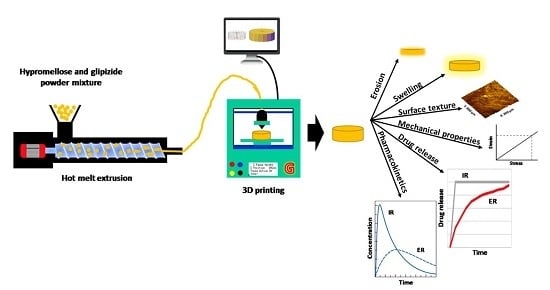
Plasticiser-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release and Pharmacokinetic Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Filaments
2.3. Physicochemical Characterisation of Filaments
2.3.1. Determination of Drug Loading
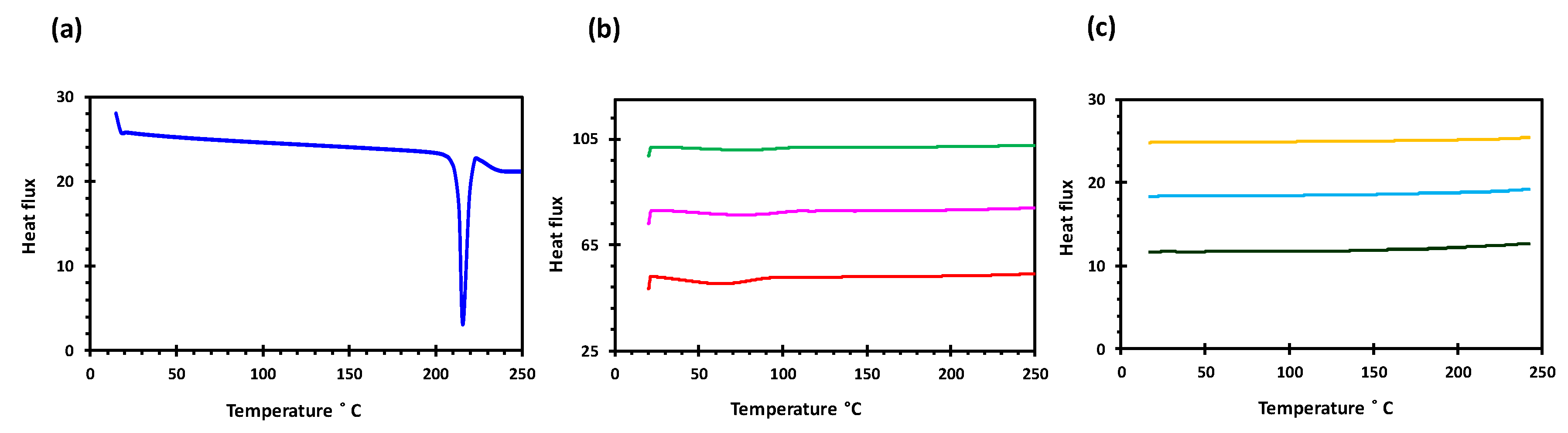
2.3.2. Differential Scanning Calorimetry (DSC)
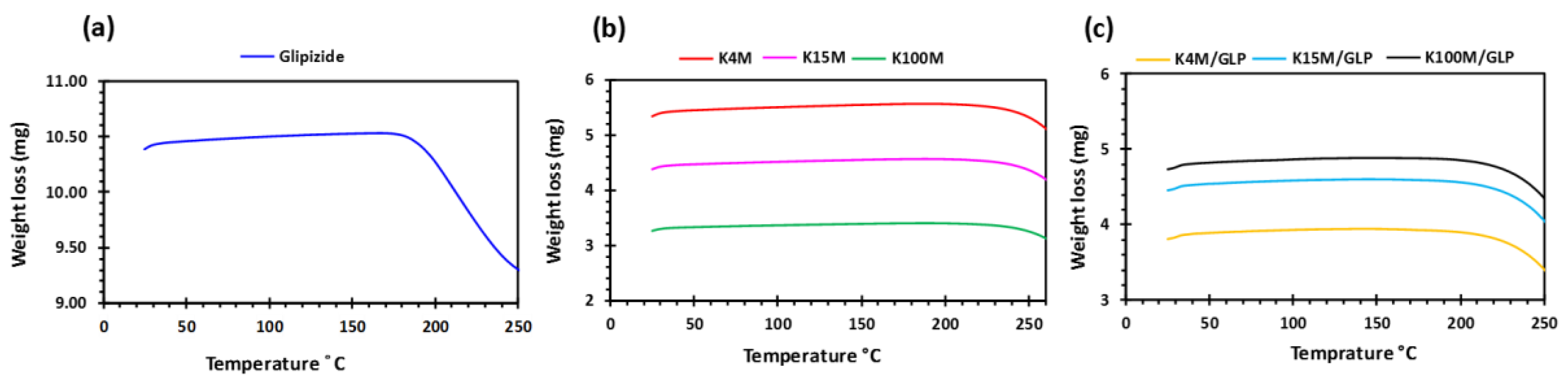
2.3.3. Thermogravimetric Analysis (TGA)
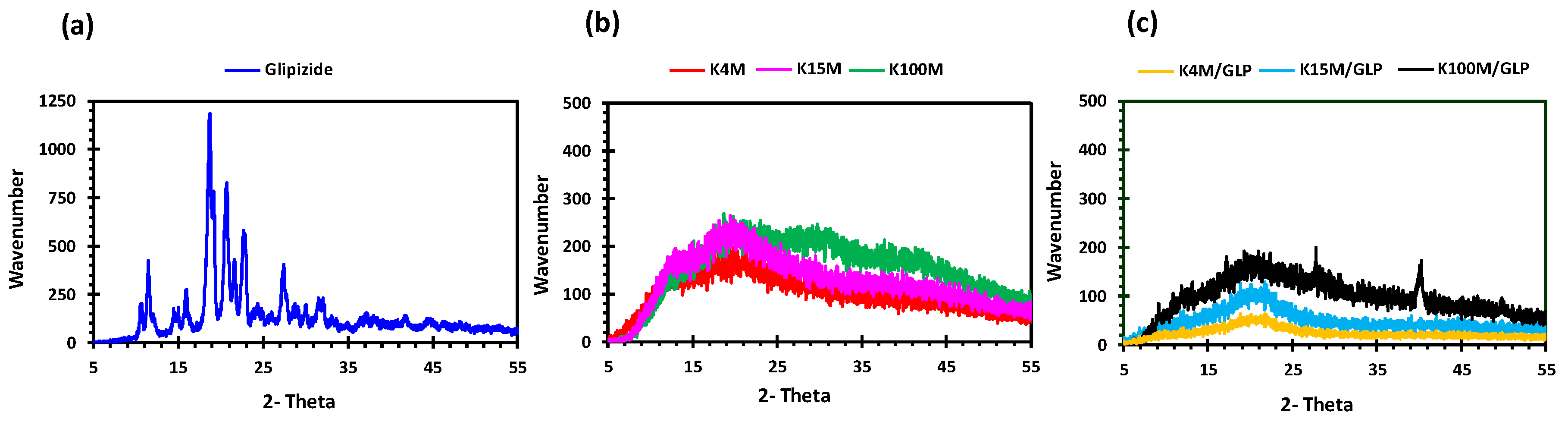
2.3.4. X-ray Powder Diffraction (XRPD)
2.3.5. Scanning Electron Microscopy (SEM)
2.3.6. Mechanical Testing
2.4. Fabrication of 3D Printed Glipizide Matrix Tablets
2.5. Characterisation of 3D Printed Matrices
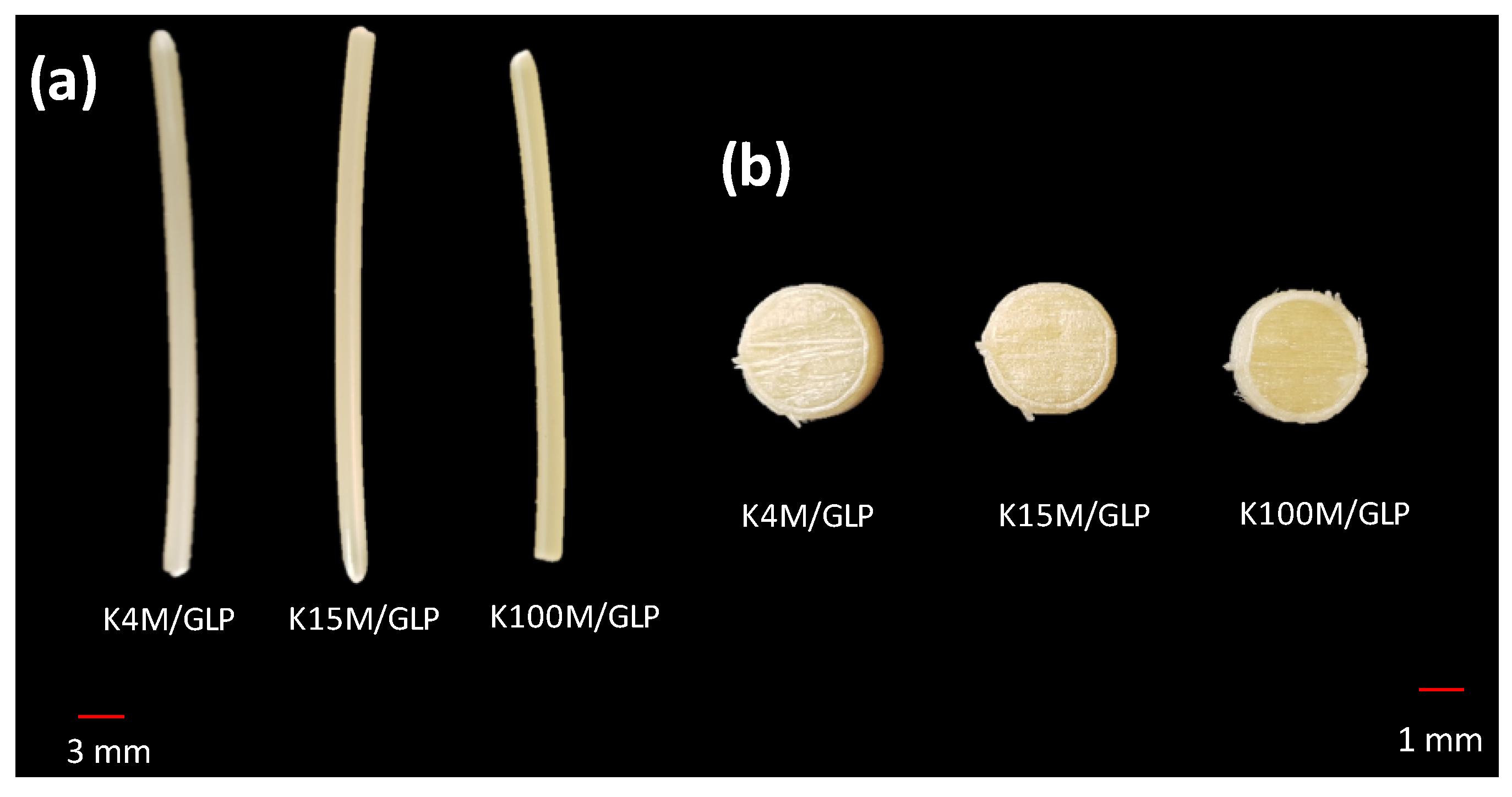
2.5.1. Geometrical, Porosity and Morphological Assessment of Matrices
2.5.2. Determination of Tablet Strength
2.5.3. Determination of Tablet Friability
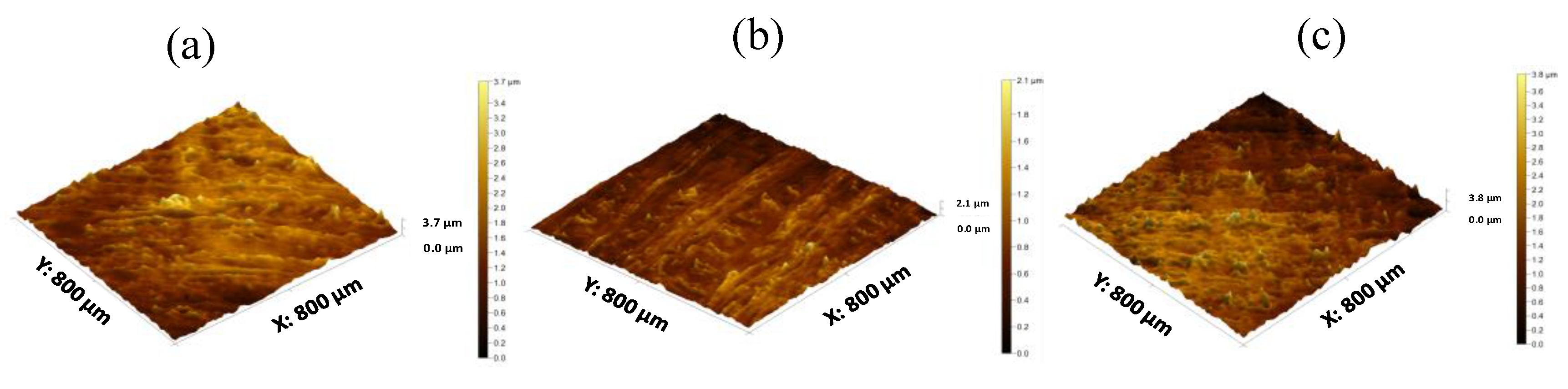
2.5.4. 3D Nanoscale Surface Texture Analysis
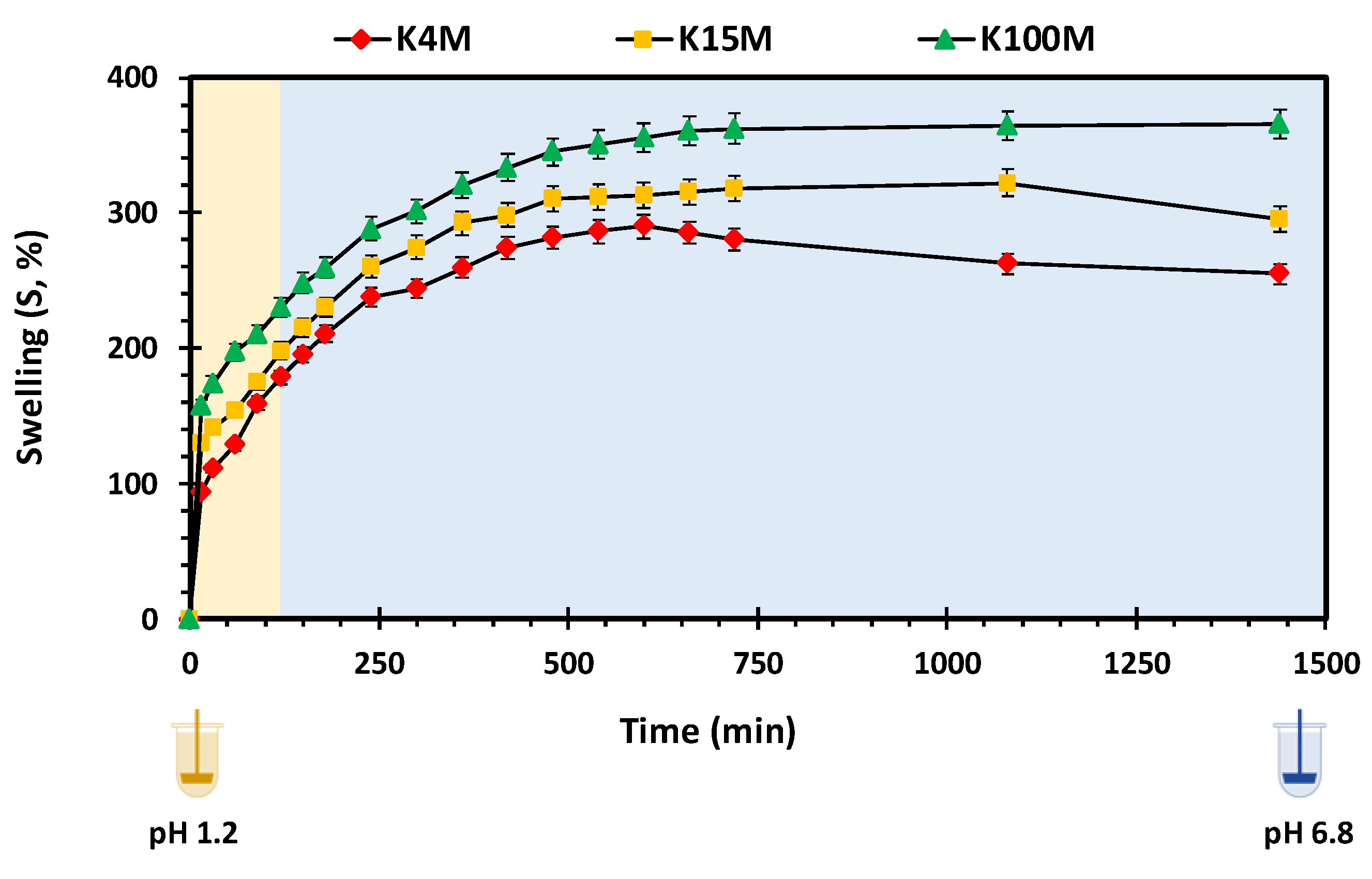
2.5.5. Swelling Studies
2.5.6. In Vitro Glipizide Dissolution Studies
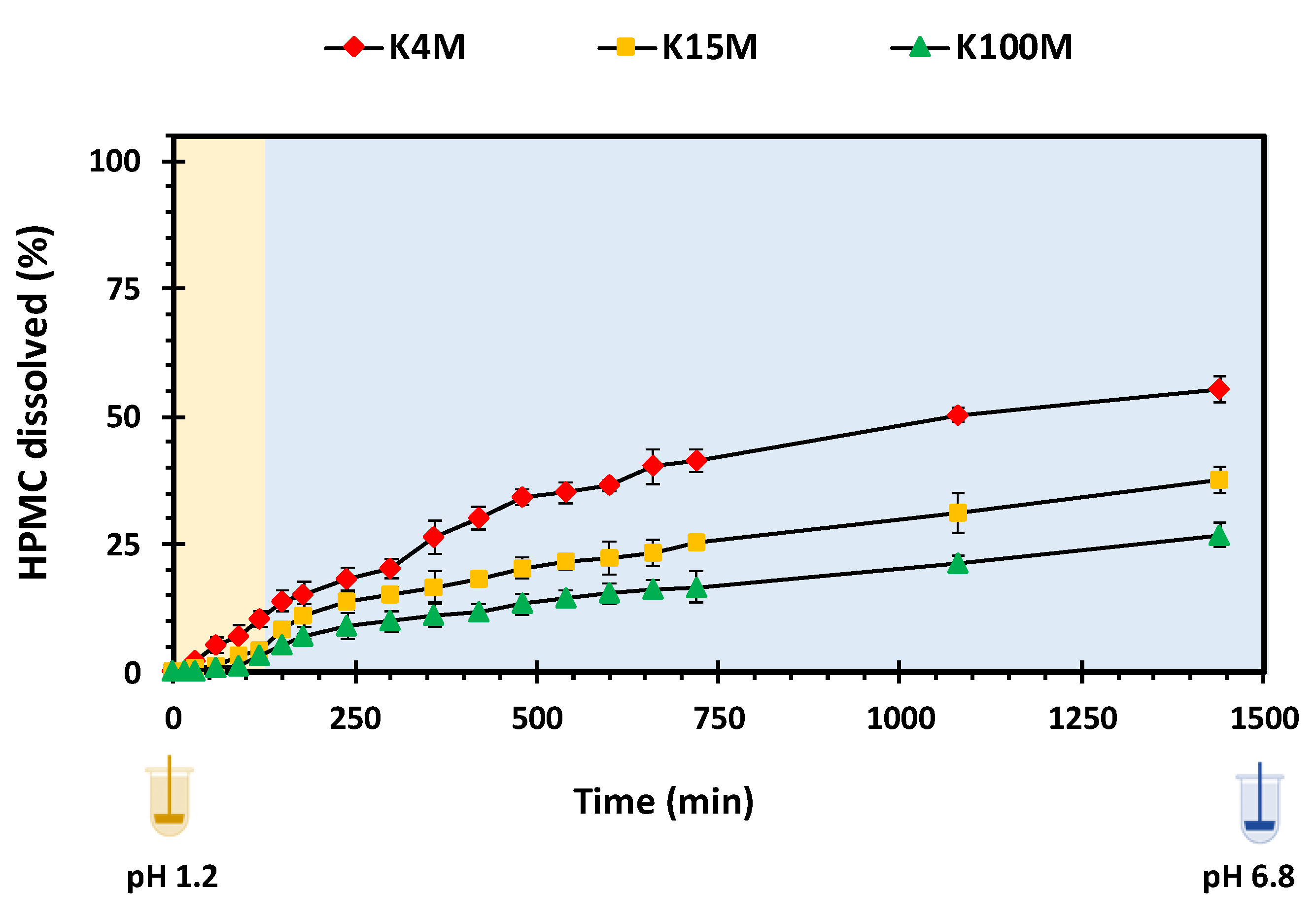
2.5.7. HPMC Dissolution and Overall Erosion Studies
2.6. Pharmacokinetic Studies
2.6.1. Animal Housing and Handling
2.6.2. In vivo Experiments
2.6.3. Quantification of Glipizide in Plasma
2.6.4. Determination of Pharmacokinetic Parameters
3. Results and Discussion
3.1. Development and Characterisation of Filaments
3.2. Development and Characterisation of 3D Printed Matrix Tablets
3.3. Swelling, Erosion, HPMC Dissolution and Drug Release Studies
3.3.1. Swelling and HPMC Dissolution Studies
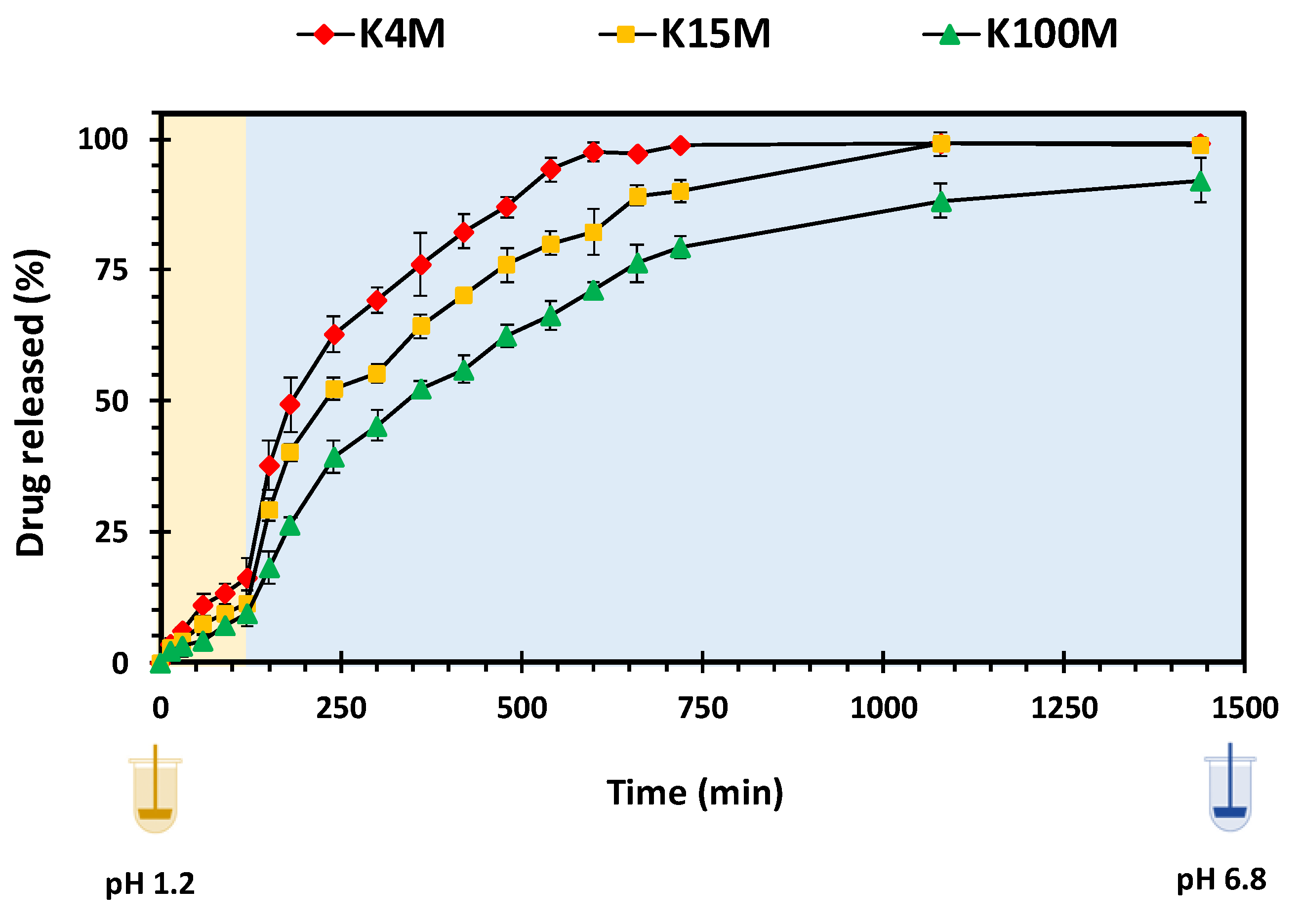
3.3.2. Drug Release Studies
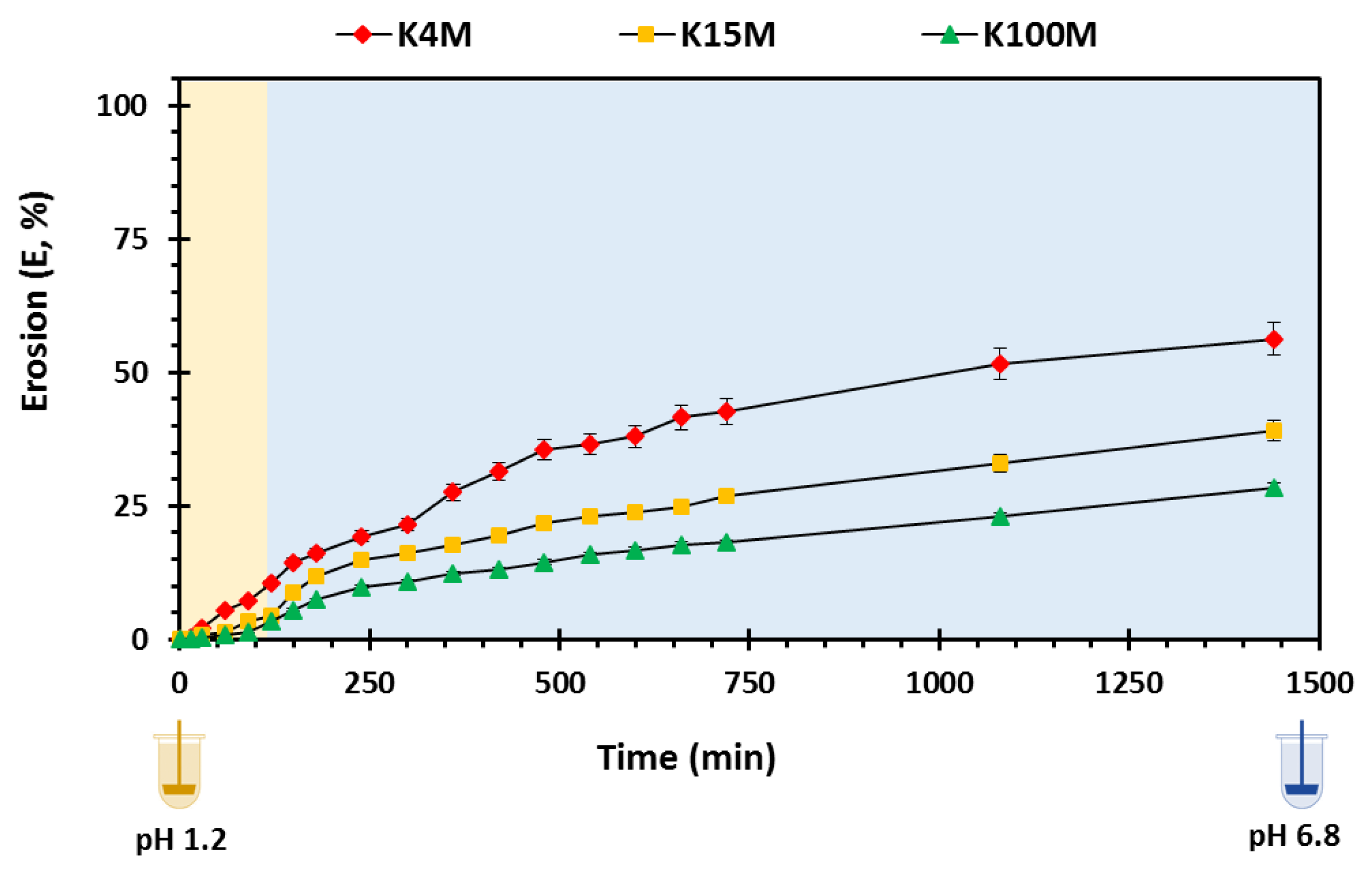
3.3.3. Erosion Studies
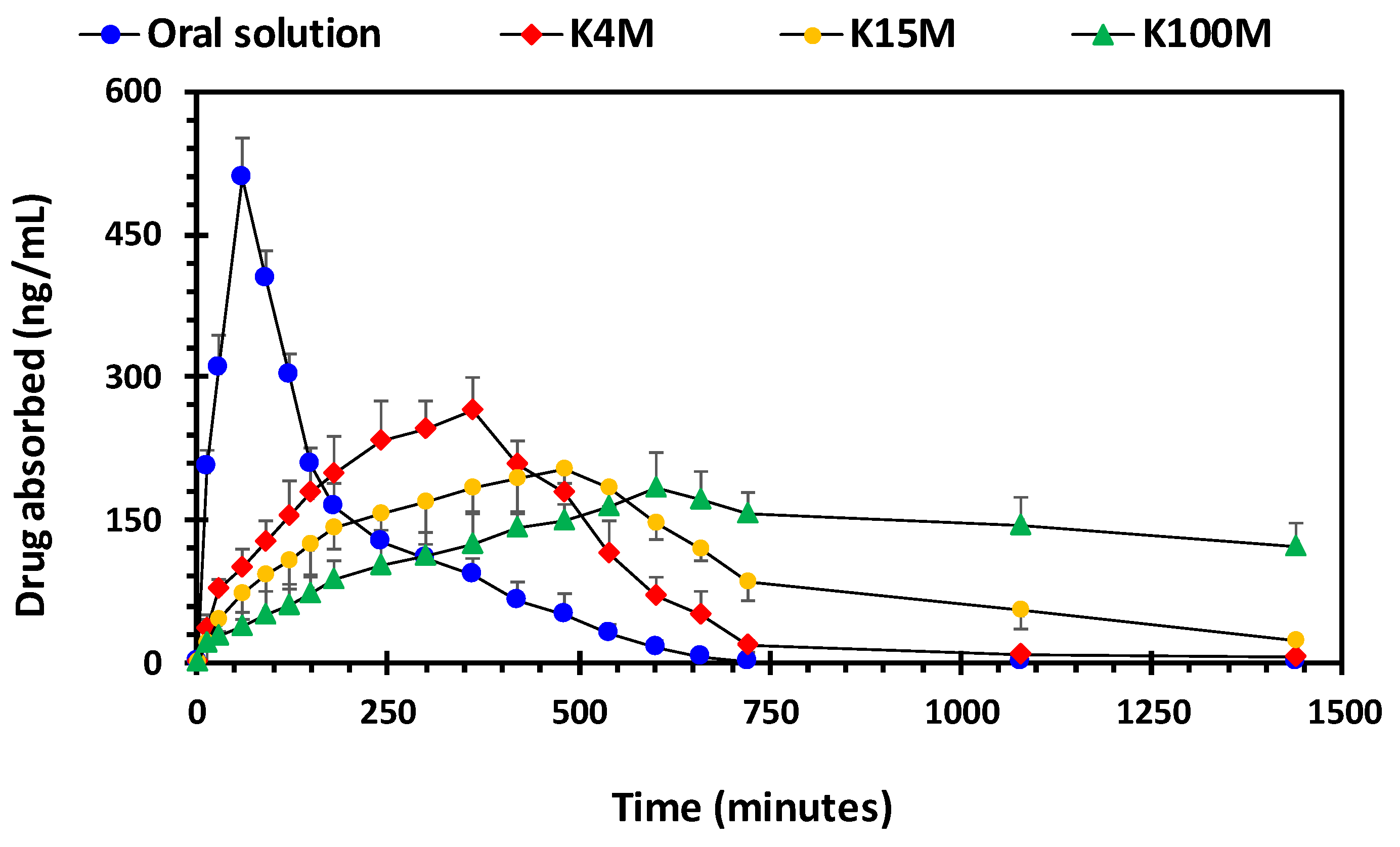
3.4. Pharmacokinetic Parameters of HPMC/Glipizide Hydrophilic Matrices
