Recovery of Bioactive Compounds from Jaboticaba Peels and Application into Zein Ultrafine Fibers Produced by Electrospinning
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Sample Preparation
2.3. Extraction Procedures
2.4. Total Phenolic (TP) Quantification
2.5. Antioxidant Activity Quantification (AA)
2.6. Total Anthocyanin (TA)
2.7. Cn-3-Glu Quantification
2.8. Antimicrobial Inhibition (AI)
2.9. Preparation of Zein Polymeric Solutions with JPE
2.10. Apparent Viscosity and Electrical Conductivity of the Polymer Solutions
2.11. Production of Zein Ultrafine Fibers by Electrospinning Technique
2.12. Morphology and Diameter Distribution of Ultrafine Fibers
2.13. Thermal Properties of Ultrafine Fibers
2.14. Functional Groups of Ultrafine Fibers
3. Results
3.1. Chemical Composition and Properties of JPE
3.2. Apparent Viscosity and Electrical Conductivity of the Polymer Solutions
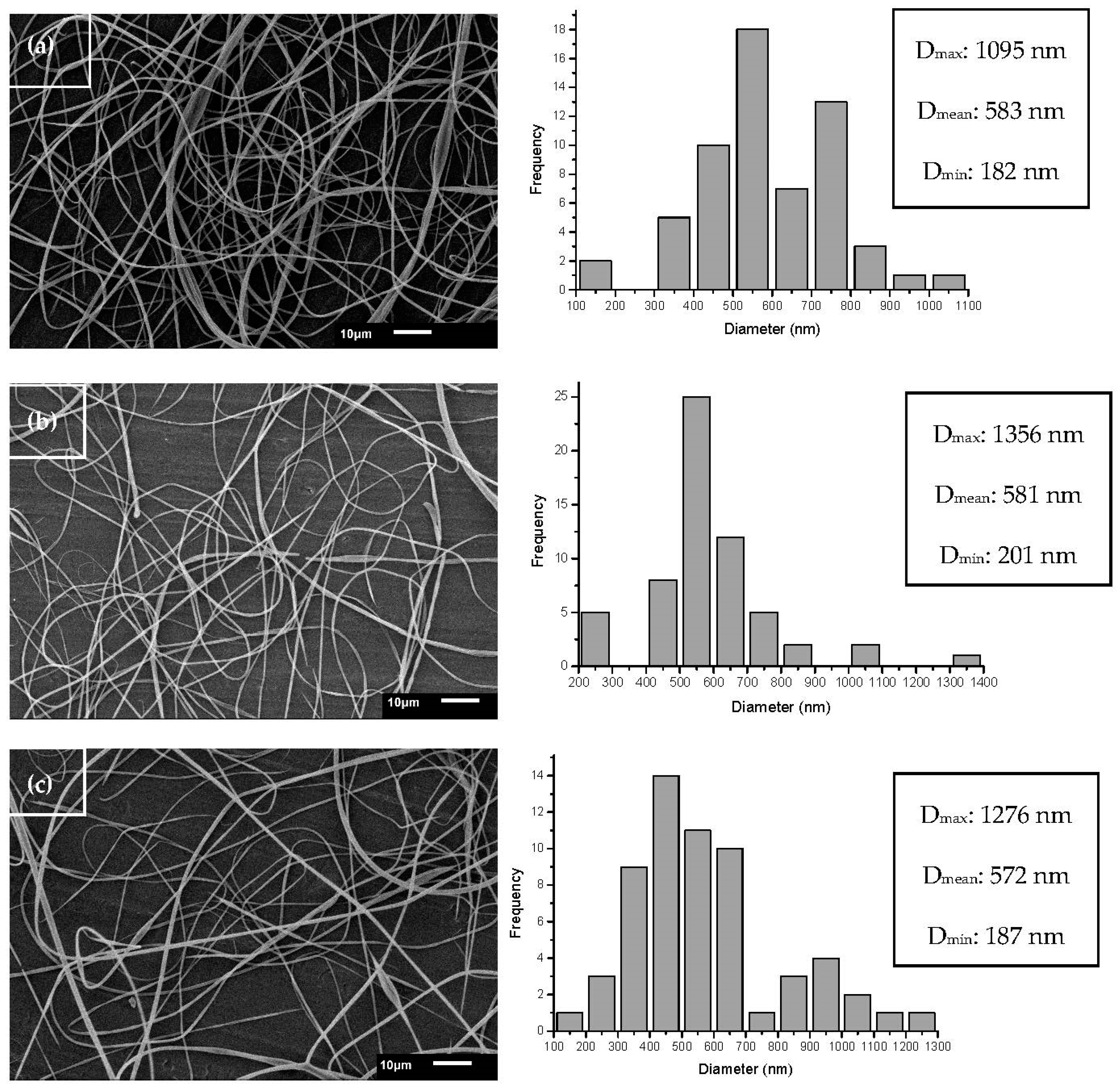
3.3. Morphology of Ultrafine Fibers
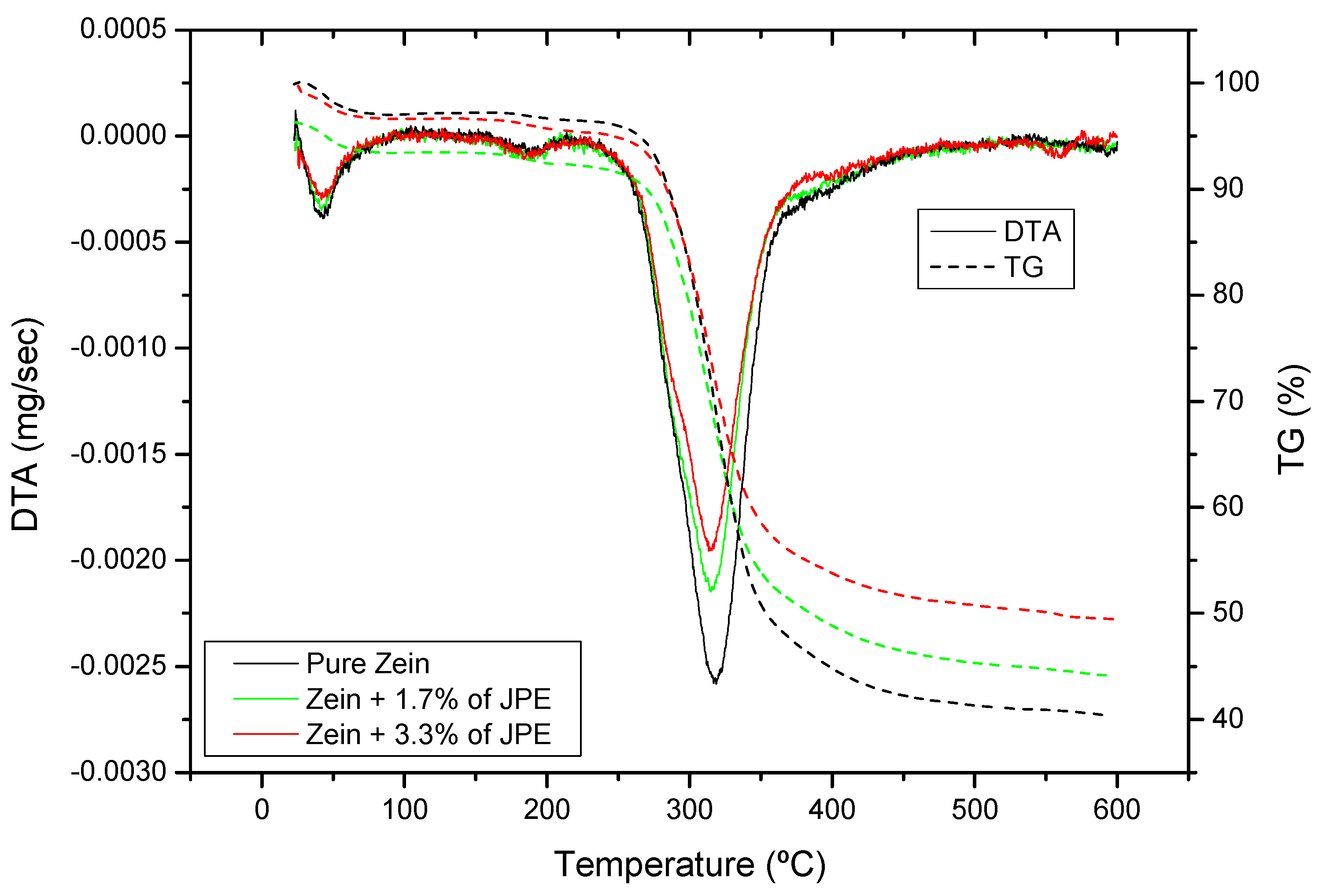
3.4. Thermal Properties of Ultrafine Fibers
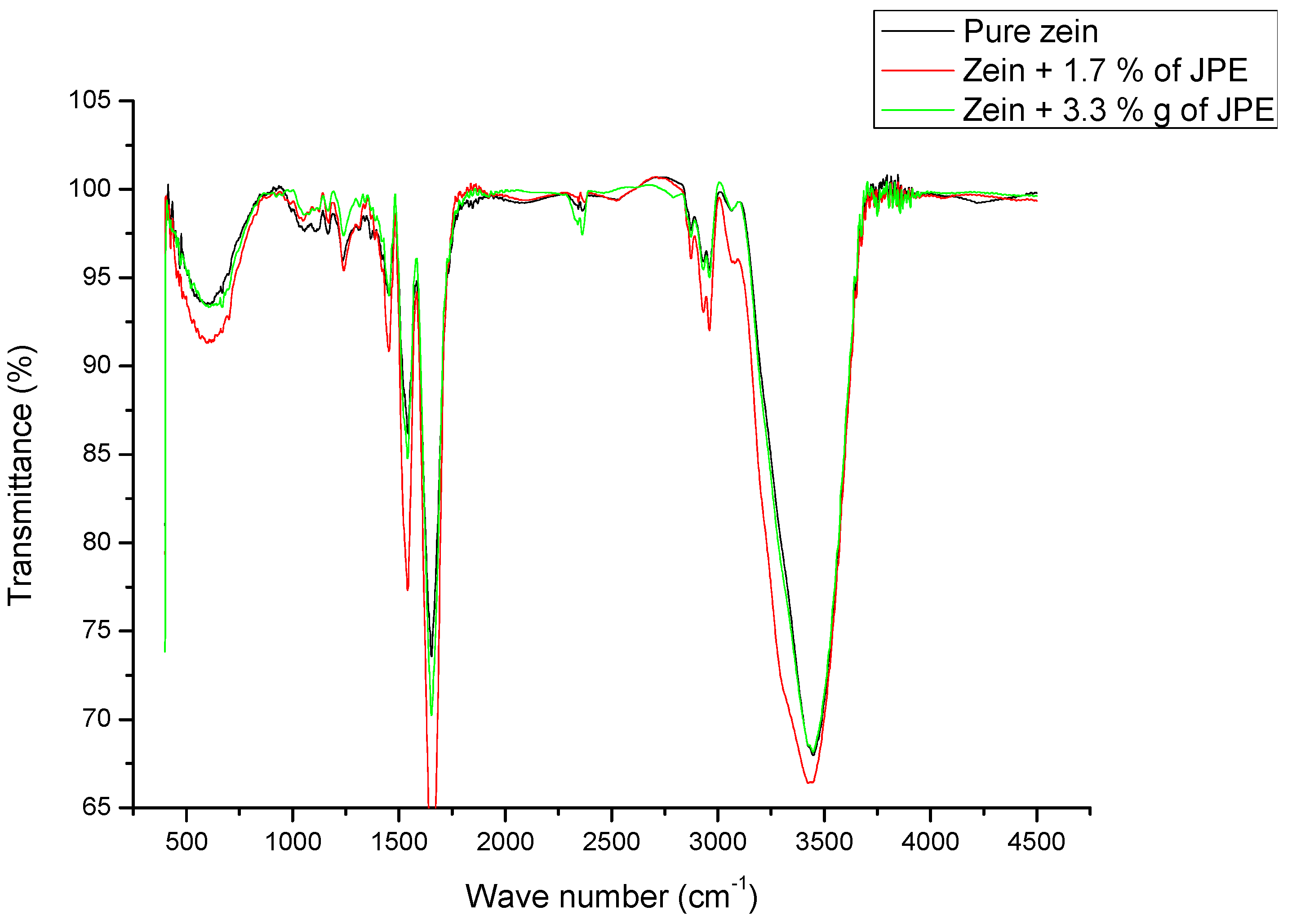
3.5. Functional Groups of Ultrafine Fibers
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leite-Legatti, A.V.; Batista, A.G.; Dragano, N.R.V.; Marques, A.C.; Malta, L.G.; Riccio, M.F.; Eberlin, M.N.; Machado, A.R.T.; de Carvalho-Silva, L.B.; Ruiz, A.L.T.G.; et al. Jaboticaba peel: Antioxidant compounds, antiproliferative and antimutagenic activities. Food Res. Int. 2012, 49, 596–603. [Google Scholar] [CrossRef]
- Neves, N.D.A.; Stringheta, P.C.; Gómez-Alonso, S.; Hermosín-Gutiérrez, I. Flavonols and ellagic acid derivatives in peels of different species of jabuticaba (Plinia spp.) identified by HPLC-DAD-ESI/MSn. Food Chem. 2018, 252, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Baseggio, A.M.; Nuñez, C.E.C.; Dragano, N.R.V.; Lamas, C.A.; Braga, P.A.D.C.; Lenquiste, S.A.; Reyes, F.G.R.; Cagnon, V.H.A.; Júnior, M.R.M. Jaboticaba peel extract decrease autophagy in white adipose tissue and prevents metabolic disorders in mice fed with a high-fat diet. PharmaNutrition 2018, 6, 147–156. [Google Scholar] [CrossRef]
- Inada, K.O.P.; Oliveira, A.A.; Revorêdo, T.B.; Martins, A.B.N.; Lacerda, E.C.Q.; Freire, A.S.; Braz, B.F.; Santelli, R.E.; Torres, A.G.; Perrone, D.; et al. Screening of the chemical composition and occurring antioxidants in jabuticaba (Myrciaria jaboticaba) and jussara (Euterpe edulis) fruits and their fractions. J. Funct. Foods 2015, 17, 422–433. [Google Scholar] [CrossRef]
