Synthesis and Characterization of Dental Nanocomposite Resins Filled with Different Clay Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Measurements
2.4. Statistical Analysis
3. Results and Discussion
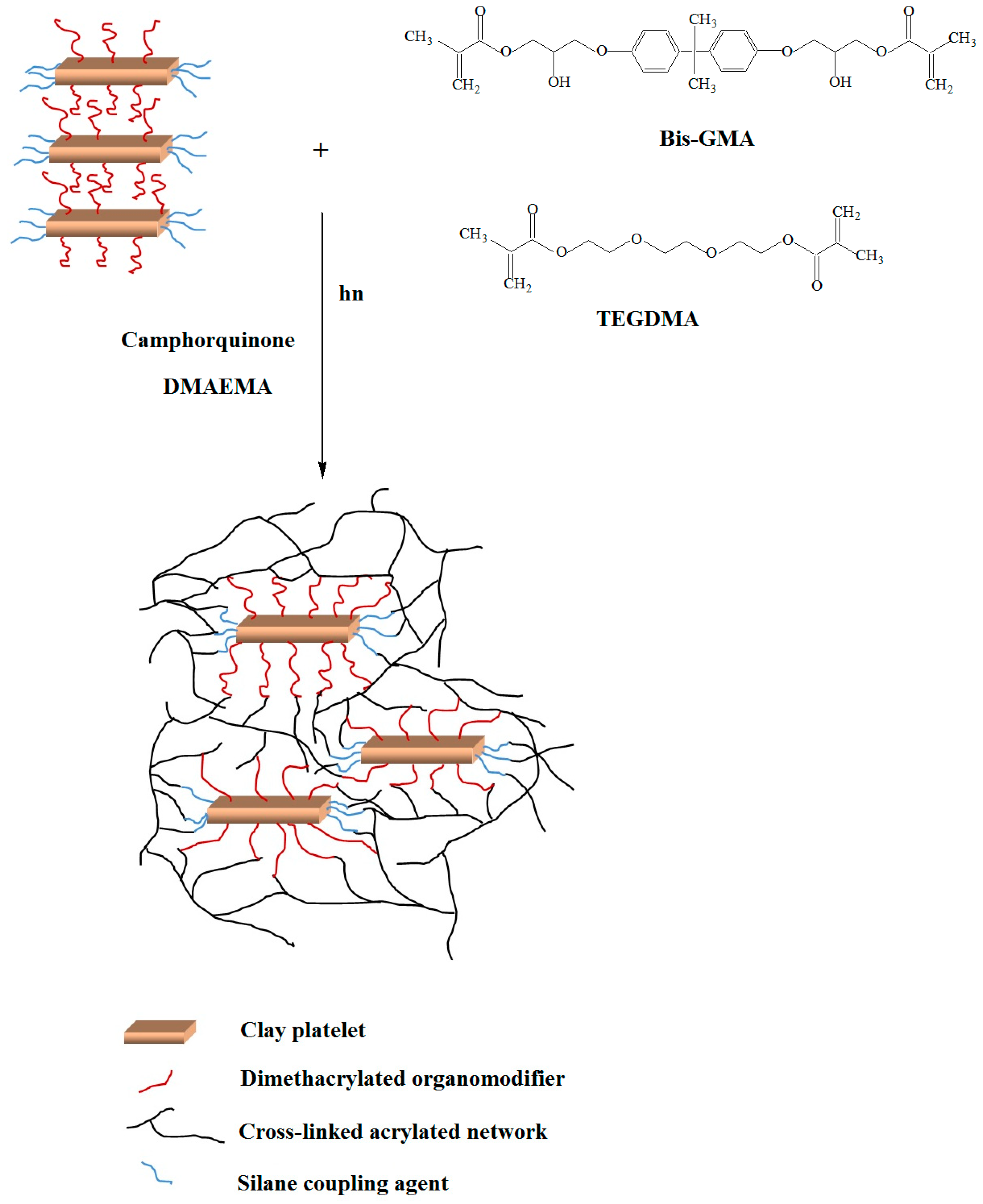
3.1. Structure and Morphology Characterization
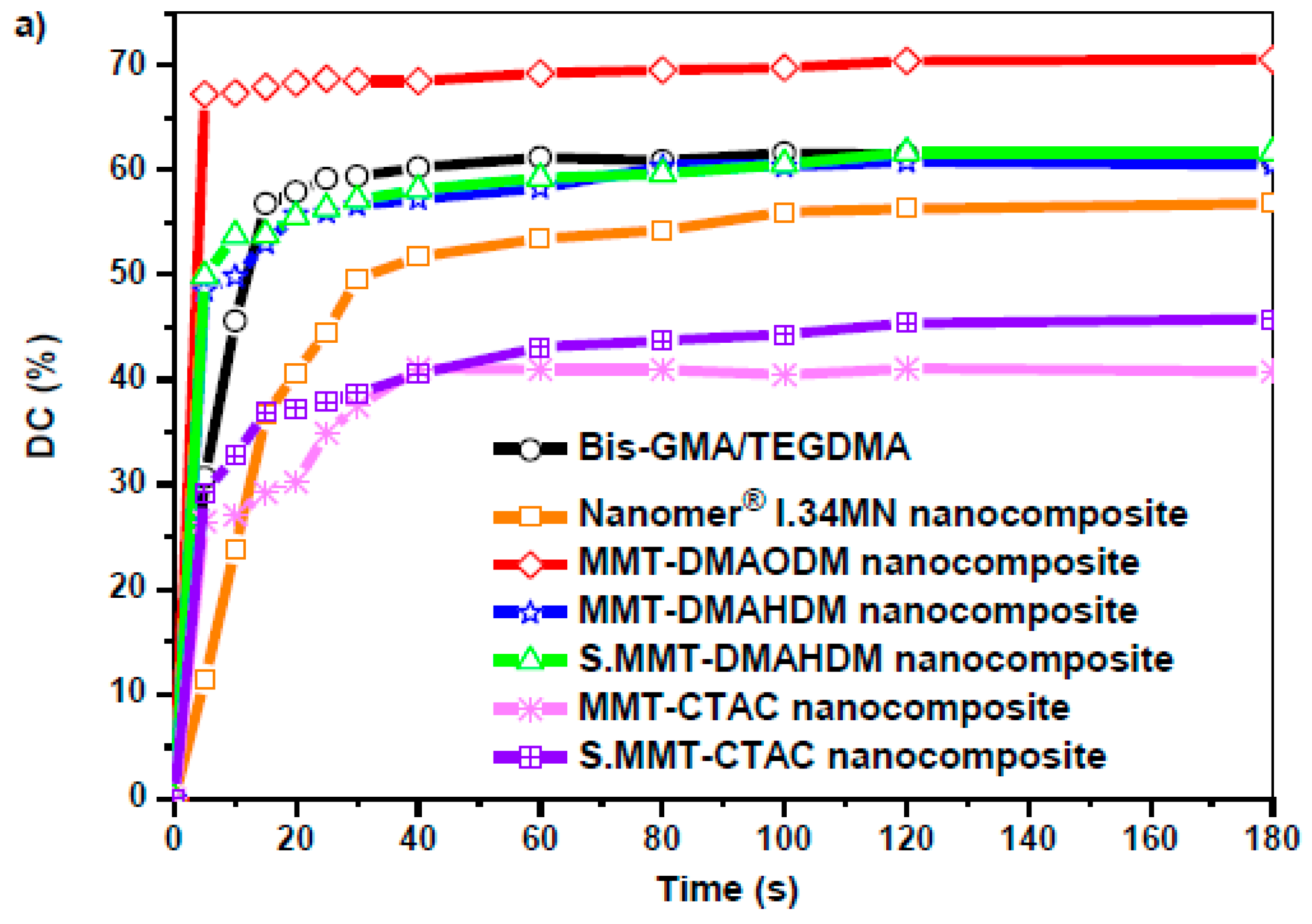
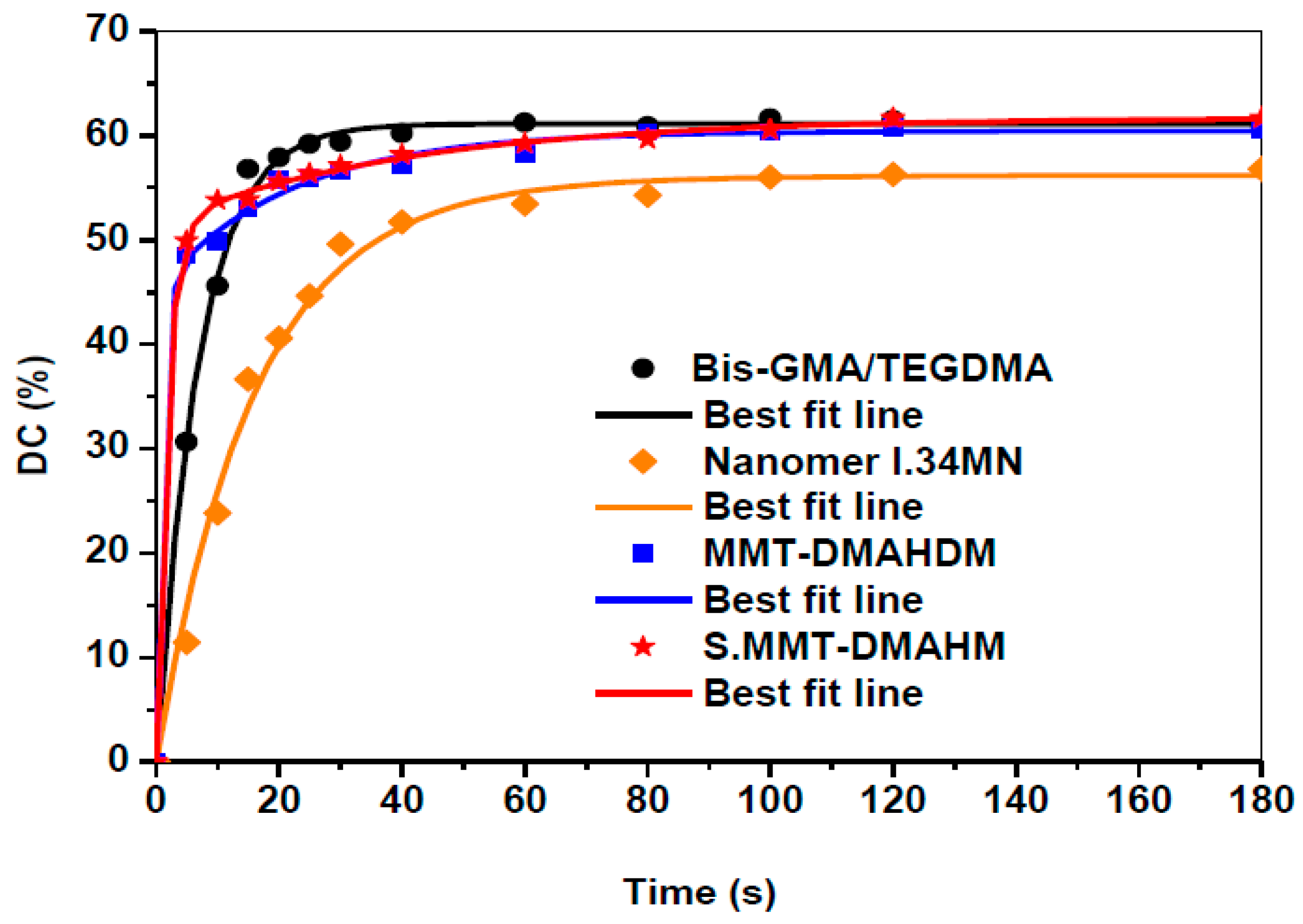
3.2. Polymerization Kinetics
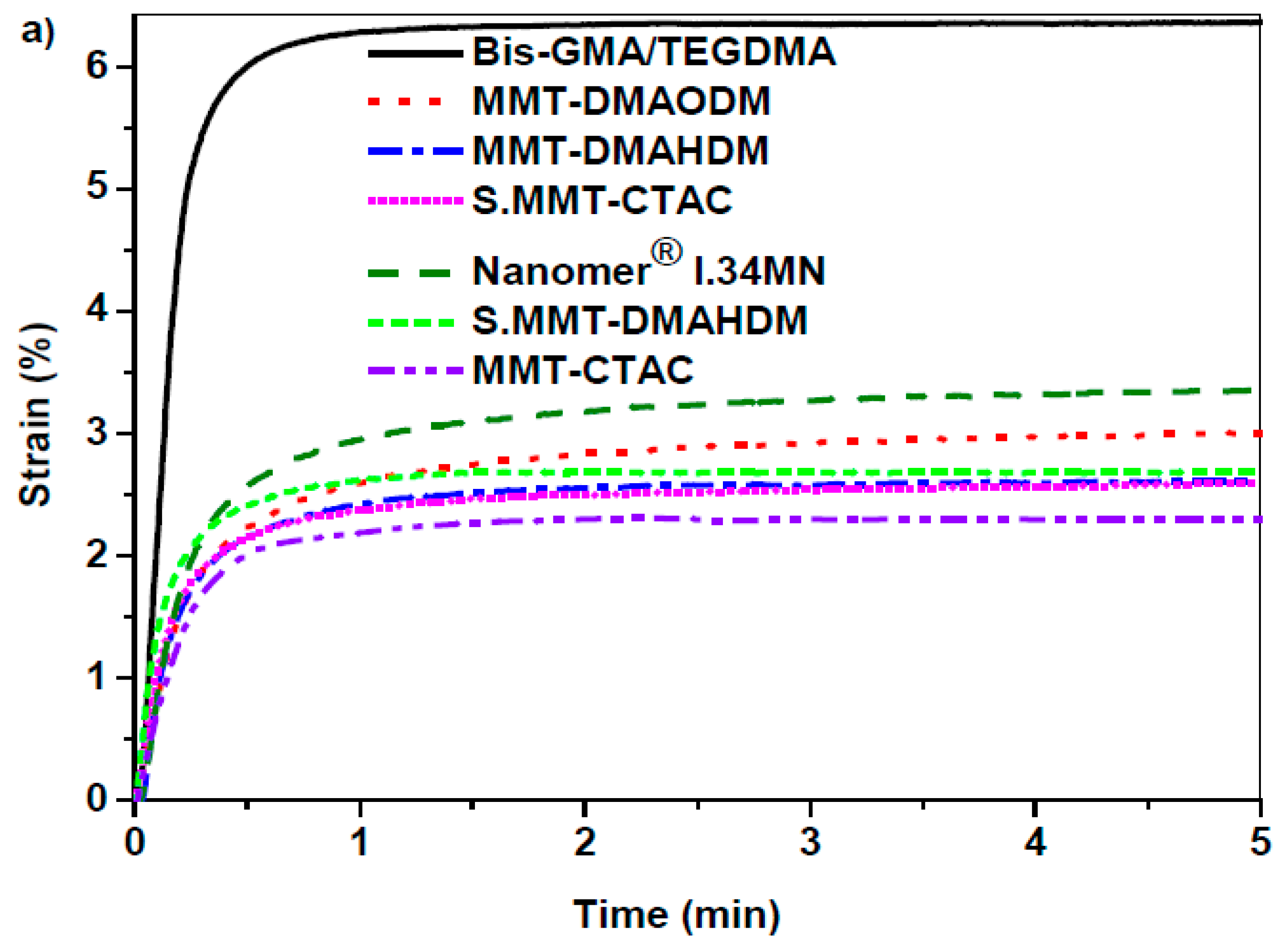
3.3. Polymerization Shrinkage Kinetics
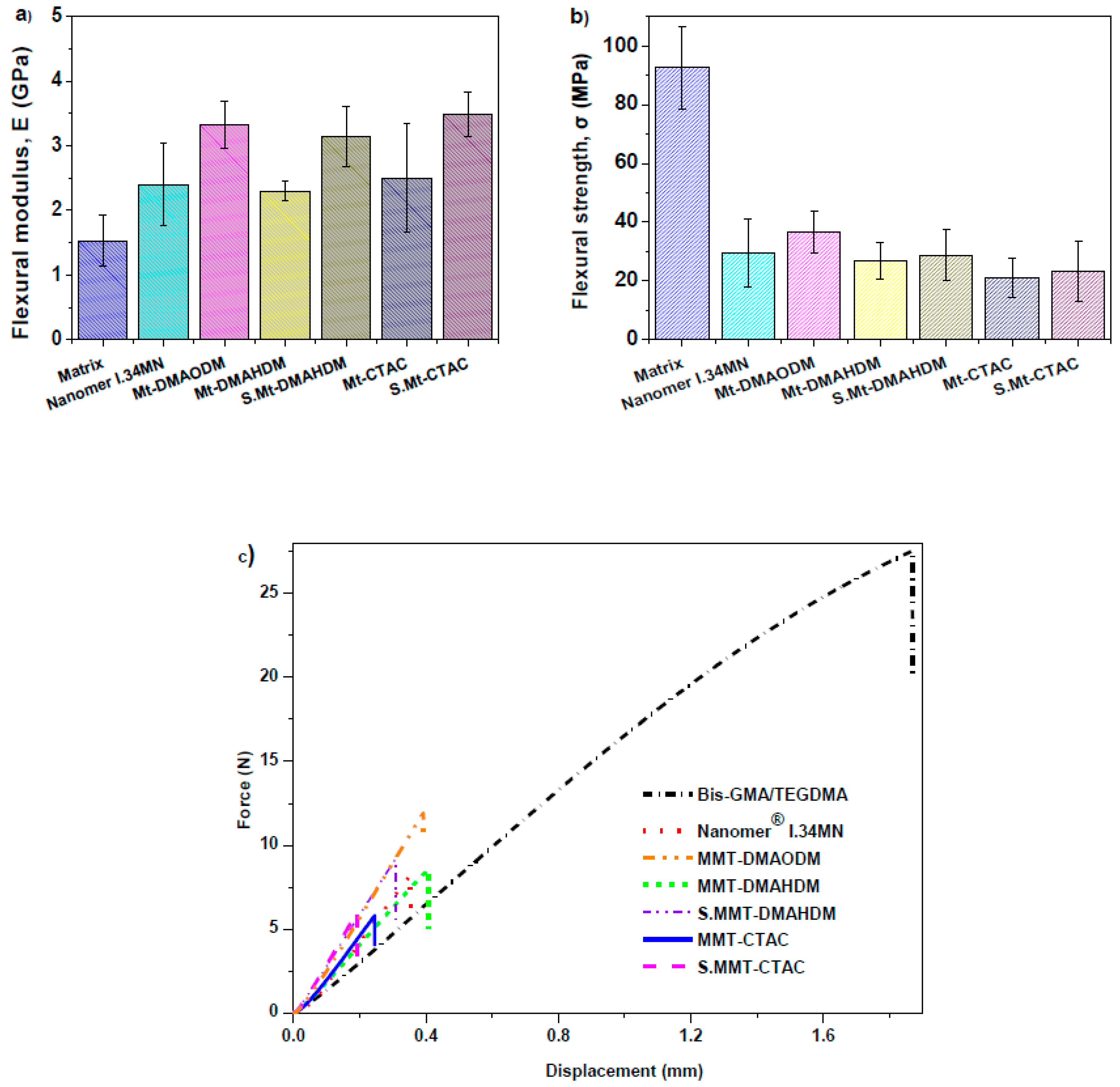
3.4. Mechanical Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bowen, R.L. Composite and sealant resins--past, present, and future. Pediatr. Dent. 1982, 4, 10–15. [Google Scholar]
- McCabe, J.; Walls, A. Applied Dental Materials, 9th ed.; Blackwell Publishing Ltd.: Oxford, UK, 2008; pp. 188–189, 206–207. [Google Scholar]
- Mai, Y.W.; Yu, Z.Z. Polymer Nanocomposites, 1st ed.; Woodhead Publishing Ltd.: Cambridge, UK, 2006; pp. 188–190, 206, 307. [Google Scholar]
- Chen, M.-H. Update on Dental Nanocomposites. J. Dent. 2010, 89, 549–560. [Google Scholar] [CrossRef]
- Van Noort, R. Introduction to Dental Materials, 4th ed.; Mosby Elsevier Ltd.: Edinburgh, UK, 2013; pp. 73–80. [Google Scholar]
- Cramer, N.; Stansbury, J.; Bowman, C. Recent advances and developments in composite dental restorative materials. J. Dent. Res. 2011, 90, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Ilie, N.; Hickel, R. Resin composite restorative materials. Aust. Dent. J. 2011, 56, 59–66. [Google Scholar] [CrossRef]
- Saharudin, K.A.; Sreekantan, S.; Basiron, N.; Khor, Y.L.; Harun, N.H.; Mydin, R.B.S.M.N.; Akil, H.M.; Seeni, A.; Vignesh, K.; Mydin, R.S.M.N. Bacteriostatic Activity of LLDPE Nanocomposite Embedded with Sol–Gel Synthesized TiO2/ZnO Coupled Oxides at Various Ratios. Polymers 2018, 10, 878. [Google Scholar] [CrossRef]
- Wang, H.-T.; Chiang, P.-C.; Tzeng, J.-J.; Wu, T.-L.; Pan, Y.-H.; Chang, W.-J.; Huang, H.-M.; Choi, I.S.; Mano, J.F. In Vitro Biocompatibility, Radiopacity, and Physical Property Tests of Nano-Fe3O4 Incorporated Poly-l-lactide Bone Screws. Polymers 2017, 9, 191. [Google Scholar] [CrossRef]
- Zagho, M.M.; Hussein, E.A.; Elzatahry, A.A. Recent Overviews in Functional Polymer Composites for Biomedical Applications. Polymers 2018, 10, 739. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wu, X.; Sun, Y.; Xie, W. POSS Dental Nanocomposite Resin: Synthesis, Shrinkage, Double Bond Conversion, Hardness, and Resistance Properties. Polymers 2018, 10, 369. [Google Scholar] [CrossRef] [PubMed]
