Valorisation of Posidonia oceanica Sea Balls (Egagropili) as a Potential Source of Reinforcement Agents in Protein-Based Biocomposites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
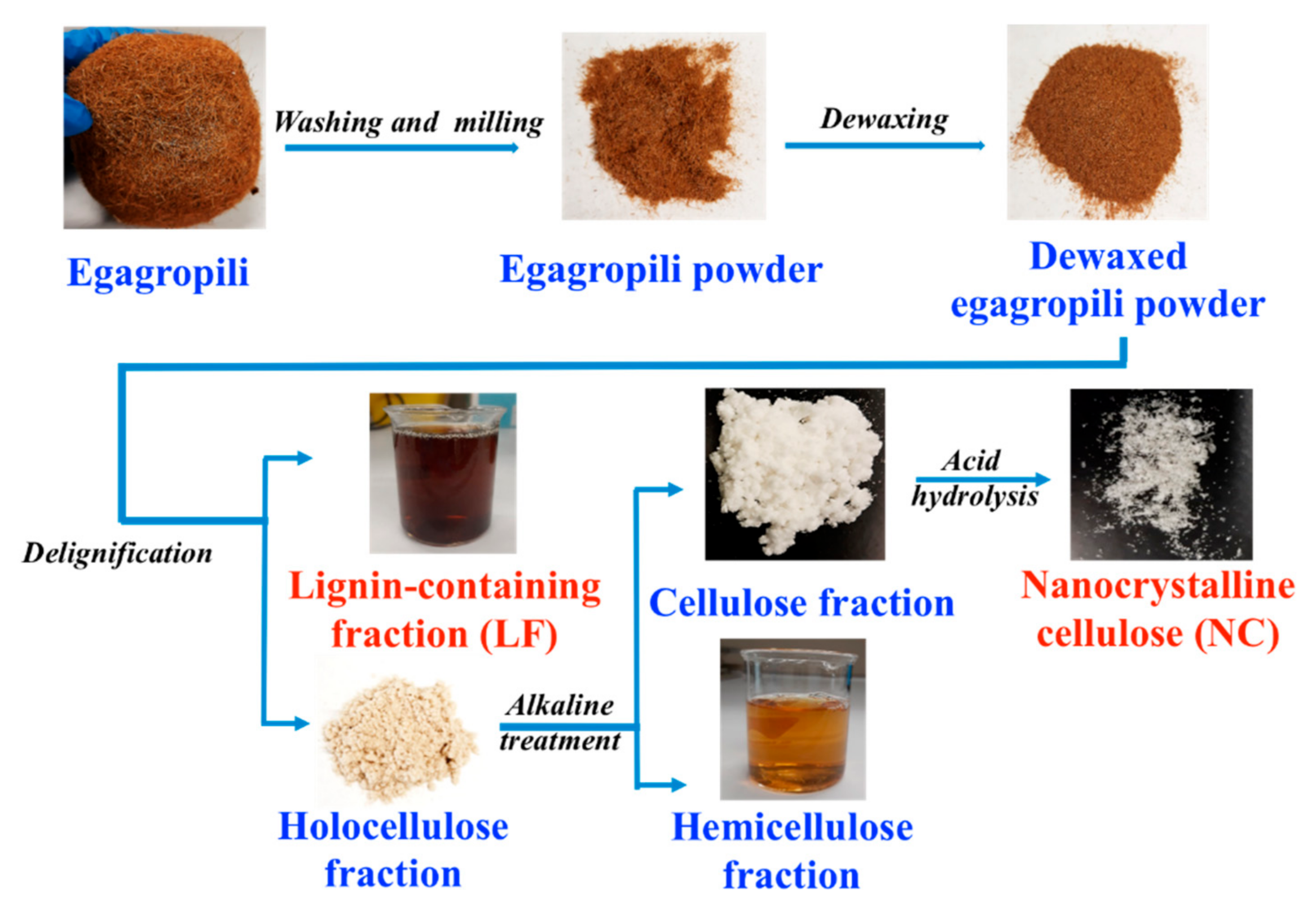
2.2. Egagropili Lignin Containing Fraction and Nanocrystalline Cellulose
2.3. Preparation of Protein-Based Composite Films
2.4. Zeta Potential and Particle Size Measurements
2.5. Film Mechanical Properties
2.6. Film Moisture Content, Swelling Ratio and Solubility
2.7. Film Water Vapor, O2 and CO2 Permeability
2.8. Scanning Electron Microscopy (SEM)
2.9. Statistical Analysis
3. Results and Discussion
3.1. Egagropili as Potential Source of Lignin and Nanocrystalline Cellulose
3.2. Surface Microstructure of Egagropili Fibers and the Derived Nanocrystalline Cellulose
3.3. Film Forming Solution Zeta Potential and Particle Size Measurements
3.4. Hemp Protein-Based Films
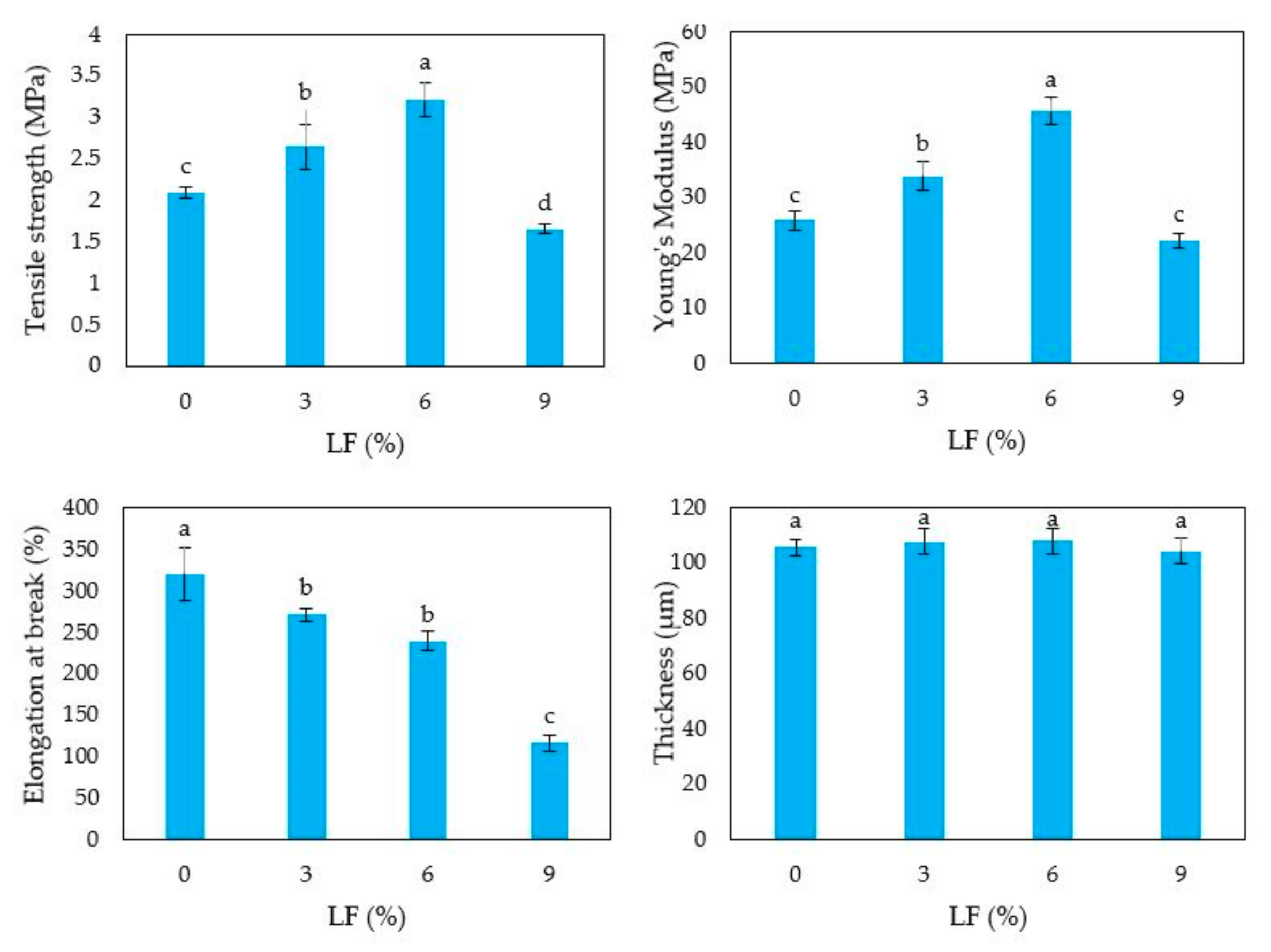
3.5. Film Mechanical Properties
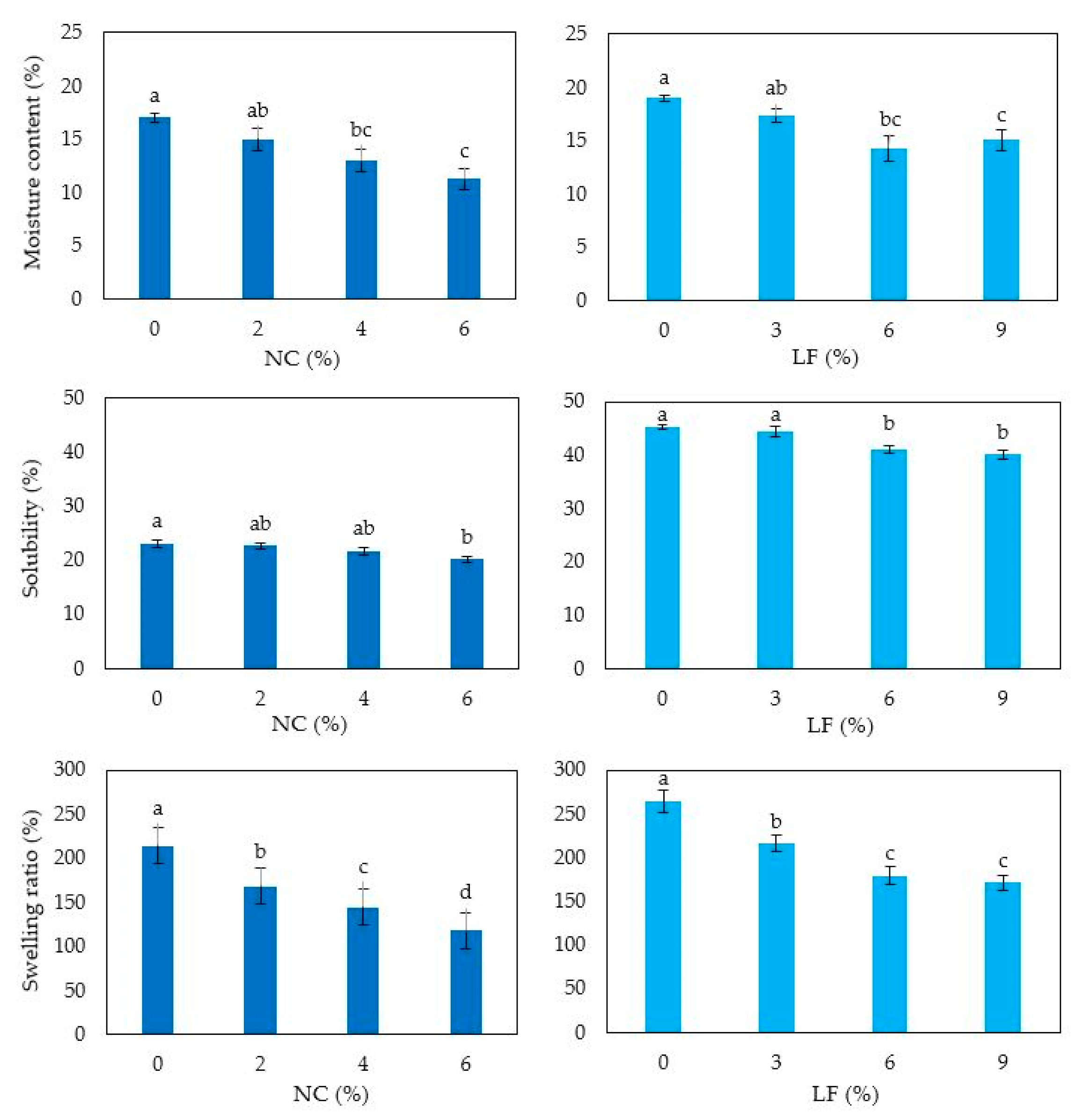
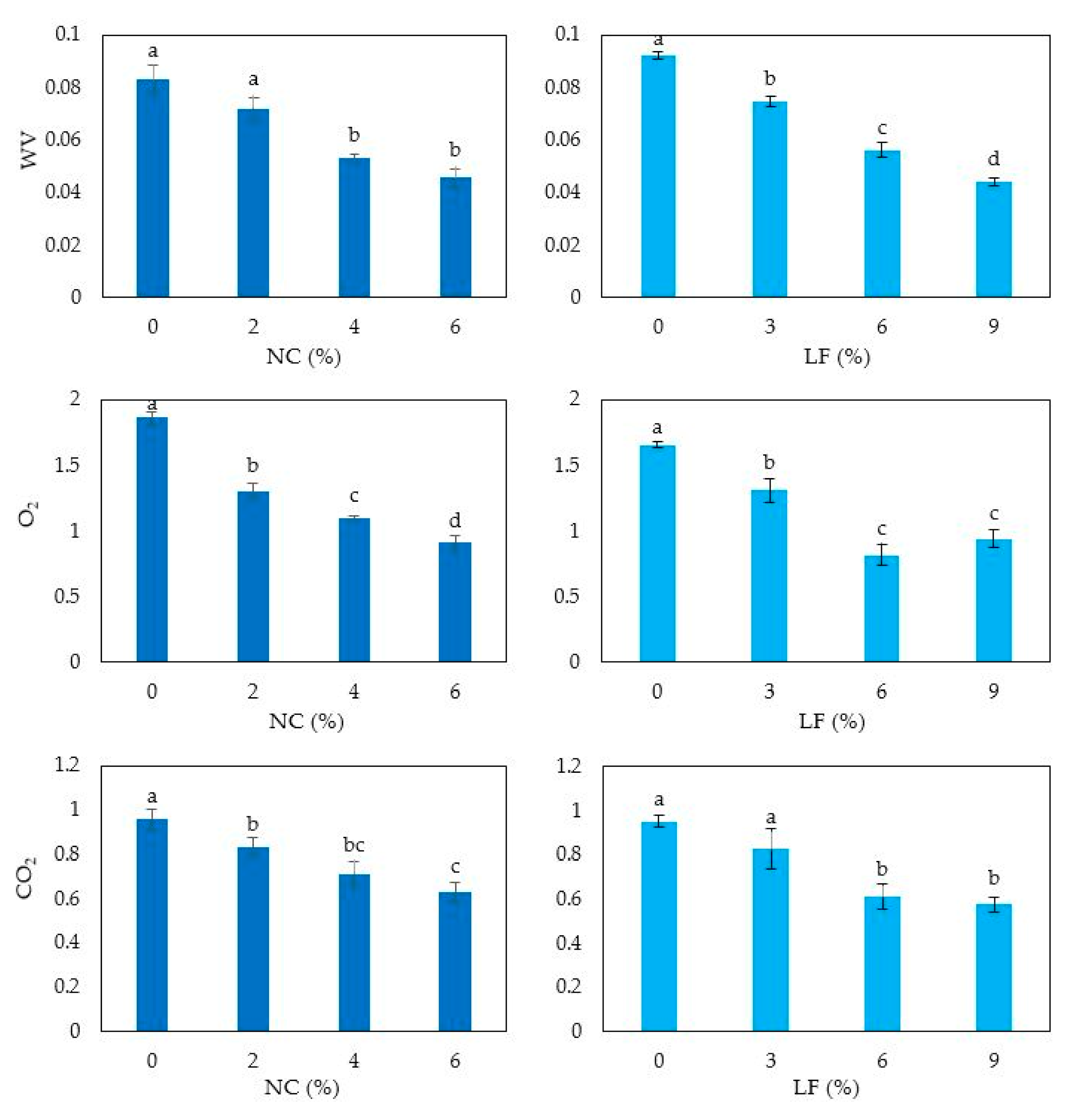
