Physical and Antioxidant Properties of Cassava Starch–Carboxymethyl Cellulose Incorporated with Quercetin and TBHQ as Active Food Packaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Film Preparation
2.3. Mechanical Properties
2.4. Fourier Transform Infrared Spectroscopy (FT-IR)
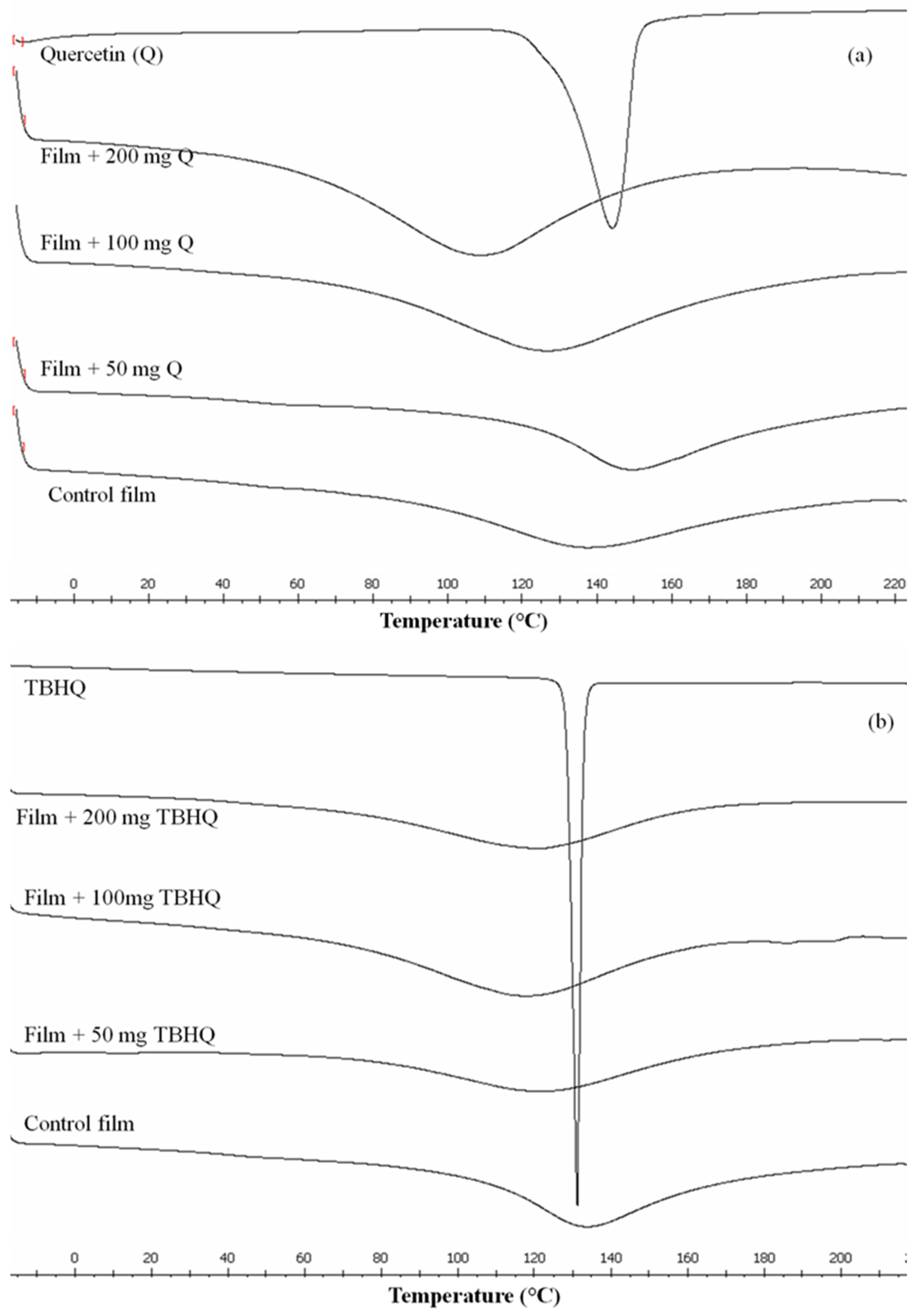
2.5. Differential Scanning Calorimetry (DSC)
2.6. X-ray Diffraction (XRD)
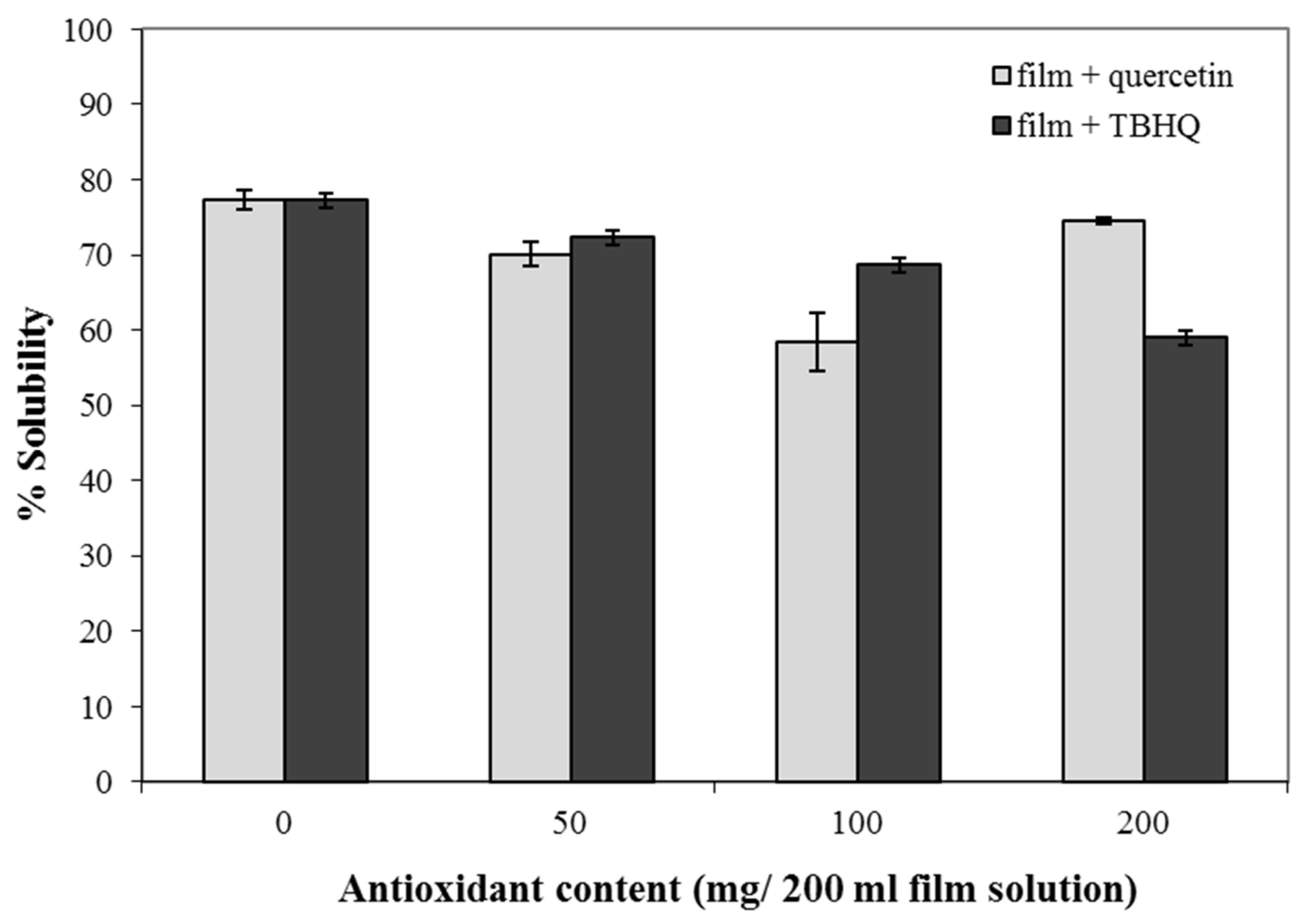
2.7. Water Solubility of Composite Films
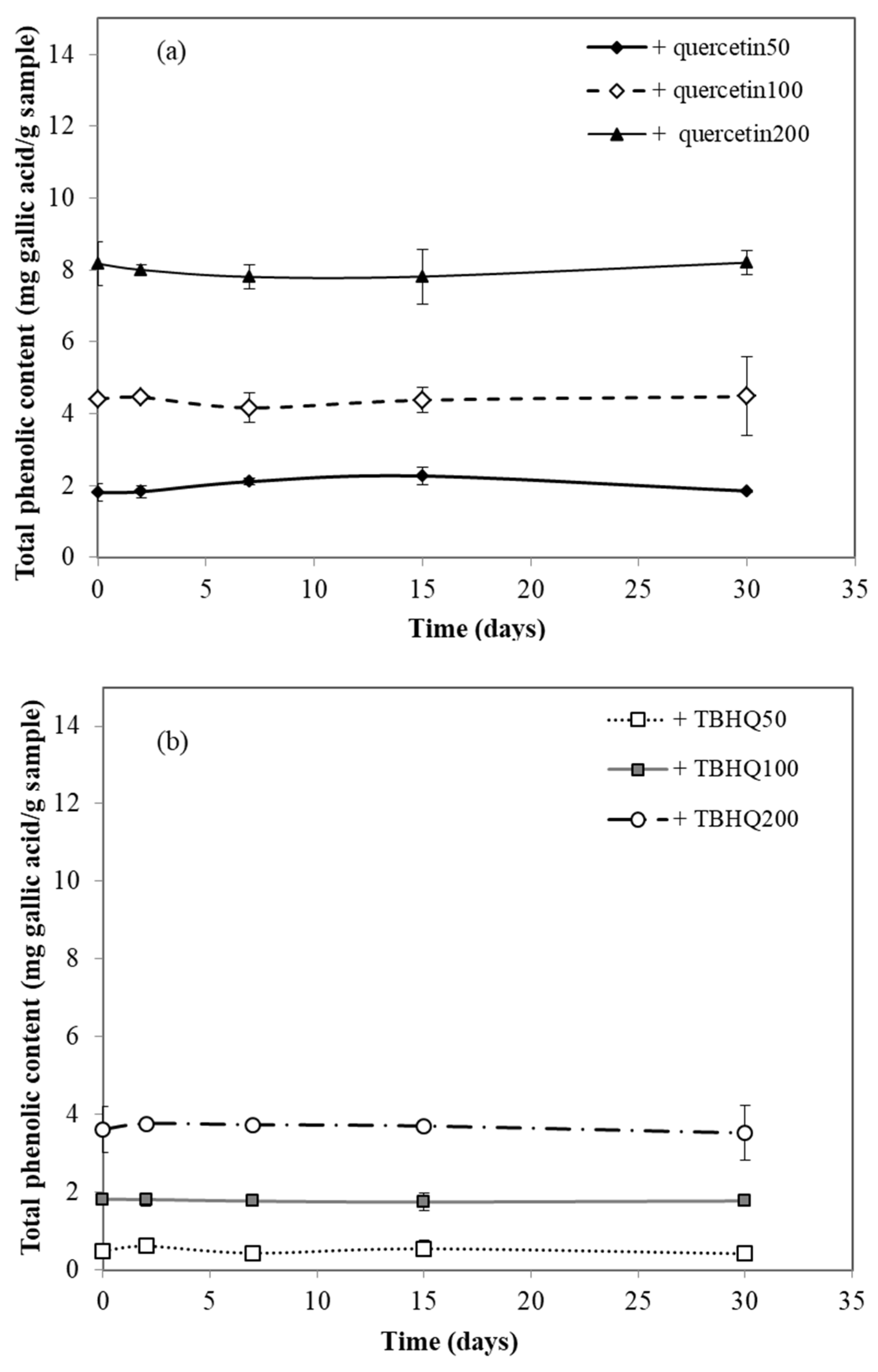
2.8. Total Phenolic Assay
2.9. Determination of Antioxidant Activity in the Composite Films
2.10. Application of Antioxidant Films on Lard
2.10.1. Effect of Antioxidant Incorporations into Cassava Starch–CMC Films on Lard Storage
2.10.2. Estimation of Peroxide Value
2.11. Application of Antioxidant Films on Fresh Pork
2.12. Statistical Analysis
3. Results and Discussion
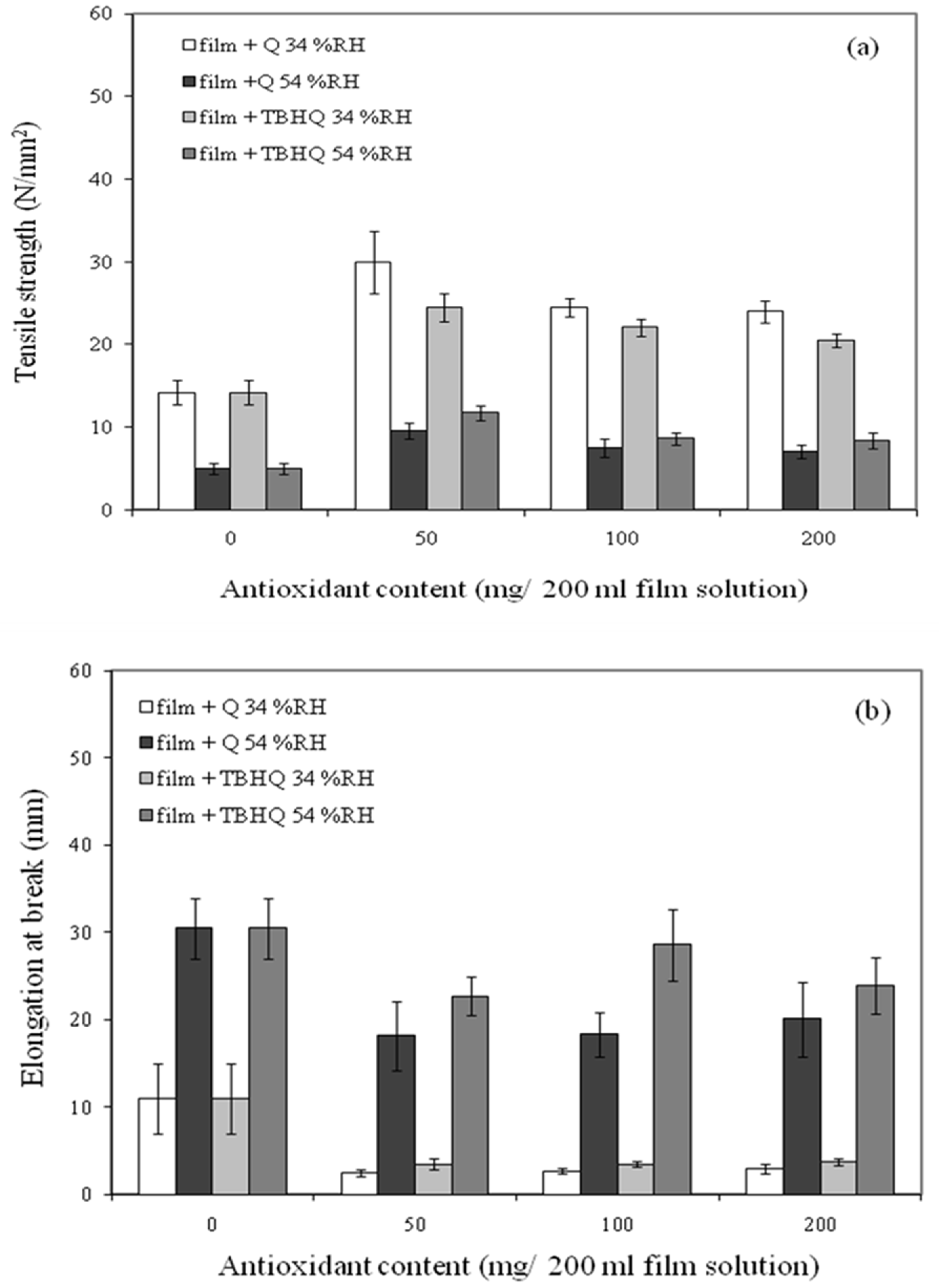
3.1. Influence of Antioxidants Concentrations on Mechanical Properties of the Composite Films
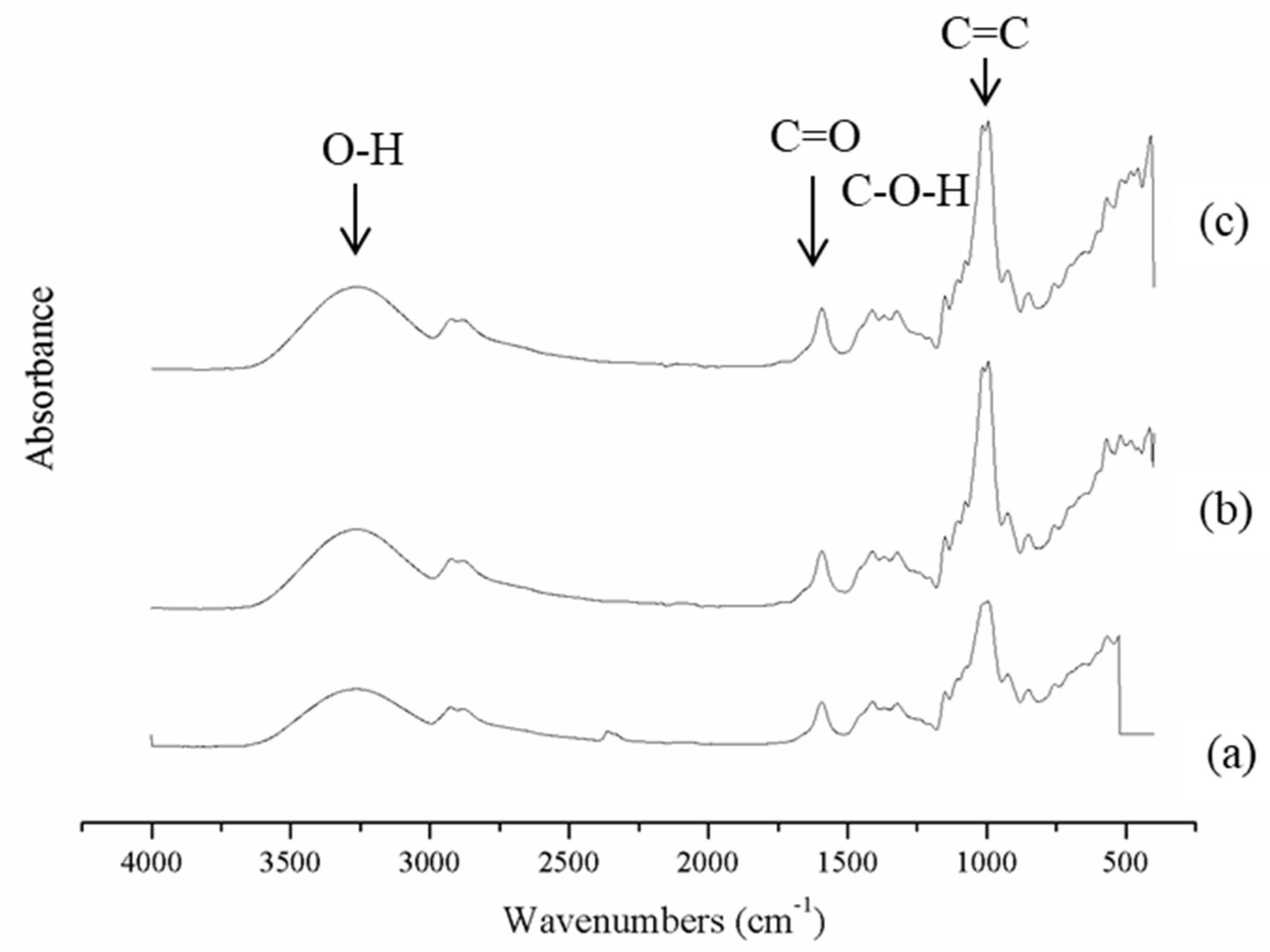
3.2. Fourier Transform Infrared Spectroscopy (FT-IR)
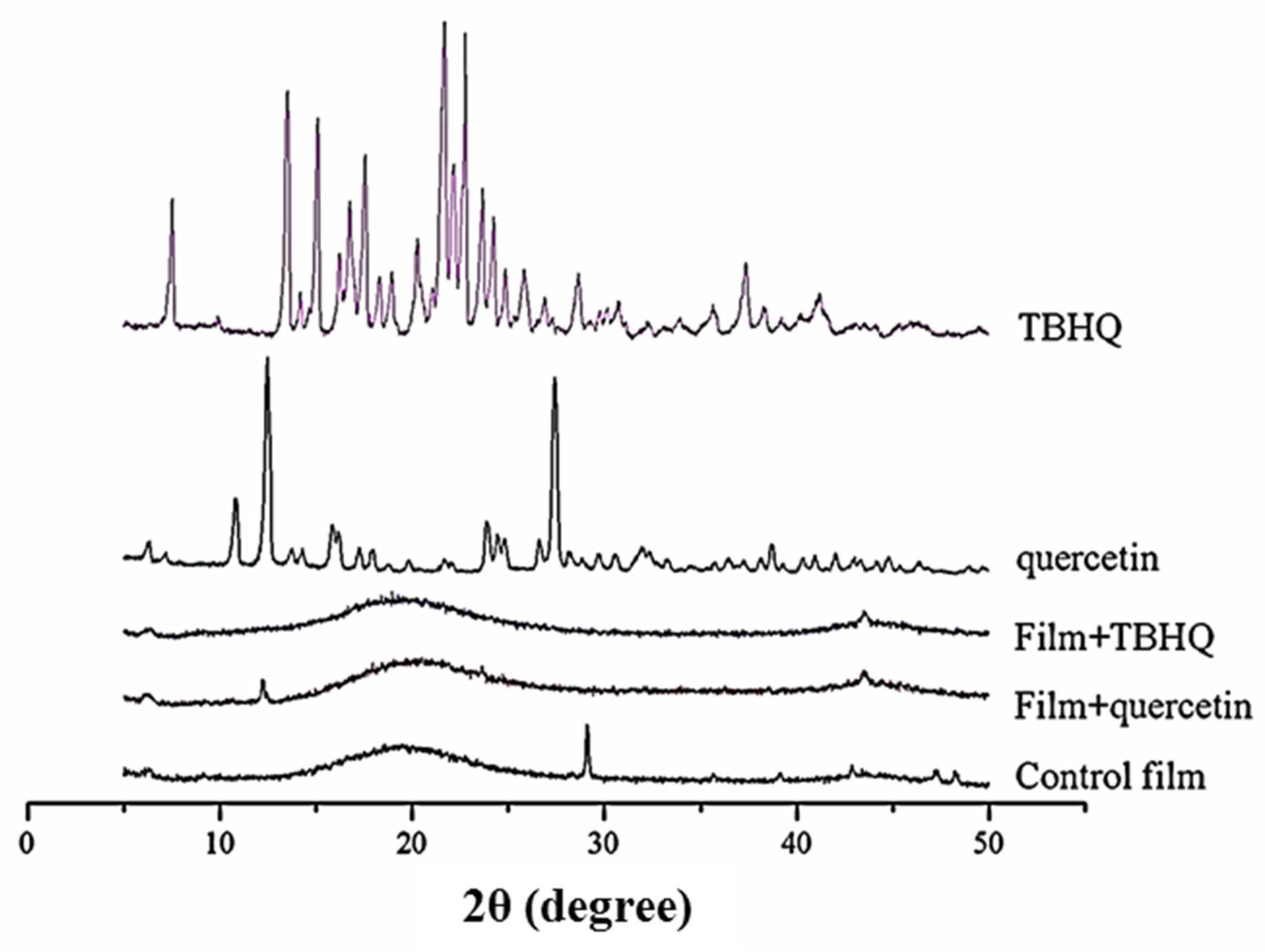
3.3. X-ray Diffraction Patterns
3.4. Thermal Properties of the Composite Films
3.5. Water Solubility of Composite Films
3.6. Total Phenolic Content Assay
3.7. Determination of Antioxidant Activity in the Composite Films
3.8. Effect of Antioxidants Incorporated into Cassava Starch–CMC Films on Lard Storage
3.9. Effect of Antioxidants Incorporated into Cassava Starch–CMC Films on Discoloration of Pork
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arvanitoyannis, I.; Biliaderis, C.G. Physical properties of polyol-plasticized edible blends made of methyl cellulose and soluble starch. Carbohydr. Polym. 1999, 38, 47–58. [Google Scholar] [CrossRef]
- Biliaderis, C.G.; Lazaridou, A.; Arvanitoyannis, I. Glass transition and physical properties of polyol-plasticised pullulan-starch blends at low moisture. Carbohydr. Polym. 1999, 40, 29–47. [Google Scholar] [CrossRef]
