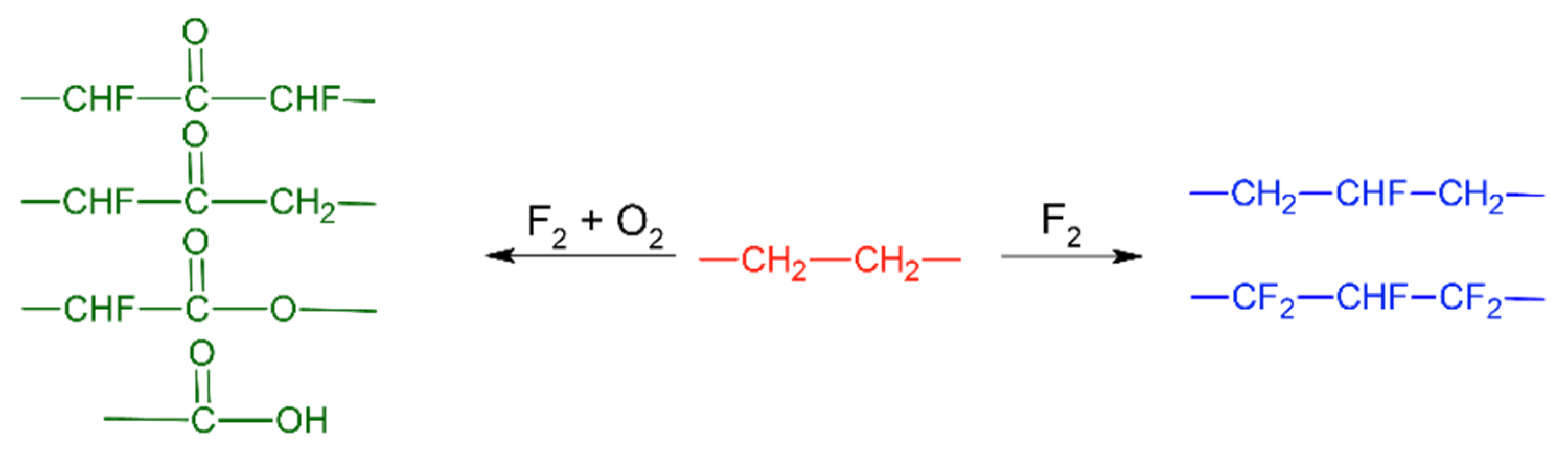Direct Fluorination as Method of Improvement of Operational Properties of Polymeric Materials
Abstract
1. Introduction
2. Surface Properties of Polymers after Direct Fluorination
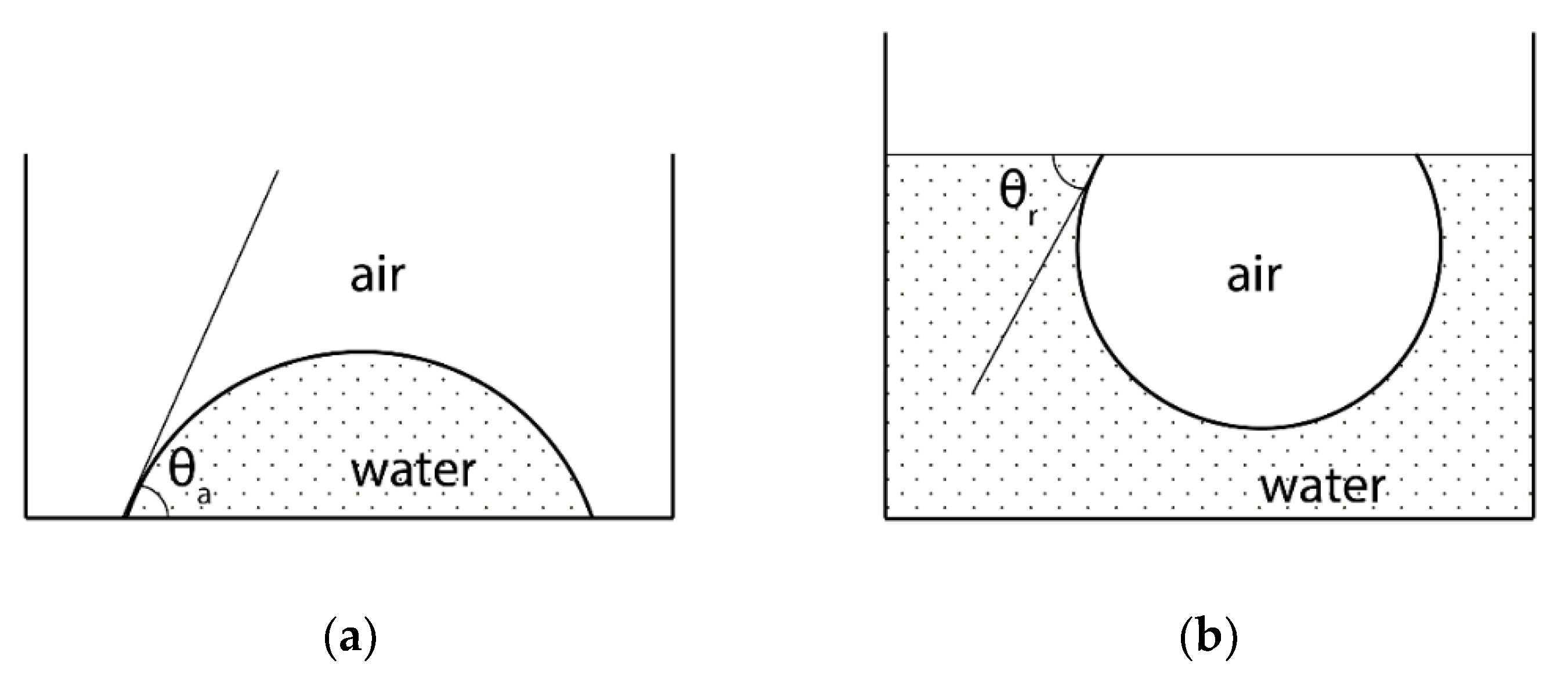
2.1. Wetting Method: The General Information
2.2. Changes in the Hydrophilicity/Hydrophobicity of the Polymer Surface as a Result of Fluorination
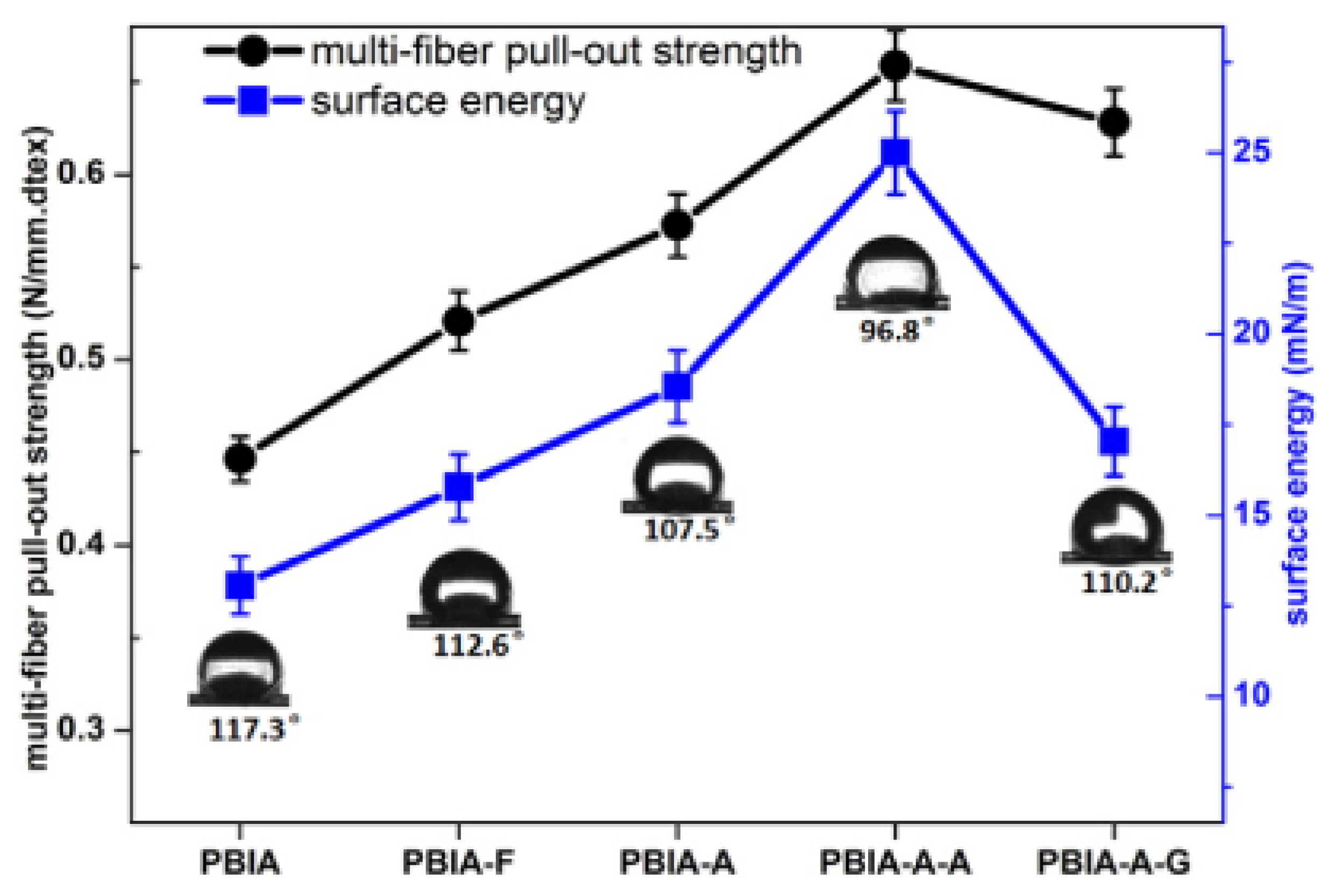
2.3. Adhesion and Specific Free Surface Energy of Fluorinated Polymers
3. Mechanical Properties of Polymers and Composites after Direct Fluorination
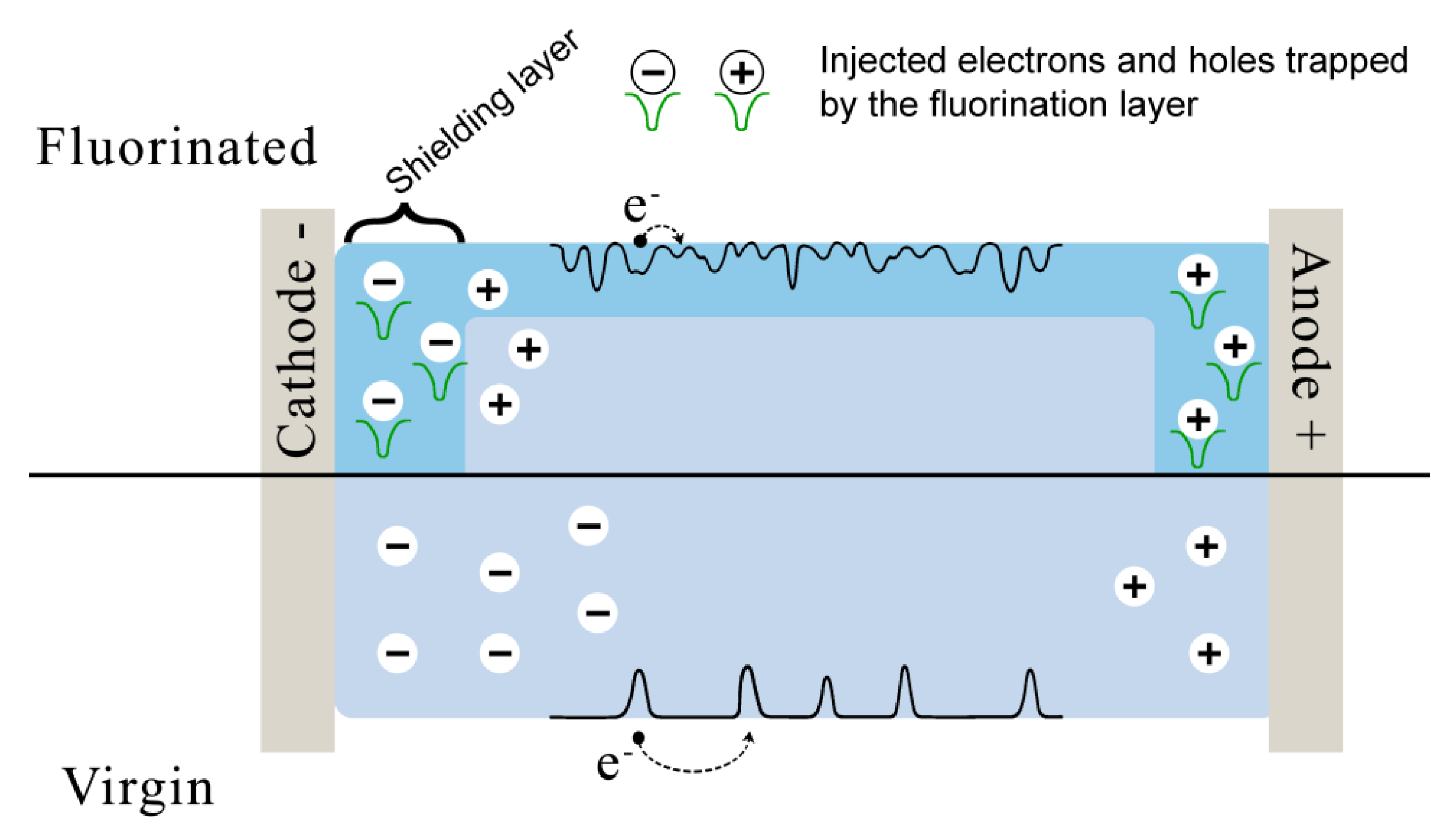
4. Electrical Properties of Polymers after Direct Surface Fluorination
5. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| CNT | carbon nanotube |
| ED-22 | epoxy resin (22–24% of epoxy groups) |
| EP | ethylene and propylene copolymer |
| FEP | tetrafluoroethylene and hexafluoropropylene copolymer |
| HDPE | high density polyethylene |
| LDPE | low density polyethylene |
| NBR | nitrile butadiene rubber |
| PBIA | poly(benzimidazole-amide) |
| PDMS | polydimethylsiloxane |
| PE | polyethylene |
| PETP | polyethylene terephthalate |
| PFA | perfluoroalkoxy alkanes |
| PP | polypropylene |
| PPO | poly(2,6-dimethyl-1,4-phenylene oxide) |
| PS | polystyrene |
| PTFE | polytetrafluoroethylene |
| PVF | polyvinyl fluoride |
| PVDF | polyvinylidene fluoride |
| PVTMS | poly(vinyl trimethylsilane) |
| TPU | thermoplastic polyurethane |
| UHMWPE | ultra-high molecular weight polyethylene |
References
- Adams, D.E.C.; Halden, R.U. Fluorinated chemicals and the impacts of anthropogenic use. In Contaminants of Emerging Concern in the Environment: Ecological and Human Health Considerations; American Chemical Society: Washington, DC, USA, 2010; Volume 1048, pp. 539–560. [Google Scholar]
- Wang, J.; Sánchez-Roselló, M.; Aceña, J.L.; del Pozo, C.; Sorochinsky, A.E.; Fustero, S.; Soloshonok, V.A.; Liu, H. Fluorine in pharmaceutical industry: Fluorine-containing drugs introduced to the market in the last decade (2001–2011). Chem. Rev. 2014, 114, 2432–2506. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.C.; Wang, L.J.; Jiang, B.; Wang, S.Y.; Wu, N.; Li, X.Q.; Shi, D.Y. Application of fluorine in drug design during 2010–2015 years: A mini-review. Mini Rev. Med. Chem. 2017, 17, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Ojima, I. Strategic incorporation of fluorine for drug discovery and development. In Modern Synthesis Processes and Reactivity of Fluorinated Compounds; Elsevier: Amsterdam, The Netherlands, 2017; pp. 499–531. [Google Scholar]
- Fujiwara, T.; O’Hagan, D. Successful fluorine-containing herbicide agrochemicals. J. Fluor. Chem. 2014, 167, 16–29. [Google Scholar] [CrossRef]
- Tressaud, A. Fluorine: A Paradoxical Element; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Ebnesajjad, S.; Morgan, R. Applications of fluorinated additives for lubricants. In Plastics Design Library: Fluoropolymer Additives, 2nd ed.; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 75–119. [Google Scholar]
- Prozhega, M.V.; Albagachiev, A.Y.; Smirnov, N.I.; Smirnov, N.N. Lubricating materials for mechanisms operating in space. J. Frict. Wear 2018, 39, 335–340. [Google Scholar] [CrossRef]
- Banerjee, S. Handbook of Specialty Fluorinated Polymers: Preparation, Properties, and Applications; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Gardiner, J. Fluoropolymers: Origin, production, and industrial and commercial applications. Aust. J. Chem. 2015, 68, 13–22. [Google Scholar] [CrossRef]
- Brothers, P.; Chapman, G.; Farnsworth, K.; Morgan, R. Melt processible perfluoroplastics for demanding applications. In Handbook of Fluoropolymer Science and Technology; Smith, D.W., Iacono, S.T., Suresh, S.I., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 599–621. [Google Scholar]
- Okamoto, Y.; Chiang, H.-C.; Merkel, T. Perfluoropolymers for gas separation membrane applications. In Fascinating Fluoropolymers and their Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 143–155. [Google Scholar]
- Yampolskii, Y.; Belov, N.; Alentiev, A. Perfluorinated polymers as materials of membranes for gas and vapor separation. J. Membr. Sci. 2020, 598, 117779. [Google Scholar] [CrossRef]
- Yampolskii, Y.P.; Belov, N.A.; Alentiev, A.Y. Fluorine in the structure of polymers: Influence on the gas separation properties. Russ. Chem. Rev. 2019, 88, 387–405. [Google Scholar] [CrossRef]
- Belov, N.; Yampolskii, Y. Gas transport in fluorine-containing polymers. In Fascinating Fluoropolymers and their Applications; Fomin, S., Ameduri, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 157–203. [Google Scholar]
- Sawada, H.; Ameduri, B. Fluorinated Polymers: Volume 1: Synthesis, Properties, Processing and Simulation; Royal Society of Chemistry: Cambridge, UK, 2016. [Google Scholar]
- McKeen, L.W. Fluorinated Coatings: Technology, History, and Applications. In Introduction to Fluoropolymers; William Andrew Publishing: Norwich, NY, USA, 2013; pp. 231–276. [Google Scholar]
- Guselnikova, O.; Elashnikov, R.; Postnikov, P.; Svorcik, V.; Lyutakov, O. Smart, piezo-responsive polyvinylidenefluoride/polymethylmethacrylate surface with triggerable water/oil wettability and adhesion. ACS Appl. Mater. Interfaces 2018, 10, 37461–37469. [Google Scholar] [CrossRef]
- Bastekova, K.; Guselnikova, O.; Postnikov, P.; Elashnikov, R.; Kunes, M.; Kolska, Z.; Švorčík, V.; Lyutakov, O. Spatially selective modification of PLLA surface: From hydrophobic to hydrophilic or to repellent. Appl. Surf. Sci. 2017, 397, 226–234. [Google Scholar] [CrossRef]