- Gilman, J.W.; Morgan, A.B.; Harris, R., Jr.; Manias, E.; Giannelis, E.P.; Wuthenow, M. Polymer layered-silicate nanocomposites: polyamide-6, polypropylene and polystyrene. In Proceedings of the New Advances in Flame Retardant Technology Conference, Tucson, AZ, USA, 27–27 October 1999. Fire Retardant Chemical Association. [Google Scholar]
- LeBaron, P. Polymer-layered silicate nanocomposites: an overview. Appl. Clay Sci. 1999, 15, 11–29. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: preparation, properties and uses of a new class of materials. Mater. Sci. Eng. R: Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Zanetti, M.; Lomakin, S.; Camino, G. Polymer layered silicate nanocomposites. Macromol. Mater. Eng. 2000, 279, 1–9. [Google Scholar] [CrossRef]
- Ahmadi, S.J.; Huang, Y.D.; Li, W. Synthetic routes, properties and future applications of polymer-layered silicate nanocomposites. J. Mater. Sci. 2004, 39, 1919–1925. [Google Scholar] [CrossRef]
- Zeng, Q.H.; Yu, A.B.; Lu, G.Q.; Paul, D.R.; Zeng, Q. Clay-Based Polymer Nanocomposites: Research and Commercial Development. J. Nanosci. Nanotechnol. 2005, 5, 1574–1592. [Google Scholar] [CrossRef]
- Pavlidou, S.; Papaspyrides, C. A review on polymer–layered silicate nanocomposites. Prog. Sci. 2008, 33, 1119–1198. [Google Scholar] [CrossRef]
- Ryu, J.G.; Lee, J.W.; Kim, H. Development of poly(methyl methacrylate)-clay nanocomposites by using power ultrasonic wave. Macromol. Res. 2002, 10, 187–193. [Google Scholar] [CrossRef]
- Diaconu, G.; Paulis, M.; Leiza, J.R. High Solids Content Waterborne Acrylic/Montmorillonite Nanocomposites by Miniemulsion Polymerization. Macromol. Eng. 2008, 2, 80–89. [Google Scholar] [CrossRef]
- Lakshmi, M.S.; Narmadha, B.; Reddy, B. Enhanced thermal stability and structural characteristics of different MMT-Clay/epoxy-nanocomposite materials. Degrad. Stab. 2008, 93, 201–213. [Google Scholar] [CrossRef]
- Dhibar, A.K.; Mallick, S.; Rath, T.; Khatua, B.B. Effect of clay platelet dispersion as affected by the manufacturing techniques on thermal and mechanical properties of PMMA-clay nanocomposites. J. Appl. Polym. Sci. 2009, 113, 3012–3018. [Google Scholar] [CrossRef]
- Chen, G.; Chen, X.; Lin, Z.; Ye, W.; Yao, K. Preparation and properties of PMMA/clay nanocomposite. J. Mater. Sci. Lett. 1999, 18, 1761–1763. [Google Scholar] [CrossRef]
- Okamoto, M.; Morita, S.; Taguchi, H.; Kim, Y.H.; Kotaka, T.; Tateyama, H. Synthesis and structure of smectic clay/poly(methyl methacrylate) and clay/polystyrene nanocomposites via in situ intercalative polymerization. Polymer 2000, 41, 3887–3890. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, B.; Xie, S.; Zhang, S. Synthesis and properties of poly(methyl methacrylate)/montmorillonite (PMMA/MMT) nanocomposites. Polym. Int. 2003, 52, 892–898. [Google Scholar] [CrossRef]
