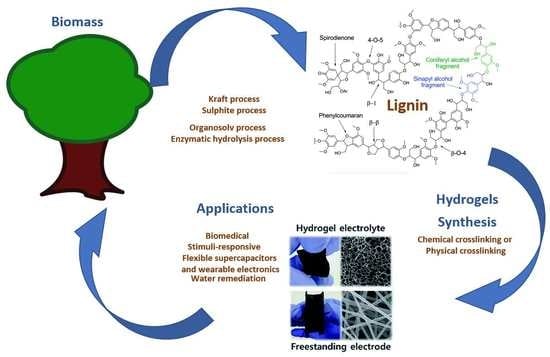
Lignin-Based Hydrogels: Synthesis and Applications
Abstract
1. Introduction
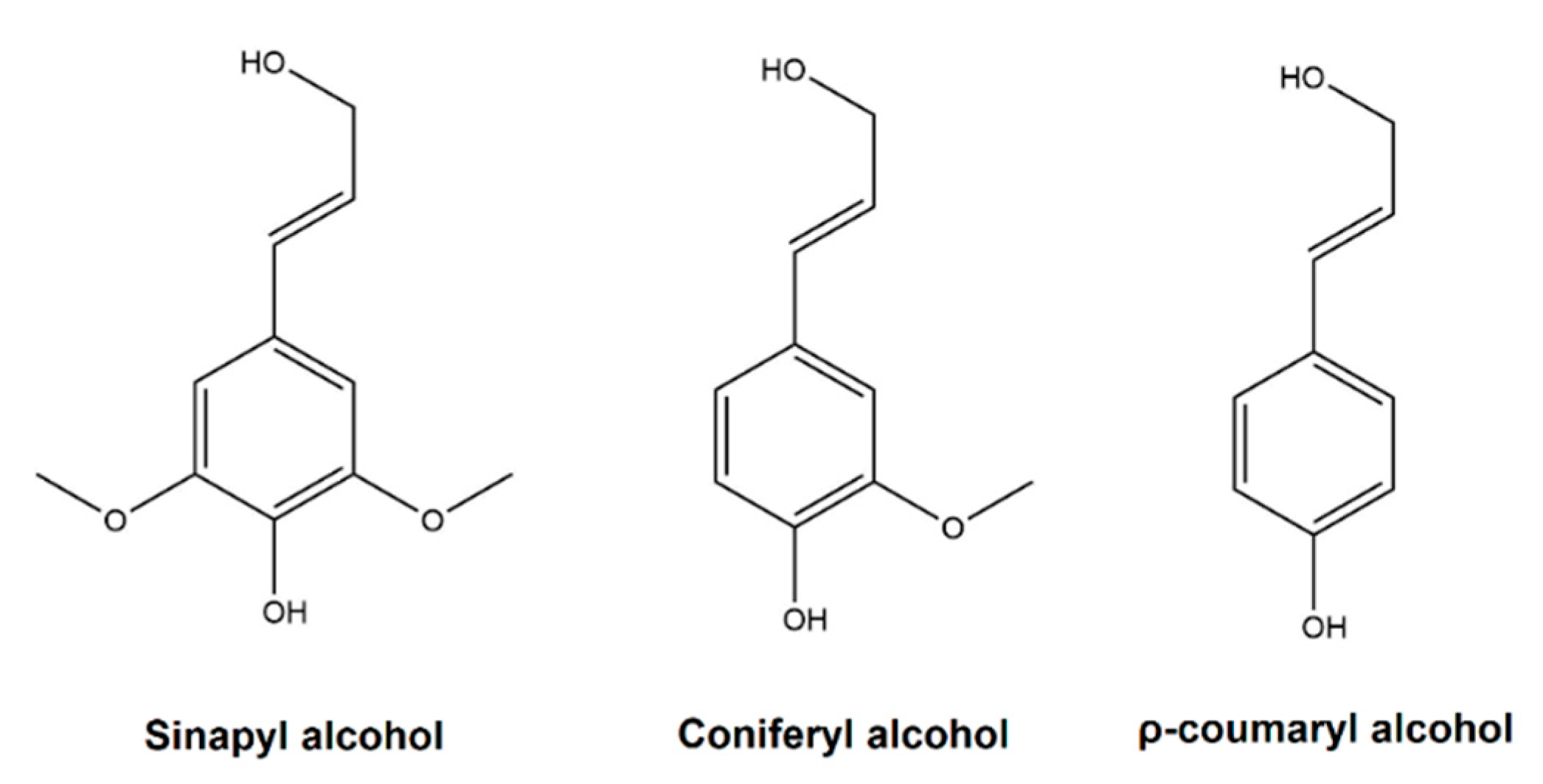
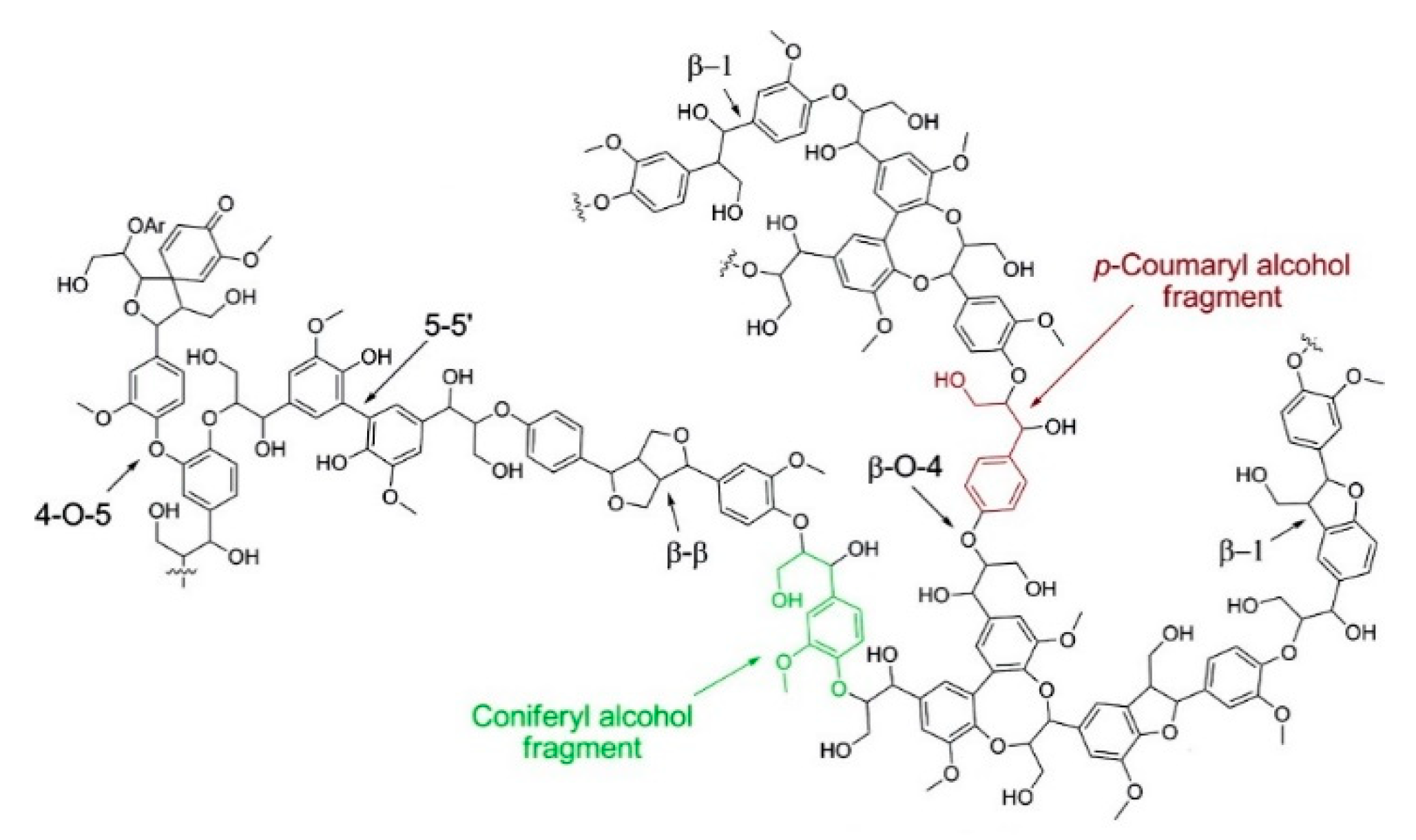
1.1. Lignin
1.1.1. Kraft Process
1.1.2. Sulphite Process
1.1.3. Organosolv Process
1.1.4. Enzymatic Hydrolysis Process
1.2. Hydrogels
2. Synthesis of Lignin-Based Hydrogels
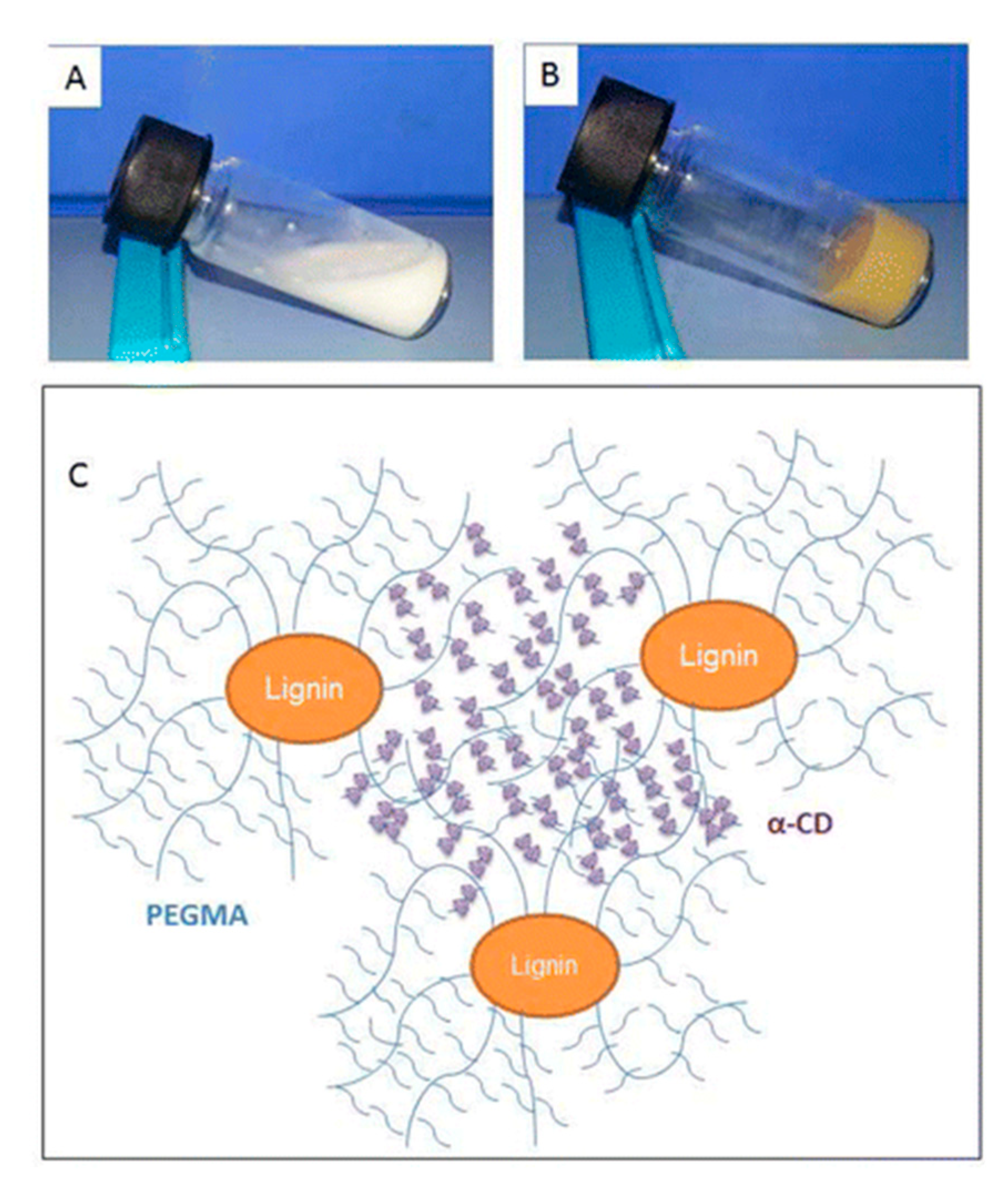
2.1. Lignin Hydrogels by Physical Interaction
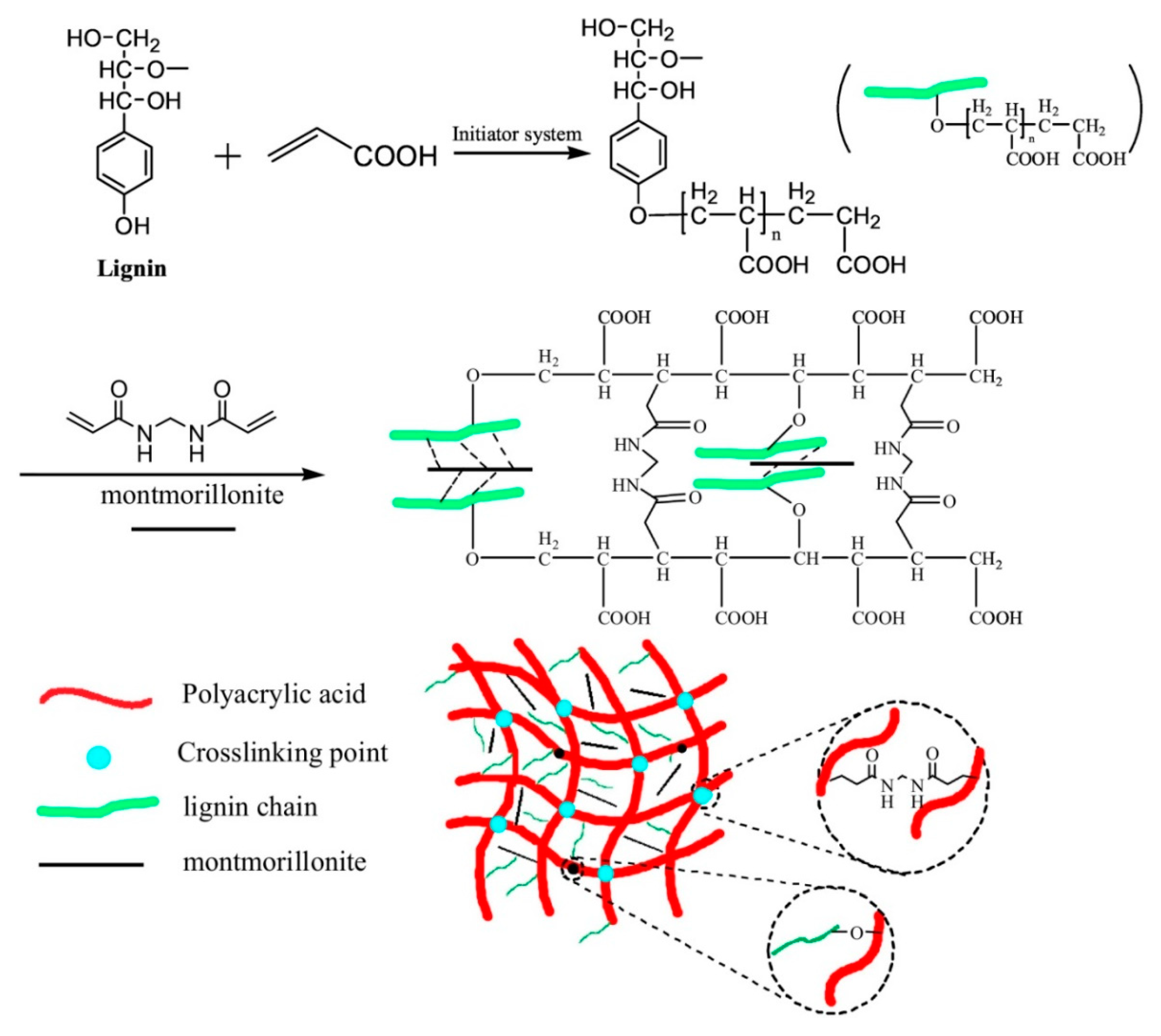
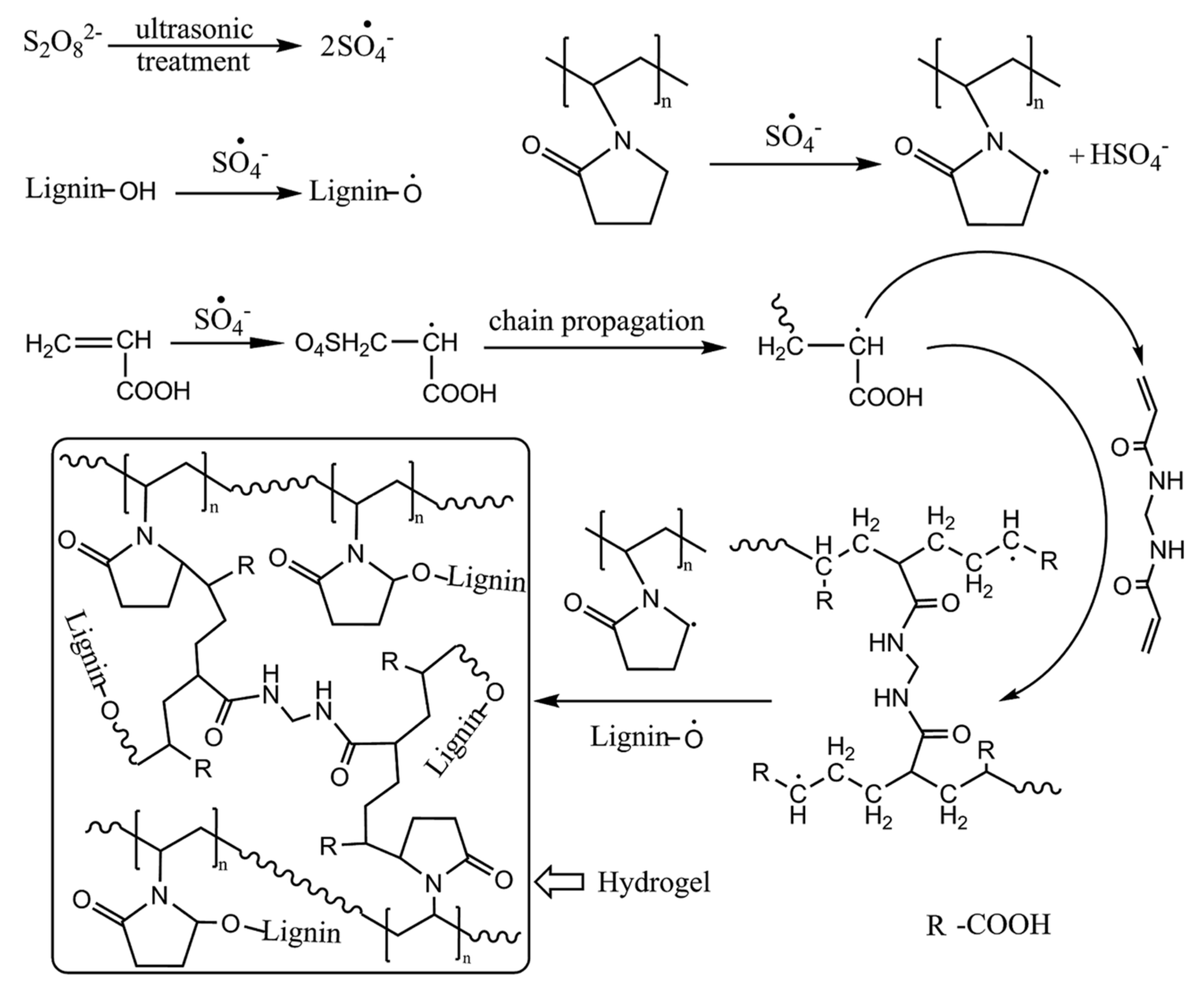
2.2. Lignin Hydrogels by Chemical Interaction
3. Applications
3.1. Stimuli-Responsive and Smart Hydrogels
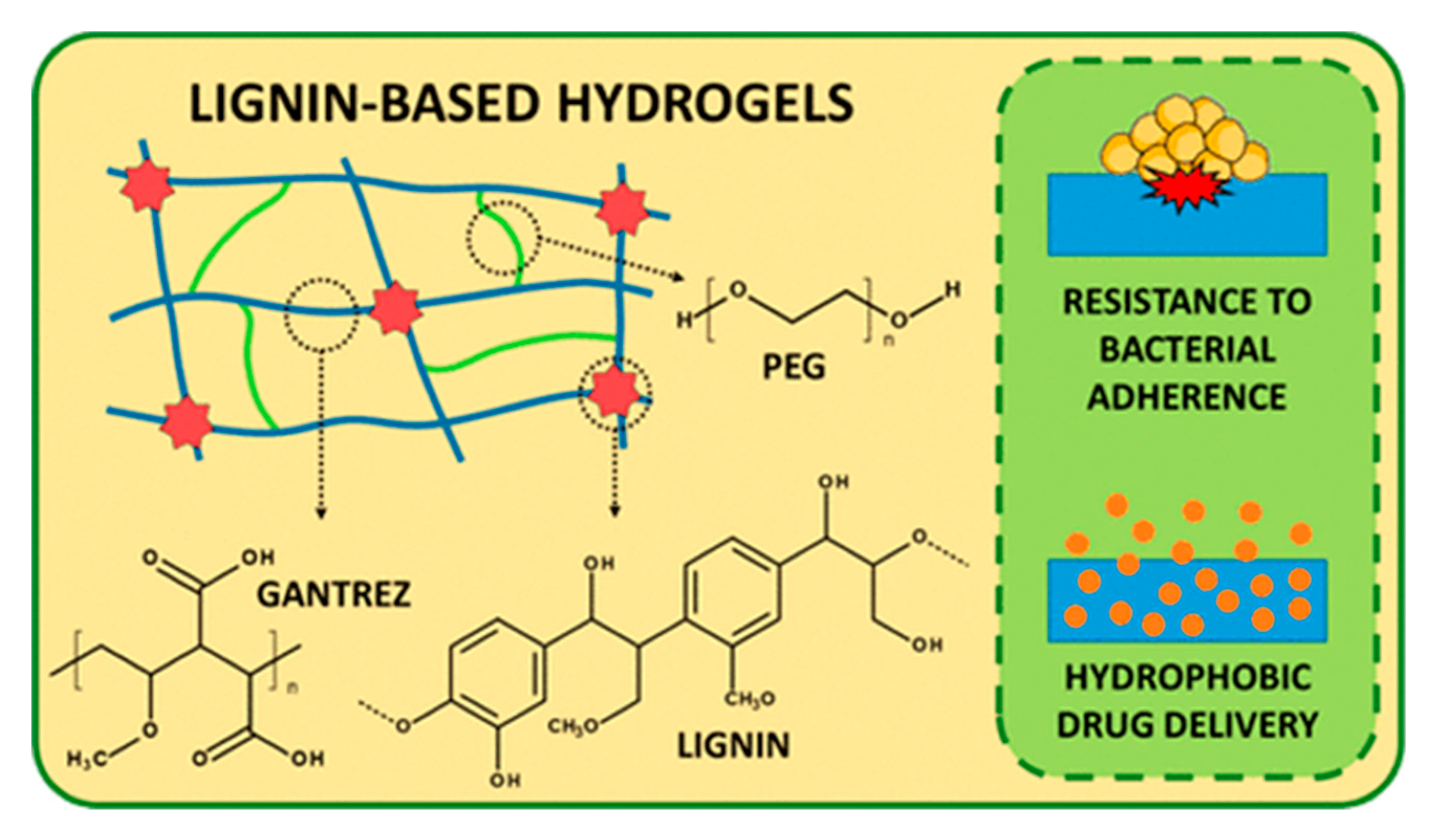
3.2. Hydrogels for Biomedical Applications
3.3. Hydrogels fo Water Remediation
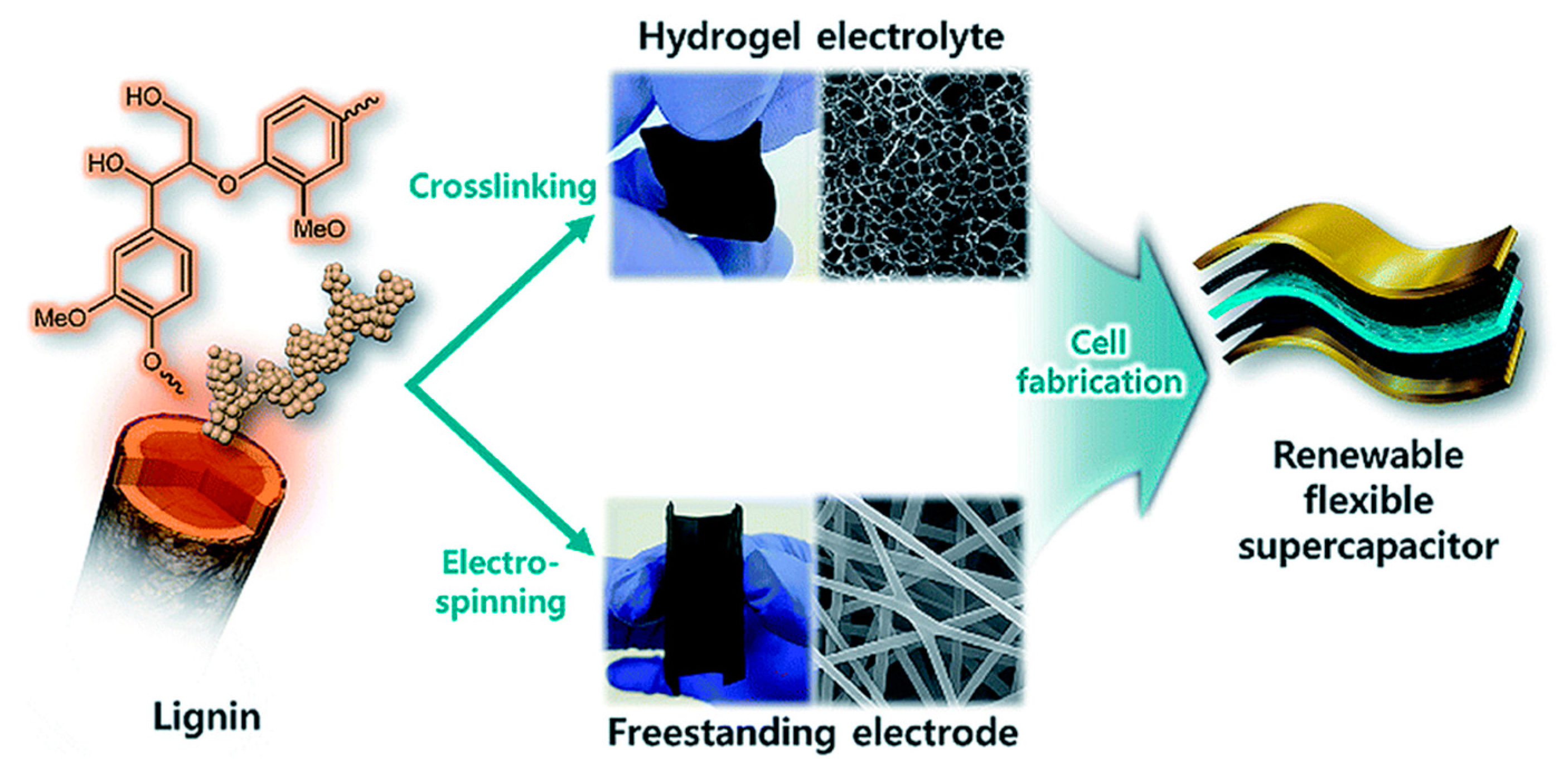
3.4. Flexible Supercapacitors and Werable Electronics
4. Future Perspective and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sasaki, M.; Goto, M. Recovery of phenolic compounds through the decomposition of lignin in near and supercritical water. Chem. Eng. Process. Process Intensif. 2008, 47, 1609–1619. [Google Scholar] [CrossRef]
- Cheng, S.; Yuan, Z.; Leitch, M.; Anderson, M.; Charles, C. Highly efficient de-polymerization of organosolv lignin using a catalytic hydrothermal process and production of phenolic resins/adhesives with the depolymerized lignin as a substitute for phenol at a high substitution ratio. Ind. Crops Prod. 2013, 44, 315–322. [Google Scholar] [CrossRef]
- Aro, T.; Fatehi, P. Production and Application of Lignosulfonates and Sulfonated Lignin. ChemSusChem 2017, 10, 1861–1877. [Google Scholar] [CrossRef]
- Alam, M.; Marghoob, S.; Prakash, V. Development and characterization of a wood adhesive using bagasse lignin. Int. J. Adhes. Adhes. 2004, 24, 485–493. [Google Scholar] [CrossRef]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The Catalytic Valorization of Lignin for the Production of Renewable Chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef]
- Alinejad, M.; Henry, C.; Nikafshar, S.; Gondaliya, A.; Bagheri, S.; Chen, N.; Singh, S.K.; Hodge, D.B.; Nejad, M. Lignin-Based Polyurethanes: Opportunities for Bio-Based Foams, Elastomers, Coatings and Adhesives. Polymers 2019, 11, 1202. [Google Scholar] [CrossRef]
- Fache, M.; Boutevin, B.; Caillol, S. Vanillin Production from Lignin and Its Use as a Renewable Chemical. ACS Sustain. Chem. Eng. 2016. [Google Scholar] [CrossRef]
- Taylor, P.; Biagiotti, J.; Puglia, D.; Kenny, J.M. A review on natural fibre-based composites-part I: Structure, processing and properties of vegetable fibres. J. Nat. Fibers 2008, 37–41. [Google Scholar]
- Brinchi, L.; Cotana, F.; Fortunati, E.; Kenny, J.M. Production of nanocrystalline cellulose from lignocellulosic biomass: Technology and applications. Carbohydr. Polym. 2013, 94, 154–169. [Google Scholar] [CrossRef]
- Hamelinck, C.N.; Van Hooijdonk, G.; Faaij, A.P. Ethanol from lignocellulosic biomass: Techno-economic performance in short-, middle- and long-term. Biomass Bioenergy 2005, 28, 384–410. [Google Scholar] [CrossRef]