- Salomão, L.C.C.; de Siqueira, D.L.; Aquino, C.F.; de Lins, L.C.R. Jabuticaba— Myrciaria spp. Exot. Fruits 2018, 237–244. [Google Scholar] [CrossRef]
- Wu, S.B.; Long, C.; Kennelly, E.J. Phytochemistry and health benefits of jaboticaba, an emerging fruit crop from Brazil. Food Res. Int. 2013, 54, 148–159. [Google Scholar] [CrossRef]
- De Carvalho Alves, A.P.; Corrêa, A.D.; Pinheiro, A.C.M.; de Oliveira, F.C. Flour and anthocyanin extracts of jaboticaba skins used as a natural dye in yogurt. Int. J. Food Sci. Technol. 2013, 48, 2007–2013. [Google Scholar] [CrossRef]
- Baldin, J.C.; Michelin, E.C.; Polizer, Y.J.; Rodrigues, I.; de Godoy, S.H.S.; Fregonesi, R.P.; Pires, M.A.; Carvalho, L.T.; Fávaro-Trindade, C.S.; de Lima, C.G.; et al. Microencapsulated jabuticaba (Myrciaria cauliflora) extract added to fresh sausage as natural dye with antioxidant and antimicrobial activity. Meat Sci. 2016, 118, 15–21. [Google Scholar] [CrossRef]
- Lima, A.D.J.B.; Corrêa, A.D.; Saczk, A.A.; Martins, M.P.; Castilho, R.O. Anthocyanins, pigment stability and antioxidant activity in jabuticaba [Myrciaria cauliflora (Mart.) O. Berg]. Rev. Bras. Frutic. 2011, 33, 877–887. [Google Scholar] [CrossRef]
- Barros, H.D.F.Q.; Baseggio, A.M.; Angolini, C.F.F.; Pastore, G.M.; Cazarin, C.B.B.; Marostica-Junior, M.R. Influence of different types of acids and pH in the recovery of bioactive compounds in Jabuticaba peel (Plinia cauliflora). Food Res. Int. 2019, 124, 16–26. [Google Scholar] [CrossRef]
- Ghomari, O.; Sounni, F.; Massaoudi, Y.; Ghanam, J.; Drissi Kaitouni, L.B.; Merzouki, M.; Benlemlih, M. Phenolic profile (HPLC-UV) of olive leaves according to extraction procedure and assessment of antibacterial activity. Biotechnol. Rep. 2019, 23, e00347. [Google Scholar] [CrossRef] [PubMed]
- Alara, O.R.; Mudalip, S.K.A.; Abdurahman, N.H.; Mahmoud, M.S.; Obanijesu, E.O.O. Data on parametric influence of microwave-assisted extraction on the recovery yield, total phenolic content and antioxidant activity of Phaleria macrocarpa fruit peel extract. Chem. Data Collect. 2019, 24, 100277. [Google Scholar] [CrossRef]
- Ćujić, N.; Šavikin, K.; Janković, T.; Pljevljakušić, D.; Zdunić, G.; Ibrić, S. Optimization of polyphenols extraction from dried chokeberry using maceration as traditional technique. Food Chem. 2016, 194, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Xu, Z.; Xiang, C.; Liu, J.; Zhou, L.; Li, T.; Yang, Z.; Ding, C. Comparative evaluation of maceration and ultrasonic-assisted extraction of phenolic compounds from fresh olives. Ultrason. Sonochem. 2017, 37, 328–334. [Google Scholar] [CrossRef]
