Responsive Adsorption of N-Isopropylacrylamide Based Copolymers on Polymer Brushes
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of PNIPAM and PAA Brushes
2.2. Synthesis of Amino-Terminated P(NIPAM-co-AA) and P(NIPAM-co-MADAP) Telomers
2.2.1. Synthesis of Amino-Terminated P(NIPAM-co-MADAP)
2.2.2. Synthesis of Amino-Terminated P(NIPAM-co-AA)
2.3. Ellipsometry
2.4. Neutron Reflectivity
3. Results and Discussion
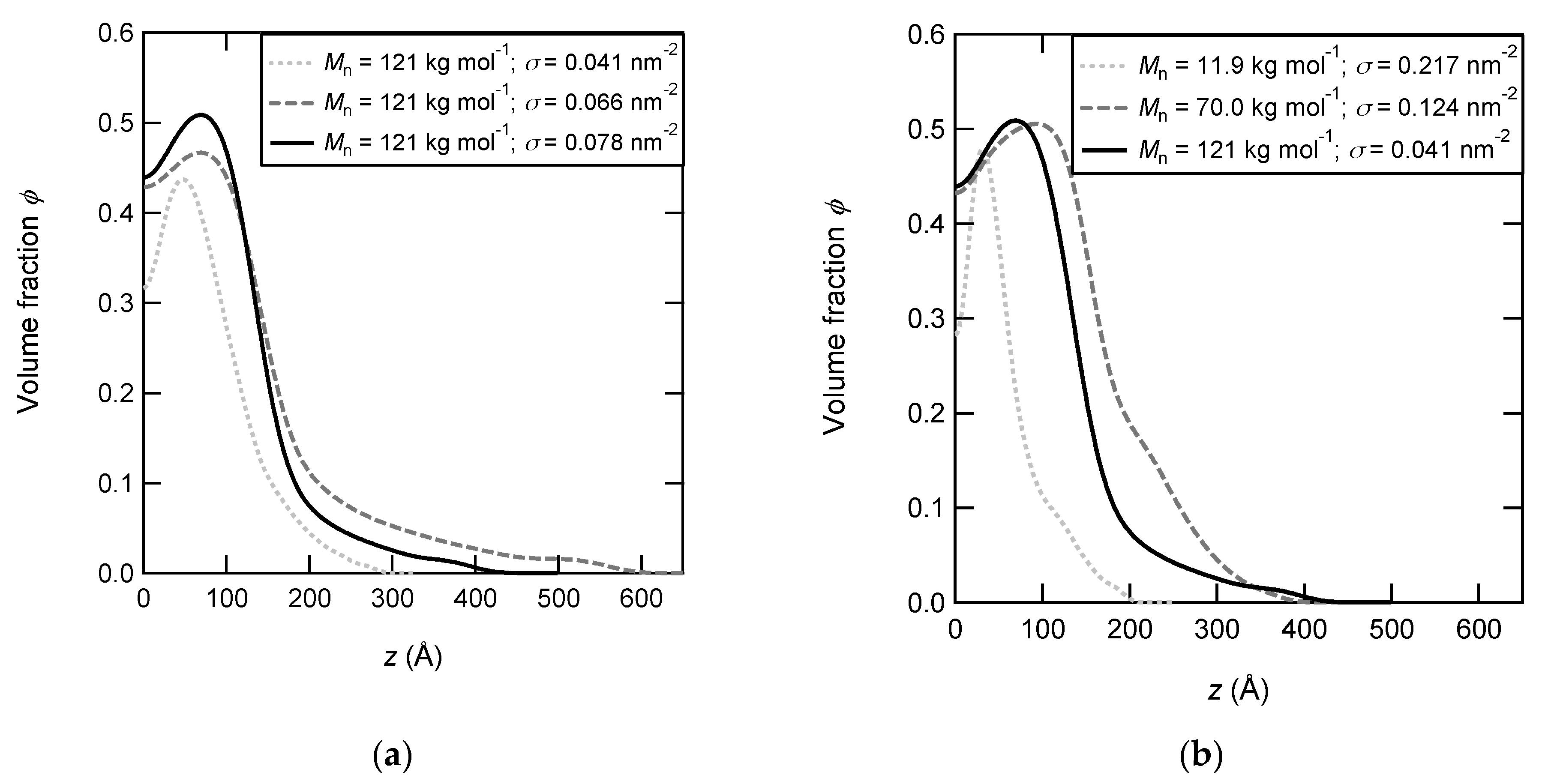
3.1. Characterization of Dry PNIPAM Brushes
3.2. Swelling Behavior of PNIPAM Brushes below LCST
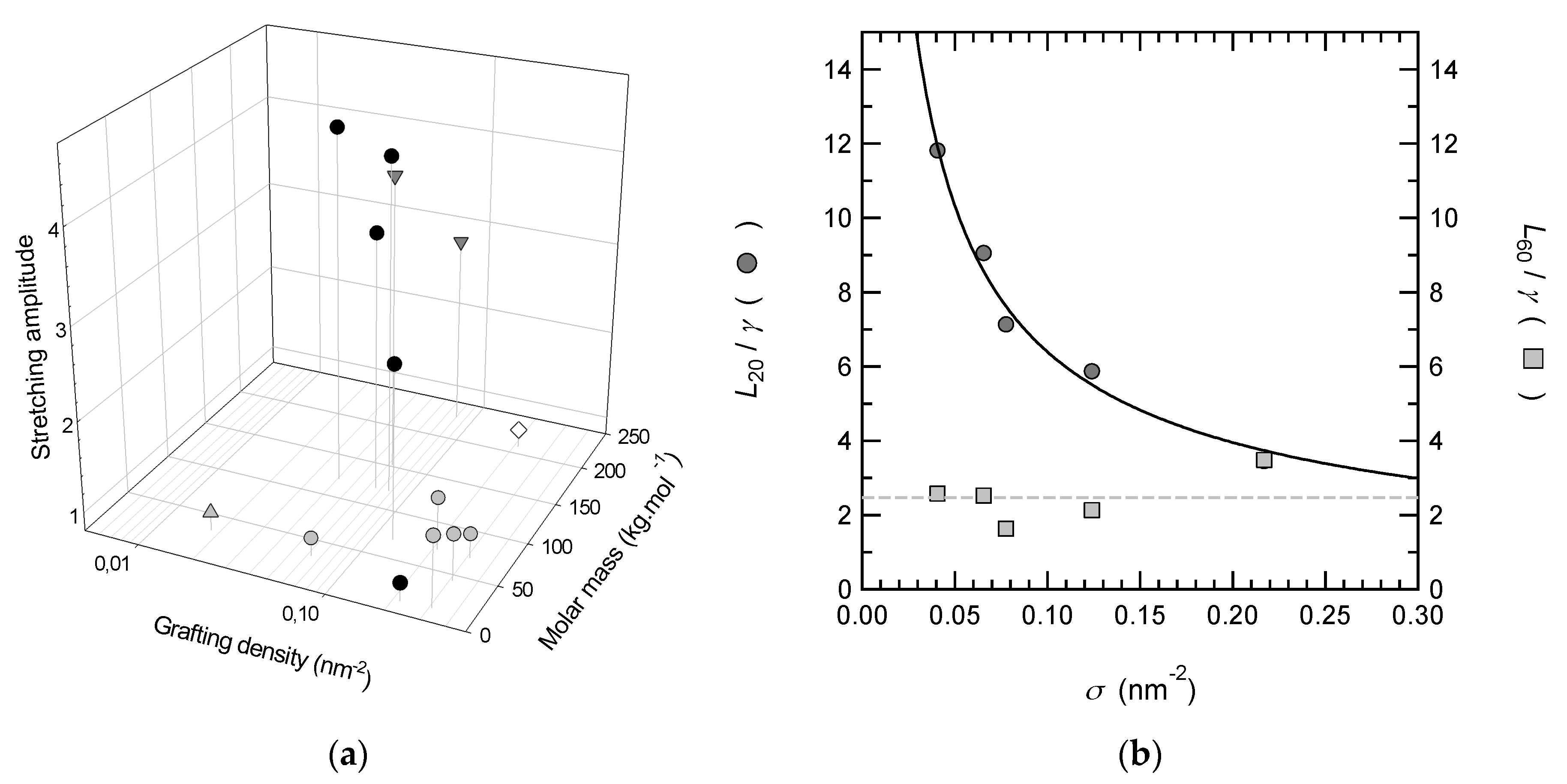
3.3. Shrinkage of PNIPAM Brushes above LCST
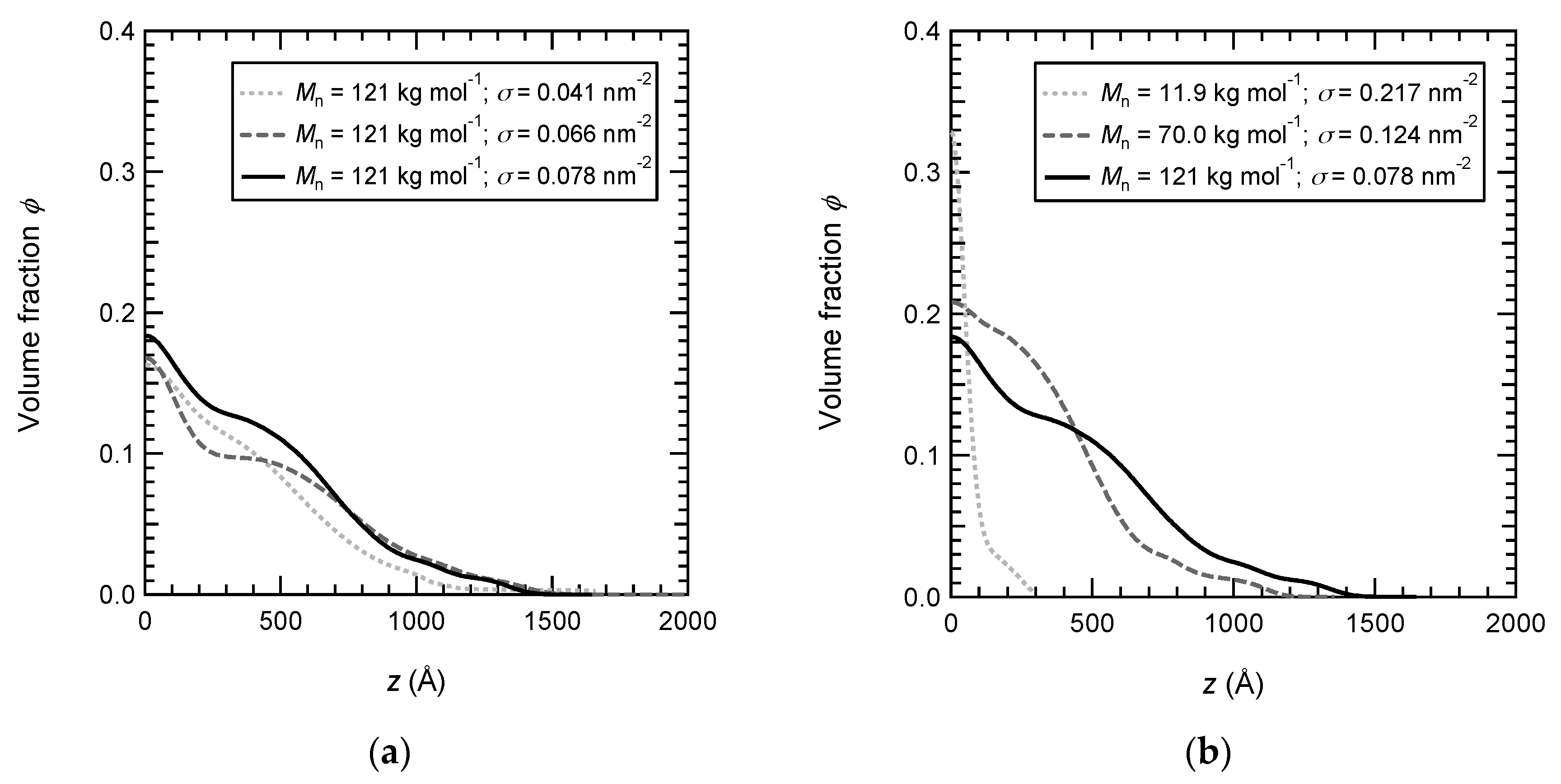
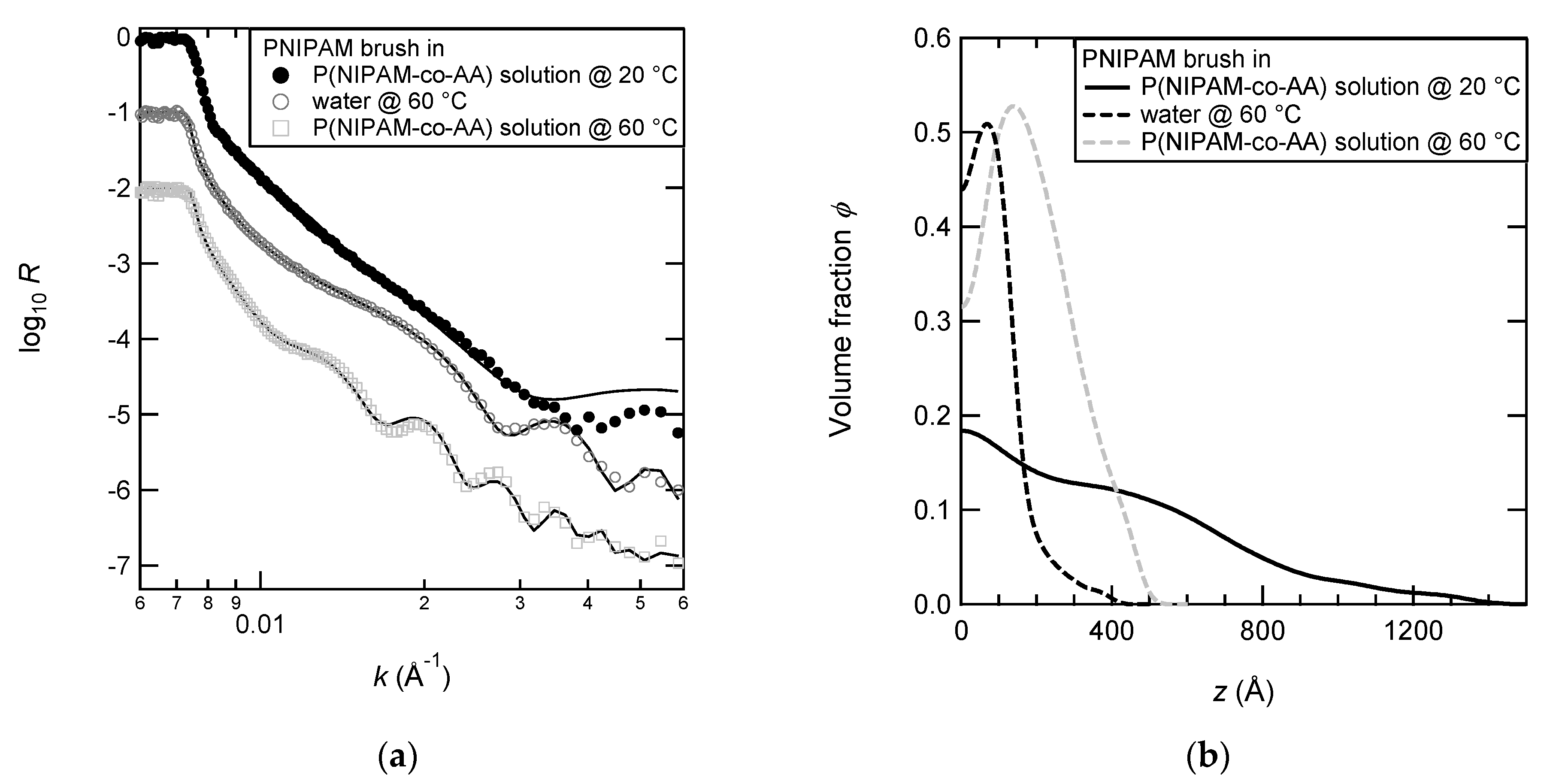
3.4. Complexation of PNIPAM Brushes with P(NIPAM-co-AA) Linear Copolymers
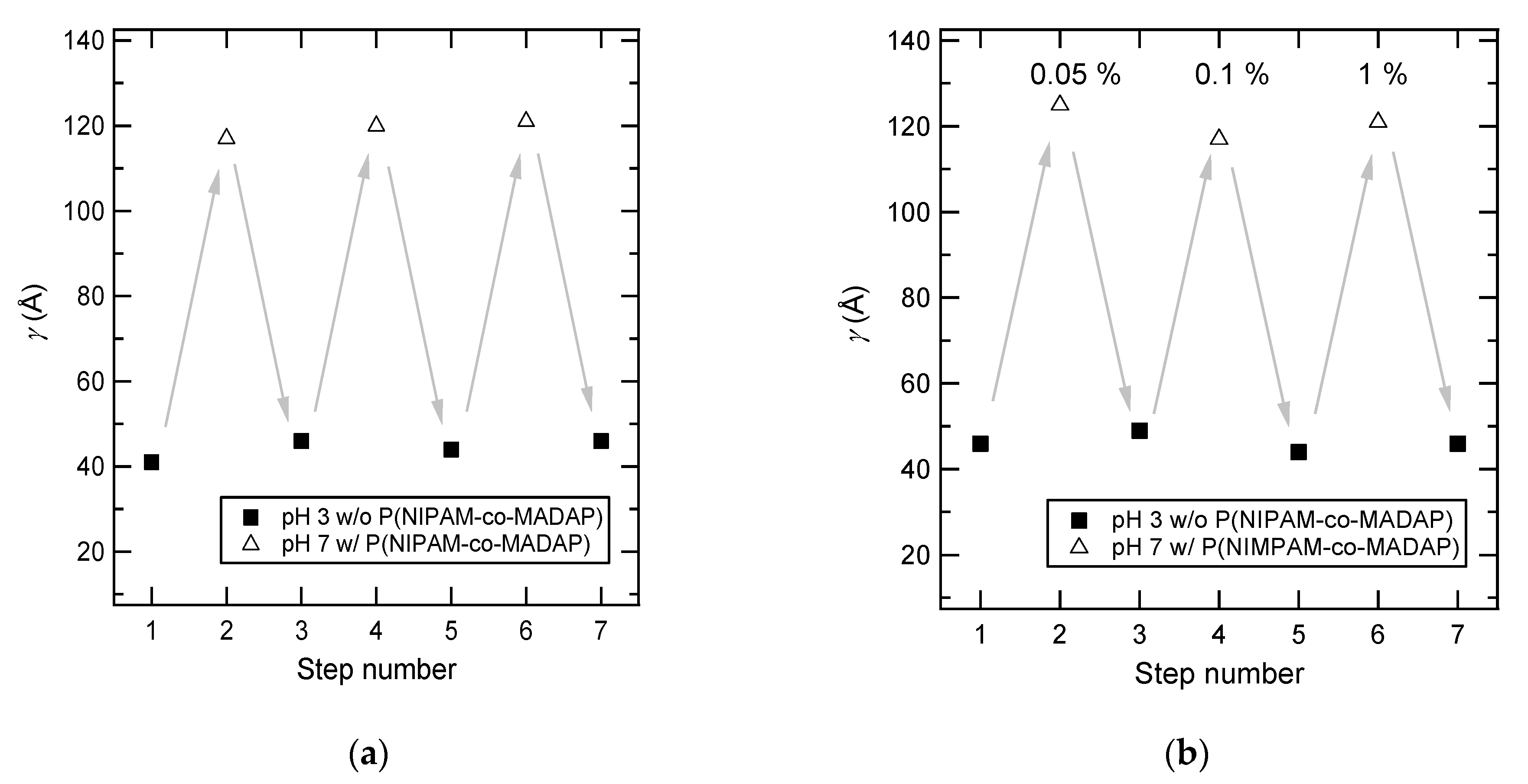
3.5. pH-Reversible Adsorption on PAA Brushes
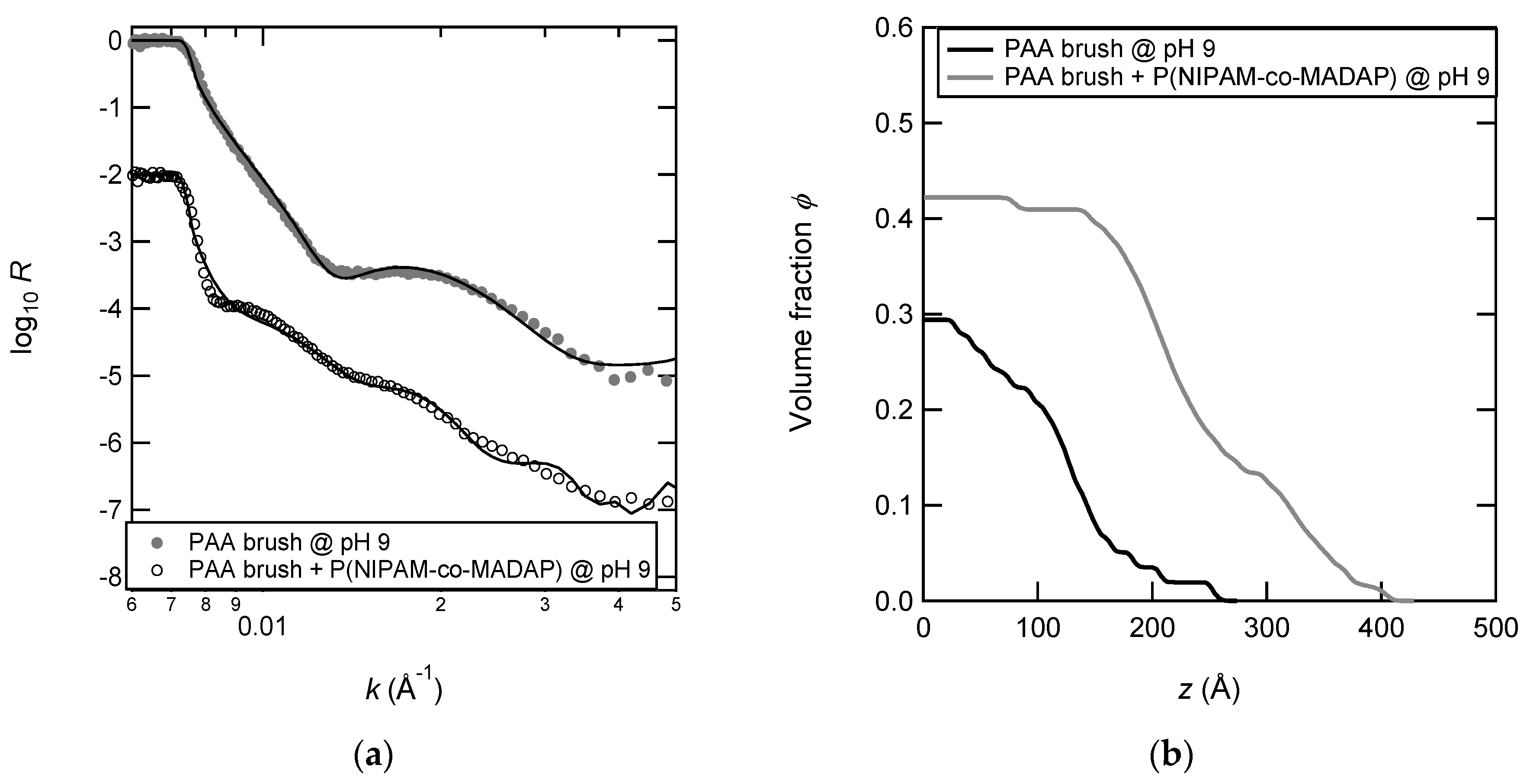
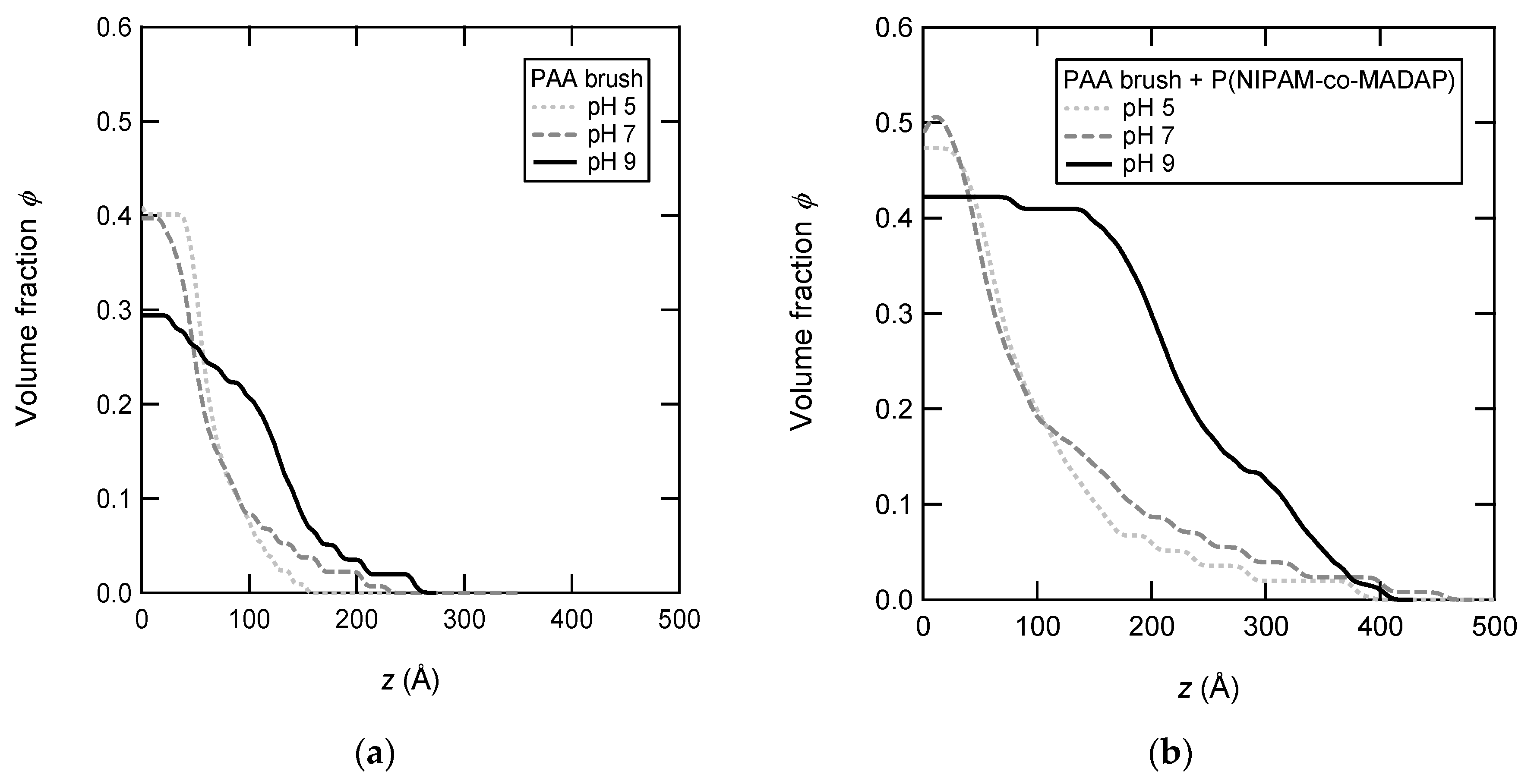
3.6. Density Profiles of PAA Brushes with the Adsorbed Copolymers
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Advincula, R.C.; Brittain, W.J.; Caster, K.C.; Rühe, J. Polymer Brushes; Wiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- Ruhe, J.; Ballauff, M.; Biesalski, M.; Dziezok, P.; Grohn, F.; Johannsmann, D.; Houbenov, N.; Hugenberg, N.; Konradi, R.; Minko, S.; et al. Polyelectrolyte brushes. In Polyelectrolytes with Defined Molecular Architecture I; Schmidt, M., Ed.; Springer: Berlin, Germany, 2004; Volume 165, pp. 79–150. [Google Scholar]
- Toomey, R.; Tirrell, M. Functional polymer brushes in aqueous media from self-assembled and surface-initiated polymers. In Annual Review of Physical Chemistry; Annual Reviews: Palo Alto, CA, USA, 2008; Volume 59, pp. 493–517. [Google Scholar]
- Weir, M.P.; Parnell, A.J. Water Soluble Responsive Polymer Brushes. Polymers 2011, 3, 2107–2132. [Google Scholar] [CrossRef]