- Norgren, M.; Edlund, H. Lignin: Recent advances and emerging applications. Curr. Opin. Colloid Interface Sci. 2014, 19, 409–416. [Google Scholar] [CrossRef]
- Chakar, F.S.; Ragauskas, A.J. Review of current and future softwood kraft lignin process chemistry. Ind. Crops Prod. 2004, 20, 131–141. [Google Scholar] [CrossRef]
- Jiang, G.; Nowakowski, D.J.; Bridgwater, A.V. Thermochimica Acta A systematic study of the kinetics of lignin pyrolysis. Thermochim. Acta 2010, 498, 61–66. [Google Scholar] [CrossRef]
- Gellerstedt, G. Softwood kraft lignin: Raw material for the future. Ind. Crops Prod. 2015, 77, 845–854. [Google Scholar] [CrossRef]
- Jang, B.W.; Gläser, R.; Liu, C.; Jang, B.W.; Gläser, R.; Liu, C.; Capel-sanchez, M.C.; Campos-martin, J.M.; Fierro, L.G.; Sci, E.E.; et al. Fuels of the future. Energy Environ. Sci. 2010, 3, 253. [Google Scholar] [CrossRef]
- El, R.; Brosse, N.; Chrusciel, L.; Sanchez, C.; Sannigrahi, P.; Ragauskas, A.; Pan, X.; Xie, D.; Yu, R.W.; Lam, D.; et al. Pretreatment of Lodgepole Pine Killed by Mountain Pine Beetle Using the Ethanol Organosolv Process: Fractionation and Process Optimization. Polym. Degrad. Stab. 2009, 94, 2609–2617. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, Q.; Lee, D. Bioresource Technology Kraft lignin biore fi nery: A perspective. Bioresour. Technol. 2018, 247, 1181–1183. [Google Scholar] [CrossRef]
- Cardoso, M.; Domingos, É.; Oliveira, D.; Laura, M. Chemical composition and physical properties of black liquors and their effects on liquor recovery operation in Brazilian pulp mills. Fuel 2009, 88, 756–763. [Google Scholar] [CrossRef]
- Kosbar, L.L.; Gelorme, J.D.; Japp, R.M.; Fotorny, W.T. Introducing Biobased Materials into the Electronics Industry. J. Ind. Ecol. 2000, 4, 93–105. [Google Scholar] [CrossRef]
- Vishtal, A.; Kraslawski, A. Challenges in industrial applications of technical lignins. BioResources 2011, 6, 3547–3568. [Google Scholar] [CrossRef]
- Fu, D.; Mazza, G.; Tamaki, Y. Lignin Extraction from Straw by Ionic Liquids and Enzymatic Hydrolysis of the Cellulosic Residues. J. Agric. Food Chem. 2010, 58, 2915–2922. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.; Bello, F.O.; Horsfall, L. Microbial enzyme systems for lignin degradation and their transcriptional regulation. Front. Biol. 2014, 9, 448–471. [Google Scholar] [CrossRef]
- Tuomela, M. Biodegradation of lignin in a compost environment: A review. Bioresour. Technol. 2000, 72, 169–183. [Google Scholar] [CrossRef]
- Huang, X.; Korµnyi, T.I.; Boot, M.D.; Hensen, E.J.M. Catalytic Depolymerization of Lignin in Supercritical Ethanol. ChemSusChem 2014, 2276–2288. [Google Scholar] [CrossRef]
- Figueiredo, P.; Lintinen, K.; Hirvonen, J.T.; Kostiainen, M.A.; Santos, H.A. Properties and chemical modifications of lignin: Towards lignin-based nanomaterials for biomedical applications. Prog. Mater. Sci. 2018, 93, 233–269. [Google Scholar] [CrossRef]