- Liu, G.-D.; Zhang, L.-C.; Qu, X.-W.; Wang, B.-T.; Zhang, Y. Tentative study on kinetics of bulk polymerization of methyl methacrylate in presence of montmorillonite. J. Appl. Sci. 2003, 90, 3690–3695. [Google Scholar] [CrossRef]
- Ray, S.S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Sci. 2003, 28, 1539–1641. [Google Scholar]
- Essawy, H.; Badran, A.; Youssef, A.; El-Hakim, A.E.-F.A. Synthesis of poly(methylmethacrylate)/montmorillonite nanocomposites via in situ intercalative suspension and emulsion polymerization. Polym. Bull. 2004, 53, 9–17. [Google Scholar] [CrossRef]
- Su, S.; Jiang, D.D.; Wilkie, C.A. Methacrylate modified clays and their polystyrene and poly(methyl methacrylate) nanocomposites. Polym. Adv. Technol. 2004, 15, 225–231. [Google Scholar] [CrossRef]
- Huskić, M.; Žigon, M. PMMA/MMT nanocomposites prepared by one-step in situ intercalative solution polymerization. Eur. J. 2007, 43, 4891–4897. [Google Scholar] [CrossRef]
- Cui, L.; Tarte, N.H.; Woo, S.I. Synthesis and properties of poly(methyl methacrylate)/clay nanocomposites prepared via in situ polymerization with Ni(acac)2catalyst. J. Appl. Sci. 2008, 110, 784–790. [Google Scholar] [CrossRef]
- Tiwari, R.R.; Khilar, K.C.; Natarajan, U. Synthesis and characterization of novel organo-montmorillonites. Appl. Clay Sci. 2004, 38, 203–208. [Google Scholar] [CrossRef]
- Karger-Kocsis, J.; Wu, C.-M.; Karger-Kocsis, J. Thermoset rubber/layered silicate nanocomposites. Status and future trends. Eng. Sci. 2004, 44, 1083–1093. [Google Scholar] [CrossRef]
- Kojima, Y.; Usuki, A.; Kawasumi, M.; Okada, A.; Kurauchi, T.; Kamigaito, O. One-pot synthesis of nylon 6–clay hybrid. J. Polym. Sci. Part A: Polym. Chem. 1993, 31, 1755–1758. [Google Scholar] [CrossRef]
- Burnside, S.D.; Giannelis, E.P. Synthesis and properties of new poly(dimethylsiloxane) nanocomposites. Chem. Mater. 1995, 7, 1597–1600. [Google Scholar] [CrossRef]
- Wang, Z.; Pinnavaia, T.J. Hybrid Organic−Inorganic Nanocomposites: Exfoliation of Magadiite Nanolayers in an Elastomeric Epoxy Polymer. Chem. Mater. 1998, 10, 1820–1826. [Google Scholar] [CrossRef]
- Sciascia, L.; Casella, S.; Cavallaro, G.; Lazzara, G.; Milioto, S.; Princivalle, F.; Parisi, F. Olive mill wastewaters decontamination based on organo-nano-clay composites. Ceram. Int. 2019, 45, 2751–2759. [Google Scholar] [CrossRef]
