XPS and FTIR Studies of Polytetrafluoroethylene Thin Films Obtained by Physical Methods
Abstract
1. Introduction
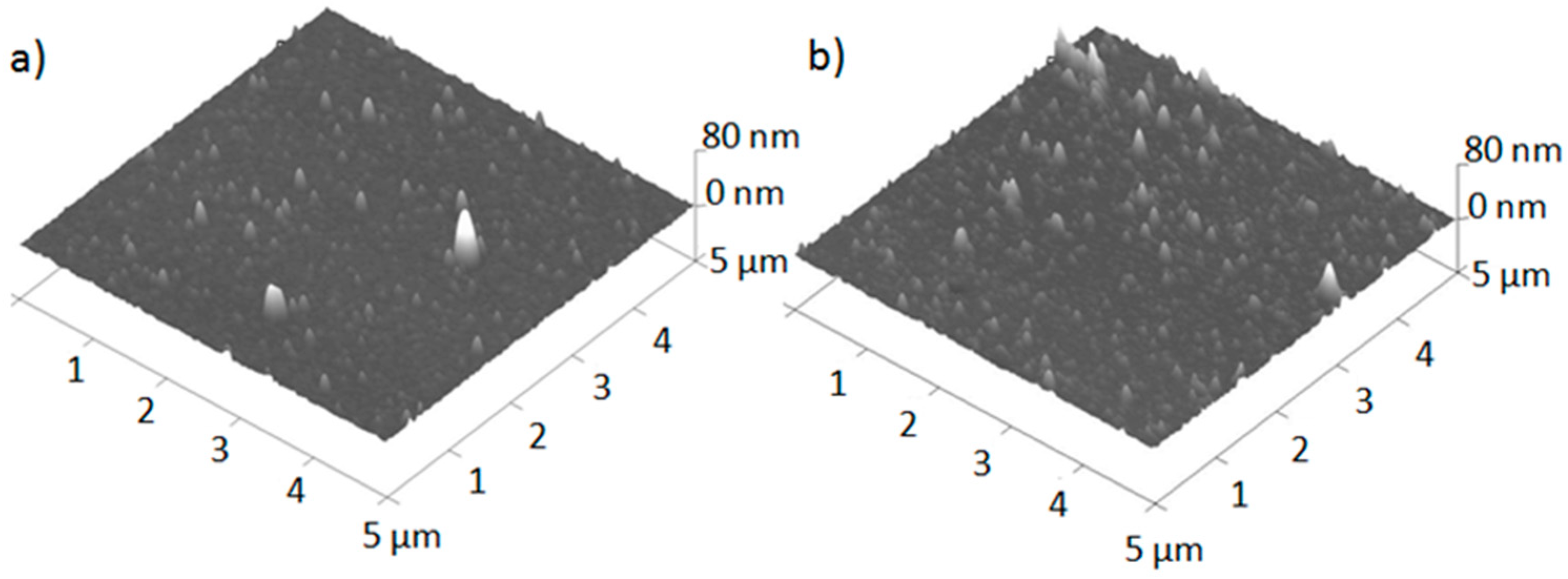
- From 10 to 200 nm, probably originating from vapor phase particle growth.
- From 0.5 to 10 µm feathery chains, which are made of aggregated type-I particles.
- Approximately 1 µm ejecta; compact and very symmetric particles, known as droplets.
- Above 4 µm ejecta; fused, open-structure “popcorn-like” material that is the result of the spallation of target “grains” (singly or in clusters), triggered by the shock wave generated by the original thermal pulse accompanying absorption.