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Tutton, R. Personalizing medicine: Futures present and past. Soc. Sci. Med. 2012, 75, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Kadry, H.; Al-Hilal, T.A.; Keshavarz, A.; Alam, F.; Xu, C.; Joy, A.; Ahsan, F. Multi-purposable filaments of HPMC for 3D printing of medications with tailored drug release and timed-absorption. Int. J. Pharm. 2018, 544, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Sandler, N.; Määttänen, A.; Ihalainen, P.; Kronberg, L.; Meierjohann, A.; Viitala, T.; Peltonen, J. Inkjet printing of drug substances and use of porous substrates-towards individualized dosing. J. Pharm. Sci. 2011, 100, 3386–3395. [Google Scholar] [CrossRef] [PubMed]
- Beebe, K.; Kennedy, A.D. Sharpening precision medicine by a thorough interrogation of metabolic individuality. Comput. Struct. Biotechnol. J. 2016, 14, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Vélez, G.F.; Wu, B.M. 3D pharming: Direct printing of personalized pharmaceutical tablets. Polym. Sci. 2016, 2, 11. [Google Scholar]
- Alhnan, M.A.; Okwuosa, T.C.; Sadia, M.; Wan, K.W.; Ahmed, W.; Arafat, B. Emergence of 3D printed dosage forms: Opportunities and challenges. Pharm. Res. 2016, 33, 1817–1832. [Google Scholar] [CrossRef] [PubMed]
- Trenfield, S.J.; Awad, A.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D printing pharmaceuticals: Drug development to frontline care. Trends Pharm. Sci. 2018, 39, 440–451. [Google Scholar] [CrossRef]
- Edgar, J.; Tint, S. Additive manufacturing technologies: 3D printing, rapid prototyping, and direct digital manufacturing. Johns. Matthey Technol. Rev. 2015, 59, 193–198. [Google Scholar] [CrossRef]
- Goyanes, A.; Buanz, A.B.; Basit, A.W.; Gaisford, S. Fused-filament 3D printing (3DP) for fabrication of tablets. Int. J. Pharm. 2014, 476, 88–92. [Google Scholar] [CrossRef]