- Rodsamran, P.; Sothornvit, R. Extraction of phenolic compounds from lime peel waste using ultrasonic-assisted and microwave-assisted extractions. Food Biosci. 2019, 28, 66–73. [Google Scholar] [CrossRef]
- Putnik, P.; Barba, F.J.; Lucini, L.; Rocchetti, G.; Montesano, D. Conventional, non-conventional extraction techniques and new strategies for the recovery of bioactive compounds from plant material for human nutrition. Food Res. Int. 2019, 123, 516–517. [Google Scholar] [CrossRef]
- Bock, N.; Dargaville, T.R.; Woodruff, M.A. Electrospraying of polymers with therapeutic molecules: State of the art. Prog. Polym. Sci. 2012, 37, 1510–1551. [Google Scholar] [CrossRef]
- Deng, L.; Zhang, X.; Li, Y.; Que, F.; Kang, X.; Liu, Y.; Feng, F.; Zhang, H. Characterization of gelatin/zein nanofibers by hybrid electrospinning. Food Hydrocoll. 2018, 75, 72–80. [Google Scholar] [CrossRef]
- Fabra, M.J.; López-Rubio, A.; Lagaron, J.M. Use of the electrohydrodynamic process to develop active/bioactive bilayer films for food packaging applications. Food Hydrocoll. 2016, 55, 11–18. [Google Scholar] [CrossRef]
- Da Silva, C.K.; Mastrantonio, D.J.D.S.; Costa, J.A.V.; de Morais, M.G. Innovative pH sensors developed from ultrafine fibers containing açaí (Euterpe oleracea) extract. Food Chem. 2019, 294, 397–404. [Google Scholar] [CrossRef]
- Wang, S.; Marcone, M.F.; Barbut, S.; Lim, L.T. Electrospun soy protein isolate-based fiber fortified with anthocyanin-rich red raspberry (Rubus strigosus) extracts. Food Res. Int. 2013, 52, 467–472. [Google Scholar] [CrossRef]
- Antunes, M.D.; Dannenberg, G.S.; Fiorentini, Â.M.; Pinto, V.Z.; Lim, L.T.; Zavareze, E.R.; Dias, A.R.G. Antimicrobial electrospun ultrafine fibers from zein containing eucalyptus essential oil/cyclodextrin inclusion complex. Int. J. Biol. Macromol. 2017, 104, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Krumreich, F.D.; Prietsch, L.P.; Antunes, M.D.; Jansen-Alves, C.; Mendonça, C.R.B.; Borges, C.D.; Zavareze, E.D.R.; Zambiazi, R.C. Avocado Oil Incorporated in Ultrafine Zein Fibers by Electrospinning. Food Biophys. 2019, 14, 383–392. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- De Almeida, C.B.; Catelam, K.T.; Cornélio, M.L.; Filho, J.F.L. Morphological and structural characteristics of zein biofilms with added xanthan gum. Food Technol. Biotechnol. 2010, 48, 19–27. [Google Scholar]
