Among thermosetting polymers, 70% of the market share is captured by epoxy resins (excluding polyurethanes) and 60% of the globally produced epoxy resins is employed in the coatings industry due to their low shrinkage during cure, good adhesion with various substrates, excellent corrosion and chemical resistance, high strength and durability, and good compatibility with numerous commercially important materials. Commercial epoxy coatings can be 1K or 2K (i.e., 1 component or 2 components, respectively, with K used as an abbreviation of the German word for component). 1K epoxy coatings contain anionic or cationic initiators (in one package), which under heat or photoirradiation, can initiate the polymerization of the epoxy monomer. In 2K epoxy coatings, generally, the component containing the epoxy resin (along with other additives like solvent(s), pigment(s), defoaming agent etc.) is termed as the Component A while the second component, termed as Component B, contains the hardener or curing agent. Component A and B are mixed before application. Thermosetting 2K epoxy formulations can be cured by compounds that are reactive towards the epoxy functional group such as, amines, thiols, organic acids, anhydrides, alcohols, and phenols. For liquid epoxy coatings intended to cure at ambient temperatures and below, amine functional curing agents are most widely used. The type of the epoxy resin and curing agent used along with the curing conditions can have a significant effect on the final properties of the coating [
1,
2].
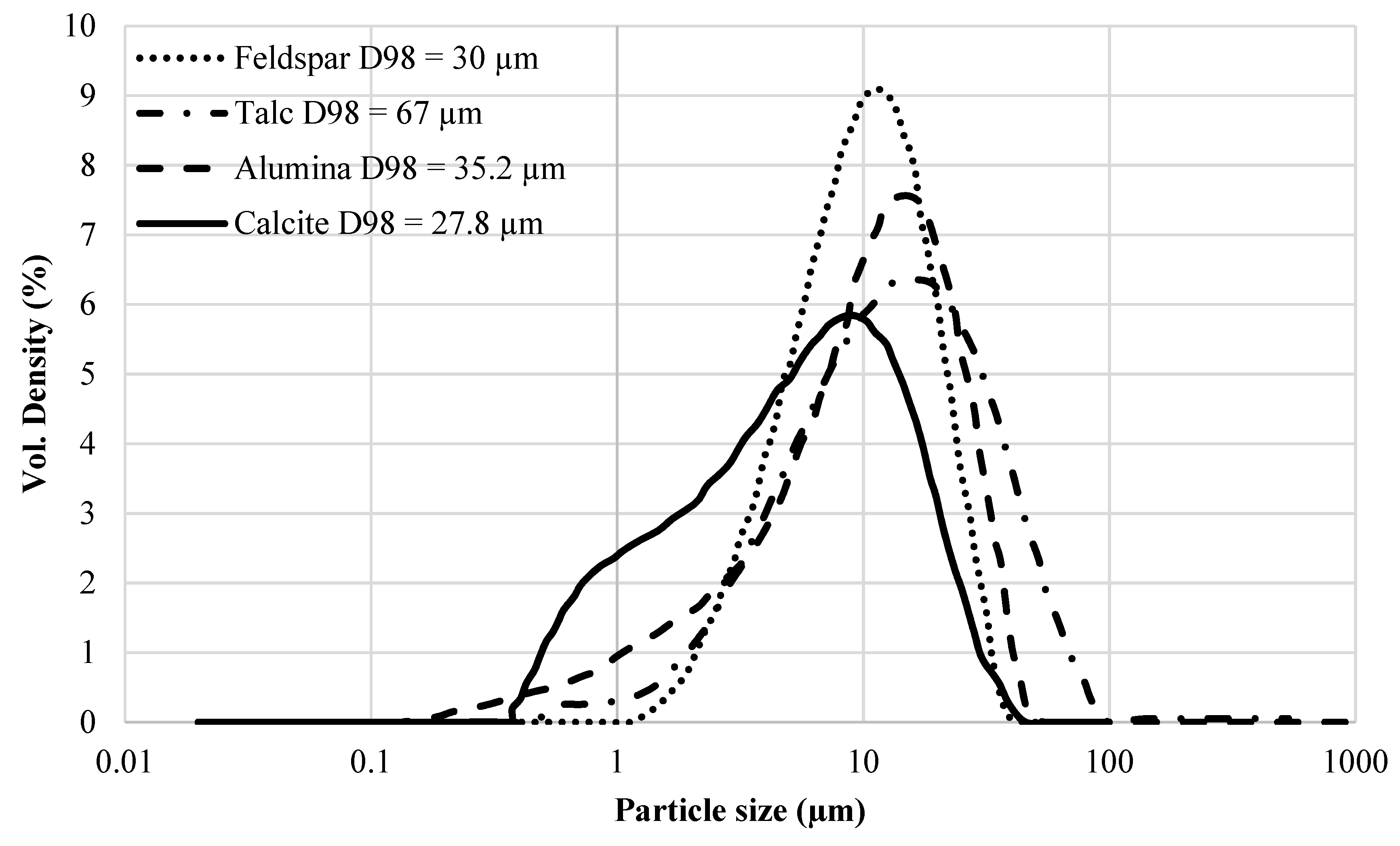
Commercial epoxy coatings contain additives and fillers which may help in easier processing during production and application, cost reduction, and improving the final properties of the coating (e.g., fracture toughness of the epoxy resin is reported to be increased by using nanosilica [
3]). Among fillers, inorganic extenders constitute a major part of the total formulation. These inorganic extenders (sometimes called extender pigments) include kaolin (aluminum silicates), calcium carbonates, talc (magnesium silicate), mica (hydrous aluminum potassium silicates), silica (silicon dioxide), wollastonite (calcium metasilicates), barite (barium sulphate), aluminum oxide etc. Generally, these extenders are inexpensive and therefore, used to reduce the production cost of coatings but their physical and chemical properties (e.g., surface treatment with chemicals like silanes) can impact the final properties of the coating. Surface area, porosity, and particle size distribution (PSD) are among the important physical properties of extenders as they impact the oil absorption of extenders which in turn can influence the processing and final properties of the coatings [
4,
5].