- Calabrese, I.; Cavallaro, G.; Scialabba, C.; Licciardi, M.; Merli, M.; Sciascia, L.; Liveri, M.L.T. Montmorillonite nanodevices for the colon metronidazole delivery. Int. J. Pharm. 2013, 457, 224–236. [Google Scholar] [CrossRef]
- Sciascia, L.; Liveri, M.L.T.; Merli, M. Kinetic and equilibrium studies for the adsorption of acid nucleic bases onto K10 montmorillonite. Appl. Clay Sci. 2011, 53, 657–668. [Google Scholar] [CrossRef]
- Mahmoodian, M.; Pourabbas, B.; Arya, A. Preparation and characterization of Bis-GMA/TEGDMA/clay nanocomposites at low filler content regimes. J. Compos. Mater. 2010, 44, 1379–1395. [Google Scholar] [CrossRef]
- Mucci, V.; Pérez, J.; Vallo, C. Preparation and characterization of light-cured methacrylate/montmorillonite nanocomposites. Polym. Int. 2011, 60, 247–254. [Google Scholar] [CrossRef]
- Terrin, M.M.; Poli, A.L.; Horn, M.A., Jr.; Neumann, M.G.; Cavalheiro, E.T.G.; Correa, I.C.; Schmitt, C.C. Effect of the loading of organomodified clays on the thermal and mechanical properties of a model dental resin. Mater. Res. 2016, 19, 40–44. [Google Scholar] [CrossRef]
- Campos, L.M.D.P.; Boaro, L.C.; Ferreira, H.P.; Dos Santos, L.K.G.; Dos Santos, T.R.; Parra, D.F. Evaluation of polymerization shrinkage in dental restorative experimental composites based: BisGMA/TEGDMA, filled with MMT. J. Appl. Sci. 2016, 133, 43543. [Google Scholar] [CrossRef]
- De Paiva Campos, L.M.; Lugão, A.B.; Vasconcelos, M.R.; Parra, D.F. Polymerization shrinkage evaluation on nanoscale-layered silicates: Bis-GMA/TEGMA nanocomposites, in photo-activated polymeric matrices. J. Appl. Polym. Sci. 2014, 131, 40010. [Google Scholar] [CrossRef]
- Campos, L.M.P.; Boaro, L.C.; Santos, T.M.R.; Marques, P.A.; Almeida, S.R.Y.; Braga, R.R.; Parra, D.F. Evaluation of flexural modulus, flexural strength and degree of conversion in BISGMA/TEGDMA resin filled with montmorillonite nanoparticles. J. Compos. Mater. 2017, 51, 927–937. [Google Scholar] [CrossRef]
- Campos, L.M.P.; Zaharescu, T.; Boaro, L.C.; Santos, L.K.G.; Santos, T.M.R.; Ferreira, H.P.; Parra, D.F. Thermal and mechanical behavior evaluation of dental composites filled with irradiated montmorillonite. J. Appl. Sci. 2017, 134, 45063. [Google Scholar] [CrossRef]
- Munhoz, T.; Fredholm, Y.; Rivory, P.; Balvay, S.; Hartmann, D.; Da Silva, P.; Chenal, J.-M. Effect of nanoclay addition on physical, chemical, optical and biological properties of experimental dental resin composites. Dent. Mater. 2017, 33, 271–279. [Google Scholar] [CrossRef]