3.6. Film Moisture Content, Solubility, Swelling Ratio and Permeability
3.7. Film Morphological Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Porta, R. The plastics sunset and the bio-plastics sunrise. Coatings 2019, 9, 526. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Ching, Y.C.; Chuah, C.H. Applications of lignocellulosic fibers and lignin in bioplastics: A review. Polymers 2019, 11, 751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Phanthong, P.; Reubroycharoen, P.; Hao, X.; Xu, G.; Abudula, A.; Guan, G. Nanocellulose: Extraction and application. Carbon Resour. Convers. 2018, 1, 32–43. [Google Scholar] [CrossRef]
- Keijsers, E.R.P.; Yılmaz, G.; van Dam, J.E.G. The cellulose resource matrix. Carbohydr. Polym. 2013, 93, 9–21. [Google Scholar] [CrossRef]
- Den Hartog, C.; Kuo, J. Taxonomy and biogeography of seagrasses. In Seagrasses: Biology, Ecology and Conservation; Larkum, A.W.D., Orth, R.J., Duarte, C.M., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 1–23. [Google Scholar]
- Fornes, A.; Basterretxea, G.; Orfila, A.; Jordi, A.; Alvarez, A.; Tintore, J. Mapping Posidonia oceanica from IKONOS. ISPRS J. Photogramm. 2006, 60, 315–322. [Google Scholar] [CrossRef]
