried wafers for potential buccal delivery of macromolecules. Drug Dev. Ind. Pharm. 2014, 40, 611–618. [Google Scholar] [CrossRef]
- Boateng, J.; Mitchell, J.; Pawar, H.; Ayensu, I. Functional characterisation and permeation studies of lyophilised thiolated chitosan xerogels for buccal delivery of insulin. Protein Pept. Lett. 2014, 21, 1163–1175. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, C.; Zheng, X.; Shao, X.; Zhang, X.; Zhang, Q.; Jiang, X. Preparation and evaluation of antigen/N-trimethylaminoethylmethacrylate chitosan conjugates for nasal immunization. Vaccine 2014, 32, 2582–2590. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.-Q.; Zhang, Z.-K.; Cheng, L.-Y.; Xiao, J.-X. Intestine-targeted delivery potency of O-carboxymethyl chitosan-coated layer-by-layer microcapsules: An in vitro and in vivo evaluation. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 105, 110129. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Sun, X.; Zhang, Z. Kidney-targeted drug delivery systems. Acta Pharm. Sin. B 2014, 4, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Vasanthakumar, T.; Rubinstein, J.L. Structure and roles of V-type ATPases. Trends Biochem. Sci. 2020, 45, 295–307. [Google Scholar] [CrossRef]
- Li, W.; Nicol, F.; Szoka, F.C. GALA: A designed synthetic pH-responsive amphipathic peptide with applications in drug and gene delivery. Adv. Drug Deliv. Rev. 2004, 56, 967–985. [Google Scholar] [CrossRef]
- El Ouahabi, A.; Thiry, M.; Pector, V.; Fuks, R.; Ruysschaert, J.M.; Vandenbranden, M. The role of endosome destabilizing activity in the gene transfer process mediated by cationic lipids. FEBS Lett. 1997, 414, 187–192. [Google Scholar]
- Ma, Z.; Yang, C.; Song, W.; Wang, Q.; Kjems, J.; Gao, S. Chitosan Hydrogel as siRNA vector for prolonged gene silencing. J. Nanobiotechnol. 2014, 12, 23. [Google Scholar] [CrossRef]
- Shi, B.; Zheng, M.; Tao, W.; Chung, R.; Jin, D.; Ghaffari, D.; Farokhzad, O.C. Challenges in DNA delivery and recent advances in multifunctional polymeric DNA delivery systems. Biomacromolecules 2017, 18, 2231–2246. [Google Scholar] [CrossRef]
- Molinaro, R.; Wolfram, J.; Federico, C.; Cilurzo, F.; Di Marzio, L.; Ventura, C.A.; Carafa, M.; Celia, C.; Fresta, M. Polyethylenimine and chitosan carriers for the delivery of RNA interference effectors. Expert Opin. Drug Deliv. 2013, 10, 1653–1668. [Google Scholar] [CrossRef]
- Boussif, O.; Lezoualc’h, F.; Zanta, M.A.; Mergny, M.D.; Scherman, D.; Demeneix, B.; Behr, J.P. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: Polyethylenimine. Proc. Natl. Acad. Sci. USA 1995, 92, 7297–7301. [Google Scholar] [CrossRef]
- Bae, Y.; Lee, Y.H.; Lee, S.; Han, J.; Ko, K.S.; Choi, J.S. Characterization of glycol chitosan grafted with low molecular weight polyethylenimine as a gene carrier for human adipose-derived mesenchymal stem cells. Carbohydr. Polym. 2016, 153, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Cui, S.; Zhao, Y.; Zhang, C.; Zhang, S.; Peng, X. Grafting chitosan with polyethylenimine in an ionic liquid for efficient gene delivery. PLoS ONE 2015, 10, e0121817. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Jin, Z.; Huang, W.; Sheng, Y.; Wang, Z.; Guo, S. Tailor-made ternary nanopolyplexes of thiolated trimethylated chitosan with pDNA and folate conjugated cis-aconitic amide-polyethylenimine for efficient gene delivery. Int. J. Biol. Macromol. 2020, 152, 948–956. [Google Scholar] [CrossRef]
- Lee, Y.H.; Park, H.I.; Choi, J.S. Novel glycol chitosan-based polymeric gene carrier synthesized by a Michael addition reaction with low molecular weight polyethylenimine. Carbohydr. Polym. 2016, 137, 669–677. [Google Scholar] [CrossRef]
- Javan, B.; Atyabi, F.; Shahbazi, M. Hypoxia-inducible bidirectional shRNA expression vector delivery using PEI/chitosan-TBA copolymers for colorectal Cancer gene therapy. Life Sci. 2018, 202, 140–151. [Google Scholar] [CrossRef]