- Liu, R.; De Leonardis, P.; Tirelli, N.; Saunders, B.R. Thermally-responsive surfaces comprising grafted poly(N-isopropylacrylamide) chains: Surface characterisation and reversible capture of dispersed polymer particles. J. Colloid Interface Sci. 2009, 340, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Lego, B.; Skene, W.G.; Giasson, S. Swelling Study of Responsive Polyelectrolyte Brushes Grafted from Mica Substrates: Effect of pH, Salt, and Grafting Density. Macromolecules 2010, 43, 4384–4393. [Google Scholar] [CrossRef]
- Kitano, H.; Kondo, T.; Suzuki, H.; Ohno, K. Temperature-responsive polymer-brush constructed on a glass substrate by atom transfer radical polymerization. J. Colloid Interface Sci. 2010, 345, 325–331. [Google Scholar] [CrossRef]
- Alfhaid, L.; Seddon, W.D.; Williams, N.H.; Geoghegan, M. Double-network hydrogels improve pH-switchable adhesion. Soft Matter 2016, 12, 5022–5028. [Google Scholar] [CrossRef] [PubMed]
- Laloyaux, X.; Mathy, B.; Nysten, B.; Jonas, A.M. Surface and Bulk Collapse Transitions of Thermo responsive Polymer Brushes. Langmuir 2010, 26, 838–847. [Google Scholar] [CrossRef]
- Nomura, A.; Okayasu, K.; Ohno, K.; Fukuda, T.; Tsujii, Y. Lubrication Mechanism of Concentrated Polymer Brushes in Solvents: Effect of Solvent Quality and Thereby Swelling State. Macromolecules 2011, 44, 5013–5019. [Google Scholar] [CrossRef]
- Takasu, K.; Kushiro, K.; Hayashi, K.; Iwasaki, Y.; Inoue, S.; Tamechika, E.; Takai, M. Polymer brush biointerfaces for highly sensitive biosensors that preserve the structure and function of immobilized proteins. Sens. Actuators B Chem. 2015, 216, 428–433. [Google Scholar] [CrossRef]
- Wu, C.Q.; Zhou, Y.D.; Wang, H.T.; Hu, J.H. P4VP Modified Zwitterionic Polymer for the Preparation of Antifouling Functionalized Surfaces. Nanomaterials 2019, 9, 706. [Google Scholar] [CrossRef]
