Investigation on the Effect of Hyperbranched Polyester Grafted Graphene Oxide on the Crystallization Behaviors of β-Nucleated Isotactic Polypropylene
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Sample Preparation
- (1)
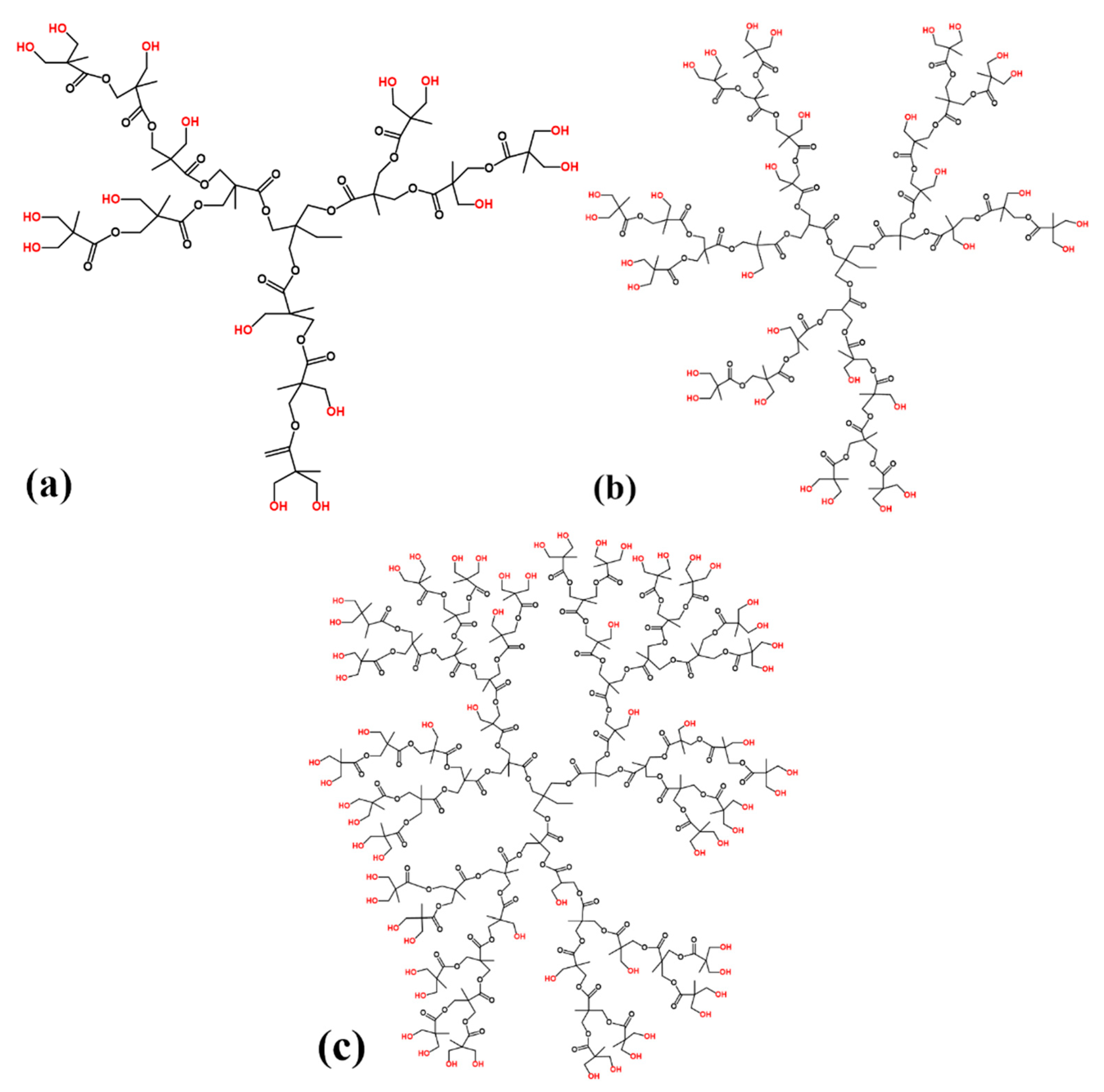
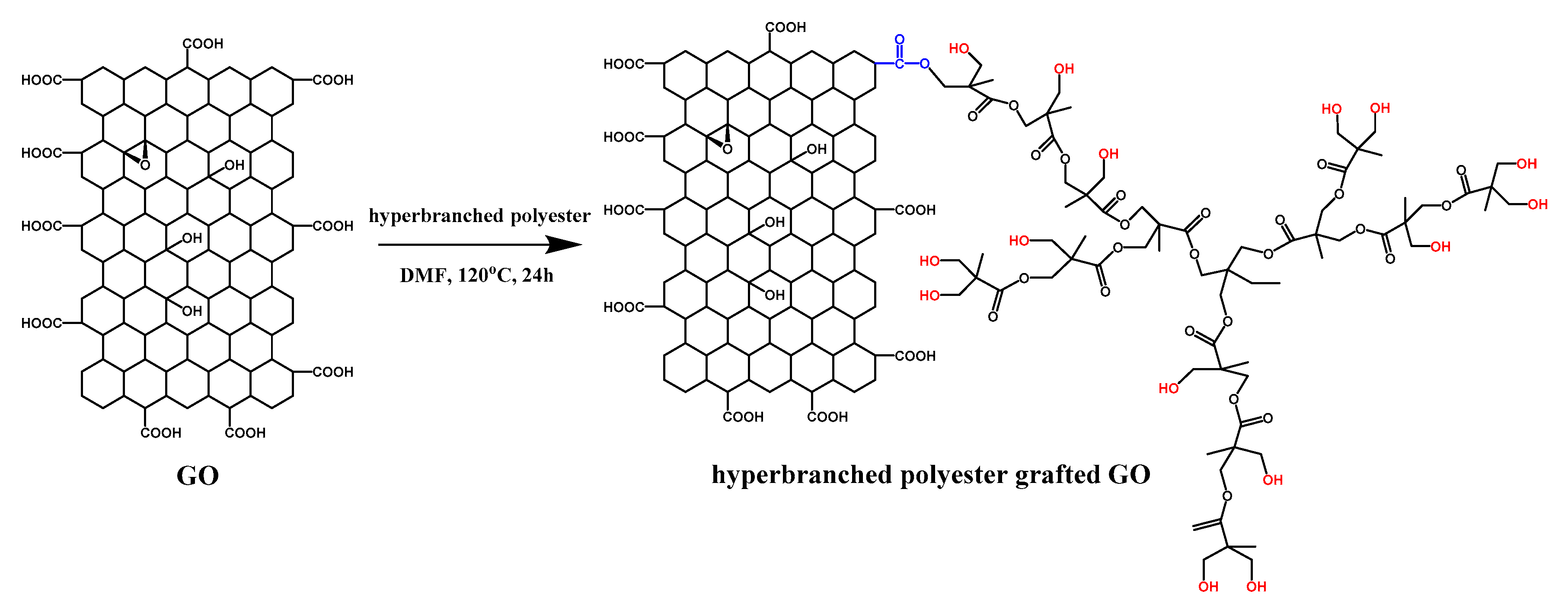
- The schematic of hyperbranched polyester grafting on graphene oxide is shown in Scheme 2. For the sake of grafting hyperbranched polyester on graphene oxide, graphene oxide (0.2 g), hyperbranched polyester (5 g), and tetrabutylammonium bromide (TBAB, 20 mg) were added in 200 mL DMF, then ultrasonically dispersed under room temperature for 1 h [25]. The obtained mixture was stirred under 120 °C for 24 h and vacuum filtered. The rinsing–filtration cycle should be repeated eight times to remove excess and physisorption of hyperbranched polyester by acetone. The resulting hyperbranched polyester grafted graphene oxide was vacuum dried under 60 °C for 12 h. Finally, hyperbranched polyester grafted GO was prepared.
- (2)
- In order to prepare hyperbranched polyester grafted iPP/GO composites, hyperbranched polyester grafted GO, iPP, and β-NA WBG-II were simultaneously added into an extruder by micro-extrusion for extrusion processing at 200 °C, with 1.0 wt % GO and 0.05 wt % WBG-II. After that, the obtained sample was shaped by a pressure-molding machine. Lastly, characterization tests were carried out [26].
2.3. Characterization Tests
2.3.1. X-ray Photoelectron Spectra (XPS)
2.3.2. Fourier Transform Infrared (FT-IR)
2.3.3. Thermogravimetric Analysis (TGA)
2.3.4. Transmission Electron Microscopy (TEM)
2.3.5. Differential Scanning Calorimetry (DSC)
2.3.6. Wide-Angle X-ray Diffraction (WAXD)
2.3.7. Non-Isothermal Crystallization Kinetics
- (1)
- To eliminate thermal histories, all samples were heated from 50 to 200 °C under 10 °C/min and kept for 5 min.