- Sun, B.; Zhao, R.; Kong, F.; Ren, Y.; Zuo, A.; Liang, D.; Zhang, J. Phosphorylatable short peptide conjugation for facilitating transfection efficacy of CS/DNA complex. Int. J. Pharm. 2010, 397, 206–210. [Google Scholar] [CrossRef]
- Zhao, R.; Sun, B.; Liu, T.; Liu, Y.; Zhou, S.; Zuo, A.; Liang, D. Optimize nuclear localization and intra-nucleus disassociation of the exogene for facilitating transfection efficacy of the chitosan. Int. J. Pharm. 2011, 413, 254–259. [Google Scholar] [CrossRef]
- Miao, J.; Yang, X.; Gao, Z.; Li, Q.; Meng, T.; Wu, J.; Yuan, H.; Hu, F. Redox-responsive chitosan oligosaccharide-SS-Octadecylamine polymeric carrier for efficient anti-Hepatitis B Virus gene therapy. Carbohydr. Polym. 2019, 212, 215–221. [Google Scholar] [CrossRef]
- Cryan, S.-A.; McKiernan, P.; Cunningham, C.M.; Greene, C. Targeting miRNA-based medicines to cystic fibrosis airway epithelial cells using nanotechnology. Int. J. Nanomed. 2013, 8, 3907. [Google Scholar] [CrossRef]
- González, J.D.; Silva-Marrero, J.I.; Metón, I.; Caballero-Solares, A.; Viegas, I.; Fernández, F.; Miñarro, M.; Fàbregas, A.; Ticó, J.R.; Jones, J.G.; et al. Chitosan-mediated shRNA knockdown of cytosolic alanine aminotransferase improves hepatic carbohydrate metabolism. Mar. Biotechnol. 2016, 18, 85–97. [Google Scholar] [CrossRef]
- Gaspar, C.; Silva-Marrero, J.I.; Fàbregas, A.; Miñarro, M.; Ticó, J.R.; Baanante, I.V.; Metón, I. Administration of chitosan-tripolyphosphate-DNA nanoparticles to knockdown glutamate dehydrogenase expression impairs transdeamination and gluconeogenesis in the liver. J. Biotechnol. 2018, 286, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Acharya, R. The recent progresses in shRNA-nanoparticle conjugate as a therapeutic approach. Mater. Sci. Eng. C 2019, 104, 109928. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Tang, C.; Yin, C. Oral delivery of shRNA based on amino acid modified chitosan for improved antitumor efficacy. Biomaterials 2015, 70, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-L.; Yao, H.-H.; Guo, L.-L.; Dong, L.; Li, S.-G.; Gu, Y.-P.; Qin, Z.-H. Selection of optimal sites for TGFB1 gene silencing by chitosan-TPP nanoparticle-mediated delivery of shRNA. Cancer Genet. Cytogenet. 2009, 190, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Avci, P.; Ahi, M.; Gazori, T.; Hamblin, M.R.; Naderi-Manesh, H. Evaluation of chitosan-tripolyphosphate nanoparticles as a p-shRNA delivery vector: Formulation, optimization and cellular uptake study. J. Nanopharm. Drug Deliv. 2013, 1, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Soliman, F.M.; Adly, M.A.; Soliman, H.A.M.; El-Matbouli, M.; Saleh, M. Recent progress in biomedical applications of chitosan and its nanocomposites in aquaculture: A review. Res. Vet. Sci. 2019, 126, 68–82. [Google Scholar] [CrossRef]
- Abdel-Ghany, H.M.; Salem, M.E. Effects of dietary chitosan supplementation on farmed fish; a review. Rev. Aquac. 2020, 12, 438–452. [Google Scholar] [CrossRef]
- Kono, M.; Matsui, T.; Shimizu, C. Effect of chitin, chitosan, and cellulose as deit supplements on the growth of cultured fish. Bull. Jpn. Soc. Sci. Fish. 1987, 53, 125–129. [Google Scholar] [CrossRef]
- Shiau, S.-Y.; Yu, Y.-P. Dietary supplementation of chitin and chitosan depresses growth in tilapia, Oreochromis niloticus×O. aureus. Aquaculture 1999, 179, 439–446. [Google Scholar] [CrossRef]
- Wu, S. The growth performance, body composition and nonspecific immunity of Tilapia (Oreochromis niloticus) affected by chitosan. Int. J. Biol. Macromol. 2020, 145, 682–685. [Google Scholar] [CrossRef]
- Fadl, S.E.; El-Gammal, G.A.; Abdo, W.S.; Barakat, M.; Sakr, O.A.; Nassef, E.; Gad, D.M.; El-Sheshtawy, H.S. Evaluation of dietary chitosan effects on growth performance, immunity, body composition and histopathology of Nile tilapia (Oreochromis niloticus) as well as the resistance to Streptococcus agalactiae infection. Aquac. Res. 2020, 51, 1120–1132. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, X.; Yang, Y.; Han, D.; Jin, J.; Xie, S. Effect of dietary chitosan on growth performance, haematology, immune response, intestine morphology, intestine microbiota and disease resistance in gibel carp (Carassius auratus gibelio). Aquac. Nutr. 2014, 20, 532–546. [Google Scholar] [CrossRef]