- Bratek-Skicki, A.; Cristaudo, V.; Savocco, J.; Nootens, S.; Morsomme, P.; Delcorte, A.; Dupont-Gillain, C. Mixed Polymer Brushes for the Selective Capture and Release of Proteins. Biomacromolecules 2019, 20, 778–789. [Google Scholar] [CrossRef]
- Balamurugan, S.; Mendez, S.; Balamurugan, S.S.; O’Brien, M.J.; Lopez, G.P. Thermal response of poly(N-isopropylacrylamide) brushes probed by surface plasmon resonance. Langmuir 2003, 19, 2545–2549. [Google Scholar] [CrossRef] [PubMed]
- Plunkett, K.N.; Zhu, X.; Moore, J.S.; Leckband, D.E. PNIPAM chain collapse depends on the molecular weight and grafting density. Langmuir 2006, 22, 4259–4266. [Google Scholar] [CrossRef] [PubMed]
- Yim, H.; Kent, M.S.; Mendez, S.; Balamurugan, S.S.; Balamurugan, S.; Lopez, G.P.; Satija, S. Temperature-dependent conformational change of PNIPAM grafted chains at high surface density in water. Macromolecules 2004, 37, 1994–1997. [Google Scholar] [CrossRef]
- Zhu, X.; Yan, C.; Winnik, F.M.; Leckband, D. End-grafted low-molecular-weight PNIPAM does not collapse above the LCST. Langmuir 2007, 23, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, Y.; Kikuchi, A.; Yamato, M.; Okano, T. Ultrathin poly(N-isopropylacrylamide) grafted layer on polystyrene surfaces for cell adhesion/detachment control. Langmuir 2004, 20, 5506–5511. [Google Scholar] [CrossRef] [PubMed]
- Canavan, H.E.; Cheng, X.H.; Graham, D.J.; Ratner, B.D.; Castner, D.G. Surface characterization of the extracellular matrix remaining after cell detachment from a thermoresponsive polymer. Langmuir 2005, 21, 1949–1955. [Google Scholar] [CrossRef]
- Canavan, H.E.; Graham, D.J.; Cheng, X.H.; Ratner, B.D.; Castner, D.G. Comparison of native extracellular matrix with adsorbed protein films using secondary ion mass spectrometry. Langmuir 2007, 23, 50–56. [Google Scholar] [CrossRef]
- Fukumori, K.; Akiyama, Y.; Yamato, M.; Kobayashi, J.; Sakai, K.; Okano, T. Temperature-responsive glass coverslips with an ultrathin poly (N-isopropylacrylamide) layer. Acta Biomater. 2009, 5, 470–476. [Google Scholar] [CrossRef]