- Guselnikova, O.; Postnikov, P.; Sajdl, P.; Elashnikov, R.; Švorčík, V.; Lyutakov, O. Functional and switchable amphiphilic pmma surface prepared by 3D selective modification. Adv. Mater. Interfaces 2018, 5, 1701182. [Google Scholar] [CrossRef]
- Reisinger, J.J.; Hillmyer, M.A. Synthesis of fluorinated polymers by chemical modification. Prog. Polym. Sci. 2002, 27, 971–1005. [Google Scholar] [CrossRef]
- Cools, P.; Astoreca, L.; Esbah Tabaei, P.S.; Thukkaram, M.; De Smet, H.; Morent, R.; De Geyter, N. Surface treatment of polymers by plasma. In Surface Modification of Polymers: Methods and Applications; Pinson, J., Thirty, D., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 31–65. [Google Scholar]
- Kharitonov, A.P. Direct fluorination of polymers—From fundamental research to industrial applications. Prog. Org. Coat. 2008, 61, 192–204. [Google Scholar] [CrossRef]
- Maity, J.; Jacob, C.; Das, C.K.; Kharitonov, A.P.; Singh, R.P.; Alam, S. Fluorinated aramid fiber reinforced polypropylene composites and their characterization. Polym. Compos. 2007, 28, 462–469. [Google Scholar] [CrossRef]
- Nazarov, V.G. Composition and dimensions of the surface and transition layers in modified polymers. Polym. Sci. Ser. B 1997, 39, 142–145. [Google Scholar]
- Kharitonov, A.P.; Simbirtseva, G.V.; Tressaud, A.; Durand, E.; Labrugère, C.; Dubois, M. Comparison of the surface modifications of polymers induced by direct fluorination and rf-plasma using fluorinated gases. J. Fluor. Chem. 2014, 165, 49–60. [Google Scholar] [CrossRef]
- Hu, H.; Xiao, M.; Wang, S.J.; Shen, P.K.; Meng, Y.Z. Surface fluorination of poly (fluorenyl ether ketone) ionomers as proton exchange membranes for fuel cell application. Fuel Cells 2011, 11, 353–360. [Google Scholar] [CrossRef]
- Nazarov, V.G.; Stolyarov, V.P.; Gagarin, M.V. Simulation of chemical modification of polymer surface. J. Fluor. Chem. 2014, 161, 120–127. [Google Scholar] [CrossRef]
- Wang, Z.; Li, S.; Li, B.; Lai, W.; Liu, Y.; Cheng, Z.; Wang, X.; Liu, X. The preparation of surface fluorinated polyethylene films with excellent properties similar to that of fluoropolymers. J. Fluor. Chem. 2017, 200, 169–178. [Google Scholar] [CrossRef]
- Hruska, Z.; Lepot, X. Surface modification of polymer webs by oxyfluorination. J. Plast. Film Sheeting 1999, 15, 235–255. [Google Scholar] [CrossRef]
- Kharitonov, A.P.; Simbirtseva, G.V.; Bouznik, V.M.; Chepezubov, M.G.; Dubois, M.; Guérin, K.; Hamwi, A.; Kharbache, H.; Masin, F. Modification of ultra-high-molecular weight polyethylene by various fluorinating routes. J. Polym. Sci. A Polym. Chem. 2011, 49, 3559–3573. [Google Scholar] [CrossRef]
- Wang, Z.; Macosko, C.W.; Bates, F.S. Tuning surface properties of poly (butylene terephthalate) melt blown fibers by alkaline hydrolysis and fluorination. ACS Appl. Mater. Interfaces 2014, 6, 11640–11648. [Google Scholar] [CrossRef] [PubMed]
- Chong, Y.; Watanabe, N. A novel composite having super hydrophobic property. J. Fluor. Chem. 1991, 54, 43. [Google Scholar] [CrossRef]
- Zha, J.; Ali, S.S.; Peyroux, J.; Batisse, N.; Claves, D.; Dubois, M.; Kharitonov, A.P.; Monier, G.; Darmanin, T.; Guittard, F.; et al. Superhydrophobicity of polymer films via fluorine atoms covalent attachment and surface nano-texturing. J. Fluor. Chem. 2017, 200, 123–132. [Google Scholar] [CrossRef]
- Cheng, Z.; Wu, P.; Gao, J.; Wang, X.; Ren, M.; Li, B.; Luo, L.; Liu, X. Structural evolution of fluorinated aramid fibers with fluorination degree and dominant factor for its adhesion property. J. Fluor. Chem. 2016, 188, 139–146. [Google Scholar] [CrossRef]
- Du Toit, F.J.; Sanderson, R.D.; Engelbrecht, W.J.; Wagener, J.B. The effect of surface fluorination on the wettability of high density polyethylene. J. Fluor. Chem. 1995, 74, 43–48. [Google Scholar] [CrossRef]
- Peyroux, J.; Dubois, M.; Tomasella, E.; Petit, E.; Flahaut, D. Enhancement of surface properties on commercial polymer packaging films using various surface treatment processes (fluorination and plasma). Appl. Surf. Sci. 2014, 315, 426–431. [Google Scholar] [CrossRef]
- Charlet, K.; Saulnier, F.; Gautier, D.; Pouzet, M.; Dubois, M.; Béakou, A. Fluorination as an effective way to reduce natural fibers hydrophilicity. In Natural Fibres: Advances in Science and Technology Towards Industrial Applications; Springer: Berlin, Germany, 2016; pp. 211–229. [Google Scholar]
- Jeong, E.; Lee, B.H.; Doh, S.J.; Park, I.J.; Lee, Y.S. Multifunctional surface modification of an aramid fabric via direct fluorination. J. Fluor. Chem. 2012, 141, 69–75. [Google Scholar] [CrossRef]
- Kharitonov, A.P.; Kharitonova, L.N. Surface modification of polymers by direct fluorination: A convenient approach to improve commercial properties of polymeric articles. Pure Appl. Chem. 2009, 81, 451–471. [Google Scholar] [CrossRef]
- Nazarov, V.G.; Doronin, F.A.; Evdokimov, A.G.; Rytikov, G.O.; Stolyarov, V.P. Oxyfluorination-controlled variations in the wettability of polymer film surfaces. Colloid J. 2019, 81, 146–157. [Google Scholar] [CrossRef]
- An, Z.; Gu, X.; Shen, R.; Tang, Y.; Zheng, F.; Zhang, Y.; Yang, L. Resistance to corona discharge of HTV silicone rubber surface layers fluorinated at different temperatures. IEEE Trans. Dielectr. Electr. Insul. 2018, 25, 729–740. [Google Scholar] [CrossRef]
- Okazoe, T.; Shirakawa, D.; Murata, K. Application of liquid-phase direct fluorination: Novel synthetic methods for a polyfluorinated coating material and a monomer of a perfluorinated polymer electrolyte membrane. Appl. Sci. 2012, 2, 327–341. [Google Scholar] [CrossRef]