- Menezes, L.R.; da Silva, E.O.; da Silva Rocha, A.C.; de Oliveira, D.C.R.S.; de Barros Campos, P.R. The applicability of organomodified nanoclays as new fillers for mechanical reinforcement of dental composites. J. Compos. Mater. 2018, 52, 963–970. [Google Scholar] [CrossRef]
- Nikolaidis, A.K.; Achilias, D.S.; Karayannidis, G.P. Effect of the type of organic modifier on the polymerization kinetics and the properties of poly(methyl methacrylate)/organomodified montmorillonite nanocomposites. Eur. J. 2012, 48, 240–251. [Google Scholar] [CrossRef]
- Nikolaidis, A.K.; Koulaouzidou, E.A.; Achilias, D.S. Synthesis and characterization of novel organomodified nanoclays for application in dental materials. Curr. Nanosci. 2018, 14, 1–12. [Google Scholar] [CrossRef]
- Watts, D.; Marouf, A. Optimal specimen geometry in bonded-disk shrinkage-strain measurements on light-cured biomaterials. Dent. Mater. 2000, 16, 447–451. [Google Scholar] [CrossRef]
- Watts, D.; Marouf, A.; Al-Hindi, A. Photo-polymerization shrinkage-stress kinetics in resin-composites: methods development. Dent. Mater. 2003, 19, 1–11. [Google Scholar] [CrossRef]
- Al Sunbul, H.; Silikas, N.; Watts, D.C. Polymerization shrinkage kinetics and shrinkage-stress in dental resin-composites. Dent. Mater. 2016, 32, 998–1006. [Google Scholar] [CrossRef]
- Rueggeberg, F.; Hashinger, D.; Fairhurst, C. Calibration of FTIR conversion analysis of contemporary dental resin composites. Dent. Mater. 1990, 6, 241–249. [Google Scholar] [CrossRef]
- Jlassi, K.; Chandran, S.; Mičušík, M.; Benna-Zayani, M.; Yagci, Y.; Thomas, S.; Chehimi, M.M. Poly(glycidyl methacrylate)-grafted clay nanofiller for highly transparent and mechanically robust epoxy composites. Eur. J. 2015, 72, 89–101. [Google Scholar] [CrossRef]
- Jlassi, K.; Kurup, S.C.C.; Poothanari, M.A.; Benna-Zayani, M.; Thomas, S.; Chehimi, M.M. Clay/polyaniline hybrid through diazonium chemistry: conductive nanofiller with unusual effects on interfacial properties of epoxy nanocomposites. Langmuir 2016, 32, 3514–3524. [Google Scholar] [CrossRef] [PubMed]
- Campos, L.M.P.; Boaro, L.C.; Santos, T.M.R.; Santos, V.J.; Grecco-Romano, R.; Santos, M.J.; Parra, D.F. Polymerization shrinkage evaluation in experimental dental composite filled with montmorillonite nanoparticles. J. Therm. Anal. Calorim. 2018, 131, 771–774. [Google Scholar] [CrossRef]
- Halvorson, R.H.; Erickson, R.L.; Davidson, C.L. The effect of filler and silane content on conversion of resin-based composite. Dent. Mater. 2003, 19, 327–333. [Google Scholar] [CrossRef]