- El-Sayed, H.S.; Barakat, K.M. Effect of dietary chitosan on challenged Dicentrarchus labrax post larvae with Aeromonas hydrophila. Russ. J. Mar. Biol. 2016, 42, 501–508. [Google Scholar] [CrossRef]
- Yan, J.; Guo, C.; Dawood, M.A.O.; Gao, J. Effects of dietary chitosan on growth, lipid metabolism, immune response and antioxidant-related gene expression in Misgurnus anguillicaudatus. Benef. Microbes 2017, 8, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, L. Effects of chitosan-supplemented diets on the growth performance, nonspecific immunity and health of loach fish (Misgurnus anguillicadatus). Carbohydr. Polym. 2019, 225, 115227. [Google Scholar] [CrossRef] [PubMed]
- Kamali Najafabad, M.; Imanpoor, M.R.; Taghizadeh, V.; Alishahi, A. Effect of dietary chitosan on growth performance, hematological parameters, intestinal histology and stress resistance of Caspian kutum (Rutilus frisii kutum Kamenskii, 1901) fingerlings. Fish Physiol. Biochem. 2016, 42, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Samarakoon, K.W.; Cha, S.-H.; Lee, J.-H.; Jeon, Y.-J. The growth, innate immunity and protection against H2O2-induced oxidative damage of a chitosan-coated diet in the olive flounder Paralichthys olivaceus. Fish. Aquat. Sci. 2013, 16, 149–158. [Google Scholar] [CrossRef]
- Ranjan, R.; Prasad, K.P.; Vani, T.; Kumar, R. Effect of dietary chitosan on haematology, innate immunity and disease resistance of Asian seabass Lates calcarifer (Bloch). Aquac. Res. 2014, 45, 983–993. [Google Scholar] [CrossRef]
- Shanthi Mari, L.S.; Jagruthi, C.; Anbazahan, S.M.; Yogeshwari, G.; Thirumurugan, R.; Arockiaraj, J.; Mariappan, P.; Balasundaram, C.; Harikrishnan, R. Protective effect of chitin and chitosan enriched diets on immunity and disease resistance in Cirrhina mrigala against Aphanomyces invadans. Fish Shellfish Immunol. 2014, 39, 378–385. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Kim, J.-S.; Balasundaram, C.; Heo, M.-S. Immunomodulatory effects of chitin and chitosan enriched diets in Epinephelus bruneus against Vibrio alginolyticus infection. Aquaculture 2012, 326–329, 46–52. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Kim, J.-S.; Balasundaram, C.; Heo, M.-S. Dietary supplementation with chitin and chitosan on haematology and innate immune response in Epinephelus bruneus against Philasterides dicentrarchi. Exp. Parasitol. 2012, 131, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J. Effects of chitosan nanoparticles on survival, growth and meat quality of tilapia, Oreochromis nilotica. Nanotoxicology 2011, 5, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Tawwab, M.; Razek, N.A.; Abdel-Rahman, A.M. Immunostimulatory effect of dietary chitosan nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.). Fish Shellfish Immunol. 2019, 88, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Naby, F.S.; Naiel, M.A.E.; Al-Sagheer, A.A.; Negm, S.S. Dietary chitosan nanoparticles enhance the growth, production performance, and immunity in Oreochromis niloticus. Aquaculture 2019, 501, 82–89. [Google Scholar] [CrossRef]
- Naiel, M.A.E.; Ismael, N.E.M.; Abd El-hameed, S.A.A.; Amer, M.S. The antioxidative and immunity roles of chitosan nanoparticle and vitamin C-supplemented diets against imidacloprid toxicity on Oreochromis niloticus. Aquaculture 2020, 523, 735219. [Google Scholar] [CrossRef]
- Abd El-Naby, A.S.; Al-Sagheer, A.A.; Negm, S.S.; Naiel, M.A.E. Dietary combination of chitosan nanoparticle and thymol affects feed utilization, digestive enzymes, antioxidant status, and intestinal morphology of Oreochromis niloticus. Aquaculture 2020, 515, 734577. [Google Scholar] [CrossRef]