2. Materials and Methods
2.1. Film Preparation
2.2. Film Characterization
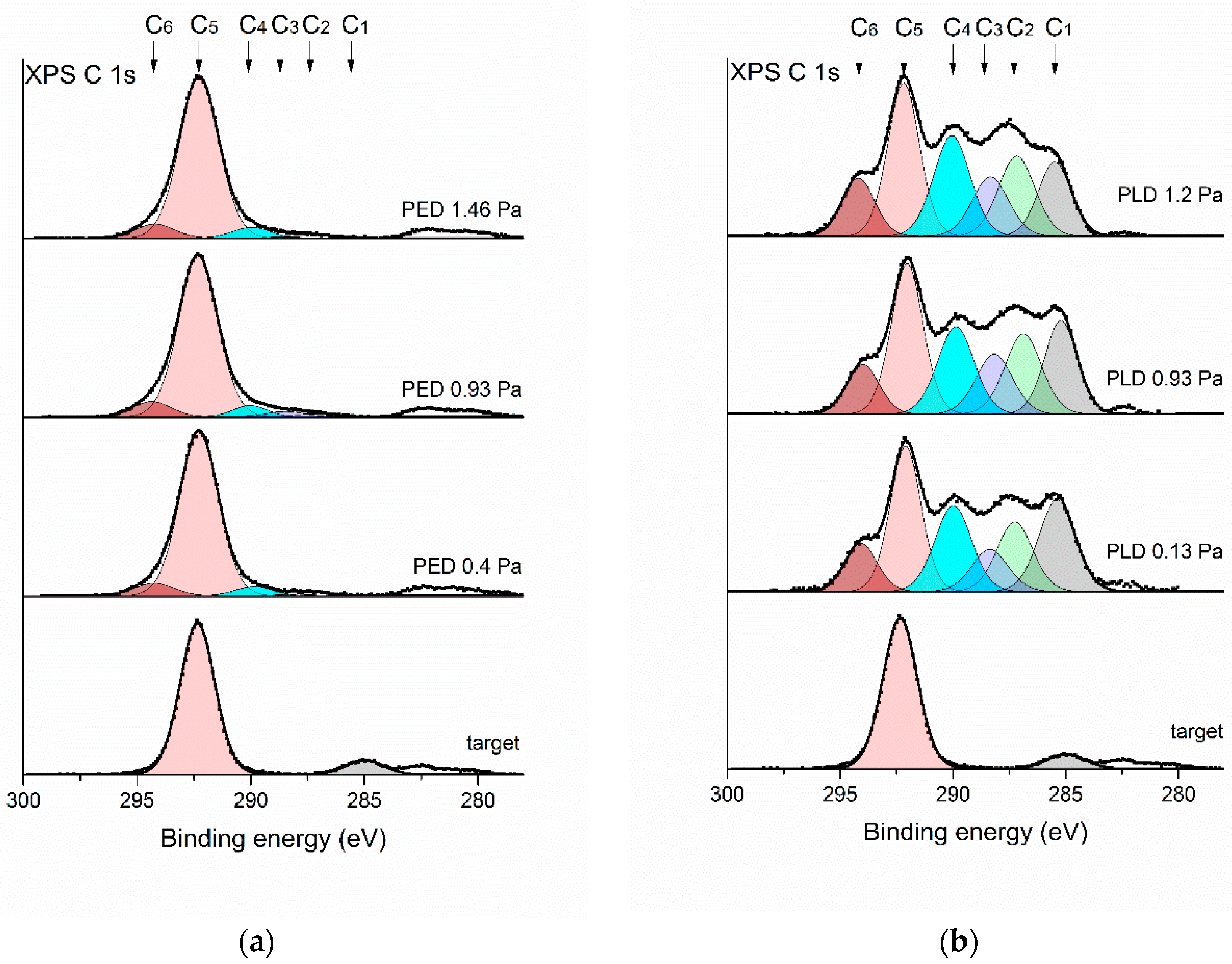
3. Results
3.1. Surface Morphology and Thickness
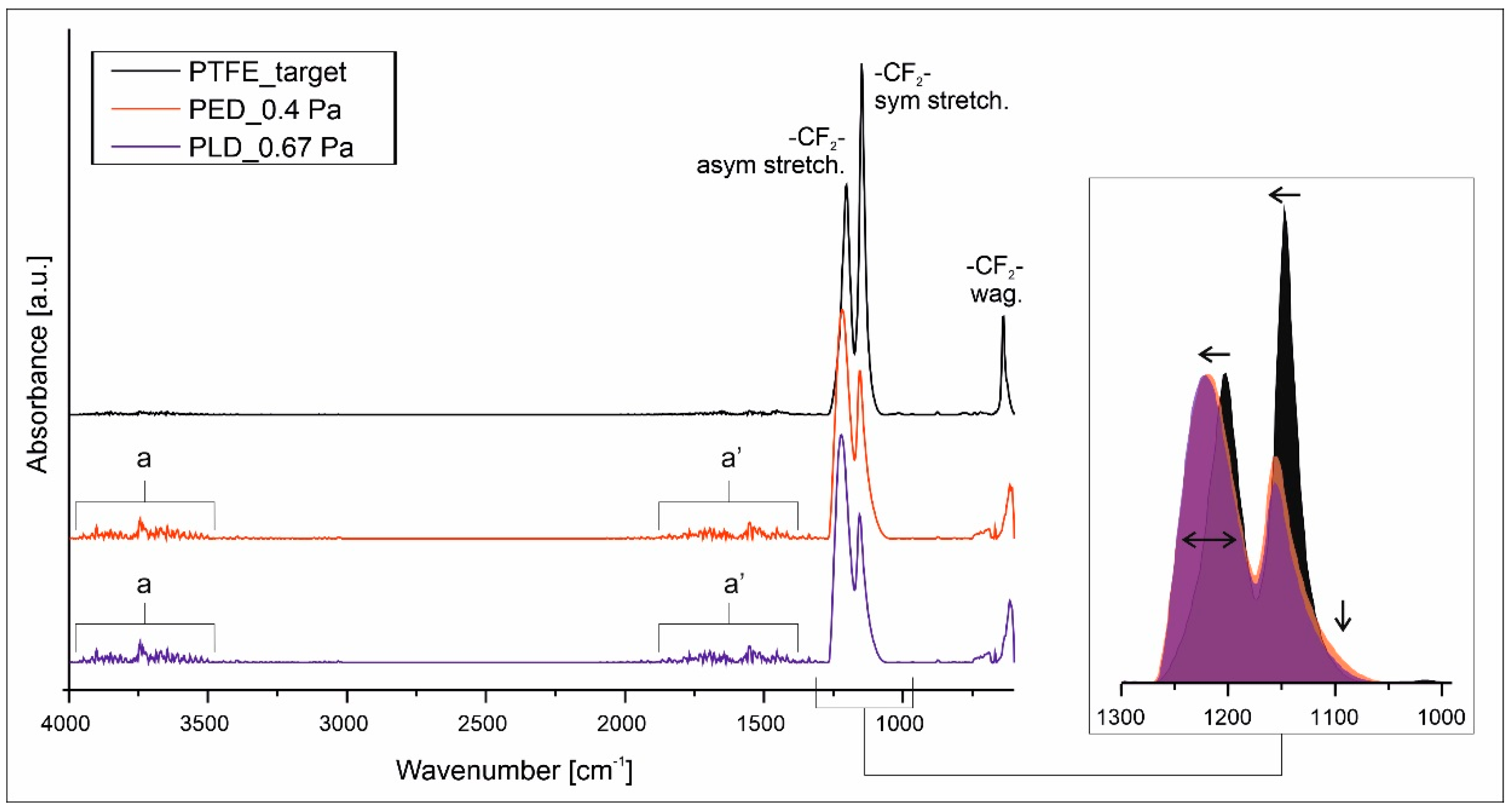
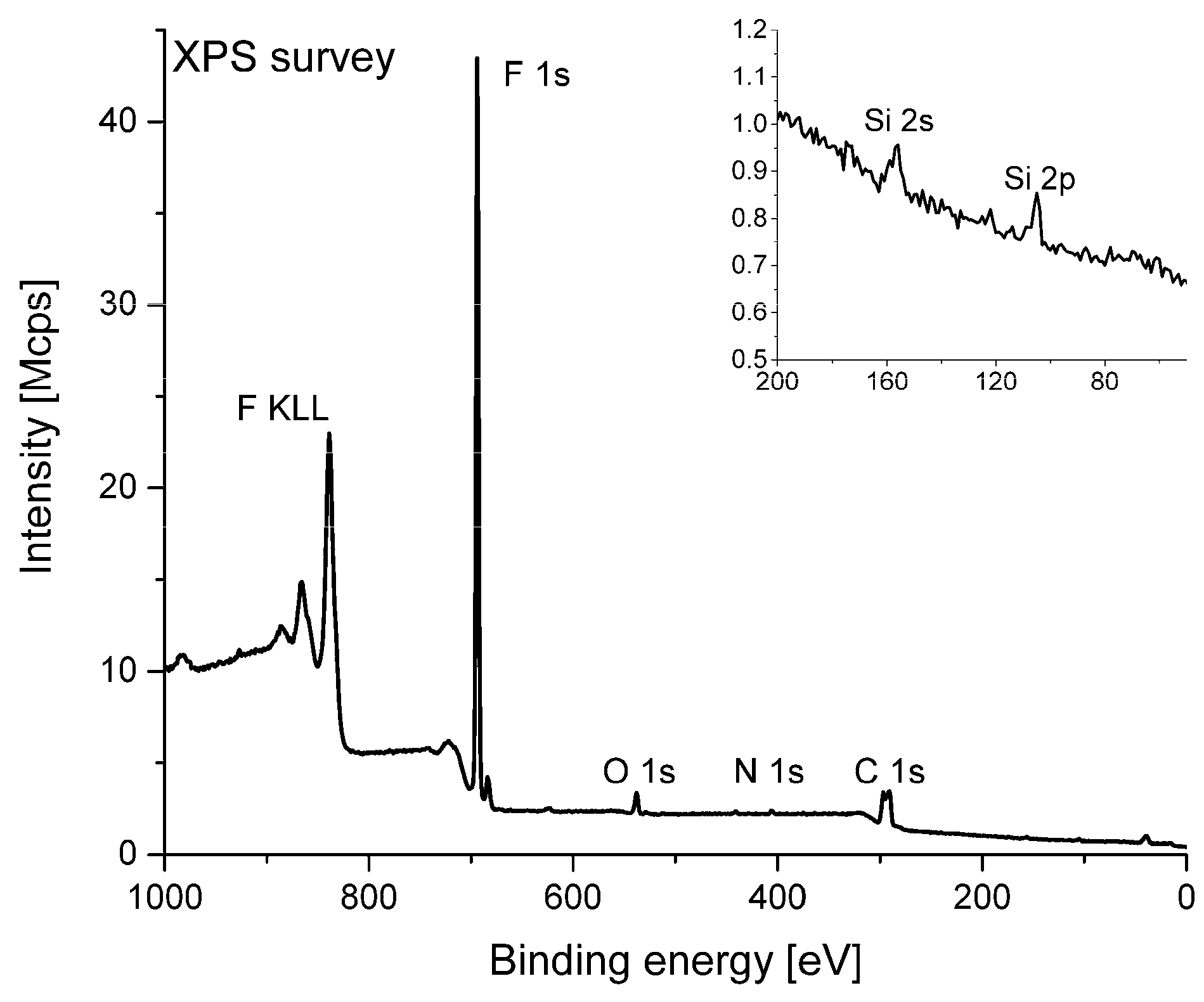
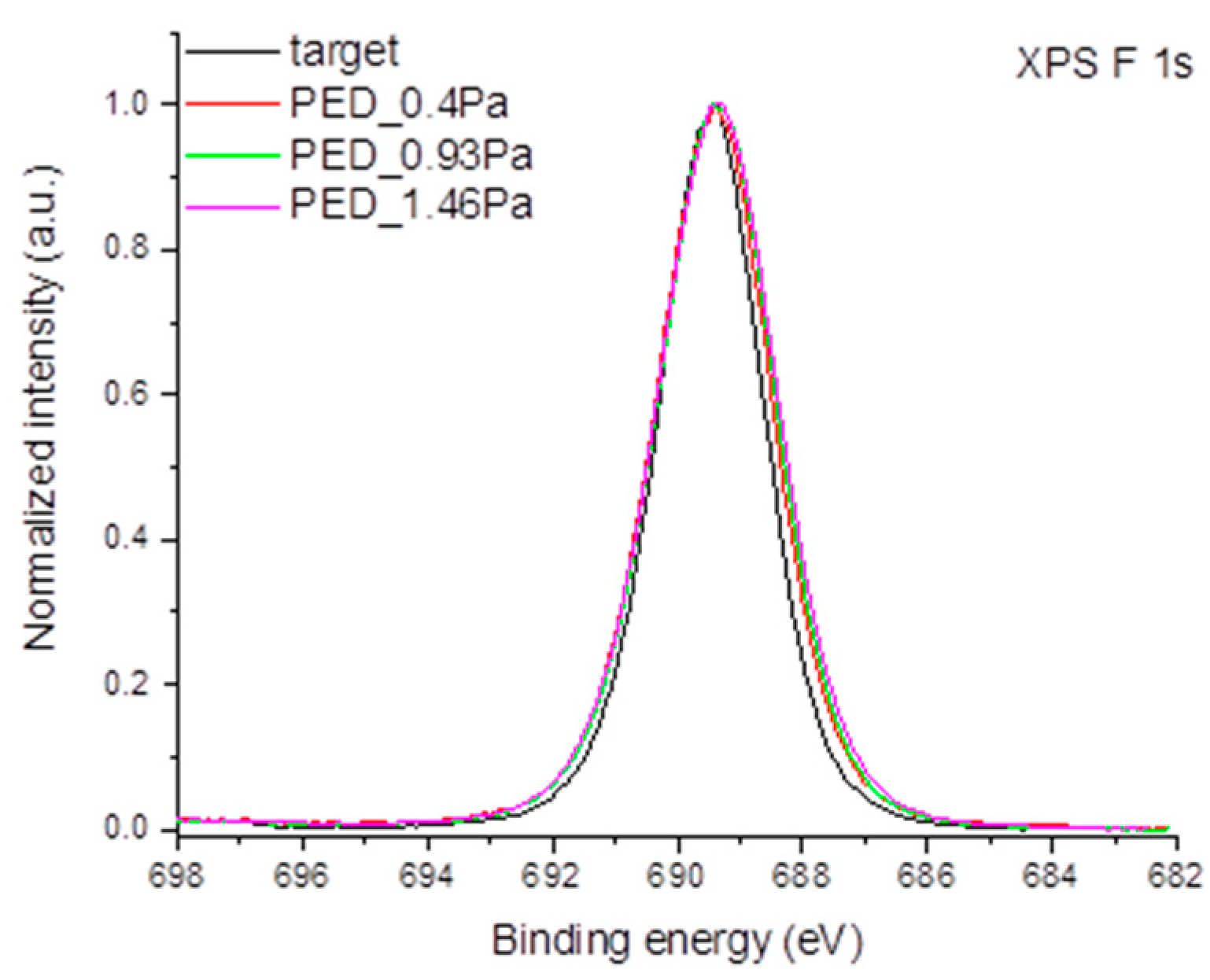
3.2. Chemical Structure Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, S.T.; Arenholz, E.; Heitz, J.; Bäuerle, D. Pulsed-laser deposition of crystalline Teflon (PTFE) films. Appl. Surf. Sci. 1998, 125, 17–22. [Google Scholar] [CrossRef]
- Henda, R.; Wilson, G.; Gray-Munro, J.; Alshekhli, O.; McDonald, A.M. Preparation of polytetrafluoroethylene by pulsed electron ablation: Deposition and wettability aspects. Thin Solid Films 2012, 520, 1885–1889. [Google Scholar] [CrossRef]
- Khan, M.S.; Franke, R.; Lehmann, D.; Heinrich, G. Physical and tribological properties of PTFE micropowder-filled EPDM rubber. Tribol. Int. 2009, 42, 890–896. [Google Scholar] [CrossRef]
- Schwödiauer, R.; Bauer-Gogonea, S.; Bauer, S.; Heitz, J.; Arenholz, E.; Bäuerle, D. Charge stability of pulsed-laser deposited polytetrafluoroethylene film electrets. Appl. Phys. Lett. 1998, 73, 2941–2943. [Google Scholar] [CrossRef]
- Hopp, B.; Smausz, T.; Kresz, N.; Nagy, P.M.; Juhász, A.; Ignácz, F.; Márton, Z. Production of biologically inert Teflon thin layers on the surface of allergenic metal objects by pulsed laser deposition technology. Appl. Phys. A Mater. Sci. Process. 2003, 76, 731–735. [Google Scholar] [CrossRef]
- Bodas, D.S.; Mandale, A.B.; Gangal, S.A. Deposition of PTFE thin films by RF plasma sputtering on 〈1 0 0〉 silicon substrates. Appl. Surf. Sci. 2005, 245, 202–207. [Google Scholar] [CrossRef]