- Benito-Gonzalez, I.; Lopez-Rubio, A.; Martınez-Sanz, M. Potential of lignocellulosic fractions from Posidonia oceanica to improve barrier and mechanical properties of bio-based packaging materials. Int. J. Biol. Macromol. 2018, 118, 542–551. [Google Scholar] [CrossRef]
- Benito-Gonzalez, I.; Lopez-Rubio, A.; Martınez-Sanz, M. High-performance starch biocomposites with celullose from waste biomass: Film properties and retrogradation behaviour. Carbohydr. Polym. 2019, 216, 180–188. [Google Scholar] [CrossRef]
- Bettaieb, F.; Khiari, R.; Hassan, M.L.; Belgacem, M.N.; Bras, J.; Dufresne, A.; Mhenni, M.F. Preparation and characterization of new cellulose nanocrystals from marine bio-mass Posidonia oceanica. Ind. Crops Prod. 2015, 72, 175–182. [Google Scholar] [CrossRef]
- Fortunati, E.; Luzi, F.; Puglia, D.; Petrucci, R.; Kenny, J.; Torre, L. Processing of PLA nanocomposites with cellulose nanocrystals extracted from Posidonia oceanica waste: Innovative reuse of coastal plant. Ind. Crops Prod. 2015, 67, 439–447. [Google Scholar] [CrossRef]