- (2)
- All samples were cooled down to 50 °C under 2, 5, 10, 20, and 40 °C/min, respectively, to get DSC cooling curves.
- (3)
- All samples were heated to 200 °C under 10 °C/min to get DSC heating curves.
3. Results and Discussion
3.1. Characterizations of Hyperbranched Polyester Grafted Graphene Oxide
3.1.1. XPS Survey Spectra
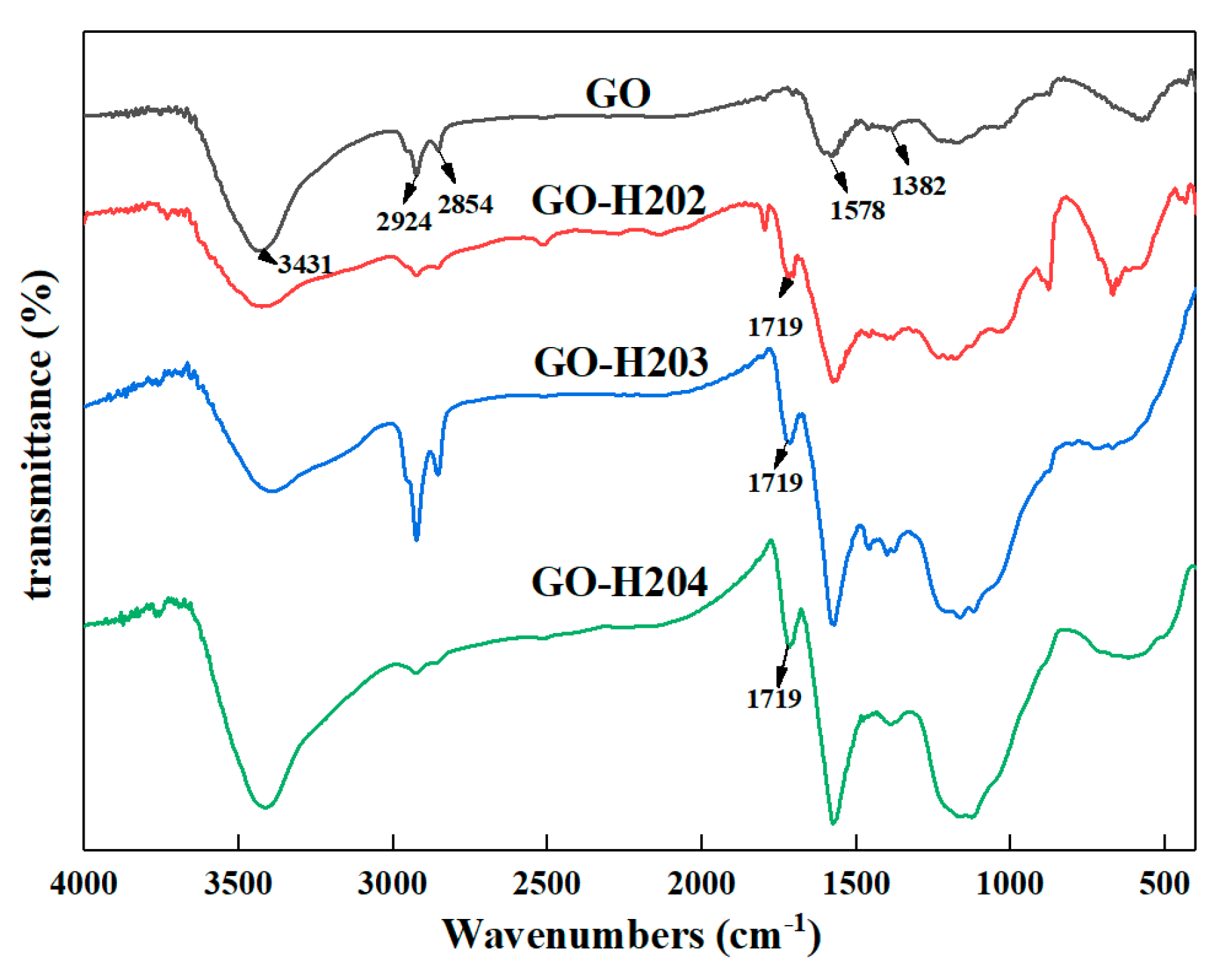
3.1.2. FT-IR Spectra
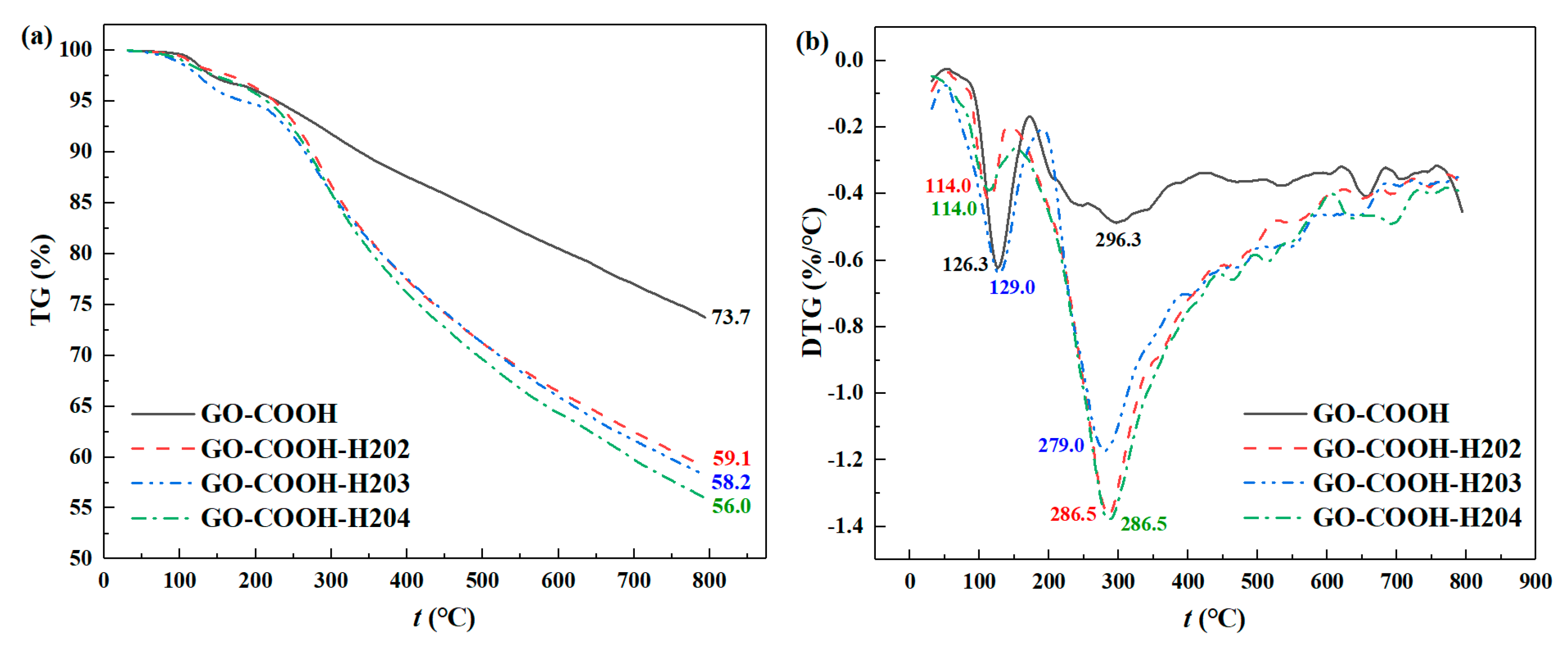
3.1.3. Thermogravimetric Analysis (TGA)
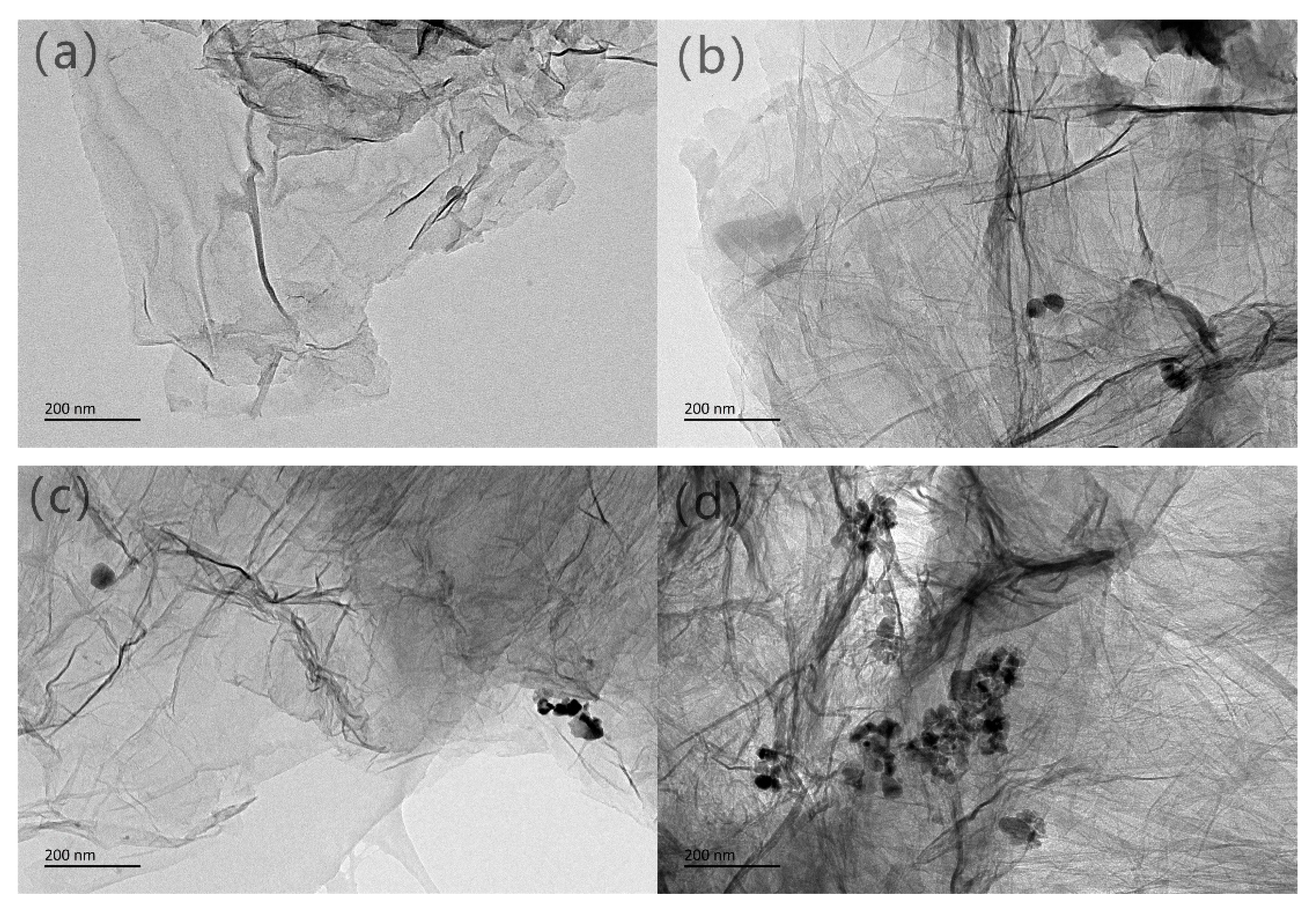
3.1.4. TEM Images
3.2. Non-Isothermal Crystallization Kinetics
3.2.1. Cooling Curves
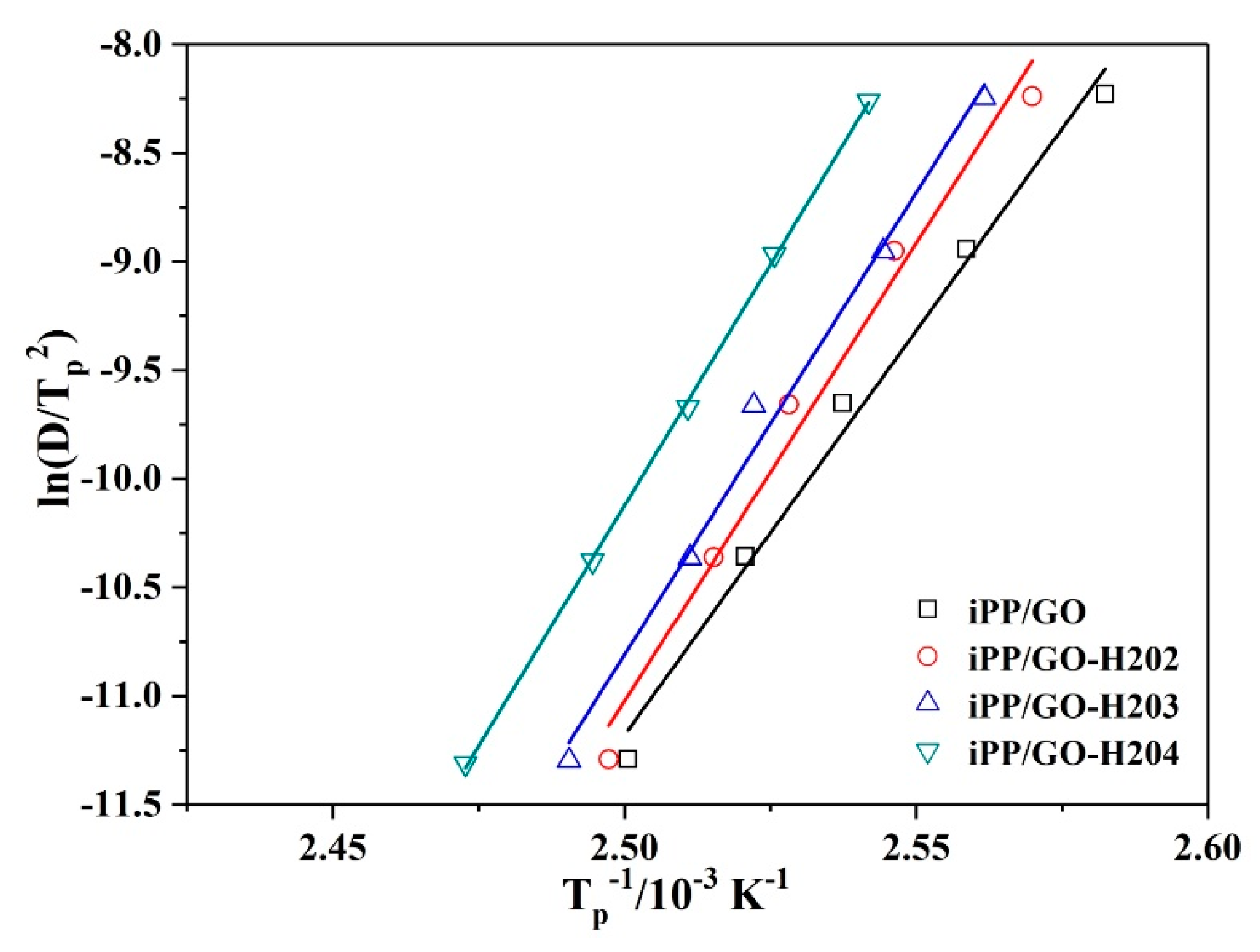
3.2.2. Crystallization Activation Energy
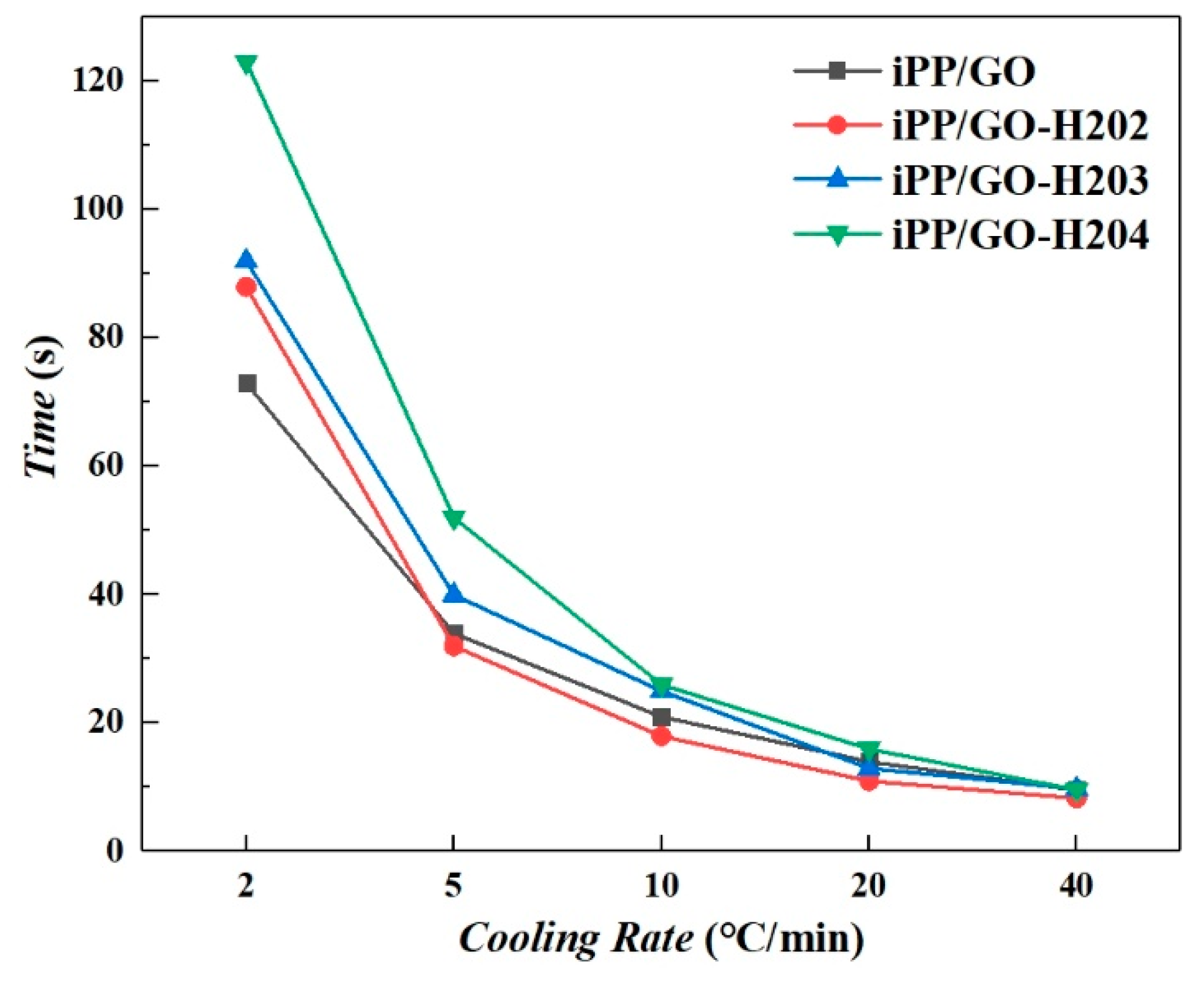
3.2.3. Half Crystallization Time
3.3. Crystallization Parameters
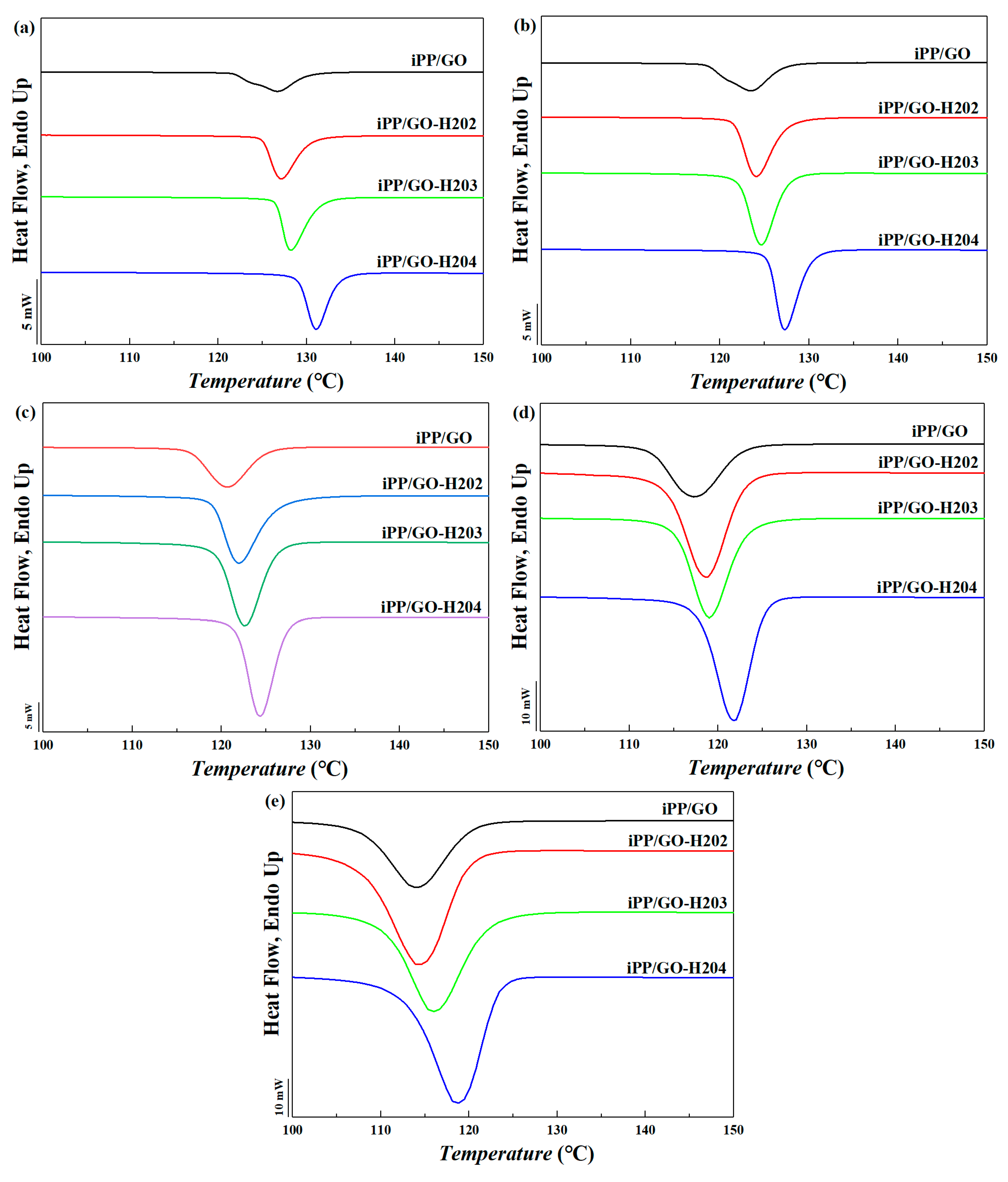
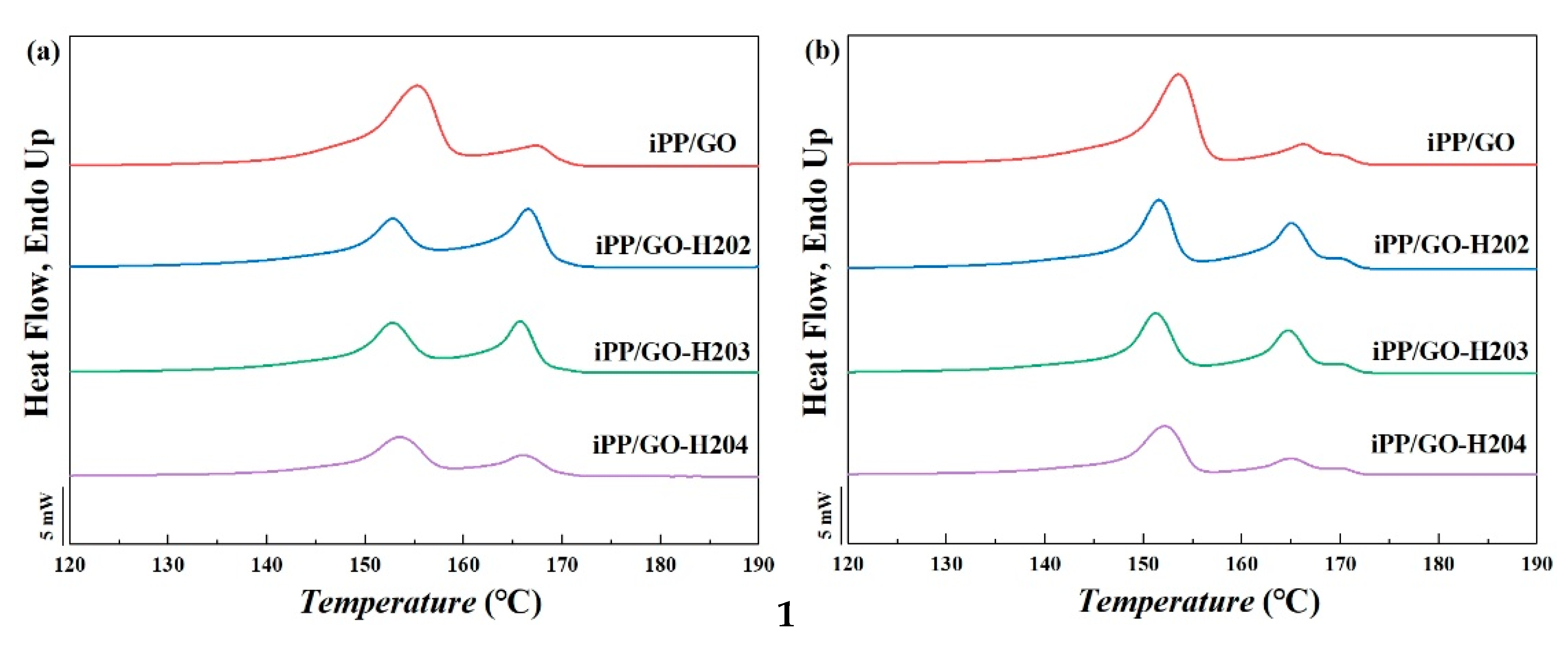
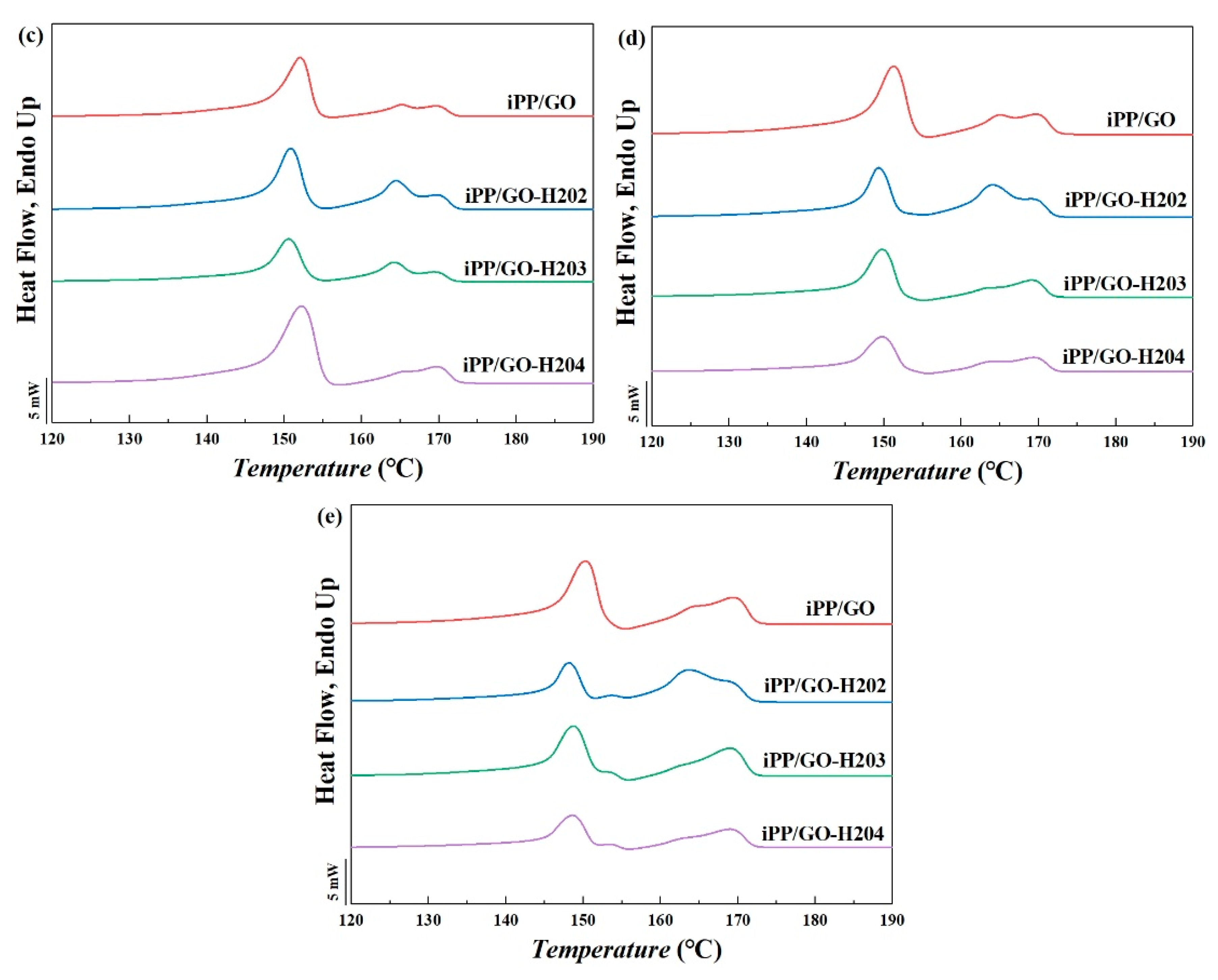
3.3.1. DSC Results
3.3.2. WAXD Results
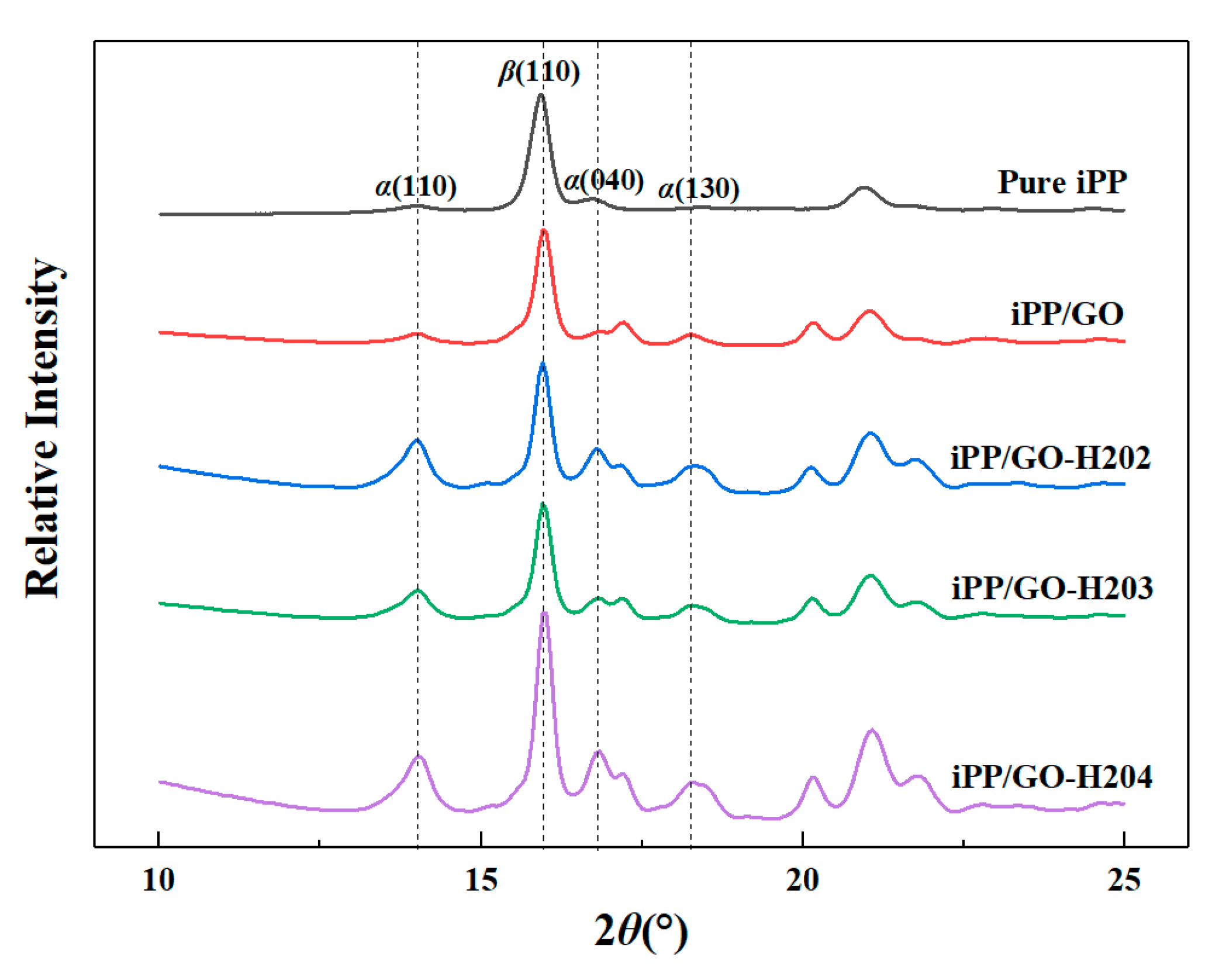
3.3.3. Mechanism Discussions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, S.-W.; Yang, W.; Bao, R.-Y.; Wang, B.; Xie, B.-H.; Yang, M.-B. The enhanced nucleating ability of carbon nanotube-supported β-nucleating agent in isotactic polypropylene. Colloid Polym. Sci. 2010, 288, 681–688. [Google Scholar] [CrossRef]
- Du, M.; Guo, B.; Wan, J.; Zou, Q.; Jia, D. Effects of halloysite nanotubes on kinetics and activation energy of non-isothermal crystallization of polypropylene. J. Polym. Res. 2009, 17, 109–118. [Google Scholar] [CrossRef]
- Chammingkwan, P.; Matsushita, K.; Taniike, T.; Terano, M. Enhancement in Mechanical and Electrical Properties of Polypropylene Using Graphene Oxide Grafted with End-Functionalized Polypropylene. Materials (Basel) 2016, 9, 240. [Google Scholar] [CrossRef]
- Dutta, S.; Pati, S.K. Novel properties of graphene nanoribbons: A review. J. Mater. Chem. 2010, 20, 8207–8223. [Google Scholar] [CrossRef]