- Favaro, L.I.; Balcão, V.M.; Rocha, L.K.; Silva, E.C.; Oliveira, J.M., Jr.; Vila, M.M.; Tubino, M. Physicochemical Characterization of a Crude Anthocyanin Extract from the Fruits of Jussara. J. Braz. Chem. Soc. 2018, 29, 2072–2088. [Google Scholar]
- Lenquiste, S.A.; Marineli, R.D.S.; Moraes, É.A.; Dionísio, A.P.; de Brito, E.S.; Maróstica, M.R. Jaboticaba peel and jaboticaba peel aqueous extract shows in vitro and in vivo antioxidant properties in obesity model. Food Res. Int. 2015, 77, 162–170. [Google Scholar] [CrossRef]
- Meira, N.A.N.; Pereira, N.P.; Maciel, L.F.; Menezes-Filho, J.A.; Oliveira, S.S.P. Development and stability testing of emulsions with Myrciaria cauliflora (Jaboticaba) Peel Extracts for Cosmetic Application. J. Appl. Cosmetol. 2018, 2, 000106. [Google Scholar]
- Palozi, R.A.C.; Guarnier, L.P.; Romão, P.V.M.; Nocchi, S.R.; dos Santos, C.C.; Lourenço, E.L.B.; Silva, D.B.; Gasparotto, F.M.; Gasparotto Junior, A. Pharmacological safety of Plinia cauliflora (Mart.) Kausel in rabbits. Toxicol. Rep. 2019, 6, 616–624. [Google Scholar] [CrossRef]
- Pitz, H.D.S.; Pereira, A.; Blasius, M.B.; Voytena, A.P.L.; Affonso, R.C.L.; Fanan, S.; Trevisan, A.C.D.; Ribeiro-Do-Valle, R.M.; Maraschin, M. In Vitro Evaluation of the antioxidant activity and wound healing properties of jaboticaba (Plinia peruviana) fruit peel hydroalcoholic extract. Oxid. Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S.B.; Bozell, J.J.; Hayes, D.G.; Zivanovic, S. Introduction of primary antioxidant activity to chitosan for application as a multifunctional food packaging material. Food Hydrocoll. 2013, 33, 207–214. [Google Scholar] [CrossRef]
- Francis, F.J.; Markakis, P.C. Food colorants: Anthocyanins. Crit. Rev. Food Sci. Nutr. 2009, 28, 273–314. [Google Scholar] [CrossRef] [PubMed]
- Cassol, L.; Rodrigues, E.; Zapata Noreña, C.P. Extracting phenolic compounds from Hibiscus sabdariffa L. calyx using microwave assisted extraction. Ind. Crops Prod. 2019, 133, 168–177. [Google Scholar] [CrossRef]
- Akbari, S.; Abdurahman, N.H.; Yunus, R.M.; Fayaz, F. Microwave-assisted extraction of saponin, phenolic and flavonoid compounds from Trigonella foenum-graecum seed based on two level factorial design. J. Appl. Res. Med. Aromat. Plants 2019, 14, 100212. [Google Scholar] [CrossRef]
- Blackhall, M.L.; Berry, R.; Davies, N.W.; Walls, J.T. Optimized extraction of anthocyanins from Reid Fruits’ Prunus avium ‘Lapins’ cherries. Food Chem. 2018, 256, 280–285. [Google Scholar] [CrossRef]
- Aspé, E.; Fernández, K. The effect of different extraction techniques on extraction yield, total phenolic, and anti-radical capacity of extracts from Pinus radiata Bark. Ind. Crops Prod. 2011, 34, 838–844. [Google Scholar] [CrossRef]