- Biederman, H. RF sputtering of polymers and its potential application. Vacuum 2000, 59, 594–599. [Google Scholar] [CrossRef]
- Stelmashuk, V.; Biederman, H.; Slavínská, D.; Zemek, J.; Trchová, M. Plasma polymer films rf sputtered from PTFE under various argon pressures. Vacuum 2005, 77, 131–137. [Google Scholar] [CrossRef]
- Oya, T.; Kusano, E. Characterization of organic polymer thin films deposited by rf magnetron sputtering. Vacuum 2008, 83, 564–568. [Google Scholar] [CrossRef]
- Drábik, M.; Polonskyi, O.; Kylián, O.; Čechvala, J.; Artemenko, A.; Gordeev, I.; Choukourov, A.; Slavínská, D.; Matolínová, I.; Biederman, H. Super-hydrophobic coatings prepared by RF magnetron sputtering of PTFE. Plasma Process. Polym. 2010, 7, 544–551. [Google Scholar] [CrossRef]
- Blanchet, G.B.; Ismat Shah, S. Deposition of polytetrafluoroethylene films by laser ablation. Appl. Phys. Lett. 1993, 62, 1026–1028. [Google Scholar] [CrossRef]
- Daoud, W.A.; Xin, J.H.; Zhang, Y.H.; Mak, C.L. Pulsed laser deposition of superhydrophobic thin Teflon films on cellulosic fibers. Thin Solid Films 2006, 515, 835–837. [Google Scholar] [CrossRef]
- Heitz, J.; Dickinson, J.T. Characterization of particulates accompanying laser ablation of pressed polytetrafluorethylene (PTFE) targets. Appl. Phys. A Mater. Sci. Process. 1999, 68, 515–524. [Google Scholar] [CrossRef]
- Smausz, T.; Hopp, B.; Kresz, N. Pulsed laser deposition of compact high adhesion polytetrafluoroethylene thin films. J. Phys. D. Appl. Phys. 2002, 35, 1859–1863. [Google Scholar] [CrossRef]