- Gillet, S.; Aguedo, M.; Petitjean, L.; Morais, A.R.C.; da Costa Lopes, A.M.; Łukasik, R.M.; Anastas, P.T. Lignin transformations for high value applications: Towards targeted modifications using green chemistry. Green Chem. 2017, 19, 4200–4233. [Google Scholar] [CrossRef]
- Nahmany, M.; Melman, A. Chemoselectivity in reactions of esterification. Org. Biomol. Chem. 2004, 2, 1563. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Recent advances in green hydrogels from lignin: A review. Int. J. Biol. Macromol. 2015, 72, 834–847. [Google Scholar] [CrossRef]
- Mishra, P.K.; Ekielski, A. The Self-Assembly of Lignin and Its Application in Nanoparticle Synthesis: A Short Review. Nanomaterials 2019, 9, 243. [Google Scholar] [CrossRef]
- Mishra, P.K.; Wimmer, R. Aerosol assisted self-assembly as a route to synthesize solid and hollow spherical lignin colloids and its utilization in layer by layer deposition. Ultrason. Sonochem. 2017, 35, 45–50. [Google Scholar] [CrossRef]
- Akhtar, M.F.; Hanif, M.; Ranjha, N.M. Methods of synthesis of hydrogels … A review. Saudi Pharm. J. 2016, 24, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Dragan, E.S. Design and applications of interpenetrating polymer network hydrogels … A review. Chem. Eng. J. 2014, 243, 572–590. [Google Scholar] [CrossRef]
- Ullah, F.; Othman, M.B.H.; Javed, F.; Ahmad, Z.; Akil, H.M. Classification, processing and application of hydrogels: A review. Mater. Sci. Eng. C 2015, 57, 414–433. [Google Scholar] [CrossRef] [PubMed]
- Gennen, S.; Grignard, B.; Thomassin, J.; Gilbert, B.; Vertruyen, B.; Jerome, C.; Detrembleur, C. Polyhydroxyurethane hydrogels: Synthesis and characterizations. Eur. Polym. J. 2016, 84, 849–862. [Google Scholar] [CrossRef]
- Peñaranda, A.; Jesús, E.; Sabino, M.A. Effect of the presence of lignin or peat in IPN hydrogels on the sorption of heavy metals. Polym. Bull. 2010, 65, 495–508. [Google Scholar] [CrossRef]
- Oveissi, F.; Yen, T.; Le, L.; Fletcher, D.F.; Dehghani, F. Tough and Processable Hydrogels Based on Lignin and Hydrophilic Polyurethane. ACS Appl. Bio. Mater. 2018, 1, 2073–2081. [Google Scholar] [CrossRef]
- Huang, S.; Shuyi, S.; Gan, H.; Linjun, W.; Lin, C.; Danyuan, X.; Zhou, H.; Lin, X.; Qin, Y. Facile fabrication and characterization of highly stretchable lignin-based hydroxyethyl cellulose self-healing hydrogel. Carbohydr. Polym. 2019, 223, 115080. [Google Scholar] [CrossRef]
- Ravishankar, K.; Venkatesan, M.; Preeth, R.; Mahalingam, A. Biocompatible hydrogels of chitosan-alkali lignin for potential wound healing applications. Mater. Sci. Eng. C 2019, 102, 447–457. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, M.; Zhang, Y.; Cao, Q.; Wang, X.; Han, Y. Novel lignin-chitosan-PVA composite hydrogel for wound dressing. Mater. Sci. Eng. C 2019, 104, 110002. [Google Scholar] [CrossRef]
- Chen, F.; Xu, M.; Wang, L.; Li, J. Preparation and characterization of organic aerogels from a lignin-resorcinol-formaldehyde copolymer. BioResources 2011, 6, 1262–1272. [Google Scholar] [CrossRef]
- Grishechko, L.I.; Amaral-labat, G.; Szczurek, A.; Fierro, V.; Kuznetsov, B.N.; Celzard, A. Microporous and Mesoporous Materials Lignin-phenol-formaldehyde aerogels and cryogels. Microporous Mesoporous Mater. 2013, 168, 19–29. [Google Scholar] [CrossRef]