- Taamalli, A.; Arráez-Román, D.; Barrajón-Catalán, E.; Ruiz-Torres, V.; Pérez-Sánchez, A.; Herrero, M.; Ibañez, E.; Micol, V.; Zarrouk, M.; Segura-Carretero, A.; et al. Use of advanced techniques for the extraction of phenolic compounds from Tunisian olive leaves: Phenolic composition and cytotoxicity against human breast cancer cells. Food Chem. Toxicol. 2012, 50, 1817–1825. [Google Scholar] [CrossRef]
- Wu, S.B.; Dastmalchi, K.; Long, C.; Kennelly, E.J. Metabolite profiling of jaboticaba (Myrciaria cauliflora) and other dark-colored fruit juices. J. Agric. Food Chem. 2012, 60, 7513–7525. [Google Scholar] [CrossRef]
- Ham, J.S.; Kim, H.Y.; Lim, S.T. Antioxidant and deodorizing activities of phenolic components in chestnut inner shell extracts. Ind. Crops Prod. 2015, 73, 99–105. [Google Scholar] [CrossRef]
- Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound phenolics in foods, a review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar] [CrossRef]
- Tauler, R.; Brown, S.D.; Walczak, B. Comprehensive Chemometrics Chemical and Biochemical Analysis, 1st ed.; Elsevier: Amsterdan, The Netherlands, 2009; ISBN 978-0-444-52702-8. [Google Scholar]
- Gurak, P.D.; De Bona, G.S.; Tessaro, I.C.; Marczak, L.D.F. Jaboticaba pomace powder obtained as a co-product of juice extraction: A comparative study of powder obtained from peel and whole fruit. Food Res. Int. 2014, 62, 786–792. [Google Scholar] [CrossRef]
- Mohd-Esa, N.; Hern, F.S.; Ismail, A.; Yee, C.L. Antioxidant activity in different parts of roselle (Hibiscus sabdariffa L.) extracts and potential exploitation of the seeds. Food Chem. 2010, 122, 1055–1060. [Google Scholar] [CrossRef]
- Özkan, G.; Sagdiç, O.; Baydar, N.G.; Kurumahmutoglu, Z. Antibacterial activities and total phenolic contents of grape pomace extracts. J. Sci. Food Agric. 2004, 84, 1807–1811. [Google Scholar] [CrossRef]
- Borrás-Linares, I.; Fernández-Arroyo, S.; Arráez-Roman, D.; Palmeros-Suárez, P.A.; Del Val-Díaz, R.; Andrade-Gonzáles, I.; Fernández-Gutiérrez, A.; Gómez-Leyva, J.F.; Segura-Carretero, A. Characterization of phenolic compounds, anthocyanidin, antioxidant and antimicrobial activity of 25 varieties of Mexican Roselle (Hibiscus sabdariffa). Ind. Crops Prod. 2015, 69, 385–394. [Google Scholar] [CrossRef]
- Souza-Moreira, T.M.; Severi, J.A.; Santos, E.; Silva, V.Y.A.; Vilegas, W.; Salgado, H.R.N.; Pietro, R.C.L.R. Chemical and antidiarrheal studies of Plinia cauliflora. J. Med. Food 2011, 14, 1590–1596. [Google Scholar] [CrossRef]