- Kresz, N.; Kokavecz, J.; Smausz, T.; Hopp, B.; Csete, M.; Hild, S.; Marti, O. Investigation of pulsed laser deposited crystalline PTFE thin layer with pulsed force mode AFM. Thin Solid Films 2004, 453–454, 239–244. [Google Scholar] [CrossRef]
- Jȩdrzejewski, R.; Piwowarczyk, J.; Kwiatkowski, K.; Baranowska, J. Polytetrafluoroethylene thin films obtained by the pulsed electron beam deposition method at different gas pressures. Polimery/Polymers 2017, 62, 743–749. [Google Scholar] [CrossRef]
- Chandra, V.; Manoharan, S.S. Pulsed electron beam deposition of highly oriented thin films of polytetrafluoroethylene. Appl. Surf. Sci. 2008, 254, 4063–4066. [Google Scholar] [CrossRef]
- Norton, M.G.; Jiang, W.; Dickinson, J.T.; Hipps, K.W. Pulsed laser ablation and deposition of fluorocarbon polymers. Appl. Surf. Sci. 1996, 96–98, 617–620. [Google Scholar] [CrossRef]
- Kwong, H.Y.; Wong, M.H.; Wong, Y.W.; Wong, K.H. Superhydrophobicity of polytetrafluoroethylene thin film fabricated by pulsed laser deposition. Appl. Surf. Sci. 2007, 253, 8841–8845. [Google Scholar] [CrossRef]
- Niemczyk, A.; Moszyński, D.; Jędrzejewski, R.; Kwiatkowski, K.; Piwowarczyk, J.; Baranowska, J. Chemical Structure of EVA Films Obtained by Pulsed Electron Beam and Pulse Laser Ablation. Polymers 2019, 11, 1419. [Google Scholar] [CrossRef] [PubMed]
- Fazullin, D.D.; Mavrin, G.V.; Sokolov, M.P.; Shaykhov, I.G. Infrared Spectroscopic Studies of the PTFE and Nylon Membranes Modified Polyaniline. Mod. Appl. Sci. 2014, 9, 242. [Google Scholar] [CrossRef]
- Rogachev, A.A.; Tamulevičius, S.; Rogachev, A.V.; Yarmolenko, M.A.; Prosycevas, I. The structure and molecular orientation of polytetrafluoroethylene coatings deposited from active gas phase. Appl. Surf. Sci. 2009, 255, 6851–6856. [Google Scholar] [CrossRef]