- Lazaridou, A.; Biliaderis, C.G. Thermophysical properties of chitosan, chitosan-starch and chitosan-pullulan films near the glass transition. Carbohydr. Polym. 2002, 48, 179–190. [Google Scholar] [CrossRef]
- Li, Y.; Shoemaker, C.F.; Ma, J.; Shen, X.; Zhong, F. Paste viscosity of rice starches of different amylose content and carboxymethylcellulose formed by dry heating and the physical properties of their films. Food Chem. 2008, 109, 616–623. [Google Scholar] [CrossRef]
- Yanishlieva-Maslarova, N.V. Inhibition oxidation. In Antioxidants in Food Practical Applications; Pokorny, J., Yanishlieva, N., Gordon, M., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2001; pp. 22–70. [Google Scholar]
- Eastman Chemical Company. Tenox TBHQ Antioxidant versus BHT in Fats and Oils. Food Thought 1995, 1–4. [Google Scholar]
- Phoopuritham, P.; Thongngam, M.; Yoksan, R.; Suppakul, P. Properties of antioxidant cellulose ether films containing selected plant extracts. In Proceedings of the 5th International Packaging Congress & Exhibition, Izmir, Turkey, 22–24 November 2007. [Google Scholar]
- Pascat, B. Study of some factors affecting permeability. In Food Packaging and Preservation: Theory and Practice; Mathlouthi, M., Ed.; Elsevier: London, UK, 1986; pp. 7–24. [Google Scholar]
- Lopez-de-Dicastillo, C.; Hernandez-Munoz, P.; Gavara, R.; Catala, R. Development of packaging films containing natural antioxidants. Characterization of their activity on stimulant and real foods. In Proceedings of the 5th International Packaging Congress & Exhibition, Izmir, Turkey, 20–23 November 2007; pp. 213–222. [Google Scholar]
- Poojary, M.M.; Putnik, P.; Bursać Kovačević, D.; Barba, F.J.; Lorenzo, J.M.; Dias, D.A.; Shpigelman, A. Stability and extraction of bioactive sulfur compounds from Allium genus processed by traditional and innovative technologies. J. Food Compos. Anal. 2017, 61, 28–39. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Pateiro, M.; Domínguez, R.; Barba, F.J.; Putnik, P.; Kovačević, D.B.; Shpigelman, A.; Granato, D.; Franco, D. Berries extracts as natural antioxidants in meat products: A review. Food Res. Int. 2018, 106, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, R.; Barba, F.J.; Gómez, B.; Putnik, P.; Bursać Kovačević, D.; Pateiro, M.; Santos, E.M.; Lorenzo, J.M. Active packaging films with natural antioxidants to be used in meat industry: A review. Food Res. Int. 2018, 113, 93–101. [Google Scholar] [CrossRef]
- Crouvisier-Urion, K.; Bodart, P.R.; Winckler, P.; Raya, J.; Gougeon, R.D.; Cayot, P.; Domenek, S.; Debeaufort, F.; Karbowiak, T. Biobased Composite Films from Chitosan and Lignin: Antioxidant Activity Related to Structure and Moisture. ACS Sustain. Chem. Eng. 2016, 4, 6371–6381. [Google Scholar] [CrossRef]
- Crouvisier-Urion, K.; Lagorce-Tachon, A.; Lauquin, C.; Winckler, P.; Tongdeesoontorn, W.; Domenek, S.; Debeaufort, F.; Karbowiak, T. Impact of the homogenization process on the structure and antioxidant properties of chitosan-lignin composite films. Food Chem. 2017, 236, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Hargens-Madsen, M.; Schnepf, M.; Hamouz, F.; Weller, C.; Roy, S. Use of edible films and tocopherols in the control of warmed over flavor. J. Am. Diet. Assoc. 1995, 95, A41. [Google Scholar] [CrossRef]
- Oussalah, M.; Caillet, S.; Salmieri, S.; Saucer, L.; Lacroix, M. Antimicrobial and antioxidant effects of milk protein-based film containing essential oils for the preservation of whole beef muscle. J. Agric. Food Chem. 2004, 52, 5598–5605. [Google Scholar] [CrossRef]
- Nisa, I.U.; Ashwar, B.A.; Shah, A.; Gani, A.; Gani, A.; Masoodi, F.A. Development of potato starch based active packaging films loaded with antioxidants and its effect on shelf life of beef. J. Food Sci. Technol. 2015, 52, 7245–7253. [Google Scholar] [CrossRef]