- Niu, B.; Zhan, L.; Shao, P.; Xiang, N.; Sun, P.; Chen, H.; Gao, H. Electrospinning of zein-ethyl cellulose hybrid nanofibers with improved water resistance for food preservation. Int. J. Biol. Macromol. 2019, 142, 592–599. [Google Scholar] [CrossRef]
- Son, W.K.; Ho Youk, J.; Seung Lee, T.; Park, W.H. Effect of pH on electrospinning of poly(vinyl alcohol). Mater. Lett. 2005, 59, 1571–1575. [Google Scholar] [CrossRef]
- Aydogdu, A.; Sumnu, G.; Sahin, S. Fabrication of gallic acid loaded Hydroxypropyl methylcellulose nanofibers by electrospinning technique as active packaging material. Carbohydr. Polym. 2019, 208, 241–250. [Google Scholar] [CrossRef]
- Gazquez, G.C.; Smulders, V.; Veldhuis, S.A.; Wieringa, P.; Moroni, L.; Boukamp, B.A.; Ten Elshof, J.E. Influence of solution properties and process parameters on the formation and morphology of YSZ and NiO ceramic nanofibers by electrospinning. Nanomaterials 2017, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Lee, C.K.; Kim, S.I. Effect of ionic salts on the processing of poly(2-acrylamido-2-methyl-1- propane sulfonic acid) nanofibers. J. Appl. Polym. Sci. 2005, 96, 1388–1393. [Google Scholar] [CrossRef]
- Bagbi, Y.; Pandey, A.; Solanki, P.R. Electrospun Nanofibrous Filtration Membranes for Heavy Metals and Dye Removal; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128139271. [Google Scholar]
- Dashdorj, U.; Reyes, M.K.; Unnithan, A.R.; Tiwari, A.P.; Tumurbaatar, B.; Park, C.H.; Kim, C.S. Fabrication and characterization of electrospun zein/Ag nanocomposite mats for wound dressing applications. Int. J. Biol. Macromol. 2015, 80, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.M.; Yu, D.G. An elevated temperature electrospinning process for preparing acyclovir-loaded PAN ultrafine fibers. J. Mater. Process. Technol. 2010, 210, 1551–1555. [Google Scholar] [CrossRef]
- Siegrist, M.; Stampfli, N.; Kastenholz, H.; Keller, C. Perceived risks and perceived benefits of different nanotechnology foods and nanotechnology food packaging. Appetite 2008, 51, 283–290. [Google Scholar] [CrossRef]
- Sorrentino, A.; Gorrasi, G.; Vittoria, V. Potential perspectives of bio-nanocomposites for food packaging applications. Trends Food Sci. Technol. 2007, 18, 84–95. [Google Scholar] [CrossRef]
- Neo, Y.P.; Ray, S.; Jin, J.; Gizdavic-Nikolaidis, M.; Nieuwoudt, M.K.; Liu, D.; Quek, S.Y. Encapsulation of food grade antioxidant in natural biopolymer by electrospinning technique: A physicochemical study based on zein-gallic acid system. Food Chem. 2013, 136, 1013–1021. [Google Scholar] [CrossRef]