- Pugmire, D.L.; Wetteland, C.J.; Duncan, W.S.; Lakis, R.E.; Schwartz, D.S. Cross-linking of polytetrafluoroethylene during room-temperature irradiation. Polym. Degrad. Stab. 2009, 94, 1533–1541. [Google Scholar] [CrossRef]
- Ignat’eva, L.N.; Buznik, V.M. IR-spectroscopic examination of polytetrafluoroethylene and its modified forms. Russ. J. Gen. Chem. 2009, 79, 677–685. [Google Scholar] [CrossRef]
- Schulze, M.; Bolwin, K.; Gülzow, E.; Schnurnberger, W. XPS analysis of PTFE decomposition due to ionizing radiation. Fresenius J. Anal. Chem. 1995, 353, 778–784. [Google Scholar] [CrossRef]
- Jędrzejewski, R.; Piwowarczyk, J.; Jędrzejewska, A.; Kwiatkowski, K.; Baranowska, J. Poly(propylene)/carbon composite thin films obtained by pulsed electron-beam deposition. Plasma Process. Polym. 2018, 15, 1700239. [Google Scholar] [CrossRef]
- Mathis, J.E.; Christen, H.M. Factors that influence particle formation during pulsed electron deposition of YBCO precursors. Phys. C Supercond. Appl. 2007, 459, 47–51. [Google Scholar] [CrossRef]
- Chandra, V.; Manoharan, S.S. Polytetrafluoroethylene (Teflon) films deposited by pulsed electron deposition technique for use in spintronics devices. Synth. React. Inorg. Met. Nano Metal Chem. 2008, 38, 288–291. [Google Scholar]
- Wang, S.; Li, J.; Suo, J.; Luo, T. Surface modification of porous poly(tetrafluoraethylene) film by a simple chemical oxidation treatment. Appl. Surf. Sci. 2010, 256, 2293–2298. [Google Scholar] [CrossRef]