- Gao, J.-Q.; Hu, Y.L.; Wang, Q.; Han, F.; Shao, J.Z. Toxicity evaluation of biodegradable chitosan nanoparticles using a zebrafish embryo model. Int. J. Nanomed. 2011, 6, 3351–3359. [Google Scholar] [CrossRef]
- Nikapitiya, C.; Dananjaya, S.H.S.; De Silva, B.C.J.; Heo, G.-J.; Oh, C.; De Zoysa, M.; Lee, J. Chitosan nanoparticles: A positive immune response modulator as display in zebrafish larvae against Aeromonas hydrophila infection. Fish Shellfish Immunol. 2018, 76, 240–246. [Google Scholar] [CrossRef]
- Qin, C.; Zhang, Y.; Liu, W.; Xu, L.; Yang, Y.; Zhou, Z. Effects of chito-oligosaccharides supplementation on growth performance, intestinal cytokine expression, autochthonous gut bacteria and disease resistance in hybrid tilapia Oreochromis niloticus ♀ × Oreochromis aureus ♂. Fish Shellfish Immunol. 2014, 40, 267–274. [Google Scholar] [CrossRef]
- Gopalakannan, A.; Arul, V. Immunomodulatory effects of dietary intake of chitin, chitosan and levamisole on the immune system of Cyprinus carpio and control of Aeromonas hydrophila infection in ponds. Aquaculture 2006, 255, 179–187. [Google Scholar] [CrossRef]
- Karlsen, Ø.; Amlund, H.; Berg, A.; Olsen, R.E. The effect of dietary chitin on growth and nutrient digestibility in farmed Atlantic cod, Atlantic salmon and Atlantic halibut. Aquac. Res. 2017, 48, 123–133. [Google Scholar] [CrossRef]
- Lin, S.-M.; Jiang, Y.; Chen, Y.-J.; Luo, L.; Doolgindachbaporn, S.; Yuangsoi, B. Effects of Astragalus polysaccharides (APS) and chitooligosaccharides (COS) on growth, immune response and disease resistance of juvenile largemouth bass, Micropterus salmoides. Fish Shellfish Immunol. 2017, 70, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.D.; Van Dang, P.; Le, A.Q.; Nguyen, T.K.L.; Pham, D.H.; Van Nguyen, N.; Nguyen, Q.H. Effect of oligochitosan and oligo-β-glucan supplementation on growth, innate immunity, and disease resistance of striped catfish (Pangasianodon hypophthalmus). Biotechnol. Appl. Biochem. 2017, 64, 564–571. [Google Scholar] [CrossRef]
- Meng, X.; Wang, J.; Wan, W.; Xu, M.; Wang, T. Influence of low molecular weight chitooligosaccharides on growth performance and non-specific immune response in Nile tilapia Oreochromis niloticus. Aquac. Int. 2017, 25, 1265–1277. [Google Scholar] [CrossRef]
- Su, P.; Han, Y.; Jiang, C.; Ma, Y.; Pan, J.; Liu, S.; Zhang, T. Effects of chitosan-oligosaccharides on growth performance, digestive enzyme and intestinal bacterial flora of tiger puffer (Takifugu rubripes Temminck et Schlegel, 1850). J. Appl. Ichthyol. 2017, 33, 458–467. [Google Scholar] [CrossRef]
- Lin, S.; Mao, S.; Guan, Y.; Luo, L.; Luo, L.; Pan, Y. Effects of dietary chitosan oligosaccharides and Bacillus coagulans on the growth, innate immunity and resistance of koi (Cyprinus carpio koi). Aquaculture 2012, 342–343, 36–41. [Google Scholar]
- Lin, S.; Mao, S.; Guan, Y.; Lin, X.; Luo, L. Dietary administration of chitooligosaccharides to enhance growth, innate immune response and disease resistance of Trachinotus ovatus. Fish Shellfish Immunol. 2012, 32, 909–913. [Google Scholar] [CrossRef]
- Liu, Y.; Xing, R.; Liu, S.; Qin, Y.; Li, K.; Yu, H.; Li, P. Effects of chitooligosaccharides supplementation with different dosages, molecular weights and degrees of deacetylation on growth performance, innate immunity and hepatopancreas morphology in Pacific white shrimp (Litopenaeus vannamei). Carbohydr. Polym. 2019, 226, 115254. [Google Scholar] [CrossRef]
- Luo, L.; Cai, X.; He, C.; Xue, M.; Wu, X.; Cao, H. Immune response, stress resistance and bacterial challenge in juvenile rainbow trouts Oncorhynchus mykiss fed diets containing chitosan-oligosaccharides. Curr. Zool. 2009, 55, 416–422. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, Y.; Zhao, X.; Wang, H.; Wang, L.; Yuan, G.; Asim, M.; Wang, W.; Zeng, L.; Liu, X.; et al. Oligochitosan stimulated phagocytic activity of macrophages from blunt snout bream (Megalobrama amblycephala) associated with respiratory burst coupled with nitric oxide production. Dev. Comp. Immunol. 2014, 47, 17–24. [Google Scholar] [CrossRef]