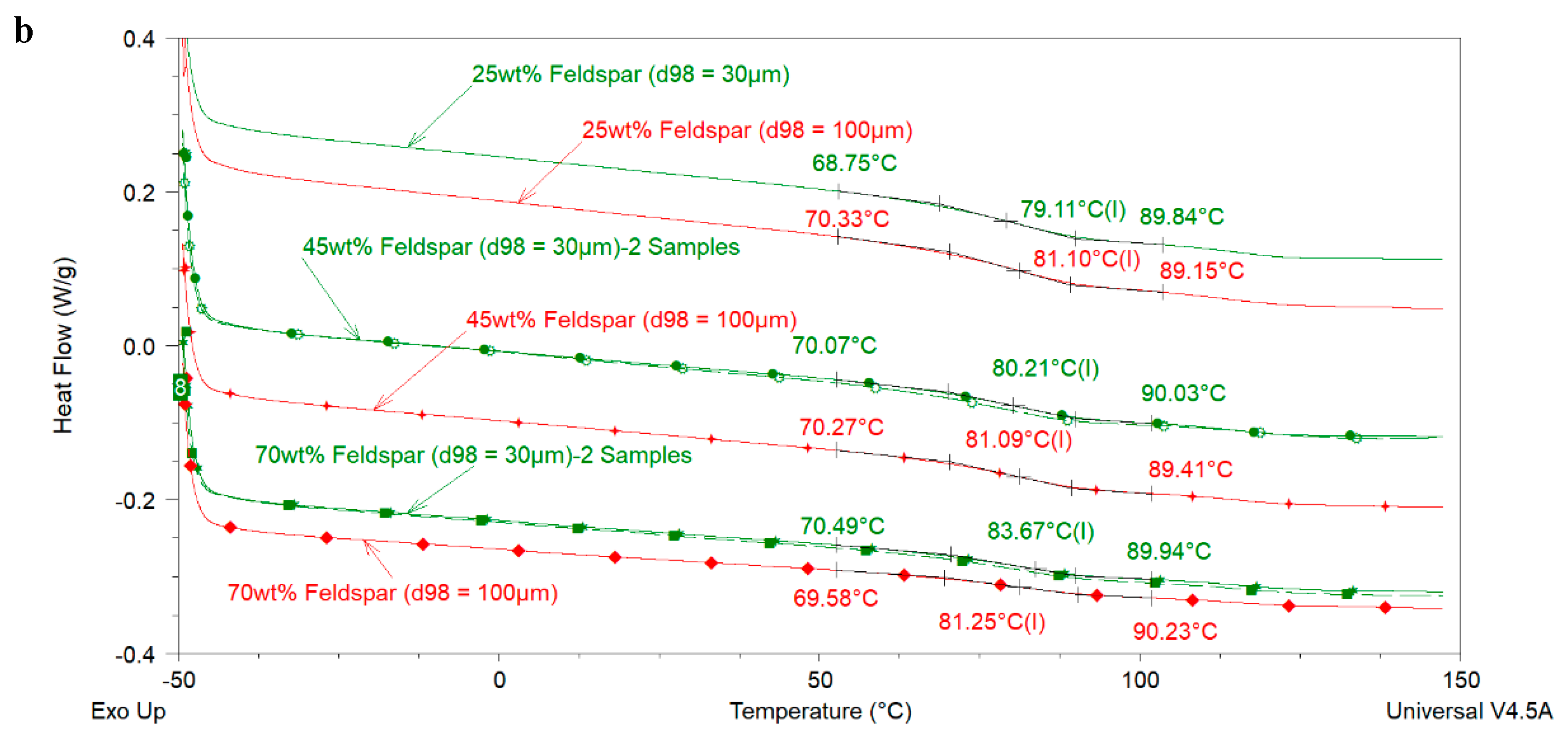
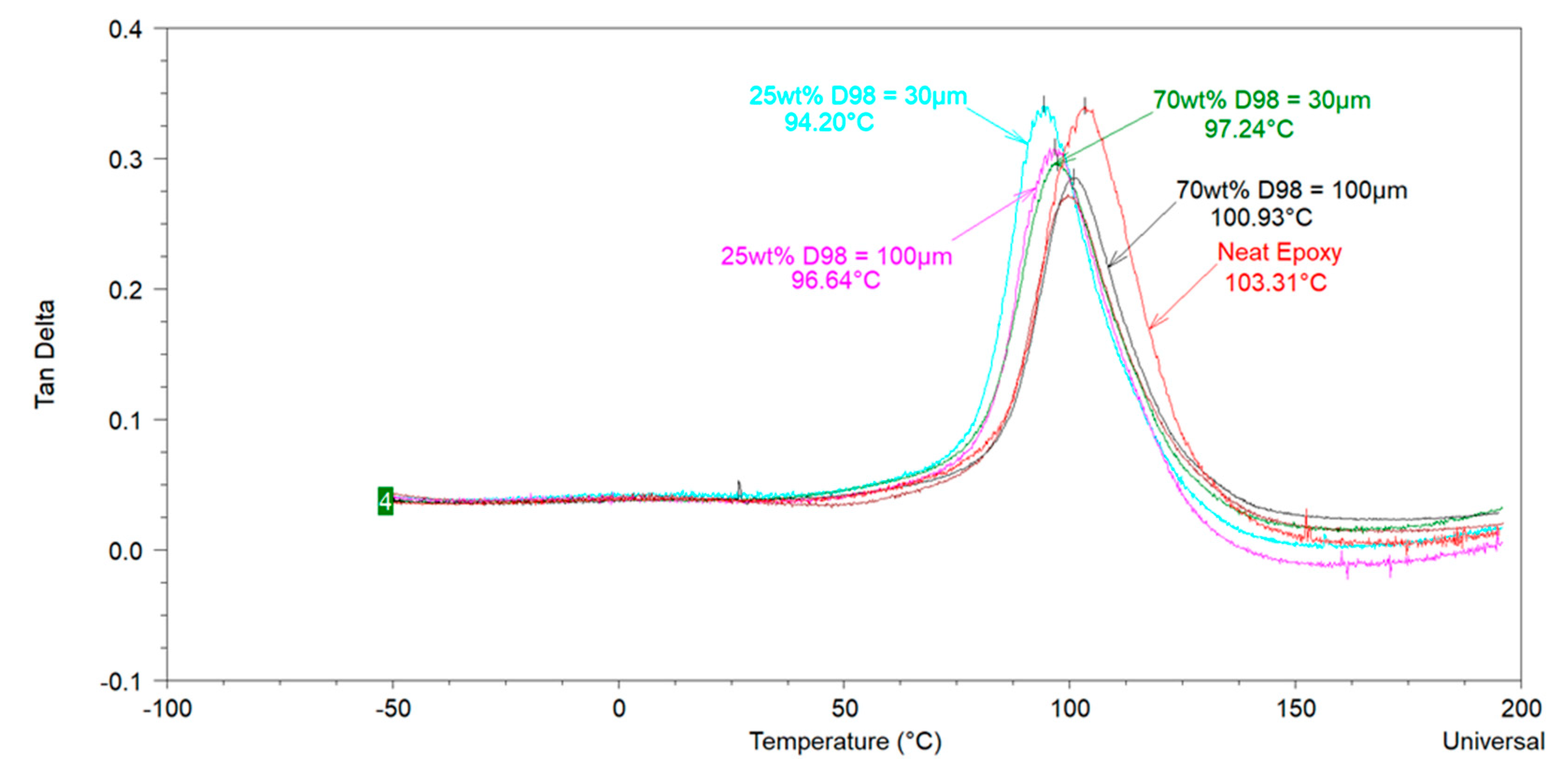
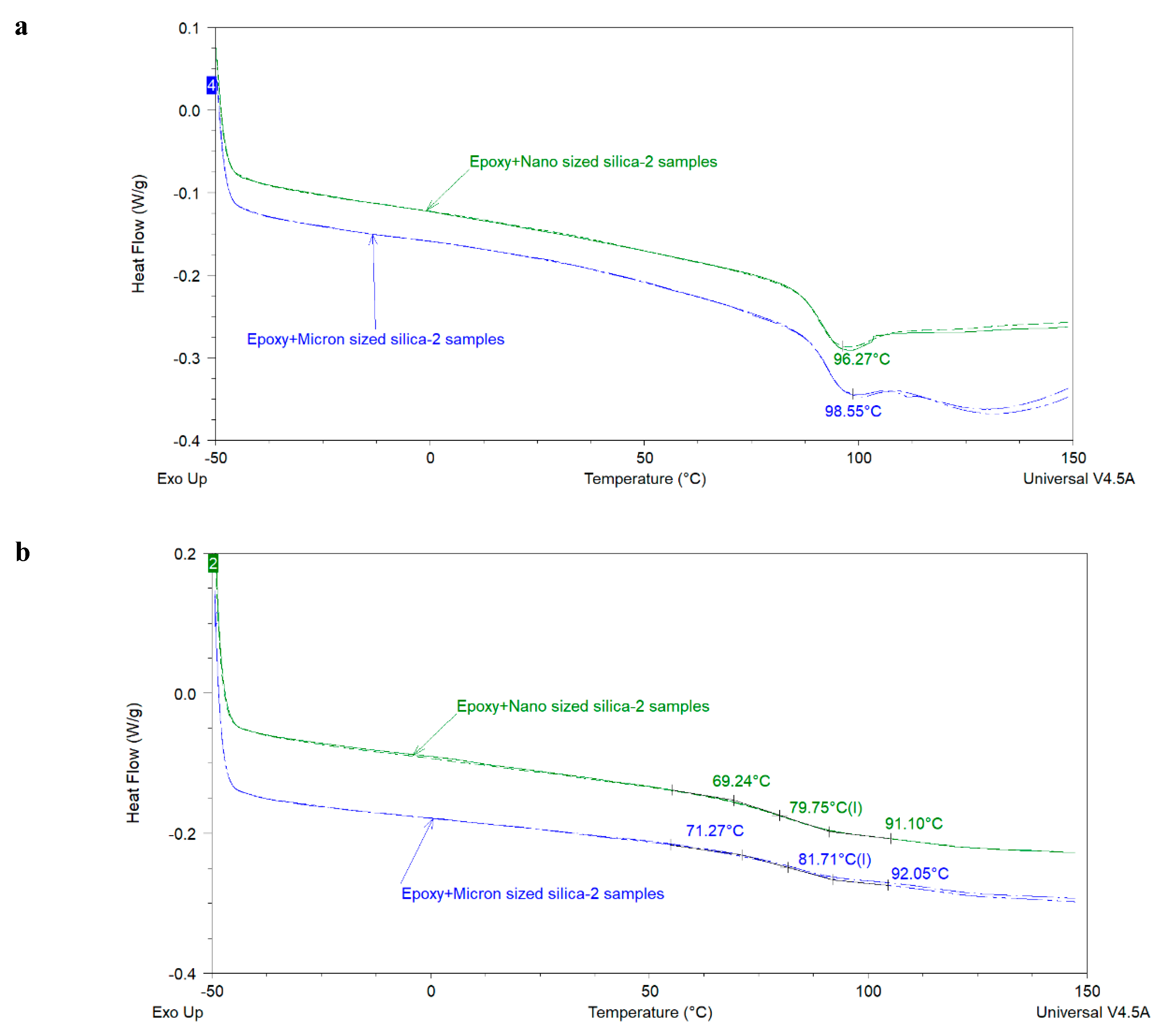
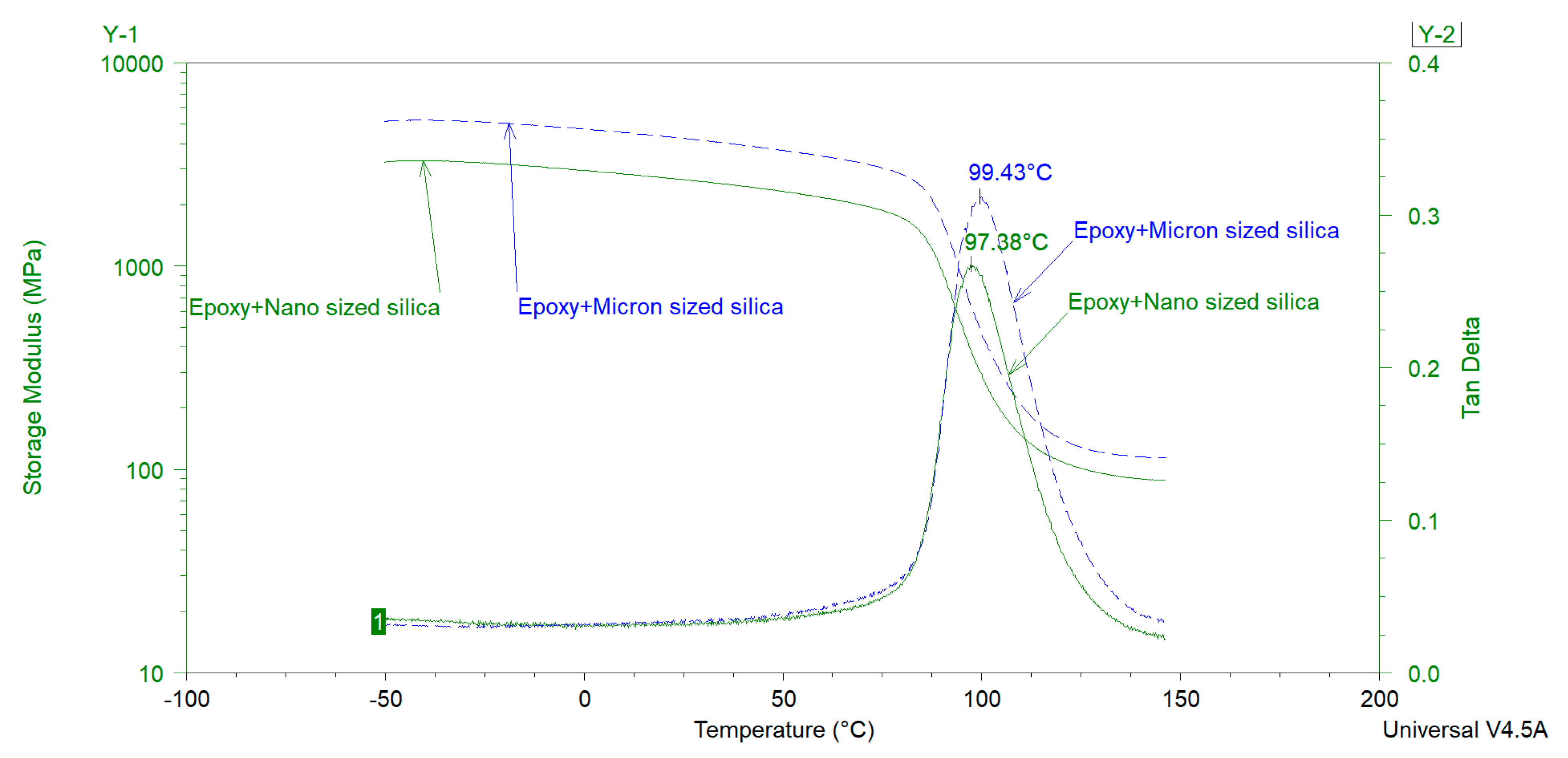
The glass transition temperature (
Tg) is an important thermal property of organic coatings which contributes in determining their application area and service life properties. Various researchers have investigated the influence of extender particle size on the
Tg of thermosetting epoxies. Sun et al. [
7] used diglycidylether of bisphenol-A (DGEBA) resin and hexahydro-4-methylphthalic anhydride (HMPA) hardener along with different particles like silica, silver, aluminum, and carbon black. These particles were of different sizes. The authors showed that at high amounts of silica, silver, and aluminum, the
Tg of cured epoxy containing nanoparticles was lower than the
Tg of the epoxy containing a similar type of microparticles which was attributed to the increase in free volume due to the enhanced resin-filler interface using nanoparticles. Liu et al. [