- Liang, C.Y.; Krimm, S. Infrared Spectra of High Polymers. III. Polytetrafluoroethylene and Polychlorotrifluoroethylene. J. Chem. Phys. 1956, 25, 563–571. [Google Scholar] [CrossRef]
- Yamada, Y.; Kurobe, T.; Yagawa, K.; Ikeda, K. X-ray photoelectron spectroscopy of sputter-deposited fluorocarbon fims. J. Mater. Sci. Lett. 1999, 18, 415–417. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Grubova, I.Y.; Chernozem, R.V.; Krause, B.; Baumbach, T.; Loza, K.; Epple, M. RF magnetron sputtering of a hydroxyapatite target: A comparison study on polytetrafluorethylene and titanium substrates. Appl. Surf. Sci. 2017, 414, 335–344. [Google Scholar] [CrossRef]
- Lei, Y.G.; Ng, K.M.; Weng, L.T.; Chan, C.M.; Li, L. XPS C 1s binding energies for fluorocarbon-hydrocarbon microblock copolymers. Surf. Interface Anal. 2003, 35, 852–855. [Google Scholar] [CrossRef]
- Golub, M.A.; Lopata, E.S.; Finney, L.S. X-ray Photoelectron Spectroscopy Study of the Effect of Hydrocarbon Contamination on Poly(tetrafluoroethylene) Exposed to a Nitrogen Plasma. Langmuir 1993, 9, 2240–2242. [Google Scholar] [CrossRef]
- Nansé, G.; Papirer, E.; Fioux, P.; Moguet, F.; Tressaud, A. Fluorination of carbon blacks: An X-ray photoelectron spectroscopy study: I. A literature review of XPS studies of fluorinated carbons. XPS investigation of some reference compounds. Carbon N.Y. 1997, 35, 175–194. [Google Scholar] [CrossRef]
- Lacoste, J.; Carlsson, D.J. Gamma-, photo-, and thermally-initiated oxidation of linear low density polyethylene: A quantitative comparison of oxidation products. J. Polym. Sci. Part A Polym. Chem. 1992, 30, 493–500. [Google Scholar] [CrossRef]
- Lippert, T. Laser Application of Polymers. Adv. Polym. Sci. 2004, 168, 51–246. [Google Scholar]
