Fabrication of Poly(ethylene glycol) Capsules via Emulsion Templating Method for Targeted Drug Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
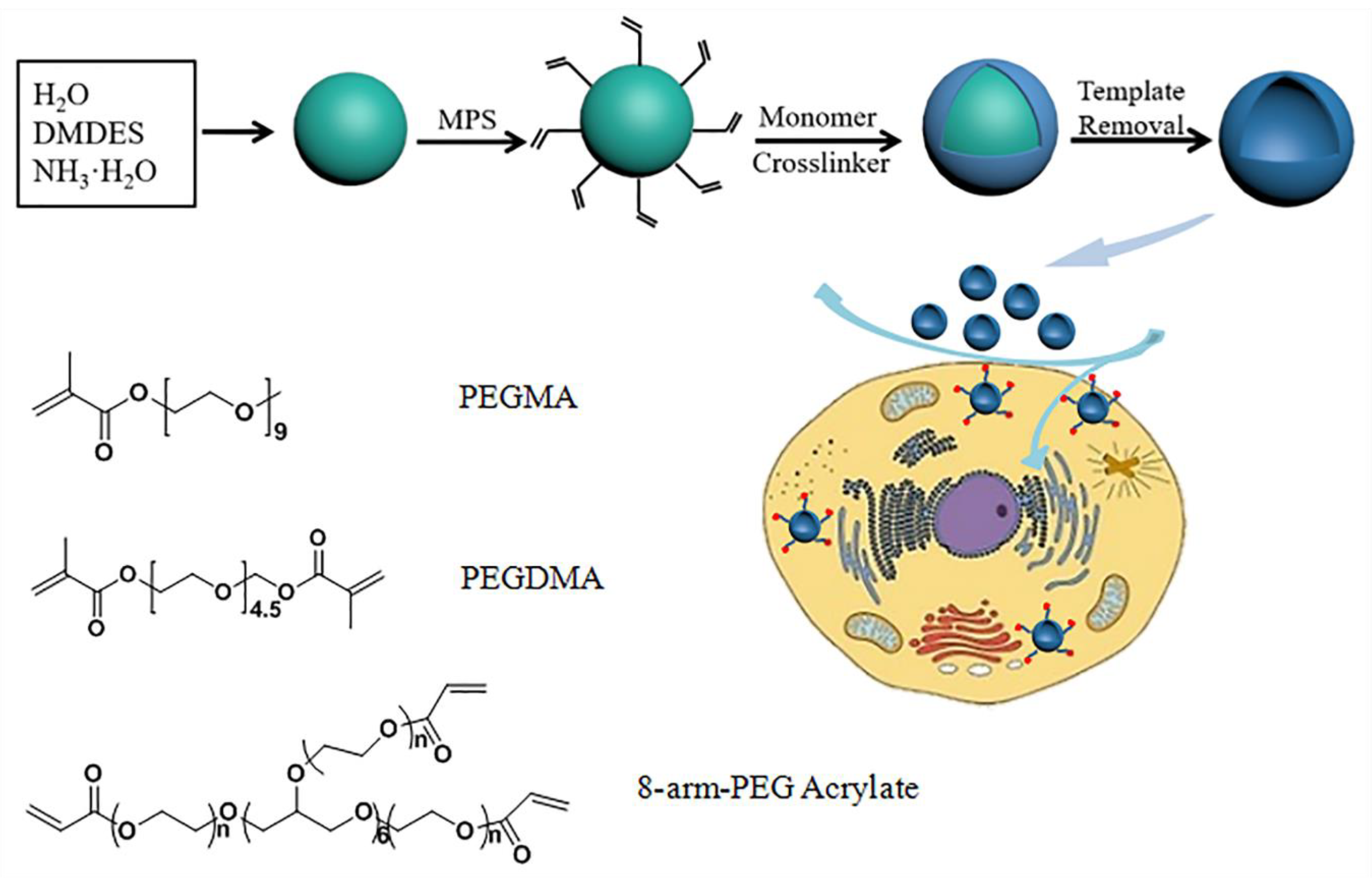
2.2. Preparation of the DMDES Emulsion Templates
2.3. Preparation of the Capsules
2.4. Preparation of the Targeting Capsules
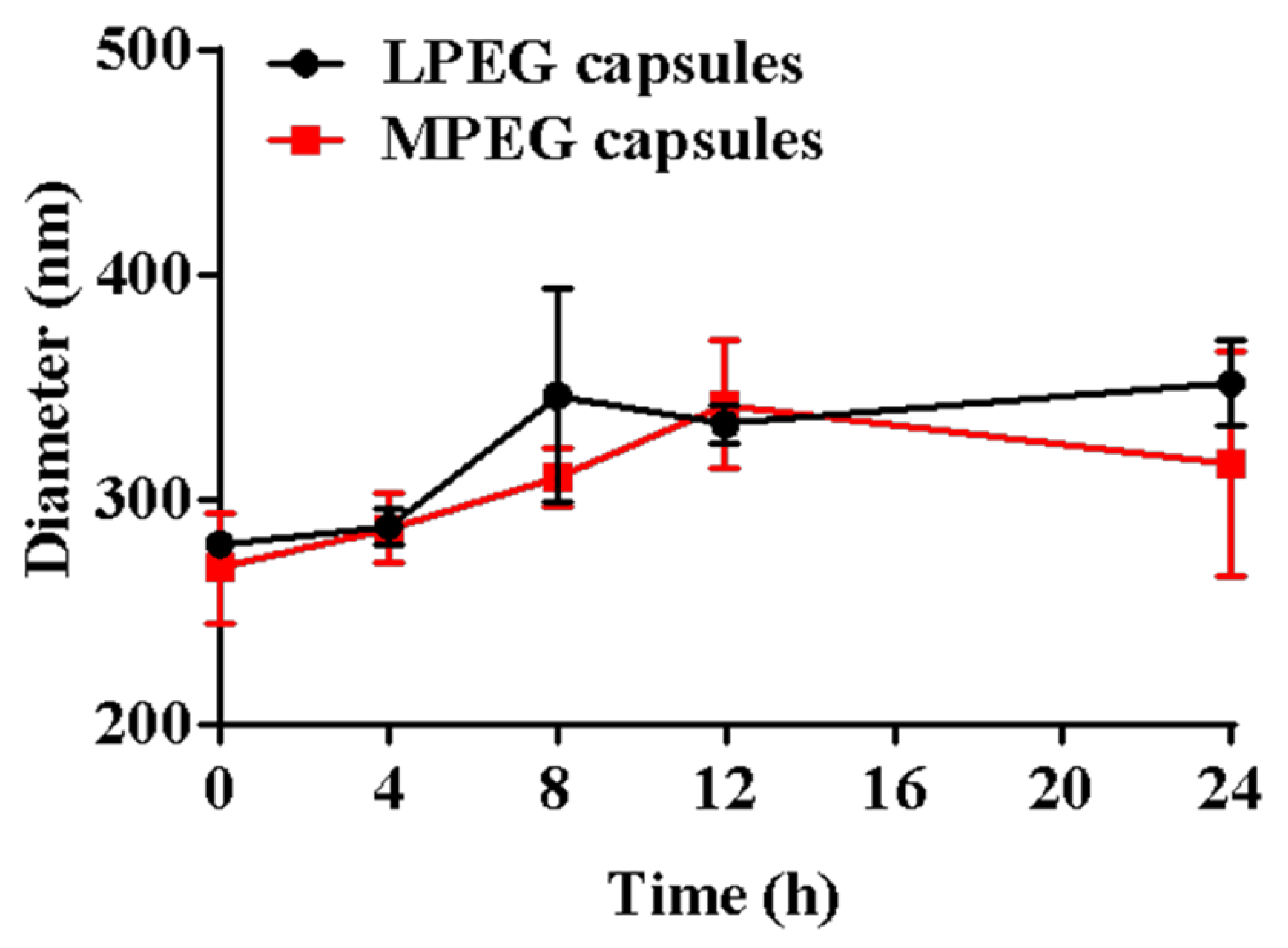
2.5. Detection of the Stability of Capsules
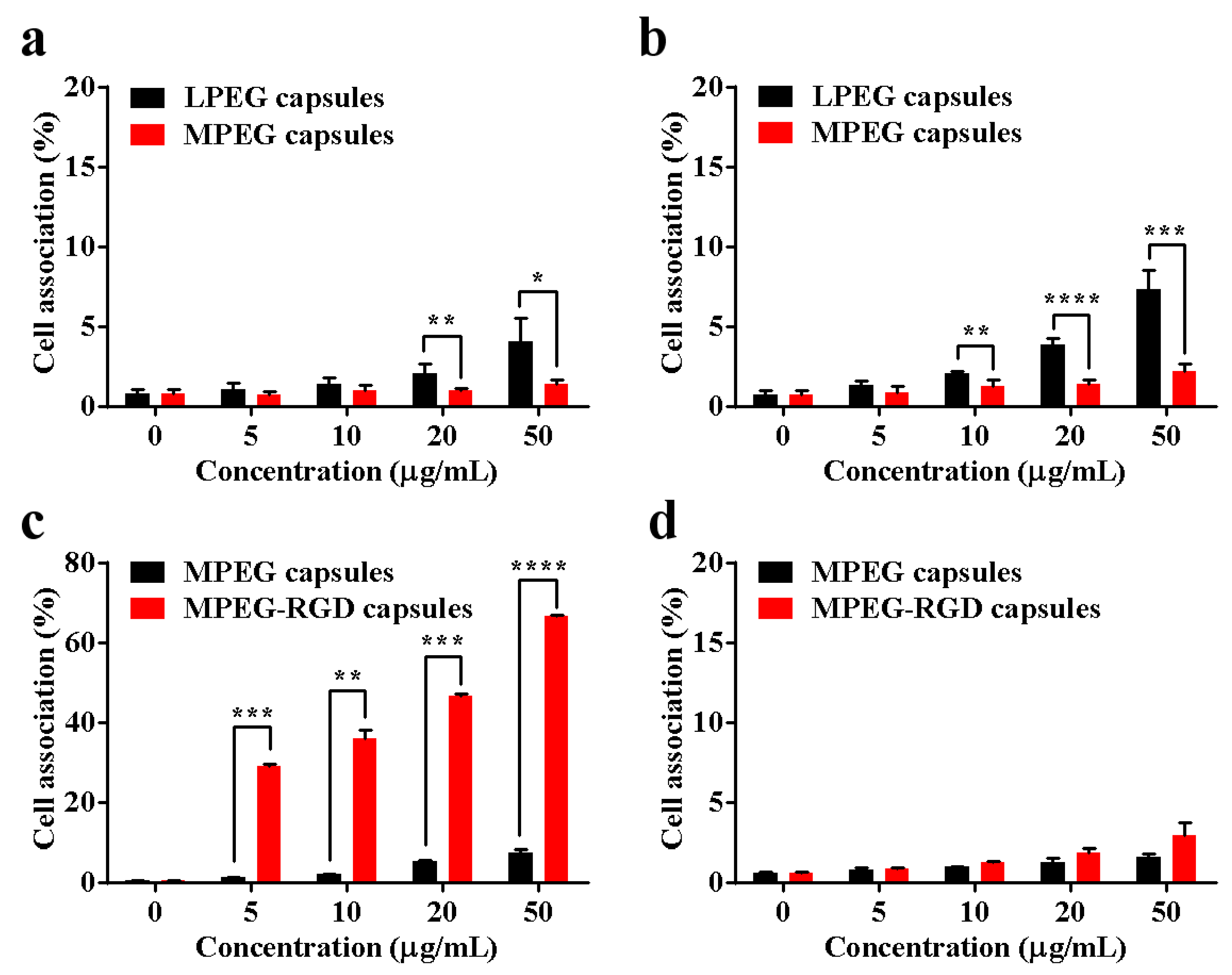
2.6. Cellular Association
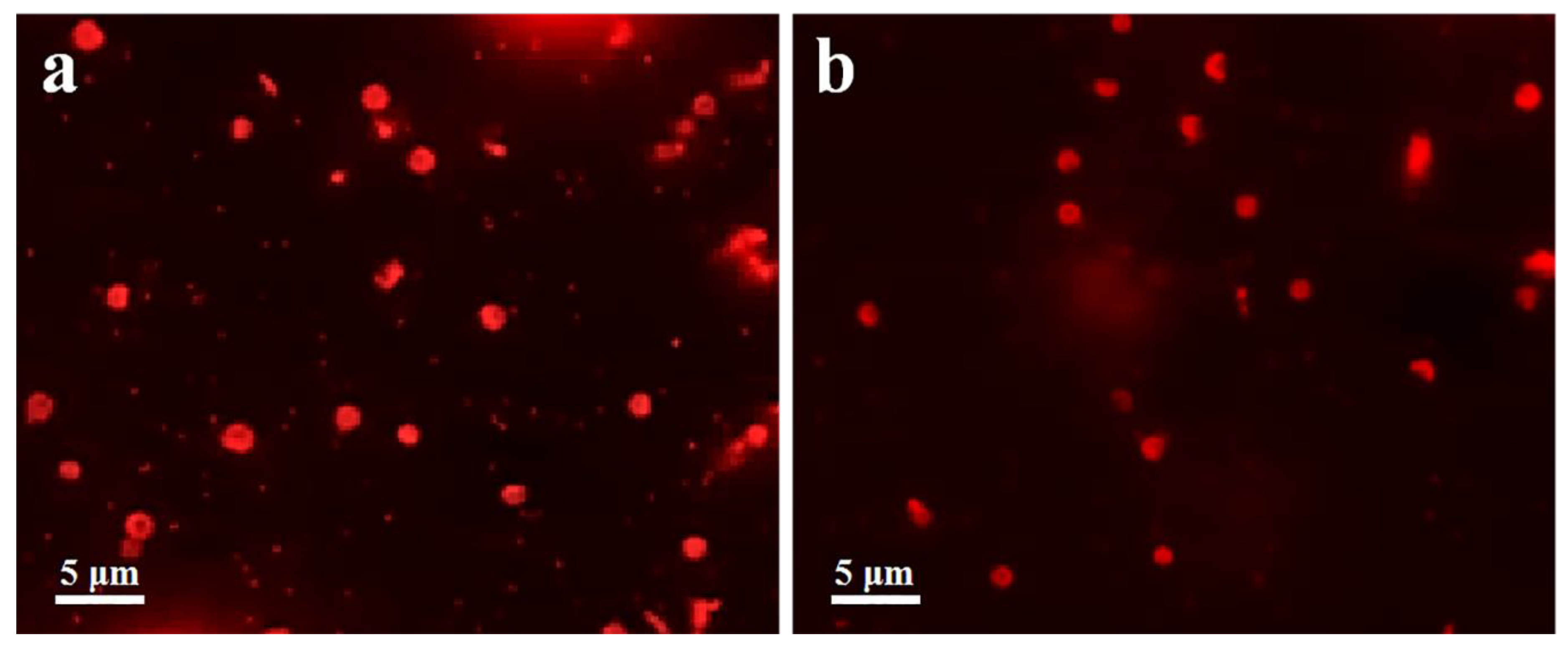
2.7. Capsules Targeting Analysis
2.8. DOX Loading
2.9. Cytotoxicity Assessment
2.10. Cellular Uptake of Capsules
2.11. Characterization Methods
3. Results and Discussion
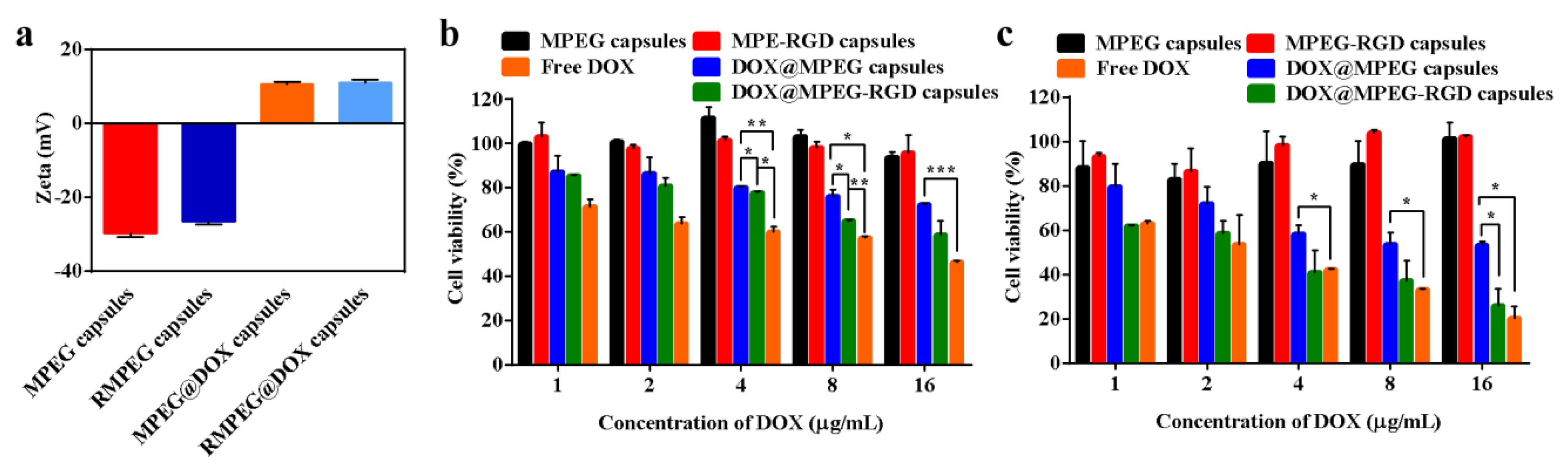
3.1. Synthesis and Characterization of Capsules
3.2. Detection of the Stability of Capsules
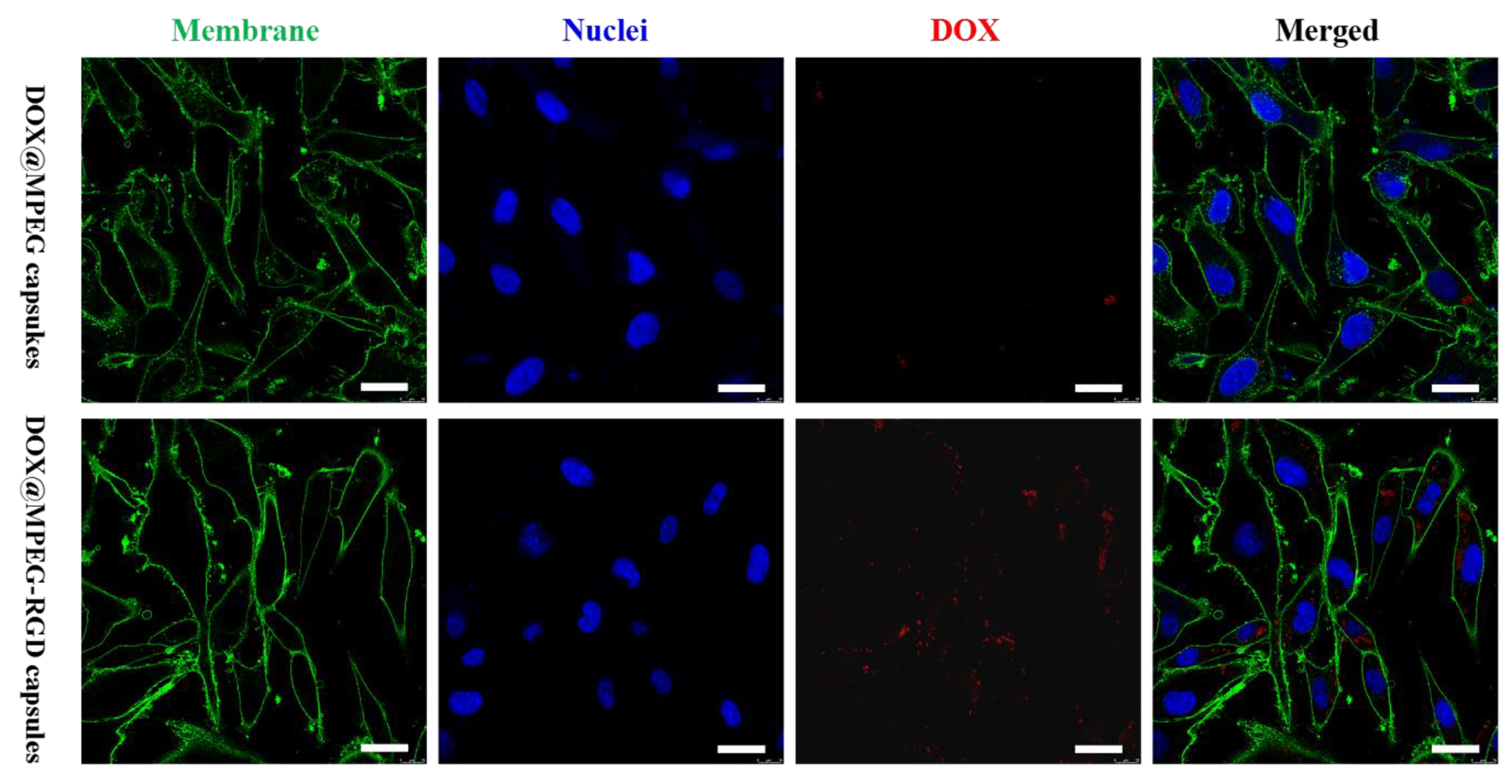
3.3. Cell Association of Capsules
3.4. DOX Loading
3.5. In Vitro Cytotoxicity Assessment of Capsules
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tyagi, P.; Santos, J.L. Macromolecule nanotherapeutics: Approaches and challenges. Drug Discov. Today 2018, 23, 1053–1061. [Google Scholar] [CrossRef]
- Biswas, A.; Shukla, A.; Maiti, P. Biomaterials for interfacing cell imaging and drug delivery: An overview. Langmuir 2019, 35, 12285–12305. [Google Scholar] [CrossRef] [PubMed]