- Yun, Y.S.; Bae, Y.H.; Kim, D.H.; Lee, J.Y.; Chin, I.-J.; Jin, H.-J. Reinforcing effects of adding alkylated graphene oxide to polypropylene. Carbon 2011, 49, 3553–3559. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Park, O.-K.; Hwang, J.-Y.; Goh, M.; Lee, J.H.; Ku, B.-C.; You, N.-H. Mechanically Strong and Multifunctional Polyimide Nanocomposites Using Amimophenyl Functionalized Graphene Nanosheets. Macromolecules 2013, 46, 3505–3511. [Google Scholar] [CrossRef]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Cui, Y.; Kundalwal, S.I.; Kumar, S. Gas barrier performance of graphene/polymer nanocomposites. Carbon 2016, 98, 313–333. [Google Scholar] [CrossRef]
- Goyal, V.; Balandin, A.A. Thermal properties of the hybrid graphene-metal nano-micro-composites: Applications in thermal interface materials. Appl. Phys. Lett. 2012, 100. [Google Scholar] [CrossRef]
- Liang, J.; Huang, Y.; Zhang, L.; Wang, Y.; Ma, Y.; Guo, T.; Chen, Y. Molecular-Level Dispersion of Graphene into Poly(vinyl alcohol) and Effective Reinforcement of their Nanocomposites. Adv. Funct. Mater. 2009, 19, 2297–2302. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Potts, J.R.; Lee, S.H.; Alam, T.M.; An, J.; Stoller, M.D.; Piner, R.D.; Ruoff, R.S. Thermomechanical properties of chemically modified graphene/poly(methyl methacrylate) composites made by in situ polymerization. Carbon 2011, 49, 2615–2623. [Google Scholar] [CrossRef]
- Li, W.; Tang, X.-Z.; Zhang, H.-B.; Jiang, Z.-G.; Yu, Z.-Z.; Du, X.-S.; Mai, Y.-W. Simultaneous surface functionalization and reduction of graphene oxide with octadecylamine for electrically conductive polystyrene composites. Carbon 2011, 49, 4724–4730. [Google Scholar] [CrossRef]
- Wang, G.; Shen, X.; Wang, B.; Yao, J.; Park, J. Synthesis and characterisation of hydrophilic and organophilic graphene nanosheets. Carbon 2009, 47, 1359–1364. [Google Scholar] [CrossRef]
- Yu, Y.; Zeng, F.; Chen, J.; Kang, J.; Yang, F.; Cao, Y.; Xiang, M. Regulating polycrystalline behavior of the β-nucleated isotactic polypropylene/graphene oxide composites by melt memory effect. Polym. Compos. 2019, 40 (Suppl. S1), E440–E448. [Google Scholar] [CrossRef]