- Aguir, C.; M’Henni, M.F. Experimental study on carboxymethylation of cellulose extracted from Posidonia oceanica. J. Appl. Polym. Sci. 2006, 99, 1808–1816. [Google Scholar] [CrossRef]
- Khiari, R.; Mhenni, M.F.; Belgacem, M.N.; Mauret, E. Valorisation of vegetal wastes as a source of cellulose and cellulose derivatives. J. Polym. Environ. 2011, 19, 80–89. [Google Scholar] [CrossRef]
- Khiari, R.; Ferchichi, I.; Mhenni, M.F. Study of liquids absorption and retention capacities of new cellulosic materials and sodium cellulose carboxylmethylate prepared from Posidonia. Fibre Polym. 2010, 11, 593–597. [Google Scholar]
- Balestri, E.; Vallerini, F.; Lardicci, C. A qualitative and quantitative assessment of the reproductive litter from Posidonia oceanica accumulated on a sand beach following a storm. Estuar. Coast. Shelf Sci. 2006, 66, 30–34. [Google Scholar] [CrossRef]
- Coletti, A.; Valerio, A.; Vismara, E. Posidonia oceanica as a renewable lignocellulosic biomass for the synthesis of cellulose acetate and glycidyl methacrylate grafted cellulose. Materials 2013, 6, 2043–2058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seggiani, M.; Cinelli, P.; Mallegni, N.; Balestri, E.; Puccini, M.; Vitolo, S.; Lardicci, C.; Lazzeri, A. New bio-composites based on polyhydroxyalkanoates and Posidonia oceanica fibres for applications in a marine environment. Materials 2017, 10, 326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R. Isolation and characterization of nanocrystalline cellulose from sugar palm fibres (Arenga Pinnata). Carbohydr. Polym. 2018, 181, 1038–1051. [Google Scholar] [CrossRef] [PubMed]
- Sanyang, M.L.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Sahari, J. Effect of sugar palm-derived cellulose reinforcement on the mechanical and water barrier properties of sugar palm starch biocomposite films. Bioresources 2016, 1, 4134–4145. [Google Scholar] [CrossRef] [Green Version]
- AACC Approved Methods of Analysis, 11th Ed. Method 02-03.Nitrogen AACC Method 46-10.01 (ARCHIVED) Crude Protein-Improved Kjeldahl Method; Cereals & Grains Association: St. Paul, MN, USA.