- Walkey, C.D.; Olsen, J.B.; Guo, H.; Emili, A.; Chan, W.C. Nanoparticle size and surface chemistry determine serum protein adsorption and macrophage uptake. J. Am. Chem. Soc. 2012, 134, 2139–2147. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Xiao, Y.; Yin, Y.; Li, G.; Peng, J. Erythrocyte membrane-camouflaged IR780 and DTX coloading polymeric nanoparticles for imaging-guided cancer photo-chemo combination therapy. Mol. Pharm. 2019, 16, 3208–3220. [Google Scholar] [CrossRef]
- Kroll, A.V.; Fang, R.H.; Zhang, L. Biointerfacing and applications of cell membrane-coated nanoparticles. Bioconjugate Chem. 2017, 28, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Li, L.; Zhao, C.; Zheng, J. Surface hydration: Principles and applications toward low-fouling/nonfouling biomaterials. Polymer 2010, 51, 5283–5293. [Google Scholar] [CrossRef]
- Ayyavoo, J.; Nguyen, T.; Jun, B.; Kim, I.; Kwon, Y. Protection of polymeric membranes with antifouling surfacing via surface modifications. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 190–201. [Google Scholar] [CrossRef]
- Otsuka, H.; Nagasaki, Y.; Kataoka, K. Characterization of aldehyde-PEG tethered surfaces: Influence of PEG chain length on the specific biorecognition. Langmuir 2004, 20, 11285–11287. [Google Scholar] [CrossRef]