- Vega-Cantú, Y.; Hauge, R.; Norman, L.; Billups, W.E. Enhancement of the chemical resistance of nitrile rubber by direct fluorination. J. Appl. Polym. Sci. 2003, 89, 971–979. [Google Scholar] [CrossRef]
- Kharitonov, A.P.; Simbirtseva, G.V.; Nazarov, V.G.; Stolyarov, V.P.; Dubois, M.; Peyroux, J. Enhanced anti-graffiti or adhesion properties of polymers using versatile combination of fluorination and polymer grafting. Prog. Org. Coat. 2015, 88, 127–136. [Google Scholar] [CrossRef]
- Shan, F.; An, Z.; Gu, X.; Shen, R.; Que, L.; Zheng, F.; Zhang, Y. Fluorination mechanisms of silicone rubbers and surface properties. In Proceedings of the 2017 International Symposium on Electrical Insulating Materials (ISEIM), Toyohashi, Japan, 11–15 September 2017. [Google Scholar]
- Kim, D.S.; Cho, H.I.; Kim, D.H.; Lee, B.S.; Lee, B.S.; Yoon, S.W.; Kim, Y.S.; Moon, G.Y.; Byun, H.; Rhim, J.W. Surface fluorinated poly (vinyl alcohol)/poly (styrene sulfonic acid-co-maleic acid) membrane for polymer electrolyte membrane fuel cells. J. Membr. Sci. 2009, 342, 138–144. [Google Scholar] [CrossRef]
- Du, B.X.; Guo, Y.G. Effects of surface fluorination on dielectric properties and surface charge behavior of water absorbed polyimide film. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 1369–1375. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, H.S.; Jin, F.L. Influence of fluorination on surface and dielectric characteristics of polyimide thin film. J. Colloid Interface Sci. 2005, 282, 238–240. [Google Scholar] [CrossRef]
- Du, B.X.; Li, J.; Du, W. Dynamic behavior of surface charge on direct- fluorinated polyimide films. IEEE Trans. Dielectr. Electr. Insul. 2013, 20, 947–954. [Google Scholar] [CrossRef]
- Du, B.X.; Li, Z.L. Hydrophobicity, surface charge and DC flashover characteristics of direct-fluorinated RTV silicone rubber. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 934–940. [Google Scholar] [CrossRef]
- An, Z.; Mao, M.; Cang, J.; Zhang, Y.; Zheng, F. Significantly improved piezoelectric thermal stability of cellular polypropylene films by high pressure fluorination and post-treatments. J. Appl. Phys. 2012, 111, 024111. [Google Scholar] [CrossRef]
- An, Z.; Zhao, M.; Yao, J.; Zhang, Y.; Xia, Z. Influence of fluorination on piezoelectric properties of cellular polypropylene ferroelectrets. J. Phys. D Appl. Phys. 2008, 42, 015418. [Google Scholar] [CrossRef]
- Xie, D.; An, Z.; Xiao, H.; Zheng, F.; Lei, Q.; Zhang, Y. Highly hydrophobic and partially conductive polydimethylsiloxane surface produced by direct fluorination and subsequent annealing. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 2968–2977. [Google Scholar] [CrossRef]
- Liu, N.; Li, Z.; Chen, G.; Chen, Q.; Li, S. Space charge dynamics Of CF4 fluorinated LDPE samples from different fluorination conditions and their DC conductivities. Mater. Res. Express 2017, 4, 075308. [Google Scholar] [CrossRef]
- Zhu, W.B.; Du, B.X.; Li, J.; Jiang, J.P.; Su, J.G.; Li, A.; He, Z.Y.; Zhang, M.M.; Huang, P.H. Effects of moisture on surface charge behavior of fluorinated oil-impregnated paper under DC and pulse voltages. In Proceedings of the IEEE 2nd International Conference on Dielectrics (ICD), Budapest, Hungary, 1–5 July 2018. [Google Scholar]
- Zhang, B.; Zhang, G.; Wang, Q.; Li, C.; He, J.; An, Z. Suppression of surface charge accumulation on Al2O3-filled epoxy resin insulator under dc voltage by direct fluorination. AIP Adv. 2015, 5, 127207. [Google Scholar] [CrossRef]
- An, Z.; Chen, W.; Chen, K.; Gao, W.; Shen, Z.; Yang, Y.; Liu, F.; Liu, X.; Zhang, Z.; Yang, W.; et al. Comparative study on direct fluorination and surface properties of alumina-filled and unfilled epoxy insulators. IEEE Trans. Dielectr. Electr. Insul. 2020, 27, 85–93. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, T.; Li, J.; Zeeshan, K.M.; He, L.; Huang, Z.; He, Y. DC breakdown and flashover characteristics of direct fluorinated epoxy/Al2O3 nanocomposites. IEEE Trans. Dielectr. Electr. Insul. 2019, 26, 702–723. [Google Scholar] [CrossRef]
- Li, C.; Hu, J.; Lin, C.; Zhang, B.; Zhang, G.; He, J. Surface charge migration and dc surface flashover of surface-modified epoxy-based insulators. J. Phys. D Appl. Phys. 2017, 50, 065301. [Google Scholar] [CrossRef]
- Li, C.; He, J.; Hu, J. Surface morphology and electrical characteristics of direct fluorinated epoxy-resin/alumina composite. IEEE Trans. Dielectr. Electr. Insul. 2016, 23, 3071–3077. [Google Scholar] [CrossRef]
- Li, C.; Hu, J.; Lin, C.; He, J. The control mechanism of surface traps on surface charge behavior in alumina-filled epoxy composites. J. Phys. D Appl. Phys. 2016, 49, 445304. [Google Scholar] [CrossRef]
- Lagow, R.J.; Wei, H.C. Direct fluorination of polymers. In Fluoropolymers 1: Synthesis; Springer: Boston, MA, USA, 2002; pp. 209–221. [Google Scholar]
- Loginov, B.A.; Kharitonov, A.P. Direct fluorination of polymer final products: From fundamental study to practical application. Russ. J. Gen. Chem. 2009, 79, 635–641. [Google Scholar] [CrossRef]
- Tressaud, A.; Durand, E.; Labrugère, C.; Kharitonov, A.P.; Kharitonova, L.N. Modification of surface properties of carbon-based and polymeric materials through fluorination routes: From fundamental research to industrial applications. J. Fluor. Chem. 2007, 128, 378–391. [Google Scholar] [CrossRef]