- Barbosa, V.; Ramires, E.C.; Razera, I.A.T.; Frollini, E. Biobased composites from tannin-phenolic polymers reinforced with coir fibers. Ind. Crops Prod. 2010, 32, 305–312. [Google Scholar] [CrossRef]
- Moomand, K.; Lim, L.T. Properties of encapsulated fish oil in electrospun zein fibres under simulated in vitro conditions. Food Bioprocess Technol. 2015, 8, 431–444. [Google Scholar] [CrossRef]
- Torres-Giner, S.; Gimenez, E.; Lagaron, J.M. Characterization of the morphology and thermal properties of Zein Prolamine nanostructures obtained by electrospinning. Food Hydrocoll. 2008, 22, 601–614. [Google Scholar] [CrossRef]
- Forato, L.A.; Bicudo, T.D.C.; Colnago, L.A. Conformation of α zeins in solid state by Fourier Transform IR. Biopolym. Biospectroscopy Sect. 2003, 72, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.A.; de Arruda, I.N.Q.; Stefani, R. Active chitosan/PVA films with anthocyanins from Brassica oleraceae (Red Cabbage) as Time-Temperature Indicators for application in intelligent food packaging. Food Hydrocoll. 2015, 43, 180–188. [Google Scholar] [CrossRef]
- Erdogan, I.; Demir, M.; Bayraktar, O. Olive leaf extract as a crosslinking agent for the preparation of electrospun zein fibers. J. Appl. Polym. Sci. 2015, 132, 1–9. [Google Scholar] [CrossRef]
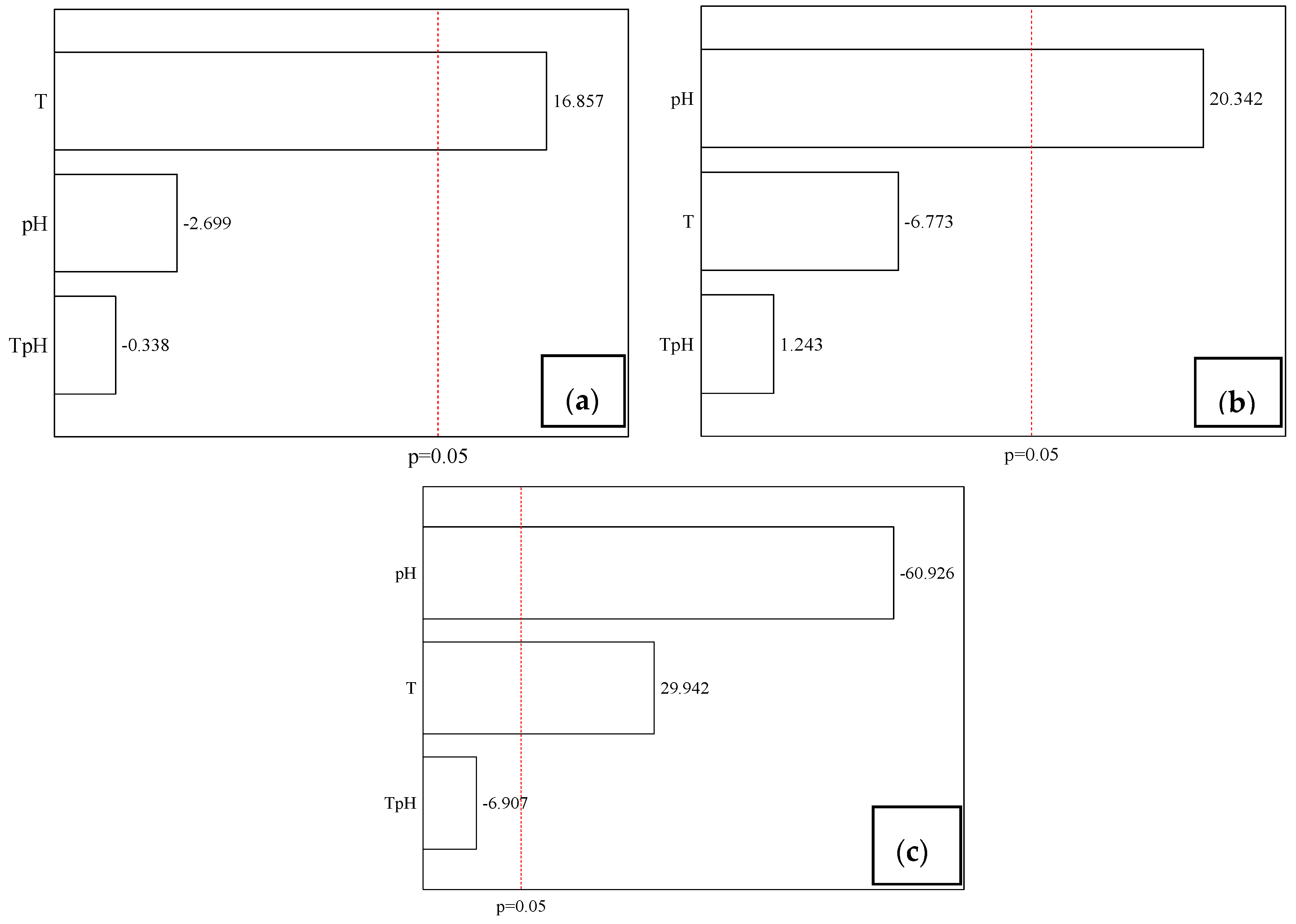
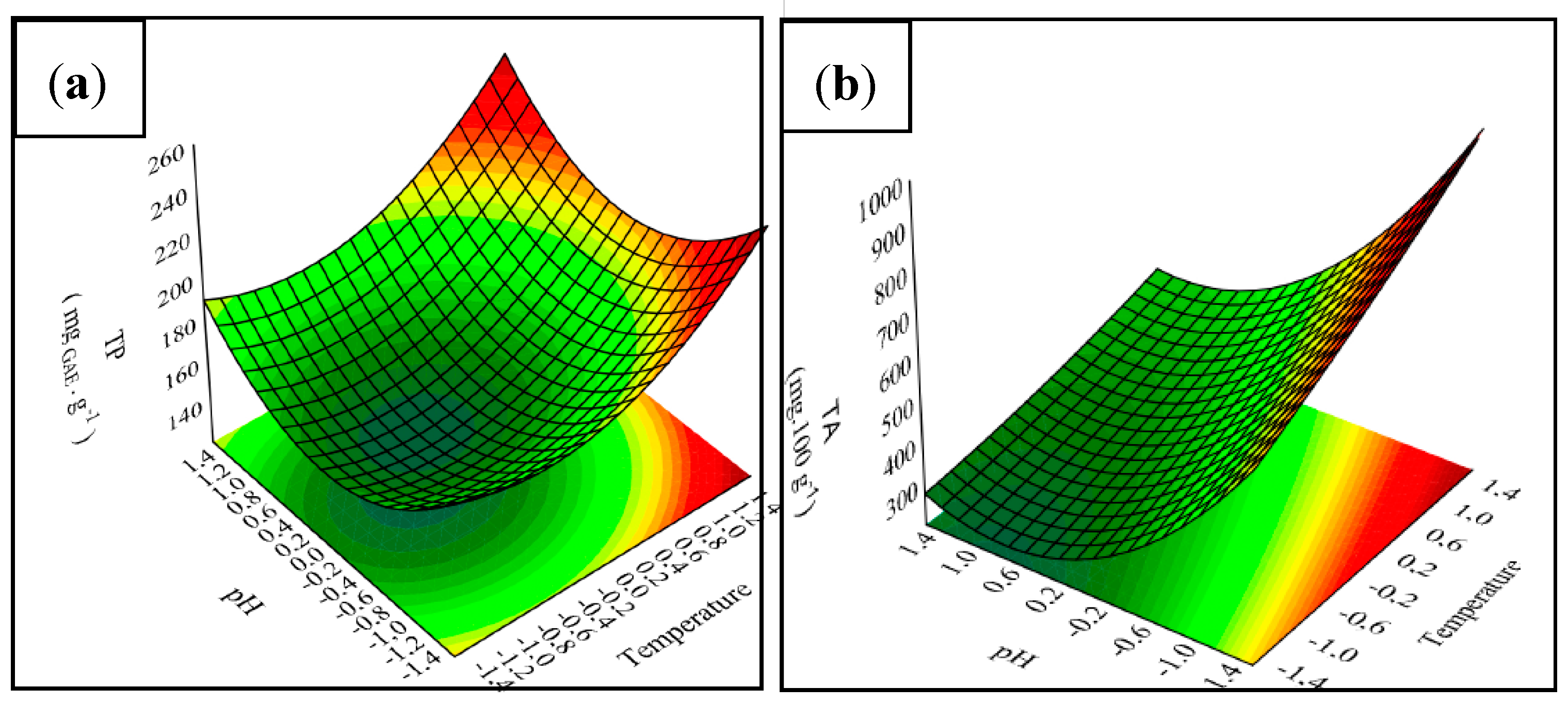
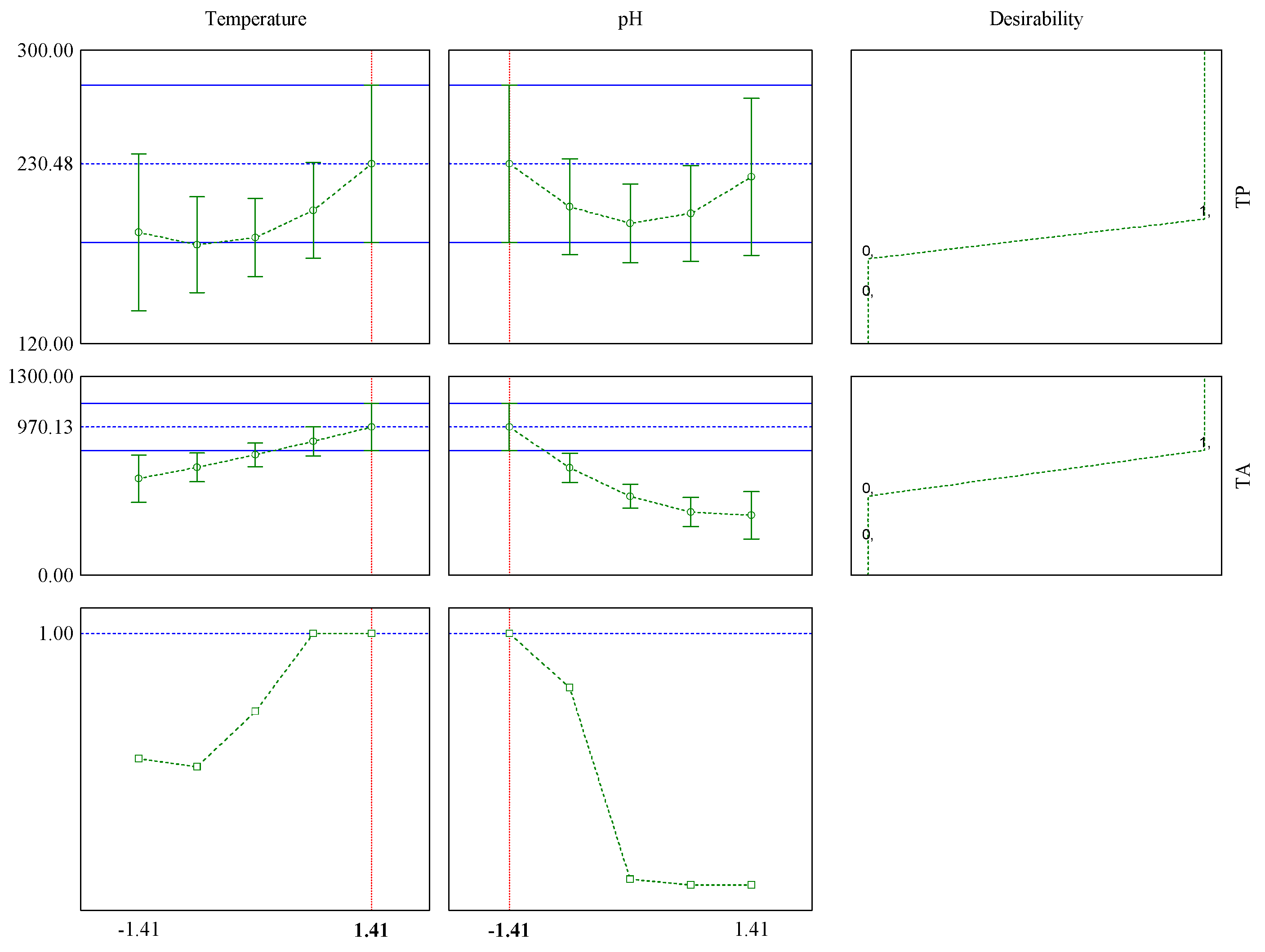
| Variables | Levels | ||||
|---|---|---|---|---|---|
| −1.41 | −1 | 0 | +1 | +1.41 | |
| T (°C) | 32 | 40 | 60 | 80 | 88 |
| pH | 1 | 2.5 | 6 | 9.5 | 11 |
| Run | T (°C) | pH | TP (mgGAE g−1 d.w.) | AA (%) | TA (mg 100 g−1 d.w.) |
|---|---|---|---|---|---|
| 1 | 40 | 2.5 | 160.58 ± 2.01 | 94.19 ± 0.30 | 513.8 ± 5.01 |
| 2 | 40 | 9.5 | 163.06 ± 1.03 | 94.85 ± 0.09 | 327.7 ± 8.52 |
| 3 | 80 | 2.5 | 189.05 ± 0.85 | 92.35 ± 0.58 | 749.27 ± 7.47 |
| 4 | 80 | 9.5 | 189.91 ± 1.11 | 94.13 ± 0.18 | 456.41 ± 13.46 |
| 5 | 32 | 6 | 154.93 ± 1.18 | 94.67 ± 0.14 | 265.11 ± 0.91 |
| 6 | 88 | 6 | 196.63 ± 1.39 | 90.52 ± 0.92 | 470.42 ± 12.56 |
| 7 | 60 | 1 | 191.78 ± 1.88 | 78.61 ± 0.21 | 819.32 ± 21.44 |
| 8 | 60 | 11 | 176.48 ± 2.12 | 94.70 ± 0.65 | 216.24 ± 4.24 |
| 9 (C) | 60 | 6 | 151.07 ± 3.06 | 93.92 ± 0.27 | 383.5 ± 3.03 |
| 10 (C) | 60 | 6 | 147.68 ± 2.45 | 94.54 ± 0.02 | 394.43 ± 0.70 |
| 11 (C) | 60 | 6 | 185.52 ± 2.06 | 92.80 ± 0.13 | 373.97 ± 8.86 |
| Variance Analysis | Sum of Squares | Degrees of Freedom | Mean of Squares | Fvalue | Ftabled | |