- Shin, K.-Y.; Hong, J.-Y.; Lee, S.; Jang, J. Evaluation of anti-scratch properties of graphene oxide/polypropylene nanocomposites. J. Mater. Chem. 2012, 22, 7871–7879. [Google Scholar] [CrossRef]
- Zhang, Z.-B.; Wu, J.-J.; Su, Y.; Zhou, J.; Gao, Y.; Yu, H.-Y.; Gu, J.-S. Layer-by-layer assembly of graphene oxide on polypropylene macroporous membranes via click chemistry to improve antibacterial and antifouling performance. Appl. Surf. Sci. 2015, 332, 300–307. [Google Scholar] [CrossRef]
- Hao, Z.; Li, L.; Liao, X.; Sheng, X.; Zhang, Y. Preparation and toughening performance investigation of epoxy resins containing carbon nanotubes modified with hyperbranched polyester. Polym. Bull. 2017, 75, 1013–1026. [Google Scholar] [CrossRef]
- Han, W.; Liao, X.; Yang, Q.; Li, G.; He, B.; Zhu, W.; Hao, Z. Crystallization and morphological transition of poly(l-lactide)–poly(ε-caprolactone) diblock copolymers with different block length ratios. RSC Adv. 2017, 7, 22515–22523. [Google Scholar] [CrossRef]
- Lu, L.; Xia, L.; Zengheng, H.; Xingyue, S.; Yi, Z.; Pan, L. Investigation on cure kinetics of epoxy resin containing carbon nanotubes modified with hyper-branched polyester. RSC Adv. 2018, 8, 29830–29839. [Google Scholar] [CrossRef]
- Hao, Z.; Li, L.; Yang, B.; Sheng, X.; Liao, X.; He, L.; Liu, P. Influences of Hyperbranched Polyester Modification on the Crystallization Kinetics of Isotactic Polypropylene/Graphene Oxide Composites. Polymers (Basel) 2019, 11, 433. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Xu, R.; Chen, J.; Kang, J.; Xiang, M.; Li, Y.; Li, L.; Sheng, X. Ordered structure effects on β-nucleated isotactic polypropylene/graphene oxide composites with different thermal histories. RSC Adv. 2019, 9, 19630–19640. [Google Scholar] [CrossRef]
- Bao, R.-Y.; Cao, J.; Liu, Z.-Y.; Yang, W.; Xie, B.-H.; Yang, M.-B. Towards balanced strength and toughness improvement of isotactic polypropylene nanocomposites by surface functionalized graphene oxide. J. Mater. Chem. A 2014, 2, 3190–3199. [Google Scholar] [CrossRef]