- Giannatsis, J.; Dedoussis, V. Additive fabrication technologies applied to medicine and health care: A review. Int. J. Adv. Manuf. Technol. 2009, 40, 116–127. [Google Scholar] [CrossRef]
- Khaled, S.A.; Burley, J.C.; Alexander, M.R.; Roberts, C.J. Desktop 3D printing of controlled release pharmaceutical bilayer tablets. Int. J. Pharm. 2014, 461, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Moulton, S.E.; Wallace, G.G. 3-dimensional (3D) fabricated polymer based drug delivery systems. J. Control. Release 2014, 193, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Goyanes, A.; Buanz, A.B.; Hatton, G.B.; Gaisford, S.; Basit, A.W. 3D printing of modified-release aminosalicylate (4-ASA and 5-ASA) tablets. Eur. J. Pharm. Biopharm. 2015, 89, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Soh, S. Printing Tablets with Fully Customizable Release Profiles for Personalized Medicine. Adv. Mater. 2015, 27, 7847–7853. [Google Scholar] [CrossRef] [PubMed]
- Goyanes, A.; Det-Amornrat, U.; Wang, J.; Basit, A.W.; Gaisford, S. 3D scanning and 3D printing as innovative technologies for fabricating personalized topical drug delivery systems. J. Control. Release 2016, 234, 41–48. [Google Scholar] [CrossRef]
- Beck, R.C.R.; Chaves, P.S.; Goyanes, A.; Vukosavljevic, B.; Buanz, A.; Windbergs, M.; Basit, A.W.; Gaisford, S. 3D printed tablets loaded with polymeric nanocapsules: An innovative approach to produce customized drug delivery systems. Int. J. Pharm. 2017, 528, 268–279. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, J.; Li, M.; Sun, K.; Liu, C.J. Three-dimensional printed acrylonitrile butadiene styrene framework coated with Cu-BTC metal-organic frameworks for the removal of methylene blue. Sci. Rep. 2014, 4, 5939. [Google Scholar] [CrossRef]
