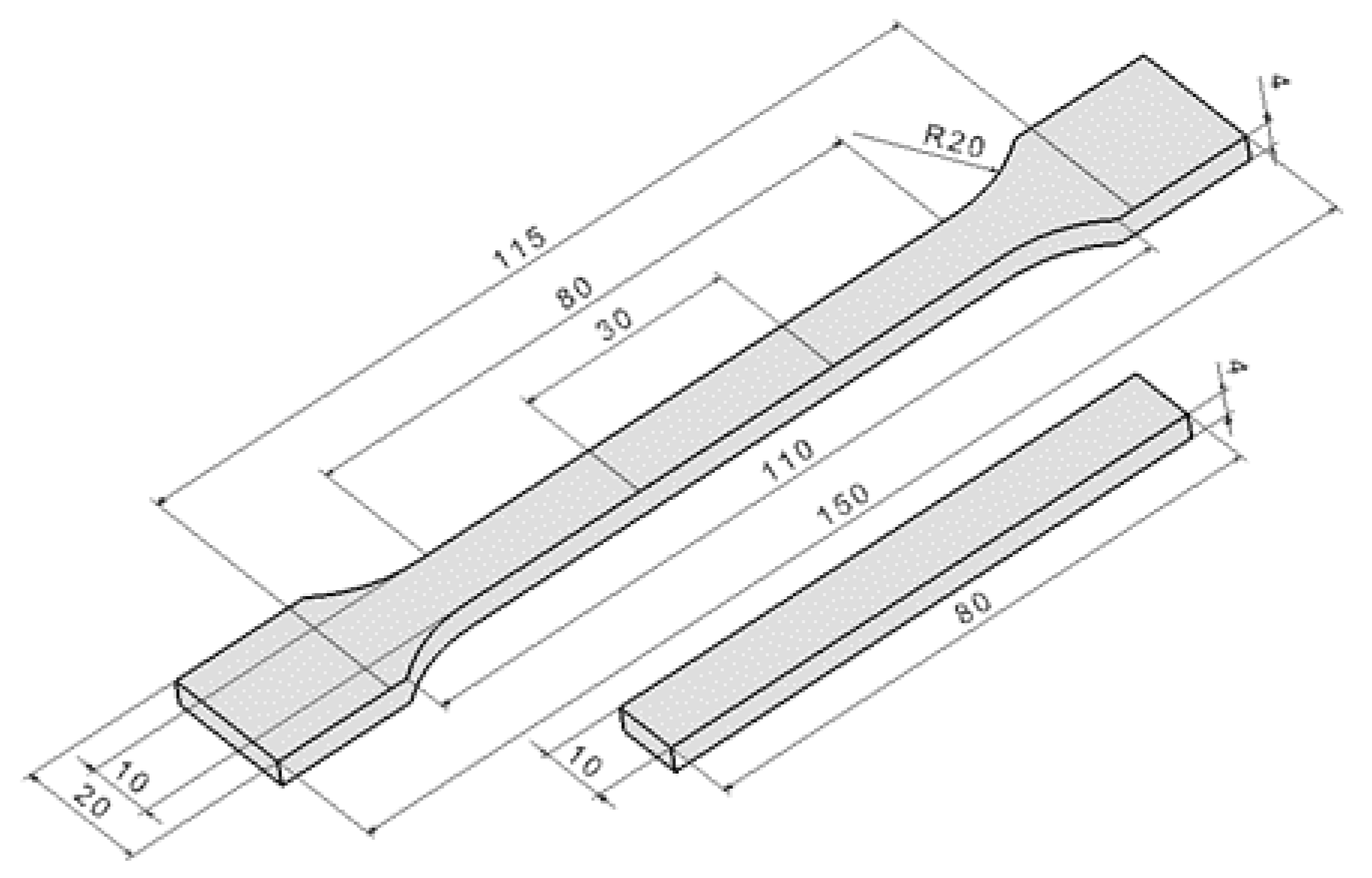
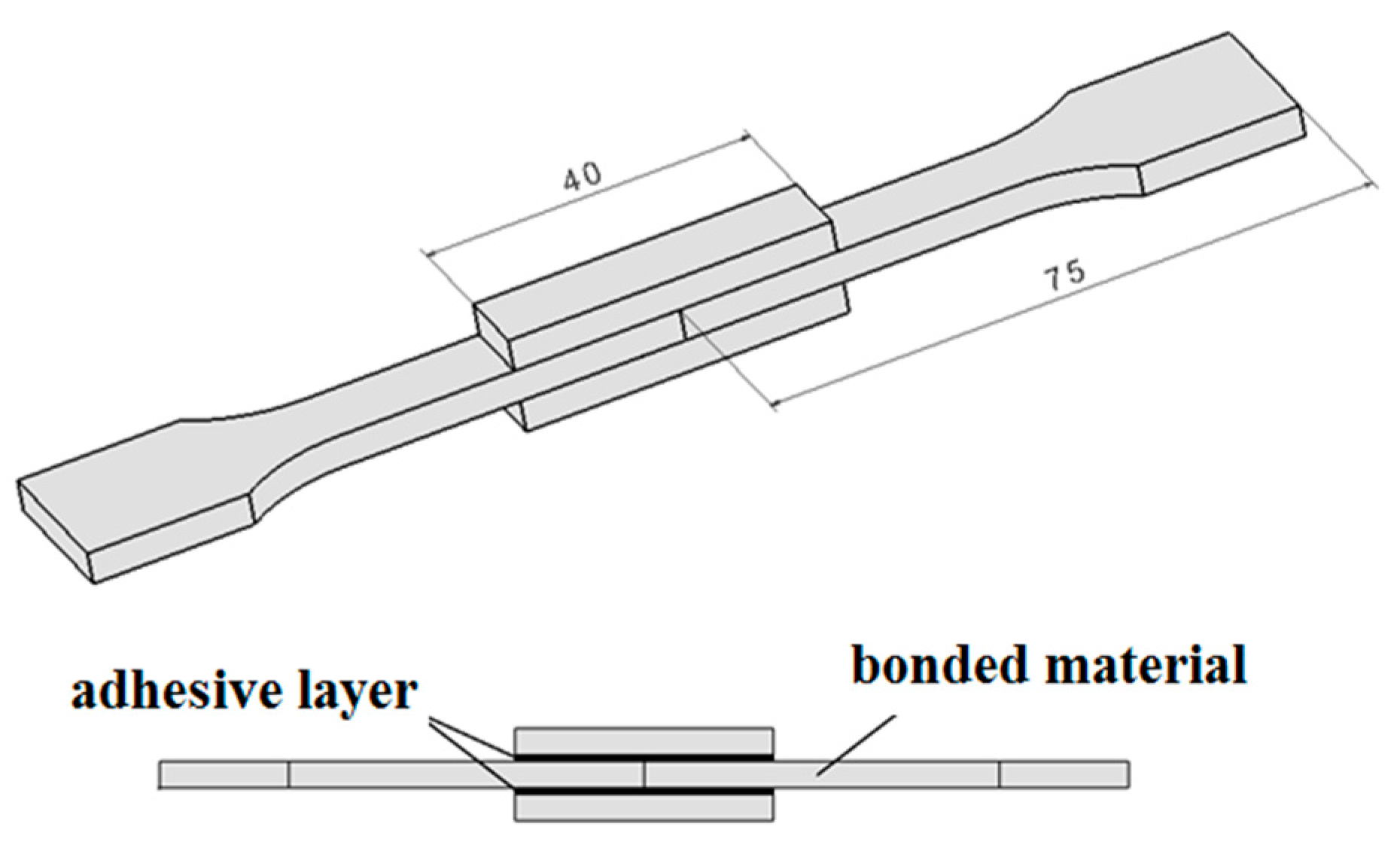
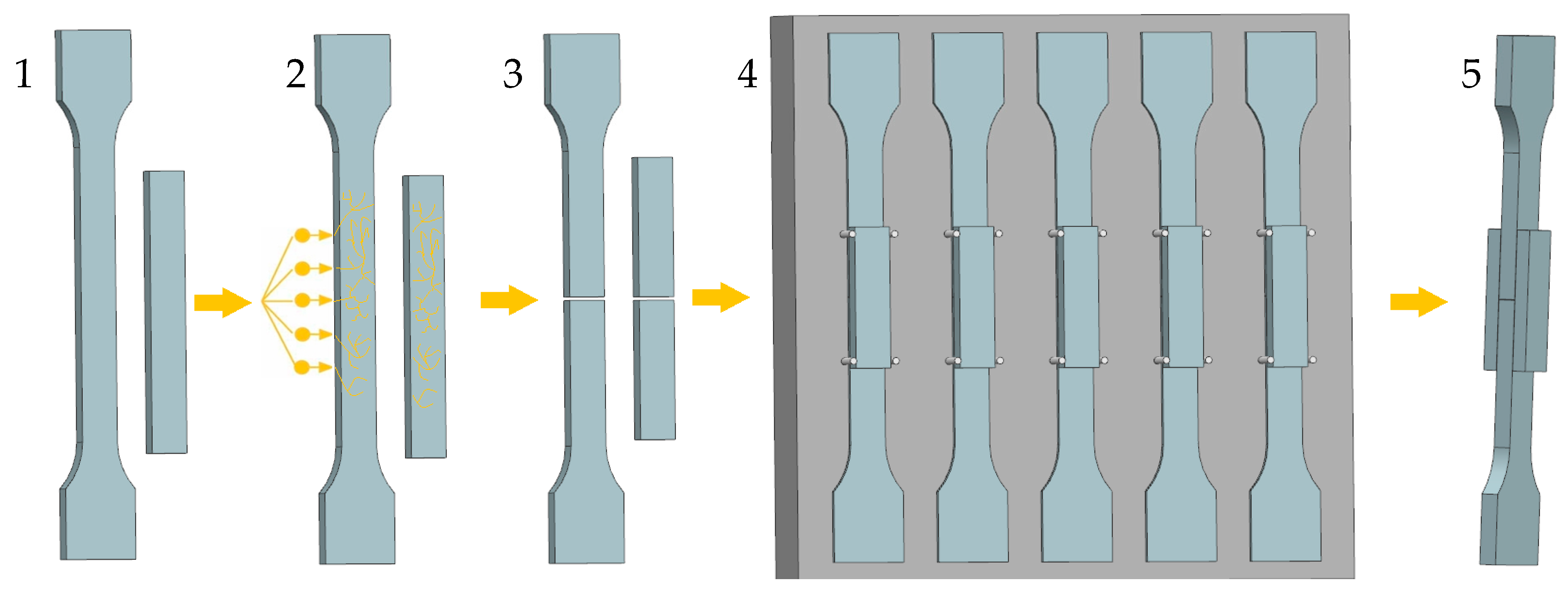
3.2. Surface-Layer Properties
The effect of β
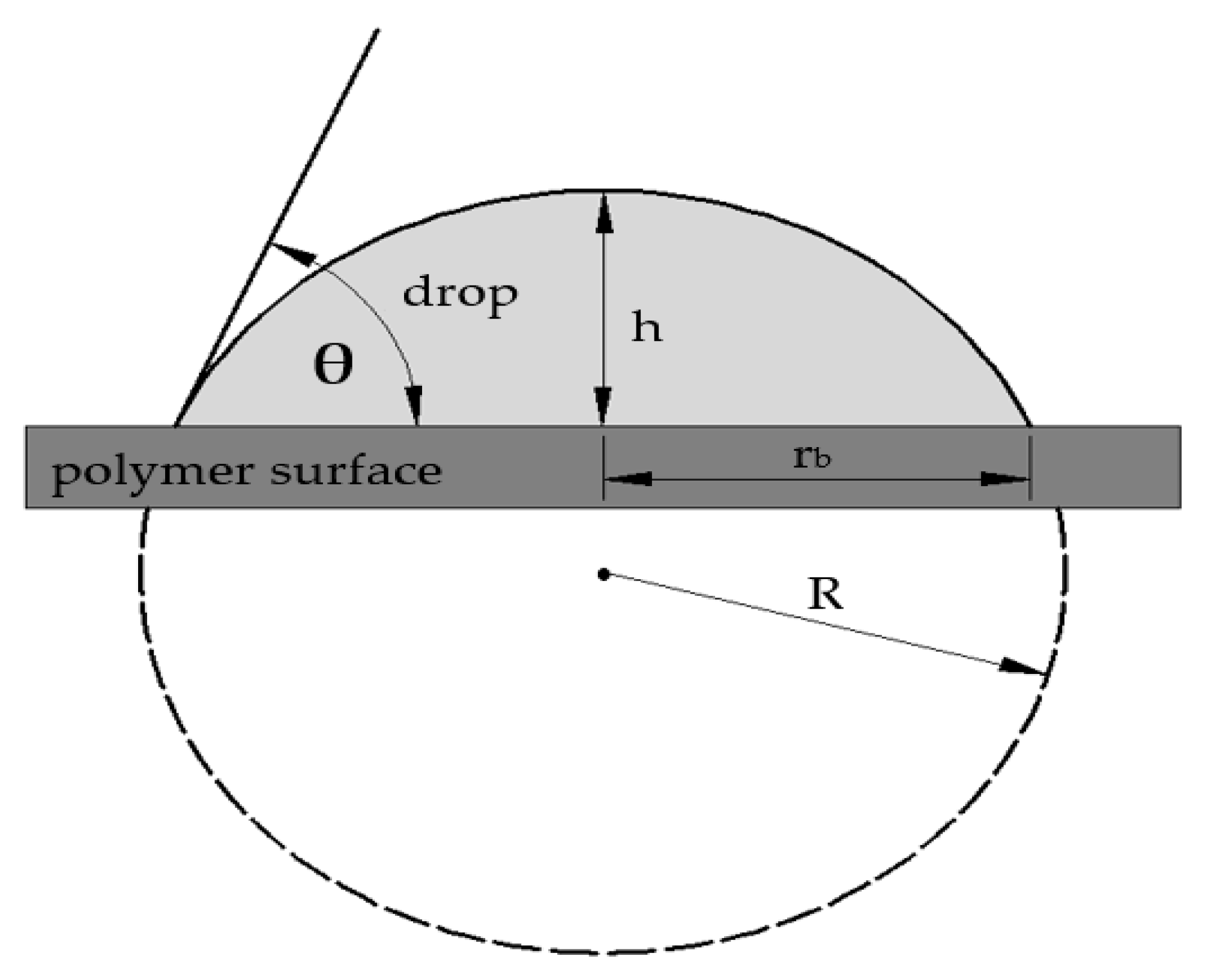
− radiation on surface properties was assessed by using the wetting contact angles, free surface energy, and relative numbers of carbonyl and hydroxyl groups. The changes in wetting contact angles of HDPE, LDPE, and PP are presented in
Table 4 and
Figure 7.
The measurement results indicated that β− radiation significantly improved the wettability of all tested materials, which was reflected by the decrease in the wetting contact angles for all the reference liquids used. The most appropriate radiation dose for HDPE and LDPE was 165 kGy, which was associated with the most significant decrease in wetting contact angle of approximately 31% to 53% compared with the unmodified material, depending on the type of liquid used. Further increasing the radiation dose no longer caused the wetting contact angles to decrease.
A significant drop in the wetting contact angle of PP was recorded at the radiation dose of 33 kGy. However, the lowest wetting contact angles were achieved at the irradiation dose of 66 kGy: 59.5°, 55.9°, and 36.6° for distilled water, glycerin, and ethylene glycol, respectively. Depending on the type of liquid used, these angles were approximately 32% to 43% smaller than those of non-irradiated PP.