8] dispersed DGEBA in a commercial product containing pre-dispersed colloidal nanosilica particles (30–31 wt%) in methylisobutylketone (MIBK) and cured the resultant formulations with 4,4- diaminodiphenylmethane (DDM) to obtain silica amounts in the range of 10% to 70%. DSC analysis of the cured samples showed that, in comparison to the neat epoxy sample, the
Tg first decreased with up to 30% nanosilica amount (i.e., 40 °C reduction in
Tg was noticed) followed by an increase in
Tg with up to 50% nanosilica amount. At 70% nanosilica content, no
Tg was observed. The authors suggested that at low extender content,
Tg decreased due to the plasticization effect of nanosilica whereas at high extender content the motion of the polymer chains was highly hindered by the particles leading to an increase in
Tg. According to Chen et al. [
9] commercially available nanosilica dispersions contain surface treated nanosilica. These authors used various analytical techniques and estimated that such nanosilica can have an organic layer on the surface of the particles which may account for 3 wt% of the dried powder. Their results showed that good dispersion of nanosilica up to 20 wt% into model epoxy-amine nanocomposite can result in 50 °C reduction in the
Tg of the cured model nanocomposite (in comparison to the system without nanosilica). Dittanet et al. [
3] showed that silica nanoparticles of sizes from 23 to 170 nm do not impact the
Tg of epoxy cured with piperidine by changing the volume concentration of nanoparticles from 0% to 30%. Note that the silica nanoparticles used by these authors were surface treated with organosilane. No other information was provided about the type of organosilane used as it would be important to understand whether the organosilane played a role in the observed
Tg independence of silica particle size or not. Similar results were obtained with respect to the
Tg of the nanocomposite when glass spheres (average diameter of 42 µm) were combined with silica nanoparticles to simulate a bimodal particle size distribution [
10]. When using epoxy resin cured with piperidine and silica nanoparticles of 20 nm and 80 nm average diameter, the results reported by Liang et al. [
11] were similar to Dittanet et al. [
3]. Preghenella et al. [
12] studied the effect of fumed nano-silica amount on the