- Holländer, J.; Genina, N.; Jukarainen, H.; Khajeheian, M.; Rosling, A.; Mäkilä, E.; Sandler, N. Three-dimensional printed pcl-based implantable prototypes of medical devices for controlled drug delivery. J. Pharm. Sci. 2016, 105, 2665–2676. [Google Scholar] [CrossRef]
- Kempin, W.; Franz, C.; Koster, L.C.; Schneider, F.; Bogdahn, M.; Weitschies, W.; Seidlitz, A. Assessment of different polymers and drug loads for fused deposition modeling of drug loaded implants. Eur. J. Pharm. Biopharm. 2017, 115, 84–93. [Google Scholar] [CrossRef]
- Pietrzak, K.; Isreb, A.; Alhnan, M.A. A flexible-dose dispenser for immediate and extended release 3D printed tablets. Eur. J. Pharm. Biopharm. 2015, 96, 380–387. [Google Scholar] [CrossRef]
- Genina, N.; Holländer, J.; Jukarainen, H.; Mäkilä, E.; Salonen, J.; Sandler, N. Ethylene vinyl acetate (EVA) as a new drug carrier for 3D printed medical drug delivery devices. Eur. J. Pharm. 2016, 90, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Chai, X.; Chai, H.; Wang, X.; Yang, J.; Li, J.; Zhao, Y.; Weimin, C.; Tao, T.; Xiang, X. Fused deposition modeling (FDM) 3D printed tablets for intragastric floating delivery of domperidone. Sci. Rep. 2017, 7, 2829. [Google Scholar] [CrossRef] [PubMed]
- Goyanes, A.; Fina, F.; Martorana, A.; Sedough, D.; Gaisford, S.; Basit, A.W. Development of modified release 3D printed tablets (printlets) with pharmaceutical excipients using additive manufacturing. Int. J. Pharm. 2017, 527, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Feng, X.; Patil, H.; Tiwari, R.V.; Repka, M.A. Coupling 3D printing with hot-melt extrusion to produce controlled-release tablets. Int. J. Pharm. 2017, 519, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Khizer, Z.; Conway, B.R.; Ghori, M.U. Surface texture analysis of plasticiser free 3D printed matrices: Before and after dissolution. Br. J. Pharm. 2019. in-press. [Google Scholar]