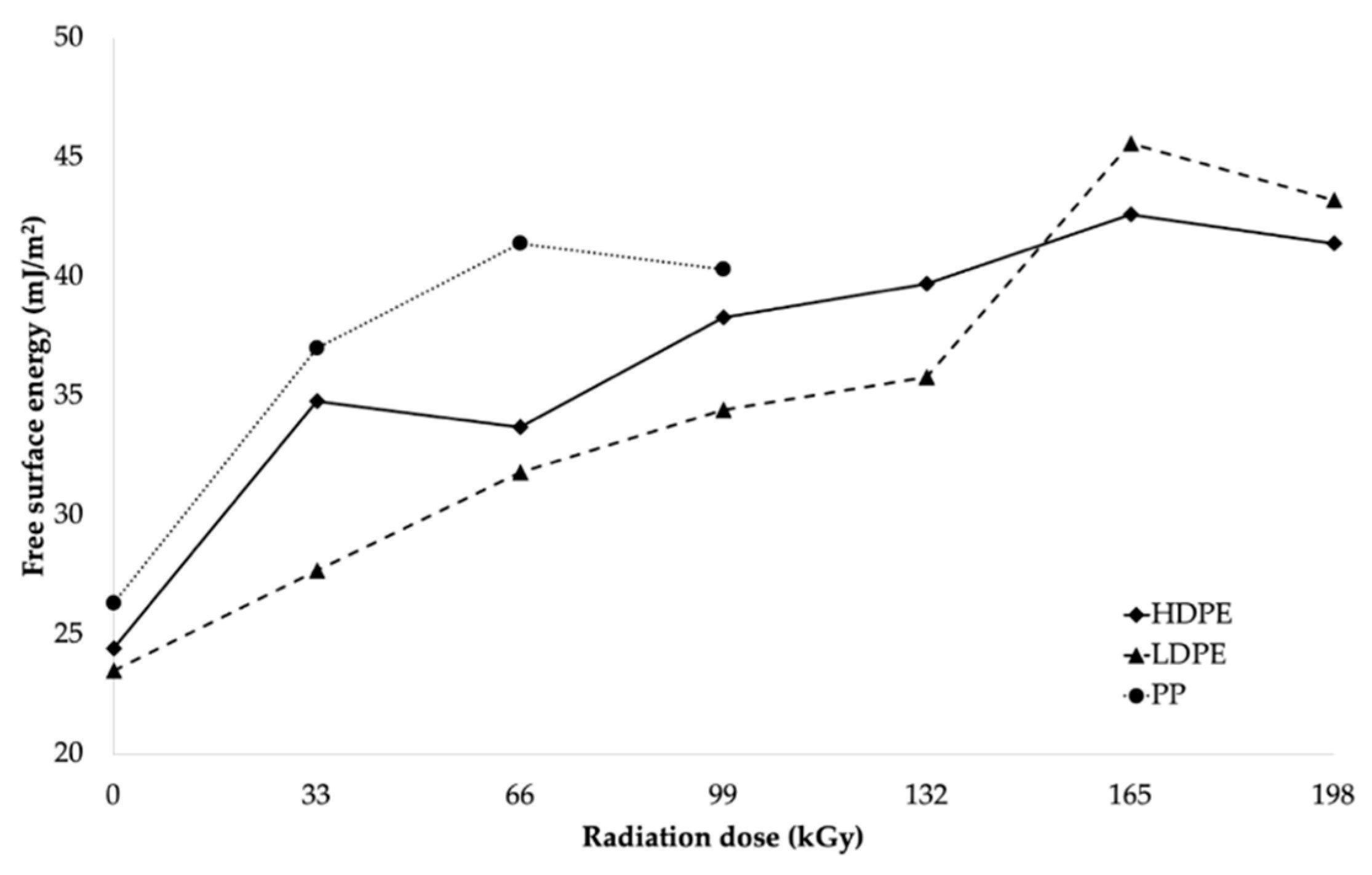
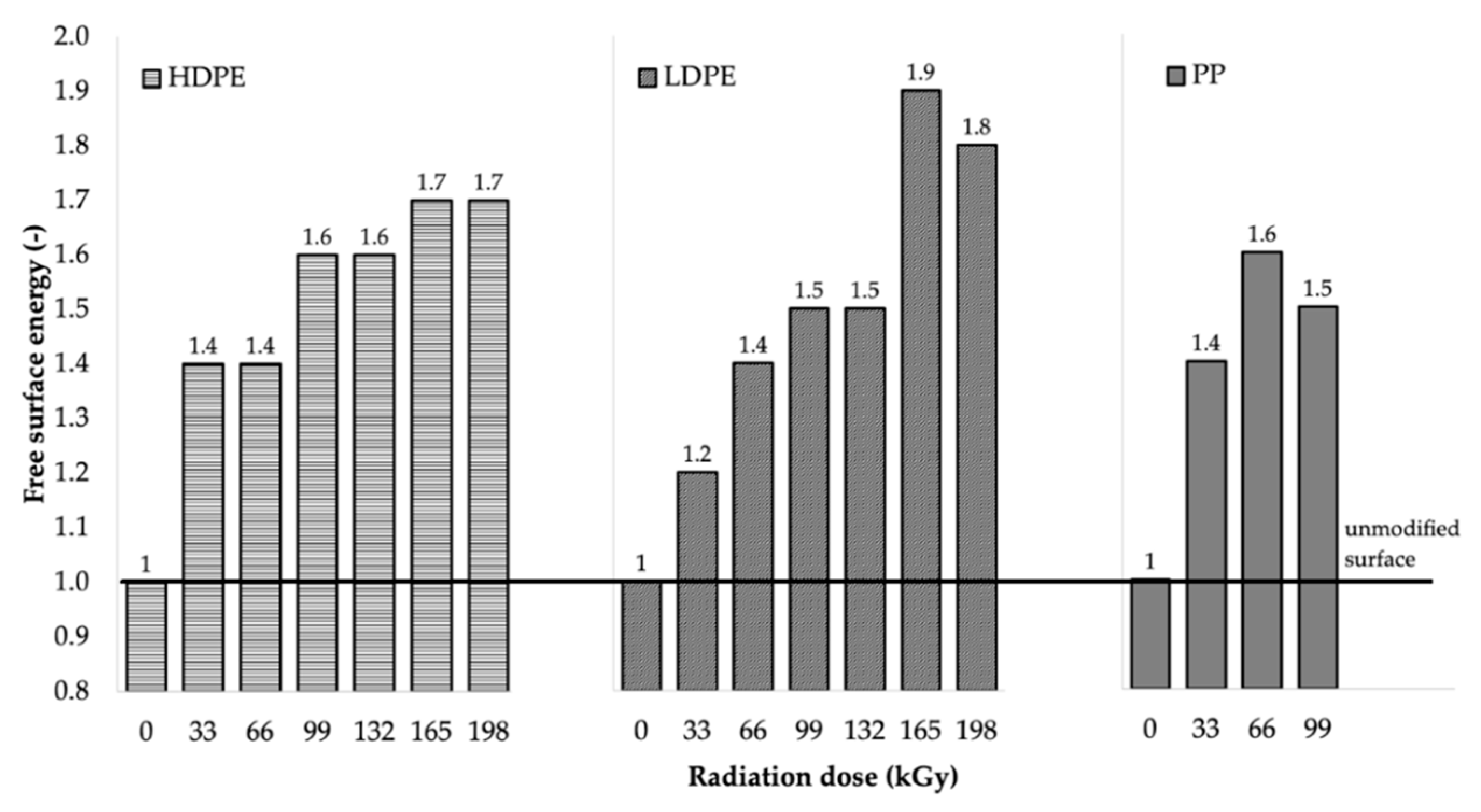
The decrease in wetting contact angles was also closely related to the change in free surface energy of the tested materials, as illustrated in
Table 5 and
Figure 8.
Table 5 summarizes the values of the free surface energy (γ
s), along with its polar (γ
p) and displacement (γ
d) components, as calculated using the OWRK method [
42,
43,
44].