- Reinhardt, M.; Kreuzer, M.; Geue, T.; Dahint, R.; Ballauff, M.; Steitz, R. Poly-acrylic Acid Brushes and Adsorbed Proteins. Z. Fur Phys. Chem. 2015, 229, 1119–1139. [Google Scholar] [CrossRef][Green Version]
- Yadav, V.; Jannes-Lizcano, Y.A.; Dewangan, N.K.; Park, N.; Li, T.H.; Robertson, M.L.; Conrad, J.C. Tuning Bacterial Attachment and Detachment via the Thickness and Dispersity of a pH-Responsive Polymer Brush. Acs Appl. Mater. Interfaces 2017, 9, 44900–44910. [Google Scholar] [CrossRef]
- Ishida, N.; Biggs, S. Effect of Grafting Density on Phase Transition Behavior for Poly(N-isopropylacryamide) Brushes in Aqueous Solutions Studied by AFM and QCM-D. Macromolecules 2010, 43, 7269–7276. [Google Scholar] [CrossRef]
- Kaholek, M.; Lee, W.K.; Ahn, S.J.; Ma, H.W.; Caster, K.C.; LaMattina, B.; Zauscher, S. Stimulus-responsive poly(N-isopropylacrylamide) brushes and nanopatterns prepared by surface-initiated polymerization. Chem. Mater. 2004, 16, 3688–3696. [Google Scholar] [CrossRef]
- Sanjuan, S.; Tran, Y. Synthesis of random polyampholyte brushes by atom transfer radical polymerization. J. Polym. Sci. Patr A Polym. Chem. 2008, 46, 4305–4319. [Google Scholar] [CrossRef]
- Suzuki, H.; Nurul, H.M.; Seki, T.; Kawamoto, T.; Haga, H.; Kawabata, K.; Takeoka, Y. Precise Synthesis and Physicochemical Properties of High-Density Polymer Brushes designed with Poly(N-isopropylacrylamide). Macromolecules 2010, 43, 9945–9956. [Google Scholar] [CrossRef]
- Turan, E.; Caykara, T. Construction of Hydroxyl-Terminated Poly(N-isopropylacrylamide) Brushes on Silicon Wafer via Surface-Initiated Atom Transfer Radical Polymerization. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 3880–3887. [Google Scholar] [CrossRef]