- Fernández-Díaz, C.; Coste, O.; Malta, E. Polymer chitosan nanoparticles functionalized with Ulva ohnoi extracts boost in vitro ulvan immunostimulant effect in Solea senegalensis macrophages. Algal Res. 2017, 26, 135–142. [Google Scholar] [CrossRef]
- Wisdom, K.S.; Bhat, I.A.; Chanu, T.I.; Kumar, P.; Pathakota, G.-B.; Nayak, S.K.; Walke, P.; Sharma, R. Chitosan grafting onto single-walled carbon nanotubes increased their stability and reduced the toxicity in vivo (catfish) model. Int. J. Biol. Macromol. 2020, 155, 697–707. [Google Scholar] [CrossRef] [PubMed]
- Alishahi, A.; Mirvaghefi, A.; Tehrani, M.R.; Farahmand, H.; Koshio, S.; Dorkoosh, F.A.; Elsabee, M.Z. Chitosan nanoparticle to carry vitamin C through the gastrointestinal tract and induce the non-specific immunity system of rainbow trout (Oncorhynchus mykiss). Carbohydr. Polym. 2011, 86, 142–146. [Google Scholar] [CrossRef]
- Jiménez-Fernández, E.; Ruyra, A.; Roher, N.; Zuasti, E.; Infante, C.; Fernández-Díaz, C. Nanoparticles as a novel delivery system for vitamin C administration in aquaculture. Aquaculture 2014, 432, 426–433. [Google Scholar] [CrossRef]
- Bhat, I.A.; Nazir, M.I.; Ahmad, I.; Pathakota, G.-B.; Chanu, T.I.; Goswami, M.; Sundaray, J.K.; Sharma, R. Fabrication and characterization of chitosan conjugated eurycomanone nanoparticles: In vivo evaluation of the biodistribution and toxicity in fish. Int. J. Biol. Macromol. 2018, 112, 1093–1103. [Google Scholar] [CrossRef]
- Wisdom, K.S.; Bhat, I.A.; Kumar, P.; Pathan, M.K.; Chanu, T.I.; Walke, P.; Sharma, R. Fabrication of chitosan nanoparticles loaded with aromatase inhibitors for the advancement of gonadal development in Clarias magur (Hamilton, 1822). Aquaculture 2018, 497, 125–133. [Google Scholar] [CrossRef]
- Bhat, I.A.; Ahmad, I.; Mir, I.N.; Yousf, D.J.; Ganie, P.A.; Bhat, R.A.H.; Gireesh-Babu, P.; Sharma, R. Evaluation of the in vivo effect of chitosan conjugated eurycomanone nanoparticles on the reproductive response in female fish model. Aquaculture 2019, 510, 392–399. [Google Scholar] [CrossRef]
- Bhat, I.A.; Ahmad, I.; Mir, I.N.; Bhat, R.A.H.; P, G.-B.; Goswami, M.; Sundaray, J.K.; Sharma, R. Chitosan-eurycomanone nanoformulation acts on steroidogenesis pathway genes to increase the reproduction rate in fish. J. Steroid Biochem. Mol. Biol. 2019, 185, 237–247. [Google Scholar] [CrossRef]
- Barakat, K.M.; El-Sayed, H.S.; Gohar, Y.M. Protective effect of squilla chitosan–silver nanoparticles for Dicentrarchus labrax larvae infected with Vibrio anguillarum. Int. Aquat. Res. 2016, 8, 179–189. [Google Scholar] [CrossRef]
- Udayangani, R.M.C.; Dananjaya, S.H.S.; Nikapitiya, C.; Heo, G.-J.; Lee, J.; De Zoysa, M. Metagenomics analysis of gut microbiota and immune modulation in zebrafish (Danio rerio) fed chitosan silver nanocomposites. Fish Shellfish Immunol. 2017, 66, 173–184. [Google Scholar] [CrossRef]
- Xia, I.F.; Cheung, J.S.; Wu, M.; Wong, K.-S.; Kong, H.-K.; Zheng, X.-T.; Wong, K.-H.; Kwok, K.W. Dietary chitosan-selenium nanoparticle (CTS-SeNP) enhance immunity and disease resistance in zebrafish. Fish Shellfish Immunol. 2019, 87, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Victor, H.; Zhao, B.; Mu, Y.; Dai, X.; Wen, Z.; Gao, Y.; Chu, Z. Effects of Se-chitosan on the growth performance and intestinal health of the loach Paramisgurnus dabryanus (Sauvage). Aquaculture 2019, 498, 263–270. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, X.; Zhang, Y.; Zhu, W.; Zhou, Y.; Yuan, G.; Liu, X.; Ai, T.; Zeng, L.; Su, J. Chitosan and anisodamine improve the immune efficacy of inactivated infectious spleen and kidney necrosis virus vaccine in Siniperca chuatsi. Fish Shellfish Immunol. 2019, 89, 52–60. [Google Scholar] [CrossRef]