After comparing the results, it can be stated that the free surface energy increased as the radiation dose increased, which resulted in the significant improvement in the adhesion properties of the surface layers of the investigated materials. From the free surface energy viewpoint, the most suitable radiation dose for LDPE and HDPE was considered to be 165 kGy, since it led to the highest increase in free surface energy for both HDPE and LDPE. The free surface energy of HDPE increased from 24.4 mJ/m
2 (for the non-irradiated material) to 42.6 mJ/m
2 (for the material irradiated using the radiation dose of 165 kGy), which represented an increase of approximately 75%. Irradiation increased the free surface energy of LDPE by approximately 94%, from 23.5 to 45.6 mJ/m
2. The radiation dose of 66 kGy was the most appropriate for PP, and it increased its free surface energy from 26.3 to 41.4 mJ/m
2, which represented an increase of approximately 57%. The free surface energy values (see
Table 5 and
Figure 8) were in good agreement with the measured wetting contact angle values.
The materials used in this study, HDPE, LDPE, and PP, are low-surface-energy polymeric materials [
2,
4]. From the obtained results, modification by using β
− radiation can be considered to be an effective method for achieving high surface energies for the studied polymers. According to the literature [
4], the high energy surface of polymeric materials must have energy higher than 40 mJ/m
2. All tested materials met this condition after being irradiated, using their respective optimal radiation doses (see
Table 5).
In addition to the total free-surface-energy value, its polar component also plays a significant role in the resultant adhesion. The influence of the dose of absorbed radiation on the polar component of the free surface energy of all materials tested is illustrated in
Figure 9. β
− radiation significantly increased the value of the polar component of the free surface energy. Not unlike the other tested properties, the highest increase in the polar component of the free surface energy for HDPE and LDPE was achieved by using the radiation dose of 165 kGy. The polar component of the free surface energy of HDPE increased from 5.4 to 27.2 mJ/m
2, which was an increase of approximately 404%. Moreover, irradiation increased the polar component of the free surface energy of LDPE from 5.8 to 33.4 mJ/m
2, which was an increase of approximately 476%. The most appropriate radiation dose for PP appeared to be 66 kGy. This increased its polar component of the free surface energy from 5.2 to 27.5 mJ/m
2, which represented an increase of approximately 429%.
In addition to wettability and free surface energy, the presence of reactive functional groups, such as carbonyl and hydroxyl, on the surface of the bonded material can significantly affect the ultimate tensile strength of bonded joints [
1,
2,
5,
20]. Infrared spectroscopy was used to determine the contributions of these groups to the ultimate tensile strength of bonded joints.
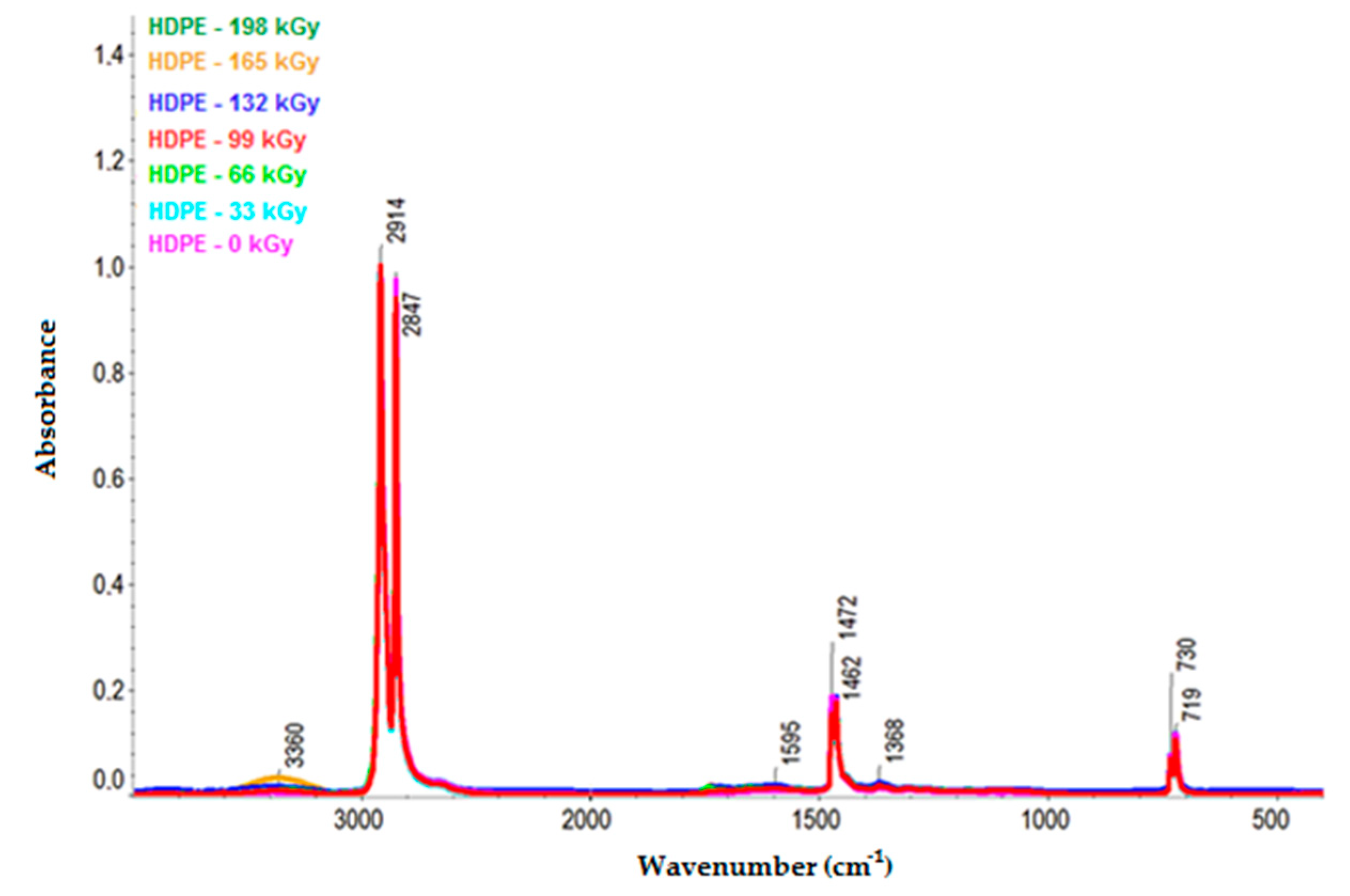
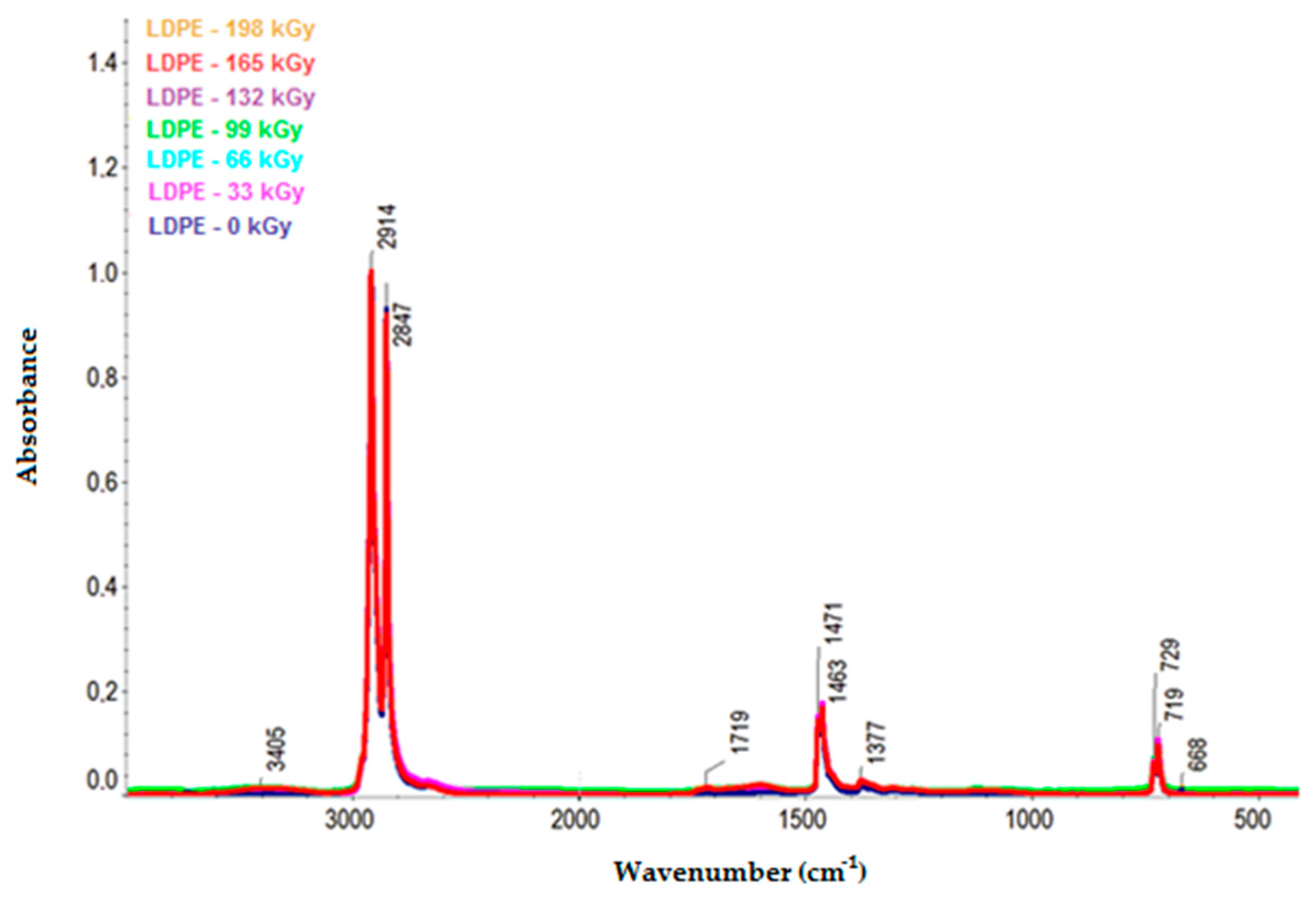
Figure 10 and
Figure 11 illustrate the infrared spectra of HDPE and LDPE, respectively. The HDPE spectra depict the decrease in intensity of the bands ascribed to aliphatic CH bonds (e.g., the negative bands at 3000–2800, ~1450, and ~1370 cm
−1) and the increase in intensity of the bands corresponding to oxygenated functional groups at 1595 and 3360 cm
−1. Furthermore, the LDPE spectra illustrate the increase in bandwidth of the oxygen-functional groups at 1719 and 3405 cm
−1.