- Wilson, K.S.; Zhang, K.; Antonucci, J.M. Systematic variation of interfacial phase reactivity in dental nanocomposites. Biomaterials 2005, 26, 5095–5103. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.S.; Antonucci, J.M. Interphase structure–property relationships in thermoset dimethacrylate nanocomposites. Dent. Mater. 2006, 22, 995–1001. [Google Scholar] [CrossRef]
- Gonçalves, F.; Kawano, Y.; Pfeifer, C.; Stansbury, J.W.; Braga, R.R. Influence of BisGMA, TEGDMA, and BisEMA contents on viscosity, conversion, and flexural strength of experimental resins and composites. Eur. J. Oral Sci. 2009, 117, 442–446. [Google Scholar] [CrossRef]
- Sideridou, I.D.; Karabela, M.M. Effect of the structure of silane-coupling agent on dynamic mechanical properties of dental resin-nanocomposites. J. Appl. Polym. Sci. 2008, 110, 507–516. [Google Scholar] [CrossRef]
- Discacciati, J.A.C.; Oréfice, R.L. Structural analysis on photopolymerized dental resins containing nanocomponents. J. Mater. Sci. 2007, 42, 3883–3893. [Google Scholar] [CrossRef]
- Cuevas-Suárez, C.E.; Pimentel-García, B.; Rivera-Gonzaga, A.; Álvarez-Gayosso, C.; Ancona-Meza, A.L.; Grazioli, G.; Zamarripa-Calderón, E. Examining the Effect of Radiant Exposure on Commercial Photopolimerizable Dental Resin Composites. Dent. J. 2018, 6, 55. [Google Scholar] [CrossRef]
- Leprince, J.G.; Palin, W.M.; Vanacker, J.; Sabbagh, J.; Devaux, J.; Leloup, G. Physico-mechanical characteristics of commercially available bulk-fill composites. J. Dent. 2014, 42, 993–1000. [Google Scholar] [CrossRef]
- Achilias, D.S. A Review of Modeling of Diffusion Controlled Polymerization Reactions. Macromol. Theory Simul. 2007, 16, 319–347. [Google Scholar] [CrossRef]
- Achilias, D.S.; Verros, G.D. Modeling of diffusion-controlled reactions in free radical solution and bulk polymerization: Model validation by DSC experiments. J. Appl. Sci. 2010, 116, 1842–1856. [Google Scholar] [CrossRef]
- Verros, G.; Latsos, T.; Achilias, D.; Achilias, D. Development of a unified framework for calculating molecular weight distribution in diffusion controlled free radical bulk homo-polymerization. Polymer 2005, 46, 539–552. [Google Scholar] [CrossRef]
- Verros, G.D.; Achilias, D.S. Modeling gel effect in branched polymer systems: Free-radical solution homopolymerization of vinyl acetate. J. Appl. Sci. 2009, 111, 2171–2185. [Google Scholar] [CrossRef]
- Menezes, L.R.; Silva, E.O. The Use of montmorillonite clays as reinforcing fillers for dental adhesives. Mater. Res. 2016, 19, 236–242. [Google Scholar] [CrossRef]
- Ratanajanchai, M.; Tanwilai, D.; Sunintaboon, P. Visible light-induced surfactant-free emulsion polymerization using camphorquinone/tertiary amine as the initiating system for the synthesis of amine-functionalized colloidal nanoparticles. J. Colloid Interface Sci. 2013, 409, 25–31. [Google Scholar] [CrossRef]