- Feldman, D.; Banu, D.; Natansohn, A.; Wang, J. Structure–properties relations of thermally cured epoxy–lignin polyblends. J. Appl. Polym. Sci. 1991, 42, 1537–1550. [Google Scholar] [CrossRef]
- Xu, C.; Ferdosian, F. Conversion of Lignin into Chemicals and Materials; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 9783662549575. [Google Scholar]
- Yin, Q.; Yang, W.; Sun, C.; Di, M. Preparation and properties of lignin-epoxy resin composite. Bioresources 2012, 7, 5737–5748. [Google Scholar] [CrossRef]
- Lindströmn, T. The colloidal behaviour of kraft lignin. Colloid Polym. Sci. 1979, 257, 277–285. [Google Scholar] [CrossRef]
- Ciolacu, D.; Oprea, A.M.; Anghel, N.; Cazacu, G.; Cazacu, M. New cellulose-lignin hydrogels and their application in controlled release of polyphenols. Mater. Sci. Eng. C 2012, 32, 452–463. [Google Scholar] [CrossRef]
- Dai, L.; Zhu, W.; Lu, J.; Kong, F.; Si, C.; Ni, Y. A lignin-containing cellulose hydrogel for lignin fractionation. Green Chem. 2019, 21, 5222–5230. [Google Scholar] [CrossRef]
- Ciolacu, D.; Cazacu, G. New Green Hydrogels Based on Lignin. J. Nanosci. Nanotechnol. 2018, 18, 2811–2822. [Google Scholar] [CrossRef]
- Wu, L.; Huang, S.; Zheng, J.; Qiu, Z.; Lin, X.; Qin, Y. Synthesis and characterization of biomass lignin-based PVA super-absorbent hydrogel. Int. J. Biol. Macromol. 2019, 140, 538–545. [Google Scholar] [CrossRef]
- Raschip, I.E.; Hitruc, G.E.; Vasile, C.; Popescu, M. Effect of the lignin type on the morphology and thermal properties of the xanthan/lignin hydrogels. Int. J. Biol. Macromol. 2013, 54, 230–237. [Google Scholar] [CrossRef]
- Li, M.; Jiang, X.; Wang, D.; Xu, Z.; Yang, M. In situ reduction of silver nanoparticles in the lignin based hydrogel for enhanced antibacterial application. Colloids Surf. B Biointerfaces 177 2019, 177, 370–376. [Google Scholar] [CrossRef]
- Nishida, M.; Uraki, Y.; Sano, Y. Lignin gel with unique swelling property. Bioresour. Technol. 2003, 88, 81–83. [Google Scholar] [CrossRef]
- Passauer, L.; Struch, M.; Schuldt, S.; Appelt, J.; Schneider, Y.; Jaros, D.; Rohm, H. Dynamic Moisture Sorption Characteristics of Xerogels from Water-Swellable Oligo (oxyethylene) Lignin Derivatives. ACS Appl. Mater. Interfaces 2012, 4, 5852–5862. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Rana, H.H.; Lee, J.Y.; Park, H.S. Renewable flexible supercapacitors based on all-lignin-based hydrogel electrolytes and nanofiber electrodes. J. Mater. Chem. A 2019, 7, 16962–16968. [Google Scholar] [CrossRef]
- El-zawawy, W.K. Preparation of hydrogel from green polymer. Polym. Adv. Technol. 2005, 16, 48–54. [Google Scholar] [CrossRef]
- El-zawawy, W.K.; Ibrahim, M.M. Preparation and Characterization of Novel Polymer Hydrogel from Industrial Waste and Copolymerization of Poly (vinyl alcohol) and Polyacrylamide. J. Appl. Polym. Sci. 2012, 124, 4362–4370. [Google Scholar] [CrossRef]