- Yang, H.-J.; Lee, J.-H.; Won, M.; Song, K.B. Antioxidant activities of distiller dried grains with solubles as protein films containing tea extracts and their application in the packaging of pork meat. Food Chem. 2016, 196, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.-Y.; Yang, J.-Y.; Lu, H.-B.; Wang, S.-S.; Yang, J.; Yang, X.-C.; Chai, M.; Li, L.; Cao, J.-X. Effect of chitosan film incorporated with tea polyphenol on quality and shelf life of pork meat patties. Int. J. Biol. Macromol. 2013, 61, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Rachtanapun, P.; Keawsuwan, N.; Kunthaprap, P.; Siriwattanapa, W. Effects of antioxidants in rice and cassava starch film on mechanical properties and study their releasing. In Proceedings of the 8th Agro-Industry Conference “Annual Meeting of Innovation for Food Industry”, Bangkok, Thailand, 15–16 June 2006. [Google Scholar]
- Tongdeesoontorn, W.; Mauer, L.J.; Wongruong, S.; Sriburi, P.; Rachtanapun, P. Effect of carboxymethyl cellulose concentration on physical properties of biodegradable cassava starch-based films. Chem. Cent. J. 2011, 5, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Pagno, C.H.; de Farias, Y.B.; Costa, T.M.H.; Rios, A.d.O.; Flôres, S.H. Synthesis of biodegradable films with antioxidant properties based on cassava starch containing bixin nanocapsules. J. Food Sci. Technol. 2016, 53, 3197–3205. [Google Scholar] [CrossRef]
- Perazzo, K.K.N.C.L.; Conceição, A.C.d.V.; dos Santos, J.C.P.; Assis, D.d.J.; Souza, C.O.; Druzian, J.I. Properties and antioxidant action of actives cassava starch films incorporated with green tea and palm oil extracts. PLoS ONE 2014, 9, e105199. [Google Scholar] [CrossRef]
- ASTM. Standard test methods for tensile properties of thin plastic sheeting D882-91 In Annual Book of ASTM; American Society for Testing and Materials: West Conshohocken, PA, USA, 1991; pp. 1–9. [Google Scholar]
- Phan, T.D.; Debeaufort, F.; Luu, D.; Voilley, A. Functional properties of edible agar-based and starch-based films for food quality preservation. J. Agric. Food Chem. 2005, 53, 973–981. [Google Scholar] [CrossRef]