- Tsagkalias, I.S.; Manios, T.K.; Achilias, D.S. Effect of Graphene Oxide on the Reaction Kinetics of Methyl Methacrylate In Situ Radical Polymerization via the Bulk or Solution Technique. Polymers 2017, 9, 432. [Google Scholar] [CrossRef] [PubMed]
- Ilie, N.; Durner, J. Polymerization kinetic calculations in dental composites: a method comparison analysis. Clin. Oral Investig. 2014, 18, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Kelly, P.; Akelah, A.; Qutubuddin, S.; Moet, A. Reduction of residual stress in montmorillonite/epoxy compounds. J. Mater. Sci. 1994, 29, 2274–2280. [Google Scholar] [CrossRef]
- Salahuddin, N.; Shehata, M. Polymethylmethacrylate–montmorillonite composites: preparation, characterization and properties. Polymer 2001, 42, 8379–8385. [Google Scholar] [CrossRef]
- Hussain, F.; Chen, J.; Hojjati, M. Epoxy-silicate nanocomposites: Cure monitoring and characterization. Mater. Sci. Eng. A 2007, 445, 467–476. [Google Scholar] [CrossRef]
- Qu, X.; Guan, T.; Liu, G.; She, Q.; Zhang, L. Preparation, structural characterization, and properties of poly(methyl methacrylate)/montmorillonite nanocomposites by bulk polymerization. J. Appl. Sci. 2005, 97, 348–357. [Google Scholar] [CrossRef]
- Noh, M.W.; Lee, D.C. Synthesis and characterization of PS-clay nanocomposite by emulsion polymerization. Polym. Bull. 1999, 42, 619–626. [Google Scholar] [CrossRef]
- Atai, M.; Pahlavan, A.; Moin, N. Nano-porous thermally sintered nano silica as novel fillers for dental composites. Dent. Mater. 2012, 28, 133–145. [Google Scholar] [CrossRef]
- Bee, S.-T.; Hassan, A.; Ratnam, C.T.; Tee, T.-T.; Sin, L.T.; Hui, D. Dispersion and roles of montmorillonite on structural, flammability, thermal and mechanical behaviours of electron beam irradiated flame retarded nanocomposite. Compos. Part B: Eng. 2014, 61, 41–48. [Google Scholar] [CrossRef]
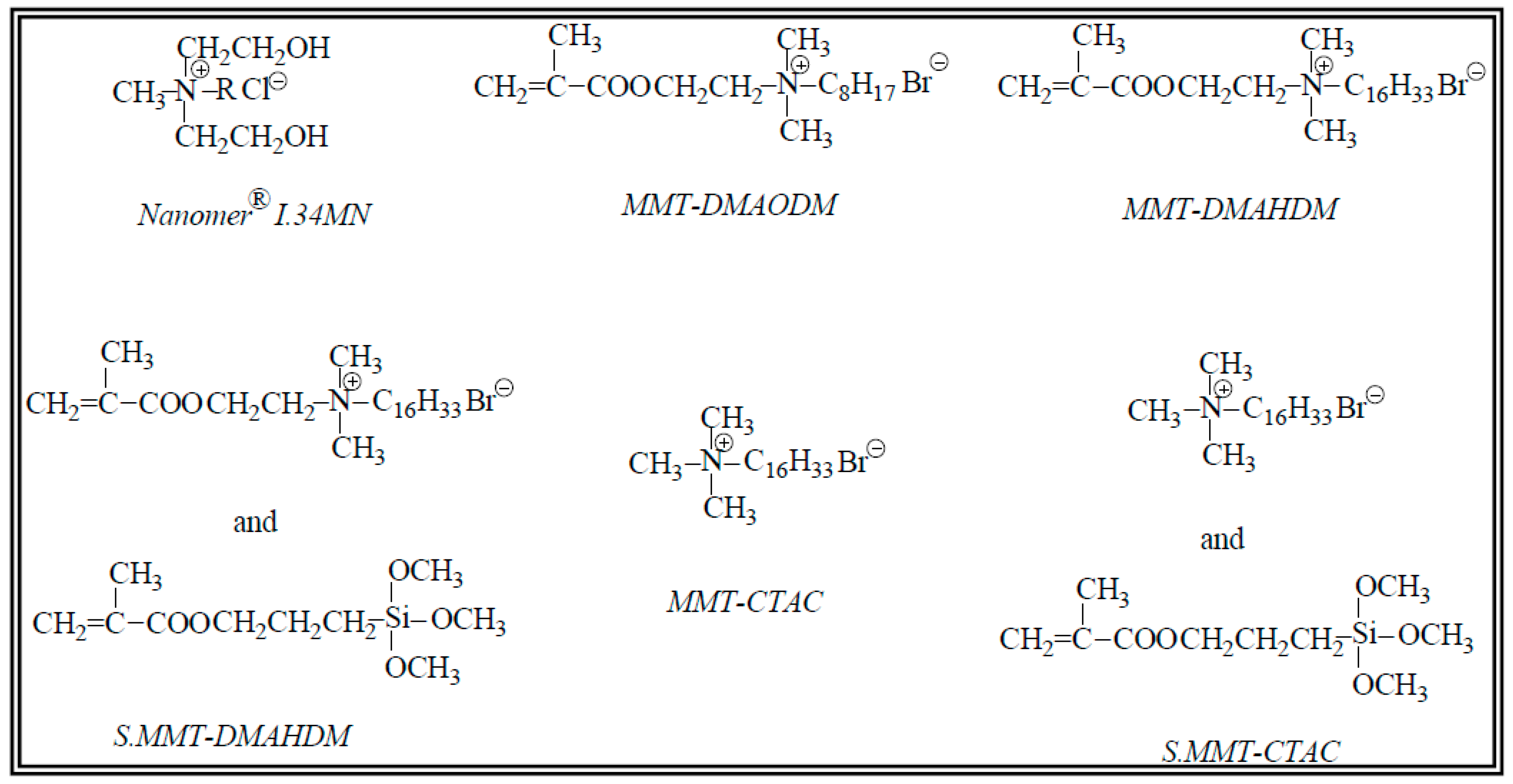
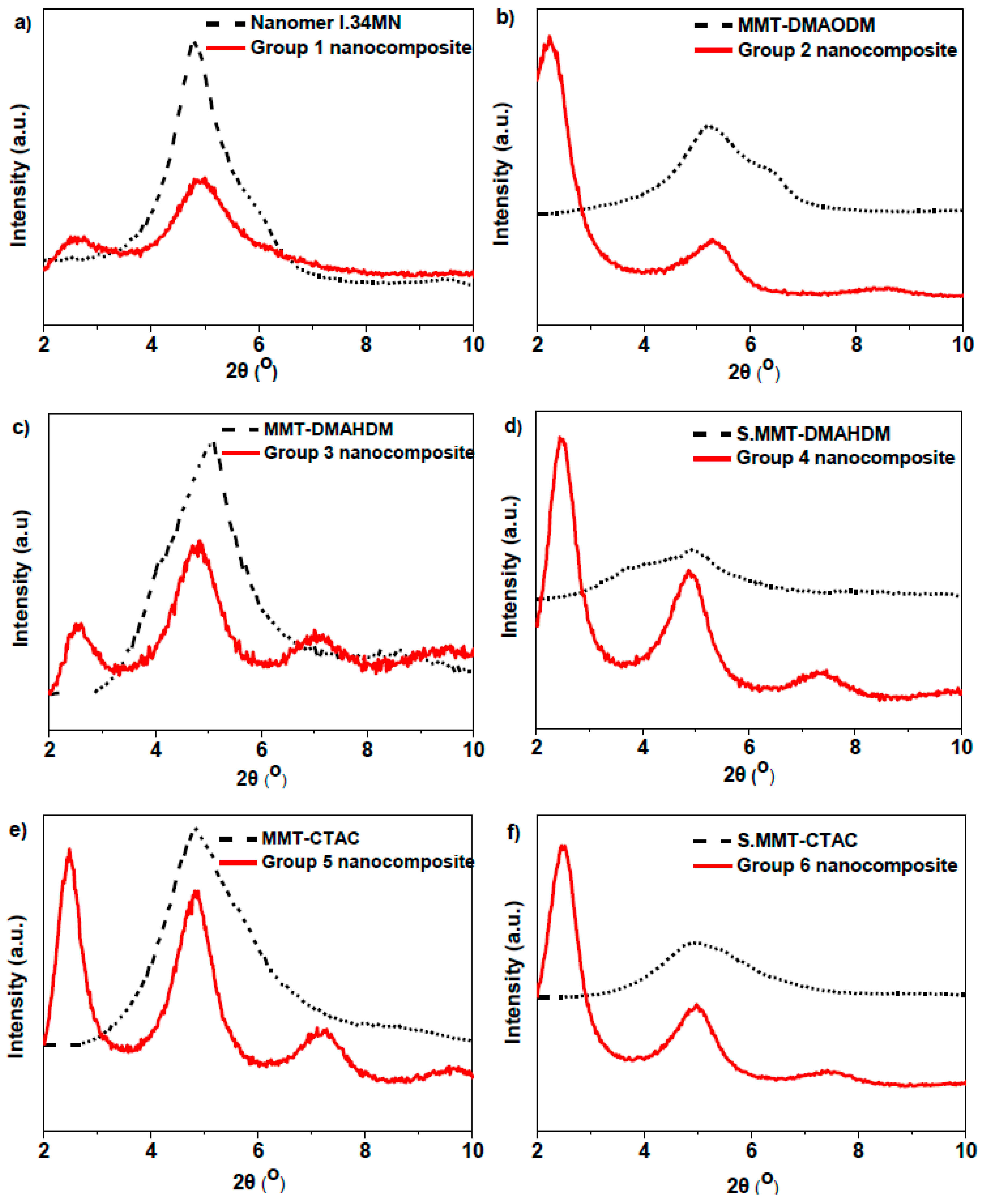

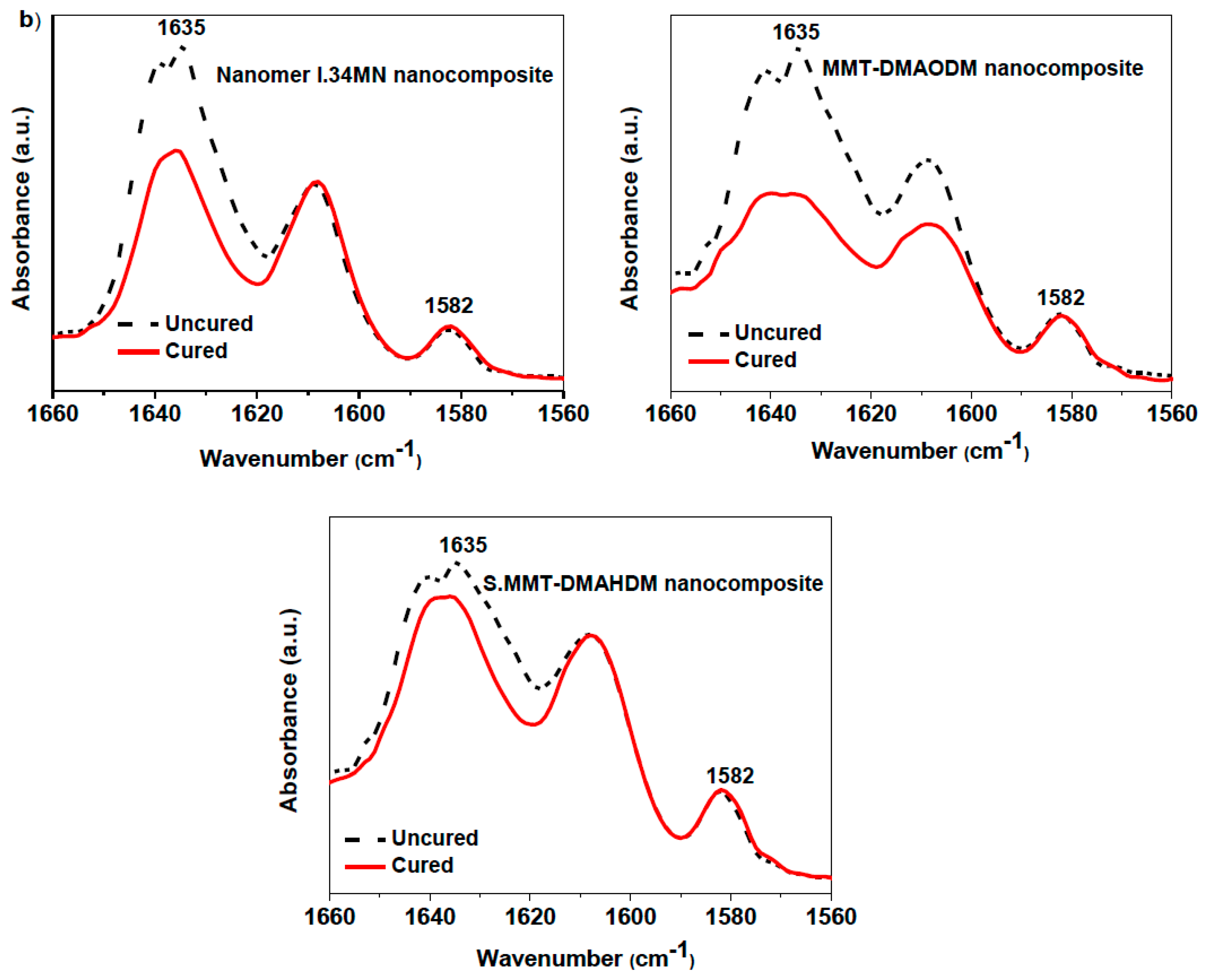

| Nanocomposite Resin | OMMT Nanofiller (50 wt %) | d001 (nm) | Δd001 (nm) | |
|---|---|---|---|---|
| Pure OMMT [49,50] | OMMT into Nanocomposite | |||
| Group 1 | Nanomer® I.34MN | 1.86 | 3.39 | 1.53 |
| Group 2 | MMT-DMAODM | 1.77 | 3.95 | 2.18 |
| Group 3 | MMT-DMAHDM | 1.73 | 3.45 | 1.72 |
| Group 4 | S.MMT-DMAHDM | 1.78 | 3.62 | 1.84 |
| Group 5 | MMT-CTAC | 1.83 | 3.56 | 1.73 |
| Group 6 | S.MMT-CTAC | 1.79 | 3.60 | 1.81 |