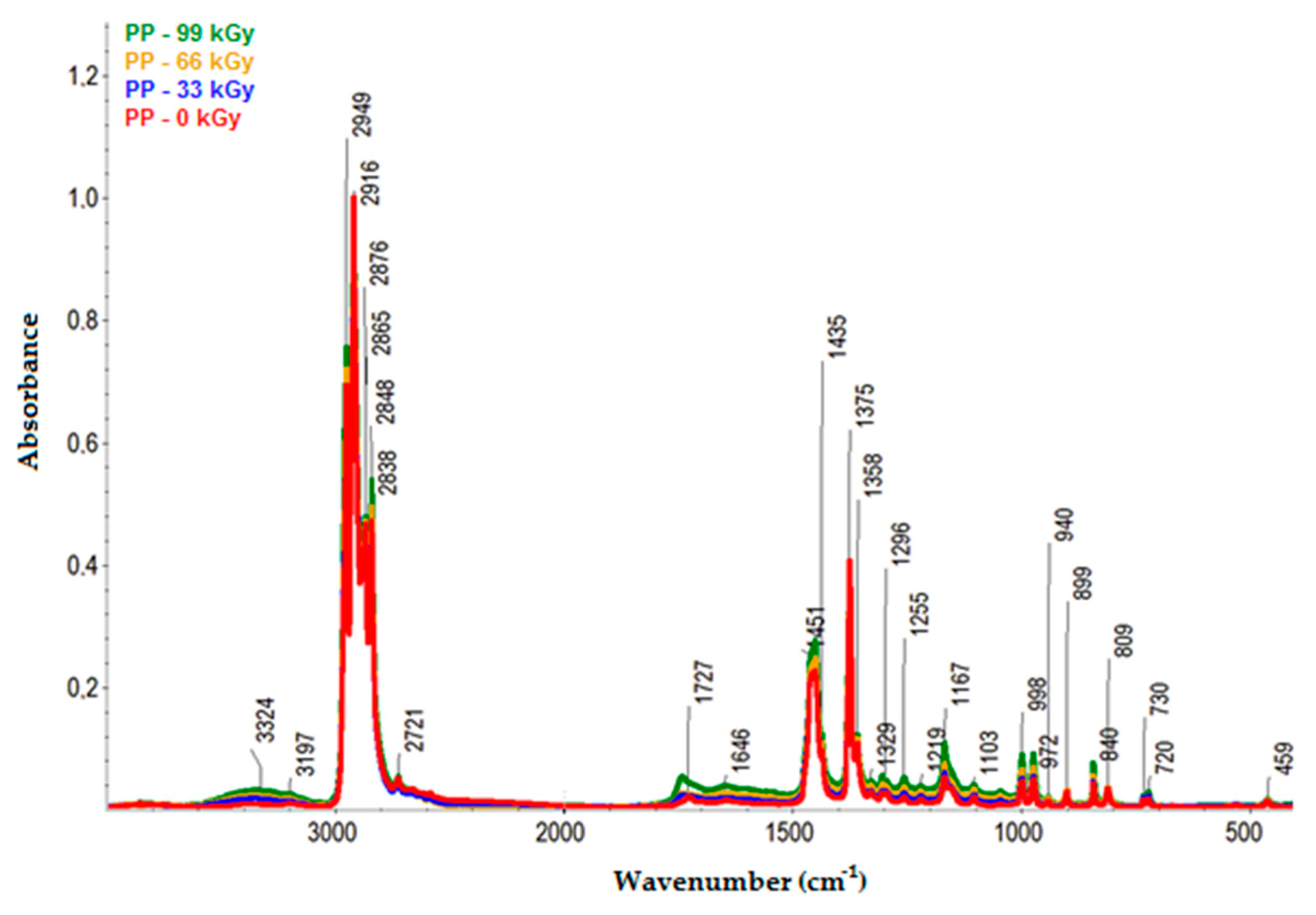
The infrared spectra of PP are presented in
Figure 12. Like HDPE and LDPE, since PP was oxidized during irradiation, hydroxyl (3600–3000 cm
−1 range), and carbonyl (1800–1500 cm
−1 range) groups were identified in its spectra.
From the infrared spectra, we concluded that irradiation caused the oxidation of CH bonds (–CH
2–) in HDPE, LDPE, and PP, and carbonyl (–CO–) and hydroxyl (–OH) groups were formed as a result. The increase in the relative numbers of oxygen functional groups is presented in
Figure 13 and
Figure 14 (for hydroxyl and carbonyl groups, respectively).
From
Figure 13 and
Figure 14, it was concluded that the relative numbers of hydroxyl and carbonyl groups in all tested materials depended on the dose of absorbed radiation. The lowest relative number of hydroxyl groups was recorded for the non-irradiated materials. The relative number of oxygen-containing groups increased as the radiation dose increased. The highest increase in the relative number of oxygen-containing groups for HDPE and LDPE was observed for the radiation dose of 165 kGy, while the radiation dose that generated the highest increase in the relative number of oxygen-containing groups for PP was 99 kGy (see
Figure 13).