- Vinogradov, I.P.; Lunk, A. Structure and chemical composition of polymer films deposited in a dielectric barrier discharge (DBD) in Ar/fluorocarbon mixtures. Surf. Coat. Technol. 2005, 200, 660–663. [Google Scholar] [CrossRef]
- Gritsenko, K.P.; Krasovsky, A.M. Thin-film deposition of polymers by vacuum degradation. Chem. Rev. 2003, 103, 3607–3649. [Google Scholar] [CrossRef]
- Rogachev, A.A.; Rogachev, A.V.; Yarmolenko, M.A.; Egorov, A.I. Morphology of polytetrafluoroethylene coatings formed from the active gas phase and its thermal modification. Russ. J. Appl. Chem. 2006, 79, 1207–1209. [Google Scholar] [CrossRef]
- Rogachev, A.A. Morphological Features of the First Growth Steps of Polymeric Coatings from the Active Gas Phase on the Activated Surface. Russ. J. Appl. Chem. 2004, 77, 281–284. [Google Scholar] [CrossRef]



| Sample | Carbon | Fluorine | Oxygen | Nitrogen | Silicon | F/C Ratio |
|---|---|---|---|---|---|---|
| Atomic percent | ||||||
| target | 29.7 | 70.3 | - | - | - | 2.4 |
| PLD 0.13Pa | 33.6 | 59.2 | 3.0 | 0.3 | 3.9 | 1.8 |
| PLD 0.93Pa | 33.0 | 63.6 | 2.3 | 0.5 | 0.6 | 1.9 |
| PLD 1.2Pa | 33.1 | 64.5 | 1.9 | 0.5 | - | 1.9 |
| PED 0.4Pa | 29.8 | 70.2 | - | - | - | 2.4 |
| PED 0.93Pa | 30.3 | 69.7 | - | - | - | 2.3 |
| PED 1.46Pa | 30.6 | 69.4 | - | - | - | 2.3 |
| Component Number on Fitted C 1s Spectrum | Position of Maximum of Component (eV) | Carbon Group or Structural Unit Corresponding to the Component (Carbon: C) |
|---|---|---|
| C1 | 285.2 ± 0.2 | Non-functionalized aliphatic carbons |
| C2 | 287.2 ± 0.2 | –CH2–CH2–CF2–, –CFR–CR2–CFR– |