- Micciulla, S.; Soltwedel, O.; Lohmann, O.; von Klitzing, R. Temperature responsive behavior of polymer brush/polyelectrolyte multilayer composites. Soft Matter 2016, 12, 1176–1183. [Google Scholar] [CrossRef]
- Bittrich, E.; Burkert, S.; Muller, M.; Eichhorn, K.J.; Stamm, M.; Uhlmann, P. Temperature-Sensitive Swelling of Poly(N-isopropylacrylamide) Brushes with Low Molecular Weight and Grafting Density. Langmuir 2012, 28, 3439–3448. [Google Scholar] [CrossRef]
- Sudre, G.; Siband, E.; Hourdet, D.; Creton, C.; Cousin, F.; Tran, Y. Synthesis and Characterization of Poly(acrylic acid) Brushes: “Grafting-Onto” Route. Macromol. Chem. Phys. 2012, 213, 293–300. [Google Scholar] [CrossRef]
- Yim, H.; Kent, M.S.; Huber, D.L.; Satija, S.; Majewski, J.; Smith, G.S. Conformation of end-tethered PNIPAM chains in water and in acetone by neutron reflectivity. Macromolecules 2003, 36, 5244–5251. [Google Scholar] [CrossRef]
- Yim, H.; Kent, M.S.; Mendez, S.; Lopez, G.P.; Satija, S.; Seo, Y. Effects of grafting density and molecular weight on the temperature-dependent conformational change of poly(N-isopropylacrylamide) grafted chains in water. Macromolecules 2006, 39, 3420–3426. [Google Scholar] [CrossRef]
- Yim, H.; Kent, M.S.; Satija, S.; Mendez, S.; Balamurugan, S.S.; Balamurugan, S.; Lopez, C.P. Study of the conformational change of poly(N-isopropylacrylamide)-grafted chains in water with neutron reflection: Molecular weight dependence at high grafting density. J. Polym. Sci. Part B Polym. Phys. 2004, 42, 3302–3310. [Google Scholar] [CrossRef]
- Annaka, M.; Yahiro, C.; Nagase, K.; Kikuchi, A.; Okano, T. Real-time observation of coil-to-globule transition in thermosensitive poly (N-isopropylacrylamide) brushes by quartz crystal microbalance. Polymer 2007, 48, 5713–5720. [Google Scholar] [CrossRef]
- Sudre, G.; Hourdet, D.; Creton, C.; Cousin, F.; Tran, Y. pH-Responsive Swelling of Poly(acrylic acid) Brushes Synthesized by the Grafting Onto Route. Macromol. Chem. Phys. 2013, 214, 2882–2890. [Google Scholar] [CrossRef]