- Zhang, Q.; Peng, H.; Kang, J.; Cao, Y.; Xiang, M. Effects of melt structure on non-isothermal crystallization behavior of isotactic polypropylene nucleated with α/β compounded nucleating agents. Polym. Eng. Sci. 2017, 57, 989–997. [Google Scholar] [CrossRef]
- Yu, Y.; Zeng, F.; Chen, J.; Kang, J.; Yang, F.; Cao, Y.; Xiang, M. Isothermal crystallization kinetics and subsequent melting behavior of β-nucleated isotactic polypropylene/graphene oxide composites with different ordered structure. Polym. Int. 2018, 67, 1212–1220. [Google Scholar] [CrossRef]
- Xu, R.; Xu, G.; Wang, J.; Chen, J.; Yang, F.; Kang, J.; Xiang, M. Influence of l-lysine on the permeation and antifouling performance of polyamide thin film composite reverse osmosis membranes. RSC Adv. 2018, 8, 25236–25247. [Google Scholar] [CrossRef]
- Wang, J.; Xu, R.; Yang, F.; Kang, J.; Cao, Y.; Xiang, M. Probing influences of support layer on the morphology of polyamide selective layer of thin film composite membrane. J. Membr. Sci. 2018, 556, 374–383. [Google Scholar] [CrossRef]
- Kang, J.; Yang, F.; Wu, T.; Li, H.; Cao, Y.; Xiang, M. Polymerization control and fast characterization of the stereo-defect distribution of heterogeneous Ziegler–Natta isotactic polypropylene. Eur. Polym. J. 2012, 48, 425–434. [Google Scholar] [CrossRef]
- Kang, J.; Zhang, J.; Chen, Z.; Yang, F.; Chen, J.; Cao, Y.; Xiang, M. Isothermal crystallization behavior of β-nucleated isotactic polypropylene with different melt structures. J. Polym. Res. 2014, 21, 506. [Google Scholar] [CrossRef]
- Lu, Q.; Dou, Q. β-Crystal formation of isotactic polypropylene induced by N, N’-dicyclohexylsuccinamide. J. Polym. Res. 2008, 16, 555–560. [Google Scholar] [CrossRef]
- Dorset, D.L.; McCourt, M.P.; Kopp, S.; Schumacher, M.; Okihara, T.; Lotz, B. Isotactic polypropylene, β-phase: A study in frustration. Polymer 1998, 39, 6331–6337. [Google Scholar] [CrossRef]
- Yu, Y.; Zeng, F.; Chen, J.; Kang, J.; Yang, F.; Cao, Y.; Xiang, M. Effects of ordered structure on non-isothermal crystallization kinetics and subsequent melting behavior of β-nucleated isotactic polypropylene/graphene oxide composites. J. Therm. Anal. Calorim. 2018, 136, 1667–1678. [Google Scholar] [CrossRef]