- Zhang, P.; Gao, Z.; Cui, J.; Hao, J. Dual-stimuli-responsive polypeptide nanoparticles for photothermal and photodynamic therapy. ACS Appl. Bio Mater. 2020, 3, 561–569. [Google Scholar] [CrossRef]
- Barraza, L.F.; Jimenez, V.A.; Alderete, J.B. Methotrexate complexation with native and PEGylated PAMAM-G4: Effect of the PEGylation degree on the drug loading capacity and release kinetics. Macromol. Chem. Phys. 2016, 217, 605–613. [Google Scholar] [CrossRef]
- Miteva, M.; Kirkbride, K.C.; Kilchrist, K.V.; Werfel, T.A.; Li, H.; Nelson, C.E.; Gupta, M.K.; Giorgio, T.D.; Duvall, C.L. Tuning PEGylation of mixed micelles to overcome intracellular and systemic siRNA delivery barriers. Biomaterials 2015, 38, 97–107. [Google Scholar] [CrossRef]
- Sousa, S.F.; Peres, J.; Coelho, M.; Vieira, T.F. Analyzing PEGylation through molecular dynamics simulations. ChemistrySelect 2018, 3, 8415–8427. [Google Scholar] [CrossRef]
- Ekladious, I.; Colson, Y.L.; Grinstaff, M.W. Polymer-drug conjugate therapeutics: Advances, insights and prospects. Nat. Rev. Drug Discovery 2019, 18, 273–294. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; De Rose, R.; Alt, K.; Alcantara, S.; Paterson, B.M.; Liang, K.; Hu, M.; Richardson, J.J.; Yan, Y.; Jeffery, C.M.; et al. Engineering poly(ethylene glycol) particles for improved biodistribution. ACS Nano 2015, 9, 1571–1580. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Björnmalm, M.; Ju, Y.; Caruso, F. Nanoengineering of poly(ethylene glycol) particles for stealth and targeting. Langmuir 2018, 34, 10817–10827. [Google Scholar] [CrossRef]
- Dai, Q.; Yan, Y.; Ang, C.-S.; Kempe, K.; Kamphuis, M.M.; Dodds, S.J.; Caruso, F. Monoclonal antibody-functionalized multilayered particles: Targeting cancer cells in the presence of protein coronas. ACS Nano 2015, 9, 2876–2885. [Google Scholar] [CrossRef]
- Hanaoka, H.; Nagaya, T.; Sato, K.; Nakamura, Y.; Watanabe, R.; Harada, T.; Gao, W.; Feng, M.; Phung, Y.; Kim, I. Glypican-3 targeted human heavy chain antibody as a drug carrier for hepatocellular carcinoma therapy. Mol. Pharm. 2015, 12, 2151–2157. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Deng, H.; Liu, Q.; Chu, L.; Zhang, Y.; Yang, C.; Zhao, X.; Huang, P.; Deng, L.; Dong, A. Integrin-targeted pH-responsive micelles for enhanced efficiency of anticancer treatment in vitro and in vivo. Nanoscale 2015, 7, 4451–4460. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Tao, L.; Li, H.; Zhao, S.; Zhang, Y.; Yang, S.; Xue, J.; Zhang, X. Hollow mesoporous carbon modified with cRGD peptide nanoplatform for targeted drug delivery and chemo-photothermal therapy of prostatic carcinoma. Colloids Surf. A Physicochem. Eng. Asp. 2019, 570, 386–395. [Google Scholar] [CrossRef]
- Su, G.; Jiang, H.; Xu, B.; Yu, Y.; Chen, X. Effects of protein corona on active and passive targeting of cyclic RGD peptide-functionalized PEGylation nanoparticles. Mol. Pharm. 2018, 15, 5019–5030. [Google Scholar] [CrossRef]
- Liu, Y.; Goebl, J.; Yin, Y. Templated synthesis of nanostructured materials. Chem. Soc. Rev. 2013, 42, 2610–2653. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Garamus, V.; Li, N.; Gong, Y.; Zhe, Z.; Tian, Z.; Zhou, A. Preparation and characterization of a pH-responsive mesoporous silica nanoparticle dual-modified with biopolymers. Colloids Surf. A Physicochem. Eng. Asp. 2018, 548, 61–69. [Google Scholar] [CrossRef]
- Wang, X.; Ma, Y.; Chen, H.; Wu, X.; Qian, H.; Yang, X.; Zha, Z. Novel doxorubicin loaded PEGylated cuprous telluride nanocrystals for combined photothermal-chemo cancer treatment. Colloids Surf. B 2017, 152, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Feng, L.; Zhu, W.; Sun, X.; Gao, M.; Zhao, H.; Chao, Y.; Liu, Z. CaCO3 nanoparticles as an ultra-sensitive tumor-pH-responsive nanoplatform enabling real-time drug release monitoring and cancer combination therapy. Biomaterials 2016, 110, 60–70. [Google Scholar] [CrossRef]