- Wu, T.; Gong, P.; Szleifer, I.; Vlcek, P.; Subr, V.; Genzer, J. Behavior of surface-anchored poly(acrylic acid) brushes with grafting density gradients on solid substrates: 1. Experiment. Macromolecules 2007, 40, 8756–8764. [Google Scholar] [CrossRef]
- Aulich, D.; Hoy, O.; Luzinov, I.; Brucher, M.; Hergenroder, R.; Bittrich, E.; Eichhorn, K.J.; Uhlmann, P.; Stamm, M.; Esser, N.; et al. In Situ Studies on the Switching Behavior of Ultrathin Poly(acrylic acid) Polyelectrolyte Brushes in Different Aqueous Environments. Langmuir 2010, 26, 12926–12932. [Google Scholar] [CrossRef]
- Dong, R.; Lindau, M.; Ober, C.K. Dissociation Behavior of Weak Polyelectrolyte Brushes on a Planar Surface. Langmuir 2009, 25, 4774–4779. [Google Scholar] [CrossRef]
- Yenice, Z.; Schoen, S.; Bildirir, H.; Genzer, J.; von Klitzing, R. Thermoresponsive PDMAEMA Brushes: Effect of Gold Nanoparticle Deposition. J. Phys. Chem. B 2015, 119, 10348–10358. [Google Scholar] [CrossRef]
- Xue, C.Y.; Yonet-Tanyeri, N.; Brouette, N.; Sferrazza, M.; Braun, P.V.; Leckband, D.E. Protein Adsorption on Poly(N-isopropylacrylamide) Brushes: Dependence on Grafting Density and Chain Collapse. Langmuir 2011, 27, 8810–8818. [Google Scholar] [CrossRef]
- Evers, F.; Reichhart, C.; Steitz, R.; Tolan, M.; Czeslik, C. Probing adsorption and aggregation of insulin at a poly(acrylic acid) brush. Phys. Chem. Chem. Phys. 2010, 12, 4375–4382. [Google Scholar] [CrossRef]
- de Vos, W.M.; Biesheuvel, P.M.; de Keizer, A.; Kleijn, J.M.; Stuart, M.A.C. Adsorption of the protein bovine serum albumin in a planar poly(acrylic acid) brush layer as measured by optical reflectometry. Langmuir 2008, 24, 6575–6584. [Google Scholar] [CrossRef]
- Siband, E.; Tran, Y.; Hourdet, D. pH- and Thermo-responsive Polymer Assemblies in Aqueous Solution. Prog. Colloid Polym. Sci. 2010, 137, 19–22. [Google Scholar] [CrossRef]