- Huang, Y.; Qin, Y.; Zhou, Y.; Niu, H.; Yu, Z.-Z.; Dong, J.-Y. Polypropylene/Graphene Oxide Nanocomposites Prepared by in Situ Ziegler−Natta Polymerization. Chem. Mater. 2010, 22, 4096–4102. [Google Scholar] [CrossRef]
- Kang, J.; Chen, Z.; Zhou, T.; Yang, F.; Chen, J.; Cao, Y.; Xiang, M. Dynamic crystallization and melting behavior of β-nucleated isotactic polypropylene with different melt structures. J. Polym. Res. 2014, 21, 384. [Google Scholar] [CrossRef]
- Blaine, R.L.; Kissinger, H.E. Homer Kissinger and the Kissinger equation. Thermochim. Acta 2012, 540, 1–6. [Google Scholar] [CrossRef]
- Kang, J.; Li, J.; Chen, S.; Zhu, S.; Li, H.; Cao, Y.; Yang, F.; Xiang, M. Hydrogenated petroleum resin effect on the crystallization of isotactic polypropylene. J. Appl. Polym. Sci. 2013, 130, 25–38. [Google Scholar] [CrossRef]
- Kang, J.; Wang, B.; Peng, H.; Li, J.; Chen, J.; Gai, J.; Cao, Y.; Li, H.; Yang, F.; Xiang, M. Investigation on the dynamic crystallization and melting behavior ofβ-nucleated isotactic polypropylene with different stereo-defect distribution-the role of dual-selectiveβ-nucleation agent. Polym. Adv. Technol. 2014, 25, 97–107. [Google Scholar] [CrossRef]
- Kang, J.; Cao, Y.; Li, H.; Li, J.; Chen, S.; Yang, F.; Xiang, M. Influence of the stereo-defect distribution on the crystallization behavior of Ziegler-Natta isotactic polypropylene. J. Polym. Res. 2012, 19. [Google Scholar] [CrossRef]
- Horvath, Z.; Sajo, I.E.; Stoll, K.; Menyhard, A.; Varga, J. The effect of molecular mass on the polymorphism and crystalline structure of isotactic polypropylene. Express Polym. Lett. 2010, 4, 101–114. [Google Scholar] [CrossRef]
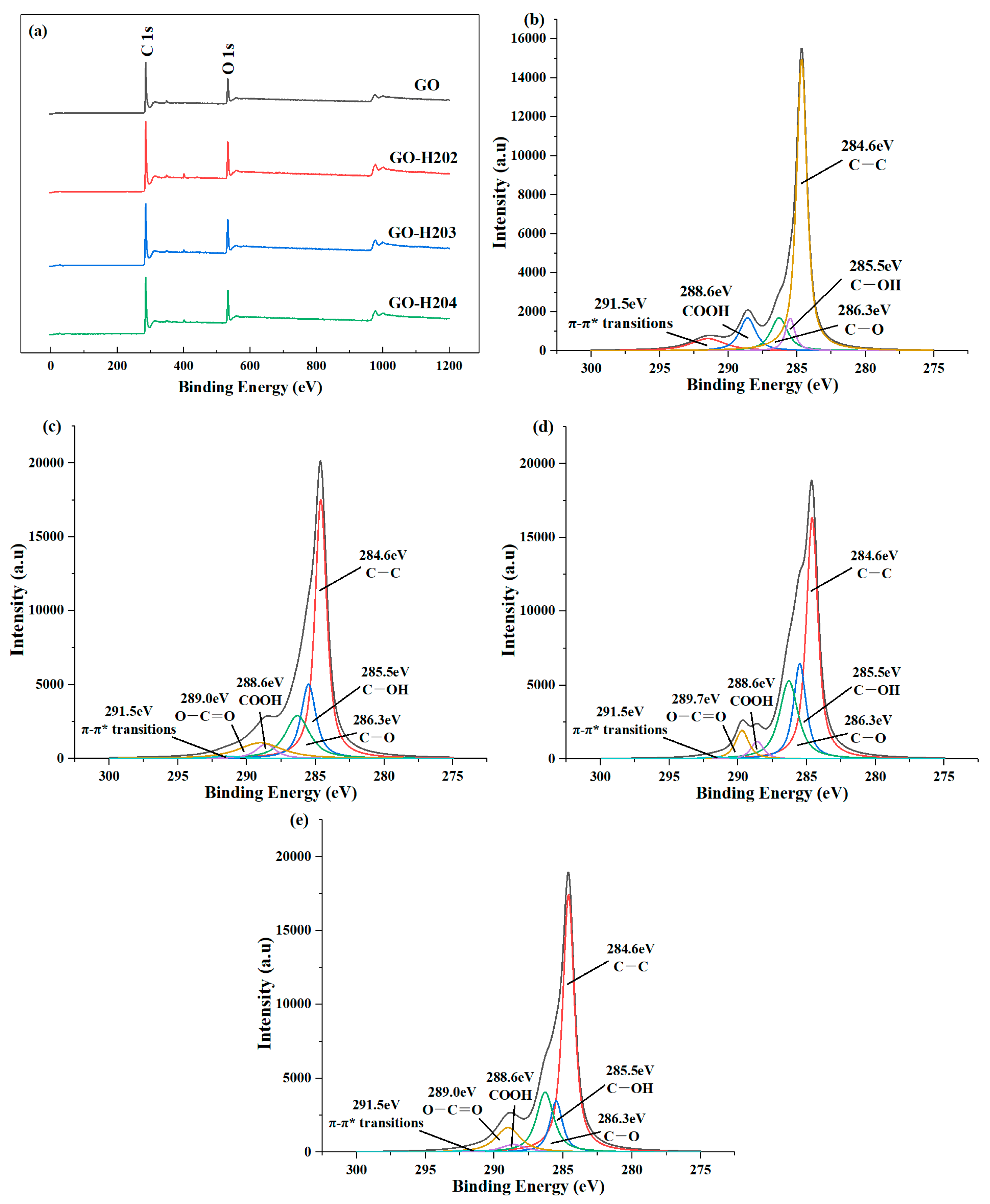

| Sample Name | O 1s (%) | C 1s (%) | C–C (%) | C–OH (%) | C–O (%) | COOH (%) | π-π* (%) | O–C=O (%) |
|---|---|---|---|---|---|---|---|---|
| 284.6 eV | 285.5 eV | 286.3 eV | 288.6 eV | 291.5 eV | 289.0 eV | |||
| GO | 28.6 | 71.4 | 62.3 | 6.4 | 11.5 | 11.0 | 8.8 | 0 |