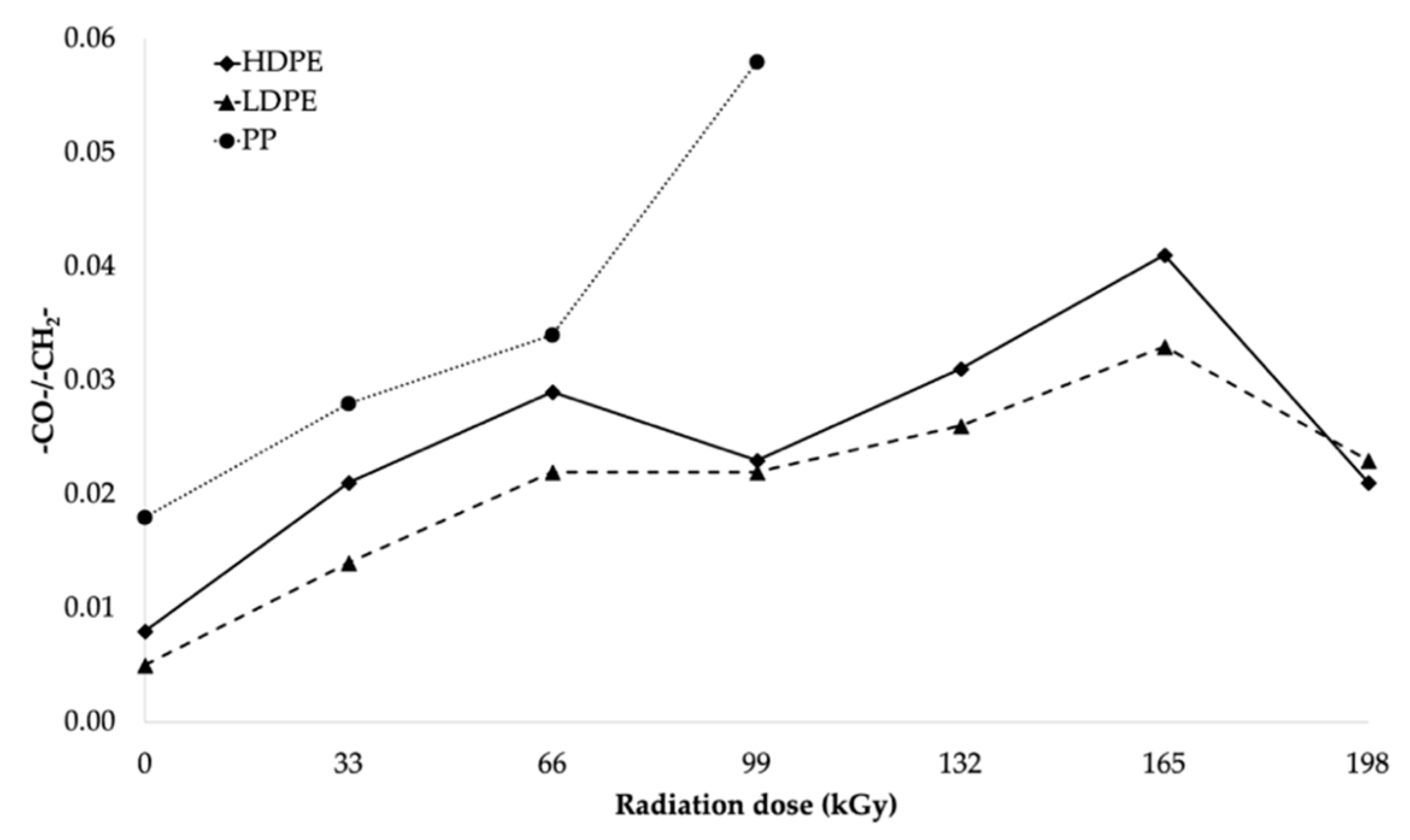
The trends in the relative number of carbonyl and hydroxyl groups (see
Figure 14 and
Figure 13, respectively) were similar. The relative number of carbonyl groups increased as the radiation dose increased. The highest increase in the relative number of oxygen-containing groups for HDPE and LDPE was observed at 165 kGy, while the highest increase for PP was observed at 99 kGy (see
Figure 14).
Similar to the changes in wetting contact angles and free surface energy, the changes in the properties of the surface layers of all tested materials were probably due to oxidation which occurred during and after beta irradiation. Oxidation is one of the secondary reactions that occurs when ionizing radiation interacts with a polymer [
21], and it results in the formation of the abovementioned carbonyl- and hydroxyl-functional groups. The kinetic processes that cause the degradation of these products are most likely controlled by atmospheric oxygen diffusion throughout the bulk polymer.
Alkyl and allyl radicals reacted with oxygen molecules to form carbonyl groups on the surfaces of samples, while, within the samples, the alkyl radicals led to crosslinking and the allyl radicals reacted with oxygen to form hydroperoxide groups. These findings were in agreement with the results reported by Murray [
47] and Hama [
48]. In addition to the oxidation, which affects the surface properties of polymers, polymer properties are also influenced by post-irradiation oxidation. Carpentieri [
49] and Costa [
50] mentioned that post-irradiation oxidation is one of the factors that influences the crystallinity and size of crystalline lamellas of polymers. This could explain, to a certain extent, some of the differences in the relative numbers of oxygenated functional groups of HDPE and LDPE (shown in
Figure 13 and
Figure 14, respectively).
A lower relative number of oxygenated functional groups was observed for LDPE, which presented lower crystallinity than HDPE. These results could be attributed to the differences in reactivity between macro-alkyl radicals, which could form in the amorphous and crystalline phases of the polymers during irradiation. Rivaton [
51] suggested that the macro-alkyl radicals that formed in the amorphous phase decayed very rapidly. Nevertheless, other macro-allyl radicals could migrate from the crystalline phase at the amorphous/crystalline interface, where they would become more accessible to oxygen, which would subsequently lead to the development of post-irradiation oxidation.
Therefore, it can be concluded that, for LDPE, post-irradiation oxidation was very low and was achieved in a relatively short time. The lamellae of LDPE are very thin lamellae compared to those of HDPE, and therefore the migration time of the macro-alkyl radicals from the crystalline to the amorphous phase was very short. Conversely, the oxidation rate of HDPE was higher, and it was reached after a relatively long time after the irradiation process. The wider crystalline lamellae of HDPE captured the macro-allyl radicals, and, thus, their migration time to the amorphous phase was longer. Hence, the migration time of the radicals from the crystalline to the amorphous phase appeared to be one of the key factors governing the oxidation process.
3.3. Load-Bearing Capacity of Bonded Joints
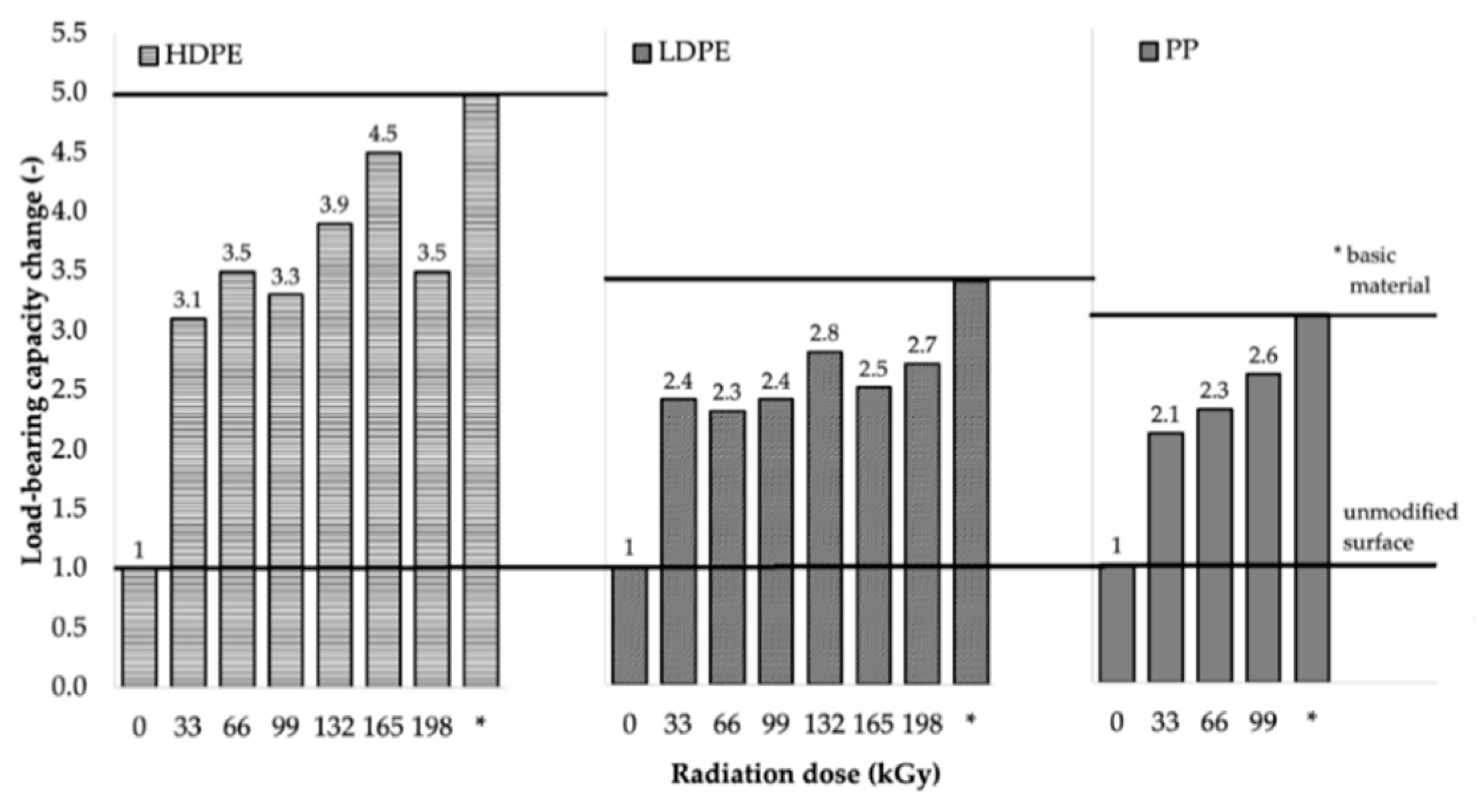
Using the measured surface-layer properties, it was concluded that beta irradiating HDPE, LDPE, and PP caused the oxidation of their surface layers, which resulted in improved wettability, increased free surface energy, and increased the relative oxygen content of the surface. Therefore, these changes became the main factors that positively influenced the adhesion properties of the tested materials, which manifested in the extreme increase of the bond strength of the bonded joints.