- Song, X.; Yang, F.; Li, L.; Zhu, A. Size-controlled/surface-functionalized polystyrene nanospheres with good biocompatibility and high encapsulation efficiency of cyclosporin A via miniemulsion polymerization in one step. Chinese. J. Chem. 2016, 34, 720–726. [Google Scholar] [CrossRef]
- Wang, Z.; Gao, H.; Zhang, Y.; Liu, G.; Niu, G.; Chen, X. Functional ferritin nanoparticles for biomedical applications. Front. Chem. Sci. Eng. 2017, 11, 633–646. [Google Scholar] [CrossRef]
- Mason, T.G.; Wilking, J.N.; Meleson, K.; Chang, C.B.; Graves, S.M. Nanoemulsions: Formation, structure, and physical properties. J. Phys.: Condens. Matter 2006, 18, 635–666. [Google Scholar] [CrossRef]
- Zoldesi, C.I.; Imhof, A. Synthesis of monodisperse colloidal spheres, capsules, and microballoons by emulsion templating. Adv. Mater. 2005, 17, 924–928. [Google Scholar] [CrossRef]
- Cui, J.; Wang, Y.; Postma, A.; Hao, J.; Hosta-Rigau, L.; Caruso, F. Monodisperse polymer capsules: Tailoring size, shell thickness, and hydrophobic cargo loading via emulsion templating. Adv. Funct. Mater. 2010, 20, 1625–1631. [Google Scholar] [CrossRef]