| C3 | 288.3 ± 0.2 | –CH2–CFR–CH2–, –CH2–CH2–CF3, C–(CFR)4 |
| C4 | 290.0 ± 0.2 | –CFR–CFR–CFR– |
| C5 | 292.1 ± 0.2 | –(CF2–CF2)n– |
| C6 | 294.2 ± 0.2 | –CFR–CF3–CR2–CF3 |
| Sample | C1 | C2 | C3 | C4 | C5 | C6 |
|---|---|---|---|---|---|---|
| target | 10 | - | - | - | 90 | - |
| PLD 0.13Pa | 20 | 15 | 9 | 19 | 28 | 9 |
| PLD 0.93Pa | 17 | 16 | 12 | 18 | 28 | 9 |
| PLD 1.2Pa | 14 | 16 | 12 | 20 | 27 | 11 |
| PED 0.4Pa | - | 2 | - | 5 | 86 | 7 |
| PED 0.93Pa | - | 2 | 3 | 5 | 82 | 8 |
| PED 1.46Pa | - | 2 | 1 | 5 | 84 | 8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piwowarczyk, J.; Jędrzejewski, R.; Moszyński, D.; Kwiatkowski, K.; Niemczyk, A.; Baranowska, J. XPS and FTIR Studies of Polytetrafluoroethylene Thin Films Obtained by Physical Methods. Polymers 2019, 11, 1629. https://doi.org/10.3390/polym11101629
Piwowarczyk J, Jędrzejewski R, Moszyński D, Kwiatkowski K, Niemczyk A, Baranowska J. XPS and FTIR Studies of Polytetrafluoroethylene Thin Films Obtained by Physical Methods. Polymers. 2019; 11(10):1629. https://doi.org/10.3390/polym11101629
Chicago/Turabian StylePiwowarczyk, Joanna, Roman Jędrzejewski, Dariusz Moszyński, Konrad Kwiatkowski, Agata Niemczyk, and Jolanta Baranowska. 2019. "XPS and FTIR Studies of Polytetrafluoroethylene Thin Films Obtained by Physical Methods" Polymers 11, no. 10: 1629. https://doi.org/10.3390/polym11101629
APA StylePiwowarczyk, J., Jędrzejewski, R., Moszyński, D., Kwiatkowski, K., Niemczyk, A., & Baranowska, J. (2019). XPS and FTIR Studies of Polytetrafluoroethylene Thin Films Obtained by Physical Methods. Polymers, 11(10), 1629. https://doi.org/10.3390/polym11101629