- Jin, Z.; Yu, Y.; Shao, S.; Ye, J.; Lin, L.; Iiyama, K. Lignin as a cross-linker of acrylic acid-grafted carboxymethyl lignocellulose. J. Wood Sci. 2010, 56, 470–476. [Google Scholar] [CrossRef]
- Sun, X.; Hao, Y.; Cao, Y.; Zeng, Q. Superadsorbent hydrogel based on lignin and montmorillonite for Cu (II) ions removal from aqueous solution. Int. J. Biol. Macromol. 2019, 127, 511–519. [Google Scholar] [CrossRef]
- Jiang, P.; Sheng, X.; Yu, S.; Li, H.; Lu, J.; Zhou, J.; Wang, H. Preparation and characterization of thermo-sensitive gel with phenolated alkali lignin. Sci. Rep. 2018, 8, 14450. [Google Scholar] [CrossRef]
- Domínguez-robles, J.; Soledad, M.; Tamminen, T.; Rodríguez, A.; Larrañeta, E.; Jääskeläinen, A. International Journal of Biological Macromolecules Lignin-based hydrogels with super-swelling capacities for dye removal. Int. J. Biol. Macromol. 2018, 115, 1249–1259. [Google Scholar] [CrossRef]
- Larrañeta, E.; Imízcoz, M.; Toh, J.X.; Irwin, N.J.; Ripolin, A.; Perminova, A.; Dom, J.; Rodr, A.; Donnelly, R.F. Synthesis and Characterization of Lignin Hydrogels for Potential Applications as Drug Eluting Antimicrobial Coatings for Medical Materials. Sustain. Chem. Eng. 2018, 6, 9037–9046. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, Z.; Guo, X.; He, Q. Ultrasonic-assisted synthesis of sodium lignosulfonate-grafted poly (acrylic acid-co-poly (vinyl pyrrolidone)) hydrogel for drug delivery. RSC Adv. 2016, 6, 35550–35558. [Google Scholar] [CrossRef]
- Zerpa, A.; Pakzad, L.; Fatehi, P. Hardwood Kraft Lignin-Based Hydrogels: Production and Performance. ACS Omega 2018, 3, 8233–8242. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Chen, F.; Wu, H. Preparation and characterization of a temperature-sensitive lignin-based hydrogel. BioResources 2011, 6, 4942–4952. [Google Scholar] [CrossRef]
- Feng, Q.; Chen, F.; Zhou, X. Preparation of thermo-sensitive hydrogels from acrylated lignin and N-isopropylacrylamide through photocrosslinking. J. Biobased Mater. Bioenergy 2012, 6, 336–342. [Google Scholar] [CrossRef]
- Gao, G.; Xu, W.Z.; Kadla, J.F. Reversible ph-responsive hydrogels of softwood kraft lignin and poly [(2-dimethylamino) ethyl methacrylate]-based polymers. J. Wood Chem. Technol. 2014, 35, 73–90. [Google Scholar] [CrossRef]
- Liu, W.; Ye, Z.; Liu, D.; Wu, Z. Hydrogels Derived from Lignin with pH Responsive and Magnetic Properties. BioResources 2018, 13, 7281–7293. [Google Scholar] [CrossRef]
- Jin, C.; Song, W.; Liu, T.; Xin, J.; Hiscox, W.C.; Zhang, J.; Liu, G.; Kong, Z. Temperature and pH Responsive Hydrogels Using Methacrylated Lignosulfonate Cross-Linker: Synthesis, Characterization, and Properties. ACS Sustain. Chem. Eng. 2018, 6, 1763–1771. [Google Scholar] [CrossRef]
- Kai, D.; Low, Z.W.; Liow, S.S.; Abdul Karim, A.; Ye, H.; Jin, G.; Li, K.; Loh, X.J. Development of Lignin Supramolecular Hydrogels with Mechanically Responsive and Self-Healing Properties. ACS Sustain. Chem. Eng. 2015, 3, 2160–2169. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Dominici, F.; Giovanale, G.; Mazzaglia, A.; Balestra, G.M.; Kenny, J.M.; Puglia, D. Effect of cellulose and lignin on disintegration, antimicrobial and antioxidant properties of PLA active films. Int. J. Biol. Macromol. 2016, 89, 360–368. [Google Scholar] [CrossRef]