- Siband, E.; Tran, Y.; Hourdet, D. Thermoresponsive Interpolyelectrolyte Complexation: Application to Macromolecular Assemblies. Macromolecules 2011, 44, 8185–8194. [Google Scholar] [CrossRef]
- Sanjuan, S.; Perrin, P.; Pantoustier, N.; Tran, Y. Synthesis and swelling behavior of pH-responsive polybase brushes. Langmuir 2007, 23, 5769–5778. [Google Scholar] [CrossRef] [PubMed]
- Burkert, S.; Bittrich, E.; Kuntzsch, M.; Muller, M.; Eichhorn, K.J.; Bellmann, C.; Uhlmann, P.; Stamm, M. Protein Resistance of PNIPAAm Brushes: Application to Switchable Protein Adsorption. Langmuir 2010, 26, 1786–1795. [Google Scholar] [CrossRef] [PubMed]
- Mendez, S.; Curro, J.G.; McCoy, J.D.; Lopez, G.P. Computational modeling of the temperature-induced structural changes of tethered poly(N-isopropylacrylamide) with self-consistent field theory. Macromolecules 2005, 38, 174–181. [Google Scholar] [CrossRef]
- Currie, E.P.K.; Norde, W.; Stuart, M.A.C. Tethered polymer chains: surface chemistry and their impact on colloidal and surface properties. Adv. Colloid Interface Sci. 2003, 100, 205–265. [Google Scholar] [CrossRef]
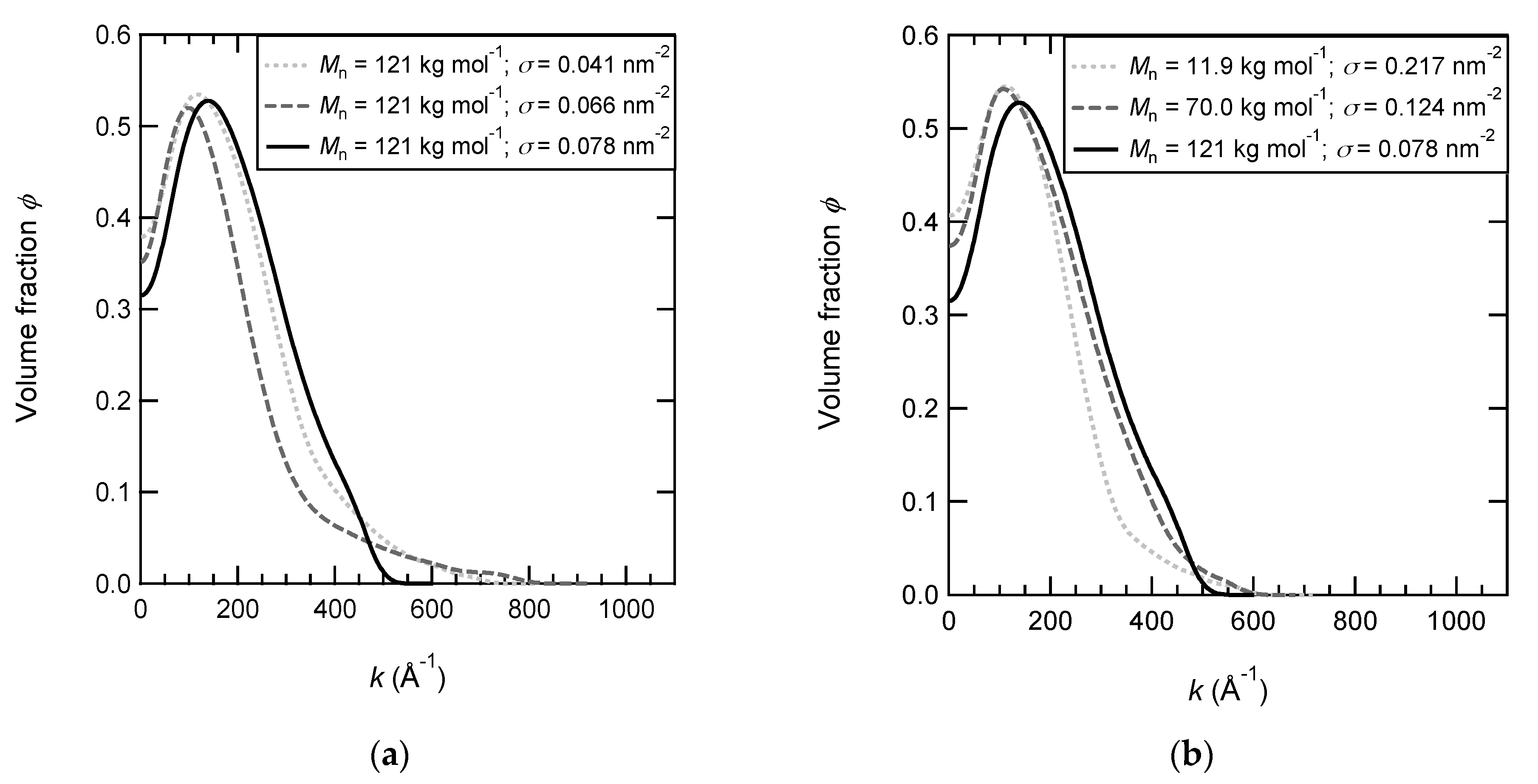

| Mn (g mol−1) | % Functionalized Polymer | Reaction Time (h) | γa (Å) | σb (nm−2) | Dc (Å) | 2R0 d (Å) | bN1/3 e (Å) |
|---|---|---|---|---|---|---|---|
| 121,000 | 100 | 144 | 113 | 0.064 | 39 | 184 | 56 |
| 121,000 | 100 | 24 | 95 | 0.054 | 43 | 184 | 56 |
| 121,000 | 10 | 24 | 59 | 0.034 | 55 | 184 | 56 |
| 121,000 | 10 | 1 | 21 | 0.012 | 91 | 184 | 56 |
| 70,000 | 100 | 144 | 104 | 0.100 | 32 | 140 | 47 |
| 70,000 | 100 | 72 | 88 | 0.085 | 34 | 140 | 47 |
| 70,000 | 100 | 24 | 72 | 0.069 | 38 | 140 | 47 |
| 70,000 | 10 | 24 | 50 | 0.048 | 46 | 140 | 47 |
| 70,000 | 10 | 1 | 19 | 0.018 | 74 | 140 | 47 |
| 11,900 | 100 | 144 | 31 | 0.217 | 24 | 58 | 26 |