- ASTM D882-Standard Test Method for Tensile Properties of Thin Plastic Sheeting; ASTM: Philadelphia, PA, USA, 1997.
- Zahedi, Y.; Fathi-Achachlouei, B.; Yousefi, A.R. Physical and mechanical properties of hybrid montmorillonite/zinc oxide reinforced carboxymethyl cellulose nanocomposites. Int. J. Biol. Macromol. 2018, 108, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Rhim, J.W.; Jaiswal, L. Bioactive agar-based functional composite film incorporated with copper sulfide nanoparticles. Food Hydrocolloid. 2019, 93, 156–166. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.W. Preparation of carbohydrate-based functional composite films incorporated with curcumin. Food Hydrocolloid. 2020, 98, 105302. [Google Scholar] [CrossRef]
- ASTM D3985-Standard Test Method for Oxygen Gas Transmission Rate through Plastic Film and Sheeting Using a Coulometric Sensor; ASTM: Philadelphia, PA, USA, 2005.
- ASTM F2476-13 Standard Test Method for the Determination of Carbon Dioxide Gas Transmission Rate (CO2TR) Through Barrier Materials Using an Infrared Detector; ASTM: Philadelphia, PA, USA, 2013.
- ASTM F1249-Standard Test Method for Water Vapor Transmission Rate through Plastic Film and Sheeting Using a Modulated Infrared Sensor; ASTM: Philadelphia, PA, USA, 2013.
- Xiao, Y.; Liu, Y.; Wang, X.; Li, M.; Lei, H.; Xu, H. Cellulose nanocrystals prepared from wheat bran: Characterization and cytotoxicity assessment. Int. J. Biol. Macromol. 2019, 140, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Tolbert, A.; Akinosho, H.; Khunsupat, R.; Naskar, A.K.; Ragauskas, A.J. Characterization and analysis of the molecular weight of lignin for biorefining studies. Biofuel. Bioprod. Bior. 2014, 8, 836–856. [Google Scholar] [CrossRef]
- Isikgor, F.H.; Becer, C.R. Lignocellulosic biomass: A sustainable platform for the production of bio-based chemicals and polymers. Polym. Chem. 2015, 6, 4497–4559. [Google Scholar] [CrossRef] [Green Version]
- Azadi, P.; Inderwildi, O.R.; Farnood, R.; King, D.A. Liquid fuels, hydrogen and chemicals from lignin: A critical review. Renew. Sust. Energ. Rev. 2013, 21, 506–523. [Google Scholar] [CrossRef]
- Liu, W.J.; Jiang, H.; Yu, H.Q. Thermochemical conversion of lignin to functional materials: A review and future directions. Green Chem. 2015, 17, 4888–4907. [Google Scholar] [CrossRef]
- Zhang, J.; Elder, T.J.; Pu, Y.; Ragauskas, A.J. Facile synthesis of spherical cellulose nanoparticles. Carbohydr. Polym. 2007, 69, 607–611. [Google Scholar] [CrossRef]
- Flaker, C.H.; Lourenço, R.V.; Bittante, A.M.; Sobral, P.J. Gelatin-based nanocomposite films: A study on montmorillonite dispersion methods and concentration. J. Food Eng. 2015, 167, 65–70. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential–what they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Zadeh, E.M.; O’Keefe, S.F.; Kim, Y.-T. Utilization of Lignin in Biopolymeric Packaging Films. ACS Omega 2018, 3, 7388–7398. [Google Scholar] [CrossRef] [Green Version]
- Alashwal, B.Y.; Bala, M.S.; Gupta, A.; Sharma, S.; Mishra, P. Improved properties of keratin-based bioplastic film blended with microcrystalline cellulose: A comparative analysis. J. King Saud Univ. Sci. 2020, 32, 853–857. [Google Scholar] [CrossRef]