- Zmejkoski, D.; Spasojević, D.; Orlovska, I.; Kozyrovska, N.; Soković, M.; Glamočlija, J.; Dmitrović, S.; Matović, B.; Tasić, N.; Maksimović, V.; et al. Bacterial cellulose-lignin composite hydrogel as a promising agent in chronic wound healing. Int. J. Biol. Macromol. 2018, 118, 494–503. [Google Scholar] [CrossRef]
- Musilová, L.; Mráček, A.; Kovalcik, A.; Smolka, P.; Minařík, A.; Humpolíček, P.; Vícha, R.; Ponížil, P. Hyaluronan hydrogels modified by glycinated Kraft lignin: Morphology, swelling, viscoelastic properties and biocompatibility. Carbohydr. Polym. 2018, 181, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Raschip, I.E.; Hitruc, E.G.; Oprea, A.M.; Popescu, M.-C.; Vasile, C. In vitro evaluation of the mixed xanthan/lignin hydrogels as vanillin carriers. J. Mol. Struct. 2011, 1003, 67–74. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, Y.; Fang, G.; Ren, S.; Fu, Y. Controlled Pesticide Release from Porous Composite Hydrogels Based on Lignin and Polyacrylic Acid. BioResources 2016, 11, 2361–2371. [Google Scholar] [CrossRef]
- Carrott, P.J.M.; Carrott, M.R. Lignin-from natural adsorbent to activated carbon: A review. Bioresour. Technol. 2007, 98, 2301–2312. [Google Scholar] [CrossRef]
- Thakur, S.; Govender, P.P.; Mamo, M.A.; Tamulevicius, S.; Mishra, Y.K.; Thakur, V.K. Progress in lignin hydrogels and nanocomposites for water purification: Future perspectives. Vacuum 2017, 146, 342–355. [Google Scholar] [CrossRef]
- Ge, Y.; Li, Z. Application of Lignin and Its Derivatives in Adsorption of Heavy Metal Ions in Water: A Review. ACS Sustain. Chem. Eng. 2018, 6, 7181–7192. [Google Scholar] [CrossRef]
- Ma, Y.; Lv, L.; Guo, Y.; Fu, Y.; Shao, Q.; Wu, T.; Guo, S.; Sun, K.; Guo, X.; Wujcik, E.K.; et al. Porous lignin based poly (acrylic acid)/organo-montmorillonite nanocomposites: Swelling behaviors and rapid removal of Pb(II) ions. Polymer 2017, 128, 12–23. [Google Scholar] [CrossRef]
- Yao, Q.; Xie, J.; Liu, J.; Kang, H.; Liu, Y. Adsorption of lead ions using a modified lignin hydrogel. J. Polym. Res. 2014, 21, 465. [Google Scholar] [CrossRef]
- Li, F.; Wang, X.; Yuan, T.; Sun, R. A lignosulfonate-modified graphene hydrogel with ultrahigh adsorption capacity for Pb(II) removal. J. Mater. Chem. A 2016, 4, 11888–11896. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, Y.; Fang, G.; Li, S.; Fu, Y. Synthesis of Acid Hydrolysis Lignin-g-Poly-(Acrylic Acid) Hydrogel Superabsorbent Composites and Adsorption of Lead Ions. BioResources 2016, 11, 5731–5742. [Google Scholar] [CrossRef]
- Parajuli, D.; Inoue, K.; Ohto, K.; Oshima, T.; Murota, A.; Funaoka, M.; Makino, K. Adsorption of heavy metals on crosslinked lignocatechol: A modified lignin gel. React. Funct. Polym. 2005, 62, 129–139. [Google Scholar] [CrossRef]