- Bian, S.; Zheng, J.; Tang, X.; Yi, D.; Wang, Y.; Yang, W. One-pot synthesis of redox-labile polymer capsules via emulsion droplet-mediated precipitation polymerization. Chem. Mater. 2015, 27, 1262–1268. [Google Scholar] [CrossRef]
- Pham, T.D.; Tran, T.T.; Le, V.A.; Pham, T.T.; Dao, T.H.; Le, T.S. Adsorption characteristics of molecular oxytetracycline onto alumina particles: The role of surface modification with an anionic surfactant. J. Mol. Liq. 2019, 287, 110900. [Google Scholar] [CrossRef]
- Lowe, A.B. Thiol-ene “click” reactions and recent applications in polymer and materials synthesis. Polym. Chem. 2010, 1, 17–36. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, F.; Ren, C.; Yang, L.; Liu, J.; Cheng, Z.; Chu, L.; Liu, J. Targeted chemo-photodynamic combination platform based on the DOX prodrug nanoparticles for enhanced cancer therapy. ACS Appl. Mater. Interfaces 2017, 9, 13016–13028. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.D.; Do, T.U.; Pham, T.T.; Nguyen, T.A.H.; Nguyen, T.K.T.; Vu, N.D.; Le, T.S.; Vu, C.M.; Kobayashi, M. Adsorption of poly(styrenesulfonate) onto different-sized alumina particles: Characteristics and mechanisms. Colloid Polym. Sci. 2019, 297, 13–22. [Google Scholar] [CrossRef]