| GO-H202 | 37.8 | 62.2 | 48.6 | 17.7 | 16.7 | 4.6 | 1.1 | 11.3 |
| GO-H203 | 39.4 | 60.6 | 45.6 | 19.6 | 23.7 | 3.7 | 1.1 | 6.3 |
| GO-H204 | 39.1 | 60.9 | 53.7 | 11.7 | 18.7 | 3.6 | 1.2 | 11.1 |
| Cooling Rate | Sample Name | Tp onset (°C) | Tp (°C) | Tp endset (°C) | Width |
|---|---|---|---|---|---|
| 2 °C/min | iPP/GO | 130.3 | 126.8 | 121.8 | 8.5 |
| iPP/GO-H202 | 130.6 | 127.3 | 125.2 | 5.4 | |
| iPP/GO-H203 | 131.6 | 128.4 | 126.8 | 4.8 | |
| iPP/GO-H204 | 133.8 | 131.3 | 129.3 | 4.5 | |
| 5 °C/min | iPP/GO | 127.3 | 123.6 | 118.7 | 8.6 |
| iPP/GO-H202 | 127.9 | 124.4 | 121.9 | 6 | |
| iPP/GO-H203 | 128 | 125.1 | 122.5 | 5.5 | |
| iPP/GO-H204 | 130.6 | 127.7 | 125.8 | 4.8 | |
| 10 °C/min | iPP/GO | 125.4 | 121 | 116.7 | 8.7 |
| iPP/GO-H202 | 126.8 | 122.4 | 119.5 | 7.3 | |
| iPP/GO-H203 | 126.9 | 123.3 | 120.2 | 6.7 | |
| iPP/GO-H204 | 128 | 125.1 | 122.4 | 5.6 | |
| 20 °C/min | iPP/GO | 123.4 | 117.7 | 112.7 | 10.7 |
| iPP/GO-H202 | 124.1 | 119.6 | 114.9 | 9.2 | |
| iPP/GO-H203 | 124.3 | 119.9 | 115.9 | 8.4 | |
| iPP/GO-H204 | 126.5 | 122.8 | 118.8 | 7.7 | |
| 40 °C/min | iPP/GO | 120.9 | 114.1 | 107.6 | 13.3 |
| iPP/GO-H202 | 121.8 | 116 | 108.6 | 13.2 | |
| iPP/GO-H203 | 123.8 | 117.2 | 110.9 | 12.9 | |
| iPP/GO-H204 | 125.3 | 120.3 | 113.6 | 11.7 |
| Sample | iPP/GO | iPP/GO-H202 | iPP/GO-H203 | iPP/GO-H204 |
|---|---|---|---|---|
| ΔE (kJ/mol) | −309.4 | −350.5 | −353.5 | −368.4 |
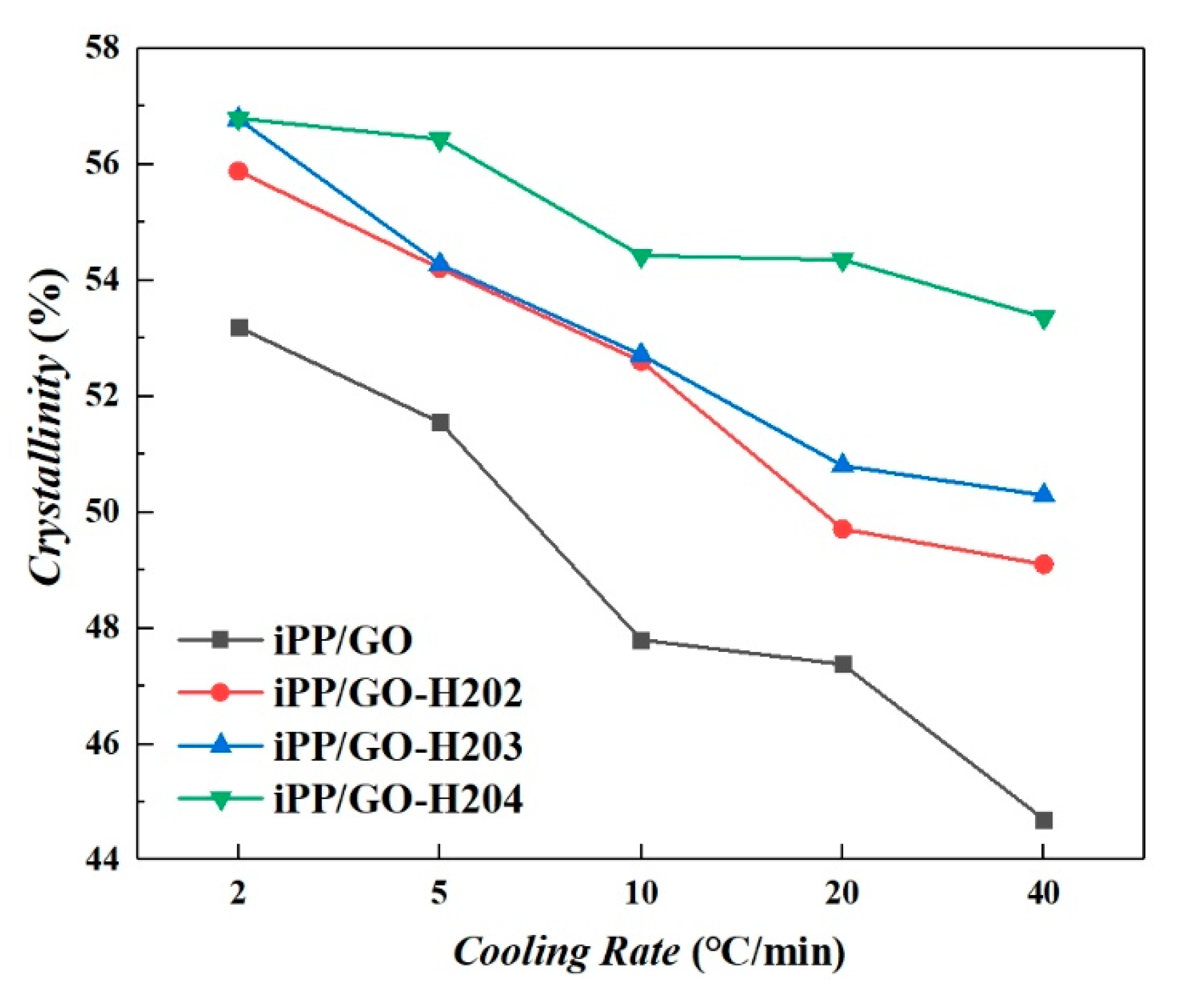
| Cooling Rate | Sample Name | Crystallinities (%) | α-Phase (%) | β-Phase (%) |
|---|---|---|---|---|
| 2 °C/min | iPP/GO | 53.2 | 14.5 | 85.5 |
| iPP/GO-H202 | 55.9 | 28.4 | 71.6 | |
| iPP/GO-H203 | 56.8 | 37.1 | 62.9 | |
| iPP/GO-H204 | 56.8 | 38.6 | 61.4 | |
| 5 °C/min | iPP/GO | 51.6 | 15.5 | 84.5 |
| iPP/GO-H202 | 54.2 | 26.7 | 73.3 | |
| iPP/GO-H203 | 54.3 | 33.6 | 66.4 | |
| iPP/GO-H204 | 56.4 | 37.8 | 62.2 | |
| 10 °C/min | iPP/GO | 47.8 | 21.7 | 78.3 |
| iPP/GO-H202 | 52.6 | 20.8 | 79.2 | |
| iPP/GO-H203 | 52.7 | 29.7 | 70.3 | |
| iPP/GO-H204 | 54.4 | 30.6 | 69.4 | |