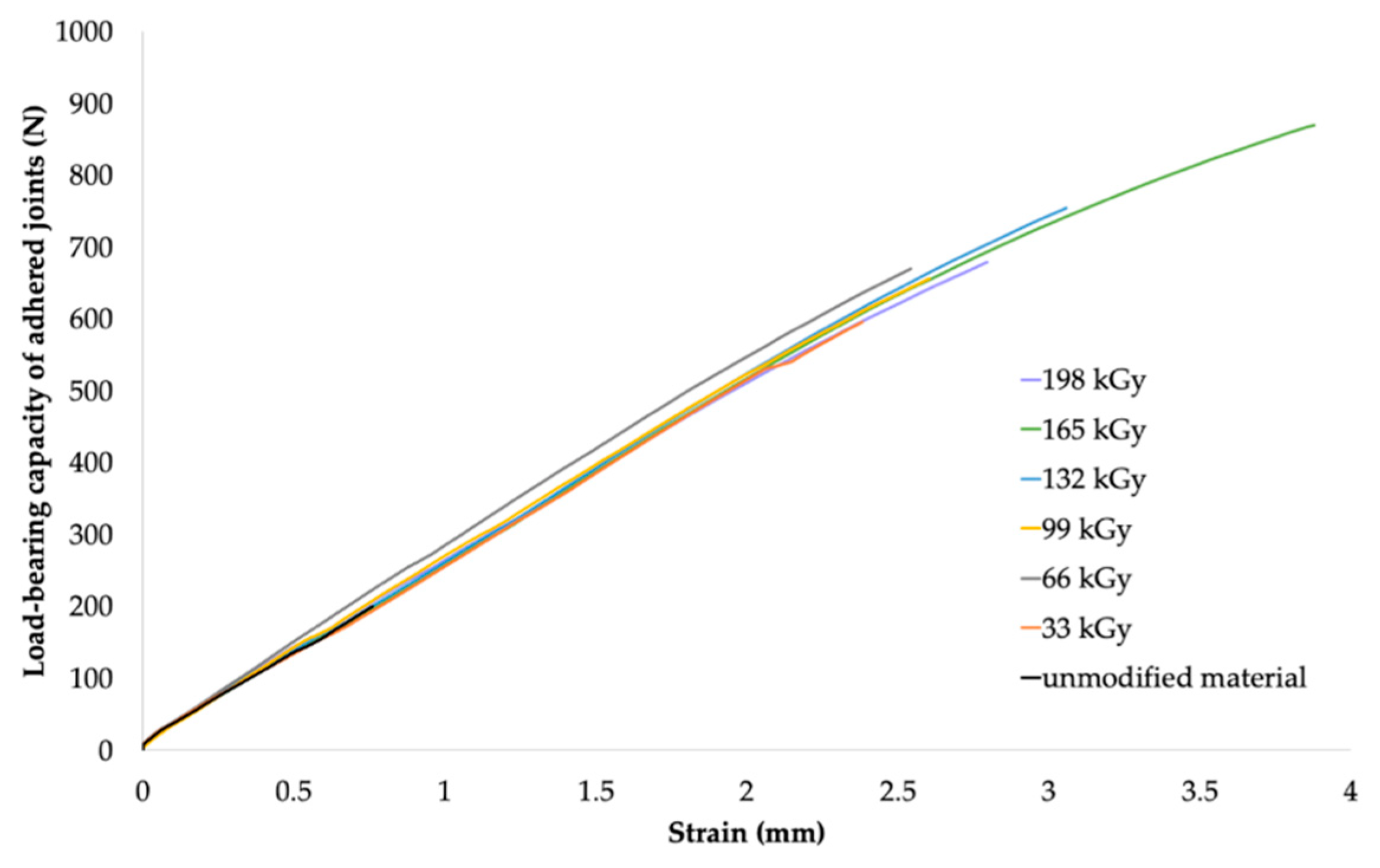
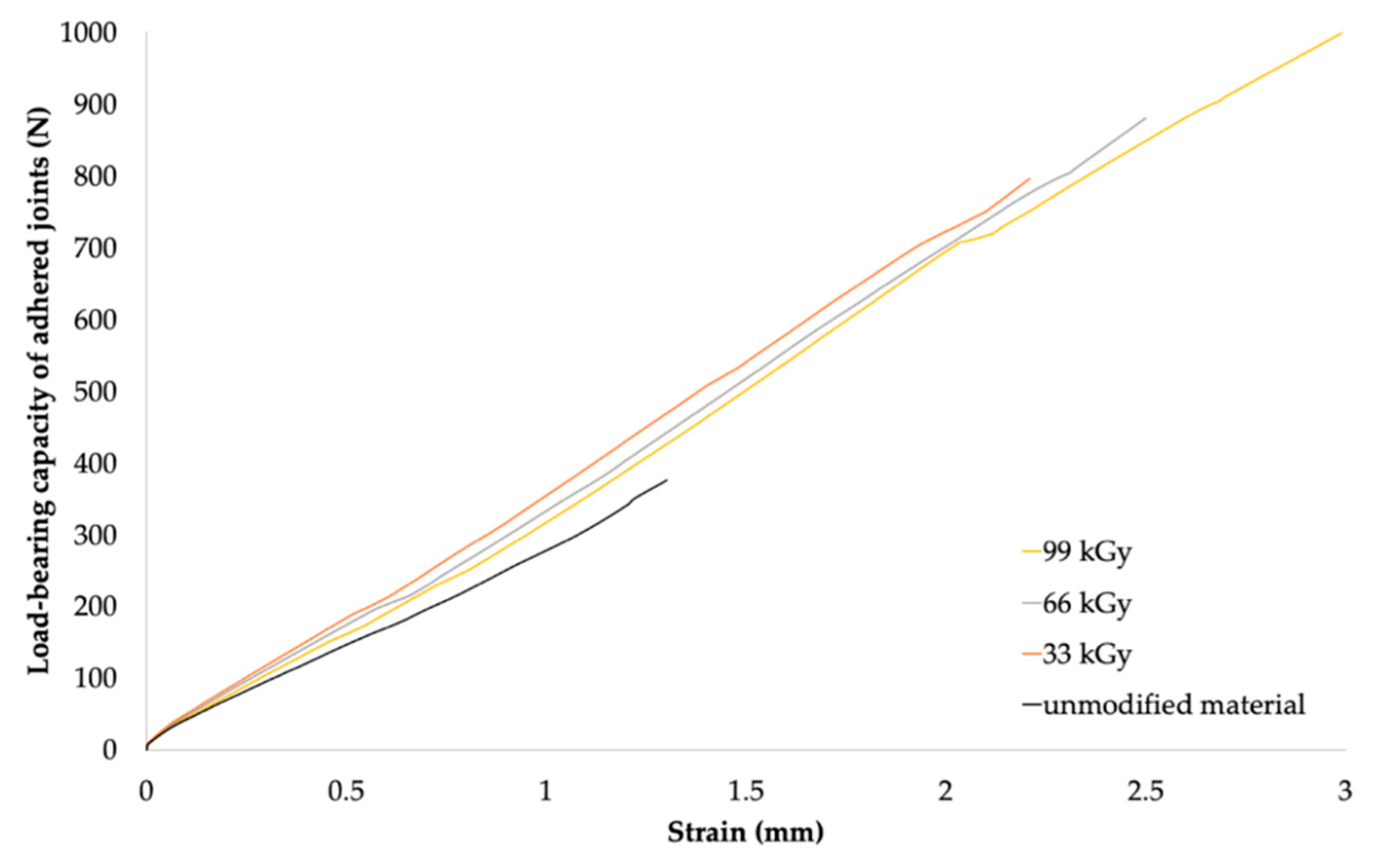
The maximum load forces that the bonded joints were able to transfer were measured to assess the load-bearing capacity of the bonded joints. The load-bearing capacities of the bonded joints of HDPE, LDPE, and PP at different absorbed radiation doses are summarized in
Table 6,
Table 7 and
Table 8, respectively. The results are presented as arithmetic means (
), and the corresponding measurement errors are presented using the standard error of the mean (
).