- Tressaud, A.; Durand, E.; Labrugčre, C.; Kharitonov, A.P.; Simbirtseva, G.V.; Kharitonova, L.N.; Dubois, M. Surface modification of polymers treated by various fluorinating media. Acta Chim. Slov. 2013, 60, 495–504. [Google Scholar] [PubMed]
- Dolzhikova, V.D.; Bogdanova, Y.G.; Majouga, A.G.; Beloglazkina, E.K.; Kudrinsky, A.A. Preparing metal-complex surfaces based on self-assembled monolayers of thiols and disulfides on gold. Russ. J. Phys. Chem. A 2017, 91, 240–245. [Google Scholar] [CrossRef]
- Adamson, A. Physical Chemistry of Surfaces; John Wiley and Sons: New York, NY, USA, 1976. [Google Scholar]
- De Gennes, P.G. Wetting: Statics and dynamics. Rev. Mod. Phys. 1985, 57, 827–863. [Google Scholar] [CrossRef]
- Ferguson, G.S.; Whitesides, G.M. Thermal reconstruction of the functionalized interface of polyethylene carboxylic acid and its derivatives. In Modern Approaches to Wettability: Theory and Applications; Plenum Press: New York, NY, USA, 1992; pp. 143–179. [Google Scholar]
- Cheng, Z.; Wu, P.; Li, B.; Chen, T.; Liu, Y.; Ren, M.; Wang, Z.; Lai, W.; Wang, X.; Liu, X. Surface chain cleavage behavior of PBIA fiber induced by direct fluorination. Appl. Surf. Sci. 2016, 384, 480–486. [Google Scholar] [CrossRef]
- Le Roux, J.D.; Paul, D.R.; Arendt, M.F.; Yuan, Y.; Cabasso, I. Surface fluorination of poly (phenylene oxide) composite membranes: Part II. Characterization of the fluorinated layer. J. Membr. Sci. 1994, 90, 37–53. [Google Scholar] [CrossRef]
- Kumeeva, T.Y.; Prorokova, N.P. Control of the sorption properties and wettability of a nonwoven polypropylene material by direct gas fluorination. Russ. J. Appl. Chem. 2019, 92, 701–706. [Google Scholar] [CrossRef]
- Pouzet, M.; Dubois, M.; Charlet, K.; Béakou, A.; Leban, J.M.; Baba, M. Fluorination renders the wood surface hydrophobic without any loss of physical and mechanical properties. Ind. Crops. Prod. 2019, 133, 133–141. [Google Scholar] [CrossRef]
- Bogdanova, Y.G.; Dolzhikova, V.D.; Alentiev, A.Y.; Karzov, I.M. Contact angle measurements in study of polymers for creation of materials. J. Int. Sci. Publ. Mater. Methods Technol. 2012, 6 Pt 2, 273–285. [Google Scholar]
- Sevastianov, V.I. Interrelation of protein adsorption and blood compatibility of biomaterials. In High Performance Biomaterials; Technomic Press: Lancaster, PA, USA, 1991; pp. 313–341. [Google Scholar]
- Shchukin, E.D.; Pertsov, A.V.; Amelina, E.A.; Zelenev, A.S. Colloid and Surface Chemistry; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Neumann, A.W.; Good, R.J.; Hope, C.J.; Sejpal, M. An equation-of-state approach to determine surface tensions of low-energy solids from contact angles. J. Colloid Interface Sci. 1974, 49, 291–304. [Google Scholar] [CrossRef]
- Chibowski, E.; Ontiveros-Ortega, A.; Perea-Carpio, R. On the interpretation of contact angle hysteresis. J. Adhes. Sci. Technol. 2002, 16, 1367–1404. [Google Scholar] [CrossRef]
- Batschinski, A.J. Investigations of the internal friction of fluids. Z. Phys. Chem. 1913, 84, 643–706. [Google Scholar]
- Ferguson, A. On a relation between surface tension and density. J. Chem. Soc. Faraday Trans. 1923, 19, 407–412. [Google Scholar] [CrossRef]
- Sugden, S. A relation between surface tension, density, and chemical composition. J. Chem. Soc. Trans. 1924, 125, 1177–1189. [Google Scholar] [CrossRef]
- Kharitonov, A.P.; Moskvin, Y.L.; Teplyakov, V.V.; Le Roux, J.D. Direct fluorination of poly (vinyl trimethylsilane) and poly (phenylene oxide). J. Fluor. Chem. 1999, 93, 129–137. [Google Scholar] [CrossRef]
- Cheng, Z.; Li, B.; Huang, J.; Chen, T.; Liu, Y.; Wang, X.; Liu, X. Covalent modification of Aramid fibers’ surface via direct fluorination to enhance composite interfacial properties. Mater. Des. 2016, 106, 216–225. [Google Scholar] [CrossRef]
- Fowkes, F.M.; Mostafa, M.A. Acid-base interactions in polymer adsorption. Ind. Eng. Chem. Prod. Res. Dev. 1978, 17, 3–7. [Google Scholar] [CrossRef]
- Kloubek, J. Development of methods for surface free energy determination using contact angles of liquids on solids. Adv. Colloid Interface Sci. 1992, 38, 99–142. [Google Scholar] [CrossRef]
- Sharma, P.K.; Rao, K.H. Analysis of different approaches for evaluation of surface energy of microbial cells by contact angle goniometry. Adv. Colloid Interface Sci. 2002, 98, 341–463. [Google Scholar] [CrossRef]
- Maity, J.; Jacob, C.; Alam, S.; Singh, R.P. Direct fluorination of Twaron fiber and preparation of PP/Twaron fiber composites using MA-g-PP as a compatibilizer. J. Compos. Mater. 2009, 43, 709–723. [Google Scholar] [CrossRef]
- Hayes, L.J. Surface energy of fluorinated surfaces. J. Fluor. Chem. 1976, 8, 69–88. [Google Scholar] [CrossRef]
- Owens, D.K.; Wendt, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Wu, S. Polymer Interface and Adhesion; Marcel Dekker: New York, NY, USA, 1982; 98p. [Google Scholar]
- Bogdanova, Y.G.; Dolzhikova, V.D.; Karzov, I.M.; Alentiev, A.Y. A new approach to the determination of adhesion properties of polymer networks. Macromol. Symp. 2012, 316, 63–70. [Google Scholar] [CrossRef]
- Bogdanova, Y.G.; Dolzhikova, V.D.; Summ, B.D. The influence of component’s chemical nature on wetting effect of mixed surfactant solutions. Vestn. Mosk. Univ. 2004, 45, 186–194. [Google Scholar]
- Pouzet, M.; Dubois, M.; Charlet, K.; Béakou, A. The effect of lignin on the reactivity of natural fibres towards molecular fluorine. Mater. Des. 2017, 120, 66–74. [Google Scholar] [CrossRef]
- Maity, J.; Kothary, P.; O’Rear, E.A.; Jacob, C. Preparation and comparison of hydrophobic cotton fabric obtained by direct fluorination and admicellar polymerization of fluoromonomers. Ind. Eng. Chem. Res. 2010, 49, 6075–6079. [Google Scholar] [CrossRef]