|---|---|---|---|---|---|---|
| TP | Regression | 3562.34 | 5 | 712.47 | 23.27 | 6.25 |
| Residual | 122.46 | 4 | 30.62 | |||
| Lack of fit | 116.72 | 3 | 38.91 | 154.20 | 215.71 | |
| Pure error | 5.75 | 1 | 5.75 | |||
| Total | 3684.8 | 9 | ||||
| AA | Regression | 122.88 | 5 | 24.58 | 1.10 | 6.25 |
| Residual | 89.10 | 4 | 22.28 | |||
| Lack of fit | 88.91 | 3 | 29.64 | 154.20 | 215.71 | |
| Pure error | 0.19 | 1 | 0.19 | |||
| Total | 211.99 | 9 | ||||
| TA | Regression | 306,582.8 | 5 | 61,316.56 | 8.93 | 6.25 |
| Residual | 27,451.5 | 4 | 6862.88 | |||
| Lack of fit | 27,379.7 | 3 | 9126.57 | 127.11 | 215.71 | |
| Pure error | 71.8 | 1 | 71.8 | |||
| Total | 334,034 | 9 |
| Scale of D | Quality Evaluation |
|---|---|
| 1.00 | No preference for improvement beyond this point |
| 1.00–0.80 | Acceptable and excellent |
| 0.80–0.63 | Acceptable and good |
| 0.63–0.40 | Acceptable but poor |
| 0.40–0.30 | Borderline |
| 0.30–0.00 | Unacceptable |
| 0.00 | Completely unacceptable |
| Polymer Solutions | Apparent Viscosity (Pa s) * | Electrical Conductivity (μs cm−1) |
|---|---|---|
| Pure zein (30%) | 0.39 b ± 0.06 | 235.9 a ± 2.7 |
| Zein 30% + 1.7% JPE | 0.21 a ± 0.08 | 249.0 b ± 1.3 |
| Zein 30% + 3.3% JPE | 0.13 a ± 0.01 | 256.4 c ± 1.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avila, L.B.; Fontes, M.R.V.; Zavareze, E.d.R.; Moraes, C.C.; Morais, M.M.; Rosa, G.S.d. Recovery of Bioactive Compounds from Jaboticaba Peels and Application into Zein Ultrafine Fibers Produced by Electrospinning. Polymers 2020, 12, 2916. https://doi.org/10.3390/polym12122916
Avila LB, Fontes MRV, Zavareze EdR, Moraes CC, Morais MM, Rosa GSd. Recovery of Bioactive Compounds from Jaboticaba Peels and Application into Zein Ultrafine Fibers Produced by Electrospinning. Polymers. 2020; 12(12):2916. https://doi.org/10.3390/polym12122916
Chicago/Turabian StyleAvila, Luisa Bataglin, Milena Ramos Vaz Fontes, Elessandra da Rosa Zavareze, Caroline Costa Moraes, Marcilio Machado Morais, and Gabriela Silveira da Rosa. 2020. "Recovery of Bioactive Compounds from Jaboticaba Peels and Application into Zein Ultrafine Fibers Produced by Electrospinning" Polymers 12, no. 12: 2916. https://doi.org/10.3390/polym12122916
APA StyleAvila, L. B., Fontes, M. R. V., Zavareze, E. d. R., Moraes, C. C., Morais, M. M., & Rosa, G. S. d. (2020). Recovery of Bioactive Compounds from Jaboticaba Peels and Application into Zein Ultrafine Fibers Produced by Electrospinning. Polymers, 12(12), 2916. https://doi.org/10.3390/polym12122916