- AOAC. AOAC SMPR 2015.009: Estimation of Total Phenolic Content Using the Folin-C Assay. J. AOAC Int. 2015, 98, 1109–1110. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Harte, B.R. Physical properties and antioxidant activity of an active film from chitosan incorporated with green tea extract. Food Hydrocoll. 2010, 24, 770–775. [Google Scholar] [CrossRef]
- Zhang, C.; Guo, K.; Ma, Y.; Ma, D.; Li, X.; Zhao, X. Incorporations of blueberry extracts into soybean-protein-isolate film preserve qualities of packaged lard. Int. J. Food Sci. Technol. 2010, 45, 1801–1806. [Google Scholar] [CrossRef]
- Jung, D.C.; Lee, S.Y.; Yoon, J.H.; Hong, K.P.; Kang, Y.S.; Park, S.R.; Park, S.K.; Ha, S.D.; Kim, G.H.; Bae, D.H. Inhibition of pork and fish oxidation by a novel plastic film coated with horseradish extract. LWT Food Sci. Technol. 2009, 42, 856–861. [Google Scholar] [CrossRef]
- Nerin, C.; Tovar, L.; Djenane, D.; Camo, J.; Salafranca, J.; Beltren, J.A.; Roncales, P. Stabilization of beef meat by a new active packaging containing natural antioxidants. J. Agric. Food Chem. 2006, 54, 7840–7846. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Xie, F.; Hasjim, J.; Witt, T.; Halley, P.J.; Gilbert, R.G. Establishing whether the structural feature controlling the mechanical properties of starch films is molecular or crystalline. Carbohydr. Polym. 2015, 117, 262–270. [Google Scholar] [CrossRef]
- Jongjareonrak, A.; Benjakul, S.; Visessanguan, W.; Tanaka, M. Antioxidative activity and properties of fish skin gelatin films incorporated with BHT and [alpha]-tocopherol. Food Hydrocoll. 2008, 22, 449–458. [Google Scholar] [CrossRef]
- Rachtanapun, P.; Wongchaiya, P. Effect of relative humidity on mechanical properties on blended chitosan-methylcellulose film. In Proceedings of the Mae Fah Luang Symposium, Chiang Rai, Thailand, 26–28 November 2008. [Google Scholar]
- Mathew, A.P.; Dufresne, A. Morphological Investigation of Nanocomposites from Sorbitol Plasticized Starch and Tunicin Whiskers. Biomacromolecules 2002, 3, 609–617. [Google Scholar] [CrossRef]