| Mn (g mol−1) | D (Å) | (Nb3)1/3 (Å) | (V0)1/3 (Å) | (VF)1/3 (Å) | L20 (Å) | (L20D2)1/3 (Å) | φ20 = γ/L20 | L60 (Å) | (L60D2)1/3 (Å) | φ60 = γ/L60 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 121,000 | 39 | 56 | 148 | 240 | 806 | 107 | 11.3 | 0.14 | 185 | 66 | 4.4 | 0.61 |
| 121,000 | 43 | 56 | 148 | 259 | 860 | 117 | 10.8 | 0.11 | 240 | 76 | 3.6 | 0.40 |
| 121,000 | 55 | 56 | 148 | 252 | 697 | 128 | 7.6 | 0.08 | 152 | 77 | 4.6 | 0.39 |
| 70,000 | 32 | 47 | 113 | 188 | 611 | 86 | 10.4 | 0.17 | 222 | 61 | 2.8 | 0.47 |
| 11,900 | 24 | 26 | 47 | 59 | 107 | 40 | 3.2 | 0.29 | 108 | 40 | 1.0 | 0.29 |
| Mn (g mol−1) | γ (Å) | σ (nm−2) | γtotal (Å) | Ltotal (Å) | γads (Å) | Lads (Å) | Rads |
|---|---|---|---|---|---|---|---|
| 121,000 | 113 | 7.76 × 10−2 | 157 | 376 | 44 | 298 | 0.39 |
| 121,000 | 95 | 6.55 × 10−2 | 132 | 349 | 37 | 263 | 0.39 |
| 121,000 | 59 | 4.07 × 10−2 | 158 | 374 | 99 | 322 | 1.70 |
| 70,000 | 104 | 12.4 × 10−2 | 154 | 354 | 50 | 257 | 0.49 |
| 11,900 | 31 | 21.7 × 10−2 | 137 | 309 | 106 | 273 | 3.46 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sudre, G.; Siband, E.; Gallas, B.; Cousin, F.; Hourdet, D.; Tran, Y. Responsive Adsorption of N-Isopropylacrylamide Based Copolymers on Polymer Brushes. Polymers 2020, 12, 153. https://doi.org/10.3390/polym12010153
Sudre G, Siband E, Gallas B, Cousin F, Hourdet D, Tran Y. Responsive Adsorption of N-Isopropylacrylamide Based Copolymers on Polymer Brushes. Polymers. 2020; 12(1):153. https://doi.org/10.3390/polym12010153
Chicago/Turabian StyleSudre, Guillaume, Elodie Siband, Bruno Gallas, Fabrice Cousin, Dominique Hourdet, and Yvette Tran. 2020. "Responsive Adsorption of N-Isopropylacrylamide Based Copolymers on Polymer Brushes" Polymers 12, no. 1: 153. https://doi.org/10.3390/polym12010153
APA StyleSudre, G., Siband, E., Gallas, B., Cousin, F., Hourdet, D., & Tran, Y. (2020). Responsive Adsorption of N-Isopropylacrylamide Based Copolymers on Polymer Brushes. Polymers, 12(1), 153. https://doi.org/10.3390/polym12010153