- Zhu, W.; Zhang, Y.; Zhang, J.; Yuan, G.; Liu, X.; Ai, T.; Su, J. Astragalus polysaccharides, chitosan and poly(I:C) obviously enhance inactivated Edwardsiella ictaluri vaccine potency in yellow catfish Pelteobagrus fulvidraco. Fish Shellfish Immunol. 2019, 87, 379–385. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, H.; Gao, Y.; Zhang, Y.; Wu, H.; Zhang, Y. Efficacy of chitosan oligosaccharide as aquatic adjuvant administrated with a formalin-inactivated Vibrio anguillarum vaccine. Fish Shellfish Immunol. 2015, 47, 855–860. [Google Scholar] [CrossRef]
- Wei, G.; Cai, S.; Wu, Y.; Ma, S.; Huang, Y. Immune effect of Vibrio harveyi formalin-killed cells vaccine combined with chitosan oligosaccharide and astragalus polysaccharides in ♀Epinephelus fuscoguttatus×♂Epinephelus lanceolatus. Fish Shellfish Immunol. 2020, 98, 186–192. [Google Scholar] [CrossRef]
- Halimi, M.; Alishahi, M.; Abbaspour, M.R.; Ghorbanpoor, M.; Tabandeh, M.R. Valuable method for production of oral vaccine by using alginate and chitosan against Lactococcus garvieae/Streptococcus iniae in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol. 2019, 90, 431–439. [Google Scholar] [CrossRef]
- Kole, S.; Qadiri, S.S.N.; Shin, S.-M.; Kim, W.-S.; Lee, J.; Jung, S.-J. Nanoencapsulation of inactivated-viral vaccine using chitosan nanoparticles: Evaluation of its protective efficacy and immune modulatory effects in olive flounder (Paralichthys olivaceus) against viral haemorrhagic septicaemia virus (VHSV) infection. Fish Shellfish Immunol. 2019, 91, 136–147. [Google Scholar] [CrossRef]
- Tandberg, J.; Lagos, L.; Ropstad, E.; Smistad, G.; Hiorth, M.; Winther-Larsen, H.C. The use of chitosan-coated membrane vesicles for immunization against salmonid rickettsial septicemia in an adult zebrafish model. Zebrafish 2018, 15, 372–381. [Google Scholar] [CrossRef]
- Dubey, S.; Avadhani, K.; Mutalik, S.; Sivadasan, S.; Maiti, B.; Girisha, S.; Venugopal, M.; Mutoloki, S.; Evensen, Ø.; Karunasagar, I.; et al. Edwardsiella tarda OmpA encapsulated in chitosan nanoparticles shows superior protection over inactivated whole cell vaccine in orally vaccinated fringed-lipped peninsula carp (Labeo fimbriatus). Vaccines 2016, 4, 40. [Google Scholar] [CrossRef]
- Wang, E.; Wang, X.; Wang, K.; He, J.; Zhu, L.; He, Y.; Chen, D.; Ouyang, P.; Geng, Y.; Huang, X.; et al. Preparation, characterization and evaluation of the immune effect of alginate/chitosan composite microspheres encapsulating recombinant protein of Streptococcus iniae designed for fish oral vaccination. Fish Shellfish Immunol. 2018, 73, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Hu, Y.; Zhou, J.; Hu, S.; Xiao, X.; Liu, X.; Su, J.; Yuan, G. Chitosan reduces the protective effects of IFN-γ2 on grass carp (Ctenopharyngodon idella) against Flavobacterium columnare infection due to excessive inflammation. Fish Shellfish Immunol. 2019, 95, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Maheshwari, D.; Philip, G.; Rana, R.; Bhatia, S.; Singh, M.; Gabrani, R.; Sharma, S.K.; Ali, J.; Sharma, R.K.; et al. Formulation and optimization of polymeric nanoparticles for intranasal delivery of lorazepam using Box-Behnken design: In vitro and in vivo evaluation. Biomed Res. Int. 2014, 2014, 156010. [Google Scholar] [CrossRef]
- Rather, M.A.; Bhat, I.A.; Gireesh-Babu, P.; Chaudhari, A.; Sundaray, J.K.; Sharma, R. Molecular characterization of kisspeptin gene and effect of nano-encapsulted kisspeptin-10 on reproductive maturation in Catla catla. Domest. Anim. Endocrinol. 2016, 56, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Yu, J.; Sun, X. Chitosan microspheres as candidate plasmid vaccine carrier for oral immunisation of Japanese flounder (Paralichthys olivaceus). Vet. Immunol. Immunopathol. 2008, 126, 220–229. [Google Scholar] [CrossRef]
- Vimal, S.; Taju, G.; Nambi, K.S.N.; Abdul Majeed, S.; Sarath Babu, V.; Ravi, M.; Sahul Hameed, A.S. Synthesis and characterization of CS/TPP nanoparticles for oral delivery of gene in fish. Aquaculture 2012, 358–359, 14–22. [Google Scholar] [CrossRef]