- Suppakul, P.; Miltz, J.; Sonneveld, K.; Bigger, S.W. Characterization of antimicrobial films containing basil extracts. Packag. Technol. Sci. 2006, 19, 259–268. [Google Scholar] [CrossRef]
- Mali, S.; Sakanaka, L.S.; Yamashita, F.; Grossmann, M.V.E. Water sorption and mechanical properties of cassava starch films and their relation to plasticizing effect. Carbohydr. Polym. 2005, 60, 283–289. [Google Scholar] [CrossRef]
- Cerruti, P.; Santagata, G.; Gomez d’Ayala, G.; Ambrogi, V.; Carfagna, C.; Malinconico, M.; Persico, P. Effect of a natural polyphenolic extract on the properties of a biodegradable starch-based polymer. Polym. Degrad. Stab. 2011, 96, 839–846. [Google Scholar] [CrossRef]
- Tong, Q.; Xiao, Q.; Lim, L.-T. Preparation and properties of pullulan–alginate–carboxymethylcellulose blend films. Food Res. Int. 2008, 41, 1007–1014. [Google Scholar] [CrossRef]
- Martins, J.T.; Cerqueira, M.A.; Vicente, A.A. Influence of α-tocopherol on physicochemical properties of chitosan-based films. Food Hydrocoll. 2012, 27, 220–227. [Google Scholar] [CrossRef]
- Rohn, S.; Rawel, H.M.; Kroll, J. Antioxidant activity of protein-bound quercetin. J. Agric. Food Chem. 2004, 52, 4725–4729. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Kosaraju, S.L. Biopolymeric delivery system for controlled release of polyphenolic antioxidants. Eur. Polym. J. 2007, 43, 2956–2966. [Google Scholar] [CrossRef]
- Curcio, M.; Puoci, F.; Iemma, F.; Parisi, O.I.; Cirillo, G.; Spizzirri, U.G.; Picci, N. Covalent insertion of antioxidant molecules on chitosan by a free radical grafting procedure. J. Agric. Food Chem. 2009, 57, 5933–5938. [Google Scholar] [CrossRef]
- Aguilar-Méndez, M.; Martin-Martínez, E.; Ortega-Arroyo, L.; Cruz-Orea, A. Application of differential scanning calorimetry to evaluate thermal properties and study of microstructure of biodegradable films. Int. J. Thermophys. 2010, 31, 595–600. [Google Scholar] [CrossRef]
- Hu, G.; Chen, J.; Gao, J. Preparation and characteristics of oxidized potato starch films. Carbohydr. Polym. 2009, 76, 291–298. [Google Scholar] [CrossRef]