- Saulnier, F.; Dubois, M.; Charlet, K.; Frezet, L.; Beakou, A. Direct fluorination applied to wood flour used as a reinforcement for polymers. Carbohydr. Polym. 2013, 94, 642–646. [Google Scholar] [CrossRef]
- Charlet, K.; Saulnier, F.; Dubois, M.; Beakou, A. Improvement of wood polymer composite mechanical properties by direct fluorination. Mater. Des. 2015, 74, 61–66. [Google Scholar] [CrossRef]
- Du Toit, F.J.; Sanderson, R.D. Surface fluorination of polypropylene: 1. Characterisation of surface properties. J. Fluor. Chem. 1999, 98, 107–114. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Istratkin, V.A.; Kumeeva, T.Y.; Vavilova, S.Y.; Kharitonov, A.P.; Bouznik, V.M. Improvement of polypropylene nonwoven fabric antibacterial properties by the direct fluorination. RSC Adv. 2015, 5, 44545–44549. [Google Scholar] [CrossRef]
- Prorokova, N.P.; Kumeeva, T.Y.; Vavilova, S.Y. Improving the wettability of polyester fabric with using direct fluorination. J. Fluor. Chem. 2019, 219, 115–122. [Google Scholar] [CrossRef]
- Schlögl, S.; Kramer, R.; Lenko, D.; Schröttner, H.; Schaller, R.; Holzner, A.; Kern, W. Fluorination of elastomer materials. Eur. Polym. J. 2011, 47, 2321–2330. [Google Scholar] [CrossRef]
- Li, B.; Gao, J.; Wang, X.; Fan, C.; Wang, H.; Liu, X. Surface modification of polypropylene battery separator by direct fluorination with different gas components. Appl. Surf. Sci. 2014, 290, 137–141. [Google Scholar] [CrossRef]
- Li, B.; Fan, C.; Wang, H.; Ren, M.; Wu, P.; Wang, X.; Liu, X. A composite with excellent tribological performance derived from oxy-fluorinated UHMWPE particle/polyurethane. RSC Adv. 2014, 18, 9321–9325. [Google Scholar] [CrossRef]
- Boinovich, L.B.; Emelyanenko, A.M. Hydrophobic materials and coatings: Principles of design, properties and applications. Russ. Chem. Rev. 2008, 77, 583. [Google Scholar] [CrossRef]
- Shen, R.; An, Z.; Yang, Y.; Liu, X.; Li, X.; Gu, X.; Zhang, Y. Preliminary study on structural integrity and properties of surface fluorinated silicone rubbers. In Proceedings of the 12th International Conference on the Properties and Applications of Dielectric Materials (ICPADM), Xian, China, 20–24 May 2018. [Google Scholar]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. J. Chem. Soc. Faraday Trans. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Gekas, V.; Persson, K.M.; Wahlgren, M.; Sivik, B. Contact angles of ultrafiltration membranes and their possible correlation to membrane performance. J. Membr. Sci. 1992, 72, 293–302. [Google Scholar] [CrossRef]
- Gao, J.; Xu, X.; Fan, C.; Wang, X.; Dai, Y.; Liu, X. Surface modification of fluoroelastomer by direct fluorination with fluorine gas. Mater. Lett. 2014, 121, 219–222. [Google Scholar] [CrossRef]
- Sawada, H.; Suzuki, T.; Takashima, H.; Takishita, K. Preparation and properties of fluoroalkyl end-capped vinyltrimethoxysilane oligomeric nanoparticles—A new approach to facile creation of a completely superhydrophobic coating surface with these nanoparticles. Colloid Polym. Sci. 2008, 286, 1569–1574. [Google Scholar] [CrossRef]
- Kim, C.S.; Kang, S.; Rhim, J.W.; Park, S.G. Synthesis of aminated poly (ether imide) for the preparation of bi-polar membranes and their application to hypochlorite production through the surface direct fluorination. Polym. Korea 2015, 39, 338–345. [Google Scholar] [CrossRef][Green Version]
- Vautard, F.; Fioux, P.; Vidal, L.; Siffer, F.; Roucoules, V.; Schultz, J.; Nardin, M.; Defoort, B. Use of plasma polymerization to improve adhesion strength in carbon fiber composites cured by electron beam. ACS Appl. Mater. Interfaces 2014, 6, 1662–1674. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, P.; Wang, X.; Yang, J.; Liu, X.Y. Effect of direct fluorination on the bonding property of ultra-high molecular weight polyethylene. Acta Polym. Sin. 2011, 5, 543–547. [Google Scholar] [CrossRef]
- Blokhin, A.N.; Dyachkova, T.P.; Maksimkin, A.V.; Stolyarov, R.A.; Suhorukov, A.K.; Burmistrov, I.N.; Kharitonov, A.P. Polymer composites based on epoxy resin with added carbon nanotubes. Fuller. Nanotub. Carbon Nanostructures 2020, 28, 45–49. [Google Scholar] [CrossRef]
- Schonhorn, H.; Hansen, R.H. Surface treatment of polymers. II. Effectiveness of fluorination as a surface treatment for polyethylene. J. Appl. Polym. Sci. 1968, 12, 1231–1237. [Google Scholar] [CrossRef]
- Kruppke, I.; Bartusch, M.; Hickmann, R.; Hund, R.D.; Cherif, C. Effects of (Oxy-) fluorination on various high-performance yarns. Molecules 2016, 21, 1127. [Google Scholar] [CrossRef]
- Li, B.; Zhang, J.; Ren, M.; Wu, P.; Liu, Y.; Chen, T.; Cheng, Z.; Wang, X.; Liu, X. Various surface functionalizations of ultra-high-molecular-weight polyethylene based on fluorine-activation behavior. RSC Adv. 2015, 5, 79081–79089. [Google Scholar] [CrossRef]
- Kharitonov, A.P.; Taege, R.; Ferrier, G.; Teplyakov, V.V.; Syrtsova, D.A.; Koops, G.H. Direct fluorination—Useful tool to enhance commercial properties of polymer articles. J. Fluor. Chem. 2005, 126, 251–263. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Q.; Du, X.; Li, L.; Li, P. Surface modification of polyethylene terephthalate films by direct fluorination. AIP Adv. 2018, 8, 125333. [Google Scholar] [CrossRef]
- Lee, S.A.; Oh, S.H.; Lee, W. The effect of direct fluorination of polydimethylsiloxane films on their surface properties. J. Colloid Interface Sci. 2009, 332, 461–466. [Google Scholar] [CrossRef]