- Li, L.; Lin, S.-L.; Deng, L.; Liu, Z.-G. Potential use of chitosan nanoparticles for oral delivery of DNA vaccine in black seabream Acanthopagrus schlegelii Bleeker to protect from Vibrio parahaemolyticus. J. Fish Dis. 2013, 36, 987–995. [Google Scholar] [CrossRef]
- Vimal, S.; Abdul Majeed, S.; Nambi, K.S.N.; Madan, N.; Farook, M.A.; Venkatesan, C.; Taju, G.; Venu, S.; Subburaj, R.; Thirunavukkarasu, A.R.; et al. Delivery of DNA vaccine using chitosan–tripolyphosphate (CS/TPP) nanoparticles in Asian sea bass, Lates calcarifer (Bloch, 1790) for protection against nodavirus infection. Aquaculture 2014, 420–421, 240–246. [Google Scholar] [CrossRef]
- Zheng, F.; Liu, H.; Sun, X.; Zhang, Y.; Zhang, B.; Teng, Z.; Hou, Y.; Wang, B. Development of oral DNA vaccine based on chitosan nanoparticles for the immunization against reddish body iridovirus in turbots (Scophthalmus maximus). Aquaculture 2016, 452, 263–271. [Google Scholar] [CrossRef]
- Bhat, I.A.; Rather, M.A.; Saha, R.; Pathakota, G.-B.; Pavan-Kumar, A.; Sharma, R. Expression analysis of Sox9 genes during annual reproductive cycles in gonads and after nanodelivery of LHRH in Clarias batrachus. Res. Vet. Sci. 2016, 106, 100–106. [Google Scholar] [CrossRef]
- Valero, Y.; Awad, E.; Buonocore, F.; Arizcun, M.; Esteban, M.Á.; Meseguer, J.; Chaves-Pozo, E.; Cuesta, A. An oral chitosan DNA vaccine against nodavirus improves transcription of cell-mediated cytotoxicity and interferon genes in the European sea bass juveniles gut and survival upon infection. Dev. Comp. Immunol. 2016, 65, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Sáez, M.I.; Vizcaíno, A.J.; Alarcón, F.J.; Martínez, T.F. Comparison of lacZ reporter gene expression in gilthead sea bream (Sparus aurata) following oral or intramuscular administration of plasmid DNA in chitosan nanoparticles. Aquaculture 2017, 474, 1–10. [Google Scholar] [CrossRef]
- Rathor, P.K.; Bhat, I.A.; Rather, M.A.; Gireesh-Babu, P.; Kumar, K.; Purayil, S.B.P.; Sharma, R. Steroidogenic acute regulatory protein (StAR) gene expression construct: Development, nanodelivery and effect on reproduction in air-breathing catfish, Clarias batrachus. Int. J. Biol. Macromol. 2017, 104, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Ahmadivand, S.; Soltani, M.; Behdani, M.; Evensen, Ø.; Alirahimi, E.; Hassanzadeh, R.; Soltani, E. Oral DNA vaccines based on CS-TPP nanoparticles and alginate microparticles confer high protection against infectious pancreatic necrosis virus (IPNV) infection in trout. Dev. Comp. Immunol. 2017, 74, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Sáez, M.I.; Vizcaíno, A.J.; Alarcón, F.J.; Martínez, T.F. Feed pellets containing chitosan nanoparticles as plasmid DNA oral delivery system for fish: In vivo assessment in gilthead sea bream (Sparus aurata) juveniles. Fish Shellfish Immunol. 2018, 80, 458–466. [Google Scholar] [CrossRef]
- Kole, S.; Kumari, R.; Anand, D.; Kumar, S.; Sharma, R.; Tripathi, G.; Makesh, M.; Rajendran, K.V.; Bedekar, M.K. Nanoconjugation of bicistronic DNA vaccine against Edwardsiella tarda using chitosan nanoparticles: Evaluation of its protective efficacy and immune modulatory effects in Labeo rohita vaccinated by different delivery routes. Vaccine 2018, 36, 2155–2165. [Google Scholar] [CrossRef]
- Silva-Marrero, J.I.; Villasante, J.; Rashidpour, A.; Palma, M.; Fàbregas, A.; Almajano, M.P.; Viegas, I.; Jones, J.G.; Miñarro, M.; Ticó, J.R.; et al. The administration of chitosan-tripolyphosphate-DNA nanoparticles to express exogenous SREBP1a enhances conversion of dietary carbohydrates into lipids in the liver of Sparus aurata. Biomolecules 2019, 9, 297. [Google Scholar] [CrossRef]
- Rao, B.M.; Kole, S.; Gireesh-Babu, P.; Sharma, R.; Tripathi, G.; Bedekar, M.K. Evaluation of persistence, bio-distribution and environmental transmission of chitosan/PLGA/pDNA vaccine complex against Edwardsiella tarda in Labeo rohita. Aquaculture 2019, 500, 385–392. [Google Scholar] [CrossRef]