- Xu, Y.X.; Kim, K.M.; Hanna, M.A.; Nag, D. Chitosan-starch composite film: Preparation and characterization. Ind. Crops Prod. 2005, 21, 185–192. [Google Scholar] [CrossRef]
- Almasi, H.; Ghanbarzadeh, B.; Entezami, A.A. Physicochemical properties of starch–CMC–nanoclay biodegradable films. Int. J. Biol. Macromol. 2010, 46, 1–5. [Google Scholar] [CrossRef]
- Arvanitoyannis, I.; Psomiadou, E.; Nakayama, A.; Aiba, S.; Yamamoto, N. Edible films made from gelatin, soluble starch and polyols, Part 3. Food Chem. 1997, 60, 593–604. [Google Scholar] [CrossRef]
- Chen, J.; Liu, C.; Chen, Y.; Chen, Y.; Chang, P.R. Structural characterization and properties of starch/konjac glucomannan blend films. Carbohydr. Polym. 2008, 74, 946–952. [Google Scholar] [CrossRef]
- Kim, S.; Baek, S.-K.; Go, E.; Song, K.B. Application of Adzuki Bean Starch in Antioxidant Films Containing Cocoa Nibs Extract. Polymers 2018, 10, 1210. [Google Scholar] [CrossRef]
- Sangsuwan, J.; Rattanapanone, N.; Rachtanapun, P. Effects of vanillin and plasticizer on properties of chitosan-methyl cellulose based film. J. Appl. Polym. Sci. 2008, 109, 3540–3545. [Google Scholar] [CrossRef]
- Kolodziejska, I.; Piotrowska, B. The water vapour permeability, mechanical properties and solubility of fish gelatin-chitosan films modified with transglutaminase or 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) and plasticized with glycerol. Food Chem. 2007, 103, 295–300. [Google Scholar] [CrossRef]
- Yang, H.; Wang, Y. Effects of concentration on nanostructural images and physical properties of gelatin from channel catfish skins. Food Hydrocoll. 2009, 23, 577–584. [Google Scholar] [CrossRef]
- Soares, R.; Scremin, F.F.; Soldi, V. Thermal stability of biodegradable films based on soy protein and corn starch. Macromol. Symp. 2005, 229, 258–265. [Google Scholar] [CrossRef]
- Atarés, L.; Pérez-Masiá, R.; Chiralt, A. The role of some antioxidants in the HPMC film properties and lipid protection in coated toasted almonds. J. Food Eng. 2011, 104, 649–656. [Google Scholar] [CrossRef]