- Zhu, Z.; Xia, Y.; Niu, G.; Liu, J.; Wang, C.; Jiang, H. Effect of direct fluorination on the mechanical and scratch performance of nitrile butadiene rubber. Wear 2017, 376, 1314–1320. [Google Scholar] [CrossRef]
- Gao, J.; Dai, Y.; Wang, X.; Huang, J.; Yao, J.; Yang, J.; Liu, X. Effects of different fluorination routes on aramid fiber surface structures and interlaminar shear strength of its composites. Appl. Surf. Sci. 2013, 270, 627–633. [Google Scholar] [CrossRef]
- Murali, R.S.; Sankarshana, T.; Sridhar, S.; Kharitonov, A.P. Fluorinated polymer membranes for separation of industrial gas mixtures. J. Polym. Mater. 2012, 29, 317–330. [Google Scholar]
- Bikerman, J.J. New ideas concerning the strength of adhesive joints of polymers. Russ. Chem. Rev. 1972, 41, 651. [Google Scholar] [CrossRef]
- Kim, J.; Jeong, E.; Lee, Y.S. Direct fluorination as a novel organophilic modification method for the preparation of Illite/polypropylene nanocomposites. J. Mater. Sci. 2012, 47, 1046–1053. [Google Scholar] [CrossRef]
- Volynskii, A.L.; Nazarov, V.G.; Yarysheva, L.M.; Stolyarov, V.P.; Tsypysheva, S.V.; Bagrov, D.V.; Rukhlya, E.G.; Yarysheva, A.Y.; Bakeev, N.F. Evaluation of the deformation strength properties of a fluorinated polymer layer on the surface of poly (ethylene terephthalate) films. Dokl. Phys. Chem. 2012, 422, 5–7. [Google Scholar] [CrossRef]
- Volynskii, L.; Bakeev, N.F. Surface Phenomena in the Structural and Mechanical Behaviour of Solid Polymers; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Panchuk, D.A.; Sadakbaeva, Z.K.; Bagrov, D.V.; Kechek’ Yan, A.S.; Bol’ Shakova, A.V.; Abramchuk, S.S.; Yarysheva, L.M.; Volynskii, A.L.; Bakeev, N.F. Specific features of surface structuring during deformation of plasma-treated polymer films. Polym. Sci. Ser. A 2010, 52, 794–800. [Google Scholar] [CrossRef][Green Version]
- Nazarov, V.G.; Volynskii, A.L.; Yarysheva, L.M.; Stolyarov, V.P.; Bakeev, N.F. Transformation of the modified layer of fluorinated polyolefins under stretching. Polym. Sci. Ser. A 2012, 54, 679–683. [Google Scholar] [CrossRef]
- Nazarov, V.G.; Stolyarov, V.P.; Novikova, S.M.; Mikhaleva, L.A.; Bablyuk, E.B.; Benda, A.P. Influence of surface fluorination on characteristics of polymeric materials. Proc. Inst. High. Educ. Issues Graph. Arts Publ. 2011, 2, 118–127. [Google Scholar]
- Maity, J.; Jacob, C.; Das, C.K.; Alam, S.; Singh, R.P. Homocomposites of chopped fluorinated polyethylene fiber with low-density polyethylene matrix. Mater. Sci. Eng. A 2008, 479, 125–135. [Google Scholar] [CrossRef]
- Maity, J.; Jacob, C.; Das, C.K.; Alam, S.; Singh, R. Direct fluorination of UHMWPE fiber and preparation of fluorinated and non-fluorinated fiber composites with LDPE matrix. Polym. Test. 2008, 27, 581–590. [Google Scholar] [CrossRef]
- Kudinov, V.V.; Buznik, V.M.; Kharitonov, A.P. Method of Obtaining Preparation for Composite Materials. RU Patent 2516526 C2, 20 May 2014. [Google Scholar]
- Pouzet, M.; Dubois, M.; Charlet, K.; Beakou, A. From hydrophilic to hydrophobic wood using direct fluorination: A localized treatment. Comptes Rendus Chim. 2018, 21, 800–807. [Google Scholar] [CrossRef]
- Mukherjee, M.; Das, C.; Kharitonov, A. Fluorinated and oxyfluorinated short Kevlar fiber-reinforced ethylene propylene polymer. Polym. Compos. 2006, 27, 205–212. [Google Scholar] [CrossRef]
- Wirti, M.; Biondo, G.R.R.; Romanzini, D.; Amico, S.C.; Zattera, A.J. The effect of fluorination of aramid fibers on vinyl ester composites. Polym. Compos. 2018, 40, 2095–2102. [Google Scholar] [CrossRef]
- Nazarov, V.G.; Nagornova, I.V.; Stolyarov, V.P.; Doronin, F.A.; Evdokimov, A.G.; Brevnov, P.N.; Zabolotnov, A.S.; Novokshonova, L.A. Effect of fluorination of ultrahigh-molecular-weight polyethylene. Russ. J. Phys. Chem. 2018, 12, 1066–1075. [Google Scholar] [CrossRef]
- Nazarov, V.G.; Stolyarov, V.P.; Doronin, F.A.; Evdokimov, A.G.; Rytikov, G.O.; Brevnov, P.N.; Zabolotnov, A.S.; Novokshonova, L.A.; Berlin, A.A. Comparison of the effects of some modification methods on the characteristics of ultrahigh-molecular-weight polyethylene and composites on its basis. Polym. Sci. Ser. A 2019, 61, 325–333. [Google Scholar] [CrossRef]
- Li, B.Y.; Fan, C.; Ren, M.M.; Wu, P.; Luo, L.B.; Wang, X.; Liu, X.Y. Surface modified UHMWPE particles by direct fluorination blended with polyurethane for enhancing its wear-resistant performance. In Materials Science Forum; Trans Tech Publications Ltd.: Freienbach, Switzerland, 2015; Volume 815, pp. 489–495. [Google Scholar]
- Li, B.; Li, M.; Fan, C.; Ren, M.; Wu, P.; Luo, L.; Wang, X.; Liu, X. The wear-resistance of composite depending on the interfacial interaction between thermoplastic polyurethane and fluorinated UHMWPE particles with or without oxygen. Compos. Sci. Technol. 2015, 106, 68–75. [Google Scholar] [CrossRef]
- Jia, E.; Yang, X.; Liu, X.; Ye, G.; Xu, J. Surface fluorination of poly(p-phenylene-1,3,4-oxadiazole) fiber. Acta Polym. Sin. 2012, 12, 385–390. [Google Scholar] [CrossRef]
- Maity, J.; Jacob, C.; Das, C.; Alam, S.; Singh, R. Direct fluorination of Twaron fiber and the mechanical, thermal and crystallization behaviour of short Twaron fiber reinforced polypropylene composites. Compos. Part A Appl. Sci. Manuf. 2008, 39, 825–833. [Google Scholar] [CrossRef]
- Mukherjee, M.; Das, C.K.; Kharitonov, A.P. Influence of fluorinated and oxy-fluorinated short kevlar fiber loading on the properties of ethylene propylene matrix composites. Mater. Manuf. Process. 2006, 21, 892–898. [Google Scholar] [CrossRef]