- Xiao, Y.; Liu, Y.; Kang, S.; Wang, K.; Xu, H. Development and evaluation of soy protein isolate-based antibacterial nanocomposite films containing cellulose nanocrystals and zinc oxide nanoparticles. Food Hydrocolloid. 2020, 106, 105898. [Google Scholar] [CrossRef]
- Salari, M.; Khiabani, M.S.; Mokarram, R.R.; Ghanbarzadeh, B.; Kafil, H.S. Development and evaluation of chitosan based active nanocomposite films containing bacterial cellulose nanocrystals and silver nanoparticles. Food Hydrocolloid. 2018, 4, 414–423. [Google Scholar] [CrossRef]
- Agustin, M.B.; Ahmmad, B.; Alonzo, S.M.M.; Patriana, F.M. Bioplastic based on starch and cellulose nanocrystals from rice straw. J. Reinf. Plast. Compos. 2014, 33, 2205–2213. [Google Scholar] [CrossRef]
- Wang, X.; Niamat, U.; Sun, X.; Guo, Y.; Chen, L.; Li, Z.; Feng, X. Development and characterization of bacterial cellulose reinforced biocomposite films based on protein from buckwheat distiller’s dried grains. Int. J. Biol. Macromol. 2017, 96, 353–360. [Google Scholar] [CrossRef]




| FFS Additive | Mean Particle Size (d.nm) | Zeta-Potential (mV) | PDI (%) |
|---|---|---|---|
| None, pH 9 | 374.50 ± 14.50 a | −22.30 ± 1.55 a | 0.58 ± 0.06 a |
| +2% NC | 397.40 ± 10.40 a,b | −21.50 ± 1.68 a | 0.56 ± 0.03 a |
| +4% NC | 466.50 ± 14.90 b,c | −20.20 ± 1.30 a | 0.58 ± 0.03 a |
| +6% NC | 552.20 ± 37.54 c | −21.80 ± 1.15 a | 0.61 ± 0.02 a |
| None, pH 12 | 324.30 ± 8.83 a | −28.50 ± 1.15 b | 0.54 ± 0.16 a |
| +3% LF | 389.80 ± 10.69 b | −27.60 ± 1.06 b | 0.61 ± 0.31 a |
| +6% LF | 394.90 ± 14.35 b | −27.10 ± 2.38 b | 0.65 ± 0.25 a |
| +9% LF | 312.70 ± 17.18 a | −28.90 ± 1.76 b | 0.64 ± 0.29 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirpoor, S.F.; Giosafatto, C.V.L.; Di Pierro, P.; Di Girolamo, R.; Regalado-González, C.; Porta, R. Valorisation of Posidonia oceanica Sea Balls (Egagropili) as a Potential Source of Reinforcement Agents in Protein-Based Biocomposites. Polymers 2020, 12, 2788. https://doi.org/10.3390/polym12122788
Mirpoor SF, Giosafatto CVL, Di Pierro P, Di Girolamo R, Regalado-González C, Porta R. Valorisation of Posidonia oceanica Sea Balls (Egagropili) as a Potential Source of Reinforcement Agents in Protein-Based Biocomposites. Polymers. 2020; 12(12):2788. https://doi.org/10.3390/polym12122788
Chicago/Turabian StyleMirpoor, Seyedeh Fatemeh, Concetta Valeria L. Giosafatto, Prospero Di Pierro, Rocco Di Girolamo, Carlos Regalado-González, and Raffaele Porta. 2020. "Valorisation of Posidonia oceanica Sea Balls (Egagropili) as a Potential Source of Reinforcement Agents in Protein-Based Biocomposites" Polymers 12, no. 12: 2788. https://doi.org/10.3390/polym12122788
APA StyleMirpoor, S. F., Giosafatto, C. V. L., Di Pierro, P., Di Girolamo, R., Regalado-González, C., & Porta, R. (2020). Valorisation of Posidonia oceanica Sea Balls (Egagropili) as a Potential Source of Reinforcement Agents in Protein-Based Biocomposites. Polymers, 12(12), 2788. https://doi.org/10.3390/polym12122788