- Parajuli, D.; Adhikari, C.R.; Kuriyama, M.; Kawakita, H.; Ohto, K.; Inoue, K.; Funaoka, M. Selective Recovery of Gold by Novel Lignin-Based Adsorption Gels. Ind. Eng. Chem. Res. 2006, 45, 8–14. [Google Scholar] [CrossRef]
- Meng, Y.; Lu, J.; Cheng, Y.; Li, Q.; Wang, H. Lignin-based hydrogels: A review of preparation, properties, and application. Int. J. Biol. Macromol. 2019, 135, 1006–1019. [Google Scholar] [CrossRef] [PubMed]
- Albadarin, A.B.; Collins, M.N.; Naushad, M.; Shirazian, S.; Walker, G.; Mangwandi, C. Activated lignin-chitosan extruded blends for efficient adsorption of methylene blue. Chem. Eng. J. 2017, 307, 264–272. [Google Scholar] [CrossRef]
- Wang, Y.; Xiong, Y.; Wang, J.; Zhang, X. Ultrasonic-assisted fabrication of montmorillonite-lignin hybrid hydrogel: Highly efficient swelling behaviors and super-sorbent for dye removal from wastewater. Colloids Surf. A Physicochem. Eng. Asp. 2017, 520, 903–913. [Google Scholar] [CrossRef]
- Feng, Q.; Li, J.; Cheng, H.; Chen, F.; Xie, Y. Synthesis and Characterization of Porous Hydrogel Based on Lignin and Polyacrylamide. BioResources 2014, 9, 4369–4381. [Google Scholar] [CrossRef][Green Version]
- Yu, C.; Wang, F.; Zhang, C.; Fu, S.; Lucia, L.A. The synthesis and absorption dynamics of a lignin-based hydrogel for remediation of cationic dye-contaminated effluent. React. Funct. Polym. 2016, 106, 137–142. [Google Scholar] [CrossRef]
- Tang, Y.; Zeng, Y.; Hu, T.; Zhou, Q.; Peng, Y. Preparation of lignin sulfonate-based mesoporous materials for adsorbing malachite green from aqueous solution. J. Environ. Chem. Eng. 2016, 4, 2900–2910. [Google Scholar] [CrossRef]
- Palchoudhury, S.; Ramasamy, K.; Gupta, R.K.; Gupta, A. Flexible Supercapacitors: A Materials Perspective. Front. Mater. 2019, 5, 83. [Google Scholar] [CrossRef]
- Dong, L.; Xu, C.; Li, Y.; Huang, Z.-H.; Kang, F.; Yang, Q.-H.; Zhao, X. Flexible electrodes and supercapacitors for wearable energy storage: A review by category. J. Mater. Chem. A 2016, 4, 4659–4685. [Google Scholar] [CrossRef]
- Weng, Z.; Su, Y.; Wang, D.-W.; Li, F.; Du, J.; Cheng, H.-M. Graphene-Cellulose Paper Flexible Supercapacitors. Adv. Energy Mater. 2011, 1, 917–922. [Google Scholar] [CrossRef]
- Xiong, C.; Zhong, W.; Zou, Y.; Luo, J.; Yang, W. Electroactive biopolymer/graphene hydrogels prepared for high-performance supercapacitor electrodes. Electrochim. Acta 2016, 211, 941–949. [Google Scholar] [CrossRef]
- Li, F.; Wang, X.; Sun, R. A metal-free and flexible supercapacitor based on redox-active lignosulfonate functionalized graphene hydrogels. J. Mater. Chem. A 2017, 5, 20643–20650. [Google Scholar] [CrossRef]
- Peng, Z.; Zou, Y.; Xu, S.; Zhong, W.; Yang, W. High-Performance Biomass-Based Flexible Solid-State Supercapacitor Constructed of Pressure-Sensitive Lignin-Based and Cellulose Hydrogels. ACS Appl. Mater. Interfaces 2018, 10, 22190–22200. [Google Scholar] [CrossRef] [PubMed]





| Lignin Role | Matrix | Crosslinker | Lignin Interaction | Ref |
|---|---|---|---|---|
| Lignin as biomass | Polymerized acrylamide | N,N′-methylenebis(acrylamide) (NMBA) | H-bonding | [35] |
| Polyurethane | − | H-bonding | [36] | |