From the results for HDPE and LDPE (shown in
Table 6 and
Table 7, respectively), it was concluded that, from the perspective of the adhesive bonds, higher radiation doses (99–198 kGy) were required for the materials that exhibited the highest increase in load-bearing strength. The maximum adhesive bond strength of HDPE (see
Table 6) was observed when Cyberbond 2028 adhesive was used at the radiation dose of 165 kGy. For such a bonded joint, the average value of the maximum load-bearing force increased from 185.1 N (for the non-irradiated material) to 840.6 N (for the material irradiated using the dose of 165 kGy), which represented an increase of approximately 354%. The most suitable combination of Cyberbond 2008 adhesive and 198 kGy radiation dose was obtained for LDPE (see
Table 7), but from a technical viewpoint, the optimal radiation dose was 165 kGy. For such a bonded joint, the average maximum load-bearing force increased from 111.8 to 375.6 N, which represented an increase of approximately 236% (at the radiation dose of 198 kGy), and from 111.8 to 372.2 N, which represented an increase of 232% at the lower radiation dose of 165 kGy.
For PP (see
Table 8), it cannot be unequivocally stated that higher radiation doses were associated with the highest increase in the average maximum load-bearing force, since extreme increases have also been achieved at the lowest radiation dose. For this type of material, the maximum bond strength of the bonded adhesive was observed by using the combination of Cyberbond 2028 adhesive and radiation dose of 99 kGy, when the average maximum load-bearing force increased from 388.8 to 1001.6 N, which represented an increase of approximately 158%. However, the highest increase in the average maximum load-bearing force of 440% was obtained using the two-component methacrylate adhesive Plexus MA300 and irradiation dose of 66 kGy.
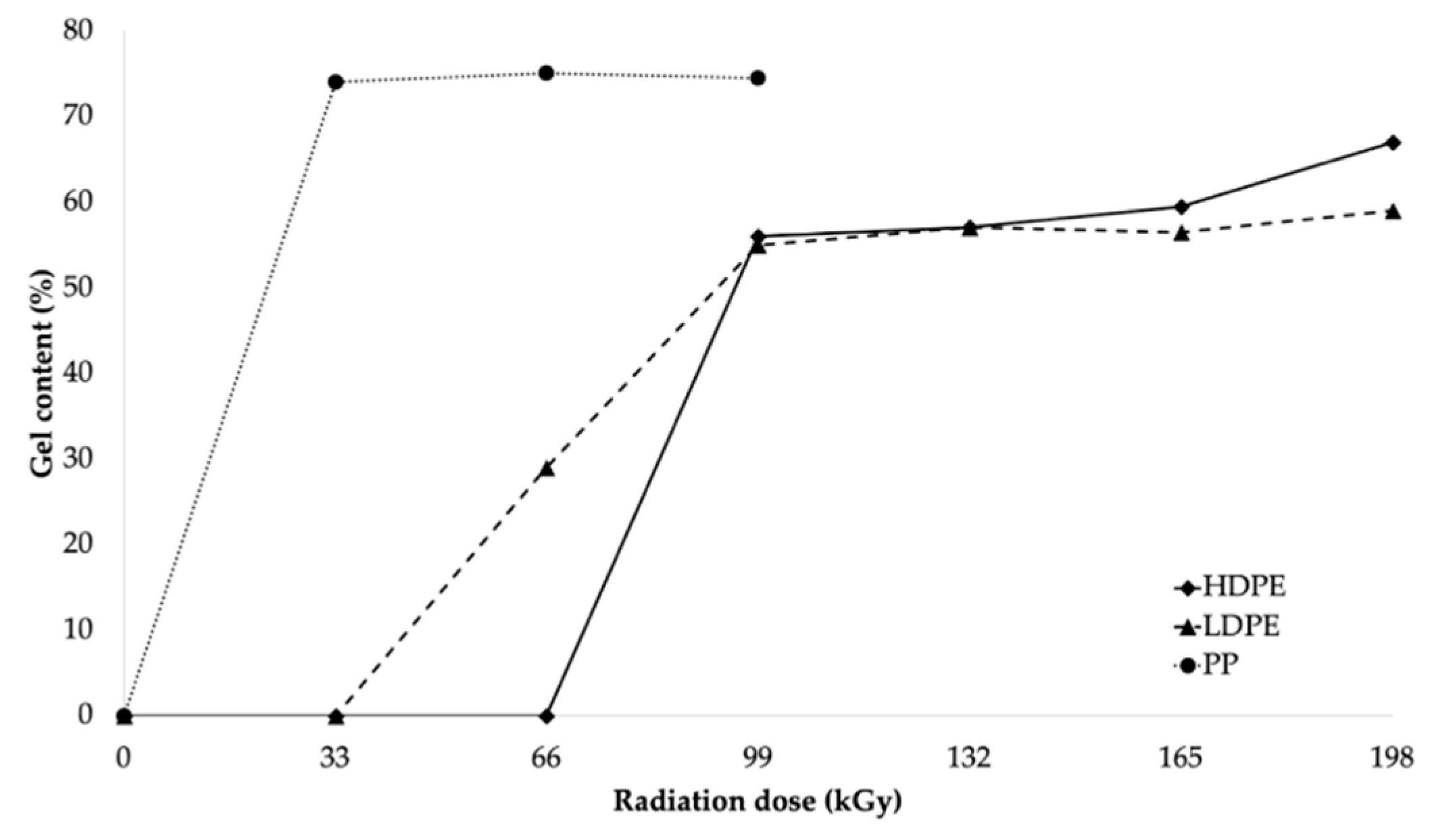
The results of the load-bearing capacity of the bonded joints were in agreement with those of the surface-layer-properties tests. Furthermore, the results were in agreement with the measured gel content, where the gel content of LDPE and HDPE was reported to be minimal at lower radiation doses (see
Figure 6), while PP presented an extreme increase in its gel content at the lowest radiation dose (see
Figure 6). These results indicated that β
− radiation appeared to be a very effective tool for increasing the load-bearing capacity of bonded HDPE, LDPE, and PP joints.