| Diameter of Emulsion Template (nm) | SDS Concentration (mg/mL) | NH3 Concentration (v/v) | DMDES Concentration (v/v) | Time (h) |
|---|---|---|---|---|
| 2000 | 0 | 2% | 2% | 24 |
| 1200 | 0 | 2% | 1% | 24 |
| 450 | 0.6 | 2% | 2% | 16 |
| 230 | 0.6 | 1% | 1% | 16 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, S.; Ding, F.; Gao, Z.; Guo, J.; Cui, J.; Zhang, P. Fabrication of Poly(ethylene glycol) Capsules via Emulsion Templating Method for Targeted Drug Delivery. Polymers 2020, 12, 1124. https://doi.org/10.3390/polym12051124
Yang S, Ding F, Gao Z, Guo J, Cui J, Zhang P. Fabrication of Poly(ethylene glycol) Capsules via Emulsion Templating Method for Targeted Drug Delivery. Polymers. 2020; 12(5):1124. https://doi.org/10.3390/polym12051124
Chicago/Turabian StyleYang, Shuang, Feng Ding, Zhiliang Gao, Jianman Guo, Jiwei Cui, and Peiyu Zhang. 2020. "Fabrication of Poly(ethylene glycol) Capsules via Emulsion Templating Method for Targeted Drug Delivery" Polymers 12, no. 5: 1124. https://doi.org/10.3390/polym12051124
APA StyleYang, S., Ding, F., Gao, Z., Guo, J., Cui, J., & Zhang, P. (2020). Fabrication of Poly(ethylene glycol) Capsules via Emulsion Templating Method for Targeted Drug Delivery. Polymers, 12(5), 1124. https://doi.org/10.3390/polym12051124