| Hydroxyethyl cellulose and polyvinyl alcohol | Borax | H-bonding | [37] | |
| Lignin as crosslinked unit | Chitosan | Lignin | Electrostatic | [38] |
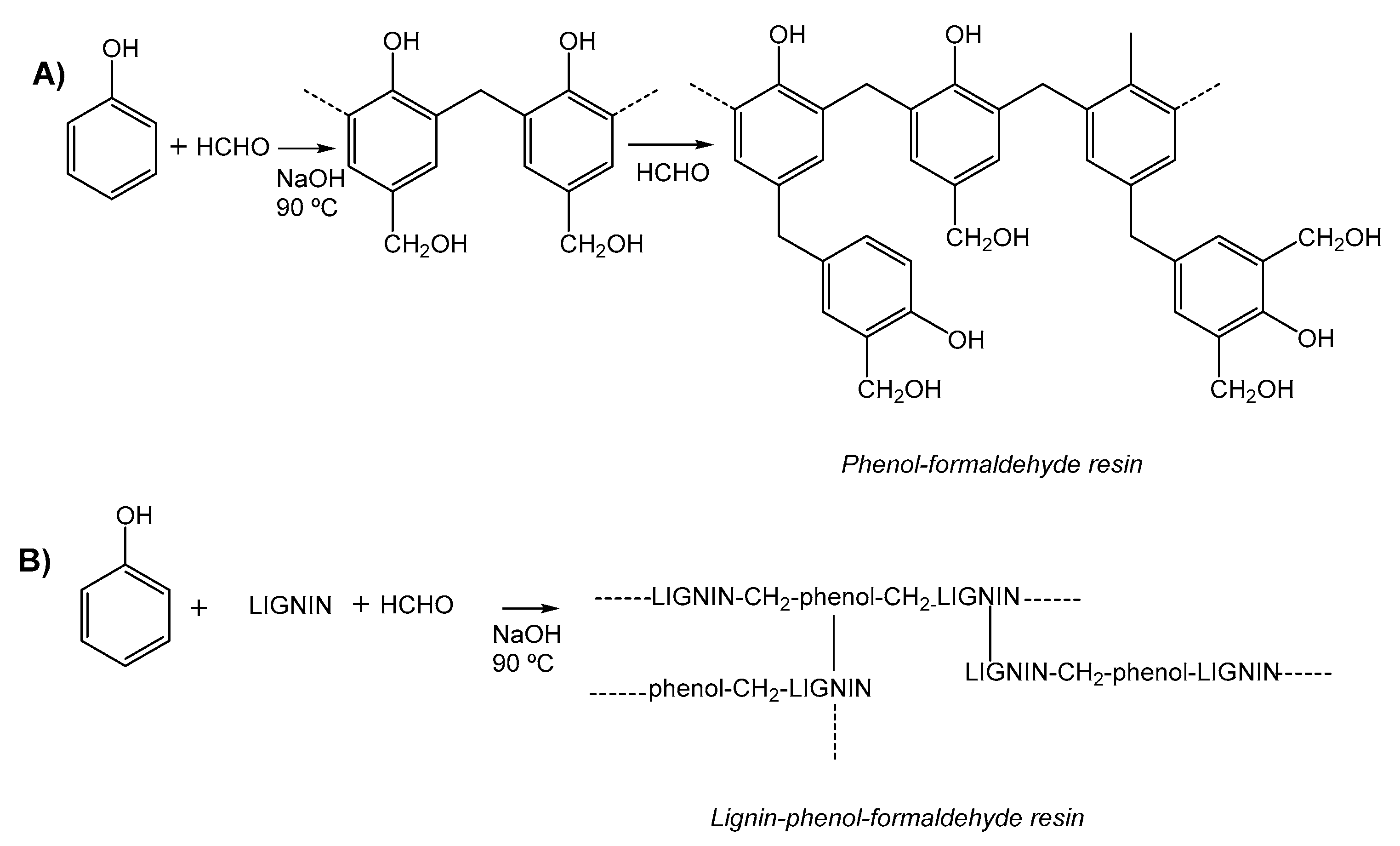
| Phenol/lignin/formaldehyde Lignin-tannin-formaldehyde | Formaldehyde | Chemical | [41] | |
| Lignin–phenol–resorcinol | Glutaraldehyde | Chemical | [40] | |
| Lignin and epoxy resin | Polyamine | Chemical | [44] | |
| Lignin | Epichlorohydrin | Chemical | [45] | |
| Cellulose and lignin | Epichlorohydrin | Chemical | [46,47] | |
| Polyvinyl alcohol and lignin or lignin-epoxy | Epichlorohydrin | Chemical/H-bonding | [48,49] | |
| Lignin | Poly (ethylene) glycol diglycidyl ether | Chemical | [52,53,54] | |
| Acrylamide-Polyvinyl alcohol-graft-lignin copolymers | −/NMBA | Chemical | [55,56] | |
| Xanthan and lignin | Epichlorohydrin | Chemical | [50] | |
| Lignin as crosslinking agent | Chitosan and polyvinyl alcohol | lignin | H-bonding/ electrostatic | [39] |
| Cellulose derivatives and polyvinyl alcohol | Lignin/epichlorohydrin | Chemical | [37] | |
| Polyvinyl alcohol | Aminated li gnin | Chemical | [51] | |
| Polymerized acrylic acid | Lignin/lignin and NMBA | Chemical | [57,58] | |
| Polymerized N-isopropylacrylamide | Lignin | Chemical | [59] | |
| Poly(methyl vinyl ether co-maleic acid) (GRANTEZ) | Lignin/Lignin and PEG | Chemical | [60,61] | |
| Polymerized acrylic acid and poly(vinyl pyrrolidone) | Lignin | Chemical | [62] |
| Smart Hydrogels | ||
|---|---|---|
| Active Hydrogels | ||
| Thermoresponsive | [63,64,65] | |
| pH-responsive | [66,67] | |
| Dual (Thermo and pH) | [68] | |
| Mechanically responsive | [69] | |
| Biomedical applications | ||
| Antibacterial | [38,72] | |
| Drug release | [61] | |
| Aroma | [73] | |
| Pesticide release | [74] | |
| Water remediation | ||
| Metals | [35,75,76,77,78,79,80,81,82,83] | |
| Dyes | [85,86,87,88,89] | |
| Supercapacitators and weareble | ||
| Supercapacitators | [54,93,94,95] | |
| Wearable electronic and 3D printing | [36] | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rico-García, D.; Ruiz-Rubio, L.; Pérez-Alvarez, L.; Hernández-Olmos, S.L.; Guerrero-Ramírez, G.L.; Vilas-Vilela, J.L. Lignin-Based Hydrogels: Synthesis and Applications. Polymers 2020, 12, 81. https://doi.org/10.3390/polym12010081
Rico-García D, Ruiz-Rubio L, Pérez-Alvarez L, Hernández-Olmos SL, Guerrero-Ramírez GL, Vilas-Vilela JL. Lignin-Based Hydrogels: Synthesis and Applications. Polymers. 2020; 12(1):81. https://doi.org/10.3390/polym12010081
Chicago/Turabian StyleRico-García, Diana, Leire Ruiz-Rubio, Leyre Pérez-Alvarez, Saira L. Hernández-Olmos, Guillermo L. Guerrero-Ramírez, and José Luis Vilas-Vilela. 2020. "Lignin-Based Hydrogels: Synthesis and Applications" Polymers 12, no. 1: 81. https://doi.org/10.3390/polym12010081
APA StyleRico-García, D., Ruiz-Rubio, L., Pérez-Alvarez, L., Hernández-Olmos, S. L., Guerrero-Ramírez, G. L., & Vilas-Vilela, J. L. (2020). Lignin-Based Hydrogels: Synthesis and Applications. Polymers, 12(1), 81. https://doi.org/10.3390/polym12010081