| Sample | Final DC (%) | Total Strain (%) | Flexural Modulus (GPa) | Flexural Strength (MPa) |
|---|---|---|---|---|
| Bis-GMA/TEGDMA | 61.60 | 6.47 ± 0.19 | 1.53 ± 0.40 a,b,c | 92.59 ± 13.94 d,e,f |
| Nanomer® I.34MN nanocomposite | 56.80 | 3.46 ± 0.37 | 2.40 ± 0.64 | 29.45 ± 11.45 |
| MMT-DMAODM nanocomposite | 70.60 | 2.80 ± 0.23 | 3.32 ± 0.36 a | 36.60 ± 7.28 |
| MMT-DMAHDM nanocomposite | 60.80 | 2.71 ± 0.13 | 2.30 ± 0.15 | 26.82 ± 6.33 d |
| S.MMT-DMAHDM nanocomposite | 61.70 | 2.86 ± 0.37 | 3.14 ± 0.47 b | 32.39 ± 3.52 |
| MMT-CTAC nanocomposite | 41.00 | 2.51 ± 0.29 | 2.50 ± 0.84 | 21.14 ± 6.79 e |
| S.MMT-CTAC nanocomposite | 45.80 | 2.54 ± 0.22 | 3.48 ± 0.34 c | 23.09 ± 10.23 f |
| FiltekTM Z350 XT (3M ESPE, St. Paul, MN, USA) [64] | 50.96 | 1.66 ± 0.15 | 9.13 ± 0.66 | 80.52 ± 15.88 |
| Tetric® N-Ceram Bulk Fill (Ivoclar-Vivadent, Schaan, Liechtenstein) [64] | 49.50 | 1.36 ± 0.08 | 7.05 ± 0.60 | 60.37 ± 11.05 |
| Tetric®Evo Ceram Bulk Fill (Ivoclar-Vivadent, Schaan, Liechtenstein) [65] | 56.70 | - | 6.10 | 94.50 |
| Grandio (Voco, Cuxhaven, Germany) [65] | 62.80 | - | 15.30 | 125.00 |
| Resin | y0 | α | b | c | d | R2 | Final DC (%) |
|---|---|---|---|---|---|---|---|
| Bis-GMA/TEGDMA | −0.2870 | 6.4892 | 0.1446 | 54.9137 | 0.1447 | 0.9972 | 61.69 |
| Nanomer® I.34MN nanocomposite | −1.8527 | 41.3735 | 0.0652 | 16.4640 | 0.0652 | 0.9929 | 55.98 |
| MMT-DMAHDM nanocomposite | 0.0000 | 15.1918 | 0.0462 | 45.2200 | 1.0484 | 0.9982 | 60.41 |
| S.MMT-DMAHDM nanocomposite | 0.0000 | 10.2364 | 0.0243 | 51.4829 | 0.5863 | 0.9995 | 61.72 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikolaidis, A.K.; Koulaouzidou, E.A.; Gogos, C.; Achilias, D.S. Synthesis and Characterization of Dental Nanocomposite Resins Filled with Different Clay Nanoparticles. Polymers 2019, 11, 730. https://doi.org/10.3390/polym11040730
Nikolaidis AK, Koulaouzidou EA, Gogos C, Achilias DS. Synthesis and Characterization of Dental Nanocomposite Resins Filled with Different Clay Nanoparticles. Polymers. 2019; 11(4):730. https://doi.org/10.3390/polym11040730
Chicago/Turabian StyleNikolaidis, Alexandros K., Elisabeth A. Koulaouzidou, Christos Gogos, and Dimitris S. Achilias. 2019. "Synthesis and Characterization of Dental Nanocomposite Resins Filled with Different Clay Nanoparticles" Polymers 11, no. 4: 730. https://doi.org/10.3390/polym11040730
APA StyleNikolaidis, A. K., Koulaouzidou, E. A., Gogos, C., & Achilias, D. S. (2019). Synthesis and Characterization of Dental Nanocomposite Resins Filled with Different Clay Nanoparticles. Polymers, 11(4), 730. https://doi.org/10.3390/polym11040730