- Wu, J.-G.; Wang, P.-J.; Chen, S.C. Antioxidant and antimicrobial effectiveness of catechin-impregnated PVA-starch film on red meat. J. Food Qual. 2010, 33, 780–801. [Google Scholar] [CrossRef]


| Films | Tm (°C) | ΔH (J/g) |
|---|---|---|
| Cassava starch–CMC | 128.65 ± 7.25 a,b | 154.14 ± 4.11 a |
| Quercetin | 128.88 ± 3.76 a,b,c | 151.00 ± 4.66 a |
| TBHQ | 131.19 ± 5.70 a | 273.56 ± 9.77 c |
| +50 mg Quercetin | 141.21 ± 6.13 e | 158.20 ± 3.38 a |
| +100 mg Quercetin | 116.09 ± 5.29 b,c,d | 211.12 ± 8.37 b |
| +200 mg Quercetin | 110.16 ± 5.66 b,c,d | 227.88 ± 7.65 b |
| +50 mg TBHQ | 110.09 ± 0.23 d | 147.39 ± 5.73 a |
| +100 mg TBHQ | 118.33 ± 2.88 c,d | 144.75 ± 3.52 a |
| +200 mg TBHQ | 119.65 ± 0.44 c,d | 149.80 ± 1.13 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tongdeesoontorn, W.; Mauer, L.J.; Wongruong, S.; Sriburi, P.; Rachtanapun, P. Physical and Antioxidant Properties of Cassava Starch–Carboxymethyl Cellulose Incorporated with Quercetin and TBHQ as Active Food Packaging. Polymers 2020, 12, 366. https://doi.org/10.3390/polym12020366
Tongdeesoontorn W, Mauer LJ, Wongruong S, Sriburi P, Rachtanapun P. Physical and Antioxidant Properties of Cassava Starch–Carboxymethyl Cellulose Incorporated with Quercetin and TBHQ as Active Food Packaging. Polymers. 2020; 12(2):366. https://doi.org/10.3390/polym12020366
Chicago/Turabian StyleTongdeesoontorn, Wirongrong, Lisa J. Mauer, Sasitorn Wongruong, Pensiri Sriburi, and Pornchai Rachtanapun. 2020. "Physical and Antioxidant Properties of Cassava Starch–Carboxymethyl Cellulose Incorporated with Quercetin and TBHQ as Active Food Packaging" Polymers 12, no. 2: 366. https://doi.org/10.3390/polym12020366
APA StyleTongdeesoontorn, W., Mauer, L. J., Wongruong, S., Sriburi, P., & Rachtanapun, P. (2020). Physical and Antioxidant Properties of Cassava Starch–Carboxymethyl Cellulose Incorporated with Quercetin and TBHQ as Active Food Packaging. Polymers, 12(2), 366. https://doi.org/10.3390/polym12020366