- Bruce, L.D.; Shah, N.H.; Malick, A.W.; Infeld, M.H.; McGinity, J.W. Properties of hot-melt extruded tablet formulations for the colonic delivery of 5-aminosalicylic acid. Eur. J. Pharm. Biopharm. 2005, 59, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Basit, A.W.; Newton, J.M.; Short, M.D.; Waddington, W.A.; Ell, P.J.; Lacey, L.F. The effect of polyethylene glycol 400 on gastrointestinal transit: Implications for the formulation of poorly-water soluble drugs. Pharm. Res. 2001, 18, 1146–1150. [Google Scholar] [CrossRef] [PubMed]
- Oosaka, K.; Tokuda, M.; Furukawa, N. Intra-gastric triacetin alters upper gastrointestinal motility in conscious dogs. World J. Gastroenterol. 2014, 20, 1054. [Google Scholar] [CrossRef][Green Version]
- Johnson, J.W. Final report on the safety assessment of acetyl triethyl citrate, acetyl tributyl citrate, acetyl trihexyl citrate, and acetyl trioctyl citrate. Int. J. Toxicol. 2002, 21, 1–17. [Google Scholar]
- Davis, S.S.; Hardy, J.G.; Fara, J.W. Transit of pharmaceutical dosage forms through the small intestine. Gut 1986, 27, 886–892. [Google Scholar] [CrossRef]
- Gordeev, E.G.; Galushko, A.S.; Ananikov, V.P. Improvement of quality of 3D printed objects by elimination of microscopic structural defects in fused deposition modeling. PLoS ONE 2018, 13, e0198370. [Google Scholar] [CrossRef] [PubMed]
- Asim, M.S.; Ghori, M.U.; Conway, B.R. Probing the impact of porosity on swelling kinetics of hydrophilic matrices. Br. J. Pharm. 2017, 2. [Google Scholar] [CrossRef]
- Sankalia, J.M.; Sankalia, M.G.; Mashru, R.C. Drug release and swelling kinetics of directly compressed glipizide sustained-release matrices: Establishment of level A IVIVC. J. Control. Release 2008, 129, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Ghori, M.; Šupuk, E.; Conway, B. Tribo-electrification and powder adhesion studies in the development of polymeric hydrophilic drug matrices. Materials 2015, 8, 1482–1498. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, Y.; Saeed, S.; Ghori, M.U.; Mahmood, T.; Yousaf, A.M.; Jamshaid, M.; Rizvi, S.A. Influence of polymer ratio and surfactants on controlled drug release from cellulosic microsponges. Int. J. Biol. Macromol. 2018, 109, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Diryak, R.; Kontogiorgos, V.; Ghori, M.U.; Bills, P.; Tawfik, A.; Morris, G.A.; Smith, A.M. Behavior of in situ cross-linked hydrogels with rapid gelation kinetics on contact with physiological fluids. Macromol. Chem. Phys. 2018, 219, 1700584. [Google Scholar] [CrossRef]
- Ghori, M.U.; Alba, K.; Smith, A.M.; Conway, B.R.; Kontogiorgos, V. Okra extracts in pharmaceutical and food applications. Food Hydrocoll. 2014, 42, 342–347. [Google Scholar] [CrossRef]
- Ghori, M.U.; Mohammad, M.A.; Rudrangi, S.R.S.; Fleming, L.T.; Merchant, H.A.; Smith, A.M.; Conway, B.R. Impact of purification on physicochemical, surface and functional properties of okra biopolymer. Food Hydrocoll. 2017, 71, 311–320. [Google Scholar] [CrossRef]
- Nep, E.I.; Asare-Addo, K.; Ghori, M.U.; Conway, B.R.; Smith, A.M. Starch-free grewia gum matrices: Compaction, swelling, erosion and drug release behaviour. Int. J. Pharm. 2015, 496, 689–698. [Google Scholar] [CrossRef]
- Ghori, M.U. Release Kinetics, Compaction and Electrostatic Properties of Hydrophilic Matrices. Ph.D. Thesis, Repository University of Huddersfield, Huddersfield, UK, December 2014; p. 37. [Google Scholar]
- Ghori, M.U.; Grover, L.M.; Asare-Addo, K.; Smith, A.M.; Conway, B.R. Evaluating the swelling, erosion, and compaction properties of cellulose ethers. Pharm. Dev. Technol. 2018, 23, 183–197. [Google Scholar] [CrossRef]