- Mukherjee, M.; Das, C.K.; Kharitonov, A.P.; Banik, K.; Mennig, G.; Chung, T.N. Properties of syndiotactic polystyrene composites with surface modified short Kevlar fiber. Mater. Sci. Eng. 2006, 441, 206–214. [Google Scholar] [CrossRef]
- Mukherjee, M.; Das, C.; Kharitonov, A. Effect of compatibilizer on the properties of fluorinated and oxy-fluorinated short kevlar fiber reinforced EP. J. Reinf. Plast. Compos. 2008, 27, 523–539. [Google Scholar] [CrossRef]
- Lv, J.; Cheng, Z.; Wu, H.; He, T.; Qin, J.; Liu, X. In-situ polymerization and covalent modification on aramid fiber surface via direct fluorination for interfacial enhancement. Compos. B Eng. 2020, 182, 107608. [Google Scholar] [CrossRef]
- Krestinin, A.V.; Kharitonov, A.P. Covalent functionalization of single-walled carbon nanotubes through the fluorination stage for integration into an epoxy composite. Polym. Sci. Ser. B 2018, 60, 516–529. [Google Scholar] [CrossRef]
- Zhao, N.; Nie, Y.; Li, S. Space charge characteristics of fluorinated polyethylene: Different effects of fluorine and oxygen. AIP Adv. 2018, 8, 045103. [Google Scholar] [CrossRef]
- Li, S. Improvement of surface flashover in vacuum. High Volt. 2020, 5, 122–133. [Google Scholar] [CrossRef]
- An, Z.; Chen, X.; Liu, C.; Zheng, F.; Zhang, Y. Significantly reduced fluorination time needed for suppression of space charge in polyethylene by increasing the fluorination temperature. J. Phys. D Appl. Phys. 2012, 45, 385303. [Google Scholar] [CrossRef]
- Liu, Y.; An, Z.; Cang, J.; Zhang, Y.; Zheng, F. Significant suppression of surface charge accumulation on epoxy resin by direct fluorination. IEEE Trans. Dielectr. Electr. Insul. 2012, 19, 1143–1150. [Google Scholar] [CrossRef]
- Zhou, R.D.; Sun, G.Y.; Song, B.P.; Guo, B.H.; Yang, N.; Mu, H.B.; Zhang, G.J. Mechanism of F2/N2 fluorination mitigating vacuum flashover of polymers. J. Phys. D Appl. Phys. 2019, 52, 375304. [Google Scholar] [CrossRef]
- An, Z.; Yin, Q.; Liu, Y.; Zheng, F.; Lei, Q.; Zhang, Y. Modulation of surface electrical properties of epoxy resin insulator by changing fluorination temperature and time. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 526–534. [Google Scholar] [CrossRef]
- Que, L.; An, Z.; Ma, Y.; Xie, D.; Zheng, F.; Zhang, Y. Improved DC flashover performance of epoxy insulators in SF6 gas by direct fluorination. IEEE Trans. Dielectr. Electr. Insul. 2017, 24, 1153–1161. [Google Scholar] [CrossRef]
- Mohamad, A.; Chen, G.; Zhang, Y.; An, Z. Surface fluorinated epoxy resin for high voltage DC application. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 101–108. [Google Scholar] [CrossRef]
- Du, B.X.; Li, X.L.; Jiang, J.P. Surface charge accumulation and decay on directfluorinated oil-impregnated paper. IEEE Trans. Dielectr. Electr. Insul. 2016, 23, 3094–3101. [Google Scholar] [CrossRef]
- Gangal, S.V.; Brothers, P.D. Perfluorinated Polymers. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Syed, H.Z. Aging Characteristics of Polytetrafluoroethylene (PTFE). Master’s Thesis, University of Windsor, Windsor, ON, Canada, 2002. [Google Scholar]
- Zhao, N.; Nie, Y.; Li, S.; Fu, M. Space charge characteristics of low density polyethylene oxyfluorinated with high concentrations of oxygen. In Proceedings of the 2015 IEEE 11th International Conference on the Properties and Applications of Dielectric Materials (ICPADM), Sydney, NSW, Australia, 19–22 July 2015. [Google Scholar]
- Jiang, Y.; An, Z.; Liu, C.; Zheng, F.; Zhang, Y. Influence of oxyfluorination time on space charge behavior in polyethylene. IEEE Trans. Dielectr. Electr. Insul. 2010, 17, 1814–1823. [Google Scholar] [CrossRef]
- An, Z.; Yang, Q.; Xie, C.; Jiang, Y.; Zheng, F.; Zhang, Y. Suppression effect of surface fluorination on charge injection into linear low density polyethylene. J. Appl. Phys. 2009, 105, 064102. [Google Scholar] [CrossRef]
- Du, B.X.; Zhu, W.B.; Li, X.L. Effects of direct fluorination on charge coupling behavior of oil-paper insulation under DC and pulse voltages. IEEE Trans. Dielectr. Electr. Insul. 2017, 24, 947–955. [Google Scholar] [CrossRef]
- Du, B.X.; Xing, Y.Q.; Jin, J.X.; Huang, P.H.; Xiao, M. Surface potential behavior of direct-fluorinated PPLP under quench condition. IEEE Trans. Appl. Supercond. 2016, 26, 1–5. [Google Scholar] [CrossRef]
- Mohamad, A. Surface Fluorinated Epoxy Resin for High Voltage DC Application. Ph.D. Thesis, University of Southampton, Southampton, UK, 2015. [Google Scholar]
- Mohamad, A.; Chen, G.; Chen, Q. Investigation of plasma-enhanced-fluorination treatment on epoxy resins for high-voltage DC applications. IEEE Trans. Dielectr. Electr. Insul. 2017, 24, 3786–3793. [Google Scholar] [CrossRef]
- An, Z.; Yang, Q.; Xie, C.; Jiang, Y.; Zheng, F.; Zhang, Y. Suppression of charge injection to linear low density polyethylene by surface fluorination modification. In Proceedings of the 2008 International Symposium on Electrical Insulating Materials (ISEIM 2008), Mie, Japan, 7–11 September 2008. [Google Scholar]
- Kong, F.; Chang, C.; Ma, Y.; Zhang, C.; Ren, C.; Shao, T. Surface modifications of polystyrene and their stability: A comparison of DBD plasma deposition and direct fluorination. Appl. Surf. Sci. 2018, 459, 300–308. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Q.; Du, X. Effect of direct fluorination on surface and electrical properties of polyimide thin films. Mater. Lett. 2018, 223, 207–209. [Google Scholar] [CrossRef]