Tg of low molecular weight DGEBA based epoxy, cured with a polyamide curing agent. The amount of silica with the same particle size was varied in the range of 3.3 to 9.2 vol%. The authors showed that the
Tg of the cured resin filled with silica is lower in comparison to neat cured epoxy up to a silica content of 6.4 vol% followed by an increase at a silica content of 9.2 vol%. It was proposed by the authors that the initial
Tg depression (up to 6.4 vol% of filler content) was probably due to the ability of the silica nanoparticles to prevent a complete cure of the epoxy which lead to a reduction in crosslinking degree. In the case of incomplete reaction during the curing period, DSC heating scans show an exothermic event which can be attributed to the heat released during the reaction of epoxy with the curing agent. No such exothermic events were presented by the authors in DSC heating scans as indication of incomplete cure of the used epoxy resin. At the highest filler content, the
Tg was increased due to the increased physical immobilization of polymer chains near the filler surface. Recently, El-Fattah et al. [
13] investigated the effect of fumed nanosilica particle size and amount on the
Tg of room temperature cured epoxy coating. By using FTIR, the authors showed that silanol groups on the silica surface chemically react with the epoxy groups due to the local heat produced during ultrasonication and that the extent of reaction in smaller particles (average particle size of 7 nm) was higher than that in larger particles (average particle size of 30 nm). However, the effect of silica particle size on the
Tg of coatings was not significant as was expected due to this reaction. At similar amounts of silica nanoparticles, the
Tg of the coating containing 7 nm diameter particles was found to be 7.5% lower than the
Tg of the neat epoxy, whereas, this difference was 4.5% for the coating containing 30 nm diameter particles. Reduction in
Tg was noticed up to 2 wt% of both silica types after which the
Tg of the coating was found to be similar to the neat epoxy coating. The authors attributed this behavior to poor dispersion at higher nanosilica amounts.
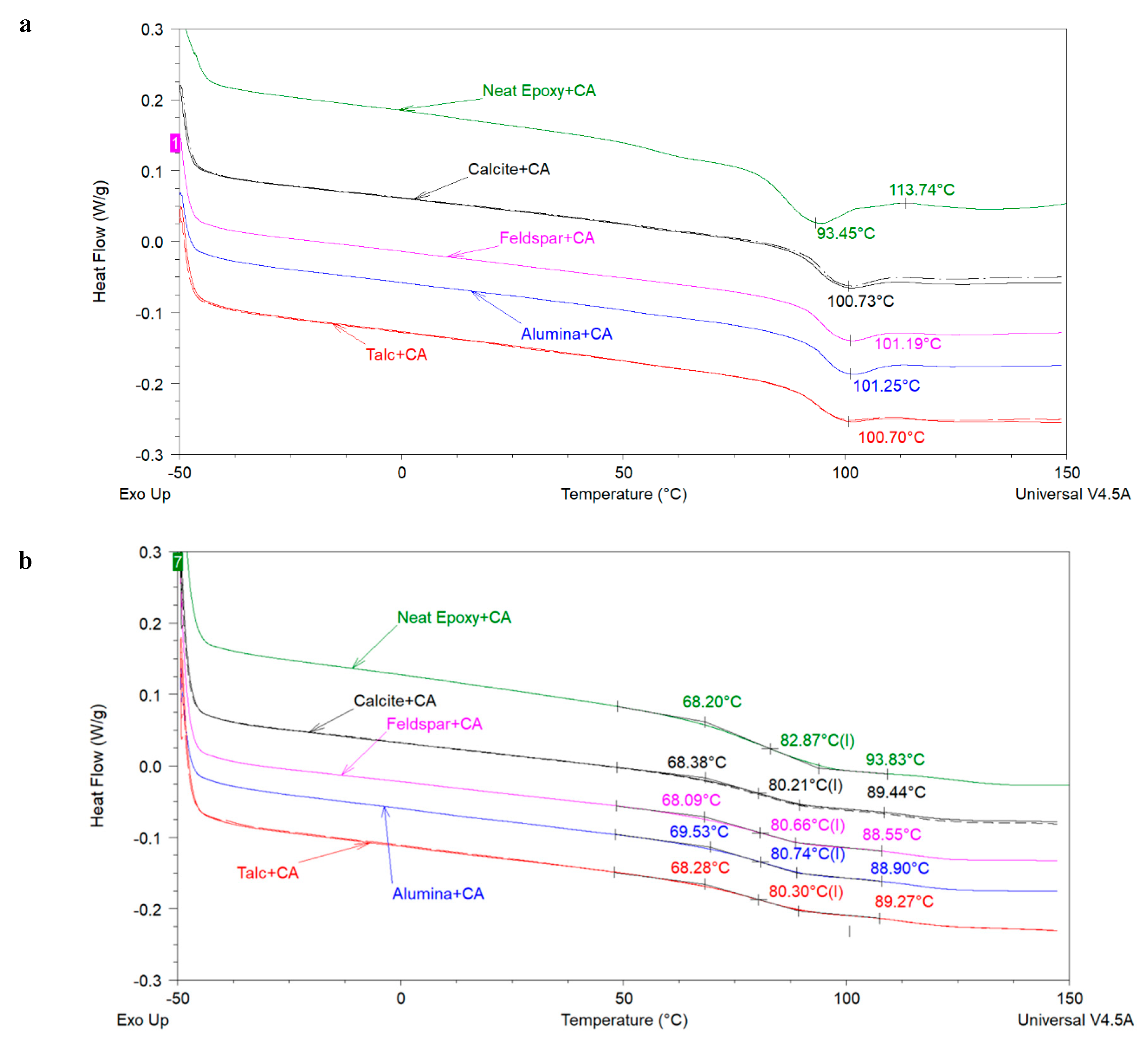