- Ghori, M.U.; Ginting, G.; Smith, A.M.; Conway, B.R. Simultaneous quantification of drug release and erosion from HPMC hydrophilic matrices. Int. J. Pharm. 2014, 465, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Nirwan, J.S.; Conway, B.R.; Ghori, M.U. In situ 3D nanoscale advanced imaging algorithms with integrated chemical imaging for the characterisation of pharmaceuticals. RSC Adv. 2019, 9, 16119–16129. [Google Scholar] [CrossRef]
- Defang, O.; Shufang, N.; Wei, L.; Hong, G.; Hui, L.; Weisan, P. In vitro and in vivo evaluation of two extended release preparations of combination metformin and glipizide. Drug. Dev. Ind. Pharm. 2005, 31, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huo, M.; Zhou, J.; Xie, S. PKSolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Comput. Methods Programs Biomed. 2010, 99, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wen, H.; Jia, D.; Guan, X.; Pan, H.; Yang, Y.; Pan, W. Preparation and investigation of controlled-release glipizide novel oral device with three-dimensional printing. Int. J. Pharm. 2017, 525, 5–11. [Google Scholar] [CrossRef]
- Korte, C.; Quodbach, J. Formulation development and process analysis of drug-loaded filaments manufactured via hot-melt extrusion for 3D-printing of medicines. Pharm. Dev. Technol. 2018, 23, 1117–1127. [Google Scholar] [CrossRef] [PubMed]
- Öblom, H.; Zhang, J.; Pimparade, M.; Speer, I.; Preis, M.; Repka, M.; Sandler, N. 3D-printed isoniazid tablets for the treatment and prevention of tuberculosis- personalized dosing and drug release. AAPS Pharmscitech. 2019, 20, 52. [Google Scholar] [CrossRef]
- Bocqué, M.; Voirin, C.; Lapinte, V.; Caillol, S.; Robin, J.J. Petro-based and bio-based plasticizers: Chemical structures to plasticizing properties. Polym. Sci. A Polym. Chem. 2016, 54, 11–33. [Google Scholar] [CrossRef]
- British Pharmacopoeia (BP). Available online: https://www.pharmacopoeia.com/ (accessed on 27 June 2019).