- An, Z.; Shan, F.; Yang, L.; Shen, R.; Gu, X.; Zheng, F.; Zhang, Y. Unusual effect of temperature on direct fluorination of high temperature vulcanized silicone rubber and properties of the fluorinated surface layers. IEEE Trans. Dielectr. Electr. Insul. 2018, 25, 190–198. [Google Scholar] [CrossRef]
- Pud, A.A.; Rogalsky, S.P.; Shapoval, G.S.; Kharitonov, A.P.; Teplyakov, V.V.; Strathmann, H.; Poncin-Epaillard, F. Reactions and stability of fluorinated poly (vinyl trimethylsilane) in electrochemical systems. Polymer 2001, 42, 1907–1913. [Google Scholar] [CrossRef]
- Rogal’skii, S.P.; Pud, A.A.; Shapoval, G.S.; Mel’nik, A.F.; Bryk, M.T. Nature of initiators for indirect electrochemical reductive degradation of polycarbonates in dimethylformamide. Theor. Exp. Chem. 2004, 40, 33–38. [Google Scholar] [CrossRef]



| Fluorinated Object | θ1, Degrees | θ2, Degrees | θ, Degrees | φ, % | Ref. |
|---|---|---|---|---|---|
| Aramid fiber | 0 | 113 | 103 | 12 | [71] |
| 0 | 117 | 97 | 23 | [84] | |
| PP (+TiO2) (composite) | 0 | 101 | 67 | 49 | [30] |
| Wood flour + polyester (composite) | 120 | 76 | 84 | 19 | [97] |
| PP (non-woven fabric) | 120 | 77 | 86 | 21 | [99] |
| 0 | 64 | 28 | |||
| Wood fiber | 120 | 51 | 98 | 68 | [94] |
| 120 | 65 | 120 | 100 | [74] | |
| Cotton fiber | 120 | 0 | 117 | 97 | [95] |
| Aminated polyetherimide (film) | 120 | 0 | 86 | 62 | [110] |
| Polybutylene terephthalate (fiber) | 170 | 126 | 156 | 82 | [32] |
| Silicone rubber (plate) | 170 | 110 | 144 | 73 | [46] |
| Carbon fiber | 170 | 110 | 153 | 85 | [34] |
| Fluorinated Object | Dispersion γdSV | Polar γpSV | Total γSV |
|---|---|---|---|
| PE | 32.0 | 1.1 | 33.1 |
| PP | 30.1 | 0 | 30.1 |
| PVF | 31.3 | 5.4 | 36.7 |
| PVDF | 23.2 | 7.1 | 30.3 |
| PTFE | 18.6 | 0.5 | 19.1 |
| 18.4 | 1.7 | 20.1 | |
| PETP | 37.8 | 3.5 | 41.3 |
| 35.6 | 9.0 | 44.6 | |
| PMMA | 35.9 | 4.3 | 40.2 |
| 29.6 | 11.5 | 41.1 |
| Polymer Matrix | Filler | Fluorination Procedure | E1, MPa | σ1, MPa | ε1, % | Ref. |
|---|---|---|---|---|---|---|
| LDPE | - | 15% F2 + He; 3 h | 72.6/111.2 | 20.5/16.3 | 647/634 | [129] |
| LDPE | UHMWPE—short fibers | 5% F2 + He; 1 h (fibers)/20% F2 + He; 1 h (surface) | 458/704/765 | 11.21/21.16/24.03 | 18.15/8.06/5.71 | [130] |
| LDPE | UHMWPE—short fibers | 10% F2 + He; 2 h | 248/737 | 10.00/28.72 | 20.31/4.25 | [131] |
| HDPE | - | 15% F2 + He; 3 h | 310/670 | 56/44 | 520/460 | [128] |
| TPU | UHMWPE—particles of 50–70 microns | 10% F2 + N2 | - | 12.5/22.3 | 523.5/892.1 | [103] |
| TPU | UHMWPE—particles of 50–70 microns | 10% F2 + N2/9% F2 + 14% O2 + N2 | - | 12.5/16.5/23.7 | 523.5/645.1/956.7 | [103] |
| - | UHMWPE—fibers | 5% F2 + He; 1 h | - | 621/797 | - | [132] |
| NBR | - | 10% F2 + N2; 1 h | - | 16/17 | - | [120] |
| NBR | - | F2 + He; 5 h /100 °C | 11.7/12.4 | 21.1/16.1 | 376.1/176.2 | [44] |
| Norsodyne G703 | wood flour | 10% F2 + N2; 3 h | 4400/4800 | 32.4/41.7 | 1.4/1.3 | [97,133] |
| EP (10/90) | Kevlar | 5% F2 + 5% air + He/5% F2 + 1% O2 + 4% N2 + He | 400/560/680 | 20/30/33 | 6.85/5.13/4.02 | [134] |
| PP | Twaron | 5% F2 + He; 1.5 h | - | 24.74/30.76 | 4.64/5.84 | [24] |
| PP | Twaron | 10% F2 + He; 2 h | 1144/1480 | 27.55/31.65 | 4.6/4.5 | [133] |
| Derakane 411–350 Ashland vinyl ester resin | Kevlar | HF | - | 364/115 | - | [135] |
| PP | Illite | F2 | - | 35/38 | - | [124] |
| ED-22 | Taunit-M CNTs, 0.1% | F2; 150 °C/250 °C | - | 77.4/89.6/69.8 | - | [113] |
| Material | (increase in %) | Ref. | ||||
|---|---|---|---|---|---|---|
| PE | - | - | - | - | 24.5 (+51%) vacuum | [151] |
| - | - | 2.4 | 6 | - | [147] | |
| Epoxy resin | 5.6 × 10−18 | 2.4 × 10−14 | - | - | - | [150] |
| 8 × 10−18 | 4 × 10−14 | - | - | - | [152] | |
| 6.2 × 10−20 | 1.25 × 10−15 | - | - | - | [58] | |
| 1.5 × 10−18 | 2.3 × 10−14 | - | - | 91.9 (+12%) SF4 | [153] | |
| - | - | 27.8 (+26%) | [59] | |||
| - | - | 15.1 (+26%) | [154] | |||
| - | - | 21.11 (+22%) | [151] | |||
| Al2O3-filled epoxy resin | 9.7 × 10−22 | 3.7 × 10−16 | - | - | - | [57] |
| 7.7 × 10−18 | 7.9 × 10−16 | - | - | - | [58] | |
| 8.8 × 10−19 | 3.68 × 10−16 | - | - | 21.1 (+7%) air 28.6 (+4%) SF4 | [60,61] | |
| - | - | - | - | 25.5 (+16%) | [59] | |
| PDMS | 3.7 × 10−18 | 4.4 × 10−14 | - | - | [54] | |
| 3.6 | 3.1 | 15.6(+4%) | [51] | |||
| Oil-impregnated paper | 5 × 10−13 | 2 × 10−11 | - | - | - | [155] |
| Polyimide | - | - | 3.3 | 2.7 | - | [48] |
| - | - | 3.1 | 2.5 | - | [49] | |
| - | - | 3.4 | 2.8 | - | [50] | |
| PTFE | <10−16 | - | 2.1 | - | 25.3 | [156,157] |
| FEP | <10−15 | - | 2 | - | - | |
| PFA | 10−18 | - | 2.03 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belov, N.A.; Alentiev, A.Y.; Bogdanova, Y.G.; Vdovichenko, A.Y.; Pashkevich, D.S. Direct Fluorination as Method of Improvement of Operational Properties of Polymeric Materials. Polymers 2020, 12, 2836. https://doi.org/10.3390/polym12122836
Belov NA, Alentiev AY, Bogdanova YG, Vdovichenko AY, Pashkevich DS. Direct Fluorination as Method of Improvement of Operational Properties of Polymeric Materials. Polymers. 2020; 12(12):2836. https://doi.org/10.3390/polym12122836
Chicago/Turabian StyleBelov, Nikolay A., Alexander Y. Alentiev, Yulia G. Bogdanova, Artem Y. Vdovichenko, and Dmitrii S. Pashkevich. 2020. "Direct Fluorination as Method of Improvement of Operational Properties of Polymeric Materials" Polymers 12, no. 12: 2836. https://doi.org/10.3390/polym12122836
APA StyleBelov, N. A., Alentiev, A. Y., Bogdanova, Y. G., Vdovichenko, A. Y., & Pashkevich, D. S. (2020). Direct Fluorination as Method of Improvement of Operational Properties of Polymeric Materials. Polymers, 12(12), 2836. https://doi.org/10.3390/polym12122836