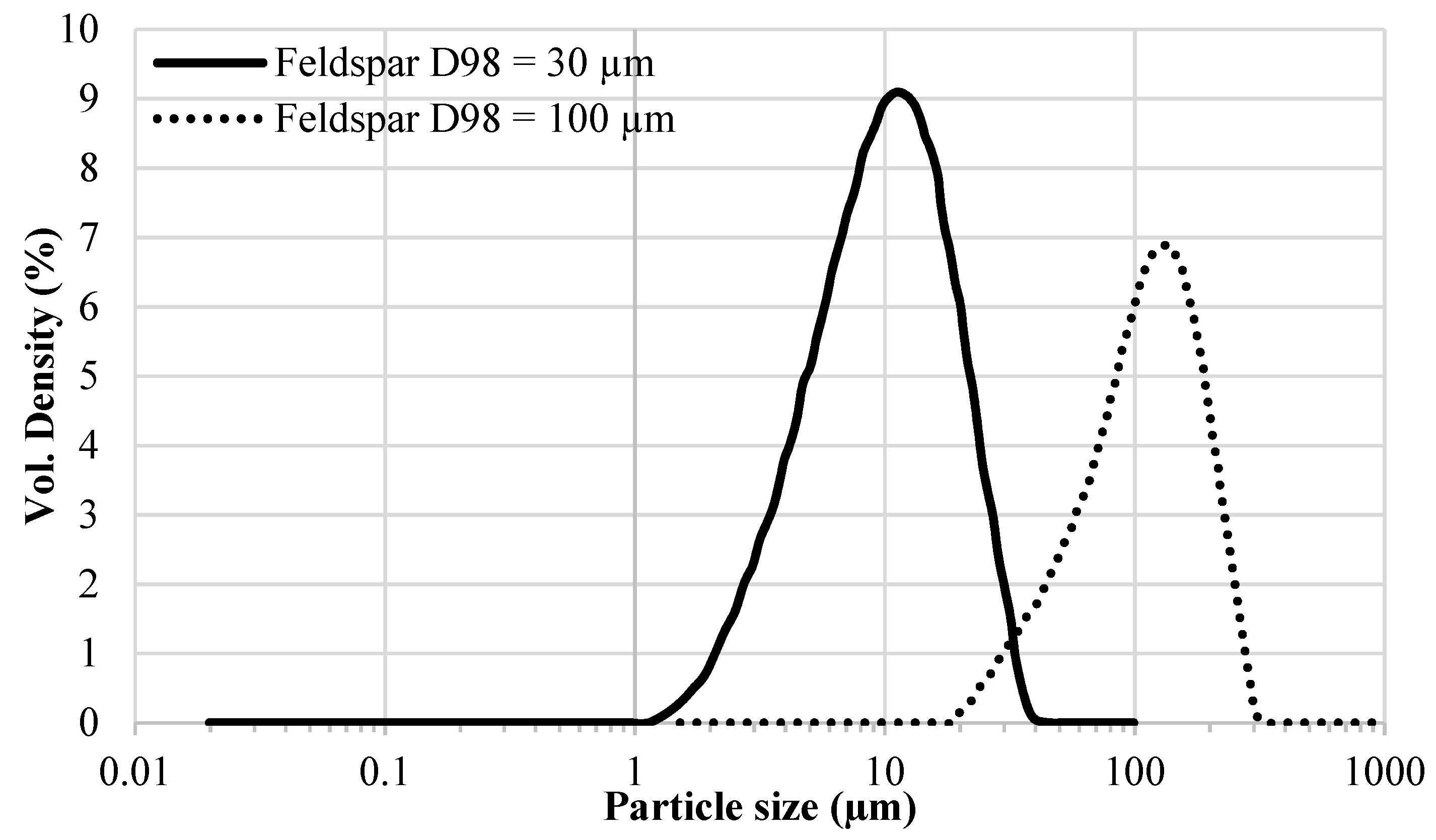
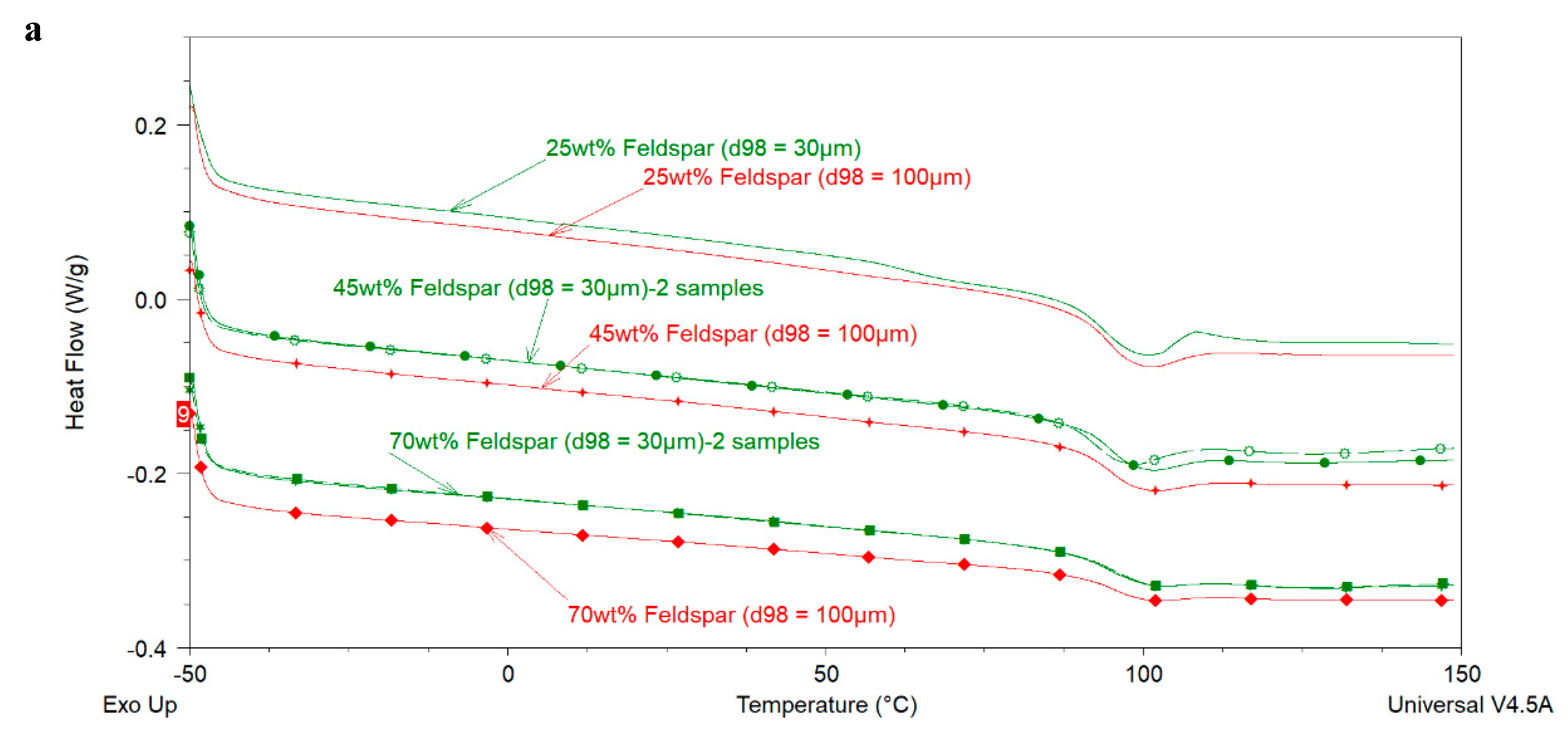
The above-mentioned literature review shows that the effect of extender particle size on the
Tg of epoxy nanocomposites is not completely understood in the model systems where mostly one extender was used. Regarding 2K organic epoxy coatings, studies showing the effect of extender particle size on the
Tg of the coatings are scarce in the open literature. In addition, several different silica types have been used in these studies. It is well-known that fumed, precipitated, and colloidal silica have very different physical properties (particle size, particle size distribution, surface area, pore volume, pore diameter, pore size distribution) which may lead to different dispersibility characteristics of these silica types [
6,
14] and, therefore, may have been a reason for the mixed results obtained by different researchers. In this work the aim is to investigate the effect of extender particle size and loading (i.e., the amount of extender in component A or in the total formulation) on the
Tg of model epoxy-amine coatings by using commercially important extenders. Furthermore, the effect of extender particle size on the
Tg of model systems with the same lamda value (
Λ) was also studied.
Λ is the ratio of pigment volume concentration (PVC) to critical pigment volume concentration (CPVC). PVC indicates the volumetric concentration of extenders (or pigments) in the coating. CPVC is representative of the PVC corresponding to the random tightest possible packing of the extender (or pigment) particles and the minimum amount of binder necessary to fill the interstices between extender particles.
Λ value less than 1 (i.e., PVC <CPVC) typically indicates that the coating film is continuous and contains only polymer (or binder) and pigments.
Λ values very close to or higher than 1 (i.e., PVC ≥CPVC) indicate that the coating film has air pockets around the extender particles due to lack of polymer. The importance and role of PVC and CPVC in coating properties was reviewed by Perera et al. [
4]. Differential Scanning Calorimetery (DSC) and Dynamic Mechanical Analysis (DMA) were mainly used in this study to measure the
Tg of the model coatings.