| 20 °C/min | iPP/GO | 47.4 | 26.3 | 73.7 |
| iPP/GO-H202 | 49.1 | 27.1 | 72.9 | |
| iPP/GO-H203 | 50.3 | 27.9 | 72.1 | |
| iPP/GO-H204 | 54.4 | 42.8 | 57.2 | |
| 40 °C/min | iPP/GO | 44.7 | 38.6 | 61.4 |
| iPP/GO-H202 | 49.7 | 39.5 | 60.5 | |
| iPP/GO-H203 | 50.8 | 43.0 | 57.0 | |
| iPP/GO-H204 | 53.4 | 49.0 | 50.9 |
| Sample | Crystallinity (%) | Amorphous (%) | αc (%) | βc (%) | Crystalline Sizes L (nm) | |||
|---|---|---|---|---|---|---|---|---|
| α (110) | α (040) | α (130) | β (110) | |||||
| Pure iPP | 48.8 | 51.2 | 13.2 | 86.8 | 10.8 | 8.2 | 7.3 | 32.9 |
| iPP/GO | 58.3 | 41.7 | 17.9 | 82.1 | 11.2 | 8.8 | 8.5 | 28 |
| iPP/GO-H202 | 65.0 | 35.0 | 30.9 | 69.1 | 11.7 | 9.9 | 8.9 | 25.7 |
| iPP/GO-H203 | 66.2 | 33.8 | 39.6 | 60.4 | 12.1 | 9.4 | 9.3 | 26.1 |
| iPP/GO-H204 | 67.3 | 32.7 | 45.3 | 54.7 | 12.5 | 10.0 | 9.3 | 26.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Jiang, X.; Fang, Y.; Chen, J.; Kang, J.; Cao, Y.; Xiang, M. Investigation on the Effect of Hyperbranched Polyester Grafted Graphene Oxide on the Crystallization Behaviors of β-Nucleated Isotactic Polypropylene. Polymers 2019, 11, 1988. https://doi.org/10.3390/polym11121988
Yu Y, Jiang X, Fang Y, Chen J, Kang J, Cao Y, Xiang M. Investigation on the Effect of Hyperbranched Polyester Grafted Graphene Oxide on the Crystallization Behaviors of β-Nucleated Isotactic Polypropylene. Polymers. 2019; 11(12):1988. https://doi.org/10.3390/polym11121988
Chicago/Turabian StyleYu, Yansong, Xi Jiang, Yiwei Fang, Jinyao Chen, Jian Kang, Ya Cao, and Ming Xiang. 2019. "Investigation on the Effect of Hyperbranched Polyester Grafted Graphene Oxide on the Crystallization Behaviors of β-Nucleated Isotactic Polypropylene" Polymers 11, no. 12: 1988. https://doi.org/10.3390/polym11121988
APA StyleYu, Y., Jiang, X., Fang, Y., Chen, J., Kang, J., Cao, Y., & Xiang, M. (2019). Investigation on the Effect of Hyperbranched Polyester Grafted Graphene Oxide on the Crystallization Behaviors of β-Nucleated Isotactic Polypropylene. Polymers, 11(12), 1988. https://doi.org/10.3390/polym11121988