- Ramos, E.A.; Relucio, J.L.V.; Torres-Villanueva, C.A.T. Gene expression in tilapia following oral delivery of chitosan-encapsulated plasmid DNA incorporated into fish feeds. Mar. Biotechnol. 2005, 7, 89–94. [Google Scholar] [CrossRef]
- Rajesh Kumar, S.; Ishaq Ahmed, V.P.; Parameswaran, V.; Sudhakaran, R.; Sarath Babu, V.; Sahul Hameed, A.S. Potential use of chitosan nanoparticles for oral delivery of DNA vaccine in Asian sea bass (Lates calcarifer) to protect from Vibrio (Listonella) anguillarum. Fish Shellfish Immunol. 2008, 25, 47–56. [Google Scholar] [CrossRef]
- Kumari, R.; Gupta, S.; Singh, A.R.; Ferosekhan, S.; Kothari, D.C.; Pal, A.K.; Jadhao, S.B. Chitosan nanoencapsulated exogenous trypsin biomimics zymogen-like enzyme in fish gastrointestinal tract. PLoS ONE 2013, 8, e74743. [Google Scholar] [CrossRef] [PubMed]
- Naylor, R.L.; Hardy, R.W.; Bureau, D.P.; Chiu, A.; Elliott, M.; Farrell, A.P.; Forster, I.; Gatlin, D.M.; Goldburg, R.J.; Hua, K.; et al. Feeding aquaculture in an era of finite resources. Proc. Natl. Acad. Sci. USA 2009, 106, 15103–15110. [Google Scholar] [CrossRef] [PubMed]
- Polakof, S.; Panserat, S.; Soengas, J.L.; Moon, T.W. Glucose metabolism in fish: A review. J. Comp. Physiol. B. 2012, 182, 1015–1045. [Google Scholar] [CrossRef]
- Rashidpour, A.; Silva-Marrero, J.I.; Seguí, L.; Baanante, I.V.; Metón, I. Metformin counteracts glucose-dependent lipogenesis and impairs transdeamination in the liver of gilthead sea bream (Sparus aurata). Am. J. Physiol. Integr. Comp. Physiol. 2019, 316, R265–R273. [Google Scholar] [CrossRef]
- Metón, I.; Egea, M.; Anemaet, I.G.; Fernández, F.; Baanante, I.V. Sterol regulatory element binding protein-1a transactivates 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase gene promoter. Endocrinology 2006, 147, 3446–3456. [Google Scholar] [CrossRef]
- Egea, M.; Metón, I.; Córdoba, M.; Fernández, F.; Baanante, I.V. Role of Sp1 and SREBP-1a in the insulin-mediated regulation of glucokinase transcription in the liver of gilthead sea bream (Sparus aurata). Gen. Comp. Endocrinol. 2008, 155, 359–367. [Google Scholar] [CrossRef]


| Preloading Diameter (nm) | Postloading Diameter (nm) | Preloading Zeta Potential (mV) | Postloading Zeta Potential (mV) | Encapsulation Efficiency | References |
|---|---|---|---|---|---|
| - | <10,000 | - | - | 94.5% | [145] |
| 30–60 | - | - | - | - | [146] |
| - | 200 | - | - | 91.5% | [147] |
| - | - | - | - | 83.6% | [148] |
| 193 ± 53 1 | 246 ± 74 1 | 32.0 ± 1.0 1 | 14.4 ± 1.3 1 | - | [80] |
| - | 146 ± 2 2 | - | 24.3 ± 0.5 2 | 92.8% ± 1.4% 2 | [149] |
| - | 133 | - | 34.3 | 63% | [150] |
| - | 50-200 | - | - | 97.5% | [151] |
| 87 | 156 | 30.3 | 36.5 | 60% | [144] |
| - | 743 | - | - | 98.6% | [152] |
| 135 | - | 26.7 | - | 86% | [153] |
| - | - | - | - | 84.2% | [154] |
| 224 ± 62 1 | Similar to preloading diameter | 33.0 ± 1.2 1 | 14.4 ± 1.3 1 | - | [81] |
| - | 750–950 | - | - | 98.6% | [155] |
| 116 | 306 | 24.7 | 18.0 | - | [156] |
| 231 ± 18 2 | 272 ± 36 2 | 31.2 ± 1.5 2 | 14.1 ± 2.3 2 | - | [157] |
| - | 267 | - | 27.1 | 87.4% | [158] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Y.; Rashidpour, A.; Almajano, M.P.; Metón, I. Chitosan-Based Drug Delivery System: Applications in Fish Biotechnology. Polymers 2020, 12, 1177. https://doi.org/10.3390/polym12051177
Wu Y, Rashidpour A, Almajano MP, Metón I. Chitosan-Based Drug Delivery System: Applications in Fish Biotechnology. Polymers. 2020; 12(5):1177. https://doi.org/10.3390/polym12051177
Chicago/Turabian StyleWu, Yuanbing, Ania Rashidpour, María Pilar Almajano, and Isidoro Metón. 2020. "Chitosan-Based Drug Delivery System: Applications in Fish Biotechnology" Polymers 12, no. 5: 1177. https://doi.org/10.3390/polym12051177
APA StyleWu, Y., Rashidpour, A., Almajano, M. P., & Metón, I. (2020). Chitosan-Based Drug Delivery System: Applications in Fish Biotechnology. Polymers, 12(5), 1177. https://doi.org/10.3390/polym12051177