- Ghori, M.U.; Conway, B.R. Hydrophilic matrices for oral control drug delivery. Am. J. Pharm. Sci. 2015, 3, 103–109. [Google Scholar]
- Vergnaud, J.M. Liquid transport controlled release processes in polymeric materials: Applications to oral dosage forms. Int. J. Pharm. 1993, 90, 89–94. [Google Scholar] [CrossRef]
- Ammar, H.O.; Salama, H.A.; Ghorab, M.; El-Nahhas, S.A.; Elmotasem, H. A transdermal delivery system for glipizide. Curr. Drug. Deliv. 2006, 3, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Jamzad, S.; Fassihi, R. Role of surfactant and pH on dissolution properties of fenofibrate and glipizide—A technical note. AAPS Pharmscitech. 2006, 7, E17–E22. [Google Scholar] [CrossRef] [PubMed]
- Khanvilkar, K.H.; Huang, Y.; Moore, A.D. Influence of hydroxypropyl methylcellulose mixture, apparent viscosity, and tablet hardness on drug release using a 23 full factorial design. Drug. Dev. Ind. Pharm. 2002, 28, 601–608. [Google Scholar] [CrossRef] [PubMed]

| Methocel® | Viscosity (cps) a | Average Molar Mass g/mol a |
|---|---|---|
| K4M | 4351 | ~88000 |
| K15M | 17129 | ~125000 |
| K100M | 79279 | ~215000 |
| Printing Temperature °C | Number of Shells | Layer Height (mm) | Raft | Support | Infill Density (%) | Speed While Extrusion (mm/sec) | Speed While Traveling (mm/sec) | Infill Pattern |
|---|---|---|---|---|---|---|---|---|
| 170 | 2 | 0.1 | No | No | 100 | 90 | 150 | Linear |
| Characteristics | K4M | K15M | K100M |
|---|---|---|---|
| Drug loading efficiency (%) | 98.7 (2.2) | 101.2 (1.1) | 99.3 (1.6) |
| Force (N) | 1.9 (0.2) | 2.2(0.6) | 2.3 (0.3) |
| Distance (mm) | 4.9 (1.2) | 5.1 (1.1) | 5.2 (1.4) |
| Stress (MPa) | 12.3 (1.1) | 14.2 (1.2) | 16.9 (1.6) |
| Strain | 0.8 (0.2) | 0.9 (0.2) | 0.80 (0.1) |
| Young modulus, E (MPa) | 14.7 (1.2) | 17.1 (1.4) | 21.1 (1.6) |
| Characteristics | K4M | K15M | K100M |
|---|---|---|---|
| Weight (mg) | 100.1 (2.2) | 99.6 (1.8) | 99.3 (2.1) |
| Diameter | 3.0 (0.1) | 3.0 (0.1) | 3.0 (0.1) |
| Thickness (mm) | 1.5 (0.1) | 1.5 (0.1) | 1.5 (0.1) |
| Drug loading in tablets (%) | 99.3 (2.6) | 100.3 (2.9) | 98.6 (1.4) |
| Porosity (%) | 2.2 (0.2) | 1.5 (0.2) | 0.8 (0.1) |
| Breaking strength of tablets (N) | 345.6 (10.3) | 480.2 (14.6) | 525.3 (9.3) |
| Friability (%) | 0 | 0 | 0 |
| Parameter | K4M | K15M | K100M |
|---|---|---|---|
| Sa (μm) | 22.6 (2.3) | 14.6 (1.9) | 16.4 (3.3) |
| Sq (μm) | 26.5 (9.4) | 20.3 (3.5) | 23.7 (4.8) |
| Sz (μm) | 75.0 (7.2) | 60.0 (9.1) | 70.0 (10.9) |
| Sp (μm) | 33.7 (6.8) | 30.7 (4.9) | 36.3 (4.0) |
| Sv (μm) | 41.3 (9.0) | 29.4 (5.3) | 33.7 (8.4) |
| Sku (μm) | 5.3 (1.1) | 4.9 (0.9) | 5.1 (1.4) |
| Ssk (μm) | −0.6 (−0.1) | −0.4 (−0.2) | −0.7 (−0.2) |
| Sds (1/μm2) | 112.4 (11.4) | 98.3 (5.6) | 104.1 (9.7) |
| Sal (μm) | 136.4 (20.4) | 111.0 (11.0) | 121.4 (9.0) |
| Str | 0.9 (0.1) | 0.9 (0.1) | 0.8 (0.1) |
| Vm (μm3/μm2) | 0.4 (0.1) | 0.3 (0.09) | 0.4 (0.1) |
| Vv (μm3/μm2) | 6.9 (2.5) | 5.2 (2.89) | 6.4 (3.2) |
| Vvv (μm3/μm2) | 0.6 (0.1) | 0.4 (0.1) | 0.3 (0.1) |
| Type of Matrix Tablet | Swelling Kinetic Parameters | Erosion Kinetic Parameters | |||
|---|---|---|---|---|---|
| KW a | n | R2 | KE b | R2 | |
| K4M | 32.96 | 0.34 | 0.99 | 0.076 | 0.98 |
| K15M | 52.20 | 0.28 | 0.97 | 0.057 | 0.96 |
| K100M | 76.51 | 0.24 | 0.98 | 0.039 | 0.97 |
| Parameters | K4M | K15M | K100M | Oral Solution |
|---|---|---|---|---|
| T max (h) | 6 (0.00) | 8(0.00) | 10 (0.00) | 1 (0.00) |
| C max (ng/mL) | 264.88 (33.69) | 202.85 (15.33) | 182.66 (38.37) | 509.17 (42.19) |
| AUCo-t (ng/mL.h) | 1959.46 (151.32) | 2325..08 (269.40) | 3082.36 (252.32) | 1499.48 (164.65) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khizer, Z.; Akram, M.R.; Sarfraz, R.M.; Nirwan, J.S.; Farhaj, S.; Yousaf, M.; Hussain, T.; Lou, S.; Timmins, P.; Conway, B.R.; et al. Plasticiser-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release and Pharmacokinetic Studies. Polymers 2019, 11, 1095. https://doi.org/10.3390/polym11071095
Khizer Z, Akram MR, Sarfraz RM, Nirwan JS, Farhaj S, Yousaf M, Hussain T, Lou S, Timmins P, Conway BR, et al. Plasticiser-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release and Pharmacokinetic Studies. Polymers. 2019; 11(7):1095. https://doi.org/10.3390/polym11071095
Chicago/Turabian StyleKhizer, Zara, Muhammad R. Akram, Rai M. Sarfraz, Jorabar Singh Nirwan, Samia Farhaj, Maria Yousaf, Tariq Hussain, Shan Lou, Peter Timmins, Barbara R. Conway, and et al. 2019. "Plasticiser-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release and Pharmacokinetic Studies" Polymers 11, no. 7: 1095. https://doi.org/10.3390/polym11071095
APA StyleKhizer, Z., Akram, M. R., Sarfraz, R. M., Nirwan, J. S., Farhaj, S., Yousaf, M., Hussain, T., Lou, S., Timmins, P., Conway, B. R., & Ghori, M. U. (2019). Plasticiser-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release and Pharmacokinetic Studies. Polymers, 11(7), 1095. https://doi.org/10.3390/polym11071095